Abstract
Increased cyclic GMP from enhanced synthesis or suppressed catabolism (e.g. PDE5 inhibition by sildenafil, SIL) activates protein kinase G (PKG) and blunts cardiac pathological hypertrophy. Suppressed calcineurin (Cn)-NFAT (nuclear factor of activated T-cells) signaling appears to be involved, though it remains unclear how this is achieved. One potential mechanism involves activation of Cn/NFAT by calcium entering via transient receptor potential canonical (TRPC) channels (notably TRPC6). Here, we tested the hypothesis that PKG blocks Cn/NFAT activation by modifying and thus inhibiting TRPC6 current to break the positive feedback loop involving NFAT and NFAT-dependent TRPC6 upregulation. TRPC6 expression rose with pressure-overload in vivo, and angiotensin (ATII) or endothelin (ET1) stimulation in neonatal and adult cardiomyocytes in vitro. 8Br-cGMP and SIL reduced ET1-stimulated TRPC6 expression and NFAT dephosphorylation (activity). TRPC6 upregulation was absent if its promoter was mutated with non-functional NFAT binding sites, whereas constitutively active NFAT triggered TRPC6 expression that was not inhibited by SIL. PKG phosphorylated TRPC6, and both T70 and S322 were targeted. Both sites were functionally relevant, as 8Br-cGMP strongly suppressed current in wild-type TRPC6 channels, but not in those with phospho-silencing mutations (T70A, S322A or S322Q). NFAT activation and increased protein synthesis stimulated by ATII or ET1 was blocked by 8Br-cGMP or SIL. However, transfection with T70A or S322Q TRPC6 mutants blocked this inhibitory effect, whereas phospho-mimetic mutants (T70E, S322E, and both combined) suppressed NFAT activation. Thus PDE5-inhibition blocks TRPC6 channel activation and associated Cn/NFAT activation signaling by PKG-dependent channel phosphorylation.
Keywords: hypertrophy, calcineurin, TRPC6, receptor-operated cation channel, sildenafil expression, protein kinase G, myocyte
Introduction
In response to sustained loading stress, the heart undergoes a complex array of cellular and molecular changes that often lead to maladaptive remodeling and heart failure [1]. The calcium/calmodulin activated phosphatase calcineurin (Cn) prominently contributes to this pathophysiology [2–4] in large part by activating the pro-hypertrophic transcription factor NFAT (nuclear factor of activated T-cells) [5]. Cn-mediated dephosphorylation of NFAT results in its nuclear translocation promoting hypertrophic growth of the heart [5]. Cn/NFAT can be negatively modulated [6–8], and among these regulators, cyclic guanylate monophosphate-protein kinase G (cGMP/PKG) signaling is intriguing given the availability of clinically effective pharmaceuticals that stimulate this pathway. Both enhancing cGMP synthesis or blocking hydrolysis by phosphodiesterase type-5 (PDE5) suppresses pathologic hypertrophy in cell culture and intact hearts [9–13]. Fiedler et al. first reported that Cn-NFAT is suppressed by cGMP/PKG stimulation in neonatal cardiomyocytes [14] and subsequent studies have found similar effects with the PDE5 inhibitor sildenafil in vitro and in vivo [10,11]. The precise mechanism remains unknown, though the finding that cGMP/PKG stimulation cannot inhibit NFAT in myocytes overexpressing Cn suggests a proximal target [11,14].
One potential mechanism involves transient receptor potential canonical (TRPC) channels. Calcium conductance via these non-voltage gated channels may contribute to various cardiovascular diseases including hypertension and hypertrophy, and recent studies have directly linked their activity to Cn/NFAT stimulation [15–18]. TRPC3 and TRPC6 are the two receptor operated channels expressed in heart, both being activated by diacylglycerol (DAG) coupled to G q-receptor signaling [19]. TRPC6 has garnered attention as it is ubiquitously expressed in vascular smooth muscle and cardiac myocytes [20,21] and expression increases in human heart failure [18,22]. TRPC6 up-regulation stimulates cardiac hypertrophy via Cn/NFAT signaling [18], and the TRPC6 promoter itself contains an NFAT-responsive sequence resulting in a positive feedback loop that further augments channel expression and activity.
TRPC channels pose an intriguing mechanism to explain PKG suppression of Cn/NFAT signaling by PKG as recent studies in non-cardiac cells have found both TRPC3 and TRPC6 can be negatively modulated by PKG phosphorylation at one or more residues [23,24]. The role of such signaling in the heart is unknown. Accordingly, we tested the hypothesis that cGMP/PKG stimulation inactivates TRPC6 channel activity and expression via channel phosphorylation, thereby inhibiting NFAT activity and NFAT-dependent TRPC6 gene upregulation. Our results show phosphorylation at either T70 or S322 and particularly their combination suppresses TRPC6 channel current, G q agonist-induced NFAT activation, and myocyte hypertrophic responses.
Material and Methods
Plasmids
pcDNA3-human TRPC6-YFP and pMALc2E-N-terminus TRPC6 (1-407aa) were provided by Dr. Craig Montell [25]. pcDNA3-mouse angiotensin II type 1 receptor (AT1R) was provided by Dr. Akiyoshi Fukamizu [26]. Constitutive active NFATc4 plasmid (NFATc4Δ317), and a TPRC6 promoter plasmid (−913mutNFAT1+2-luc) lacking functional NFAT binding sites in the 5′-TRPC6 promoter region, were provided by Dr. Eric N. Olson and Dr. Koichiko Kuwahara [18]. pGL4.30-NFAT-RE firefly luciferase vector (NFAT-luc), pGL4.75-CMV Renilla luciferase vector (CMV-Rluc) and pGL4.74-TK (tymidine kinase) Renilla luciferase (TK-Rluc) vector were purchased from Promega. To replace amino acids at PKG-phosphorylation candidate sites, we used a PCR-based site mutagenesis kit (QuikChange, Stratagene) following manufacturer’s protocol using pcDNA3-human TRPC6 YFP or pMALc2E-N-terminus TRPC6 as a template. The primers used to make mutations are provided in supplemental methods.
Pharmaceuticals
Sidenafil (SIL) was administered in vivo at 200 mg/kg/min. This SIL dose has been previously shown to yield free plasma levels in the 30–50nM range in mice, well within the range selective for PDE5 and far below that for PDE1, PDE2 or PDE3 [27]. For the in vitro studies, we employed 1 mM SIL, a dose previously shown to selectively inhibit PDE5 in isolated myocytes based on co-administration in cells with genetically silenced PDE5. Other inhibitors and their respective doses are described where applicable or in supplemental methods.
Isolated cardiac myocytes
Primary cultures of neonatal rat cardiac ventricular (NRVM) myocytes were prepared as previously described [11,26]. Adult mouse ventricular myocytes (AMVM) were isolated from C57Bl/6 mice and prepared using the method of O’Connell with minor modifications [13,28], (See supplemental methods)
Transfection and Luciferase assay in NRVM
Transfection of plasmid DNA was performed with Lipofectamine 2000 (Invitrogen) per manufacturer’s protocol (with Plus Reagent and OptiMEM (Invitrogen) to enhance transfection efficiency to 30–50%). After 4–24-h stimulation, cells were harvested with cell lysis buffer (Promega), and luciferase activity measured by GloMax 96 Microplate Luminometer (Promega) using Dual-Luciferase assay kit (Promega). Adenovirus (AdV) infection was performed in cells placed in serum-free medium using vectors and protocols previously reported [13]. Virus encoded gene silencing PDE5A RNA coupled to a CMV promotor and GFP tag. Control virus contained the GFP tag.
Real-time RT-PCR
RNA was extracted with TRIzol (Invitrogen) per manufacturer’s protocol. Complementary DNAs (cDNAs) were synthesized using the TaqMan Reverse Transcription Reagent (Applied Biosystems). Real-time quantitative PCR was performed using a 7900HT Sequence Detection System (Applied Biosystems). Sample duplicates were subjected to real-time PCR. TaqMan primer and probes for rat/mouse Gapdh, Trpc1, Trpc3 and Trpc6 were from Applied Biosystems. Expression data are calculated from the cycle threshold (Ct) value using the ΔΔCt method for quantification. Gene expression was normalized to that for Gapdh.
Protein Immunoblot
Protein was extracted from cultured myocytes and whole heart extract using lysis buffer (Cell Signaling) (see supplemental methods) Bands were detected by chemiluminescence, and band intensity expressed in relative units. GAPDH was used as a loading control, and data normalized to GAPDH density.
NFATc4 phosphorylation
In vivo and in vitro NFATc4 phosphorylation was determined as reported [29] with minor modifications. Briefly, 200ug protein lysate was immunoprecipitated overnight with rabbit anti-NFATc4 antibody (Santa Cruz) and protein A/G-agarose (Santa Cruz). Immunoprecipitates were then probed with mouse monoclonal anti-phosphoserine antibody (Sigma). After stripping, the same membrane was re-probed with anti-NFATc4 antibody, and results shown as the ratio of phosphorylated to total protein.
Transverse aortic occlusion (TAC) and chronic PDE5a inhibitor treatment
Male C57BL/6 mice (8–11 weeks, Jackson Laboratories) were subjected to trans-aortic constriction using a 27G needle or to sham procedure as previously described (11) (see also supplemental methods). SIL treatment was provided orally (SIL, 200 mg/kg/day) in solid food (soft diet, Bioserv) as described. The Johns Hopkins Medical Institutions Animal Care and Use Committee approved the protocol.
Patch-clamp studies in HEK Cells
Patch-clamp studies were performed in HEK293T cells transfected with or without AT1R cDNA(26) and pcDNA3-human TRPC6 YFP (1 μg for both), using Lipofectamine2000 (Invitrogen) [30]. Cells were treated with or without 200nM ATII, and ± 8Br-cGMP (0.1–1mM (both Sigma). Borosilicate glass capillary pipettes (World Precision Instr.) were prepared with ≈ 3 MΩ resistance (when filled with internal solution) and typical series resistance compensation of > 90%. Transfected cells were first identified by YFP fluorescence. Currents were recorded in a whole-cell configuration using an Axopatch 200A amplifier (Axon Instruments), digitized and stored to a computer using custom software [31]. Currents were elicited by voltage step-pulse or ramp protocol from −100mV to +100mV (500ms duration) applied every 2s from holding potential of −60mV. Data were filtered at 1 kHz and sampled at 5 kHz. The bath solution contained (in mM) 140 NaCl, 5 CsCl2, 2 CaCl2, 1 MgCl2, 10 HEPES and 10 glucose (pH 7.4 with NaOH). The pipette solution contained (in mM) 5 NaCl, 40 CsCl2, 80 Cs-glutamate, 5 Mg-ATP, 5 EGTA, 1.5 CaCl2 (free [Ca2+]i=100 nM) and 10 HEPES (pH 7.2 with CsOH) [25]. Data were obtained at room temperature.
In vitro TRPC6 Phosphorylation Assay
MBP-TRPC6 fusion protein was purified by amylase resin (New England Biolab Inc.) according to manufacturer’s instructions. In addition to wild type TRPC6 (NT-TRPC6WT), NT-TRPC6T70A and NT-TRPC6S322Q were generated. Purified synthetic proteins were assayed in a reaction mixture containing 30mM Tris-HCL (pH7.4), 10 μM cGMP (Sigma), 2mM magnesium acetate, 1mM [γ-32P]ATP (500 to 800 cpm/pmol) (Perkin-Elmer), 0.3mg/mL of bovine serum albumin, 3mM 2-mercaptoethanol and 0.1–1 μg/mL active bovine PKG-1α (Calbiochem) [32]. After 10 min incubation at 30°C, reaction mixtures were subjected to SDS-PAGE (NuPAGE, Invitrogen), transferred to nitrocellulose, and phosphorylation visualized by autoradiography.
Statistics
All values are expressed as mean ± SEM. Comparisons between groups were performed using 1- or 2-way analysis of variance, with a Tukey test for multiple comparisons when appropriate.
Results
Sildenafil blocks cardiac TRPC6 expression induced by pressure-overload
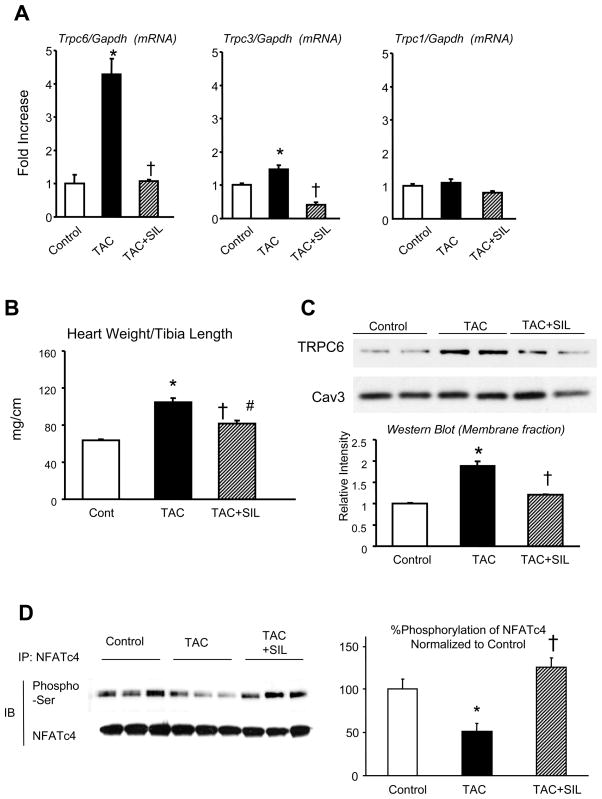
Myocardial gene expression of Trpc1, 3, and 6 was assessed by real-time RT-PCR at baseline and after pressure-overload induced by trans-aortic constriction (TAC, 7 days). Trpc6 expression rose >4 fold and Trpc3 increased 50% (both p<0.05), whereas Trpc1 was unaltered (Fig. 1A). SIL suppressed the rise in both Trpc6 and Trpc3 expression (open bars), accompanied by an anti-hypertrophic effect (Fig. 1B) consistent with prior reports [11]. Given the most prominent response was in Trpc6, we focused on this species next testing if it corresponded to increased protein expression. Protein levels in membrane fractions doubled with TAC, and this was also suppressed by SIL to near control levels (Fig. 1C). TAC resulted in NFATc4 de-phosphorylation consistent with Cn activation, and this was suppressed by SIL (Fig. 1D) supporting upstream targeting of the transcription factor by SIL. Similar suppression of FNATc4 de-phosphorylation by SIL was documented in neonatal rat ventricular myocytes (NRVM) exposed to ET1 (Supplemental figure).
Figure 1.
Influence of pressure-overload and PDE5 inhibition (sildenafil, SIL) on TRPC6 expression. A) Tprc6, 3, and 1, mRNA normalized to Gapdh were assessed in mice subjected to 7-days of transverse aortic constriction (TAC). Results are shown normalized to normal controls (n=4 for each group, * p<0.01 versus control and TAC1w; † p<0.001 vs TAC1w; ‡ p<0.05 vs TAC1w). B) SIL effect on cardiac hypertrophy (heart weight/tibia length) after TAC; n=4–5/group; * p<0.01 versus control; †-p<0.01 versus TAC; # p=0.028 versus control). C) Western blot of TRPC6 in membrane fraction from whole heart tissue isolate. Caveolin3 (Cav3) was used as a protein loading control; (n=4/group; *-p<0.05 versus control; †-p<0.01 versus 1-wk TAC hearts) D) SIL blunts NFAT dephosphorylation induced by TAC. NFATc4 was immune-precipitated and isolated then proved with a phospho-Ser specific antibody. Summary data for ratio of phosphorylated/total NFATc4 is shown at the right (n=4/group; *-p<0.05 versus control; †-p<0.01 versus 1-wk TAC hearts).
PDE5A inhibition and cGMP/PKG block TRPC6 expression in cardiomyocytes
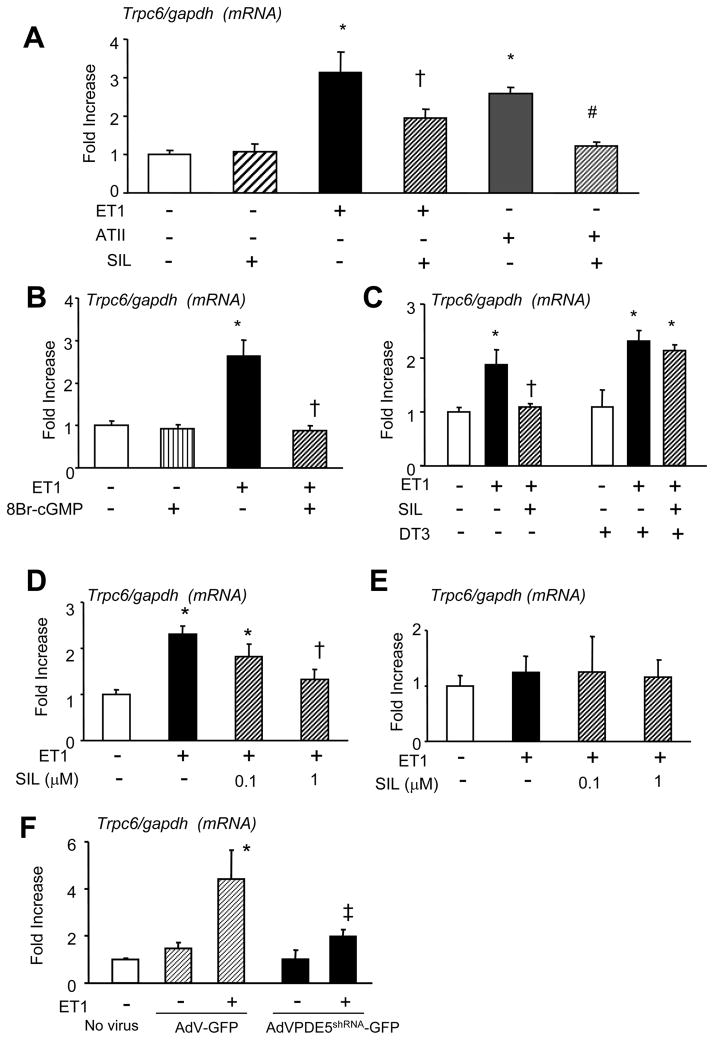
To dissect mechanisms for SIL effects on TRPC6 expression, we conducted studies in cultured neonatal and adult cardiomyocytes. ET1 or ATII stimulation enhanced Trpc6 gene expression in NRVM, and this was blocked by SIL (Fig. 2A) or 8-Br-cGMP (Fig. 2B). Neither SIL nor 8Br-cGMP altered basal Trpc6 expression by themselves. SIL was ineffective in cells with PKG co-inhibited using DT-3 (0.2 μM) [33], supporting the role of PKG activation to the response (Fig. 2C). Importantly, SIL also suppressed ET1-induced Trpc6 expression in adult cardiomyocytes (Fig. 2D). We also examined Trpc3 expression and found it unchanged by ET1 with or without SIL in these cells (Fig. 2E). To further confirm test the specific role of PDE5 inhibition, cells were transfected with a silencing RNA (AdV-PDE5shRNA-GFP; with AdV-GFP as the control) that markedly reduces PDE5 expression [13]. AdV-PDE5shRNA inhibited ET1-induced Trpc6 (Fig. 2F) in AMVM similar to the response with SIL.
Figure 2.
Effect of SIL or 8Br-cGMP on Trpc6 mRNA expression in neonatal rat (A–C) and adult mouse (D and E) cultured cardiomyocytes. A) Trpc6 mRNA expression normalized to Gapdh rose with 0.1 μM endothelin-1(ET1) or 1 μM angiotensin II (ATII), and both changes were suppressed by co-incubation with 1 μM SIL for 24hrs. (n=5–12/group; * p<0.01 vs vehicle (Veh), †p<0.05 vs ET1; ‡p<0.01 vs ATII). B) ET1 stimulated Trpc6 mRNA expression is suppressed by co-treatment of 8Br-GMP (1 mM, n=5–6/group, *p<0.002 vs other groups) for 24hrs. C) SIL-mediated suppression of ET1 activated Trpc6 expression is blocked by inhibiting PKG activity with DT3 (n=4/group; * p<0.05 vs Veh; †p<0.05 vs ET1). D) SIL (0.1, 1 μM) suppresses ET-1 triggered Trpc6 gene expression in adult mouse cardiac myocytes. (24 hr incubation study, n=9/group, * p<0.01 versus vehicle control, † p<0.05 versus ET1 stimulation. E) In contrast, ET-1 did not stimulate Trpc3 expression in these cells. F) PDE5 gene-silencing blocks ET1-stimulated Trpc6 gene expression. Cells were transfected with (AdVPDE5shRNA GFP) or control (AdV-GFP) [13] were stimulated by 0.1 μM ET1 for 24hrs (n=3/group, * p<0.001 for ET1 effect, ‡ p<0.05 for effect from PDE5 gene silencing on ET1 response, 2-way ANOVA).
SIL blocks TRPC6 expression by NFAT dependent mechanisms
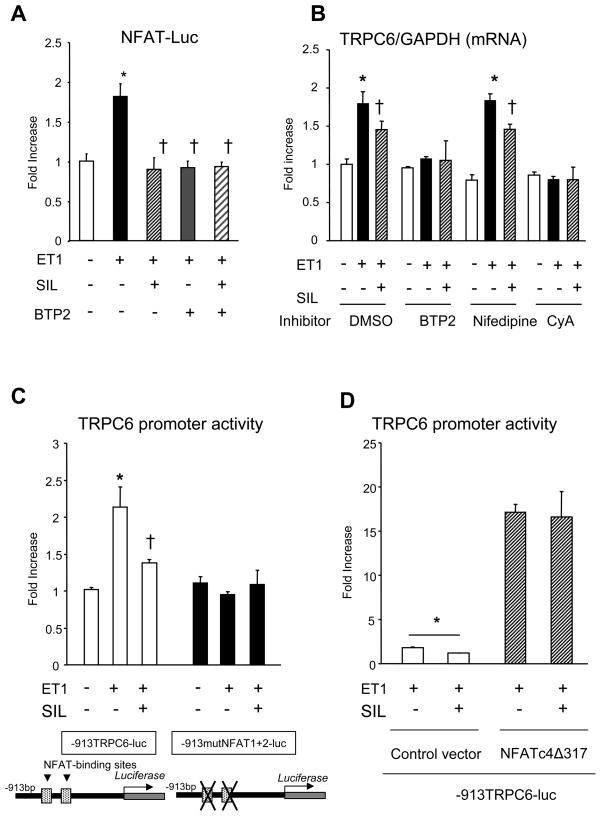
We next tested the influence of SIL on coupling between TRPC6 activation and Cn/NFAT. SIL fully inhibited ET1 stimulated NFAT (NFAT-luciferase reporter) a response identical to that with a non-selective TRPC channel blocker 3,5-bistrifluoromethyl pyrazole inhibitor 2 (BTP2) [22]. There was no additive effect by their combination. BTP2 as well as the Cn inhibitor cyclosporine A both fully blocked the ability of ET1 to stimulate Trpc6 gene expression, and SIL had no further impact in their presence (Fig 3B). In contrast, blocking the L-type Ca2+ channel (LTCC) with nifedipine neither diminished ET1-stimulated TRPC6 expression nor altered the capacity of SIL to suppress expression, indicating independence from L-type channels.
Figure 3.
Effect of SIL on cardiomyocyte NFAT activation and TRPC6 expression. A) Activation of NFAT (luciferase reporter assay, NFAT-Luc) by ET1 (0.1 μM) in NRVM is prevented by SIL (1 μM) or by the TRP-channel blocker BTP2 (1 μM), with no additive effects from their combination (4-hr incubation, n=3/group, *P<0.01 versus other groups). B) ET-1 stimulated Trpc6 expression is prevented by the Cn inhibitor cyclosporin A (1 μM) and TRP channel antagonist BTP2, but not LTCC blocker nifedipine (1 μM). SIL still suppress expression in the presence of nifedipine; studies performed in NRVM; DMSO, dimetyl sulfoxide (n=3/group, *P<0.01 versus vehicle, †p<0.01 versus 24hr ET1 stimulation.) C) TRPC6 promoter activity in constructs with (−913TRPC6-luc) or without (−913 mutNFAT1+2TRPC6-luc) NFAT binding sites, generated as described(18). N=6/group, *P<0.01 versus vehicle, †p<0.01 versus ET1 stimulation. 4hour stimulation D) SIL suppression of TRPC6 promoter activity with or without constitutive active form of NFATc4: NFATc4Δ317. *P<0.05. 4hr stimulation.
To test whether SIL-mediated suppression of Trpc6 gene expression was NFAT-dependent, NRVM were transfected with a Trpc6-luciferase promoter construct that had or lacked functional NFAT binding domains (−913 TRPC6-luc, −913mutNFAT1+2-luc) [18]. ET1 stimulated TRPC6 transcription and SIL-suppression of the response was absent if the promoter lacked functional NFAT binding sites (Fig. 3C). In contrast, cells transfected with a constitutively active NFATc4 (NFATc4Δ317) displayed increased TRPC6 promoter activity that was not inhibited by SIL.
TRPC6 S322 and T70 are phosphorylated by PKG
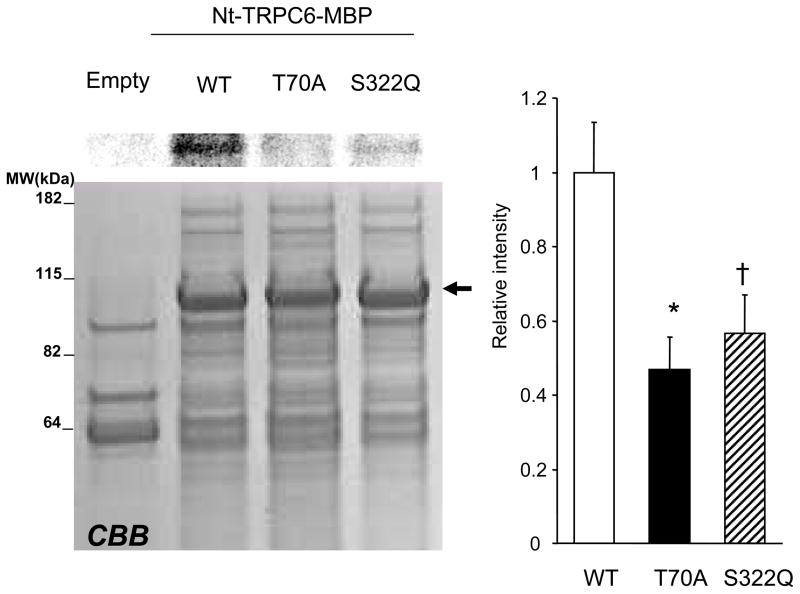
TRPC3 has been reported to be phosphorylated by PKG at T11 and S263 [23], sites analogous to T70 and S322 in human TRPC6 (T69 and S321 in rat or mouse). Recent data using an immortalized vascular cell line confirmed changes at T70 in TRPC6 [24]. Given the different cell types, we examined both sites by performing in vitro radiolabeled phosphorylation assays. When recombinant N-terminus TRPC6 synthetic peptide (NT-TRPC6) was used as the target, PKG-mediated phosphorylation was readily observed (Fig 4). However, expression of either T70A or S322Q phospho-silenced mutant forms of recombinant TRPC6 resulted in reduced phosphorylation, supporting targeting by PKG at both sites.
Figure 4.
PKG phosphorylation of N-terminus human TRPC6 protein. Left panel shows representative in vitro PKG phosphorylation of TRPC6 fused with maltose binding protein (Nt-TRPC6-MBP) with wild type (WT) or phospho-silenced mutants. CBB: Coomassie Brilliant Blue staining of the synthetic peptides was used for the phosphorylation assay, MW: molecular weight. ; top panel shows autoradiogram data, and right panel summary of radiogram for each condition. (n=4/group, *P<0.01 versus WT, † p<0.05 versus WT).
S322 and T70 Sites Modulate cGMP/PKG-suppression of TRPC6 channel current
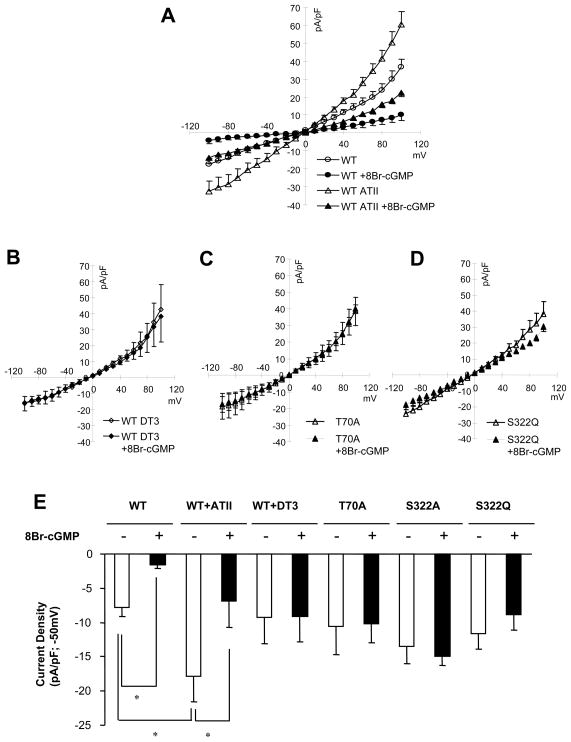
To test cGMP-PKG regulation of TRPC6 channel conductance and functionality of both phosphorylation sites, patch clamp studies were performed in HEK293T cells. These cells express basal PKG-1α but not TRPC6, and so werefirst transfected with human TRPC6 (wild type:WT). Under rest conditions, a typical TRPC6 current-voltage dependence was observed, and this could be markedly suppressed by exposure to 1mM 8Br-cGMP (Fig. 5A). Rest current was enhanced in cells co-transfected with the angiotensin receptor (AT1R) and subsequently exposed to ATII, consistent with receptor activated TRPC6 stimulation. 8Br-cGMP also markedly attenuated this current. 8Br-cGMP had no impact if PKG was co-inhibited with DT3, supporting the importance of PKG activation (Fig. 5B).
Figure 5.
cGMP-mediated TRPC6 current inhibition in HEK293T cells expressing wild-type (WT) or mutant TRPC6. A) Averaged current-voltage (I–V) relationship in cells expressing wild-type TRPC6 channels in absence (open circle; N=6) or presence (closed circle; N=5) of 8Br-cGMP (1mM) and in ATII (200nM) stimulated cells with (open triangle; N=5) or without (closed triangle; N=5) 8Br-cGMP. ATII type I receptor (AT1R) was co-transfected into HEK cells for ATII experiments. In both conditions, cGMP depressed TRPC6 current. B) Inhibition of PKG with DT3 (0.2 μM) prevents suppression of TRPC6 current by cGMP. C,D) I-V relationships in HEK cells expressing T70A or S322Q mutant TRPC6 channels (phospho-silenced) with and without co-treatment by 8Br-cGMP. (n=5–6/group). In both instances, the inhibition of channel current by cGMP was absent. E) Summary data for the preceding studies (also for S322A mutant) displaying averaged density of TRPC6 current measured at −50mV from a holding potential −60mV. * p<0.05 versus comparison as depicted.
We next tested whether transfection with human TRPC6 containing phospho-silencing mutations (S322A, S322Q, or T70A) also prevented cGMP-suppression of TRPC6 current. These proteins had a slightly increased resting current relative to WT though these differences were not statistically significant. Notably however, each of these mutated forms displayed no modulation by 8Br-cGMP, indicating both sites could regulate PKG modulation of TRPC6 (Fig 5C–E).
S322 and T70 Sites Modulate cGMP/PKG-suppression of ATII-stimulated NFAT
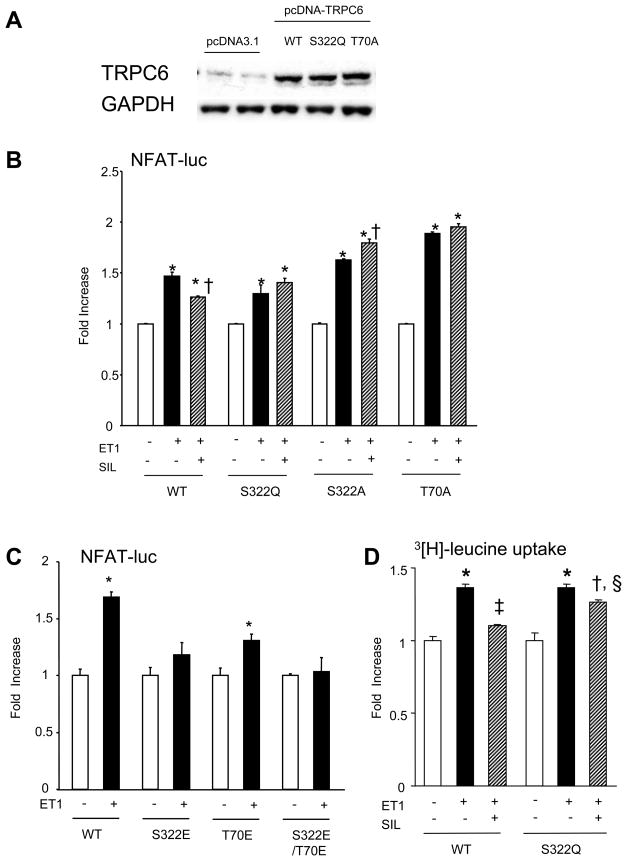
We next tested the impact of genetically targeting TRPC6 with phospho-mimetic or silencing mutations on the activation of NFAT. NRVMs were transfected with a NFAT activity reporter and either wild-type or mutant TRPC6. Fig 6A shows example immunoblots confirming robust and similar protein expression with WT and mutated TRPC6 plasmids versus negligible protein with transfection of the control (pcDNA3.1). In cells transfected with TRPC6-WT, SIL suppressed ET1-induced NFAT activity, whereas SIL was ineffective in S322A, S322Q, or T70A transfected cells (Fig. 6B). The opposite was observed in myocytes transfected with phospho-mimetic mutants (S322E, T70E) in which NFAT activation by ET1 was suppressed. NFAT activation was fully blocked with the double mutant. The impact of mutant TRPC6 on myocyte growth was further tested by radiolabeled leucine incorporation. SIL suppressed ET1 stimulated protein synthesis in TRPC6-WT transfected cells, but did so less effectively in cells expressing the S322Q mutant (p<0.02 for interaction effect, 2-way ANOVA, Fig. 6D).
Figure 6.
Effect of TRPC6 phospho-mutants on the efficacy of SIL to block NFAT activation by ET1. A) Example Western blot showing robust expression of various TRPC6 forms after transient transfection of TRPC6 plasmids in NRVM. B) NFAT luciferase assay in NRVM transfected with WT of phospho-silenced TRPC6 mutants (S322Q, S322A, or T70A). SIL suppression of NFAT activation by ET1 was absent with each mutant. (n=3/group for both, * p<0.01 versus vehicle, †-p<0.01 vs ET1). C) In contrast, expression of phospho-mimetic mutants of either S322E or T70E reduced ET1-induced NFAT activation, and the double mutant (S322E/T70E) fully prevented activation. (n=6/group for both, * p<0.01 versus vehicle). D) NRVM expressing WT-TRPC6 undergo enhanced protein synthesis (leucine incorporation) with ET1 that is blunted by SIL. In contrast, cells expressing the S322Q-TRPC6 have less impact from SIL. (n=5–6/group, *P<0.01 versus vehicle, † p<0.01 vs vehicle, p<0.01 vs ET1; ‡p<0.005 vs vehicle, p<0.01 vs ET1; § p<0.02 for interaction of TRPC6 mutation and ET1 effect (2-way ANOVA). 4hr stimulation.
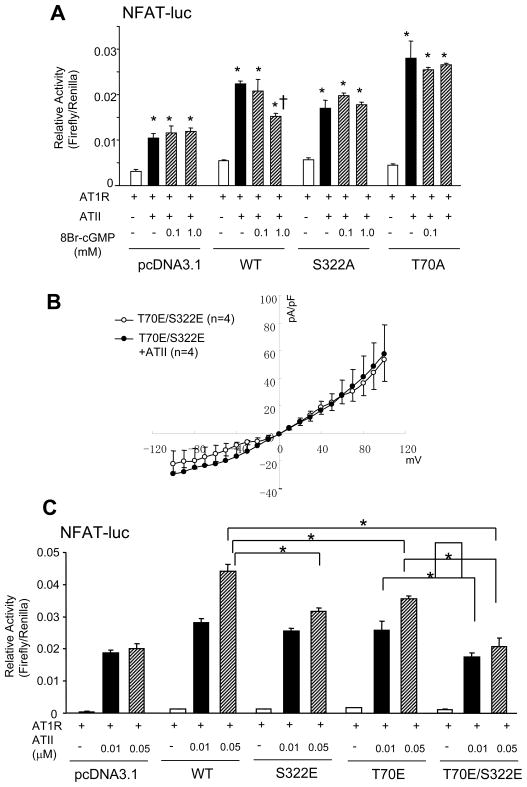
Additional studies were performed in HEK293T cells co-transfected with TRPC6 (WT and mutants) and AT1R. cGMP itself did not inhibit basal NFAT activity in HEK cells (data not shown). Interestingly, without WT-TRPC6 transfection, HEK cells do not exhibit an 8Br-cGMP-dependent decline in ATII-mediated NFAT activation, but this is observed once cells were co-transfected (Fig. 7A), highlighting the specificity of TRPC6-PKG targeting for the response. Expression of phospho-silenced mutants prevented cGMP-inhibition (Fig. 7A), as in NRVM. Functional effects in the double phospho-mimetic mutant were examined by patch-clamp, and in contrast to WT, the T70E/S322E mutant exhibited no receptor-stimulated (ATII) response (Fig. 7B). Furthermore, NFAT activated by ATII was significantly attenuated in cells expressing the S322E or T70E mutation, and the double mutant reduced activation to the level observed in cells transfected with control (empty) vector. This indicates that pseudo-phosphorylation of both sites yields a functional null channel.
Figure 7.
Effect of TRPC mutants on NFAT activation in HEK293T cells. A) NFAT activity in cells transfected with WT or phospho-silenced TRPC6 mutants. Cells were co-transfected with ATII type 1a receptor (AT1R) and stimulated with 0.01 μM ATII in the presence or absence of 8Br-cGMP (0.1–1mM, 4–6 hr incubation). Without TRPC6 co-transfection, 8Br-cGMP did not suppress NFAT activation induced by ATII. This suppression was also absent in cells expressing S322A or T70A mutants. B) Averaged current-voltage (I-V) relationship in cells expressing a phospho-mimetic mutant of TRPC6 channel (S322E/T70E double mutant) and AT1-R in the absence (open circle; N=4) or presence (closed circle; N=4) of ATII (200nM). C) Effect of TRPC6 phospho-mimetic mutations on NFAT activation from AII in HEK cells co-transfected with AT1-R. Co-transfection with WT-TRPC6 resulted in an augmented NFAT activation and ATII dose response. This was blunted with S322E and T70E mutants, and completely suppressed (to empty vector control levels) with the double mutant. N=6 of each group († p<0.01 versus WT-0.05 μM ATII stimulation; # P<0.05 versus S322E or T70E with 0.05 μM ATII; ¶ P<0.05 versus WT-0.01 μM ATII stimulation). 4hr stimulation.
Discussion
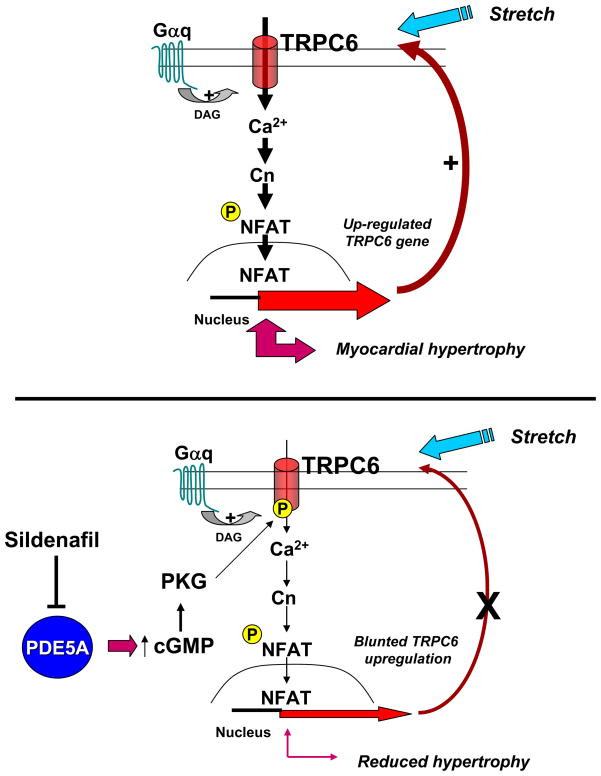
Both pharmacological and genetic suppression of PDE5 can suppress pathological cardiac hypertrophic growth and Cn-NFAT activation in cardiac myocytes and the intact heart. The present study reveals a novel mechanism underlying this modulation that involves the inhibition of TRPC6 channel activation by PKG-dependent channel inactivation. The proposed scheme is depicted in Figure 8. Upon activation by either a Gq-coupled agonist or mechanical stretch, TRPC6 channels are stimulated resulting in entry of a strategic Ca2+ pool. This activates Ca2+-calmodulin dependent calcineurin which in turn de-phosphorylates NFAT sending it to the nucleus where it activates a hypertrophic gene program. The TRPC6 promotor contains NFAT response elements and so is itself upregulated, providing a positive feedback loop to further stimulate TRPC6 current and related signaling (Fig 8, upper panel). In the presence of activated PKG (Fig 8, lower panel), which we achieved by inhibiting PDE5 or with exogenous cGMP, TRPC6 current is inactivated. We demonstrated that this depends upon targeted phosphorylation of either of two residues, T70 or S322, that can have additive effects. As depicted in the lower panel, phosphorylation suppresses channel current, reducing consequent NFAT activation that in turn diminishes TRPC6 upregulation (positive feedback). Our data shows that the suppressive influence of PKG activation via PDE5 inhibition is targeted upstream of NFAT dephosphoryation, and that a dual phospho-mimetic mutant channel functions is an effective null, displaying negligible current response or consequent NFAT activation from Gq agonists.
Figure 8.
Schematic summarizing the signaling involving TRPC6-Cn-NFAT in the absence (top) and presence (bottom) of activated PKG (due to enhanced cGMP generation or blocked catabolism). See text for details.
Our results are consistent with recent studies highlighting a central role of TRPC channel currents, in particular TRPC6 and TRPC3, in modulating Cn/NFAT activation. We concentrated on TRPC6 given its greater up-regulation with pressure-overload, but admittedly other channels such as TRPC3 or TRPC1 which have both been linked to hypertrophic responses could contribute as well. BTP2 is a non-specific inhibitor of TRPC channels [22], so its suppression of TRPC6 activation does not necessarily rule out regulation by other members of the TRPC family. Intriguingly, PKG was first shown to phosphorylate and suppress TRPC3 activity in a heterologous cell system, though whether this occurs and its impact in myocytes remains to be determined. Furthermore, recent data suggests TRPC1 and TRPC3 can form hetermers that are NO (cGMP) inhibited, so PKG might impact TRPC1 as well. Thus, these other targets may have been co-suppressed by SIL. Further work is needed to define the role of TRPC3 (or TRPC1/3) PKG-modulation in the heart.
While our data strongly support a primary role of TRPC6-mediated signaling to Cn/NFAT activation, they do not rule out other potential sources of calcium entry. For example, Fiedler et al found that Cn-NFAT activation was suppressed by PKG and also by inhibiting the LTCC with verapamil [14]. As PKG can blunt LTCC current in part by phosphorylation of specific sites on the α1c and β2 subunit [34], this would seem a logical and potential contributor to PKG modulation. Moreover, there is potentially interaction between this modification and calcineurin activity, as Cn increased LTCC current by de-phosphorylating Ser1928 (α1-subunit) [35] one of the PKG targets. In contrast, we found nifedipine at a dose sufficient to block LTCC in NRVM [36] had no impact on ET1-mediated TRPC6 gene or the capacity of SIL to suppress it. This suggests that the TRPC6 positive-feedback loop is largely independent of LTCC activity. We have also shown that SIL blunts β-adrenergic stimulation (which activates the LTCC), yet does so without altering the whole cell Ca2+. This suggests that SIL-mediated PKG activation may not target the channel. The relative impact of LTCC versus other Ca2+ sources could also vary with the specific stimuli, e.g. ET1 versus phenylephrine, that activate different receptors and may differently modulate the LTCC [37,38]. Furthermore, LTCC transcription is tightly autoregulated [39] unlike TRPC6 that has a positive feedback loop enhancing its role under pathophysiologic conditions.
In their study of A7r5 vascular cells, Takahashi et al. [24] found that PKG modified TRPC6 only at T69 (analogous to T70 in the human gene), though functional consequences of S321 (S322 in human) change was not performed. Yet, prior work with TRPC3, which is highly homologous to TRPC6, found PKG phosphoryation at both analogous sites to those in the current study [23]. Importantly, we we show a functional role for both sites, with similar influences on channel conductance and coupling to NFAT activation, and use several relevant cell systems with both gain and loss of function mutagenesis. Importantly, the pseudo-phosphorylation mutant combining both residues (T70E/S322E) had greater impact than both alone, and fully blocked TRPC6 activation effects. Differences in cell type, functional assays, and/or substrate for the phosphorylation assay (e.g. synthetic peptide versus immuno-precipitation product), may underlie the discrepancy. We do not yet know if PKG is the sole modulator at these sites, or if this phosphorylation impacts other known PTMs of the channel [40].
Our study has several limitations. One is that we present lack definitive evidence that the post-translational changes identified in TRPC6 from PKG activation are occurring in vivo. Phospho-protein antibodies are under development, thought this may prove somewhat difficult as even antibodies to total protein remain less than ideal. We are presently developing mass spectroscopy methods to identify phosphorylation at these specific residues and survey for other potential sites, and these would potentially be applicable to cardiac tissue with further optimization. We did not perform loss-of expression studies to confirm a role of endogenous TRPC6 in cardiomyocyte NFAT activation as this has already been reported [18,41]. Rather, we viewed the fact that TRPC6 substantially increases with cardiac stress as justifying studies using enhanced expression that also facilitates analysis of phosphorylation deficient and mimetic forms. Mice with knock-in mutations of TRPC6 are planned to assess their impact in vivo.
The finding that PKG can directly suppress TRPC6 channel activity may have implications for cardiac disease therapy, but also in other conditions where TRPC6 hyperfunction has been observed. For example, mutations that increase TRPC6 function or its expression have been recently described in human focal glomerulosclerosis [42] and pulmonary hypertension [43], and TRPC channel upregulation may also play a role in muscular dystrophy [44]. Sildenafil is currently used to treat pulmonary hypertension, and recent studies have suggested potential benefits in dystrophic disorders [45]. It is intriguing to speculate that TRPC targeting by PKG may play a role. Type-specific TRPC channel antagonists are being developed and along with organ targeted inducible gene deletion models, they should provide novel insights regarding the pathophysiologic role of these channel. Our results show that enhancing cGMP/PKG activity, in particular by PDE5 inhibition, can also suppress TRPC6 activity may prove relevant to the use of such inhibitors for a variety of human diseases.
Supplementary Material
Acknowledgments
We thank Craig Montell for his helpful advice and providing the several TRPC6 plasmids. We thank Koichiro Kuwahara for his helpful advice and providing the several TRPC6 promoter plasmid. This study was supported by National Institutes of Health Grants: HL-89297, HL-59408, HL-84946 (DAK), HL-077180 (DAK, GFT, TA), HL-07227 (DAK,MZ) and American Heart Association Mid-Atlantic Fellowship Grant (MZ, NK), and a grant from the Japan Heart Foundation (NK).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Mudd JO, Kass DA. Tackling heart failure in the twenty-first century. Nature. 2008;451:919–928. doi: 10.1038/nature06798. [DOI] [PubMed] [Google Scholar]
- 2.Molkentin JD, Lu JR, Antos CL, Markham B, Richardson J, Robbins J, Grant SR, Olson EN. A calcineurin-dependent transcriptional pathway for cardiac hypertrophy. Cell. 1998;93:215–228. doi: 10.1016/s0092-8674(00)81573-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bueno OF, Wilkins BJ, Tymitz KM, Glascock BJ, Kimball TF, Lorenz JN, Molkentin JD. Impaired cardiac hypertrophic response in Calcineurin Abeta -deficient mice. Proc Natl Acad Sci U S A. 2002;99:4586–4591. doi: 10.1073/pnas.072647999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hsu S, Nagayama T, Koitabashi N, Zhang M, Zhou L, Bedja D, Gabrielson KL, Molkentin JD, Kass DA, Takimoto E. Phosphodiesterase 5 inhibition blocks pressure overload-induced cardiac hypertrophy independent of the calcineurin pathway. Cardiovasc Res. 2009;81:301–309. doi: 10.1093/cvr/cvn324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hogan PG, Chen L, Nardone J, Rao A. Transcriptional regulation by calcium, calcineurin, and NFAT. Genes Dev. 2003;17:2205–2232. doi: 10.1101/gad.1102703. [DOI] [PubMed] [Google Scholar]
- 6.Ni YG, Berenji K, Wang N, Oh M, Sachan N, Dey A, Cheng J, Lu G, Morris DJ, Castrillon DH, Gerard RD, Rothermel BA, Hill JA. Foxo transcription factors blunt cardiac hypertrophy by inhibiting calcineurin signaling. Circulation. 2006;114:1159–1168. doi: 10.1161/CIRCULATIONAHA.106.637124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hill JA, Rothermel B, Yoo KD, Cabuay B, Demetroulis E, Weiss RM, Kutschke W, Bassel-Duby R, Williams RS. Targeted inhibition of calcineurin in pressure-overload cardiac hypertrophy. Preservation of systolic function. J Biol Chem. 2002;277:10251–10255. doi: 10.1074/jbc.M110722200. [DOI] [PubMed] [Google Scholar]
- 8.Li HH, Kedar V, Zhang C, McDonough H, Arya R, Wang DZ, Patterson C. Atrogin-1/muscle atrophy F-box inhibits calcineurin-dependent cardiac hypertrophy by participating in an SCF ubiquitin ligase complex. J Clin Invest. 2004;114:1058–1071. doi: 10.1172/JCI22220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Calderone A, Thaik CM, Takahashi N, Chang DL, Colucci WS. Nitric oxide, atrial natriuretic peptide, and cyclic GMP inhibit the growth-promoting effects of norepinephrine in cardiac myocytes and fibroblasts. J Clin Invest. 1998;101:812–818. doi: 10.1172/JCI119883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nagayama T, Hsu S, Zhang M, Koitabashi N, Bedja D, Gabrielson KL, Takimoto E, Kass DA. Sildenafil stops progressive chamber, cellular, and molecular remodeling and improves calcium handling and function in hearts with pre-existing advanced hypertrophy caused by pressure overload. J Am Coll Cardiol. 2009;53:207–215. doi: 10.1016/j.jacc.2008.08.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Takimoto E, Champion HC, Li M, Belardi D, Ren S, Rodriguez ER, Bedja D, Gabrielson KL, Wang Y, Kass DA. Chronic inhibition of cyclic GMP phosphodiesterase 5A prevents and reverses cardiac hypertrophy. Nat Med. 2005;11:214–222. doi: 10.1038/nm1175. [DOI] [PubMed] [Google Scholar]
- 12.Tsai EJ, Kass DA. Cyclic GMP signaling in cardiovascular pathophysiology and therapeutics. Pharmacol Ther. 2009;122:216–238. doi: 10.1016/j.pharmthera.2009.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang M, Koitabashi N, Nagayama T, Rambaran R, Feng N, Takimoto E, Koenke T, O’Rourke B, Champion HC, Crow MT, Kass DA. Expression, activity, and pro-hypertrophic effects of PDE5A in cardiac myocytes. Cell Signal. 2008;20:2231–2236. doi: 10.1016/j.cellsig.2008.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fiedler B, Lohmann SM, Smolenski A, Linnemuller S, Pieske B, Schroder F, Molkentin JD, Drexler H, Wollert KC. Inhibition of calcineurin-NFAT hypertrophy signaling by cGMP-dependent protein kinase type I in cardiac myocytes. Proc Natl Acad Sci U S A. 2002;99:11363–11368. doi: 10.1073/pnas.162100799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Inoue R, Jensen LJ, Shi J, Morita H, Nishida M, Honda A, Ito Y. Transient receptor potential channels in cardiovascular function and disease. Circ Res. 2006;99:119–131. doi: 10.1161/01.RES.0000233356.10630.8a. [DOI] [PubMed] [Google Scholar]
- 16.Nilius B, Owsianik G, Voets T, Peters JA. Transient receptor potential cation channels in disease. Physiol Rev. 2007;87:165–217. doi: 10.1152/physrev.00021.2006. [DOI] [PubMed] [Google Scholar]
- 17.Nakayama H, Wilkin BJ, Bodi I, Molkentin JD. Calcineurin-dependent cardiomyopathy is activated by TRPC in the adult mouse heart. FASEB J. 2006;20:1660–1670. doi: 10.1096/fj.05-5560com. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kuwahara K, Wang Y, McAnally J, Richardson JA, Bassel-Duby R, Hill JA, Olson EN. TRPC6 fulfills a calcineurin signaling circuit during pathologic cardiac remodeling. J Clin Invest. 2006;116:3114–3126. doi: 10.1172/JCI27702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hofmann T, Obukhov AG, Schaefer M, Harteneck C, Gudermann T, Schultz G. Direct activation of human TRPC6 and TRPC3 channels by diacylglycerol. Nature. 1999;397:259–263. doi: 10.1038/16711. [DOI] [PubMed] [Google Scholar]
- 20.Inoue R, Okada T, Onoue H, Hara Y, Shimizu S, Naitoh S, Ito Y, Mori Y. The transient receptor potential protein homologue TRP6 is the essential component of vascular alpha(1)-adrenoceptor-activated Ca(2+)-permeable cation channel. Circ Res. 2001;88:325–332. doi: 10.1161/01.res.88.3.325. [DOI] [PubMed] [Google Scholar]
- 21.Nishida M, Hara Y, Yoshida T, Inoue R, Mori Y. TRP channels: molecular diversity and physiological function. Microcirculation. 2006;13:535–550. doi: 10.1080/10739680600885111. [DOI] [PubMed] [Google Scholar]
- 22.Bush EW, Hood DB, Papst PJ, Chapo JA, Minobe W, Bristow MR, Olson EN, McKinsey TA. Canonical transient receptor potential channels promote cardiomyocyte hypertrophy through activation of calcineurin signaling. J Biol Chem. 2006;281:33487–33496. doi: 10.1074/jbc.M605536200. [DOI] [PubMed] [Google Scholar]
- 23.Kwan HY, Huang Y, Yao X. Regulation of canonical transient receptor potential isoform 3 (TRPC3) channel by protein kinase G. Proc Natl Acad Sci U S A. 2004;101:2625–2630. doi: 10.1073/pnas.0304471101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Takahashi S, Lin H, Geshi N, Mori Y, Kawarabayashi Y, Takami N, Mori MX, Honda A, Inoue R. Nitric oxide-cGMP-protein kinase G pathway negatively regulates vascular transient receptor potential channel TRPC6. J Physiol. 2008;586:4209–4223. doi: 10.1113/jphysiol.2008.156083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kwon Y, Hofmann T, Montell C. Integration of phosphoinositide- and calmodulin-mediated regulation of TRPC6. Mol Cell. 2007;25:491–503. doi: 10.1016/j.molcel.2007.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Koitabashi N, Arai M, Tomaru K, Takizawa T, Watanabe A, Niwano K, Yokoyama T, Wuytack F, Periasamy M, Nagai R, Kurabayashi M. Carvedilol effectively blocks oxidative stress-mediated downregulation of sarcoplasmic reticulum Ca2+-ATPase 2 gene transcription through modification of Sp1 binding. Biochem Biophys Res Commun. 2005;328:116–124. doi: 10.1016/j.bbrc.2004.12.139. [DOI] [PubMed] [Google Scholar]
- 27.Kass DA, Champion HC, Beavo JA. Phosphodiesterase type 5: expanding roles in cardiovascular regulation. Circ Res. 2007;101:1084–1095. doi: 10.1161/CIRCRESAHA.107.162511. [DOI] [PubMed] [Google Scholar]
- 28.O’Connell TD, Rodrigo MC, Simpson PC. Isolation and culture of adult mouse cardiac myocytes. Methods Mol Biol. 2007;357:271–296. doi: 10.1385/1-59745-214-9:271. [DOI] [PubMed] [Google Scholar]
- 29.Bubikat A, De Windt LJ, Zetsche B, Fabritz L, Sickler H, Eckardt D, Godecke A, Baba HA, Kuhn M. Local atrial natriuretic peptide signaling prevents hypertensive cardiac hypertrophy in endothelial nitric-oxide synthase-deficient mice. J Biol Chem. 2005;280:21594–21599. doi: 10.1074/jbc.M501103200. [DOI] [PubMed] [Google Scholar]
- 30.Ishida J, Asada S, Daitoku H, Fujiwara K, Kon Y, Sugaya T, Murakami K, Nakajima T, Kasuya Y, Fukamizu A. Expression and characterization of mouse angiotensin II type 1a receptor tagging hemagglutinin epitope in cultured cells. Int J Mol Med. 1999;3:263–270. doi: 10.3892/ijmm.3.3.263. [DOI] [PubMed] [Google Scholar]
- 31.Aiba T, Hesketh GG, Barth AS, Liu T, Daya S, Chakir K, Dimaano VL, Abraham TP, O’Rourke B, Akar FG, Kass DA, Tomaselli GF. Electrophysiological consequences of dyssynchronous heart failure and its restoration by resynchronization therapy. Circulation. 2009;119:1220–1230. doi: 10.1161/CIRCULATIONAHA.108.794834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Glass DB, Smith SB. Phosphorylation by cyclic GMP-dependent protein kinase of a synthetic peptide corresponding to the autophosphorylation site in the enzyme. J Biol Chem. 1983;258:14797–14803. [PubMed] [Google Scholar]
- 33.Dostmann WR, Taylor MS, Nickl CK, Brayden JE, Frank R, Tegge WJ. Highly specific, membrane-permeant peptide blockers of cGMP-dependent protein kinase Ialpha inhibit NO-induced cerebral dilation. Proc Natl Acad Sci U S A. 2000;97:14772–14777. doi: 10.1073/pnas.97.26.14772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yang L, Liu G, Zakharov SI, Bellinger AM, Mongillo M, Marx SO. Protein kinase G phosphorylates Cav1.2 alpha1c and beta2 subunits. Circ Res. 2007;101:465–474. doi: 10.1161/CIRCRESAHA.107.156976. [DOI] [PubMed] [Google Scholar]
- 35.Tandan S, Wang Y, Wang TT, Jiang N, Hall DD, Hell JW, Luo X, Rothermel BA, Hill JA. Physical and functional interaction between calcineurin and the cardiac L-type Ca2+ channel. Circ Res. 2009;105:51–60. doi: 10.1161/CIRCRESAHA.109.199828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pignier C, Potreau D. Characterization of nifedipine-resistant calcium current in neonatal rat ventricular cardiomyocytes. Am J Physiol Heart Circ Physiol. 2000;279:H2259–H2268. doi: 10.1152/ajpheart.2000.279.5.H2259. [DOI] [PubMed] [Google Scholar]
- 37.Kilts JD, Lin SS, Lowe JE, Kwatra MM. Selective activation of human atrial Galpha12 and Galpha13 by Galphaq-coupled angiotensin and endothelin receptors. J Cardiovasc Pharmacol. 2007;50:299–303. doi: 10.1097/FJC.0b013e3180a72632. [DOI] [PubMed] [Google Scholar]
- 38.Uchi J, Sasaki H, Morimoto S, Kusakari Y, Shinji H, Obata T, Hongo K, Komukai K, Kurihara S. Interaction of alpha1-adrenoceptor subtypes with different G proteins induces opposite effects on cardiac L-type Ca2+ channel. Circ Res. 2008;102:1378–1388. doi: 10.1161/CIRCRESAHA.107.167734. [DOI] [PubMed] [Google Scholar]
- 39.Schroder E, Byse M, Satin J. L-type calcium channel C terminus autoregulates transcription. Circ Res. 2009;104:1373–1381. doi: 10.1161/CIRCRESAHA.108.191387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sharif-Naeini R, Folgering JH, Bichet D, Duprat F, Delmas P, Patel A, Honore E. Sensing pressure in the cardiovascular system: Gq-coupled mechanoreceptors and TRP channels. J Mol Cell Cardiol. 2009 doi: 10.1016/j.yjmcc.2009.03.020. [DOI] [PubMed] [Google Scholar]
- 41.Onohara N, Nishida M, Inoue R, Kobayashi H, Sumimoto H, Sato Y, Mori Y, Nagao T, Kurose H. TRPC3 and TRPC6 are essential for angiotensin II-induced cardiac hypertrophy. EMBO J. 2006;25:5305–5316. doi: 10.1038/sj.emboj.7601417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Winn MP, Conlon PJ, Lynn KL, Farrington MK, Creazzo T, Hawkins AF, Daskalakis N, Kwan SY, Ebersviller S, Burchette JL, Pericak-Vance MA, Howell DN, Vance JM, Rosenberg PB. A mutation in the TRPC6 cation channel causes familial focal segmental glomerulosclerosis. Science. 2005;308:1801–1804. doi: 10.1126/science.1106215. [DOI] [PubMed] [Google Scholar]
- 43.Yu Y, Keller SH, Remillard CV, Safrina O, Nicholson A, Zhang SL, Jiang W, Vangala N, Landsberg JW, Wang JY, Thistlethwaite PA, Channick RN, Robbins IM, Loyd JE, Ghofrani HA, Grimminger F, Schermuly RT, Cahalan MD, Rubin LJ, Yuan JX. A Functional Single-Nucleotide Polymorphism in the TRPC6 Gene Promoter Associated With Idiopathic Pulmonary Arterial Hypertension. Circulation. 2009 doi: 10.1161/CIRCULATIONAHA.108.782458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kruger J, Kunert-Keil C, Bisping F, Brinkmeier H. Transient receptor potential cation channels in normal and dystrophic mdx muscle. Neuromuscul Disord. 2008;18:501–513. doi: 10.1016/j.nmd.2008.04.003. [DOI] [PubMed] [Google Scholar]
- 45.Khairallah M, Khairallah RJ, Young ME, Allen BG, Gillis MA, Danialou G, Deschepper CF, Petrof BJ, Des RC. Sildenafil and cardiomyocyte-specific cGMP signaling prevent cardiomyopathic changes associated with dystrophin deficiency. Proc Natl Acad Sci U S A. 2008;105:7028–7033. doi: 10.1073/pnas.0710595105. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.