Abstract
The mechanisms of transscleral iontophoresis have been investigated previously with small molecules in rabbit sclera. The objective of the present study was to examine transscleral iontophoretic transport of charged macromolecules across excised human sclera. Passive and 2-mA iontophoretic transport experiments were conducted in side-by-side diffusion cells with human sclera. The effects of iontophoresis upon transscleral transport of model permeants bovine serum albumin (BSA) and polystyrene sulfonic acid (PSS) as well as a model drug bevacizumab (BEV) were determined. Passive and iontophoretic transport experiments of tetraethylammonium (TEA) and salicylic acid (SA) and passive transport experiments of the macromolecules served as the controls. The results of iontophoresis enhanced transport of TEA and SA across human sclera were consistent with those in a previous rabbit sclera study. For the iontophoretic transport of macromolecules BSA and BEV, higher iontophoretic fluxes were observed in anodal iontophoresis as compared to passive and cathodal iontophoresis. This suggests the importance of electroosmosis. For the polyelectrolyte PSS, higher iontophoretic flux was observed in cathodal iontophoresis compared to anodal iontophoresis. Both electroosmosis and electrophoresis affected iontophoretic fluxes of the macromolecules; the relative contributions of electroosmosis and electrophoresis were a function of molecular size and charge of the macromolecules.
Keywords: Electrophoresis, Electroosmosis, Charged macromolecules, Human sclera
1. Introduction
The treatment of diseases such as age-related macular degeneration, diabetic retinopathy, and uveitis has been a challenge to ophthalmologists (Urtti, 2006) . The rapid progress of drug discovery has led to the development of biologicals for the treatment of these eye diseases (Hofman et al., 2000; Ranta and Urtti, 2006). However, most of these new bioactive agents have high molecular weights and are usually administered via intravitreal injections. Multiple injections are common in these treatments and increase the potential of adverse effects such as retinal detachment, vitreous hemorrhage, and endophthalmitis (Jager et al., 2004). A safe and effective method is required to deliver these bioactives to the posterior segment of the eye.
Transscleral iontophoresis has shown a viable modality to deliver therapeutic drugs for the treatment of eye diseases (Myles et al., 2005). Transscleral iontophoresis is a noninvasive technique in which a low electric current is used to drive a drug into and across a tissue (Eljarrat-Binstock and Domb, 2006; Eljarrat-Binstock et al., 2008b). The flux enhancing mechanisms of iontophoretic drug delivery include electrophoresis, electroosmosis, and electro-permeabilization (Srinivasan et al., 1989; Sims et al., 1991; Kasting, 1992; Edwards and Langer, 1994; Eljarrat-Binstock and Domb, 2006). There are a number of ocular iontophoresis studies focusing on the delivery of small therapeutic agents into the eye and on its safety (Behar-Cohen et al., 1996; Behar-Cohen et al., 2002). It has been demonstrated that macromolecules with multiple charges and molecular weight (MW) between 4 to 150 kDa could be transported into the sclera by an electric field (Davies et al., 2003). Because both electroosmosis and electrophoresis could play a significant role in iontophoretic flux enhancement of these macromolecules, the optimization of transscleral iontophoretic delivery of macromolecules requires the understanding of these two mechanisms and their relative contributions in transscleral iontophoresis.
Studies of iontophoresis for ocular delivery of both charged and neutral molecules using rabbit as an animal model have been reported (Eljarrat-Binstock and Domb, 2006; Frucht-Pery et al., 2006; Eljarrat-Binstock et al., 2007; Eljarrat-Binstock et al., 2008a). The mechanisms of transscleral iontophoretic transport of small molecules have also been investigated using rabbit sclera (Li et al., 2005). However, human sclera data for studying the transport behavior of charged macromolecules are limited in the literature. A systematic examination of transscleral iontophoretic transport behavior of macromolecules with human sclera is required to optimize transscleral iontophoretic drug delivery.
Bevacizumab (Avastin®), a humanized recombinant monoclonal antibody, is the first anti-angiogenic protein approved by FDA. As an anti-human vascular endothelial growth factor (VEGF) agent, intravitreal injection of bevacizumab has been shown to be beneficial in off-label treatment of age-related macular degeneration, choroidal neovascularization, and diabetic retinopathy (Mordenti et al., 1999; Michels et al., 2005). Due to the potential adverse effects of intravitreal injections, transscleral iontophoresis could be an alternative method and enhance the delivery of bevacizumab via the transscleral route.
The purpose of the present study was to study the transport of charged macromolecules of 60 to 150 kDa in transscleral iontophoresis using human cadaveric sclera in vitro. In the present study, the following questions will be addressed: (1) how effective is iontophoresis in the delivery of these charged macromolecules across human sclera in vitro and (2) what are the mechanisms controlling the iontophoretic transport of the macromolecules across human sclera in vitro? Answering these questions could help pharmaceutical researchers better predict transscleral iontophoretic transport and develop an effective iontophoresis system for the delivery of macromolecules to the eye.
2. Materials and methods
2.1. Materials
Phosphate buffered saline (PBS, pH 7.4, consisting of 0.01 M phosphate buffer, 0.0027 M potassium chloride, 0.137 M sodium chloride) was prepared by dissolving PBS tablets (Sigma-Aldrich, St. Louis, MO) in distilled, deionized water. 14C-tetraethylammonium (TEA, MW 130 Da) bromide (1–5 mCi/mmol) and 14C-salicylic acid (SA, 40–60 mCi/mmol, MW 138 Da) were purchased from PerkinElmer Life and Analytical Sciences (Boston, MA). All radiolabelled chemicals had purity of at least 97%. Fluorescein isothiocyanate (FITC) labelled dextrans (DEX) of average MW 4 and 20 kDa (degree of substitution 0.004–0.005 mol FITC/mol of glucose) and bovine serum albumin (BSA, purity > 98%, MW 68 kDa) were purchased from Sigma-Aldrich (St. Louis, MO). Poly (styrene sulfonic acid) sodium salt (PSS, MW 67 kDa, Mw/Mn < 1.2) was purchased from Polysciences, Inc. (Warrington, PA). Bevacizumab (BEV, Avastin®, 100 mg in 4 mL, MW 149 kDa) was from Genentech, Inc. (Oceanside, CA). Agarose (low EEO, molecular biological grade) was purchased from Fisher Scientific (Fair Lawn, NJ). 0.15 M TEA solution was prepared by reacting tetraethylammonium hydroxide (20% w/w, Acros Organics, Morris Plains, NJ) with hydrochloric acid, and 0.15 M SA solution was prepared by dissolving sodium salicylate (Acros Organics, Morris Plains, NJ) in distilled, deionized water. The TEA and SA solutions were adjusted to pH 7.4 with hydrochloric acid or sodium hydroxide.
2.2. Preparation of the sclera
Cadaver eyes were obtained from Dayton Eye Bank (Dayton, OH) and Moran Eye Center at the University of Utah (Salt Lake City, UT). The tissues were stored in moisture chamber at 2–4°C not more than six months. Before the experiments the tissue was thawed at room temperature and adhering tissues on the sclera including the retina and choroid were removed with a pair of forceps. The sclera was then rinsed with PBS, cut into small pieces, and equilibrated in PBS at room temperature for 30 min. The use of human tissues was approved by the Institutional Review Board at the University of Cincinnati, Cincinnati, OH.
2.3. Passive and iontophoresis transport experiments
Passive transport, anodal iontophoresis, and cathodal iontophoresis were conducted for all the permeants used in the present study. Experiments were first conducted with low MW permeants TEA and SA. These experiments allowed the comparison of the present results using human sclera with those in previous studies using rabbit sclera (Li et al., 2005). Experiments of macromolecules were then studied using BSA, PSS, and BEV. BSA has a low charge to MW ratio similar to those of therapeutic peptides/proteins, and PSS has a high charge to MW ratio similar to those of oligonucleotides and small interfering RNAs (siRNA). BEV is a model drug that has clinical significance.
In the experiments, the sclera was sandwiched between the two half-cells of a side-by-side diffusion cell (with an effective diffusion area of approximately 0.2 cm2) with the choroid side facing the receptor chamber. The diffusion cell was placed in a circulating water bath at 36 ± 1°C. The volume of the donor and receptor solutions was 1.5 mL. PBS was used as the receptor solution for all transport experiments except for BEV, in which 0.05% (w/v) BSA in PBS was used. In the transport studies of TEA and SA, trace amounts of radiolabelled TEA or SA (2–10 µCi/mL) were added into the donor solution of PBS. In the transport studies of BSA and PSS, approximately 1.7 mg/mL of BSA in PBS, and 15 mg/mL PSS in PBS were the donor solutions, respectively. In the transport study of BEV, the donor solution was prepared by diluting Avastin® (25 mg/mL) with PBS containing 0.05% (w/v) BSA to a final concentration of 2.5 mg/mL. In all iontophoresis experiments, a 2-mA current was applied across the sclera with a constant current iontophoretic device (Phoresor II Auto, Model PM 850, Iomed, Inc., Salt Lake City, UT) using Ag/AgCl (cathode) and Ag (anode) as driving electrodes. The electrodes were immersed in the donor and receptor solutions except in the BEV study. In the BEV study, the donor electrode was placed in an electrode chamber containing PBS, which was connected to the donor chamber containing the donor solution through a salt bridge (5% w/v agarose in PBS). The salt bridge was stored in the donor solution before use. This salt bridge setup would prevent the interactions between BEV and the electrode during iontophoresis and the precipitation of BEV in the donor chamber. In all iontophoretic transport experiments, the electrodes were replaced before the AgCl on the Ag/AgCl electrode was depleted to avoid any changes in the pH of the donor and receptor solutions during iontophoresis. The compositions of the donor and receptor solutions could be affected by ion transport and the formation of electrochemical products at the electrodes. For example, significant depletion of the chloride ion in the donor chamber during anodal iontophoresis could lead to AgCl precipitation in the donor solution. In the anodal iontophoresis experiments, no AgCl precipitation was observed in the donor chamber, indicating the presence of enough chloride ions in the system for the electrochemical reaction at the anode electrode. Ten microliters of the donor solution and 1 mL of the receptor solution were taken at predetermined time intervals for assay. Fresh receptor solution of 1 mL was then added into the receptor to maintain a constant volume in the receptor. The durations of the passive and iontophoretic transport experiments ranged from 2 to 96 h. The long duration experiments were performed to study the mechanisms of iontophoresis under steady-state conditions and therefore the duration was dependent on the permeants used.
In an additional study, the same tissues were used in the passive and iontophoresis experiments with small molecules (TEA and SA) without dissembling the diffusion cells. In this study, a second passive transport experiment after iontophoresis was conducted with the same tissue so the results of these passive experiments before and after iontophoresis could be compared to examine possible irreversible electro-permeabilization effects of iontophoresis upon cadaver human sclera. Another study was also performed with small molecules (TEA and SA) using 0.15 M TEA and 0.15 M SA in deionized water (instead of PBS) as the donor in the TEA and SA experiments, respectively. These studies provided the same experimental conditions as in a previous rabbit sclera study (Li et al., 2005) for direct comparison of the results in the present human and previous rabbit studies. Passive transport experiments of macromolecules DEX of molecular weights 4 and 20 kDa with human sclera were also performed. The duration of the experiments was 24–48 h, and the donor was 2 mg/mL DEX in PBS. The results in these experiments were used, along with the TEA, SA, BSA, PSS, and BEV data, to determine the effective pore size of the transport pathways in the sclera.
2.4. Assay methods
Radiolabelled TEA and SA samples were analyzed by a liquid scintillation counter (Beckman coulter LS 6500, Fullerton, CA). The samples were mixed with 10 mL of liquid scintillation cocktail (Ultima Gold™, PerkinElmer Life and Analytical Sciences, Shelton, CT) and assayed by the liquid scintillation counter. Samples of DEX 4 kDa were analyzed using a spectrophotometer (UV-mini 1240, Shimadzu, Kyoto, Japan) at 493 nm. DEX 20 kDa, BSA, and PSS samples were analyzed using high performance liquid chromatography (HPLC). The HPLC system (Prominence, Shimadzu, Columbia, MD) consisted of CBM-20A system controller, LC-20AT solvent delivery module, SIL-20A autosampler, and SPD-20A UV-Vis detector. A size exclusion column (TSKgel™ G4000 PWXL, 7.8 mm × 300 mm; Tosoh Biosciences, Montgomeryville, PA) was used at room temperature. The mobile phase was distilled, deionized water and was delivered at a flow rate of 0.5 mL/min and detection was at 490 nm for DEX 20 kDa and 280 nm for BSA and PSS. The concentration of BEV was quantified by enzyme-linked immunosorbent assay (ELISA) as described previously (Krohne et al., 2008) with modifications. Briefly, samples were diluted with 0.05% (w/v) BSA in PBS and were analyzed in duplicates. 100 µL of each sample was incubated in 96-well plates coated with human immunoglobulin G (IgG)-specific goat IgG (BD Biocoat; BD Biosciences, San Jose, CA) for 1 h at room temperature. Subsequently, the wells were incubated for 1 h with biotinylated recombinant human VEGF165 (Fluorokine biotinylated human VEGF; R&D Systems, Minneapolis, MN) diluted in 0.05% (w/v) BSA in PBS. This was followed by the addition of 100 µL peroxidase-conjugated streptavidin (BD Biosciences, San Jose, CA) diluted 1:2000 in 0.05% (w/v) BSA in PBS and incubated for 1 h. Peroxidase activity was determined after the incubation with 100 µL peroxidase substrate solutions (BD Biosciences, San Jose, CA) for 10 min. Absorbance was quantified in a SpectraMax 250 (GMI, Inc., Ramsey, MN) microplate reader at 450 nm with the subtraction of reference absorbance at 650 nm. The detection limit of the assay was 30 ng/mL.
2.5. Theory and equation
The cumulative amount of a permeant transported across the sclera (Q) was plotted against time (t). The steady-state flux of the permeant (J) was calculated from the slope of the linear portion of the plot (ΔQ/Δt) under sink conditions, correcting for the effective diffusion area (AD). The steady-state permeability coefficient (P) was calculated by normalizing the flux by the donor concentration (CD). In the iontophoresis experiments, P was the effective permeability coefficient under the particular iontophoresis condition for comparison to the passive control. Enhancement factor (E) is calculated by the ratio of permeability coefficient of iontophoretic transport to that of passive transport.
| (1) |
| (2) |
The steady-state iontophoretic flux (JΔψ) of the permeant through a homogeneous porous membrane can be described by the modified Nernst-Planck model (Li et al., 2005), a model appropriate for constant current iontophoresis when the electrical resistance of the membrane and the voltage applied across the membrane are relatively constant during iontophoresis.
| (3) |
where ψ is the electric potential in the membrane, F is the Faraday constant, Rgas is the gas constant, T is the temperature, ν is the average velocity of the convective solvent flow, ε is the combined porosity and tortuosity factor of the membrane, and C, x, z, and D are the concentration, the position in the membrane, the charge number, and the diffusion coefficient of the permeant, respectively. H is the hindrance factor for simultaneous Brownian diffusion and migration driven by the electric field and W is the hindrance factor for permeant transport via convective solvent flow during iontophoresis. Assuming cylindrical pore geometry in the membrane and using asymptotic centerline approximation, the hindrance factor H can be expressed as (Deen, 1987):
| (4) |
where λi is the ratio of permeant radius (rs) to the pore radius (Rp) and Kt is:
| (5) |
and a1 = −1.217, a2 = 1.534, a3 = −22.51, a4 = −5.612, a5 = −0.3363, a6 = −1.216, a7 = 1.647. When λi is <0.4, Eq. 4 is equivalent to the commonly used Renkin equation. The effective pore radius of the membrane can be calculated from the ratio of the permeability coefficients of the permeants (of different molecular sizes) obtained from the passive transport experiments using Eqs. 4–6.
| (6) |
2.6. Statistical analysis
All experiments were conducted with a minimum of four replicates using sclera from different human donors. The means ± standard deviations (SD) of the data are presented, and the statistical differences were determined by Student’s t-tests. Differences were considered to be significant at a level of p < 0.05. Power of the test was also performed in paired comparisons to avoid type II error in testing the null hypothesis.
3. Results and discussion
3.1. Passive transport of permeants
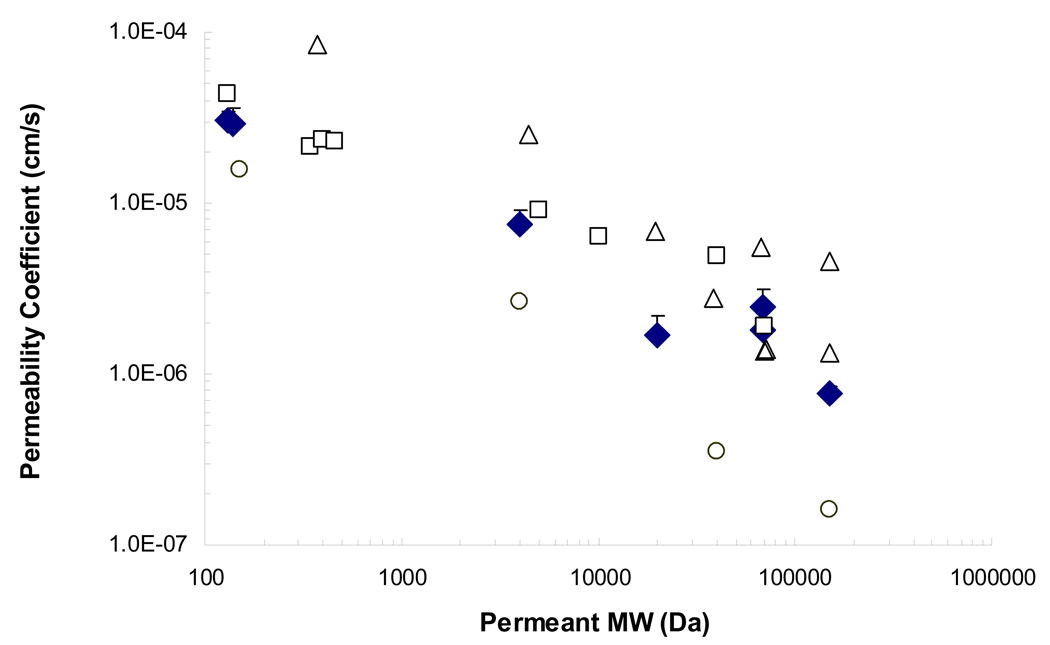
The passive permeability coefficients of human sclera for the permeants TEA, SA, DEX of MW 4 and 20 kDa, BSA, PSS, and BEV were calculated using Eq. 2 and presented in Fig. 1. TEA and SA have comparable passive permeability coefficients due to their similar MW. Likewise, the passive permeability coefficients of BSA and PSS are approximately the same as they have similar MW. The passive permeability coefficient decreases (from 3 × 10−5 to 8 × 10−7 cm/s) when the MW of the permeant increases (from around 130 Da to 150 kDa), consistent with previous trends of a general inverse relationship between the permeability coefficient and MW of permeants (Olsen et al., 1995; Prausnitz and Noonan, 1998; Ambati et al., 2000; Nicoli et al., 2009). Fig. 1 also provides the comparison between passive permeability coefficients of the permeants in the present study and those from previous studies. The passive permeability coefficient values of macromolecules in the present study are close to the values in a previous human sclera study (Olsen et al., 1995), generally lower than those in the rabbit sclera (Ambati et al., 2000), and higher than those in the porcine sclera studies (Nicoli et al., 2009).
Figure 1.
Comparison of the relationships between passive permeability coefficients and permeant MW for human sclera in the present study and those for human, rabbit, and porcine sclera in the literature. Symbols: closed diamonds, experimental passive permeability with human sclera in the present study (mean and standard deviation, n ≥ 4); open squares, previous human passive permeability data (Olsen et al., 1995); open triangles, rabbit passive permeability data (Ambati et al., 2000); open circles, porcine passive permeability data (Nicoli et al., 2009).
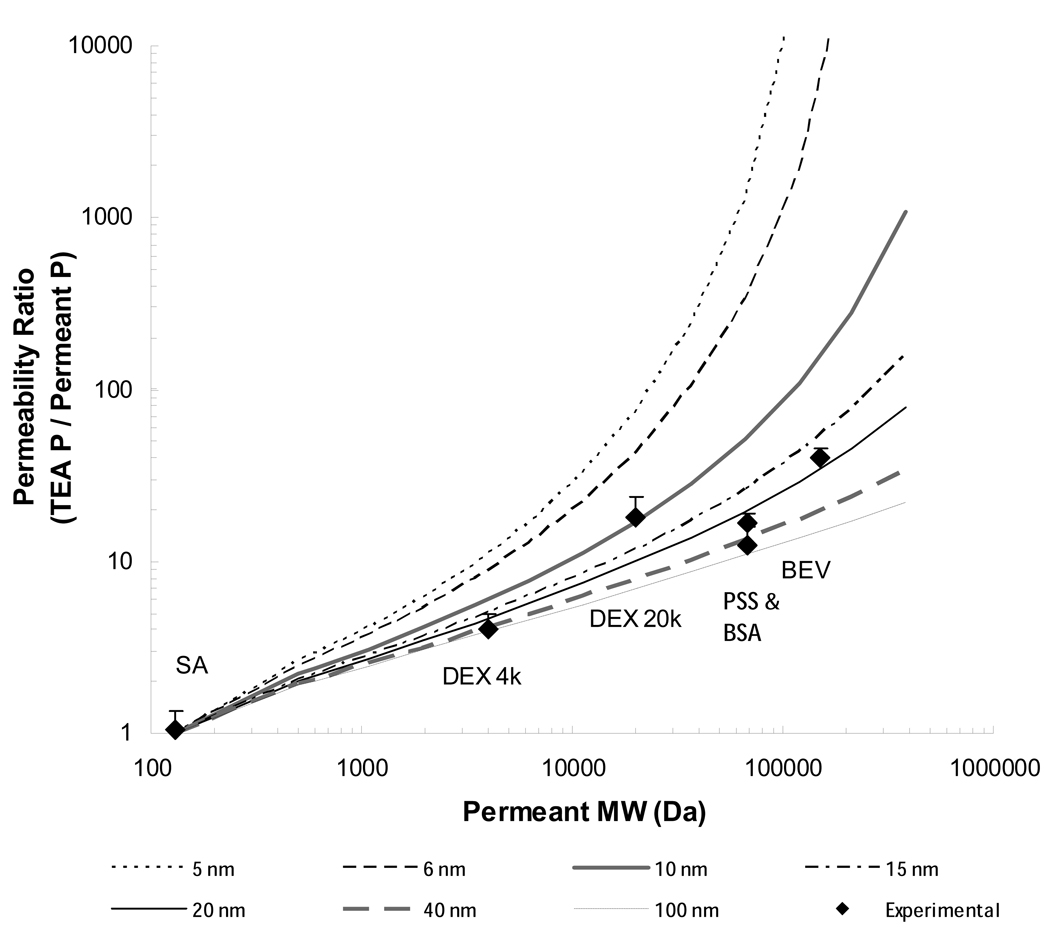
Fig. 2 is a plot of the passive permeability coefficient ratio of TEA to the permeants versus permeant MW in the present study. The lines in the figure represent the theoretical calculations of Eqs. 4−6. Using the experimental passive permeability coefficient ratios of TEA to BSA, TEA to PSS, TEA to BEV, and TEA to the DEXs in Fig. 2, the average effective pore radius of human sclera was estimated to be around 10–40 nm.
Figure 2.
Permeability coefficient ratios of permeant vs. permeant MW. Symbols: experimental permeability coefficient ratios of TEA to SA, TEA to BSA, TEA to PSS, TEA to BEV, and TEA to DEXs with MW 4 and 20 kDa (DEX 4k and DEX 20k). The lines represent the theoretical calculations of the permeant permeability ratio vs. permeant MW at different effective membrane pore radius using Eqs. 4–6.
3.2. Iontophoretic transport of small charged permeants
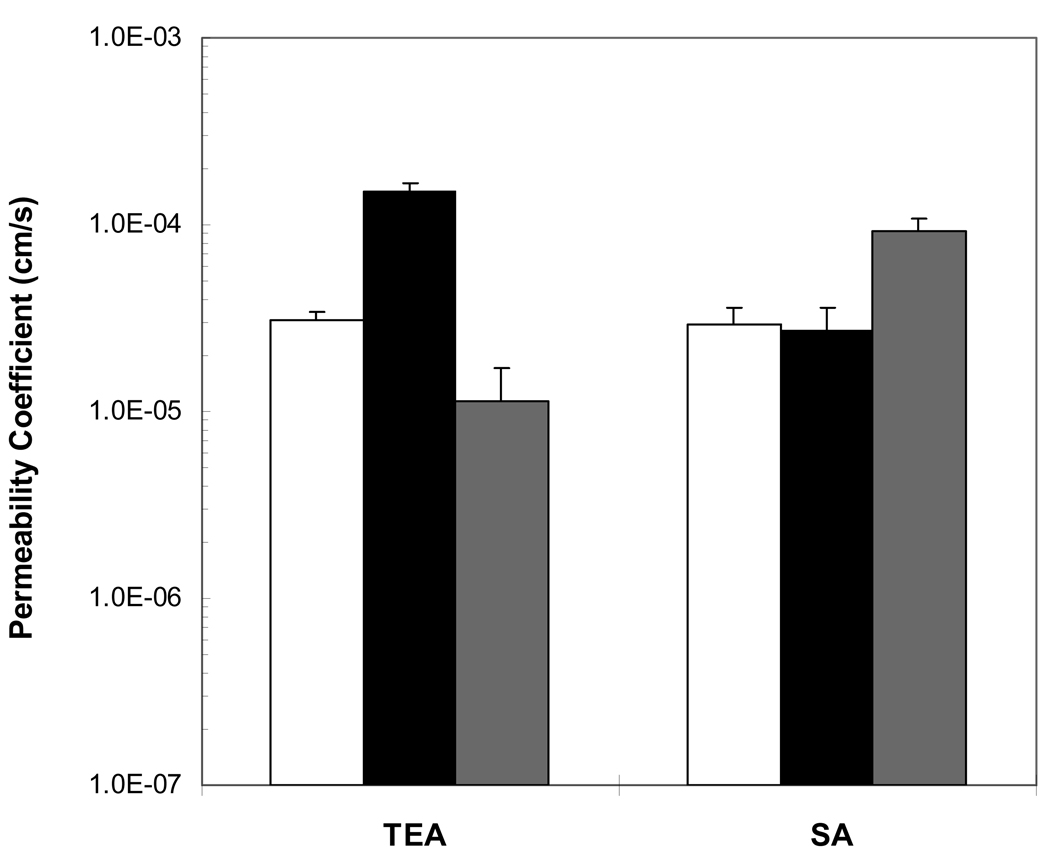
The passive and effective iontophoretic permeability coefficients of TEA and SA are presented in Fig. 3. The figure shows significantly higher effective iontophoretic permeability coefficients of TEA during anodal iontophoresis and of SA during cathodal iontophoresis compared to those of their respective passive transport (p < 0.05, Student’s t-test), suggesting significant flux enhancement due to iontophoresis. The higher effective iontophoretic permeability coefficient of TEA during anodal iontophoresis when the donor electrode had the same polarity as the permeant relative to those of passive and cathodal transport suggests that electrophoresis was the main flux enhancing mechanism for transscleral iontophoresis of small charged molecules. The SA results with higher effective cathodal iontophoretic permeability coefficient relative to those of passive and anodal transport are consistent with this finding. Iontophoresis enhanced the fluxes of TEA and SA by approximately 5- and 3-fold during anodal and cathodal iontophoresis, respectively (Table 1). The differences in the iontophoretic transport enhancement of TEA and SA can be attributed to the effect of electroosmosis, consistent with previous finding of transscleral electroosmosis and the negatively charged sclera at physiological pH (Li et al., 2005).
Figure 3.
Effective permeability coefficients of TEA and SA obtained in the passive (open bars), anodal iontophoretic (black bars), and cathodal iontophoretic (gray bars) transport experiments with human sclera. Data represent the mean and standard deviation, n ≥ 4. The Student’s t-test analyses show significant differences between the iontophoresis and passive data (p < 0.05) and between the cathodal iontophoresis and anodal iontophoresis data (p < 0.05).
Table 1.
Enhancement factors of charged small and macromolecules a
| Experiment | TEA | SA | BSA | PSS | BEV |
|---|---|---|---|---|---|
| Anodal iontophoresis | 4.9 | 0.9 | 58 | 7 | 32 |
| Cathodal iontophoresis | 0.4 | 3.2 | 19 | 12 | 8 |
Data represent the mean of n ≥ 4 samples.
The effective permeability coefficients of passive and iontophoretic transport of the small permeants for human sclera in the present study are consistent with those obtained previously with rabbit sclera (Li et al., 2005), suggesting that the rabbit sclera can be an appropriate model for transscleral transport of small molecules in vitro. In addition, no significant difference (p > 0.05) in the passive permeability coefficients of TEA and SA before and after iontophoresis (3.1 ± 0.5 and 3.3 ± 0.4 × 10−5 cm/s, mean ± SD, n = 6, for TEA and SA, respectively, after iontophoresis) was observed. This suggests minimal irreversible impact of the electric field on the barrier properties of the tissue. If electro-permeabilization of human sclera had occurred, the data show that the effect was reversible and the tissue regained its original properties when the application of electric current was terminated.
3.3. Iontophoretic transport of charged macromolecules
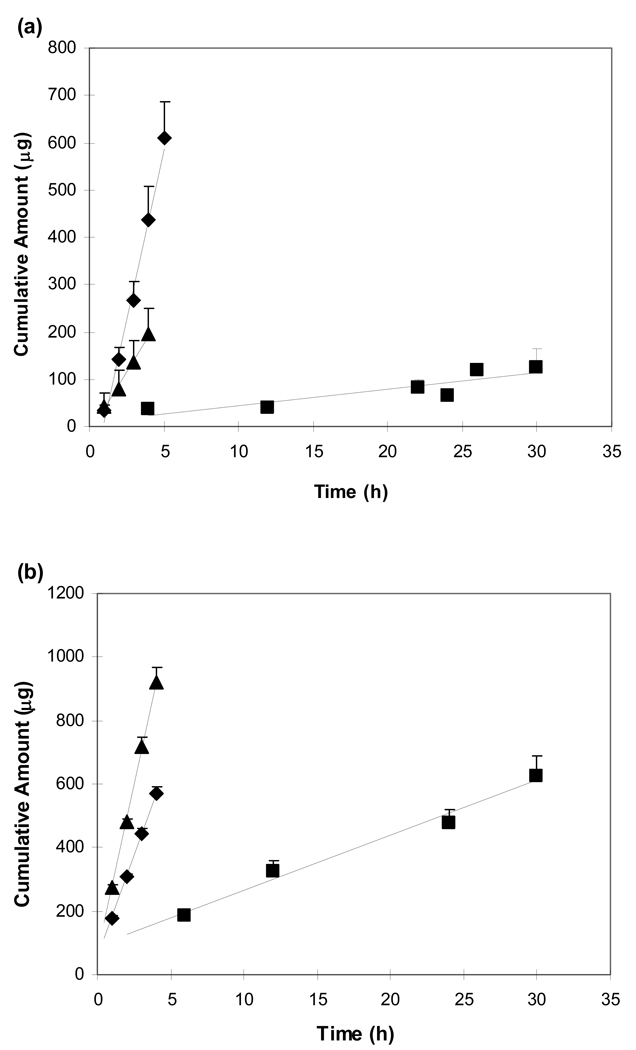
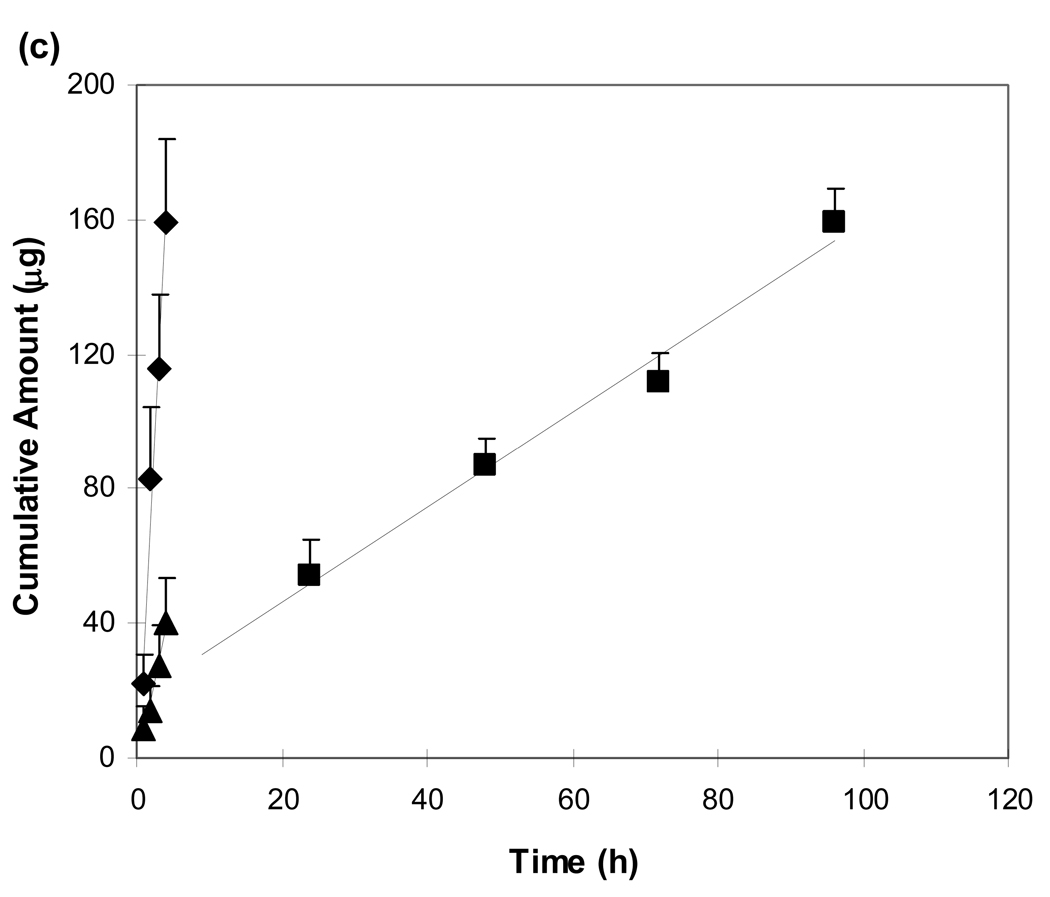
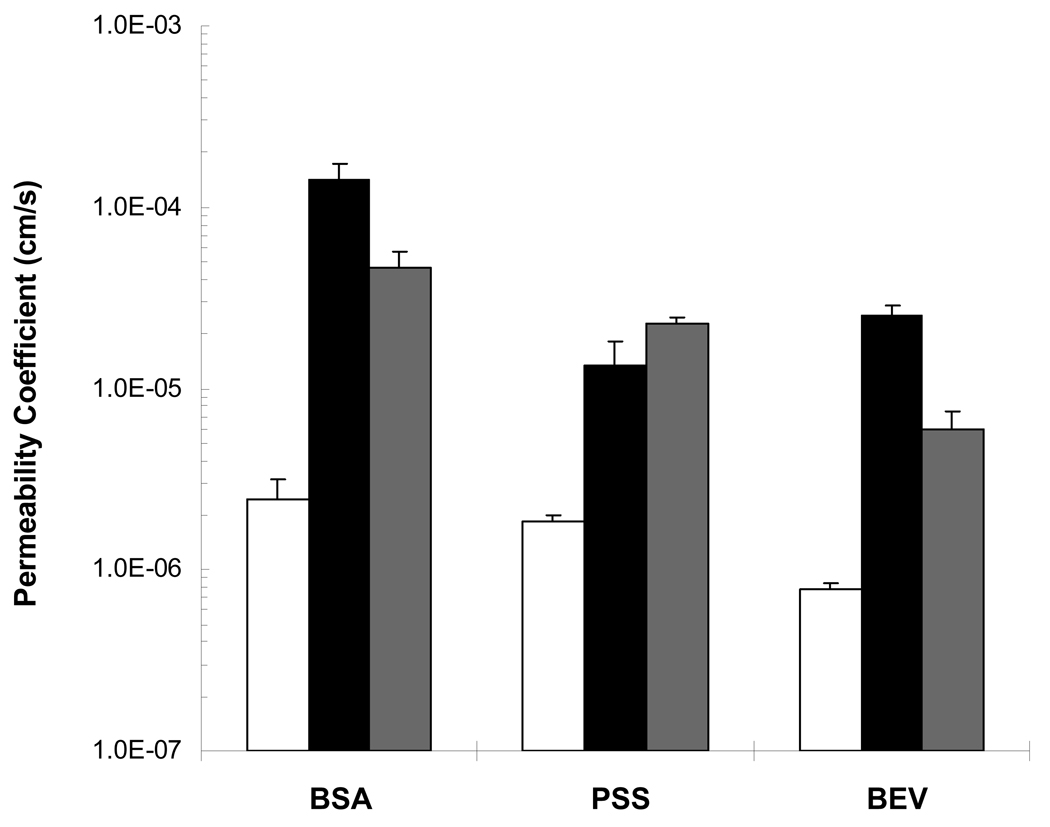
Fig. 4 presents the cumulative amounts of BSA, PSS, and BEV transported across the sclera in passive transport, anodal iontophoresis, and cathodal iontophoresis. The effective permeability coefficients are summarized in Fig. 5. For BSA, higher effective permeability coefficient was observed during anodal iontophoresis than that during cathodal iontophoresis (p < 0.05, Student’s t-test). The observation that the transport of the macromolecule was enhanced even when the polarity of the electrode in the donor was opposite to the charge of the permeant, i.e., anodal iontophoresis of BSA, suggests the dominant effect of electroosmosis for the macromolecule. The significance of electroosmosis on transscleral iontophoretic transport of BSA is similar to the findings in previous skin studies, in which BSA transport was enhanced across human skin by anodal iontophoresis due to the dominant electroosmosis effect upon macromolecules of low charge density (Pikal, 1992). For BEV, similarly higher effective permeability coefficient (p < 0.05, Student’s t-test) was observed during anodal iontophoresis compared to that during passive transport and cathodal iontophoresis. The significant anodal iontophoretic transport enhancement is likely due to the dominant effect of electroosmosis in the system similar to the result in the BSA transport study. Different from the BSA and BEV results, significantly higher effective permeability coefficient (p < 0.05, Student’s t-test) was observed in cathodal iontophoresis of PSS as compared to those in passive transport and anodal iontophoresis. The higher flux enhancement of PSS during cathodal iontophoresis was likely a result of the dominant effect of electrophoresis (versus dominant electroosmosis for BSA and BEV).
Figure 4.
Representative plots of cumulative amount transported across human sclera vs. time for (a) BSA, (b) PSS, and (c) BEV in passive transport (squares), anodal iontophoresis (diamonds), and cathodal iontophoresis (triangles). Data represent the mean and standard deviation, n ≥ 4 for each condition.
Figure 5.
Effective permeability coefficients of BSA, PSS, and BEV in the passive (open bars), anodal iontophoretic (black bars), and cathodal iontophoretic (gray bars) transport experiments with human sclera. Data represent the mean and standard deviation, n ≥ 4. The Student’s t-test analyses show significant differences between the iontophoresis and passive data (p < 0.05) and between the cathodal iontophoresis and anodal iontophoresis data (p < 0.05).
The iontophoretic transport enhancement of macromolecules depends on the net effect of transport enhancements due to electrophoresis and electroosmosis. At pH 7.4 in the present study, the human sclera was expected to be net negatively charged (Li et al., 2005) and electroosmosis favored the transport from the anode to cathode in anodal iontophoresis. The electrophoresis transport enhancement of negatively charged macromolecules is anticipated from the cathode to anode in cathodal iontophoresis, which is a function of the charge to mass ratio of the macromolecule. Given the similar MW of BSA and PSS, the difference in the iontophoretic transport results of BSA and PSS is due to the charge to mass ratio of the macromolecules. BSA has a lower charge to mass ratio than PSS, which is a polyelectrolyte with high negative charges. Other factors that can affect iontophoretic transport of macromolecules but have not been discussed here include molecular shape and electrophoretic and relaxation effects (Bockris and Reddy, 1998). It should be noted that the fluxes of BSA, PSS, and BEV were enhanced by both anodal and cathodal iontophoresis. The effects of electrophoresis in cathodal iontophoresis of BSA and BEV and the effect of electroosmosis in anodal iontophoresis of PSS could be significant as indicated by the flux enhancement observed in cathodal iontophoresis of BSA and BEV and anodal iontophoresis of PSS, respectively. Other mechanisms such as changes in the sclera properties caused by the macromolecules and/or electric field during iontophoresis might also be involved.
3.4. Transscleral drug delivery
Several studies have shown that transscleral iontophoresis is safe and efficient for ocular drug delivery and could take advantage of the large surface area of the sclera (Behar-Cohen et al., 2002; Eljarrat-Binstock et al., 2008a). Previous studies have shown that the sclera allows the penetration of macromolecules such as DEX of 70–150 kDa (Ambati et al., 2000; Nicoli et al., 2009) and transscleral delivery depends on the size and shape of the permeant (Olsen et al., 1995; Cruysberg et al., 2002; Maurice, 2002). Consistent with the previous findings, the results in the present study show a similar relationship between passive permeability coefficient and molecular size for the charged permeants. The present iontophoresis data suggest that iontophoretic flux enhancement of small charged permeants was governed by electrophoresis along with small contribution of electroosmosis. For BSA and BEV, macromolecules with low charge to mass ratios, electroosmosis is the major contributing mechanism in the iontophoretic transport of the macromolecules. Anodal iontophoresis could be a promising strategy to deliver BEV through human sclera for the treatment of retinal eye diseases. This is consistent with the findings of neutral macromolecules in a previous study (Nicoli et al., 2009).
The model permeants employed in the present study have similar MW as those of therapeutic proteins/ peptides and oligonucleotides that could be used in the treatment of posterior eye diseases. It is therefore believed that proteins of up to 150 kDa can be effectively delivered by anodal iontophoresis but highly negatively charged macromolecules such as oligonucleotides and siRNA are more effectively delivered by cathodal iontophoresis. As electroosmosis can be affected by formulation factors such as solution ionic strength and pH and the presence of surfactants, a key to effective iontophoretic delivery of charged macromolecules is to understand the interplay of these factors on transscleral electroosmosis in future studies.
4. Conclusions
The iontophoretic transport of macromolecules has been studied by using human sclera in vitro. Transport of the macromolecules was significantly affected by electroosmosis as such the transport of negatively charged BSA was enhanced from the anode to cathode. The effect of electroosmosis in iontophoretic transport of polyelectrolyte such as PSS was relatively less important than that of electrophoresis. No irreversible effect of the electric field upon the barrier properties of human sclera was observed under the experimental conditions studied. The iontophoresis results with human sclera are comparable to those of rabbit sclera. Transscleral iontophoresis of biologicals could provide new therapeutic approaches for treating posterior eye diseases. Understanding the different mechanisms involved in transscleral iontophoresis of these macromolecules would help researchers optimize this drug delivery method.
Acknowledgements
This research was supported by NIH grant EY 015181. The authors thank Dr. Paul S. Bernstein at Moran Eye Center Utah for generously supplying us with some of the sclera used in this study.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ambati J, Canakis CS, Miller JW, Gragoudas ES, Edwards A, Weissgold DJ, Kim I, Delori FC, Adamis AP. Diffusion of high molecular weight compounds through sclera. Invest. Ophthalmol. Vis. Sci. 2000;41:1181–1185. [PubMed] [Google Scholar]
- Behar-Cohen F, El Aouni A, Gautier S, David G, Davis J, Chapon P, Parel JM. Transscleral coulomb-controlled iontophoresis of methylprednisolone into the rabbit eye: Influence of duration of treatment, current intensity and drug concentration on ocular tissue and fluid levels. Exp. Eye Res. 2002;74:51–59. doi: 10.1006/exer.2001.1098. [DOI] [PubMed] [Google Scholar]
- Behar-Cohen FF, Parel JM, Pouliquen Y, Thillaye-Goldenberg B, Goureau O, Heydolph S, Courtois Y, De Kozak Y. Iontophoresis of dexamethasone in the treatment of endotoxin-induced-uveitis in rats. Exp Eye Res. 1997;65:533–545. doi: 10.1006/exer.1997.0364. [DOI] [PubMed] [Google Scholar]
- Bockris JOM, Reddy AKN. Modern Electrochemistry. 2nd. New York: Kluwer Academic/Plenum Publishers; 1998. [Google Scholar]
- Cruysberg LPJ, Nuijts RMMA, Geroski DH, Koole LH, Hendrikse F, Edelhauser HF. In vitro human scleral permeability of fluorescein, dexamethasonefluorescein, methotrexate-fluorescein and rhodamine 6G and the use of a coated coil as a new drug delivery system. J. Ocul. Pharmaco. Ther. 2002;18:559–569. doi: 10.1089/108076802321021108. [DOI] [PubMed] [Google Scholar]
- Davies JB, Ciavatta VT, Boatright JH, Nickerson JM. Delivery of several forms of DNA, DNA-RNA hybrids, and dyes across human sclera by electrical fields. Mol. Vis. 2003;9:569–578. [PubMed] [Google Scholar]
- Deen WM. Hindered transport of large molecules in liquid-filled pores. AlChE. J. 1987;33:1409–1425. [Google Scholar]
- Edwards DA, Langer R. A linear-theory of transdermal transport phenomena. J. Pharm. Sci. 1994;83:1315–1334. doi: 10.1002/jps.2600830925. [DOI] [PubMed] [Google Scholar]
- Eljarrat-Binstock E, Domb AJ. Iontophoresis: A non-invasive ocular drug delivery. J. Control. Release. 2006;110:479–489. doi: 10.1016/j.jconrel.2005.09.049. [DOI] [PubMed] [Google Scholar]
- Eljarrat-Binstock E, Domb AJ, Orucov F, Dagan A, Frucht-Pery J, Pe'er J. In vitro and in vivo evaluation of carboplatin delivery to the eye using hydrogel-iontophoresis. Curr. Eye Res. 2008a;33:269–275. doi: 10.1080/02713680701871140. [DOI] [PubMed] [Google Scholar]
- Eljarrat-Binstock E, Domb AJ, Orucov F, Frucht-Pery J, Pe'er J. Methotrexate delivery to the eye using transscleral hydrogel iontophoresis. Curr. Eye Res. 2007;32:639–646. doi: 10.1080/02713680701528674. [DOI] [PubMed] [Google Scholar]
- Eljarrat-Binstock E, Orucov F, Frucht-Pery J, Pe'er J, Domb AJ. Methylprednisolone delivery to the back of the eye using hydrogel iontophoresis. J. Ocul. Pharmacol Ther. 2008b;24:344–350. doi: 10.1089/jop.2007.0097. [DOI] [PubMed] [Google Scholar]
- Frucht-Pery J, Raiskup F, Mechoulam H, Shapiro M, Eljarrat-Binstock E, Domb A. Iontophoretic treatment of experimental pseudomonas keratitis in rabbit eyes using gentamicin-loaded hydrogels. Cornea. 2006;25:1182–1186. doi: 10.1097/01.ico.0000243959.14651.18. [DOI] [PubMed] [Google Scholar]
- Hofman P, Blaauwgeers HGT, Tolentino MJ, Adamis AP, Cardozo BJN, Vrensen GFJM, Schlingemann RO. VEGF-A induced hyperpermeability of blood-retinal barrier endothelium in vivo is predominantly associated with pinocytotic vesicular transport and not with formation of fenestrations. Curr. Eye Res. 2000;21:637–645. [PubMed] [Google Scholar]
- Jager RD, Aiello LP, Patel SC, Cunningham ET. Risks of intravitreous injection: A comprehensive review. Retina. 2004;24:676–698. doi: 10.1097/00006982-200410000-00002. [DOI] [PubMed] [Google Scholar]
- Kasting GB. Theoretical models for iontophoretic delivery. Adv. Drug Deliv. Rev. 1992;9:177–199. [Google Scholar]
- Krohne TU, Eter N, Holz FG, Meyer CH. Intraocular pharmacokinetics of bevacizumab after a single intravitreal injection in humans. Am. J. Ophthalmol. 2008;146:508–512. doi: 10.1016/j.ajo.2008.05.036. [DOI] [PubMed] [Google Scholar]
- Li SK, Zhang YH, Zhu HG, Higuchi WI, White HS. Influence of asymmetric donor-receiver ion concentration upon transscleral iontophoretic transport. J. Pharm. Sci. 2005;94:847–860. doi: 10.1002/jps.20293. [DOI] [PubMed] [Google Scholar]
- Maurice DM. Drug delivery to the posterior segment from drops. Surv. Ophthalmol. 2002;47:S41–S52. doi: 10.1016/s0039-6257(02)00326-0. [DOI] [PubMed] [Google Scholar]
- Michels S, Rosenfeld PJ, Puliafito CA, Marcus EN, Venkatraman AS. Systemic bevacizumab (Avastin) therapy for neovascular age-related macular degeneration - Twelve-week results of an uncontrolled open-label clinical study. Ophthalmology. 2005;112:1035–1047. doi: 10.1016/j.ophtha.2005.02.007. [DOI] [PubMed] [Google Scholar]
- Mordenti J, Cuthbertson RA, Ferrara N, Thomsen K, Berleau L, Licko V, Allen PC, Valverde CR, Meng YG, Fei DT, Fourre KM, Ryan AM. Comparisons of the intraocular tissue distribution, pharmacokinetics, and safety of 125I-labeled full-length and Fab antibodies in rhesus monkeys following intravitreal administration. Toxicol. Pathol. 1999;27:536–544. doi: 10.1177/019262339902700507. [DOI] [PubMed] [Google Scholar]
- Myles ME, Neumann DM, Hill JM. Recent progress in ocular drug delivery for posterior segment disease: Emphasis on transscleral iontophoresis. Adv. Drug Deliv. Rev. 2005;57:2063–2079. doi: 10.1016/j.addr.2005.08.006. [DOI] [PubMed] [Google Scholar]
- Nicoli S, Ferrari G, Quarta M, Macaluso C, Santi P. In vitro transscleral iontophoresis of high molecular weight neutral compounds. Eur. J. Pharm. Sci. 2009;36:486–492. doi: 10.1016/j.ejps.2008.11.012. [DOI] [PubMed] [Google Scholar]
- Olsen TW, Edelhauser HF, Lim JI, Geroski DH. Human scleral permeability - Effects of age, cryotherapy, transscleral diode-laser, and surgical thinning. Invest. Ophthalmol. Vis. Sci. 1995;36:1893–1903. [PubMed] [Google Scholar]
- Pikal MJ. The role of electroosmotic flow in transdermal iontophoresis. Adv. Drug Deliv. Rev. 1992;9:201–237. doi: 10.1016/s0169-409x(00)00138-1. [DOI] [PubMed] [Google Scholar]
- Prausnitz MR, Noonan JS. Permeability of cornea, sclera, and conjunctiva: A literature analysis for drug delivery to the eye. J. Pharm. Sci. 1998;87:1479–1488. doi: 10.1021/js9802594. [DOI] [PubMed] [Google Scholar]
- Ranta VP, Urtti A. Transscleral drug delivery to the posterior eye: Prospects of pharmacokinetic modeling. Adv. Drug Deliv. Rev. 2006;58:1164–1181. doi: 10.1016/j.addr.2006.07.025. [DOI] [PubMed] [Google Scholar]
- Sims SM, Higuchi WI, Srinivasan V. Interaction of electric-field and electroosmotic effects in determining iontophoretic enhancement of anions and cations. Int. J. Pharm. 1991;77:107–118. [Google Scholar]
- Srinivasan V, Higuchi WI, Sims SM, Ghanem AH, Behl CR. Transdermal iontophoretic drug delivery : mechanistic analysis and application to polypeptide delivery. J. Pharm. Sci. 1989;78:370–375. doi: 10.1002/jps.2600780506. [DOI] [PubMed] [Google Scholar]
- Urtti A. Challenges and obstacles of ocular pharmacokinetics and drug delivery. Adv. Drug Deliv. Rev. 2006;58:1131–1135. doi: 10.1016/j.addr.2006.07.027. [DOI] [PubMed] [Google Scholar]