Abstract
We previously established that the inhibitory receptor LILRB4 mitigates LPS-induced, neutrophil-dependent pathologic effector mechanisms in inflammation. We now report that LILRB4 on dendritic cells (DCs) counterregulates development of an adaptive Th2 immune response and ensuing inflammation in a model of allergic pulmonary inflammation initiated by inhalation sensitization with OVA and LPS followed by airway challenge with OVA. We found that Lilrb4−/− mice had significantly exacerbated eosinophilic pulmonary inflammation as assessed in bronchoalveolar lavage and lung tissue, as well as elevated levels of OVA-specific IgE and Th2 cytokines produced by OVA-restimulated lymph node cells. LILRB4 was preferentially expressed on MHC class IIhighCD86high OVA-bearing DCs in lung-draining lymph nodes after sensitization or challenge. Moreover, the lymph nodes of Lilrb4−/− mice had significantly more of these mature DCs after challenge with OVA, which was accompanied by significantly more IL-4-producing lymphocytes, compared with Lilrb4+/+ mice. Sensitization of naïve Lilrb4+/+ mice by transfer of OVA-LPS-pulsed Lilrb4−/− bone marrow-derived DCs (BMDCs) was sufficient to confer exacerbated allergic lung pathology upon challenge with OVA, compared with mice that received Lilrb4+/+ BMDCs. Our findings establish that maturation and migration of pulmonary DCs to lymph nodes in response to Ag and an innate immune stimulus is associated with upregulated expression of LILRB4. In addition, this receptor attenuates the number of these mature DCs and attendant IL-4-producing lymphocytes in the lymph nodes, and accordingly, the ability of DCs to elicit pathologic Th2 pulmonary inflammation.
Keywords: Dendritic Cells, Inflammation, Allergy, Lung
Introduction
The ITIM-bearing inhibitory receptor LILRB4 suppresses the activation of mast cells induced by passive administration of IgE and Ag or stem cell factor leading to a reduction in the ensuing anaphylactic reactions in mouse models (1,2). In addition, LILRB4 diminishes neutrophil-dependent pathologic inflammation induced by LPS in the thrombohemorrhagic Shwartzman reaction and an effector phase model of immune complex arthritis induced by the combination of anti-type II collagen mAbs and LPS (3-5). LPS traditionally has been associated with innate immune responses to bacterial infections initiated by TLR4 signaling that can lead to Th1 polarized adaptive immune responses driven by the production of IL-12 (6). The ability of Th1 polarization to prevent or suppress Th2 responses (7) led to the hypothesis that exposure to microbial components that foster Th1 polarization would reduce the incidence of allergic pulmonary disorders. Indeed, exposure of children to environments high in LPS early in life has been associated with a reduced risk for subsequent development of bronchial asthma (8-10). Conversely, environmental LPS levels have been directly correlated with the severity of asthma in older subjects (11,12). In a number of animal models, exposure to LPS near the time of sensitization to experimental Ags introduced intraperitoneally with adjuvants reduces pulmonary inflammation and/or airway hyperresponsiveness (AHR) induced by Ag challenge in the airway (13-15). In contrast, inhalation of LPS before or during sensitization with inhaled OVA can result in augmented Ag-specific serum IgE levels and lung eosinophilia upon airway challenge with OVA (16,17). The ability to detect a contribution for LPS in the development of the Th2 adaptive immune response and pulmonary inflammation may reflect the fact that sensitization with OVA alone via the airway is tolerogenic (18-21), and an innate immune “danger” signal such as LPS is necessary to convert the tolerogenic response to a pathologic immune and inflammatory response.
Nevertheless, the contribution of LPS to models in which both sensitization and challenge occur through the airway is not restricted to inducing Th2 inflammation. An elegant study, in which the major variable was the dose of LPS administered during airway sensitization to OVA, established that the amount of LPS materially alters the nature of the pulmonary inflammation that ensues upon challenge with OVA. A relatively low dose of LPS (300 ng delivered in three intranasal instillations with a total of 300 μg of OVA) leads to iconic Th2 pulmonary inflammation upon OVA challenge characterized by elevated serum levels of Ag-specific IgE and IgG1, the absence of serum IL-12, eosinophilia and goblet cell metaplasia in the lung, and preferential production of Th2 cytokines IL-5 and IL-13 by Ag-restimulated lymph node cells in a TL4- and MyD88-dependent manner (17,22). In contrast, the administration of 1000-fold more LPS during sensitization with the same dose of OVA leads to generation of OVA-specific serum IgG2a, serum IL-12, neutrophilic pulmonary inflammation, and release of IFN-γ from Ag-restimulated lymph node cells, all of which are characteristic of Th1 responses (17). LPS in the low-dose protocol made an essential contribution to the migration of pulmonary DCs to the draining intrathoracic lymph nodes (17,23) and likely to MyD88-dependent upregulation of MHC class II and CD86 on pulmonary DCs during sensitization, the latter favoring Th2 polarization in the absence of IL-12 (22,23). Indeed, LPS also contributes importantly to the recruitment of Ag-specific Th2 cells to the lung in a similar model (24).
A better understanding of the mechanisms that regulate the ability of LPS to influence the development of allergic pulmonary inflammation would help explain the complex contributions of this innate and ubiquitous molecule to asthma. Given that LILRB4 counterregulates LPS-mediated inflammation in several neutrophil-dependent acute effector phase models (3-5), we considered the possibility that LILRB4 might also counterregulate the development of the LPS-dependent, adaptive Th2 response to inhaled OVA through expression on DCs (25). We report here that expression of LILRB4 was strikingly upregulated on DCs bearing OVA and high levels of MHC class II and CD86 in the lung-draining lymph nodes of mice after inhalation of OVA and low-dose LPS. In addition, after challenge with OVA, Lilrb4−/− mice have significantly exacerbated eosinophilic lung inflammation in the OVA/low-dose LPS model, as well as elevated levels of Ag-specific serum IgE and Th2 cytokines produced by OVA-restimulated intrathoracic lymph node cells. The greater pathologic Th2 response in Lilrb4−/− mice was preceded by the appearance of a significantly greater number of mature, OVA-bearing DCs in the lung-draining lymph nodes as well as IL-4-producing lymphocytes likely accumulated there in response to the OVA-bearing DCs. Moreover, sensitization of Lilrb4+/+ mice by transfer of Lilrb4−/− OVA/LPS-pulsed BMDCs to Lilrb4+/+ mice was sufficient to reiterate upon Ag-challenge the exacerbated allergic lung pathology observed in Lilrb4−/− mice sensitized by inhalation of OVA-LPS. Hence, LILRB4 on DCs can temper the course of a pathologic adaptive immune response in vivo and ensuing pulmonary Th2 inflammation.
Materials and Methods
Animals
Lilrb4+/+ and Lilrb4−/− mice were generated on the BALB/c background as previously described (4). Mice were maintained in a specific pathogen-free barrier facility. Female mice, 6–12 weeks old, were used for experiments. The use of mice for these studies was reviewed and approved by the Animal Care and Use Committee of the Dana-Farber Cancer Institute.
LPS depletion and measurement
For some experiments, Endotoxin Detoxi-gel (Pierce Biotechnology) was used according to the manufacturer’s instructions to remove >99% of the contaminating LPS in the administered OVA (Grade V; Sigma-Aldrich) solution. For other experiments, OVA was administered without prior detoxification. However, as determined with the limulus amebocyte assay (Cambrex), in all cases <2 ng of contaminating LPS per mouse was delivered by the OVA solutions during sensitization and challenge.
Sensitization and challenge protocol
Mice were lightly anesthetized with isoflurane and then sensitized intranasally with 50 μl of PBS alone or containing 100 μg of LPS-depleted OVA and 100 ng of LPS (from E. coli 055:B5; Sigma-Aldrich #L4524) on days 0, 1, and 2 (17). On days 14, 15, 18, and 19, mice were lightly anesthetized with isoflurane and challenged intranasally with 25 μg of OVA in 50 μl of PBS. In some experiments, the challenges were delivered in 30 μl with comparable results. Mice were sacrificed for analyses on day 21 by i.p. injection of an overdose of pentobarbital.
Analysis of BAL
A 22-g plastic cannula was inserted into the upper third of the trachea and secured with cotton thread. BAL fluid was obtained by infusion and withdrawal of 0.7 ml of calcium- and magnesium-free Hanks’ Balanced Salt Solution (HBSS) containing 0.1 mM EDTA or Dulbecco’s PBS containing 0.5 mM EDTA; this was repeated three times to achieve a total instillation of 2.1 ml. Cells were pelleted and washed by centrifugation, counted with a hemacytometer, and spun onto slides with a Shandon Cytospin (Thermo Scientific). The slides were stained with Dif-Quik (Dade Behring), and differential cell counts were determined on the basis of cell morphology and staining characteristics by counting at least 200 cells per sample.
Lung mononuclear cell isolation
Lungs were perfused with 10 ml of ice-cold calcium- and magnesium-free HBSS through the right ventricle, removed intact, and chopped into 1-mm fragments with 18-mm scalpel blades. Lung fragments were incubated in 500 U/ml collagenase (CLS-IV, Worthington Biochemical) and 0.02 mg/ml DNAse I (Sigma-Aldrich) at 37°C for 60 min with agitation at 200 rpm. The resulting suspensions were strained through metal sieves to remove debris, and mononuclear cells (>97% pure) were isolated from the interface of a Nycoprep (Axis-Shield) gradient (300 x g for 20 min at 4°C). Cells were washed with PBS, counted, and subjected to flow cytometric analysis.
Flow cytometry
Cells were resuspended in PBS containing 0.5% FBS or BSA and 0.05% sodium azide at 4°C and incubated with anti-FcγRII/III mAb (eBioscience) and mouse IgG (Sigma) for 20 min on ice to block FcγR. Cells were then incubated with saturating concentrations of fluorochrome conjugated anti-CD4, CD8, CD11c, CD19, CD86, or MHC class II mAbs (eBioscience), or mAb B23.1 prepared as previously described (5), for 30 min at 4°C, washed by centrifugation, and analyzed on a Becton Dickinson FACSCanto flow cytometer, using FACSDiva acquisition software and FlowJo analysis software (Treestar).
Isolation of intrathoracic lymph node cells for Ag restimulation and flow cytometry
For the Ag restimulation assay, cells were isolated from the parathymic, hilar, and paratracheal lymph nodes by mechanical dispersion through mesh and were cultured at 2 ×106 cells/ml with 0.1 mg/ml of LPS-depleted OVA for 72 h. The concentrations of cytokines in culture supernatants were measured with ELISA kits (eBioscience). For flow cytometry of DCs, the residual tissue fragments on the mesh were added to the filtrate that was then digested with collagenase and DNAse I at 37°C for 20 min with agitation at 200 rpm. The reaction was stopped with an equal volume of PBS containing 2 mM EDTA, and cells were then washed by centrifugation in PBS containing 1 mM EDTA.
Measurements of anti-OVA Ig in sera
For quantification of anti-OVA IgE levels, anti-mouse IgE (2 μg/ml; clone R35–72) (BD Biosciences) was incubated in 96-well flat bottom Maxisorp plates (Thermo Fisher) overnight at 4°C. The plates were washed with PBS containing 0.05% Tween-20 and blocked with PBS containing 10% heat-inactivated FBS (Sigma-Aldrich). Serially-diluted sera samples were added, and the plates were incubated for 1 h at 37°C. Biotinylated OVA (2 μg/ml) was added to each well, and the plates were incubated for 1h at 37°C. Plates were washed several times and incubated with HRP-conjugated streptavidin for 1 h at 37°C. The plates were washed several times, substrate solution (tetramethylbenzidine and hydrogen peroxide) was added, and the plates were incubated for 30 min at room temperature. The reactions were terminated by the addition of 1 M H2SO4, and the absorbances at 570 nm were subtracted from the absorbances at 450 nm. OVA-specific IgE concentrations in experimental samples were calculated by comparison with those of a serum containing high-titer anti-OVA IgE generated by repeated i.p injection and then intranasal administration of OVA in BALB/c mice. The concentration of anti-OVA IgE in that serum was initially determined by comparison with a monoclonal anti-OVA IgE (26) provided by Dr. Lester Kobzik (Harvard School of Public Health). Anti-OVA IgE levels in the BMDC transfer experiments were determined with an ELISA kit from MD Biosciences.
For quantification of OVA-specific IgG1, plates were coated with OVA (10 μg/ml), the serially-diluted sera were added, the plates was incubated with biotinylated anti-mouse IgG1 (clone A85-1) (BD Biosciences), and HRP-conjugated streptavidin. Anti-OVA mouse IgG1 mAb (clone OVA-14; Sigma-Aldrich) was used as the standard.
Analysis of Ag-bearing DCs in intrathoracic lymph nodes
In one set of experiments, mice received 100 μg of Alexa Fluor 647-labeled OVA (AF-OVA) (Invitrogen) and 100 ng of LPS intranasally on day 0; mice were analyzed on day 1. In other experiments, mice received 100 μg of unlabeled OVA and 100 ng of LPS intranasally on days 0, 1, and 2; some were analyzed on day 14, whereas others were challenged with 25 μg of AF-OVA on day 14 and analyzed on day 15. In each type of experiment, some mice received unlabeled OVA rather than AF-OVA to serve as negative controls for defining AF-OVA positivity in flow cytometry. On the days of analysis, mice were euthanized, and their pulmonary and systemic circulation was perfused with 10 ml of HBSS. Four draining intrathoracic lymph nodes (two right parathymic, one paratracheal, and one right hilar) were harvested per animal, pooled, and disaggregated to single cell suspensions with the flat end of a tuberculin syringe on a 70 μm cell strainer (Costar). The cells were centrifuged, resuspended in 3 ml of pre-warmed RPMI containing 5% FBS, and digested with collagenase and DNAse as described above for lung, except that the incubation time was 20 minutes. The digestion was stopped by the addition of 3 ml of ice-cold PBS containing 2 mM EDTA, the cells were washed with 5 ml of PBS containing 1mM EDTA, and the cells were resuspended in 500 μl of cold PBS.
Quantification of IL-4-producing cells by ELISPOT assay
Mice were sensitized by intranasal instillation of 100 μg OVA and 100 ng LPS on days 0, 1, and 2. On day 14, mice were challenged by intranasal instillation of 25 μg of OVA, and on day 15, four intratracheal lymph node cells were isolated from each mouse as described above. Four serial 2-fold dilutions of cells, starting with 8 × 105 cells/ml, were cultured in 96-well sterile multiscreen tissue culture plates (Millipore) coated with anti-capture antibody (eBioscience), and cells were stimulated with 50 ng/ml of PMA and 1 μM ionomycin for 36 h. IL-4 spots were detected with an ELISPOT kit (eBioscience) according to the manufacturer’s instructions and were counted using a dissecting microscope. The frequency of IL-4 producing cells was calculated and multiplied by the total number of lymph node cells to determine the total number of IL-4-producing cells.
Generation and adoptive transfer of BMDCs
BMDCs from Lilrb4+/+ and Lilrb4−/− mice were generated according to the method of Lutz, et al. (27). Cells were flushed from the femurs of euthanized mice with 10 ml cold RPMI 1640 and filtered through a 70-μm cell strainer. The cells were then plated in extra deep 100 × 25 mm Petri dishes (Thermo Fisher) at 4 × 105 cells/ml in 10 ml RPMI 1640 supplemented with 10% FBS, 42.9 mM 2-mercaptoethanol, 10,000 U/ml Pen-Strep, 200 mM L-glutamine, MEM non-essential amino acids (R10 medium), and 20 ng/ml mouse recombinant GM-CSF (Peprotech). On day 3 of culture, 10 ml of fresh R10 medium with 20 ng/ml GM-CSF was added to each dish. On days 6 and 8, half the volume of each dish was removed, centrifuged, resuspended in 10 ml/dish of fresh R10 medium with 20 ng/ml GM-CSF, and added back to the original dish. On day 9, non-adherent cells were harvested, washed with PBS at 4°C, and re-plated in 35 × 10 mm Petri dishes (Thermo Fisher) at 1 × 106 cells/ml in 3 ml/dish R10 medium with 20 ng/ml GM-CSF, 100 μg/ml OVA, and 100 ng/ml LPS. After overnight incubation, non-adherent cells were harvested, washed by centrifugation three times with PBS, and resuspended in PBS at 1 × 105 cells/50 μl. Lilrb4+/+ BALB/c recipients were lightly anesthetized with isoflurane and received 1 × 105 Lilrb4+/+ or Lilrb4−/− Ag-loaded BMDCs in 50 μl PBS intranasally. On days 14, 15, 18 and 19, mice were challenged with intranasal instillations of 25 μg OVA in 30 μl PBS or given PBS alone. Mice were euthanized and analyzed 48 h after the last instillation.
Lung Histology
The left lungs of mice were fixed for at least 8 h in 4% paraformaldehyde, embedded in glycolmethacrylate, and 2.5 μm sections were cut, as described previously (28). Areas of inflammation and total areas in eight sections from each lung were quantified by digital image analysis to determine the percent inflamed area. Eosinophilia was revealed by staining sections with Congo red (28,29). Other sections were stained with Periodic acid-Schiff (PAS) for enumeration of goblet cells in the bronchial epithelium of at least four independent bronchovascular bundles from each mouse. Only comparable, large caliber, pre-terminal bronchi (diameter 200–220 μm) were examined, since minimal goblet cell metaplasia occurs in terminal bronchioles (30). The length of basal lamina of each bronchus was measured by Image J (NIH image analysis software), and the data are expressed as the number of goblet cells per mm of basal lamina.
Statistical analyses
Statistical significance was determined using Student’s unpaired, two-tailed t-test or the Mann-Whitney test. P values <0.05 were considered to be significant.
Results
Effects of LILRB4 deficiency on Th2 pulmonary inflammation in an LPS-dependent model
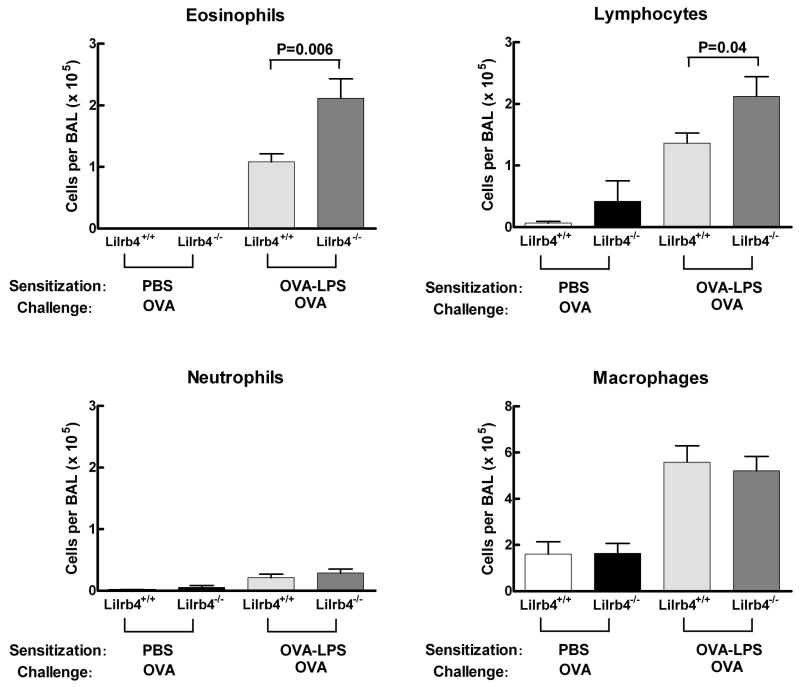
To detect contributions of LILRB4 to pulmonary inflammation, we chose a model of airway sensitization and challenge to OVA that depends on the presence of relatively low levels of LPS during the sensitization phase to elicit Th2 inflammation in the lung (17). On days 0, 1, and 2, Lilrb4+/+ and Lilrb4−/− mice received intranasal instillations of phosphate-buffered saline (PBS) alone or containing 100 μg of LPS-depleted OVA and 100 ng of highly purified LPS. On days 14, 15, 18, and 19, all mice received 25 μg of LPS-depleted OVA intranasally, and mice were euthanized for analysis on day 21 (17). The infiltration of inflammatory cells into the lungs was assessed by analysis of cells in BAL. The BAL from Lilrb4+/+ and Lilrb4−/− mice that were sensitized with OVA-LPS and challenged with OVA (OVA-LPS/OVA mice) developed an eosinophilia that did not occur in the BAL fluid from mice that received PBS during the sensitization phase (PBS/OVA mice) (Fig. 1). However, the number of eosinophils in the BAL of Lilrb4−/− OVA-LPS/OVA mice was significantly greater (P=0.006) than in Lilrb4+/+ mice. In addition, the number of BAL lymphocytes induced in OVA-LPS/OVA mice was also significantly greater (P=0.04) in Lilrb4−/− mice compared with Lilrb4+/+ mice, whereas there were no differences in the numbers of neutrophils or macrophages. These data indicate that the absence of LILRB4 leads to significantly exacerbated Th2 pulmonary inflammation in this model.
Fig 1. BAL cell numbers and differentials from Lilrb4+/+ and Lilrb4−/− mice.
Lilrb4+/+ and Lilrb4−/− and mice received PBS alone or with 100 μg of OVA and 100 ng of LPS intranasally on days 0, 1, and 2. Mice were challenged with 25 μg of LPS-depleted OVA intranasally on days 14, 15, 18, and 19. BAL was collected on day 21, cells in the BAL were counted, cytospin slides were prepared and stained with Diff-Quik, and leukocyte differential counts were determined for each mouse and multiplied by the number of recovered cells. Data are expressed as mean ± SEM, n=4, 5, 12, and 12 for the four columns from left-to-right, respectively.
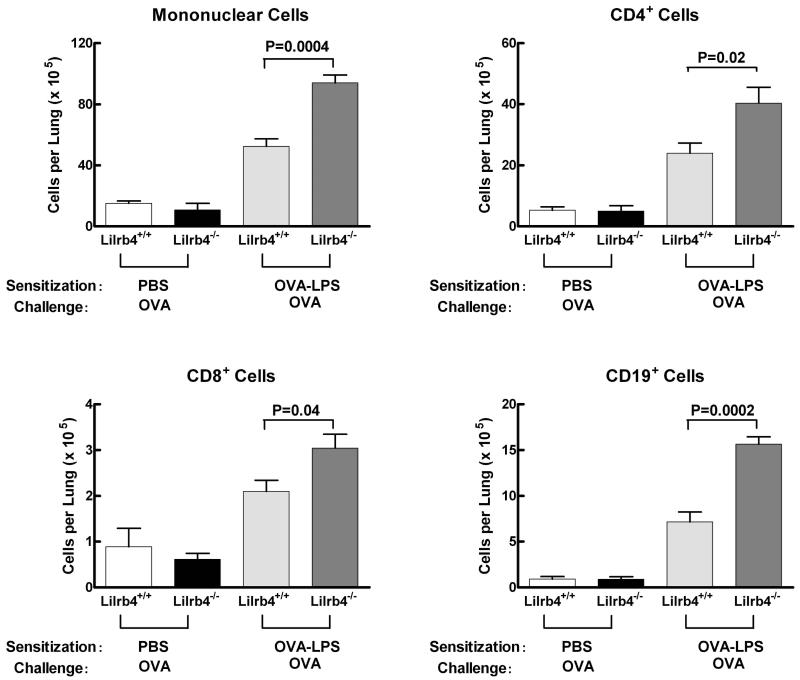
To quantify pulmonary inflammation in a second manner, the lungs of Lilrb4+/+ and Lilrb4−/− mice at day 21 were perfused to remove intravascular cells, minced, digested with collagenase and DNAse I, and parenchymal mononuclear cells were isolated by density gradient centrifugation. Greater numbers of mononuclear cells were recovered from both Lilrb4+/+ and Lilrb4−/− OVA-LPS/OVA mice compared with PBS/OVA mice (Fig. 2), demonstrating that the inflammation induced in this model was not limited to cells recovered with BAL (Fig. 1). Moreover, the number of lung mononuclear cells obtained from Lilrb4−/− mice was significantly greater than from Lilrb4+/+ animals (P=0.0004) (Fig. 2). The mononuclear cells were phenotyped by flow cytometry to define their composition. The lung mononuclear cells from Lilrb4−/− mice had significantly more CD4+ cells (P=0.02), CD8+ cells (P=0.04), and CD19+ (P=0.0002) cells (Fig. 2). Hence, the greater number of lung MNCs in Lilrb4−/− OVA-LPS/OVA mice at day 21 included both T and B lymphocytes. These changes primarily reflected the greater number of total lung MNCs in Lilrb4−/− mice rather than selective increases in particular populations of lymphocytes. Therefore, as revealed by significantly increased cellularity in both BAL and parenchymal lung mononuclear cells, the absence of LILRB4 leads to exacerbated pathologic Th2 pulmonary inflammation.
Fig. 2. Cell numbers and surface phenotypes of parenchymal lung mononuclear cells from Lilrb4+/+ and Lilrb4−/− mice.
Mice were sensitized and challenged as described in the legend for Fig. 1. Lungs were perfused, cut into fragments, and digested with collagenase and DNAse I. The mononuclear cells dispersed from each lung were isolated on Nycoprep gradients and counted. Fc receptors on the cells were blocked with mouse IgG and anti-FcγRIIB/III, the percentage of cells expressing CD4, CD8, or CD19 was determined by flow cytometry with directly fluorochrome-labeled mAbs, and the total number of cells expressing each receptor was calculated. Data are expressed as mean ± SEM, n=2, 2, 5, and 5 for the four columns from left-to-right, respectively.
Effects of LILRB4 deficiency on serum Ig levels and Ag-recall responses
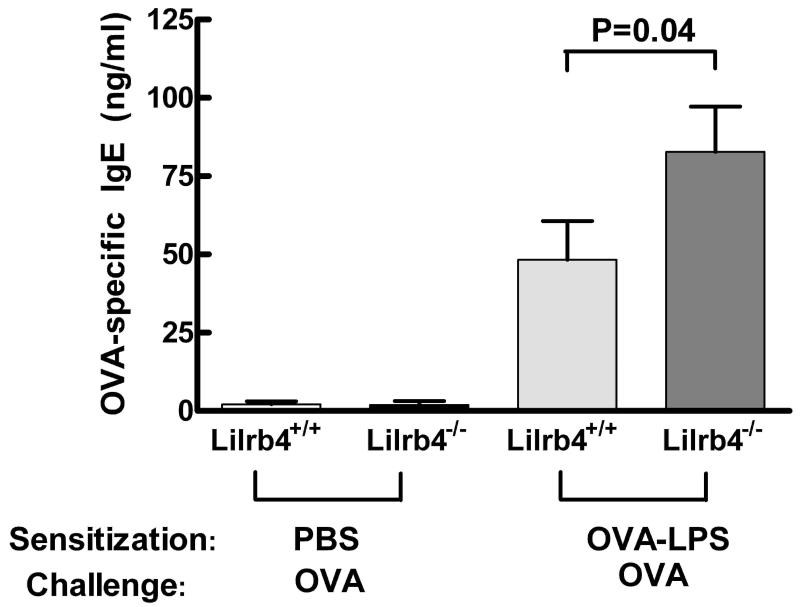
Our previous studies identified counterregulatory effects of LIRB4 on mast cells and neutrophils in passive, effector phase models of inflammation (1-5). The augmented pulmonary inflammation induced by active immunization in the OVA-LPS model suggested that LILRB4 might contribute to regulating the development of adaptive immune responses. We therefore determined whether the Ag-specific IgE response was elevated in the absence of LILRB4. Serum samples were isolated from mice at D21, and the levels of OVA-specific IgE were quantified with an ELISA. IgE was essentially undetectable in Lilrb4+/+ and Lilrb4−/− PBS/OVA mice, but the levels were elevated in OVA-LPS/OVA mice (Fig. 3). Moreover, sera from Lilrb4−/− mice contained a significantly greater amount of Ag-specific IgE compared with sera from Lilrb4+/+ animals (P=0.04). In addition, there was a trend towards higher levels of OVA-specific IgG1, a Th2-associated Ig subclass in mice (31) in Lilrb4−/− vs. Lilrb4+/+ OVA-LPS/OVA mice (2.4 ± 0.3 and 1.6 ± 0.3 μg/ml; n=16 and 17, respectively; P=0.08). These data indicate that the absence of LILRB4 led to an enhanced Th2 polarized humoral response.
Fig. 3. Serum IgE levels in Lilrb4+/+ and Lilrb4−/− mice.
Mice were sensitized and challenged as described in the legend for Fig. 1. Levels of anti-OVA IgE were measured by ELISA with an anti-mouse IgE mAb as the capture reagent and biotinylated OVA/horseradish peroxidase (HRP)-Streptavidin/TMB as the detecting reagents. Data are expressed as mean ± SEM, n=8, 10, 16, and 16 for the four columns from left-to-right, respectively.
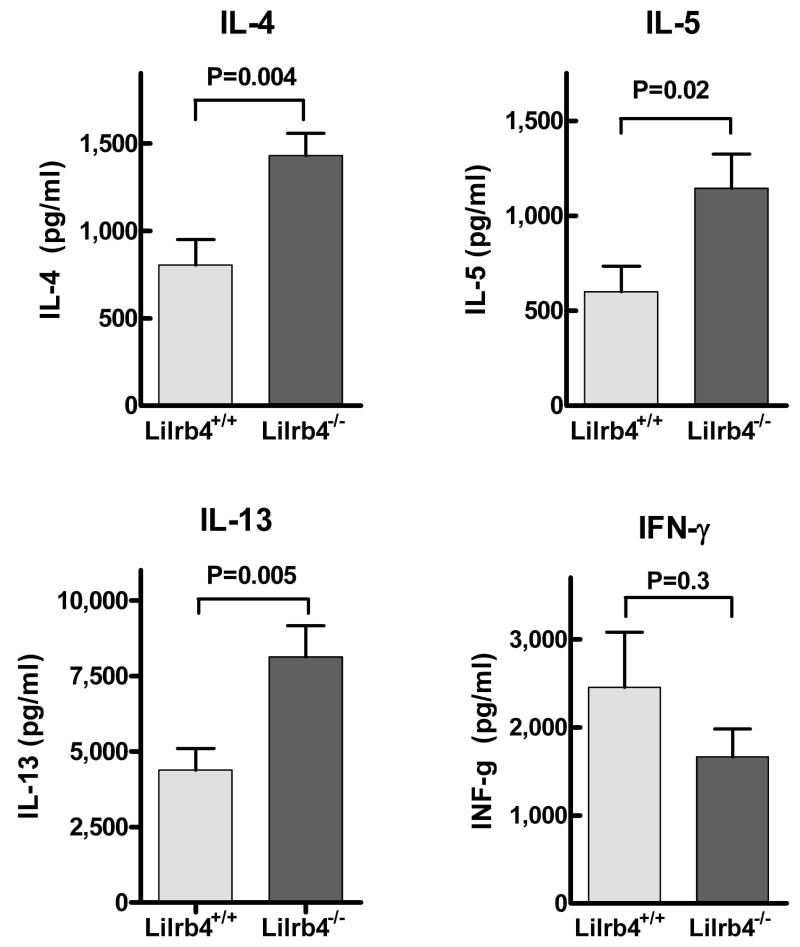
To examine a cellular adaptive immune response, we isolated intrathoracic lymph node cells from mice on D21, restimulated the lymph node cells with OVA, and measured cytokine production 72 h later with ELISAs. The secretion of IL-4, IL-5, and IL-13 were all significantly greater in Lilrb4−/− cultures compared with Lilrb4+/+ cultures (P=0.004, 0.02, and 0.005, respectively) (Fig. 4). In contrast, there was a trend towards less production of IFN-γ in the Lilrb4−/− cultures. These data indicate that there was an enhancement of the Th2-polarized T cell response in cells from Lilrb4−/− mice.
Fig. 4. Secretion of cytokines by Ag-restimulated lymph node cells from Lilrb4+/+ and Lilrb4−/− mice.
Mice were sensitized and challenged as described in the legend for Fig. 1. Intrathoracic lymph nodes were removed, cells were dispersed by mechanical disruption of the tissue, cells were cultured at 2 × 106cell /ml with 0.1 mg/ml of LPS-depleted OVA for 3 d, and mediators in the supernatants were measured with ELISAs. Data are expressed as mean ± SEM, n=13 and 11 (IL-4), 12 and 9 (IL-5), 13 and 11 (IL-13), and 9 and 8 (IFN-γ) for each Lilrb4+/+ and Lilrb4−/− pair, respectively.
Expression of LILRB4 on intrathoracic lymph node DCs
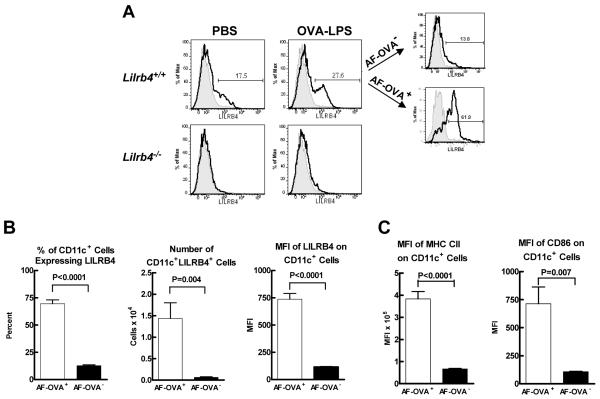
Although the preceding data indicated that LILRB4 can negatively regulate a pulmonary adaptive immune response, we did not detect expression of LILRB4 on lung or lymph node T cells in this model (data not shown). We then considered the possibility that LILRB4 is expressed on DCs in the pulmonary system and might influence lymphocytes in lung-draining lymph nodes. To address this possibility, we instilled PBS alone or containing 100 μg of AF-OVA and 100 ng of LPS intranasally into Lilrb4+/+ and Lilrb4−/− mice. Intrathoracic lymph node cells were isolated 18 h later as described above, and the binding of mAb B23.1 anti-LILRB4 (32) to CD11c+ DCs was assessed with flow cytometry. In mice that received PBS alone, 17 ± 0.15% of the CD11c+ cells expressed LILRB4, and the total number of CD11c+LILRB4+ cells was 0.5 × 104 ± 0 (n=2); LILRB4 was not detected on cells from Lilrb4−/− mice (representative histograms are shown in Fig. 5A). In contrast, mice that received AF-OVA and LPS had a greater percentage (26 ± 1.8%) (Fig. 5A) and total number (2.0 ± 0.3 × 104; n=8) of CD11c+ cells that expressed LILRB4+ cells than mice that received PBS alone. Notably, whereas 70 ± 3.6% of CD11c+AF-OVA+ cells expressed LILRB4, a significantly lower 12 ± 0.7% of CD11c+AF-OVA− cells expressed LILRB4 (representative histograms in Fig. 5A; compiled data in Fig. 5B). In addition, the number of LILRB4+CD11c+AF-OVA+ cells in the lymph nodes was a significant 22-fold greater than the number of LILRB4+CD11c+AF-OVA− cells, and the MFI value for LILRB4 on the OVA-bearing cells was a significant 6.4-fold greater than on the cells that did not have OVA (Fig. 5B). Furthermore, the MFI values for MHC class II and CD86 were a significant 5.7- and 6.6-fold greater, respectively, on CD11c+AF-OVA+ cells compared with CD11c+AF-OVA− cells (Fig. 5C). Because the vast majority of OVA-bearing DCs in the intrathoracic lymph nodes of mice after intranasal instillation of OVA and LPS are derived from the airway (23), our data indicate that OVA-bearing DCs that had migrated from the lung to the draining lymph nodes and matured expressed LILRB4.
Fig. 5. Expression of LILRB4 on intrathoracic lymph node DCs.
PBS alone or containing 100 μg of AF-OVA and 100 ng of LPS were instilled intranasally into Lilrb4+/+ and Lilrb4−/− mice, four intrathoracic lymph nodes was removed from each mouse 18 h later, the cells were dispersed enzymatically from the tissue and counted, FcγR on the cells were blocked as described in the legend for Fig. 2, and the binding of anti-LILRB4 (bold histograms) and isotype control (light, shaded histograms) to CD11c+ DCs was measured by flow cytometry. The cells in Lilrb4+/+ mice that had received AF-OVA and LPS were further gated into AF-OVA+ and AF-OVA− cells, and the percent, number, and MFI of LILRB4+ cells was determined for the two cell populations. Representative flow cytometry histograms (A) and compiled data (B) are shown. The expression of MHC class II and CD86 on the AF-OVA+ and AF-OVA− populations was determined in parallel (C). Data are expressed as mean ± SEM, n=6 (B) and n=4 (C).
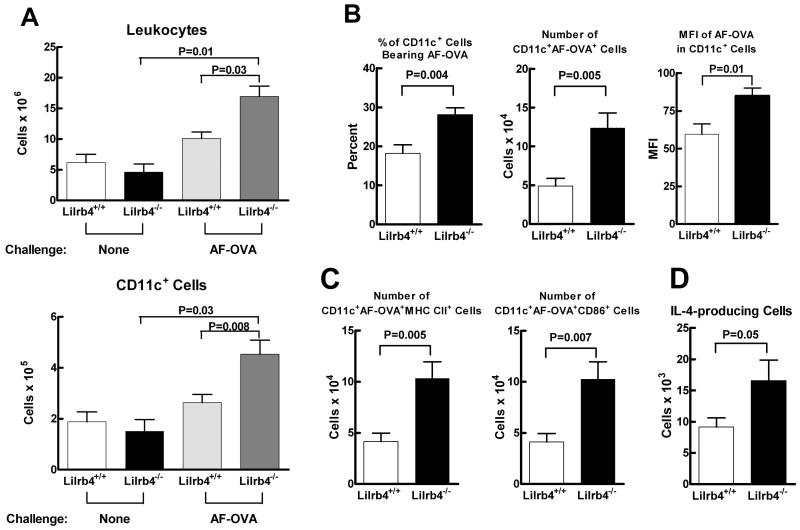
To determine whether the absence of LILRB4 affected lymph nodes DCs 18 h after instillation of AF-OVA and LPS, we compared these cells in Lilrb4+/+ and Lilrb4−/− mice. There were no significant differences in the percentages or numbers of CD11c+ cells, CD11c+AF-OVA+ cells, or in the expression levels of AF-OVA, MHC class II, and CD86 on CD11c+ cells in the two strains of mice (n=4–13). Nevertheless, the preferential expression of LILRB4 on OVA-bearing DCs that express high levels of MHC class II and CD86 in the draining lymph nodes of Lilrb4+/+ mice raised the possibility that LILRB4 would influence the response of pulmonary DCs in sensitized mice to challenge with OVA. We therefore sensitized Lilrb4+/+ and Lilrb4−/− mice by daily intranasal instillation of 100 μg of OVA-LPS on days 0 through 2, and the intrathoracic lymph nodes cells of one group of mice were analyzed on day 14. Another group of sensitized mice was challenged with 25 μg of AF-OVA on day 14 and their lymph node cells were analyzed on day 15. Before challenge, there were no differences in either the numbers of leukocytes or CD11c+ cells in the lymph nodes of Lilrb4+/+ and Lilrb4−/− mice (Fig. 6A). After the single challenge in Lilrb4+/+ mice, there was no significant increase in either of these parameters, whereas there were significant 4- and 3-fold increases in the numbers of leukocytes and CD11c+ cells, respectively, in the lymph nodes of Lilrb4−/− mice, and these numbers were significantly greater than those of challenged Lilrb4+/+ mice (Fig. 6A). Moreover, there was a significantly greater percentage and number of CD11c+ cells bearing AF-OVA in Lilrb4−/− mice, as well as a significantly greater MFI of AF-OVA in the CD11c+ cells (Fig. 6B), indicative of greater Ag content. The AF-OVA+ cells from Lilrb4+/+ mice, when compared with AF-OVA− cells, expressed significantly greater levels of both MHC class II (MFI of 4,095 ± 354 versus 1,466 ± 19, respectively; P<0.0001, n=7) and CD86 (MFI of 1,051 ± 85 vs. 155 ± 4.9, respectively; P<0.0001, n=7), and although there were no differences in the MFI values of these cell populations in Lilrb4−/− mice (data not shown, n=7), there were significantly more AF-OVA+MHC class IIhighCD86high cells in the lymph nodes of Lilrb4−/− mice (Fig. 6C).
Fig. 6. Intrathoracic lymph node cell DCs and IL-4 production after a single challenge of Lilrb4+/+ and Lilrb4−/− mice.
Mice were sensitized as described in the legend for Fig. 1. On day 14, intrathoracic lymph node cells were isolated from Lilrb4+/+ and Lilrb4−/− mice as described in the legend for Fig. 5, and the numbers of leukocytes (A, upper panel) and CD11c+ cells (A, lower panel) were determined by cell counting and flow cytometry. Another group of sensitized Lilrb4+/+ and Lilrb4−/− mice received 25 μg of AF-OVA intranasally on day 14, and lymph nodes cells were analyzed on day 15 as on day 14 with the addition of measurements of the percentages, numbers, and MFI of AF-OVA+ cells (B) and the numbers of MHC class II+ and CD86+ cells (C) by flow cytometry. Lymph node cells were isolated on day 15 from an additional group of sensitized Lilrb4+/+ and Lilrb4−/− mice that had been challenged with 25 μg of unlabeled OVA on day 14, and the numbers of IL-4 producing cells was determined with an ELISPOT assay (D). Data are expressed mean ± SEM, n=2, 2, 9, and 9 for the four columns from left-to-right (A); n=7 (B and C); and n=8 (D).
To determine whether the greater number of mature DCs bearing greater amounts of Ag in the lymph nodes of Lilrb4−/− mice affected the magnitude of the Th2 response in the lymph nodes, mice were sensitized daily with OVA-LPS three times, challenged on day 14 with unlabeled OVA, and the frequency of IL-4-producing cells in the lymph nodes was determined with an ELISPOT assay. In accordance with the greater number of mature, OVA-bearing DCs in the LNs of Lilrb4−/− mice, there was a significantly greater number of IL-4-producing cells in the LNs of these mice compared with Lilrb4+/+ animals (Fig. 6D).
Effects of the absence of LIRB4 on DCs on Ag-induced Th2 pulmonary inflammation
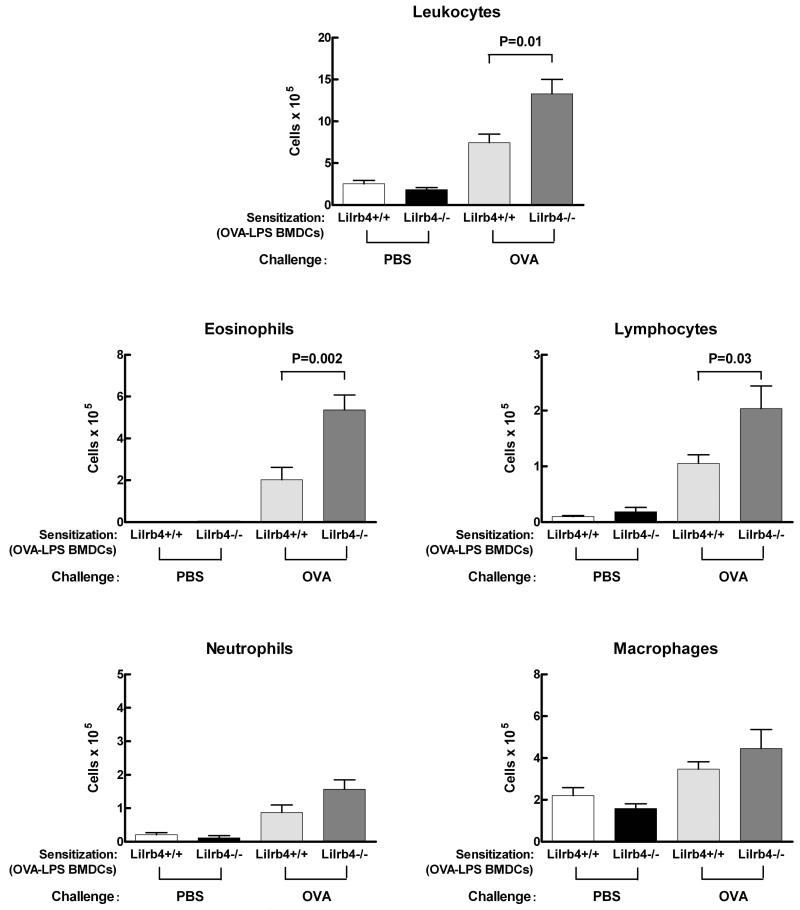
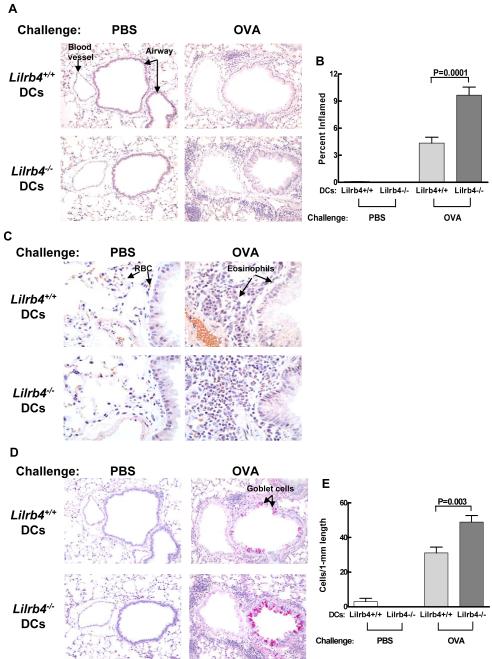
Because LILRB4 is expressed on cell populations other than DCs that could influence the progression of pulmonary inflammation, we sought to determine whether the absence of LILRB4 on DCs alone is sufficient to cause exacerbated pulmonary inflammation in the OVA-LPS model. It has been reported that BMDCs pulsed with OVA or OVA-LPS transfer Ag sensitization to naïve mice (33,34), that BMDCs express LILRB4, and that the absence of this receptor on these culture-derived cells results in greater activation of T cells in vitro (25). We therefore cultured bone marrow cells from Lilrb4+/+ and Lilrb4−/− mice in GM-CSF to generate BMDCs (27). After 9 days of culture, the cells were cultured with medium alone or were pulsed with 100 μg/ml OVA and 100 ng/ml LPS for 18 h. As assessed by flow cytometry, Lilrb4+/+ but not Lilrb4−/− CD11c+ BMDCs expressed LILRB4 before and after Ag pulse (data not shown). BMDCs from Lilrb4+/+ and Lilrb4−/− mice were instilled intranasally (1 × 105 per mouse) into separate groups of Lilrb4+/+ recipients. Thus, the sensitization step in these Lilrb4+/+ mice was replaced with adoptive transfer of Lilrb4+/+ or Lilrb4−/− DCs pulsed with OVA-LPS, and in mice that received Lilrb4−/− cells, only the OVA-LPS-pulsed DCs lacked expression of LILRB4. On days 14, 15, 18, and 19, mice were challenged with 25 μg of OVA intranasally. As determined by analysis of BAL on day 21, Lilrb4+/+ mice that had been sensitized with Lilrb4−/− DCs had significantly greater numbers of total leukocytes, eosinophils, and lymphocytes in their BAL compared with mice that received Lilrb4+/+ DCs (P=0.01, 0.002, and 0.03, respectively), but not neutrophils or macrophages (Fig. 7). In addition, there was more infiltration of inflammatory cells around the bronchovascular bundles in the lungs of Lilrb4+/+ mice that had received Lilrb4−/− DCs compared with mice that had received Lilrb4+/+ cells (Fig. 8A), and the percent of tissue that was inflamed was significantly greater (P=0.001) as determined by quantitative digital image analysis of tissue sections stained with hematoxylin and eosin (Fig. 8B). Tissue eosinophilia in the infiltrates was markedly more pronounced in animals that received Lilrb4−/− DCs as revealed in tissue sections stained with Congo Red (Fig. 8C); indeed the extent of eosinophilia was so great in recipients of Lilrb4−/− DCs that it was not possible to quantify the cell numbers. Goblet cell metaplasia (Fig. 8D), as quantified by the number of goblet cells per length of bronchial epithelium stained with PAS, was also significantly greater in recipients of Lilrb4−/− DCs compared with Lilrb4+/+ DCs (Fig. 8E) (P=0.03). Recipients of Lilrb4−/− DCs also had 1.7-fold higher levels of serum OVA-specific IgE than mice that received Lilrb4+/+ DCs (n=10), an increment identical to that of Lilrb4−/− versus Lilrb4+/+ OVA-LPS/OVA mice (Fig. 3). The combined data indicate that the absence of LILRB4 on DCs was sufficient to confer exacerbated Th2 pulmonary responses in this model.
Fig. 7. Effects of transferred Lilrb4+/+ and Lilrb4−/− BMDCs on BAL cells in Lilrb4+/+ mice.
BMDCs from Lilrb4+/+ or Lilrb4−/− mice were pulsed for 18 h with 100 μg/ml OVA and 100 ng/ml LPS, and 1 × 105 cells in 50 μl of PBS were instilled intranasally into Lilrb4+/+ mice. On days 14, 15, 18, and 19, mice were instilled intranasally with 30 μl of PBS alone or containing 25 μg of OVA, and the mice were euthanized on day 21. The numbers of the different cell populations in BAL fluid was determined as described in the legend for Fig.1.
Fig. 8. Effects of transferred Lilrb4+/+ and Lilrb4−/− BMDCs on lung inflammation and goblet cell hyperplasia in Lilrb4+/+ mice.
Lungs from mice treated as described in the legend for Fig. 7 were processed for histology and tissue sections were stained with chloroacetate esterase to quantify inflammation (A, B), Congo Red to demonstrate tissue eosinophilia (C), and periodic acid-Schiff stain (D, E) to quantify goblet cells. Quantitative data in B and E are expressed as mean ± SEM, n=5, 5, 10, and 10 for the four columns from left-to-right, respectively.
Discussion
We have established that LILRB4 counterregulates the development of a Th2-polarized adaptive immune response and the ensuing allergic pulmonary inflammation induced by Ag sensitization in the presence of a relatively low concentration of LPS followed by Ag challenge in the airway. Moreover, we have found that LILRB4 is expressed on mature, Ag-bearing DCs in lung-draining intrathoracic lymph nodes during sensitization to Ag, and that Lilrb4−/− mice have a greater number of these DCs and IL-4-producing lymphocytes in their lymph nodes after a single challenge with Ag that initiates the pulmonary pathology that ensues after additional exposure to Ag. The absence of LILRB4 on DCs alone was sufficient to induce the exacerbated eosinophilic inflammation in this model, demonstrating that LILRB4 on DCs can contribute significantly to the counterregulatory process afforded by this ITIM-bearing receptor. Hence, LILRB4 dampens not only IgE-induced mast cell activation that elicits immediate hypersensitivity reactions (1) and neutrophil-dependent pathologic inflammation induced by LPS (3) and the combination of IgG immune complexes and LPS (4,5), but also plays an important role in regulating the amplification of an innate immune signal from DCs to the adaptive immune response of T cells.
Our studies confirmed that this model induces Th2 pulmonary inflammation in BALB/c wild-type mice as defined by the preferential infiltration of eosinophils rather than neutrophils into the lung as detected in BAL (Fig. 1). Moreover, we found that Lilrb4−/− mice had significantly greater numbers of both eosinophils and lymphocytes in the BAL compared with Lilrb4+/+ mice (Fig. 1), demonstrating that the absence of LILRB4 can intensify the development of an immunization-induced Th2 response. Accordingly, the number of interstitial mononuclear cells isolated from the lungs of Lilrb4−/− mice was significantly greater than from Lilrb4+/+ mice and consisted of significantly greater numbers of CD4+ and CD8+ T cells, as well as CD19+ B cells (Fig. 2). Hence, LILRB4 in wild-type mice suppressed the induced allergic pulmonary inflammation without altering its hallmark Th2 characteristics.
The inhibitory influence of LILRB4 on the development of the Th2-polarized adaptive immune response was revealed by the presence of significantly augmented levels of OVA-specific IgE in the serum of Lilrb4−/− mice compared with Lilrb4+/+ mice (Fig. 3). We have previously reported that when Lilrb4+/+ and Lilrb4−/− mice are immunized by i.p. injection of OVA adsorbed to alum, there are no differences in the levels of OVA-specific Igs, including IgE (1). Because Th2-polarized immune responses initiated by immunization with OVA/alum can develop in the absence of TLR4 (17) and MyD88 (22), our ability to detect more Ag-specific IgE in the sera of Lilrb4−/− mice sensitized with OVA-LPS in the absence of alum may reflect the TLR4-dependence of the response and its counterregulation by LILRB4 in vivo. Additional evidence that LILRB4 counterregulated the adaptive Th2 response to inhaled OVA-LPS is that upon rechallenge of intrathoracic lymph node cells with Ag, cells from Lilrb4−/− mice produced significantly more IL-4, IL-5, and IL-13, but not more IFN-γ, compared with Lilrb4+/+ mice (Fig. 4). These findings indicate that LILRB4 counterregulates the development of a pathologic adaptive immune response initiated by an innate immune signal that renders an otherwise harmless and tolerizing molecule immunogenic when inhaled (17,35).
A potential contribution for LILRB4 on DCs in this model was appreciated through the finding that a single intranasal instillation of OVA-LPS induced a substantial increase in the number of DCs expressing this molecule in the lung-draining intrathoracic lymph nodes of Lilrb4+/+ mice one day later (Fig. 5). Moreover, there was a striking preferential expression of LILRB4 on OVA-bearing DCs that expressed high levels of MHC class II and CD86. The vast majority of OVA-bearing DCs in lung-draining lymph nodes one day after intranasal instillation of OVA are derived from the airway mucosa, and the number of these cells increases when LPS is administered with OVA, as does their expression of CD86 and ability to present Ag to T cells in vivo (23). Hence, our data reveal that functionally mature pulmonary DCs that have the appropriate localization and capacity to present Ag to T cells preferentially express LILRB4, consistent with a counterregulatory role for this receptor on DCs in the development of allergic lung inflammation.
Although the absence of LILRB4 did not affect the number and/or phenotype of DCs in the intrathoracic lymph nodes of mice during sensitization to OVA-LPS, we considered the possibility that the receptor might influence the response of DCs in sensitized animals to subsequent inhalation of OVA. Airway challenge of Ag-sensitized animals results in increased numbers of airway DCs and migration of the cells to the lung-draining lymph nodes, particularly in the first 24 h after challenge (36-39). Similarly, there is a rapid increase in the number of DCs in the bronchial mucosa of individuals with allergic asthma in response to an inhaled allergen challenge (40). Pulmonary DCs that respond to Ag challenge in sensitized animals contribute importantly to the development and continuation of Th2 airway inflammation and AHR. Depletion of lung DCs at the time of Ag challenge substantially reduces the number of pulmonary T cells and their ability to generate Th2 cytokines in response to Ag, as well as preventing the development of eosinophilic airway inflammation, goblet cell hyperplasia, and AHR (41,42). Because of these key contributions of pulmonary DCs to the pathology induced by inhalation challenge, we compared the DCs of OVA-LPS-sensitized Lilrb4+/+ and Lilrb4−/− mice 1 day after a single challenge with OVA. We found that a significantly greater percentage and number of OVA-bearing DCs expressing high levels of MHC class II and CD86 appeared in the intrathoracic lymph nodes of Lilrb4−/− mice compared with Lilrb4+/+ mice (Fig. 6). In addition, the amount of OVA in the lymph node DCs of Lilrb4−/− mice was greater, which is notable because upregulation of Ag uptake by airway DCs is associated with the progression of allergic airway inflammation (43). We also found that the lymph nodes of challenged Lilrb4−/− mice had significantly more IL-4 producing cells (Fig. 6), which is consistent with the greater number of Ag-bearing mature DCs available to present Ag to memory T cells traversing the lymph nodes after the challenge. Overall, our findings in the lymph nodes of Lilrb4−/− mice are thus consonant with the exacerbated eosinophilic (Figs. 1 and 2), Th2 polarized (Figs. 3 and 4) inflammation that ensues with continued Ag challenge in these mice.
To address whether the expression of LILRB4 on DCs is sufficient to regulate pathologic Th2 responses, we pulsed BMDCs generated from Lilrb4+/+ and Lilrb4−/− mice with OVA in the presence of LPS. Lilrb4+/+ mice were then sensitized by intranasal transfer of the DCs and subsequently challenged by intranasal instillation of OVA. In this adoptive transfer system, Lilrb4+/+ mice that received Lilrb4−/− DCs lacked LILRB4 only on those DCs that would present Ag to T cells in the recipients. The transfer of Lilrb4+/+ DCs led to a robust Th2 pulmonary response to Ag challenge as measured by influx of eosinophils and lymphocytes into BAL (Fig. 7), as well as eosinophilic inflammation near bronchovascular bundles and goblet cell metaplasia in the airway epithelium (Fig. 8). All of these hallmark characteristics of allergic pulmonary inflammation were significantly greater in Lilrb4+/+ mice that had received Lilrb4−/− Ag-pulsed DCs (Figs. 7 and 8), demonstrating that the absence of LILRB4 on DCs alone was sufficient to induce the exacerbated response. It was recently reported that LILRB4 on mouse DCs can counterregulate their ability to activate T cells in vitro, and that the allogeneic graft-versus-host reaction is more severe in Lilrb4−/− recipients, in parallel with greater expression of CD86 on splenic DCs from Lilrb4−/− mice (25). Our in vivo DC transfer system directly establishes that LILRB4 on DCs can contribute importantly to negative regulation of a pathologic allergic airway response that is dependent on the generation of a Th2-polarized adaptive immune response (17).
The ability to discern an effect of LILRB4 on the adaptive immune response in models of allergic pulmonary inflammation may depend on the nature of the model and/or genetic contributions. For example, when Lilrb4−/− mice (on the C57BL/6 background) were sensitized by i.p. injection of ragweed extract and alum followed by intratracheal and intranasal challenges with ragweed, there were no significant differences between Lilrb4+/+ and Lilrb4−/− mice in the levels of serum IgE and IgG1, the amounts of IL-5 or IL-13 secreted from Ag-restimulated splenocytes, and lung inflammation as assessed histologically, although the levels of IL-4 and IL-13 in the BAL were significantly greater in Lilrb4−/− mice (44). The inability to detect a difference in cytokine production by Ag-restimulated lymphocytes from Lilrb4+/+ and Lilrb4−/− mice may have reflected the use of splenocytes rather than lymphocytes from the lung-draining intrathoracic lymph nodes, inasmuch as we detected significantly greater secretion of Th2 cytokines by Ag-rechallenged lymph node cells of Lilrb4−/− mice in the OVA-LPS model (Fig. 4). In addition, our ability to detect a contribution of LILRB4 to the negative regulation of lymphocyte Th2 cytokine production as well as serum IgE levels (Fig. 3) may reflect the fact that in our model, both the sensitizations and challenges were performed in the airway, without the use of i.p. injection of alum that substantially alters the mechanism by which DCs initiate Th2 immune responses (45).
The ability of LILRB4 to negatively regulate DC biology is not limited to mouse cells and models. A human LILRB4 homolog is expressed on DCs (46,47), is upregulated on populations that have been induced to exert negative regulatory functions on effector T cell activation, and contributes to the inhibitory process directly or via regulatory T cells (48-51). Indeed, human LILRB4 fosters allograft acceptance in a humanized mouse model (52), a finding consistent with the exacerbated rejection of allografts in Lilrb4−/− mice (25). It was recently reported that the extracellular domains of mouse and human LILRB4 bind collagen (53), thus identifying another similarity between the human and mouse receptors. Furthermore, LPS upregulates LILRB4 expression on human DCs (54,55) suggesting that, as we have found in the mouse, this innate immune signal induces a counterregulatory response via increased expression of LILRB4.
In summary, we have established that the immune response to inhaled Ag includes an increase in mature, Ag-bearing DCs expressing LILRB4 in lung-draining lymph nodes and that in the absence of LIRB4 there are increased numbers of these cells and cells producing the key Th2 polarizing cytokine IL-4. Concomitantly, a significant exacerbation of the pathologic adaptive immune response and ensuing pulmonary inflammation occur in Lilrb4−/− mice, an effect that can be reiterated when LILRB4 is absent only on DCs. Given the fundamental roles that DCs play in the initiation and perpetuation of allergic airway disease, and the effects of LPS in modulating responses to inhaled Ag in both mice and humans, our findings establish that LILRB4 on DCs can play an important role in counterregulating those processes that are key determinants in the development of allergic airway disease.
Footnotes
Supported by grants AI07306, AI41144, and HL36110 from the National Institutes of Health
- AHR
- airway hyperresponsiveness
- BAL
- bronchoalveolar lavage
- BMDCs
- bone marrow-derived dendritic cells
- DCs
- dendritic cells
- HBSS
- Hanks’ balanced salt solution
- ITIMs
- immunoreceptor tyrosine-based inhibitory motifs
- LILRB4
- leukocyte Ig-like receptor B4
- PAS
- Periodic acid-Schiff
References
- 1.Daheshia M, Friend DS, Grusby MJ, Austen KF, Katz HR. Increased severity of local and systemic anaphylactic reactions in gp49B1-deficient mice. J. Exp. Med. 2001;194:227–233. doi: 10.1084/jem.194.2.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Feldweg AF, Friend DS, Zhou JS, Kanaoka Y, Daheshia M, Li L, Austen KF, Katz HR. gp49B1 suppresses stem cell factor-induced mast cell activation-secretion and attendant inflammation in vivo. Eur. J. Immunol. 2003;33:2262–2268. doi: 10.1002/eji.200323978. [DOI] [PubMed] [Google Scholar]
- 3.Zhou JS, Friend DS, Feldweg AF, Daheshia M, Li L, Austen KF, Katz HR. Prevention of lipopolysaccharide-induced microangiopathy by gp49B1: Evidence for an important role for gp49B1 expression on neutrophils. J. Exp. Med. 2003;198:1243–1251. doi: 10.1084/jem.20030906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhou JS, Friend DS, Lee DM, Li L, Austen KF, Katz HR. gp49B1 deficiency is associated with increases in cytokine and chemokine production and severity of proliferative synovitis induced by anti-type II collagen mAbs. Eur. J. Immunol. 2005;35:1530–1538. doi: 10.1002/eji.200425895. [DOI] [PubMed] [Google Scholar]
- 5.Zhou JS, Xing W, Friend DS, Austen KF, Katz HR. Mast cell deficiency in KitW-sh mice does not impair antibody-mediated arthritis. J. Exp. Med. 2007;204:2797–2802. doi: 10.1084/jem.20071391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Magram J, Connaughton SE, Warrier RR, Carvajal DM, Wu CY, Ferrante J, Stewart C, Sarmiento U, Faherty DA, Gately MK. IL-12-deficient mice are defective in IFNγ production and type 1 cytokine responses. Immunity. 1996;4:471–481. doi: 10.1016/s1074-7613(00)80413-6. [DOI] [PubMed] [Google Scholar]
- 7.Manetti R, Parronchi P, Giudizi MG, Piccinni MP, Maggi E, Trinchieri G, Romagnani S. Natural killer cell stimulatory factor (interleukin 12 [IL-12]) induces T helper type 1 (Th1)-specific immune responses and inhibits the development of IL-4-producing Th cells. J. Exp. Med. 1993;177:1199–1204. doi: 10.1084/jem.177.4.1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.von Mutius E, Braun-Fahrlander C, Schierl R, Riedler J, Ehlermann S, Maisch S, Waser M, Nowak D. Exposure to endotoxin or other bacterial components might protect against the development of atopy. Clin. Exp. Allergy. 2000;30:1230–1234. doi: 10.1046/j.1365-2222.2000.00959.x. [DOI] [PubMed] [Google Scholar]
- 9.Riedler J, Braun-Fahrlander C, Eder W, Schreuer M, Waser M, Maisch S, Carr D, Schierl R, Nowak D, von Mutius E. Exposure to farming in early life and development of asthma and allergy: a cross-sectional survey. Lancet. 2001;358:1129–1133. doi: 10.1016/S0140-6736(01)06252-3. [DOI] [PubMed] [Google Scholar]
- 10.Braun-Fahrlander C, Riedler J, Herz U, Eder W, Waser M, Grize L, Maisch S, Carr D, Gerlach F, Bufe A, Lauener RP, Schierl R, Renz H, Nowak D, von Mutius E. Environmental exposure to endotoxin and its relation to asthma in school-age children. N. Engl. J. Med. 2002;347:869–877. doi: 10.1056/NEJMoa020057. [DOI] [PubMed] [Google Scholar]
- 11.Michel O, Ginanni R, Duchateau J, Vertongen F, Le Bon B, Sergysels R. Domestic endotoxin exposure and clinical severity of asthma. Clin. Exp. Allergy. 1991;21:441–448. doi: 10.1111/j.1365-2222.1991.tb01684.x. [DOI] [PubMed] [Google Scholar]
- 12.Michel O, Kips J, Duchateau J, Vertongen F, Robert L, Collet H, Pauwels R, Sergysels R. Severity of asthma is related to endotoxin in house dust. Am. J. Respir. Crit. Care Med. 1996;154:1641–1646. doi: 10.1164/ajrccm.154.6.8970348. [DOI] [PubMed] [Google Scholar]
- 13.Tulic MK, Wale JL, Holt PG, Sly PD. Modification of the inflammatory response to allergen challenge after exposure to bacterial lipopolysaccharide. Am. J. Respir. Cell Mol. Biol. 2000;22:604–612. doi: 10.1165/ajrcmb.22.5.3710. [DOI] [PubMed] [Google Scholar]
- 14.Gerhold K, Blumchen K, Bock A, Seib C, Stock P, Kallinich T, Lohning M, Wahn U, Hamelmann E. Endotoxins prevent murine IgE production, T(H)2 immune responses, and development of airway eosinophilia but not airway hyperreactivity. J. Allergy Clin. Immunol. 2002;110:110–116. doi: 10.1067/mai.2002.125831. [DOI] [PubMed] [Google Scholar]
- 15.Velasco G, Campo M, Manrique OJ, Bellou A, He H, Arestides RS, Schaub B, Perkins DL, Finn PW. Toll-like receptor 4 or 2 agonists decrease allergic inflammation. Am. J. Respir. Cell Mol. Biol. 2005;32:218–224. doi: 10.1165/rcmb.2003-0435OC. [DOI] [PubMed] [Google Scholar]
- 16.Wan GH, Li CS, Lin RH. Airborne endotoxin exposure and the development of airway antigen-specific allergic responses. Clin. Exp. Allergy. 2000;30:426–432. doi: 10.1046/j.1365-2222.2000.00730.x. [DOI] [PubMed] [Google Scholar]
- 17.Eisenbarth SC, Piggott DA, Huleatt JW, Visintin I, Herrick CA, Bottomly K. Lipopolysaccharide-enhanced, toll-like receptor 4-dependent T helper cell type 2 responses to inhaled antigen. J. Exp. Med. 2002:1645–1651. doi: 10.1084/jem.20021340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Holt PG, Batty JE, Turner KJ. Inhibition of specific IgE responses in mice by pre-exposure to inhaled antigen. Immunology. 1981;42:409–417. [PMC free article] [PubMed] [Google Scholar]
- 19.van Halteren AG, van der Cammen MJ, Cooper D, Savelkoul HF, Kraal G, Holt PG. Regulation of antigen-specific IgE, IgG1, and mast cell responses to ingested allergen by mucosal tolerance induction. J. Immunol. 1997;159:3009–3015. [PubMed] [Google Scholar]
- 20.Seymour BW, Gershwin LJ, Coffman RL. Aerosol-induced immunoglobulin (Ig)-E unresponsiveness to ovalbumin does not require CD8+ or T cell receptor (TCR)-γ/δ+ T cells or interferon (IFN)-γ in a murine model of allergen sensitization. J. Exp. Med. 1998;187:721–731. doi: 10.1084/jem.187.5.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tsitoura DC, DeKruyff RH, Lamb JR, Umetsu DT. Intranasal exposure to protein antigen induces immunological tolerance mediated by functionally disabled CD4+ T cells. J. Immunol. 1999;163:2592–2600. [PubMed] [Google Scholar]
- 22.Piggott DA, Eisenbarth SC, Xu L, Constant SL, Huleatt JW, Herrick CA, Bottomly K. MyD88-dependent induction of allergic Th2 responses to intranasal antigen. J. Clin. Invest. 2005;115:459–467. doi: 10.1172/JCI22462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wikstrom ME, Batanero E, Smith M, Thomas JA, von Garnier C, Holt PG, Stumbles PA. Influence of mucosal adjuvants on antigen passage and CD4+ T cell activation during the primary response to airborne allergen. J. Immunol. 2006;177:913–924. doi: 10.4049/jimmunol.177.2.913. [DOI] [PubMed] [Google Scholar]
- 24.Jung YW, Schoeb TR, Weaver CT, Chaplin DD. Antigen and lipopolysaccharide play synergistic roles in the effector phase of airway inflammation in mice. Am. J. Pathol. 2006;168:1425–1434. doi: 10.2353/ajpath.2006.050986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kasai S, Inui M, Nakamura K, Kakizaki Y, Endo S, Nakamura A, Ito S, Takai T. A novel regulatory role of gp49B on dendritic cells in T-cell priming. Eur. J. Immunol. 2008;38:2426–2437. doi: 10.1002/eji.200737550. [DOI] [PubMed] [Google Scholar]
- 26.Hamada K, Suzaki Y, Goldman A, Ning YY, Goldsmith C, Palecanda A, Coull B, Hubeau C, Kobzik L. Allergen-independent maternal transmission of asthma susceptibility. J. Immunol. 2003;170:1683–1689. doi: 10.4049/jimmunol.170.4.1683. [DOI] [PubMed] [Google Scholar]
- 27.Lutz MB, Kukutsch N, Ogilvie AL, Rossner S, Koch F, Romani N, Schuler G. An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. J. Immunol. Methods. 1999;223:77–92. doi: 10.1016/s0022-1759(98)00204-x. [DOI] [PubMed] [Google Scholar]
- 28.Friend DS, Gurish MF, Austen KF, Hunt J, Stevens RL. Senescent jejunal mast cells and eosinophils in the mouse preferentially translocate to the spleen and draining lymph node, respectively, during the recovery phase of helminth infection. J. Immunol. 2000;165:344–352. doi: 10.4049/jimmunol.165.1.344. [DOI] [PubMed] [Google Scholar]
- 29.Grouls V, Helpap B. Selective staining of eosinophils and their immature precursors in tissue sections and autoradiographs with Congo red. Stain Technol. 1981;56:323–325. doi: 10.3109/10520298109067335. [DOI] [PubMed] [Google Scholar]
- 30.Hayashi T, Ishii A, Nakai S, Hasegawa K. Ultrastructure of goblet-cell metaplasia from Clara cell in the allergic asthmatic airway inflammation in a mouse model of asthma in vivo. Virchows Archiv. 2004;444:66–73. doi: 10.1007/s00428-003-0926-8. [DOI] [PubMed] [Google Scholar]
- 31.Vitetta ES, Ohara J, Myers CD, Layton JE, Krammer PH, Paul WE. Serological, biochemical, and functional identity of B cell-stimulatory factor 1 and B cell differentiation factor for IgG1. J. Exp. Med. 1985;162:1726–1731. doi: 10.1084/jem.162.5.1726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Katz HR, Benson AC, Austen KF. Activation- and phorbol ester-stimulated phosphorylation of a plasma membrane glycoprotein antigen expressed on mouse IL-3-dependent mast cells and serosal mast cells. J. Immunol. 1989;142:919–926. [PubMed] [Google Scholar]
- 33.Lambrecht BN, De Veerman M, Coyle AJ, Gutierrez-Ramos JC, Thielemans K, Pauwels RA. Myeloid dendritic cells induce Th2 responses to inhaled antigen, leading to eosinophilic airway inflammation. J. Clin. Invest. 2000;106:551–559. doi: 10.1172/JCI8107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kuipers H, Hijdra D, de Vries VC, Hammad H, Prins JB, Coyle AJ, Hoogsteden HC, Lambrecht BN. Lipopolysaccharide-induced suppression of airway Th2 responses does not require IL-12 production by dendritic cells. J. Immunol. 2003;171:3645–3654. doi: 10.4049/jimmunol.171.7.3645. [DOI] [PubMed] [Google Scholar]
- 35.Akbari O, DeKruyff RH, Umetsu DT. Pulmonary dendritic cells producing IL-10 mediate tolerance induced by respiratory exposure to antigen. Nature Immunol. 2001;2:725–731. doi: 10.1038/90667. [DOI] [PubMed] [Google Scholar]
- 36.van Rijt LS, Prins JB, Leenen PJ, Thielemans K, de Vries VC, Hoogsteden HC, Lambrecht BN. Allergen-induced accumulation of airway dendritic cells is supported by an increase in CD31hiLy-6Cneg bone marrow precursors in a mouse model of asthma. Blood. 2002;100:3663–3671. doi: 10.1182/blood-2002-03-0673. [DOI] [PubMed] [Google Scholar]
- 37.Vermaelen K, Pauwels R. Accelerated airway dendritic cell maturation, trafficking, and elimination in a mouse model of asthma. Am. J. Respir. Cell Mol. Biol. 2003;29:405–409. doi: 10.1165/rcmb.2003-0008OC. [DOI] [PubMed] [Google Scholar]
- 38.Strickland DH, Stumbles PA, Zosky GR, Subrata LS, Thomas JA, Turner DJ, Sly PD, Holt PG. Reversal of airway hyperresponsiveness by induction of airway mucosal CD4+CD25+ regulatory T cells. Journal of Experimental Medicine. 2006;203:2649–2660. doi: 10.1084/jem.20060155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Huh JC, Strickland DH, Jahnsen FL, Turner DJ, Thomas JA, Napoli S, Tobagus I, Stumbles PA, Sly PD, Holt PG. Bidirectional interactions between antigen-bearing respiratory tract dendritic cells (DCs) and T cells precede the late phase reaction in experimental asthma: DC activation occurs in the airway mucosa but not in the lung parenchyma. Journal of Experimental Medicine. 2003;198:19–30. doi: 10.1084/jem.20021328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jahnsen FL, Moloney ED, Hogan T, Upham JW, Burke CM, Holt PG. Rapid dendritic cell recruitment to the bronchial mucosa of patients with atopic asthma in response to local allergen challenge. Thorax. 2001;56:823–826. doi: 10.1136/thorax.56.11.823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lambrecht BN, Salomon B, Klatzmann D, Pauwels RA. Dendritic cells are required for the development of chronic eosinophilic airway inflammation in response to inhaled antigen in sensitized mice. J. Immunol. 1998;160:4090–4097. [PubMed] [Google Scholar]
- 42.van Rijt LS, Jung S, Kleinjan A, Vos N, Willart M, Duez C, Hoogsteden HC, Lambrecht BN. In vivo depletion of lung CD11c+ dendritic cells during allergen challenge abrogates the characteristic features of asthma. J. Exp. Med. 2005;6:981–991. doi: 10.1084/jem.20042311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.von Garnier C, Wikstrom ME, Zosky G, Turner DJ, Sly PD, Smith M, Thomas JA, Judd SR, Strickland DH, Holt PG, Stumbles PA. Allergic airways disease develops after an increase in allergen capture and processing in the airway mucosa. J. Immunol. 2007;179:5748–5759. doi: 10.4049/jimmunol.179.9.5748. [DOI] [PubMed] [Google Scholar]
- 44.Norris HH, Peterson ME, Stebbins CC, McConchie BW, Bundoc VG, Trivedi S, Hodges MG, Anthony RM, Urban JF, Jr., Long EO, Keane-Myers AM. Inhibitory receptor gp49B regulates eosinophil infiltration during allergic inflammation. J. Leukoc. Biol. 2007;82:1531–1541. doi: 10.1189/jlb.1106667. [DOI] [PubMed] [Google Scholar]
- 45.Kool M, Soullie T, van Nimwegen M, Willart MA, Muskens F, Jung S, Hoogsteden HC, Hammad H, Lambrecht BN. Alum adjuvant boosts adaptive immunity by inducing uric acid and activating inflammatory dendritic cells. J. Exp. Med. 2008;205:869–882. doi: 10.1084/jem.20071087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cella M, Dohring C, Samaridis J, Dessing M, Brockhaus M, Lanzavecchia A, Colonna M. A novel inhibitory receptor (ILT3) expressed on monocytes, macrophages, and dendritic cells involved in antigen processing. J. Exp. Med. 1997;185:1743–1751. doi: 10.1084/jem.185.10.1743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cella M, Jarrossay D, Facchetti F, Alebardi O, Nakajima H, Lanzavecchia A, Colonna M. Plasmacytoid monocytes migrate to inflamed lymph nodes and produce large amounts of type I interferon. Nat. Med. 1999;5:919–923. doi: 10.1038/11360. [DOI] [PubMed] [Google Scholar]
- 48.Chang CC, Ciubotariu R, Manavalan JS, Yuan J, Colovai AI, Piazza F, Lederman S, Colonna M, Cortesini R, Dalla-Favera R, Suciu-Foca N. Tolerization of dendritic cells by T(S) cells: the crucial role of inhibitory receptors ILT3 and ILT4. Nat. Immunol. 2002;3:237–243. doi: 10.1038/ni760. [DOI] [PubMed] [Google Scholar]
- 49.Manavalan JS, Rossi PC, Vlad G, Piazza F, Yarilina A, Cortesini R, Mancini D, Suciu-Foca N. High expression of ILT3 and ILT4 is a general feature of tolerogenic dendritic cells. Transplant Immunol. 2003;11:245–258. doi: 10.1016/S0966-3274(03)00058-3. [DOI] [PubMed] [Google Scholar]
- 50.Vlad G, Piazza F, Colovai A, Cortesini R, Della PF, Suciu-Foca N, Manavalan JS. Interleukin-10 induces the upregulation of the inhibitory receptor ILT4 in monocytes from HIV positive individuals. Hum. Immunol. 2003;64:483–489. doi: 10.1016/s0198-8859(03)00040-5. [DOI] [PubMed] [Google Scholar]
- 51.Beinhauer BG, McBride JM, Graf P, Pursch E, Bongers M, Rogy M, Korthauer U, De Vries JE, Aversa G, Jung T. Interleukin 10 regulates cell surface and soluble LIR-2 (CD85d) expression on dendritic cells resulting in T cell hyporesponsiveness in vitro. Eur. J. Immunol. 2004;34:74–80. doi: 10.1002/eji.200324550. [DOI] [PubMed] [Google Scholar]
- 52.Vlad G, D’Agati VD, Zhang QY, Liu Z, Ho EK, Mohanakumar T, Hardy MA, Cortesini R, Suciu-Foca N. Immunoglobulin-like transcript 3-Fc suppresses T-cell responses to allogeneic human islet transplants in hu-NOD/SCID mice. Diabetes. 2008;57:1878–1886. doi: 10.2337/db08-0054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Jiang L, Barclay AN. New assay to detect low-affinity interactions and characterization of leukocyte receptors for collagen including leukocyte-associated Ig-like receptor-1 (LAIR-1) Eur. J. Immunol. 2009;39:1167–1175. doi: 10.1002/eji.200839188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Velten FW, Duperrier K, Bohlender J, Metharom P, Goerdt S. A gene signature of inhibitory MHC receptors identifies a BDCA3(+) subset of IL-10-induced dendritic cells with reduced allostimulatory capacity in vitro. Eur. J. Immunol. 2004;34:2800–2811. doi: 10.1002/eji.200324732. [DOI] [PubMed] [Google Scholar]
- 55.Ju XS, Hacker C, Scherer B, Redecke V, Berger T, Schuler G, Wagner H, Lipford GB, Zenke M. Immunoglobulin-like transcripts ILT2, ILT3 and ILT7 are expressed by human dendritic cells and down-regulated following activation. Gene. 2004;331:159–164. doi: 10.1016/j.gene.2004.02.018. [DOI] [PubMed] [Google Scholar]