Abstract
The application of small RNA in therapy has been hindered by the lack of an efficient and safe delivery system to target specific cells. Packaging RNA (pRNA), part of the DNA-packaging motor of bacteriophage phi29(φ29), was manipulated by RNA nanotechnology to make chimeric RNAs that form dimers via interlocking right- and left-hand loops. Fusing pRNA with receptor-binding RNA aptamer, folate, small interfering RNA (siRNA), ribozyme, or another chemical group did not disturb dimer formation or interfere with the function of the inserted moieties. Incubation of cancer cells with the pRNA dimer, one subunit of which harbored the receptor-binding moiety and the other harboring the gene-silencing molecule, resulted in their binding and entry into the cells, and subsequent silencing of anti/proapoptotic genes. The chimeric pRNA complex was found to be processed into functional double-stranded siRNA by Dicer (RNA-specific endonuclease). Animal trials confirmed the suppression of tumorigenicity of cancer cells by ex vivo delivery. It has been reported [Shu, D., Moll, W.-D., Deng, Z., Mao, C., and Guo, P. (2004). Nano Lett. 4:1717–1724] that RNA can be used as a building block for bottom-up assembly in nanotechnology. The assembly of protein-free 25-nm RNA nanoparticles reported here will allow for repeated long-term administration and avoid the problems of short retention time of small molecules and the difficulties in the delivery of particles larger than 100 nm.
OVERVIEW SUMMARY
The successful use of small RNA for therapeutic purposes requires a safe and efficient delivery system capable of targeting specific cells. φ29 motor RNA (pRNA) was manipulated to produce chimeric RNAs that formed dimers through connection by right- and left-hand loops. Fusion of the pRNA with a variety of therapeutic and chemical compounds did not impede the formation of dimers or interfere with moiety function. Incubation of cells with dimer, one subunit of which carried a gene-silencing molecule and the other a receptor-binding moiety, resulted in successful binding, entry, and silencing of apoptotic genes. Animal trials further confirmed suppression of the tumorigenicity of cancer cells by ex vivo delivery. The dimeric RNA chimera was processed into double-stranded small interfering RNA (siRNA) by Dicer. These protein-free 25-nm nanoparticles will allow for repeated and long-term administration escaping immunoresponse and avoid the short retention time of smaller molecules and the undeliverability of larger molecules.
INTRODUCTION
The development of molecular gene therapy is one of the most promising applications of modern biological science. Small interfering RNA (siRNA) (Elbashir et al., 2001; Brummelkamp et al., 2002; Carmichael, 2002; Li et al., 2002; McCaffrey et al., 2002), ribozymes (Blount and Uhlenbeck, 2002; Hoeprich et al., 2003), and antisense RNA (Kumar and Carmichael, 1998) have shown significant potential as novel molecular approaches to downregulate specific gene expression. siRNA was also successfully used for knocking down human immunodeficiency virus (HIV)-related gene expression (Novina et al., 2002; Akkina et al., 2003). To be successfully applied in the treatment of cancer and infectious diseases, siRNAs must be able to (1) enter cells despite size limitations on membrane penetration, (2) survive degradation by nucleases within the cells, (3) traffic into the appropriate cell compartment, (4) fold correctly in the cell, if fused to a carrier, and (5) home to specific cells. Hence, the development of a safe, efficient, specific, and nonpathogenic system for the delivery of therapeutic RNA is highly desirable.
We discovered a 117-nucleotide bacteriophage φ29-encoded packaging RNA (pRNA) that plays a novel and essential role in packaging DNA into procapsids (Fig. 1) (Guo et al., 1987). Six copies of pRNA form a hexameric ring to drive the DNA-packaging motor (Trottier and Guo, 1997; Guo et al., 1998; Zhang et al., 1998). pRNA dimers are the building blocks of hexamers (Chen et al., 2000). Hand-in-hand interaction of the right and left interlocking loops can be manipulated to produce desired stable dimers, trimers, or hexamers (Guo et al., 1998; Zhang et al., 1998; Chen et al., 1999; Shu et al., 2003). The size of pRNA dimer is about 25 nm (Hoeprich and Guo, 2002; Shu et al., 2004).
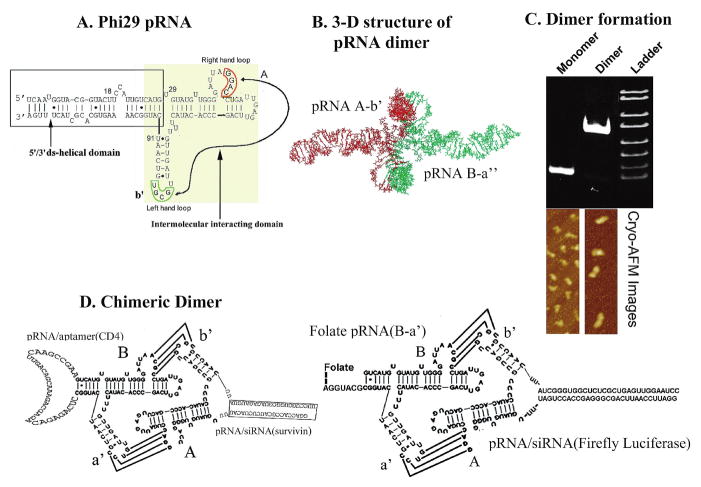
FIG. 1.
Sketch of sequence and structure of pRNA chimeras. (A) φ29 pRNA sequence and secondary structure. The right- and left-hand loops are circled in orange and green, respectively. The double-stranded helical domain on the 5′/3′ ends is framed, and the domain for dimer formation is shaded. The curved line points to the two interacting loops. (B) Three-dimensional structure of pRNA dimer. (C) Native polyacrylamide gel showing monomer and dimer of the pRNA chimeras exhibiting different migration rates. Below the gel are cryo-AFM images of ′29 pRNA monomer and dimer. The colors reflect the thickness and height of the molecule; the brighter the color, the thicker or taller the molecule. (D) Design of chimeric pRNA dimers harboring foreign moieties (see Nomenclature of RNA Subunits, under Results).
Our work indicates that RNA, and especially pRNA, can serve as a building block to build nanomaterials via bottom-up assembly (Shu et al., 2004). The structural and molecular features of φ29 pRNA allow its easy manipulation, making it possible to redesign its parts as gene-targeting and delivery vehicles. The pRNA molecule contains intermolecular interaction domains and a 5′/3′ helical domain (Fig. 1) (Zhang et al., 1995; Garver and Guo, 1997; Chen et al., 1999, 2000). Replacement or insertion of the 5′/3′ helical domain does not interfere with dimer formation (Chen et al., 1999).
The feasibility of these ideas was tested by the construction of chimeric pRNA dimers. One subunit of the dimer contained a receptor-binding RNA aptamer or folate for cell recognition, and the other harbored a moiety of siRNA, ribozyme, or chemical group. The dimers were delivered to specific cells to silence the genes for green fluorescent protein (GFP), luciferase, survivin, and other pro/anti-apoptotic members of the BCL-2 family in a variety of cancer cells.
MATERIALS AND METHODS
In vitro synthesis and physical characterization of RNA
RNAs were prepared as described (Zhang et al., 1995). DNA oligonucleotides (oligos) were synthesized with the desired sequences and used to produce double-stranded DNA by polymerase chain reaction (PCR). The DNA products containing the T7 promoter were cloned into plasmids or used as a substrate for direct in vitro transcription. All pRNA chimeras were treated with calf intestinal alkaline phosphatase (CIP) to remove the 5′-phosphate and eliminate PKR (protein kinase RNA-activated) and interferon effects (Kim et al., 2004) or synthesized in the presence of SH-AMP, biotin–AMP, or CoA (Li et al., 2005; Khaled et al., 2005). Methods of electrophoresis and cryo-atomic force microscopy (cryo-AFM) for the detection of pRNA dimer have been described (Chen et al., 2000; Shu et al., 2003). Magnesium (10 mM) was included in all buffers to maintain the folding of pRNA and the formation of dimers (Chen et al., 2000; Mat-Arip et al., 2001).
Transfection assay for monomeric chimeric pRNA subunits
For Drosophila S2 cells, various siRNAs and GFP-coding plasmid pMT-GFP were cotransfected in a 24-well plate, using Cellfectin (Invitrogen, Carlsbad, CA). The expression of GFP was induced by overnight incubation with CuSO4 at 0.5 mM (Li et al., 2002).
For luciferase assay of monomer pRNA/siRNA, various chimeric siRNAs were cotransfected into mouse fibroblast PA317-PAR cells with both plasmid DNA pGL3 encoding firefly luciferase and pRL-TK (Promega, Madison, WI) encoding Renilla luciferase. Luciferase activities were measured in a dual reporter assay system (Promega) 1 day after transfection.
For survivin knockdown assay, MDA-231, PC-3, A-549, T47D, and MCF-7 cells were transfected with various RNAs at 20 pmol/well in 24-well plates, using Lipofectamine2000 (Invitrogen). The next day, cells were observed under a phase-contrast microscope and scored on the basis of viability.
For pro-B FL5.12A cells, 107 cells were resuspended in 500 μl of RPMI 1640 with 10% fetal bovine serum (FBS). For D1 cells, the cells were resuspended in hypo-osmolar buffer. Electroporation was performed with an ElectroSquarePorator (ECM 830; BTX Molecular Delivery Systems/Harvard Apparatus, Holliston, MA). FL5.12A cells were electroporated at 200 V (three pulses) and D1 cells were electroporated at 180 V (one pulse). After a short incubation on ice, cells were resuspended in 10 ml of complete medium with cytokine and incubated for 2 hr. Cell viability was measured by trypan blue assay before transfection with Mirus reagent (Mirus Bio, Madison, WI). Cells (106) were incubated with the RNA constructs (100 nM) overnight, washed twice with Hanks’ balanced salt solution, and incubated for an additional 24 to 48 hr in the medium with or without cytokine.
Chimeric pRNA/siRNA processing by Dicer
Chimeric siRNA was incubated with purified recombinant RNA-specific endonuclease (Dicer; Genlantis, San Diego, CA) for 2 hr at 37°C. RNA was labeled with 32P at the 5′ end, using T4 polynucleotide kinase (New England BioLabs).
Functional delivery assays for chimeric pRNA dimer harboring CD4 aptamer and survivin siRNA
CD4hi, CD4lo, and CD4neg cells were seeded in a 96-well plate. Cells (5 × 104 per sample) were washed once. Cells were incubated with 100 nM RNA dimer for 30 min. After rinsing, cells were further incubated for 24 or 48 hr, with or without cytokines. Cell viability was measured by trypan blue assay.
CD4 receptor-binding assay by confocal microscopy
D1 cells were grown in RPMI 1640 with 10% FBS, penicillin–streptomycin (50 IU/ml), 0.1% 2-mercaptoethanol, and interleukin-7 (IL-7, 50 ng/ml). FL5.12A cells were grown in complete medium supplemented with IL-3 (2 ng/ml). For the expression of CD4 in D1 cells, the L3T4 (mouse CD4) insert was subcloned into pcDNA 6/V5-HisB (Invitrogen). Stable lines were selected by antibiotic resistance. D1-CD4 cells, expressing high levels of CD4, were further isolated by fluorescence-activated cell sorting (FACS). The D1-CD4 cell line was maintained in complete medium supplemented with IL-7 (50 ng/ml) and blasticidin HCl (2.5 mg/ml). Coverslips coated with poly-L-lysine (200 μg/ml) were incubated overnight with cells. Before fixing, coverslips with cells were rinsed and treated for 30 min in a 65 nM solution of dimeric RNA complex. Coverslips with cells were fixed with 4% paraformaldehyde and mounted in Gel/Mount (Biomeda). Images were captured with a confocal microscope (LSM 510 NLO; Carl Zeiss).
Specific gene knockdown mediated by folate receptor
Folate dimer was prepared by mixing folate-pRNA(7–106) B-a′ and pRNA/siRNA(firefly luciferase) A-b′ with 10 mM Mg2+. KB cells were seeded in a six-well plate in folate-free medium. After being washed with phosphate-buffered saline (PBS) containing MgCl2, the premixed dimer RNA (1.75 μM) was then added to the cells and incubated for 3 hr at 37°C. RNase inhibitor (SUPERase · IN, 1 unit/μl; Ambion, Austin, TX) was added to the binding buffer. After incubation, free RNA was washed off and pGL3 and pRL-TK plasmids were introduced into the cells, using Lipofectamine2000 (Promega). Luciferase activities were measured the next day.
Double-labeling and flow cytometry
MCF-7 cells were transfected with RNA samples at a 100 nM concentration. Cells were stained with annexin V and propidium iodide (PI) followed by flow cytometry assay.
Animal studies
Pathogen-free male 5-week-old athymic nude mice (Harlan, Indianapolis, IN) were housed in a specific pathogen-free environment. Food supplies and instruments were autoclaved, and all manipulations were performed in a laminar flow hood. Animals were randomly assigned to experimental groups. Cells were inoculated subcutaneously at the right axilla of the forelimb.
RESULTS
Nomenclature of RNA subunits
To simplify description of the construction of RNA complexes, upper-case and lower-case letters are used to represent the right- and left-hand loops of the pRNA, respectively (Fig. 1A). Matched letters indicate complementarity, whereas different letters indicate noncomplementary loops. For example, pRNA(A-b′) contains right-hand loop A (5′G45G46A47C48) and left-hand loop b′ (3′U85G84C83G82), which can pair with the left-hand loop a′ (3′C85C84U83G82) and right-hand loop B (5′A45C46G47C48), respectively, of pRNA(B-a′) (Fig. 1). pRNA/aptamer(CD4) denotes a pRNA chimera that harbors an aptamer that binds CD4, and pRNA/siRNA(GFP) represents a pRNA chimera that harbors an siRNA targeting green fluorescent protein (GFP). pRNA/ribozyme(survivin) represents a chimeric pRNA harboring a hammerhead ribozyme against survivin.
Construction of chimeric pRNA subunits harboring foreign moieties
Construction of chimeric pRNA harboring siRNA
pRNA contains a double-stranded helical domain at 5′/3′ end and an intermolecular binding domain, which fold independently of each other. Complementary modification studies have revealed that altering the primary sequences of any nucleotide of the helical region does not affect pRNA structure and folding as long as the two strands are paired (Fig. 1) (Zhang et al., 1994). Extensive studies revealed that siRNA is a double-stranded RNA helix (Elbashir et al., 2001; Brummelkamp et al., 2002; Carmichael, 2002; Li et al., 2002). To test whether it is possible to replace the helical region in pRNA with double-stranded siRNA, a variety of chimeric pRNAs with different targets were constructed to carry siRNA connected to nucleotides 29 and 91 of the pRNA (Fig. 1), resulting in pRNA/siRNA(GFP), pRNA/siRNA(luciferase), pRNA/siRNA(BAD), and pRNA/siRNA(survivin) that were competent in gene sliencing (see below).
Construction of chimeric pRNA harboring receptor-binding aptamer or ribozyme
To achieve specific delivery of therapeutic complexes, it is often necessary to incorporate a moiety that recognizes signature molecules on cell surfaces. In comparison with antibodies and phage-displayed peptides, RNA aptamer is an attractive alternative because it avoids the induction of immune responses (Goldsby et al., 2002). Using the SELEX (systematic evolution of ligands by exponential enrichment) approach (Gold, 1995; Ellington and Szostak, 1990), a number of RNA aptamers were obtained that specifically recognize a particular cell surface receptor such as CD4 (Kraus et al., 1998). One CD4-binding RNA aptamer was chosen to construct chimeric pRNA/aptamer(CD4) via a mutual 5′/3′ end connection (Fig. 1D). The pRNA vector was reorganized into a circularly permuted form, with the nascent 5′ and 3′ ends relocated to residues 71 and 75, respectively, of the original pRNA sequence. The 71/75 end is located in a tightly folded area to bury and protect the ends from exonuclease degradation in vivo (Hoeprich et al., 2003). Similar rules were followed to construct the chimeric pRNA/ribozyme(survivin). All aptamers or ribozymes fused to the pRNA were functional (see below).
Construction of chimeric pRNA harboring folate
Folate receptors are overexpressed in various types of tumors such as human nasopharyngeal epidermal carcinoma, but are generally absent in normal adult tissues. Many therapeutic reagents such as low molecular weight drugs, antisense oligonucleotides, and protein toxins have been conjugated to folate and then delivered to tumor cells (Sudimack and Lee, 2000; Lu and Low, 2003). The same strategy was employed in this study to deliver siRNA to folate receptor-overexpressing tumor cells. The folate molecule was incorporated into the 5′ end of RNA and formed a dimer with a pRNA/siRNA chimera to achieve specific delivery (Fig. 1D). To increase the accessibility of the folate molecule to folate receptor on the cell surface, folate-labeled RNA was designed to be a 5′ overhang, in which nucleotides 107 to 117 of pRNA were truncated.
Processing of chimeric pRNA/siRNA complex into double-stranded siRNA by Dicer
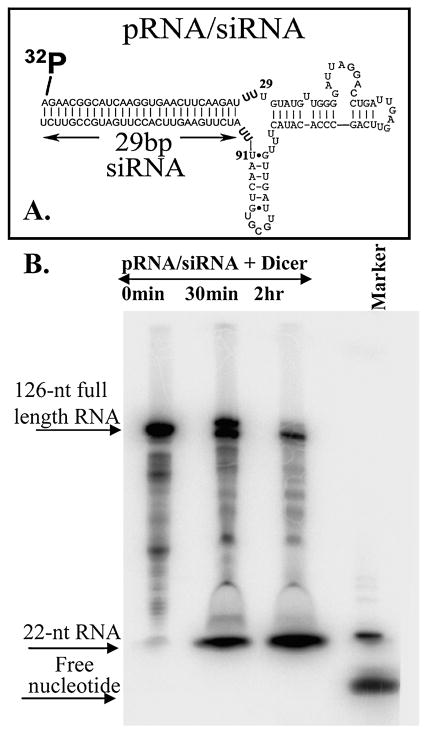
To determine whether the chimeric complexes could be processed into functional siRNA, chimeric pRNA/siRNA was subjected to treatment with purified recombinant Dicer, which is well known for its function of processing long double-stranded RNA into 22-bp siRNAs in vitro and in vivo (Hannon, 2002). The chimeric pRNA/siRNA complex used as the substrate in this study harbored a 29-bp double-stranded siRNA connected to the pRNA intermolecular interaction domain from nucleotides 29 to 91 (Fig. 2A). Two additional uridines were used to link the siRNA to the pRNA domain to help enhance processing efficiency by increasing the ΔG for the folding of the loop. Purified chimeric pRNA/siRNA complex was labeled at the 5′ end with 32P and incubated with Dicer, and the digestion product was then analyzed on denaturing PAGE/urea gels. As shown in Fig. 2, digestion of pRNA/siRNA by Dicer for 30 min to 2 hr resulted in the production of 22-base siRNA with high efficiency. This result confirms that the chimeric pRNA/siRNA was cleaved and released the functional double-stranded siRNA located at the 5′/3′ ends.
FIG. 2.
Processing of chimeric pRNA/siRNA complex by Dicer. The structures of pRNA/siRNA and pRNA vector are shown in (A) and (B). Processing of pRNA/siRNA into 22-bp siRNAs by recombinant Dicer. The chimeric pRNA/siRNA with 5′-end 32P labeling was incubated with purified recombinant Dicer for 30 min and 2 hr, respectively, and then separated on a denaturing PAGE/urea gel. A radiolabeled 22-nucleotide RNA was used as a molecular weight marker.
Functional assay of chimeric pRNA subunits by transfection pRNA/siRNA(GFP)
To test the function of pRNA/siRNA(GFP), GFP-expressing plasmid was cotransfected with various RNA chimeras into cells. Fluorescence microscopy revealed that pRNA/siRNA(GFP) effectively inhibited GFP gene expression in a dose-dependent manner (Fig. 3A). In contrast, such inhibitory effects were not observed with a control construct containing site-directed mutations within siRNA sequences. Nonspecific inhibition by pRNA vector was ruled out through a control (Fig. 3B) with the vector alone (nucleotides 23–97). The effect of gene silencing on mRNA level was further demonstrated by Northern blot (Fig. 3D). In both assays, chimeric pRNA/siRNA(GFP) exhibited equivalent or superior inhibitory effects on GFP gene expression compared with chemically synthesized double-stranded siRNA(GFP) (Fig. 3C and D).
FIG. 3.
Functional assay of chimeric pRNA/siRNA(GFP) by transfection. (A–C) Fluorescence microscopy images showing the silencing of GFP gene by transfection. (A) Dose-dependent silencing of GFP gene by chimeric pRNA/siRNA(GFP) (left column). A mutant pRNA/siRNA (right column) served as negative control. (B) GFP expression of cells transfected with various RNAs: (a) no RNA; (b) synthesized double-stranded siRNA(GFP); (c) double-stranded siRNA(LacZ) control; (d) pRNA/siRNA(GFP); (e) pRNA/siRNA(mutant); (f) pRNA vector alone. (C) Comparison of the performance of (a) chimeric pRNA/siRNA(GFP) and (b) conventional double-stranded siRNA(GFP) at the same molar concentration; (c) control with no siRNA treatment. (D) Northern blot to examine the effect of chimeric pRNA/siRNA(GFP) on GFP mRNA level after transfection. Lanes 1 and 2 show the effects of two different constructs of pRNA/siRNA(GFP); lane 3, double-stranded siRNA; lane 4, cells without RNA treatment. rRNA was used as loading control.
pRNA/siRNA(luciferase)
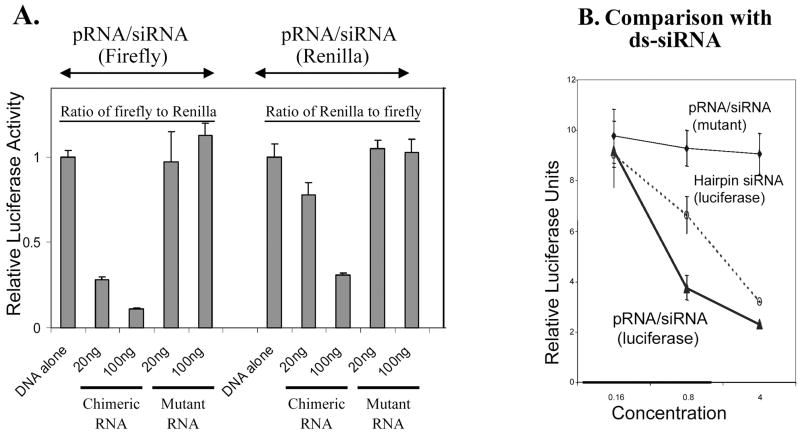
In addition to the GFP-specific chimeric siRNA, pRNA/siRNA constructs against luciferase were also constructed and tested. Two chimeric pRNA/siRNA constructs targeting either firefly luciferase or Renilla luciferase were introduced into cells by transient transfection in separate experiments, and the expression levels of both luciferases were then measured simultaneously in a dual reporter assay. When the targeted luciferase was examined, the nontargeted luciferase served as the internal control. As shown in Fig. 4A, each construct was found to suppress its target gene efficiently and specifically. No silencing of the luciferase genes occurred when mutations were introduced into the siRNA of the pRNA complexes. In addition, the silencing effect of pRNA/siRNA(firefly) was found to be more efficient than that of hairpin siRNA(firefly) alone (Fig. 4B).
FIG. 4.
Functional assay of chimeric pRNA/siRNA targeting luciferase by transfection. (A) Dual reporter luciferase assay showing the specific knockdown of firefly luciferase or Renilla luciferase expression by pRNA/siRNA(firefly) or pRNA/siRNA(Renilla), respectively, in a dose-dependent manner. (B) Comparison of the activities of conventional hairpin siRNA(luciferase) and pRNA/siRNA(luciferase). pRNA/siRNA(mutant) with mutations in siRNA sequences was included as a nonspecific control.
pRNA/siRNA(survivin) and pRNA/ribozyme(survivin) knock down antiapoptosis factor survivin and initiate cell death
To evaluate the effectiveness of therapeutic RNA molecules in treating cancer, it is necessary to suppress genes involved in tumor development and progression. Survivin was chosen as a target because it inhibits apoptosis and is detected only in cancer cells and not in normal adult cells. It has been shown that the suppression of survivin induces the apoptosis of cancer cells (Grossman et al., 2001; Pennati et al., 2003).
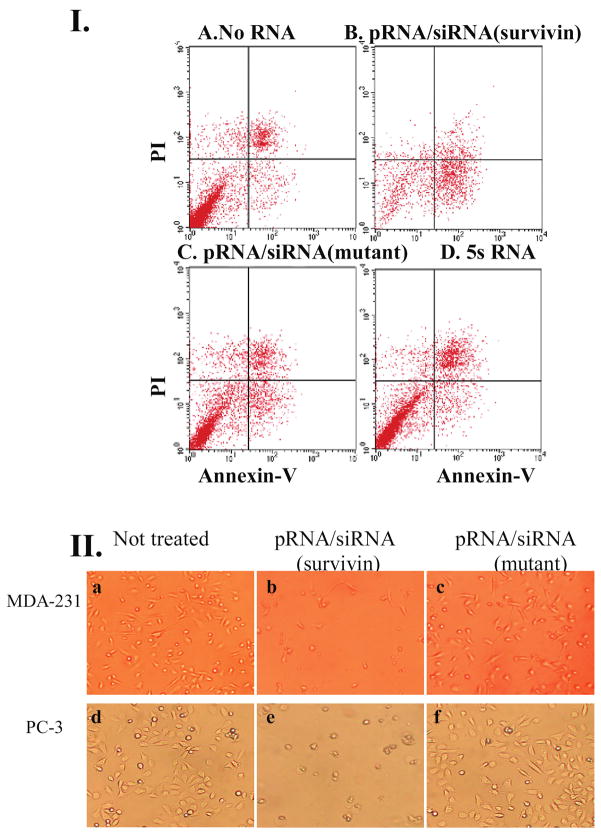
The function and specificity of pRNA/siRNA(survivin) were first examined in breast and prostate cancer cells. The apoptosis of breast cancer cells transfected with pRNA/siRNA(survivin) was assessed with annexin V–propidium iodide (PI) double-staining followed by flow cytometry analysis. As shown in Fig. 5.I.B, cells transfected with pRNA/siRNA(survivin) were shown at a much higher percentage in the bottom right area representing apoptotic cells, compared with those treated with mutant chimeric siRNA or 5S RNA as negative controls (Fig. 5.I.A, C, and D). The effects of chimeric siRNA on cell survival were also evaluated by cell morphology studies in breast cancer cell line MDA-231 and prostate cancer cell line PC-3. After transfection, the majority of cells shrank and were detached from the cell culture plate, whereas the control pRNA/siRNA(mutant) did not induce cell death (Fig. 5.II).
FIG. 5.
Apoptosis and cell death induced by transfection of chimeric pRNA harboring siRNA targeting survivin. (I) Breast cancer MCF-7 cells were transfected with pRNA/siRNA(survivin) and apoptosis was monitored by PI–annexin V double-labeling followed by flow cytometry. Cells in the bottom right quadrant represent apoptotic cells. (II) Breast cancer cells (MDA-231) and prostate cancer cells (PC-3) were transfected with 20 pmol of pRNA/siRNA(survivin) in 24-well plates and images were taken 24 hr after transfection. The mutant pRNA/siRNA was transfected in parallel as a negative control.
The knockdown effect by pRNA/ribozyme(survivin) was also tested. When several human cancer cell lines were transfected with pRNA/ribozyme(survivin), more than 80% of the cells were dead 24 hr after transfection.
pRNA/siRNA(BAD) silences proapoptosis factor and prevents cell death
Inducing cell death by knocking down anti-apoptosis factors would be ideal in cancer therapy, but it might not be sufficient to demonstrate the specificity of the RNA delivery strategy, because cell death can potentially be caused by nonspecific effects, if any occur. To rule out the possibility of nonspecific cytotoxicity, proapoptosis factor BAD in the Bcl-2 family (Khaled et al., 2001) was selected as the target, because silencing proapoptotic factors would prevent apoptosis instead of causing cell death (Kumar and Vaux, 2002). Thus, the knockdown effect from siRNA could be distinguished from the nonspecific toxicity of the RNA components. To this end, IL-3-dependent pro-B cell line FL5.12A was employed. Withdrawal of IL-3 could induce the expression of BAD, leading to apoptosis of FL5.12A cells (Khaled and Durum, 2002). pRNA/siRNA(BAD) was constructed and introduced into the pro-B cell line, and Western blot indicated that there was a significant decrease in BAD protein (Fig. 6B), whereas the mutant controls and pRNA vector alone resulted in only minor decreases in BAD protein compared with untreated cells. The viability assay revealed that the transfection of pRNA/siRNA(BAD) protected FL5.12A cells from death on IL-3 removal, and did not cause cell death in the presence of IL-3 (Fig. 6A). These results demonstrate that chimeric pRNA/siRNA can specifically silence the expression of targeted proapoptotic genes and prevent growth factor withdrawal-induced cell death. In contrast, cell death was induced by pRNA/siRNA(Survivin) in the presence of IL-3, and exacerbated on IL-3 withdrawal, compared with the mutant chimeric siRNA control (Fig. 6C).
FIG. 6.
Functional assay of pRNA/siRNA chimera targeting proapoptotic factor BAD. (A) pRNA/siRNA(BAD) and control siRNAs (10 nM) were introduced into pro-B cells by electroporation, combined with a transfection reagent on day 1. Cells were washed to remove IL-3 on day 2 and assayed for viability on day 3. (B) BAD protein levels were compared in cells transfected with chimeric siRNA(BAD) or two mutant controls containing different mutations within siRNA sequences. Control cells were treated with pRNA alone. Cell lysates were prepared (Khaled et al., 2001) on day 3 and proteins were separated by 12% SDS–PAGE followed by Western blot with BAD antibody (Cell Signaling Technology, Beverly, MA). Numbers below the panel indicate the remaining BAD level, expressed as a percentage compared with control cells. (C) Pro-B cells transfected with pRNA/siRNA(survivin) or mutant were grown in complete medium containing IL-3 or deprived of IL-3. The impact of RNA on cell morphology was observed by microscopy.
Assembly of pRNA dimers composed of chimeric subunits harboring individual functional subunits
Effective gene therapy requires at least two features: specific cell recognition and the silencing of specific gene(s) in cells. Construction of a RNA molecule with both functionalities would satisfy these requirements, but direct fusion or conjugation of one RNA with two or more moieties could lead to misfolding and loss of function. Construction of RNA dimer is an alternative approach for achieving these two goals.
Extensive studies of φ29 pRNA revealed that two pRNA subunits form a dimer through the interaction of the complementary left- and right-hand loops of pRNA monomers (Guo et al., 1998; Zhang et al., 1998; Chen et al., 1999). The extendable 5′/3′ ends of dimer provide two separated sites to carry extra sequences without altering the secondary structure of the pRNA vectors or the inserted sequences (Shu et al., 2004). The dimer formation mechanism was therefore used to construct the RNA complex to deliver functional moieties for (1) specific recognition mediated by receptor-binding RNA aptamer or folate, and (2) regulation of cell functions (growth, death, physiology, etc.) mediated by siRNA or ribozyme.
The sequence responsible for intermolecular pRNA/pRNA interaction is located between residues 23 and 97 (Chen et al., 1999). Chimeric monomer subunits were intentionally designed to possess either A-b′ or B-a′ to match with each other. When pRNA A-b′ and B-a′ were mixed in a 1:1 molar ratio in the presence of 10 mM Mg2+, pRNA dimers were produced with high efficiency, as confirmed by gel electrophoresis and cryo-AFM imaging (Fig. 1C). In AFM images, the monomer exhibits a checkmark shape and the dimer is twice as large as the monomer. Dimers were generated from monomer subunits despite the replacement of the 5′/3′ helix with double-stranded siRNA or connection of the 5′/3′ end to an aptamer or ribozyme. The correct folding of pRNA chimera was also confirmed by φ29 in vitro assembly inhibition assay as previously reported (Guo et al., 1987, 1998).
Functional assay of chimeric pRNA dimer by incubation
Binding assay of chimeric pRNA containing CD4 aptamer
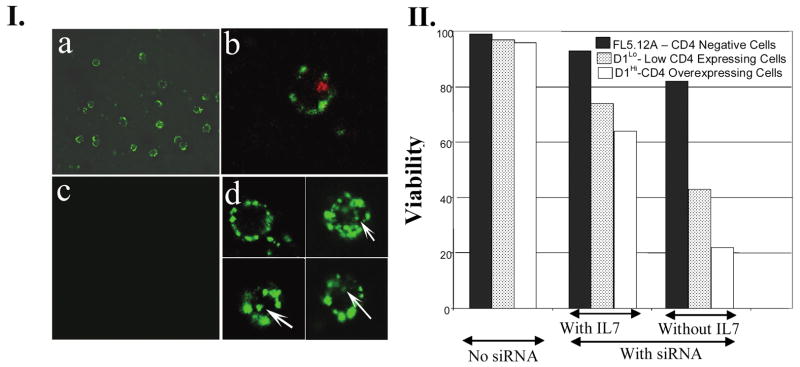
CD4 is a receptor displayed on the surface of certain T lymphocytes. A CD4-overexpressing T cell line (referred to as CD4hi) was developed from a murine IL-7-dependent pro-T cell line D1 (referred to as CD4low) (Kim et al., 2003) that normally expresses undetectable levels of endogenous CD4. Incubation of CD4hi cells with RNA dimer composed of pRNA(A-b′)/aptamer(CD4) and pRNA(B-a′)/FITC (fluorescein isothiocyanate) revealed strong and specific binding (Fig. 7.I), because binding was not detected in FL5.12A cells with no CD4 expression (Fig. 7.I.c) or FITC–dimer without aptamer(CD4) (data not shown).
FIG. 7.
Specific delivery of chimeric pRNA/siRNA by CD4 receptor. (I) Green circles corresponding to the binding of FITC-labeled pRNA dimer containing CD4-binding aptamer to lymphocytes were shown by confocal microscopy (a) and the entry of RNA was shown as a green spot inside the cell (d). Texas Red-labeled transferrin was used as a positive control of internalization (b). No binding was observed in the control cell line without CD4 expression (c). (II) Incubation of RNA dimer led to the specific suppression of cell viability, as measured by trypan blue exclusion assay. Three different cell lines with no, low, or high CD4 expression level were incubated with the pRNA/siRNA(survivin)-pRNA/aptamer(CD4) complex in the presence or absence of IL-7.
Incubation with dimer harboring both aptamer(CD4) and siRNA(survivin)
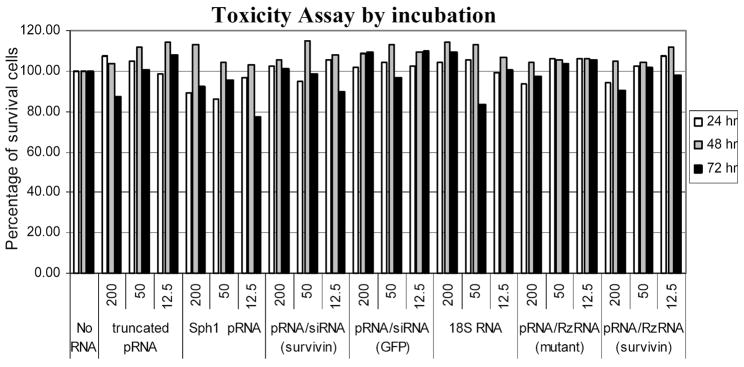
The strategy of RNA dimer-mediated gene delivery is that the receptor-binding moiety mediates cell recognition and subsequent internalization, and the siRNA is then released to downregulate specific genes. Dimers containing both pRNA/siRNA(survivin) and pRNA/aptamer(CD4) were incubated with cells with different levels of CD4 expression. The CD4hi cells responded most strongly to the pRNA dimeric complex, which showed more than 30% reduction in cell viability in the presence of IL-7. Upon removal of IL-7, both CD4low and CDhi D1 cells exhibited severe cell death compared with CD4-negative FL5.12A cells. The level of cell death was correlated with the expression level of CD4. These results suggest that CD4-mediated entry of survivin siRNA led to the suppression of cell viability and that such effects were dependent on the level of CD4 molecules on the cell surface. When incubated simply with cancer cells, pRNA/siRNA(survivin) or pRNA/ribozyme(survivin) did not cause cell death for up to 72 hr in the absence of transfection reagent (Fig. 8). This indicates that the chimeric pRNA did not show nonspecific cytotoxicity on incubation.
FIG. 8.
Toxicity assay of chimeric pRNA targeting survivin. HeLaT4 cells were seeded in 24-well plates so that they will be 30–50% confluent at the time of incubation. Twenty-four hours after seeding, the indicated amount of RNA was added into each well. Cells were further incubated in incubator for 24, 48, and 72 hr and the number of viable cells was counted by hemacytometer. The relative survival rates displayed were obtained by dividing the number of surviving cells in each treatment by the number of surviving cells without treatment with RNA.
Binding assay of chimeric pRNA conjugated with folate
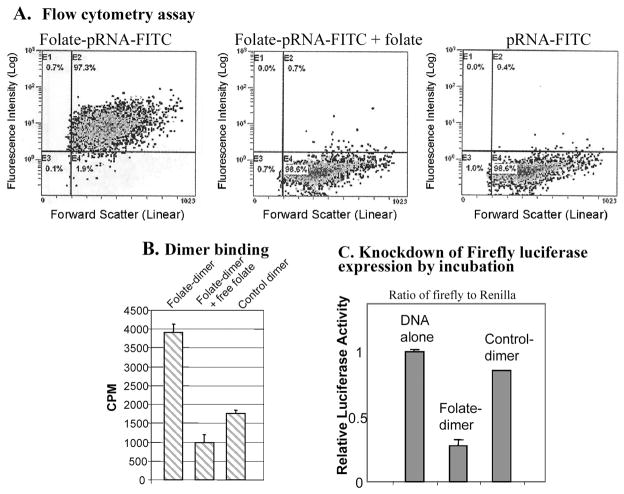
Specific cell binding of pRNA/folate monomer was demonstrated by flow cytometry (Fig. 9A). When human nasopharyngeal epidermal carcinoma KB cells with endogenously overexpressed folate receptors were incubated with FITC-labeled pRNA/folate, 97.3% of the cells were FITC positive. Adding free folate as a blocking reagent to the incubation buffer decreased the FITC-positive cells to only 0.7% (Fig. 9A), suggesting that the binding of pRNA to cells is specific and dependent on folate receptor. FITC-labeled pRNA without folate conjugation did not exhibit binding.
FIG. 9.
Specific delivery of chimeric pRNA/siRNA by folate-pRNA. (A) Flow cytometry analyses of the binding of FITC-labeled folate-pRNA to KB cells. Left: Cells were incubated with folate-pRNA labeled with FITC. Middle: Cells were preincubated with free folate, which served as a blocking agent to compete with folate-pRNA for binding to the receptor. Right: Binding was also tested using folate-free pRNA labeled with FITC as a negative control. The percentages of FITC-positive cells are shown in the top right quadrants. (B) Specific binding of folate-pRNA dimer to KB cells. After incubation of cells with the [3H]folate-pRNA dimer in the presence (middle column) or absence (left column) of free folate, cells were isolated and subjected to scintillation counting. The right-hand column represent 3H-labeled dimer without folate labeling as a negative control. (C) In a knockdown assay by incubation, folate-chimeric dimer complex containing pRNA(B-a′)/folate and pRNA(A-b′)/siRNA(firefly) was incubated with KB cells for 3 hr to allow the binding and entry of RNA. The luciferase level was measured the next day in the dual reporter system. The control dimer was identical to the folate dimer except for its lack of folate labeling.
The possibility of using folate to carry dimeric siRNA chimera was also demonstrated through the use of radiolabeled RNA. First, the RNA dimer was generated by mixing equal amounts of pRNA(A-b′) and 3H-labeled pRNA(B-a′) in the presence of Mg2+. The folate-dimer showed much stronger binding compared with the control dimer without folate labeling (Fig. 9B). When free folate was included as a blocking reagent, the binding of folate-labeled heterodimer RNA to cells diminished.
Functional assay for pRNA/siRNA(firefly or Renilla) after delivery to cells by incubation
After the specific knockdown of chimeric siRNA against luciferase (Fig. 4A) and the specific binding of folate-labeled dimer (Fig. 9A) were demonstrated, the gene-silencing effects by incubation were further investigated. To determine whether folate-mediated targeting of chimeric siRNA to folate receptor-overexpressing cells can lead to the entry of the RNA complex and knockdown of a specific gene, dimers composed of pRNA/siRNA(firefly) and pRNA/folate were incubated with folate receptor-positive KB cells, to allow the binding and entry of RNA mediated by folate. Dual reporter assay revealed a dramatic decrease in firefly luciferase expression after incubation (Fig. 9C). A control RNA without folate labeling, on the other hand, did not significantly interfere with target protein expression. The specificity of pRNA/siRNA(firefly) in dimers was demonstrated when Renilla luciferase was used as an internal control.
Animal trials to demonstrate specific suppression of tumorigenicity of cancer cells by ex vivo delivery of chimeric siRNA against survivin
Animal trials were conducted to test the specificity by ex vivo delivery, using a dimer containing both pRNA(A-b′)/folate and pRNA(B-a′)/siRNA(survivin). The potential of this RNA dimer to suppress tumor formation was tested in athymic nude mice. Nasopharyngeal epidermal carcinoma (KB) cells were incubated with dimeric RNA with or without the blocking reagent, free folate, before being introduced into the nude mice by axillary injection. The mice receiving cells alone developed tumors within 3 weeks, whereas none of the mice receiving cells pretreated with the dimers with pRNA(A-b′)/folate and pRNA(B-a′)/siRNA(survivin) developed tumors (Table 1). The inhibition of tumor formation is specific because the control dimer RNA without folate conjugation used in control mice groups did not affect tumor development.
Table 1.
Animal Trial in Epidermal Cancer Therapya
| Treatment group | No. of cancerous animals/no. of animals tested |
|---|---|
| 1: No pRNA | 4/8 |
| 2: Dimer [folate-pRNA(B-a′) + pRNA/siRNA(survivin)(A-b′)] | 0/7 |
| 3: Dimer [pRNA(B-a′) + pRNA/mutant-siRNA(A-b′)] | 7/8 |
| 4: Dimer [pRNA(B-a′) + pRNA/siRNA(survivin)(A-b′)] | 6/8 |
KB cells were maintained in folate-free medium RPMI 1640.
Cells were preincubated with pRNA complex for 3 hr before being used for animal injection. After rinsing twice with PBS containing 10 mM MgCl2, cells were collected into a centrifuge tube. Each mouse was inoculated with 2.5 × 103 cells in 0.1 ml of medium. Shown are the results of in vivo testing of mice receiving tumor xenografts along with the chimeric pRNA complex. One mouse in group 2 produced a plaque within 1 week, much earlier than any of the other mice in any group, and therefore given these special circumstances it was treated as an outlier and there are seven mice recorded for group 2 instead of eight.
DISCUSSION
φ29 pRNA has a tendency to form dimers, which are the building blocks of hexamers (Fig. 10), as a result of the interaction of interlocking loops of each pRNA. This paper has demonstrated the production of dimers to deliver therapeutic RNA to specific cells. In the future, chimeric hexamers could also be assembled via hand-in-hand interaction. Thus, because there are six chimeric pRNAs in the hexamer, there would be six positions available to carry molecules for cell recognition, therapy, and detection. Besides the receptor-binding aptamers, siRNA, ribozyme, and folate reported here, other materials such as fluorescent dyes, heavy metal, quantum dots, fluorescent beads, or radioisotopes can also be conjugated for the detection of cancer signatures at different stages of development. The reported methods for conjugating folate and FITC could be used for the conjugation of chemical drugs, and endosome-disrupting chemicals could be added to promote the release of siRNA from the endosome after delivery to improve therapeutic efficacy (Fig. 10). Nucleotide derivatives such as 2′-fluoro-2′-deoxy-CTP, 2′-fluoro-2′-deoxy-UTP, or Spiegelmer will be incorporated into the RNA to produce stable in vitro RNA transcripts that are resistant to RNase digestion (Soutschek et al., 2004).
FIG. 10.
The potential use of pRNA hexamers as polyvalent gene delivery vectors. Six copies of pRNA have been found to form a hexameric ring to drive the DNA-packaging motor of bacterial virus φ29. There would therefore be six positions available to carry foreign moieties for targeting, therapy, and detection.
This polyvalent RNA complex can also potentially be used to treat chronic viral infections, such as those caused by HIV and hepatitis B virus, by targeting the specific virus glycoproteins incorporated on the infected cell surface. It is well established in the scientific community that RNAs do not induce a detectable immune response except when complexed with proteins (Madaio et al., 1984; Goldsby et al., 2002). The use of RNA as a delivery vehicle could avoid the problems of immune response and the rejection of protein vectors after repeated long-term drug administration. The use of such a 25 nm RNA complex would provide a longer turnover time in the body than other small molecules would offer. The toxicity or other adverse effects of the pRNA chimera were further mitigated by the conjugation of a small DNA oligo (Manuscript in preparation).
Acknowledgments
This work was supported by NIH grants, ROI GM59944, and DOD prostate cancer grant W81XWH-050158 and DOD breast cancer grant DAMD17-03-1-0589 to P.G. We thank Guangxin Shen and Paul Robison for the flow cytometry assays; Suresh Mittal, David Riese, Phillip Low, and Shou-wei Ding for providing cell lines; we thank Taejin Lee for luciferase assays and Anoop Tiwari and Jeremy Hall for their assistance in the preparation of this manuscript; we thank Annette Khaled, Carol Koons, Richard Roll, Alex Cole, and Faqing Huang for their contributions to this manuscript.
References
- AKKINA R, BANERJEA A, BAI J, ANDERSON J, LI MJ, ROSSI J. siRNAs, ribozymes and RNA decoys in modeling stem cell-based gene therapy for HIV/AIDS. Anticancer Res. 2003;23:1997–2005. [PubMed] [Google Scholar]
- BLOUNT KF, UHLENBECK OC. The hammerhead ribozyme. Biochem Soc Trans. 2002;30:1119–1122. doi: 10.1042/bst0301119. [DOI] [PubMed] [Google Scholar]
- BRUMMELKAMP TR, BERNARDS R, AGAMI R. A system for stable expression of short interfering RNAs in mammalian cells. Science. 2002;296:550–553. doi: 10.1126/science.1068999. [DOI] [PubMed] [Google Scholar]
- CARMICHAEL GG. Medicine: Silencing viruses with RNA. Nature. 2002;418:379–380. doi: 10.1038/418379a. [DOI] [PubMed] [Google Scholar]
- CHEN C, ZHANG C, GUO P. Sequence requirement for hand-in-hand interaction in formation of pRNA dimers and hexamers to gear φ29 DNA translocation motor. RNA. 1999;5:805–818. doi: 10.1017/s1355838299990350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHEN C, SHENG S, SHAO Z, GUO P. A dimer as a building block in assembling RNA: A hexamer that gears bacterial virus φ29 DNA-translocating machinery. J Biol Chem. 2000;275:17510–17516. doi: 10.1074/jbc.M909662199. [DOI] [PubMed] [Google Scholar]
- ELBASHIR SM, HARBORTH J, LENDECKEL W, YALCIN A, WEBER K, TUSCHL T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001;411:494–498. doi: 10.1038/35078107. [DOI] [PubMed] [Google Scholar]
- ELLINGTON AD, SZOSTAK JW. In vitro selection of RNA molecules that bind specific ligands. Nature. 1990;346:818–822. doi: 10.1038/346818a0. [DOI] [PubMed] [Google Scholar]
- GARVER K, GUO P. Boundary of pRNA functional domains and minimum pRNA sequence requirement for specific connector binding and DNA packaging of phage φ29. RNA. 1997;3:1068–1079. [PMC free article] [PubMed] [Google Scholar]
- GOLD L. The SELEX process: a surprising source of therapeutic and diagnositc compounds. Harvey Lect. 1995;91:47–57. [PubMed] [Google Scholar]
- GOLDSBY RA, KINDT TJ, OSBORNE BA, KUBY J. Immunology. W.H. Freeman and Company; New York: 2002. Antigens; pp. 57–61. [Google Scholar]
- GROSSMAN D, KIM PJ, SCHECHNER JS, ALTIERI DC. Inhibition of melanoma tumor growth in vivo by survivin targeting. Proc Natl Acad Sci USA. 2001;98:635–640. doi: 10.1073/pnas.230450097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GUO P, ERICKSON S, ANDERSON D. A small viral RNA is required for in vitro packaging of bacteriophage f29 DNA. Science. 1987;236:690–694. doi: 10.1126/science.3107124. [DOI] [PubMed] [Google Scholar]
- GUO P, ZHANG C, CHEN C, TROTTIER M, GARVER K. Inter-RNA interaction of phage φ29 pRNA to form a hexameric complex for viral DNA transportation. Mol Cell. 1998;2:149–155. doi: 10.1016/s1097-2765(00)80124-0. [DOI] [PubMed] [Google Scholar]
- HANNON GJ. RNA interference. Nature. 2002;418:244–251. doi: 10.1038/418244a. [DOI] [PubMed] [Google Scholar]
- HOEPRICH S, GUO P. Computer modeling of three-dimensional structure of DNA-packaging RNA (pRNA) monomer, dimer, and hexamer of φ29 DNA. J Biol Chem. 2002;277:20794–20803. doi: 10.1074/jbc.M112061200. [DOI] [PubMed] [Google Scholar]
- HOEPRICH S, ZHOU Q, GUO S, QI G, WANG Y, GUO P. Bacterial virus φ29 pRNA as a hammerhead ribozyme escort to destroy hepatitis B virus. Gene Ther. 2003;10:1258–1267. doi: 10.1038/sj.gt.3302002. [DOI] [PubMed] [Google Scholar]
- KHALED AR, DURUM SK. Lymphocide: Cytokines and the control of lymphoid homeostasis. Nat Rev Immunol. 2002;2:817–830. doi: 10.1038/nri931. [DOI] [PubMed] [Google Scholar]
- KHALED AR, REYNOLDS DA, YOUNG HA, THOMPSON CB, MUEGGE K, DURUM SK. Interleukin-3 withdrawal induces an early increase in mitochondrial membrane potential unrelated to the Bcl-2 family: Roles of intracellular pH, ADP transport, and F0F1-ATPase. J Biol Chem. 2001;276:6453–6462. doi: 10.1074/jbc.M006391200. [DOI] [PubMed] [Google Scholar]
- KHALED A, GUO S, LI F, GUO P. Controllable self-assembly of nanoparticles for specific delivery of multiple therapeutic molecules to cancer cells using RNA nanotechnology. Nano letters. 2005 doi: 10.1021/nl051264s. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- KIM DH, LONGO M, HAN Y, LUNDBERG P, CANTIN E, ROSSI JJ. Interferon induction by siRNAs and ssRNAs synthesized by phage polymerase. Nat Biotechnol. 2004;22:321–325. doi: 10.1038/nbt940. [DOI] [PubMed] [Google Scholar]
- KIM K, KHALED AR, REYNOLDS D, YOUNG HA, LEE CK, DURUM SK. Characterization of an interleukin-7-dependent thymic cell line derived from a p53−/− mouse. J Immunol Methods. 2003;274:177–184. doi: 10.1016/s0022-1759(02)00513-6. [DOI] [PubMed] [Google Scholar]
- KRAUS E, JAMES W, BARCLAY AN. Cutting edge: Novel RNA ligands able to bind CD4 antigen and inhibit CD4+ T lymphocyte function. J Immunol. 1998;160:5209–5212. [PubMed] [Google Scholar]
- KUMAR M, CARMICHAEL GG. Antisense RNA: Function and fate of duplex RNA in cells of higher eukaryotes. Microbiol Mol Biol Rev. 1998;62:1415–1434. doi: 10.1128/mmbr.62.4.1415-1434.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KUMAR S, VAUX DL. Apoptosis: A Cinderella caspase takes center stage. Science. 2002;297:1290–1291. doi: 10.1126/science.1076118. [DOI] [PubMed] [Google Scholar]
- LI H, LI WX, DING SW. Induction and suppression of RNA silencing by an animal virus. Science. 2002;296:1319–1321. doi: 10.1126/science.1070948. [DOI] [PubMed] [Google Scholar]
- LI N, YU C, HUANG F. Novel cyanine-AMP conjugates for efficient 5′ RNA fluorescent labeling by one-step transcription and replacement of [gamma-32P]ATP in RNA structural investigation. Nucleic Acids Res. 2005;33:e37. doi: 10.1093/nar/gni036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LU Y, LOW PS. Immunotherapy of folate receptor-expressing tumors: Review of recent advances and future prospects. J Control Release. 2003;91:17–29. doi: 10.1016/s0168-3659(03)00215-3. [DOI] [PubMed] [Google Scholar]
- MADAIO MP, HODDER S, SCHWARTZ RS, STOLLAR BD. Responsiveness of autoimmune and normal mice to nucleic acid antigens. J Immunol. 1984;132:872–876. [PubMed] [Google Scholar]
- MAT-ARIP Y, GARVER K, CHEN C, SHENG S, SHAO Z, GUO P. Three-dimensional interaction of φ29 pRNA dimer probed by chemical modification interference, cryo-AFM, and cross-linking. J Biol Chem. 2001;276:32575–32584. doi: 10.1074/jbc.M100045200. [DOI] [PubMed] [Google Scholar]
- McCAFFREY AP, MEUSE L, PHAM TT, CONKLIN DS, HANNON GJ, KAY MA. RNA interference in adult mice. Nature. 2002;418:38–39. doi: 10.1038/418038a. [DOI] [PubMed] [Google Scholar]
- NOVINA CD, MURRAY MF, DYKXHOORN DM, BERESFORD PJ, RIESS J, LEE SK, COLLMAN RG, LIEBERMAN J, SHANKAR P, SHARP PA. siRNA-directed inhibition of HIV-1 infection. Nat Med. 2002;8:681–686. doi: 10.1038/nm725. [DOI] [PubMed] [Google Scholar]
- SHU D, HUANG L, HOEPRICH S, GUO P. Construction of φ29 DNA-packaging RNA (pRNA) monomers, dimers and trimers with variable sizes and shapes as potential parts for nanodevices. J Nanosci Nanotech. 2003;3:295–302. doi: 10.1166/jnn.2003.160. [DOI] [PubMed] [Google Scholar]
- PENNATI M, BINDA M, COLELLA G, FOLINI M, CITTI L, VILLA R, DAIDONE MG, ZAFFARONI N. Radiosensitization of human melanoma cells by ribozyme-mediated inhibition of survivin expression. J Invest Dermatol. 2003;120:648–654. doi: 10.1046/j.1523-1747.2003.12082.x. [DOI] [PubMed] [Google Scholar]
- SHU D, MOLL D, DENG Z, MAO C, GUO P. Bottom-up assembly of RNA arrays and superstructures as potential parts in nanotechnology. Nano Lett. 2004;4:1717–1724. doi: 10.1021/nl0494497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SOUTSCHEK J, AKINC A, BRAMLAGE B, CHARISSE K, CONSTIEN R, DONOGHUE M, ELBASHIR S, GEICK A, HADWIGER P, HARBORTH J, JOHN M, KESAVAN V, LAVINE G, PANDEY RK, RACIE T, RAJEEV KG, ROHL I, TOUDJARSKA I, WANG G, WUSCHKO S, BUMCROT D, KOTELIANSKY V, LIMMER S, MANOHARAN M, VORNLOCHER HP. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature. 2004;432:173–178. doi: 10.1038/nature03121. [DOI] [PubMed] [Google Scholar]
- SUDIMACK J, LEE RJ. Targeted drug delivery via the folate receptor. Adv Drug Deliv Rev. 2000;41:147–162. doi: 10.1016/s0169-409x(99)00062-9. [DOI] [PubMed] [Google Scholar]
- TROTTIER M, GUO P. Approaches to determine stoichiometry of viral assembly components. J Virol. 1997;71:487–494. doi: 10.1128/jvi.71.1.487-494.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ZHANG CL, LEE CS, GUO P. The proximate 5′ and 3′ ends of the 120-base viral RNA (pRNA) are crucial for the packaging of bacteriophage f29 DNA. Virology. 1994;201:77–85. doi: 10.1006/viro.1994.1267. [DOI] [PubMed] [Google Scholar]
- ZHANG CL, TELLINGHUISEN T, GUO P. Confirmation of the helical structure of the 5′/3′ termini of the essential DNA packaging pRNA of phage f29. RNA. 1995;1:1041–1050. [PMC free article] [PubMed] [Google Scholar]
- ZHANG F, LEMIEUX S, WU X, ST-ARNAUD S, McMURRAY CT, MAJOR F, ANDERSON D. Function of hexameric RNA in packaging of bacteriophage φ29 DNA in vitro. Mol Cell. 1998;2:141–147. doi: 10.1016/s1097-2765(00)80123-9. [DOI] [PubMed] [Google Scholar]