Abstract
The pharmacokinetics of orally administered pefloxacin were studied to evaluate the bio-enhancing effect of the herbal bio-enhancer, trikatu, in mountain Gaddi goats (n = 6). The findings of the study revealed a decreased plasma concentration (p > 0.05) of pefloxacin following trikatu administration during the absorption phase (10, 15, 20 min post pefloxacin administration). In contrast, the plasma concentrations of pefloxacin were significantly higher at 4, 6, 8 and 12 h (during the elimination phase) of the pefloxacin administration. The findings of the investigation revealed higher values for the area under the curve, the area under the first moment of the plasma drug concentration time curve, the mean residential time, the total duration of pharmacological action and bioavailability. Trikatu treatment, however, significantly reduced the elimination half life (t1/2β) and zero time intercept of the elimination phase. The apparent volume of distribution based on the total area under the plasma drug concentration curve [(Vd(area)] and the apparent volume of distribution based on the zero time plasma concentration intercept of the elimination phase [Vd(B)] were significantly higher in trikatu treated animals indicating a better penetration of the drug. Based on the MIC of 0.8 µg/ml of pefloxacin, a priming dose of 6.0 mg/kg and a maintenance dose of 2.21 mg/kg is required to be administered at 8 h intervals. For practical purposes in goats this would mean a priming dose of 6 mg/kg and a maintenance dose of 2 mg/kg given by the oral route, to be repeated at 8 h intervals.
Keywords: goats, oral administration, pefloxacin, pharmacokinetics, trikatu
Introduction
Fluoroquinolones are now becoming the antibacterials of choice in veterinary therapeutics. The fluoroquinolones have also become popular as there are minimal chances of bacterial resistance with these agents. The pharmacokinetics of pefloxacin are clearly described in various species [1,8,10,15]. Inspection of the pharmacokinetic behavior in various animal species reveals that the drug is poorly absorbed in goats following its oral administration. One practical approach to enhance the bioavailability is by using herbal bio-enhancers with the antibacterial agents. Piper longum pretreatment has been successfully employed as a bio-enhancer with orally administered oxytetracycline in WLH poultry birds [7]. In the present study, the effect of the herbal bio-enhancer, trikatu (equal parts of Piper longum, Piper nigrum and Zingiber officinale) on the pharmacokinetic behaviour of pefloxacin in mountain Gaddi goats is presented.
Materials and Methods
The study was conducted on six adult healthy mountain Gaddi goats weighing 18-22 kg. The same animals were used for both treatments after a wash out period of 15 days. The goats were kept under close observation before beginning the experiments to enable them to acclimatize to the new environment. In the first treatment pefloxacin (Pelwin; Wokhardt, India) was given at the dose rate of 20 mg/kg body weight whereas in second treatment animals were orally administered trikatu (piperine contents of trikatu 2.02% (w/w)) at a dose rate of 2 g/kg for 14 days and on day 15, pefloxacin was administered at the dose rate of 20 mg/kg. 5 ml blood samples were collected from each animal in heparinized tubes at 10, 15, 20, 30, 45, 60, 120, 240, 360, 480 and 720 min post drug administration and were centrifuged at 3,000 rpm for 10 min for plasma separation. The plasma samples were stored at -20℃ for microbial assay. The assay was completed within 1 week.
The concentration of microbiologically active pefloxacin (parent & metabolites) in plasma were determined by the agar plate diffusion method [4] using E. coli (strain MTCC-443) as the test organism with the modification that large petri plates were used instead of large glass plates and only three punches were made for the microbial assay.
The plasma concentration-time profile of pefloxacin for each goat was used to determine the pharmacokinetics. Different pharmacokinetic parameters were analysed using the "method of least square" and the "method of residual yields" as described by Gibaldi and Perrier [5]. Estimation of the area under the concentration-time curve (AUC) and the area under the first moment of the plasma drug concentration time curve (AUMC) were based on the trapezoidal rule [5]. The compartmental analysis of plasma concentration vs. time was carried out using the mono-exponential equation:
Cp(t) = Be-βt - Ae-Kat
Where Cp(t) is the plasma drug concentration at time t, B is the zero time intercept of the regression line of the elimination phase, A is the zero time plasma drug concentration intercept of the regression line of the absorption phase, Ka is the absorption rate constant, β is the overall elimination rate constant, t is time and e is the natural logarithm base.
The dosage regimen for maintaining MIC of 0.1-0.8 µg/ml for most organisms [13] at dosage interval of 4, 6 and 8 h was derived as per the method described by Notari [11].
The difference between the means of the two treatments was determined by student's t-test [14] and the data were analyzed using GraphPad Instat (GraphPad Software, USA).
Results
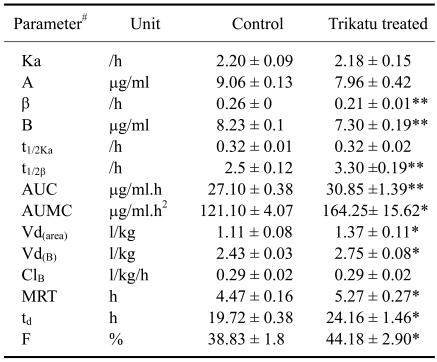
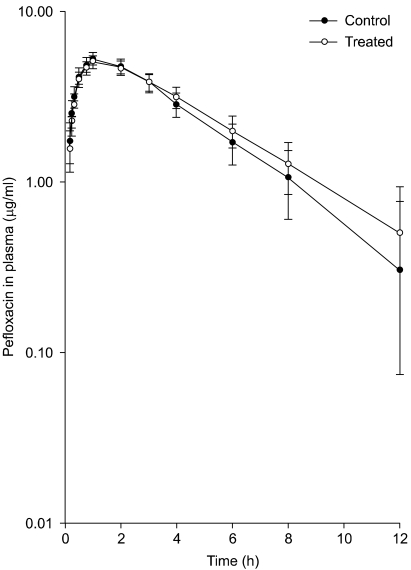
The results of the study revealed that the mean plasma concentrations of pefloxacin were consistently lower in trikatu treated goats up to 2 h. The values were significantly lower at 10 min, 15 min and 20 min post drug administration (Table 1 and Fig. 1). The values at 20 min post drug administration were 3.17 µg/ml in control animals against 2.87 µg/ml in trikatu treated goats. In contrast, the plasma concentrations of pefloxacin were significantly higher at 4, 6, 8 and 12 h post pefloxacin administration. The plasma levels were 1.28 µg/ml in trikatu treated animals at 8 h whereas the plasma levels were 1.07 µg/ml in control animals.
Table 1.
Comparative pharmacokinetic profile of pefloxacin administered orally (20 mg/kg) in control and trikatu treated goats (mean ± SE)
#Pharmacokinetic parameters as described by Gibaldi and Perrier [5], Ka: absorption rate constant, A: zero time plasma drug concentration intercept of regression line of absorption phase, β: overall elimination rate constant, B: zero time intercept of the regression line of the elimination phase, t1/2Ka: absorption half life, t1/2β: elimination half life, AUC: area under the curve, AUMC: area under the first moment of the plasma drug concentration time curve, MRT: mean residential time, td: total duration of pharmacological action, ClB: total body clearance; Vd(area): volume of distribution based on the total area under the plasma drug concentration time curve; Vd(B): volume of distribution based on the zero time plasma drug concentration intercept of the elimination phase. *p ≤ 0.05, **p ≤ 0.01.
Fig. 1.
Comparative semilogarithmic plot of plasma concentration - time profile of pefloxacin in control and trikatu treated goats following oral administration (20 mg/kg).
The values of the pharmacokinetic determinants in the control and trikatu treated animals are given in Table 2. The values of the absorption rate constant (Ka) and zero time intercept of the absorption phase (A1) revealed a non significant difference between the two groups.
Table 2.
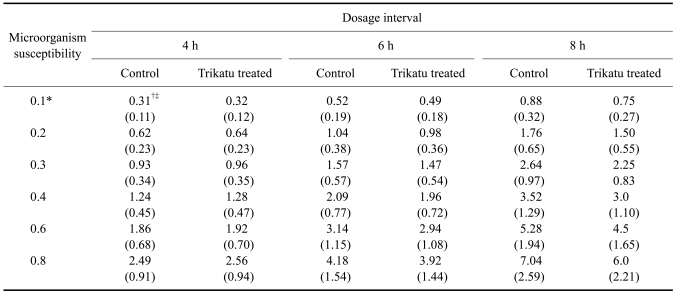
Dosage regimen of pefloxacin, calculated on the basis of pharmacokinetic values obtained following its oral administration (with and without trikatu treatment) in goats at dosage intervals for micro-organisms of different susceptibilities
*Values are expressed as µg/ml. †Values given are expressed as mg/kg body weight. ‡Values given are loading doses and the values in parenthesis are maintenance doses.
The findings of the present study further revealed significantly higher values (30.85 ± 1.39 µg/ml.h in trikatu treated animals against 27.10 µg/ml.h in control animals) for the area under curve (AUC), AUMC, the mean residential time (MRT) and the total duration of pharmacological action (td) in the trikatu treated goats. The mean residential time was 5.27 ± 0.27 h in trikatu treated animals against 4.47 ± 0.16 h in control animals. Similarly, the total duration of the td was 24.16 ± 1.46 h in trikatu treated animals against 19.72 ± 0.38 h in control animals. The bioavailability of the drug was also significantly higher (p < 0.05) in trikatu treated goats (44.18 ± 2.90% against 38.33 ± 1.80% in control animals).
Prior treatment of the goats with trikatu significantly reduced the elimination half life (t1/2β) and the zero time intercept of the elimination phase (B). The elimination half life was 3.30 ± 0.19 /h in trikatu treated against 2.5 ± 0.12/h in control animals. The total body clearance (ClB), however, did not show any significant difference in the trikatu treated goats.
The indicators of drug distribution in the body i.e. apparent volume of distribution [Vd(area)] and volume of distribution based on the zero time plasma drug concentration intercept of the elimination phase [Vd(B)] were significantly higher in the trikatu treated goats. The values of Vd(area) in trikatu treated goats were 1.37 ± 0.11 l/kg whereas the values of Vd(area) were 1.11 ± 0.08 l/kg in control animals.
The oral priming and maintenance doses of pefloxacin based on the minimum inhibitory concentration (MIC) of 0.1, 0.2, 0.3, 0.4 and 0.6 and 0.8 µg/ml for most of the organisms of clinical significance in the control and trikatu treated goats are given in Table-2. On the basis of the data for microorganisms possessing a minimum inhibitory concentration of 0.8 µg/ml, the priming dose of 7.0 mg/kg and maintenance dose of 2.59 mg/kg should be repeated at 8 h intervals. However, following trikatu administration the oral priming and maintenance doses would be 6.0 and 2.21 mg/kg, respectively and for practical purposes a priming dose of 6.0 mg/kg and maintenance dose of 2.0 mg/kg would be required to be administered following trikatu co-administration.
Discussion
Pefloxacin has excellent activity against a wide range of gram positive and gram negative microorganisms and it is being successfully used in the treatment of various human and animal diseases. Pefloxacin is partially metabolized in the liver to norfloxacin, which is a potent anti microbial agent [10]. Due to its favourable pharmacokinetic properties such as better bioavailability, better volume of distribution and minimal chances of resistance, it is one of the favoured anti bacterial agents for treatment of animal diseases. The irregular absorption from the gastrointestinal tract of goats limits its therapeutic utility. In goats only about 42% of the drug administered orally is available to the systemic circulation [8].
Trikatu is an ayurvedic preparation containing an equal ratio of long pepper (Piper longum), black pepper (Piper nigrum) and ginger (Zingiber officinale). Piperine (1-piperoyl piperidine), an amide alkaloid, from a different species of pepper is mainly responsible for enhancing the bioavailability of concurrently administered drugs [3,7,9]. Ginger in the preparation, may also aid the absorption of drugs through thermogenesis.
The observed plasma values of pefloxacin in goats by oral route were best described by a one compartment model. This model has also been used by previous workers to describe the pharmacokinetics of pefloxacin in various domestic species [8]. The findings of the present study indicated that plasma concentrations of the drug remained lower up to 2 h post drug administration with consistently significantly lower concentrations up to 20 min. In contrast, in our earlier studies in birds [7], higher plasma values of oxytetracycline were observed following Piper longum treatment during the absorption phase. The findings of the present study in goats therefore ruled out the possibility of vasodilatation of enteric vessels [2] during absorption. The findings of the present study agreed with those of Lala et al. [6] who also observed decreased plasma levels of declofenac sodium during the absorption phase in rabbits.
The decreased elimination of the drug was adequately supported by the higher values of t1/2β in the present study. However, the total body clearance (ClB) remained unaffected in the trikatu treated goats. Further, higher values of the apparent volume of distribution values based on the apparent volume of distribution based on the total area under the plasma drug concentration time curve [Vd(area)] indicated a better penetration of the drug in various body tissues. This was supported by the higher value for the apparent volume of distribution based on the zero time plasma concentration intercept of elimination phase [Vd(B)] The higher values of AUC, AUMC, and MRT in the piperine treated goats were indicative of enhanced systemic availability of pefloxacin following trikatu administration.
The pharmacokinetic profile of pefloxacin following trikatu administration revealed an enhancement of bioavailability. The enhanced bioavailability was not due to increased absorption but because of decreased elimination as evidenced by the higher values of t1/2β. The decreased elimination could either be due to suppression of drug metabolizing activities [12] or due to decreased renal excretion.
In the present study td and MRT were also indicative of enhanced systemic availability of pefloxacin in trikatu treated goats.
Based on the MIC data, only a marginal reduction in the dose (6-15%) was possible following trikatu administration. Nonetheless, trikatu administration enhanced the duration of antimicrobial action by about 22%.
Acknowledgments
We express our sincere thanks to the Dean, College of Veterinary and Animal Sciences, CSK Himachal Pradesh Agricultural University, Palampur- 176 062 (HP) India, for providing the necessary facilities to carry out the present investigation.
References
- 1.Abd El-Aty AM, Goudah A. Some pharmacokinetic parameters of pefloxacin in lactating goats. Vet Res Commun. 2002;26:553–561. doi: 10.1023/a:1020243615928. [DOI] [PubMed] [Google Scholar]
- 2.Annamalai AR, Manavalan R. Effects of Trikatu and its individual components and piperine on gastrointestinal tract: Trikatu - A bioavailability enhancer. Indian Drugs. 1989;27:595–604. [Google Scholar]
- 3.Atal CK. A breakthrough in drug bioavailability- a clue from age old wisdom of Ayurveda. Indian Drug Manuf Asso Bull. 1979;10:483–484. [Google Scholar]
- 4.Bennett JV, Brodie JL, Benner EJ, Kirby WM. Simplified, accurate method for antibiotic assay of clinical specimens. Appl Microbiol. 1966;14:170–177. doi: 10.1128/am.14.2.170-177.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gibaldi M, Perrier D. Pharmacokinetics. 2nd ed. New York: Marcel-Dekker; 1982. pp. 45–109. [Google Scholar]
- 6.Lala LG, D'Mello PM, Naik SR. Pharmacokinetic and pharmacodynamic studies on interaction of "Trikatu" with diclofenac sodium. J Ethnopharmacol. 2004;91:277–280. doi: 10.1016/j.jep.2003.12.027. [DOI] [PubMed] [Google Scholar]
- 7.Singh M, Varshneya C, Telang RS, Srivastava AK. Alteration of pharmacokinetics of oxytetracycline following oral administration of Piper longum in hens. J Vet Sci. 2005;6:197–200. [PubMed] [Google Scholar]
- 8.Malik JK, Rao GS, Ramesh S, Muruganandan S, Tripathi HC, Shukla DC. Pharmacokinetics of pefloxacin in goats after intravenous and oral administration. Vet Res Commun. 2002;26:141–149. doi: 10.1023/a:1014047702196. [DOI] [PubMed] [Google Scholar]
- 9.Mathur P, Valpandium T, Sengupta S, Gupta SK. Effect of piperine on analgesic activity of Nimesulide: a possible pharmacokinetic interaction. Indian J Pharm. 1998;30:204. [Google Scholar]
- 10.Montay G, Goueffon Y, Roquet F. Absorption, distribution, metabolic fate, and elimination of pefloxacin mesylate in mice, rats, dogs, mon keys, and humans. Antimicrob Agents Chemother. 1984;25:463–472. doi: 10.1128/aac.25.4.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Notari RE. Biopharmaceutics and clinical pharmacokinetics: an introduction. 4th ed. New York: Marcel-Dekker; 1987. pp. 221–270. [Google Scholar]
- 12.Reen RK, Wiebel FJ, Singh J. Piperine inhibits aflatoxin B1 induced cytotoxicity and genotoxicity in V79 Chinese hamster cells genetically engineered to express rat cytochrome P450 2B1. J Ethnopharmacol. 1997;58:165–173. doi: 10.1016/s0378-8741(97)00104-9. [DOI] [PubMed] [Google Scholar]
- 13.Sharma AK, Khosla R, Kela AK, Mehta VL. Fluoroquinolones: Antimicrobial agents of the 90's. Indian J Pharm. 1994;26:249–261. [Google Scholar]
- 14.Snedecor GW, Cochran WG. Statistical Methods. 6th ed. Ames: Iowa State University Press; 1967. pp. 59–65. [Google Scholar]
- 15.Srivastava AK, Dumka VK, Deol SS. Disposition kinetics and urinary excretion of pefloxacin after intravenous injection in crossbred calves. Vet Res Commun. 2000;24:189–196. doi: 10.1023/a:1006408415431. [DOI] [PubMed] [Google Scholar]