Abstract
Selecting the most efficient recombinant adeno-associated virus (rAAV) serotype for airway gene therapy has been difficult due to cross-specific differences in tropism and immune response between humans and animal models. Chimpanzees—the closest surviving genetic relative of humans—provide a valuable opportunity to select the most effective serotypes for clinical trials in humans. However, designing informative experiments using this protected species is challenging due to limited availability and experimental regulations. We have developed a method using Renilla luciferase (RL) and firefly luciferase (FL) reporters to directly index the relative transduction and immune response of two promising rAAV serotypes following lung coinfection. Analysis of differential luciferase activity in chimpanzee airway brushings demonstrated a 20-fold higher efficiency for rAAV1 over rAAV5 at 90 days, a finding that was similar in polarized human airway epithelia. T-cell responses to AAV5 capsid were stronger than AAV1 capsid. This dual vector indexing approach may be useful in selecting lead vector serotypes for clinical gene therapy and suggests rAAV1 is preferred for cystic fibrosis.
Introduction
The pursuit of a safe and effective gene therapy for cystic fibrosis (CF) has been impeded by the inability of existing preclinical models to accurately predict the efficiency of gene transfer in human airway epithelial cells or the magnitude of immune responses to gene transfer vectors. Many studies have used rodent or lagomorph species to predict in vivo efficacy of gene transfer and immune responses. A lesser, but significant, body of data has been generated with a variety of nonhuman primate species.1,2,3,4,5,6 These species are more closely related to humans genetically, yet they still vary widely in their relative permissiveness for a variety of viruses and viral vectors.
The use of differentiated airway epithelia cultured at an air–liquid interface and related techniques has allowed for rapid access to differentiated airway epithelial cells from a variety of rodent and primate species.7,8 These culture models reproduce many aspects of native airway cell morphology, gene expression, and function.9 Recently, differentiated airway epithelial cell cultures have been used to evaluate the relative efficiency of recombinant adeno-associated viral (rAAV) vectors across a range of species.10,11,12,13 Interestingly, recent studies have demonstrated disparate results between human airway epithelia and those derived from a variety of lower nonhuman primates and rodents.10,13 In particular, rAAV5-based vectors perform very well in mice and in differentiated airway cultures from mouse and lower primate species, whereas rAAV1-based vectors perform significantly better in human airway cultures. Preclinical studies in lower primates (monkey) using rAAV2 virus have also demonstrated clear efficacy for prolonged transgene expression in vivo in the lung3 and in vitro in monkey polarized airway epithelia.10 However, clinical trials using a rAAV2 vector have failed to give rise to transgene-derived mRNA in human CF lungs14 and also perform poorly in human polarized airway epithelia.10 This has raised questions about the manner in which clinical rAAV vectors are tested and chosen for human trials. Faced with an increasing number of rAAV serotypes,15 it is unclear what models may be best to predict clinical efficacy.
Chimpanzees are by far the closest genetic relatives to Homo sapiens (Figure 1). In fact, chimpanzees are more closely related to humans than they are to gorillas, and the relatedness to monkey species is much more distant. One might predict, therefore, that chimpanzees would serve as a superior model for predicting the behavior of viruses in humans than would other nonhuman primate species. Experience with viral pathogens would tend to substantiate that hypothesis. Specifically, important human pathogens such as hepatitis C virus and respiratory syncytial virus have only been successfully modeled in chimpanzees, in terms of their full infectious life cycle.16,17
Figure 1.
Illustration of a primate evolutionary tree. From this tree it is evident that chimpanzees are more closely related to modern humans than are any of the other primates. Chimpanzees and modern humans diverged from a common ancestor around 7 million years ago, this diagram also shows that chimpanzees are more closely related to humans than they are to any other primate including gorillas.
Based on these observations, we sought to determine whether we could validate the findings from differentiated human airway epithelial cell cultures, with regard to the relative efficiency of gene transfer among the most newly available rAAV serotypes that might be used for CF gene therapy. A wide range of studies have suggested that rAAV vectors crosspackaged or “pseudotyped” into capsids of rAAV5, rAAV1, or rAAV6 could be much more efficient than the original rAAV2 vectors for delivery of reporter genes or of CF transmembrane conductance regulator to the airway. We chose to compare rAAV5 and rAAV1, excluding rAAV6 from this analysis because it is so closely related to rAAV1.18 Because of the regulated nature of chimpanzee studies, requiring studies be done in a small number of animals without terminal procedures, we developed a dual reporter system which allows for comparative assessment of vector performance following coadministration of two different vector serotypes to the same animal. This approaches used two similar reporters [firefly luciferase (FL) and Renilla luciferase (RL)] driven from the same promoter and allowed for simultaneous detection within the same tissue biopsy. The findings presented here from these in vivo chimpanzee studies are consistent with those obtained from human differentiated airway cell cultures and strongly suggest that rAAV1 has significant advantages, both in terms of overall higher efficiency and lower immunogenicity.
Results
Validation of dual luciferase reporters in polarized human airway epithelia
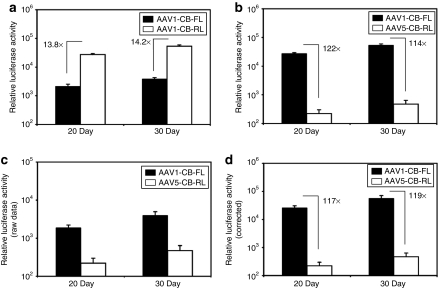
Before initiating studies in chimpanzees, it was important that the dual reporter vector approach be validated for accuracy in polarized human airway epithelia. The goal of these studies was to (i) determine differences in the specific activities of the FL and RL transgenes so direct comparisons could be made in a single sample, and (ii) to assure that coinfection of epithelia with two independent rAAV serotypes did not alter the efficiency of transduction of each serotype alone. To assess relative differences in the specific activity of the two luciferase reporters, rAAV1-CB-FL and rAAV1-CB-RL vectors were used to independently infect polarized human airway epithelia and the relative luciferase activities were assessed at 20 and 30 days after infection (Figure 2a). Results from these experiments demonstrated that RL was 14-fold more sensitive than FL. Hence, when comparing RL to FL activities within the same sample, a correction of 1 FL unit = 14 RL units was used.
Figure 2.
Validation of FL and RL dual reporter AAV1 and AAV5 vectors in primary human polarized airway epithelia. 5 × 109 particles of each of the indicated rAAV vectors were applied alone or in combination to the apical membrane of polarized human airway epithelia and luciferase expression was assayed at 20 and 30 days post-infection. 1/50th of the crude cell lysate was used for luciferase activity measurement. Data represents the mean (±SEM) relative luciferase activity from six independent infections. (a) rAAV1 vectors encoding the FL (AAV1-CB-FL) or RL (AAV1-CB-RL) reporters were independently infected into polarized airway epithelia. These findings were used to calculate the differences in specific activity between the two luciferase transgenes using an average of both 20 and 30 day time points; RL has a 14-fold higher sensitivity than FL (1 FL unit = 14 RL units). (b) The comparison of rAAV1 and rAAV5 transduction with a single-RL reporter. Human airway epithelia were infected independently with either AAV1-CB-RL or AAV5-CB-RL vectors and transgene expression was assessed at the indicated time points. (c,d) The dual-luciferase reporter assay was applied to compare the transduction efficiency following coinfection of human airway epithelia with AAV1-CB-FL and AAV5-CB-RL. The raw data for relative light units from FL and RL measurement are shown in c and the corrected relative luciferase activities from the two different vectors are shown in d using a correction of 1 FL unit = 14 RL units as determined in a. Fold differences (~120-fold) in the efficiency of transduction for rAAV1 and rAAV5 vectors using the dual reporter assays (d) closely matched the data from the single reporter assays shown in b. FL, firefly luciferase; rAAV, recombinant adeno-associated virus; RL, Renilla luciferase.
We next sought to evaluate the differences in efficiency of rAAV1 and rAAV5 vectors to transduce polarized human airway epithelia using the same RL reporter encoded into each vector serotype (Figure 2b). Results from these experiments demonstrated that rAAV1 was ~120-fold more effective at transducing human airway epithelia in comparison to rAAV5. Such differences are similar to previous comparisons of rAAV1 and rAAV5 using a FL reporter.11 Lastly, we tested whether this 120-fold difference in efficiency would be similarly obtained using the dual reporter vectors proposed for use in the chimp study. In this context, primary human airway epithelia were coinfected with rAAV1-CB-FL and rAAV5-CB-RL vectors (the same vectors used for the chimp studies). Figure 2c shows the raw data from these experiments, whereas Figure 2d shows the data corrected for the 14-fold difference in the specific activity of the two luciferase reporters. Results from these dual reporter experiments demonstrated an ~120-fold higher efficiency of rAAV1 as compared to rAAV5, a finding that closely mirrored results of single infection studies with the two serotypes using the same RL reporter (Figure 2b). These findings demonstrated that indeed the dual reporter system could be used to accurately assess relative transduction of two independent rAAV serotypes in the same sample.
Direct comparison of efficiency of rAAV1 and rAAV5 vectors in the chimpanzee airway in vivo
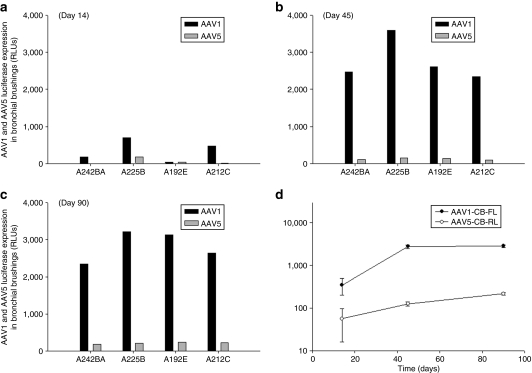
As in vitro polarized airway epithelia cannot fully model all aspects of in vivo gene deliver to the lung, we sought to assess differences in rAAV1 and rAAV5 transduction in four adult chimpanzees using the dual reporter system. A single mixture containing 1 × 1013 vg particles of each rAAV1-CB-FL and rAAV5-CB-RL virus was administered to chimpanzees by fiberoptic bronchoscopy with direct aerosol administration, using a Penn–Century MicroSprayer device (Penn–Century, Philadelphia, PA) positioned in the distal trachea. Bronchial brush biopsies were obtained before vector administration and on days 14, 45, and 90 after vector administration. Protein extracts of the brush biopsy samples were then assayed for both FL and RL activities. By day 14 after vector administration, a significant elevation of luciferase activity from each vector was noted, with rAAV1 rising faster than rAAV5 (Figure 3). By day 45, an ~20-fold greater level of rAAV1 transduction over rAAV5 was observed and this difference in luciferase expression persisted to day 90, which was the last point at which biopsies were taken (Figure 3).
Figure 3.
In vivo direct comparison AAV1 and AAV5 vectors in chimpanzee airways after aerosol delivery with a FL and RL dual reporter assay. The airways from four chimpanzees were aerosolized with a mixture of AAV1-CB-FL (1 × 1013 vg) and AAV5-CB-RL (1 × 1013 vg) with the use of a bronchoscopic microsprayer. Bronchial brushings were taken at (a) day 14, (b) day 45, and (c) day 90 to determine expression levels with a dual reporter luciferase assay. (c) Time course showing average luciferase activity for AAV1 and AAV5 expression in the four chimpanzees ± SEM. FL, firefly luciferase; AAV, adeno-associated virus; RL, Renilla luciferase; RLU, relative luminescence unit.
Immune responses to AAV capsid components
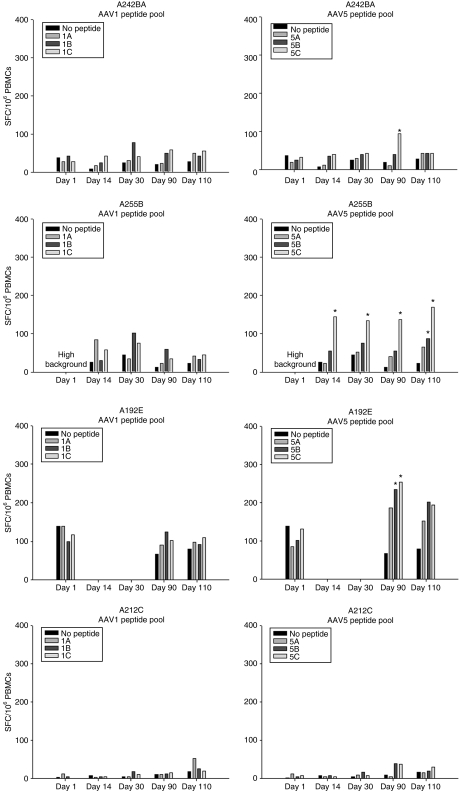
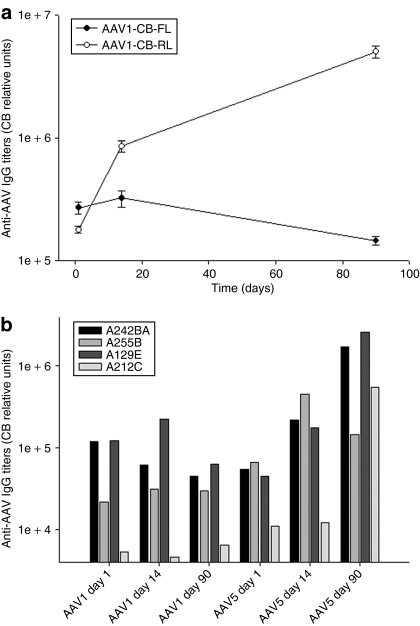
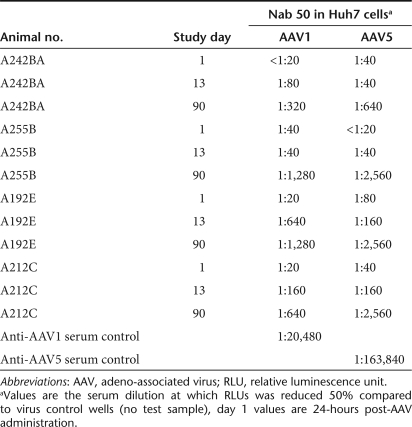
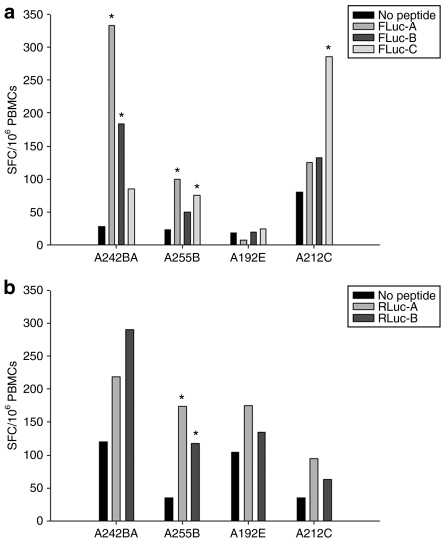
In order to determine whether there were serotype specific differences in the immune responses to either of the AAV capsids, antibodies to each were determined by enzyme-linked immunosorbent assay and neutralizing antibody assay, and cellular responses were measured using a interferon-γ enzyme-linked immunosorbent spot (ELISPOT) assay. There was a clear difference noted between the serotypes in terms of the cellular responses to AAV capsids. AAV5 elicited a significant ELISPOT response in three of the four chimpanzees (Figure 4). Two of the chimpanzees demonstrated transient responses; one persisted from days 14 to 110, which was the last time point for these assays. In contrast, zero of the four animals responded to AAV1 capsid peptide pools (Figure 4). Similarly, total and neutralizing antibody responses were more robust against AAV5 capsid in comparison to AAV1 capsid components. These differences were sustained through day 90, which was the last time point for which data is available (Figure 5 and Table 1). In order to determine whether differences in immunogenicity to the transgene could account for the observed differences in expression, an interferon-γ ELISPOT on peripheral blood mononuclear cells (PBMCs) at day 110 post-delivery was performed using either a FL or RL peptide library. Whereas AAV1 capsid did not elicit a positive response as shown above, three out of four chimpanzees mounted positive responses to one or more of the firefly peptide pools, while the Renilla gene delivered by rAAV5 only caused a significant response in one out of four chimps (Figure 6). This suggests that immune clearance targeting the expressed transgene product could not account for the lower expression observed with rAAV5, however, it remains unclear whether the rAAV1 delivered firefly is indeed more immunogenic or whether the higher expression of the transgene that ensued post-transduction may have positioned it for a more robust immune response.
Figure 4.
Representative ex vivo IFN-γ ELISPOT assay results. Representative raw data is shown in terms of the numbers of SFCs per million PBMCs at each of the time points indicated. Each graph represents PBMCs from a single chimpanzee stimulated with either an AAV1 or AAV5 peptide pool as shown in the legend. PBMCs were subjected to an IFN-γ ELISPOT assay by stimulation with the respective AAV capsid pools or medium alone (no peptide). Each set of asterisks indicate responses that are statistically above background. Samples were compared using Student's t-tests, *P ≤ 0.05. (*Indicate responses that are over threefold of response from medium control). AAV, adeno-associated virus; IFN-γ ELISPOT, interferon-γ enzyme-linked immunosorbent spot; PBMC, peripheral blood mononuclear cell; SFC, spot forming cell.
Figure 5.
Humoral responses to AAV1 and AAV5 capsids after airway aerosol delivery. Total serum antibody levels to either AAV1 or AAV5 whole capsids were assayed by an indirect ELISA after coating plates with either AAV1 or AAV5 viral particles (a) group averages (b) individual chimpanzee values. AAV, adeno-associated virus; ELISA, enzyme-linked immunosorbent assay; IgG, immunoglobin G.
Table 1.
Neutralizing antibodies to AAV1 and AAV5 in Huh7 cells
Figure 6.
Representative ex vivo IFN-γ ELISPOT assay results against transgene peptide library. Representative raw data is shown in terms of the numbers of SFCs per million PBMCs at day 110 post-rAAV delivery. PBMCs were subjected to an IFN-γ ELISPOT assay by stimulation with (a) the firefly luciferase or (b) Renilla luciferase peptide pool. Each set of asterisks indicate responses that are statistically above background (no peptide). Samples were compared using Student's t-tests, *P < 0.05. (*Indicate responses that are over threefold of response from medium control). AAV, adeno-associated virus; IFN-γ ELISPOT, interferon-γ enzyme-linked immunosorbent spot; PBMC, peripheral blood mononuclear cell; SFC, spot forming cell.
Discussion
The results presented here demonstrate clear advantages for rAAV1 over rAAV5 in the chimpanzee airway, both in terms of a superior level of gene transfer and reduced immunogenicity of the rAAV1 capsid. Our working hypothesis is that these two findings are independent of one another. If the differences in transduction were due solely to the lack of effector T-cell responses to rAAV1, we would not have expected the findings to mirror those observed in the human airway epithelial cell cultures, where no immune cells are present. If these do represent two independent advantages, they would both operate in favor of improved efficacy of rAAV1-based vectors for gene therapy of CF.
The work represented here in chimpanzees is validated by studies of these serotypes in well-differentiated human airway epithelia, suggesting that both human polarized airway epithelia and the dual reporter coinfection approach can predict efficacy of rAAV vectors in vivo. For example, in vivo analysis demonstrated a ~20-fold higher level of rAAV1 transduction over rAAV5, whereas in vitro analysis in human airway epithelia demonstrated an ~120-fold differential (rAAV1 > rAAV5).
Previous studies have suggested that heparin binding can direct activation of T cells against AAV2 capsid.19 Because AAV1 and AAV5 do not bind heparin,20,21 one might have predicted that both serotypes would elicit a lesser effector T-cell response. Given the higher T-cell response found against AAV5 capsids, these findings suggest that alternative nonheparin binding epitopes also influence serotype-specific T-cell responses in higher primates.
As with respiratory syncytial virus, the in vivo behavior of respiratory syncytial virus in chimpanzees was consistent with that observed in human cells, whereas the behavior in lower primate airway cells was not.16 This discrepancy could be due to differences in the sequence, structure, or distribution of cell surface proteins and glycoproteins that might serve as receptors for the various serotypes.
As the number of AAV serotypes available for use as vectors continues to increase, it is essential that model systems be evaluated as to their predictive value with regard to the ultimate safety and efficacy of each vector in humans. In order to truly fulfill these characteristics, a model system must ultimately be shown to yield results that are consistent with late phase clinical trial results. Based on that reasoning, the predictive utility of the chimpanzee model in evaluating new AAV serotypes will not truly be known until such clinical data becomes available with serotype vectors that have gone through chimpanzee-based testing. Until that time, the consistency between in vivo results in chimpanzees and results in human cell culture model systems, suggest a reasonable likelihood of a high predictive value for these models.
Materials and Methods
Plasmid construction and viral packaging. pAV-CMV-flag-luc containing AAV2 ITR and flag-tagged FL reporter was obtained from Targeted Genetics (Seattle, WA). The cytomegalovirus promoter in pAV-CMV-flag-luc was replaced with the cytomegalovirus enhancer linked β-actin promoter segment from pTR2CB-cftrD264, the resultant AAV proviral plasmid was designated as pAV2-CBA-flag-FL. RL gene was derived from the plasmid pSVRL (Promega, Madison, WI). An HA-tag was fused in-framed to the N-terminus of the RL gene by PCR cloning. The RL AAV proviral vector (called pAV2-CBA-HA-Rlplus) was constructed by replacing flag-tagged FL gene with the HA-tagged RL in pAV2-CBA-flag-FL, together with a 750-bp SacI segment from LacZ gene as stuffer. Incorporation of the stuffer was used to keep the viral genome of the Renilla reporter virus similar in length with that of FL vector.
Vectors were packaged and purified using a standard co-transfection technique in human embryonic kidney-293 cells and produced as previously described.22 Briefly, the vectors were produced via calcium phosphate based co-transfection of plasmid DNA into human embryonic kidney-293 cells. Following the transfection reaction, the cells were incubated for ~66 hours at 37 °C, 5% CO2, and 85% relative humidity. The cells were harvested following the incubation period and subjected to three freeze/thaw cycles (freezing in dry ice/ethanol and thawing at 37 °C). The resultant cell lysates were treated with magnesium and benzonase then incubated for 30 minutes at 37 °C. After the benzonase digest the lysates were centrifuged at 4,000 g for 20 minutes to remove cellular debris.
The partially clarified lysates were applied to the top of the iodixanol step gradient and ultracentrifuged at 350,000 g for 1 hour at 18 °C. Approximately 5 ml of the gradient at the 60–40% interface is retrieved by way of side-puncture of the centrifugation tube and aspiration using a syringe. The iodixanol aspirates were pooled and diluted 1:1 with a low salt wash buffer to decrease the high viscosity of the iodixanol solution before application to the chromatographic media. Once diluted and following column equilibration the sample was loaded onto an GE HiTrap column containing “Q Sepharose High Performance” anion exchange media (5-ml bed volume), at a volumetric flow rate of 5 ml/min. Immediately after the sample is completely loaded the column, it is washed with 10 column volumes of low salt buffer. The virus containing fraction is eluted by increasing the sodium chloride concentration to 350 mmol/l. The eluent (sublot) was collected and tested for purity and titer by polyacrylamide gel electrophoresis/silver stain and dot blot before pooling. The sublots were stored at −80 °C until all sublots were completed and tested.
The sublots were pooled before buffer exchange and concentration. The buffer exchange and concentrations were preformed using “high-performance centrifugal concentrators”. The buffer exchange continued until an estimated 99.4% exchange into the final stock buffer. Once the buffer exchange and concentration was complete the final stock was sterile filtered by passing through a 0.22-µm syringe filter before final fill of the vials used for QC testing and the study material.
Infection of polarized airway epithelia. Polarized human airway epithelia were generated from lung transplant airway tissue by growing airway epithelia on 12-mm Millicell membrane inserts (Millipore, Bedford, MA) followed by differentiation at an air–liquid interface as previously described.11 5 × 109 particles of rAAV1 and/or rAAV5 were diluted in culture medium to a 50-µl volume and used to infect each Millicell insert from the apical membrane for a 16-hour period. Following infection, the media and virus were removed and cultures were returned to an air–liquid interface with fresh basolateral media. Luciferase assays were performed at 20–30 days post-infection. Several vector combinations were used to standardize the specific activity of the RL and FL reporters. In this context, rAAV1-based vectors were generated for both the RL (rAAV1-CB-RL) and FL reporters (rAAV1-CB-FL). Independent and coinfection with rAAV1-CB-RL, rAAV5-CB-RL, and/or rAAV1-CB-FL was used to determine the specific activity of the luciferase reporters and also confirm that coinfection with the two serotypes did not alter transduction efficiency. A second set of experiments was designed to mimic the preclinical trail in chimpanzees. In this context, rAAV1-CB-FL and rAAV5-CB-RL vectors were used for coinfection of human airway epithelia under the conditions described above and these results were compared to those following individual infections with each vector. It should also be noted that we found no evidence for crossreactivity of the luciferin substrates for RL and FL. This was assessed in human airway epithelia infected individually with the two reporter viruses and assessed for both RL and FL enzymatic activities.
Animal care approvals and oversight. This study was conducted in accordance with all applicable animal care guidelines at an American Association for Accreditation of Laboratory Animal Care accredited facility, and was preapproved by the University of Louisiana at Lafayette Institutional Animal Care and Use Committee and Institutional Biosafety Committee. All animals were housed with appropriate social stimulation and veterinary oversight.
rAAV aerosol airway delivery and sample collection. Directed vector delivery to the tracheobronchial tree was accomplished as previously described.1 Briefly a MicroSprayer (Penn–Century) inserted through a 3.5-mm flexible fiberoptic bronchoscope (Olympus, Melville, NY) using universal precautions and general anesthetic techniques. The chimpanzees were first sedated with general anesthesia with isoflurane, intubated with a 5.0-mm endotracheal tube, and given supplemental oxygen. Lidocaine (1%) was instilled onto the carina to minimize coughing. The MicroSprayer was advanced ~3 mm beyond the tip of the bronchoscope to visualize aerosolization of the vector into the main-stem bronchi.
At serial time points, brushes were collected from each animal for luciferase assays. For sampling the airway epithelium, standard Olympus bronchoscopy brushes were passed through the suction port of the bronchoscope. These bronchoscopy brushes were used for sampling of epithelial cells within the bronchial lumen by abrading the airway epithelium, previously exposed to vector and under direct vision, and then the brushes were placed immediately into transport medium for analysis.
Dual luciferase assay. Polarized cultured epithelial cells or brushed chimpanzee bronchial samples were lysed in 200 µl of Passive Lysis Buffer (Promega). FL and RL activities in the cell lysates were measured with the Promega Dual-luciferase Reporter Assay System in 20/20 luminometer (Turner BioSystem, Sunnyvale, CA) following manufacturer's instruction. The time points for chimpanzee sample assays were 14, 34, and 90 days post-AAV infection and the predosing negative control samples were collected 1 day before aerosol delivery of AAV.
Anti-AAV antibody enzyme-linked immunosorbent assay. Anti-AAV1 and anti-AAV5 antibodies were assayed using a previously published enzyme-linked immunosorbent assay method,23 modified for use on chimpanzee serum by substituting an appropriate rabbit antihuman immunoglobin G secondary antibody. AAV-neutralizing antibodies were assayed as previously described.24
Blood sample collection, PBMC isolation, and in vitro culture of PBMCs. The blood samples were collected through venipuncture using heparin as anticoagulant and shipped overnight to the testing lab at the University of Pennsylvania. The PBMCs were isolated using Ficoll–Paque gradient centrifugation (GE-Amersham Biosciences, Pittsburgh, PA). All samples were processed upon receiving and evaluated in ELISPOT assays as fresh samples.
Interferon-γ ELISPOT assay. Interferon-γ ELISPOT assays were performed using previously described protocols.25 PBMCs were added at the density of 2 × 105 cells/well and stimulated for 18–20 hours at 37 °C with peptide libraries derived from the AAV1 or AAV5-VP1 protein at the concentration of 2 µg/ml. Medium alone served as a negative control, and phytohemagglutinin as a positive control. The peptide libraries were synthesized as 15-mers with 10-amino-acid overlap with the preceding peptide (Mimotopes, Raleigh, NC). The AAV1-VP1 peptide library was divided into three pools such that pool 1A contains the first 50 peptides of AAV1 VP1, pool 1B contains peptides 51–100 and pool 1C contains peptides 101–146. The same scheme was applied to the AAV5-VP1 peptide library. Spots were counted with an ELISPOT reader (AID, San Diego, CA). Peptide-specific cells were represented as spot forming cells per 106 PBMCs. A positive response to any peptide pool was arbitrarily defined as three times over background (medium alone control) and the response was over 55 spot forming cell per 106 PBMCs.
Acknowledgments
This work was supported by NIH HL58340 (J.F.E.), DK54759 (J.F.E.), and a sponsored research grant from Targeted Genetics Corporation (J.F.E.). J.F.E. and Z.Y. do not hold financial interests in Targeted Genetics Corporation and do not receive any form of personal financial income from this company.
REFERENCES
- Fischer AC, Beck SE, Smith CI, Laube BL, Askin FB, Guggino SE, et al. Successful transgene expression with serial doses of aerosolized rAAV2 vectors in rhesus macaques. Mol Ther. 2003;8:918–926. doi: 10.1016/j.ymthe.2003.08.015. [DOI] [PubMed] [Google Scholar]
- Engelhardt JF, Simon RH, Yang Y, Zepeda M, Weber-Pendleton S, Doranz B, et al. Adenovirus-mediated transfer of the CFTR gene to lung of nonhuman primates: biological efficacy study. Hum Gene Ther. 1993;4:759–769. doi: 10.1089/hum.1993.4.6-759. [DOI] [PubMed] [Google Scholar]
- Conrad CK, Allen SS, Afione SA, Reynolds TC, Beck SE, Fee-Maki M, et al. Safety of single-dose administration of an adeno-associated virus (AAV)-CFTR vector in the primate lung. Gene Ther. 1996;3:658–668. [PubMed] [Google Scholar]
- Afione SA, Conrad CK, Kearns WG, Chunduru S, Adams R, Reynolds TC, et al. In vivo model of adeno-associated virus vector persistence and rescue. J Virol. 1996;70:3235–3241. doi: 10.1128/jvi.70.5.3235-3241.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernandez YJ, Wang J, Kearns WG, Loiler S, Poirier A., and , Flotte TR. Latent adeno-associated virus infection elicits humoral but not cell-mediated immune responses in a nonhuman primate model. J Virol. 1999;73:8549–8558. doi: 10.1128/jvi.73.10.8549-8558.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirmule N, Xiao W, Truneh A, Schnell MA, Hughes JV, Zoltick P, et al. Humoral immunity to adeno-associated virus type 2 vectors following administration to murine and nonhuman primate muscle. J Virol. 2000;74:2420–2425. doi: 10.1128/jvi.74.5.2420-2425.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bals R, Xiao W, Sang N, Weiner DJ, Meegalla RL., and , Wilson JM. Transduction of well-differentiated airway epithelium by recombinant adeno-associated virus is limited by vector entry. J Virol. 1999;73:6085–6088. doi: 10.1128/jvi.73.7.6085-6088.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gruenert DC, Finkbeiner WE., and , Widdicombe JH.1995Culture and transformation of human airway epithelial cells Am J Physiol 2683 Pt 1): L347–L360. [DOI] [PubMed] [Google Scholar]
- Bernacki SH, Nelson AL, Abdullah L, Sheehan JK, Harris A, Davis CW, et al. Mucin gene expression during differentiation of human airway epithelia in vitro. Muc4 and muc5b are strongly induced. Am J Respir Cell Mol Biol. 1999;20:595–604. doi: 10.1165/ajrcmb.20.4.3442. [DOI] [PubMed] [Google Scholar]
- Liu X, Luo M, Trygg C, Yan Z, Lei-Butters DC, Smith CI, et al. Biological Differences in rAAV Transduction of Airway Epithelia in Humans and in Old World Non-human Primates. Mol Ther. 2007;15:2114–2123. doi: 10.1038/sj.mt.6300277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Z, Lei-Butters DC, Liu X, Zhang Y, Zhang L, Luo M, et al. Unique biologic properties of recombinant AAV1 transduction in polarized human airway epithelia. J Biol Chem. 2006;281:29684–29692. doi: 10.1074/jbc.M604099200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seiler MP, Miller AD, Zabner J., and , Halbert CL. Adeno-associated virus types 5 and 6 use distinct receptors for cell entry. Hum Gene Ther. 2006;17:10–19. doi: 10.1089/hum.2006.17.10. [DOI] [PubMed] [Google Scholar]
- Liu X, Yan Z, Luo M., and , Engelhardt JF. Species-specific differences in mouse and human airway epithelial biology of recombinant adeno-associated virus transduction. Am J Respir Cell Mol Biol. 2006;34:56–64. doi: 10.1165/rcmb.2005-0189OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss RB, Rodman D, Spencer LT, Aitken ML, Zeitlin PL, Waltz D, et al. Repeated adeno-associated virus serotype 2 aerosol-mediated cystic fibrosis transmembrane regulator gene transfer to the lungs of patients with cystic fibrosis: a multicenter, double-blind, placebo-controlled trial. Chest. 2004;125:509–521. doi: 10.1378/chest.125.2.509. [DOI] [PubMed] [Google Scholar]
- Gao G, Vandenberghe LH., and , Wilson JM. New recombinant serotypes of AAV vectors. Curr Gene Ther. 2005;5:285–297. doi: 10.2174/1566523054065057. [DOI] [PubMed] [Google Scholar]
- Crowe JE Jr, Collins PL, London WT, Chanock RM., and , Murphy BR. A comparison in chimpanzees of the immunogenicity and efficacy of live attenuated respiratory syncytial virus (RSV) temperature-sensitive mutant vaccines and vaccinia virus recombinants that express the surface glycoproteins of RSV. Vaccine. 1993;11:1395–1404. doi: 10.1016/0264-410x(93)90168-w. [DOI] [PubMed] [Google Scholar]
- Bukh J. A critical role for the chimpanzee model in the study of hepatitis C. Hepatology. 2004;39:1469–1475. doi: 10.1002/hep.20268. [DOI] [PubMed] [Google Scholar]
- Wu Z, Miller E, Agbandje-McKenna M., and , Samulski RJ. Alpha2,3 and alpha2,6 N-linked sialic acids facilitate efficient binding and transduction by adeno-associated virus types 1 and 6. J Virol. 2006;80:9093–9103. doi: 10.1128/JVI.00895-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenberghe LH, Wang L, Somanathan S, Zhi Y, Figueredo J, Calcedo R, et al. Heparin binding directs activation of T cells against adeno-associated virus serotype 2 capsid. Nat Med. 2006;12:967–971. doi: 10.1038/nm1445. [DOI] [PubMed] [Google Scholar]
- Chen S, Kapturczak M, Loiler SA, Zolotukhin S, Glushakova OY, Madsen KM, et al. Efficient transduction of vascular endothelial cells with recombinant adeno-associated virus serotype 1 and 5 vectors. Hum Gene Ther. 2005;16:235–247. doi: 10.1089/hum.2005.16.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zabner J, Seiler M, Walters R, Kotin RM, Fulgeras W, Davidson BL, et al. Adeno-associated virus type 5 (AAV5) but not AAV2 binds to the apical surfaces of airway epithelia and facilitates gene transfer. J Virol. 2000;74:3852–3858. doi: 10.1128/jvi.74.8.3852-3858.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zolotukhin S, Potter M, Zolotukhin I, Sakai Y, Loiler S, Fraites TJ, Jr, et al. Production and purification of serotype 1, 2, and 5 recombinant adeno-associated viral vectors. Methods. 2002;28:158–167. doi: 10.1016/s1046-2023(02)00220-7. [DOI] [PubMed] [Google Scholar]
- Hauswirth WW, Aleman TS, Kaushal S, Cideciyan AV, Schwartz SB, Wang L, et al. Treatment of leber congenital amaurosis due to RPE65 mutations by ocular subretinal injection of adeno-associated virus gene vector: short-term results of a phase I trial. Hum Gene Ther. 2008;19:979–990. doi: 10.1089/hum.2008.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin J, Calcedo R, Vandenberghe LH, Figueredo JM., and , Wilson JM. Impact of preexisting vector immunity on the efficacy of adeno-associated virus-based HIV-1 Gag vaccines. Hum Gene Ther. 2008;19:663–669. doi: 10.1089/hum.2008.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu TM, Dubey SA, Mehrotra DV, Freed DC, Trigona WL, Adams-Muhler L, et al. Evaluation of cellular immune responses in subjects chronically infected with HIV type 1. AIDS Res Hum Retroviruses. 2007;23:67–76. doi: 10.1089/aid.2006.0114. [DOI] [PubMed] [Google Scholar]