Abstract
Reelin is an extracellular matrix protein synthesized in cerebellar granule cells that plays an important role in Purkinje cell positioning during cerebellar development and in modulating adult synaptic function. In the cerebellum of schizophrenia (SZ) and bipolar (BP) disorder patients, there is a marked decrease (≈50%) of reelin expression. In this study we measured Purkinje neuron density in the Purkinje cell layer of cerebella of 13 SZ and 17 BP disorder patients from the McLean 66 Cohort Collection, Harvard Brain Tissue Resource Center. The mean number of Purkinje neurons (linear density, neurons per millimeter) was 20% lower in SZ and BP disorder patients compared with nonpsychiatric subjects (NPS; n = 24). This decrease of Purkinje neuron linear density was unrelated to postmortem interval, pH, drugs of abuse, or to the presence, dose, or duration of antipsychotic medications. A comparative study in the cerebella of heterozygous reeler mice (HRM), in which reelin expression is down-regulated by ≈50%, showed a significant loss in the number of Purkinje cells in HRM (10–15%) compared with age-matched (3–9 months) wild-type mice. This finding suggests that lack of reelin impairs GABAergic Purkinje neuron expression and/or positioning during cerebellar development.
Keywords: bipolar disorder, granular cells, glutamic acid decarboxylase, schizophrenia
A number of behavioral, neuropathologic, neuroimaging, and neurochemical studies suggest that the cerebellum may be defective in schizophrenia (SZ) (for a review see ref. 1). We (2, 3) and others (4) have reported that cerebella from SZ and bipolar (BP) disorder patients exhibit a 50% decrease in the expression of glutamic acid decarboxylase 67 (GAD67), which is a major GABA synthesizing enzyme present in cerebellar Purkinje neurons. This decrease suggests that these GABAergic principal neurons may be damaged. We also reported that in the cerebellar glutamatergic granule cells of SZ and BP disorder patients, there is a marked decrease of reelin mRNA levels (2, 3).
A decrease of reelin levels in the cerebellum of SZ patients has been confirmed in other studies (4). In addition, the reduction of reelin mRNA is negatively correlated with the expression of the transcription factor semaphorin 3A in the cerebella of subjects with SZ (5).
Reelin is a large (≈400 kDa) extracellular matrix protein synthesized by GABAergic interneurons in the telencephalon and by glutamatergic granule cells in the cerebellum (6–9). It is secreted extracellularly by constitutive mechanisms (10) and is believed to play a significant role in glutamate receptor homeostasis and spine formation in the adult brain (11–14).
The heterozygous reeler mouse (HRM), which expresses ≈50% of the amount of reelin present in brains of wild-type mice (WTM), displays certain behavioral and anatomic abnormalities reminiscent of those found in SZ patients. These include (i) prepulse inhibition deficits, which are a measure of sensory motor gating of the CNS (15), (ii) contextual fear conditioning deficits, which are a measure of a hippocampal/amygdala-dependent function (16), and (iii) long-term potentiation amplitude and maintenance (17). HRM also exhibit a down-regulation of GAD67 expression and GABA turnover rate in several brain areas and an increase of neural packing density (13, 18, 19). Furthermore, HRM exhibit a marked reduction in dendritic spine density in the frontal cortex and hippocampal pyramidal neurons (11, 19, 20).
In HRM there is also evidence of a deficit in the number of cerebellar Purkinje neurons compared with age-matched WTM (21). Hence, we inferred that in the mammalian cerebellum, reelin, which is synthesized by granule cells and secreted extracellularly at the axon terminals of the parallel fibers, adheres to integrin or very-low-density lipoproteins (VLDL) and apolipoprotein E2 (APO-E2) receptors located on Purkinje neuron cell bodies and dendrites. Thus, reelin stimulates dendritic protein synthesis, promotes dendritic spine expression and maturation, and determines Purkinje neuron positioning in the Purkinje cell layer (11, 22).
Because reelin is markedly decreased in various telencephalic areas and in the cerebellum of SZ and BP disorder patients (2, 3, 8, 9), here we studied whether Purkinje neurons are decreased in the cerebellar Purkinje cell layer of these patients.
In addition, to support the concept that the reduced number of cerebellar Purkinje neurons in SZ and BP patients is related to reelin down-regulation, we have confirmed earlier findings in HRM (21) that linked reelin haploinsufficiency to a reduction in cerebellar Purkinje neurons.
Results
Density of Cerebellar Purkinje Neurons Is Reduced in SZ and BP Disorder Patients.
Table 1 describes the demographic characteristics of the patient cohort used in this study. The cohort included nonpsychiatric subjects (NPS), SZ, and BP disorder patients. By applying Fisher’s exact test, it was established that SZ and BP disorder patient groups did not differ significantly from the NPS group in age, gender, postmortem interval (PMI), fixation date, or brain pH. Measurements of the number of Purkinje neurons (linear density, cells per millimeter) in the cerebellum from these three cohorts showed a statistically significant decrease (≈20%) in the SZ and BP disorder groups compared with the NPS group (Table 2).
Table 1.
Demographic characteristics of brain samples*† obtained from the Harvard Brain Tissue Resource Center
| Patient cohort |
|||
| Characteristic | NPS (n = 24) | SZP (n = 13) | BP (n = 17) |
| Male/female | 17/7 | 10/3 | 10/7 |
| Hemisphere (left/right) | 9/15 | 4/9 | 9/8 |
| Age (y) | 60 ± 17 | 56 ± 19 | 60 ± 18 |
| Postmortem interval (h) | 20 ± 6.1 | 22 ± 5.0 | 20 ± 9.8 |
| Fixation days × 103 | 1.8 ± 0.6 | 2.2 ± 0.4 | 2.0 ± 0.6 |
| Brain pH | 6.4 ± 0.2 | 6.4 ± 0.2 | 6.5 ± 0.2 |
| Age of illness onset (y) | — | 23 ± 11.1 | 37 ± 16 |
| Duration of illness (y) | — | 33 ± 19 | 26 ± 18 |
| Suicide | 0 | 1 (8) | 2 (12) |
| Abuse or dependence | 0 | 6 (47) | 8 (47) |
| CNS medications‡ | |||
| Atypical antipsychotics | — | 5 (46) | 6 (43) |
| Typical antipsychotics | — | 5 (46) | 3 (21) |
| Typical and atypical | — | 1 (9) | 0 |
| Mood stabilizers | — | 1 (9) | 11 (79) |
| Antidepressants | — | 4 (36) | 5 (36) |
Values are mean ± SD or number (percentage)
*Formaldehyde-fixed cerebellum.
†The psychiatric diagnoses were established by two senior psychiatrists on the basis of clinical and family histories and according to Feighner et al. (41) for SZ and The Diagnostic and Statistical Manual of Mental Disorders IV criteria for BP.
‡There was only one subject taking both typical and atypical antipsychotics in the SZ group, and this was not included in the medication categories of typical or atypical. The rows “typical, “atypical,” and “typical and atypical” in this table are mutually exclusive. In the SZ group there were one subject taking both mood stabilizer and atypical antipsychotics; two taking both antidepressant and typical antipsychotics; two taking both antidepressant and atypical antipsychotics; and one taking antidepressant and typical and atypical antipsychotics. No subject was taking both mood stabilizers and antidepressants. In the BP group, there were three subjects taking both mood stabilizers and typical antipsychotics; three subjects taking both mood stabilizers and atypical antipsychotics; one taking both antidepressant and typical antipsychotics; two taking both antidepressants and atypical antipsychotics; and four subjects taking both mood stabilizers and antidepressants.
Table 2.
Reduced cerebellar Purkinje neuron density in SZ and BP disorder patients
| Subjects | Purkinje neuron linear density (neurons/mm) |
| NPS (n = 24) | 4.8 ± 0.56 |
| SZ (n = 13) | 3.9 ± 0.50* |
| BP (n = 17) | 4.0 ± 0.64* |
Values are mean ± SD. For statistical analysis we used a mixed model (F2.422 = 32.45; P < 0.0001), followed by Tukey comparisons.
*NPS vs. SZ (P < 0.001) and NPS vs. BP (P < 0.001).
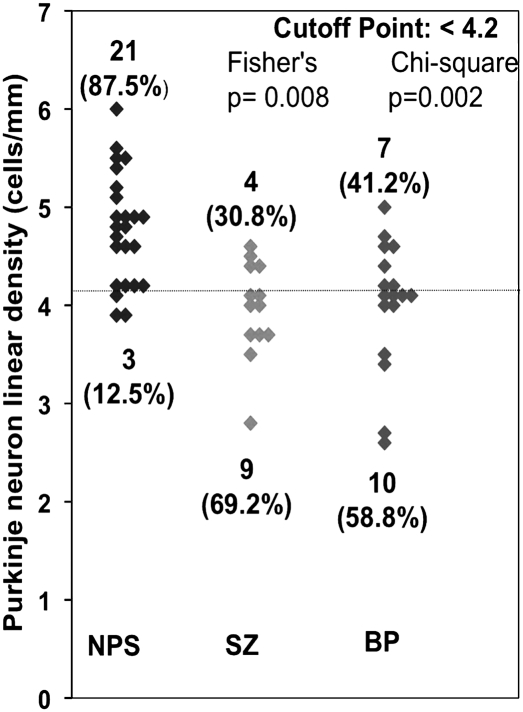
As shown in Fig. 1, an analysis of SZ and BP disorder patient cohorts using the χ2 or Fisher exact test showed that 30.8% (P = 0.0008) of SZ patients and 41.2% (P = 0.002) of BP disorder patients had Purkinje neuron linear density equal to or slightly higher than 4.2, whereas the Purkinje neuron linear density was above this level in 87.5% of NPS. Our mixed models on Purkinje neuron linear density failed to find that background variables had an effect on Purkinje neuron linear density, except for the difference in diagnosis. This included age [F1,421 = 1.51, nonsignificant (NS) when treated as continuous variable; F2,420 = 1.61, NS when treated as categoric variable], gender (F1,421 = 2.61, NS), PMI (F1,421 = 2.75, NS), hemisphere (F1,421 = 1.56, NS), brain pH (F1,421 = 3.32, NS), suicide (F1,421 = 0.31, NS), age of onset of illness (F1,214 = 0.24, NS), other cause of death (F4,410 = 2.09, NS), or type of medication (F3,188 = 1.01, NS).
Fig. 1.
Plot of Purkinje cell number from non-psychiatric subjects (NPS) (n = 24), schizophrenia (SZ) (n = 13) and bipolar (BP) disorder (n = 17) patients. In parentheses is the percentage of subjects with Purkinje cell number above and below cutoff point of 4.2. Note that 30.8% and 41.2% of the SZ and BP disorder patients had Purkinje neuron linear density ≥4.2 neurons/mm vs. 87.5% of NPS. To compare SZ vs. NPS, Fisher's exact test was used (P = 0.0008), whereas to compare BP vs. NPS, we used a chi-square test (P = 0.002).
In our cohort, several patients had a history of current alcohol abuse or other substances of abuse (Table 1). The mean Purkinje neuron density was virtually identical whether subjects with history of alcohol abuse or other drugs of abuse were included or excluded. Furthermore, our mixed model did not find any significant change in this variable (F1,405 = 0.16, NS). We did not find any significant effect of type of antipsychotic treatment or antipsychotic dosage, lithium, or other mood stabilizers (i.e., valproic acid) on Purkinje neuron density. The autopsy records showed no gross or microscopic cerebellar atrophy or degeneration, with the exception of one male BP disorder patient in whom the Purkinje cell layer expressed a modest Bergmann gliosis.
Reelin Is Reduced in Cerebellar Granular Cells of SZ and BP Disorder Patients.
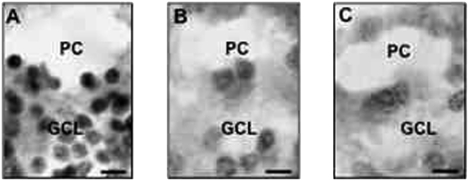
In previous studies, we (2, 3) and others (4) reported that in the cerebellum of SZ and BP disorder patients, there is a marked (≈50%) decrease of reelin mRNA and protein expression. Fig. 2 depicts a typical example of reelin in situ hybridization study in the cerebellar cortex of NPS (Fig. 2A), SZ (Fig. 2B), and BP (Fig. 2C) disorder patients. It is evident that reelin mRNA is primarily expressed in granule cells but not in Purkinje neurons.
Fig. 2.
Reelin mRNA in situ hybridization in human cerebellar cortex from an NPS (A), a SZ (B), and a BP disorder patient (C). Note the decrease of reelin mRNA signals in the granular cell layer (GCL) in SZ and BP disorder patients. Purkinje cells (PC) lack a reelin mRNA signal. (Scale bar: 10 μm.)
In addition, Table 3 shows that reelin mRNA expression is down-regulated in cerebellar granule cells of SZ and BP patients who also show a decrease in the linear density of Purkinje neurons. In the same samples, the number of cerebellar granule cells stained with Nissl failed to change (Table 3).
Table 3.
Density of Purkinje neurons, reelin mRNA-positive, and Nissl-positive granular neurons in cerebellum of SZ and BP disorder patients
| Groups | Purkinje neurons (per mm) | Reelin mRNA-positive granular neurons (×104/mm2) | Nissl-positive granular neurons (×104/mm2) |
| NPS | 4.7 ± 0.23 | 2.2 ± 0.16 | 2.6 ± 0.41 |
| SZ | 3.5 ± 0.32* | 1.4 ± 0.15* | 2.5 ± 0.10 |
| BP | 3.1 ± 0.28* | 1.4 ± 0.11* | 2.5 ± 0.12 |
All values are mean ± SEM of three subjects per group.
*Statistically significant difference at P ≤ 0.01; one-way ANOVA followed by Bonferroni multiple comparison.
Cerebellar Purkinje Neurons Are Decreased in HRM.
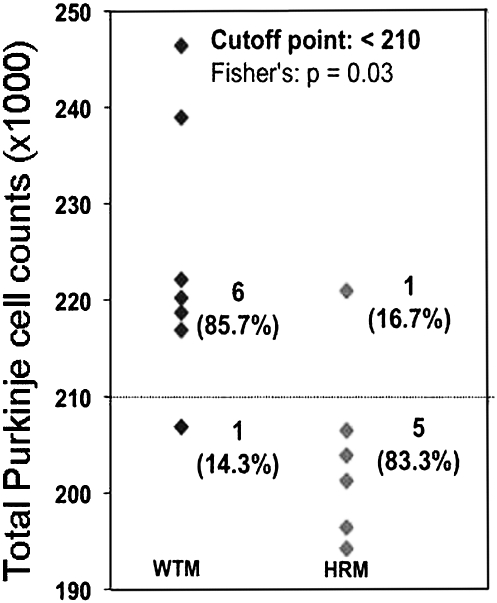
To evaluate whether the reduced number of Purkinje neurons in SZ and BP disorder patients is due to an insufficient expression of reelin, we measured Purkinje neuron density in the whole cerebellum of WTM and HRM. HRM differ from WTM because they express reelin haploinsufficiency. In line with previous results, showing that reelin mRNA is reduced by ≈50% (from 0.34 ± 0.025 to 0.17 ± 0.006 fmol/μg RNA) in the cerebellum of HRM (15), the present data show that the number of Purkinje neurons per cerebellum in adult (aged 3–9 months) HRM [203 ± 3.9 × 103 (mean ± SE), n = 11] is significantly lower (Student’s t test; t1, 20 = 2.66, P < 0.05) than in WTM of corresponding ages (224 ± 5.1 × 103, n = 11). As shown in Fig. 3, only 16.7% of the HRM had Purkinje neuron cerebellar density ≥210 × 103, vs. 85.7% of the WTM (P = 0.03). There were no significant differences in the cerebellum weight between WTM and HRM of any age.
Fig. 3.
Plot of total Purkinje neuron number from wild-type mice (WTM) (n = 7) and heterozygous reeler mice (HRM) (n = 6). In parentheses is the percentage of mice with Purkinje cell number above and below cutoff point of 210 × 103. Note that only 16.7% of HRM had Purkinje neuron density ≥210 × 103 vs. 85.7% of WTM. To compare HRM vs. WTM, Fisher's exact test was used (P = 0.03).
Discussion
Purkinje Neurons Are Decreased in Cerebellar Purkinje Cell Layers of SZ and BP Disorder Patients.
The Purkinje neuron decrease in SZ and BP patients is statistically independent of those shown in brain degenerative disorders. For example, with the exception of one BP disorder patient, there was no evidence in our samples of gross or microscopic (i.e., Bergmann gliosis) cerebellar damage or overt histologic signs of Alzheimer’s pathology that correlated with the reduction of Purkinje neurons. Furthermore, we did not find a statistically significant correlation between Purkinje neuron number and drugs of abuse, lithium, mood stabilizers (i.e., valproic acid), gender, type or dose of neuroleptic administered, or duration of psychopathology.
Our data agree with previous neuropathologic studies conducted in small cohorts of SZ patients that reported a decrease in Purkinje neurons in the anterior cerebellar vermis (23, 24). Another study (25) reported decreased cerebellar Purkinje neuron size in the superior vermis of elderly SZ patients. Atrophy of the anterior cerebellar vermis was initially reported by Weinberger et al. (26) and subsequently confirmed by others (27–29).
However, in other SZ patient studies in which the Purkinje neurons were counted in the lateral cerebellar hemispheres (30) or Purkinje neurons of the entire cerebellum were stereologically counted (31), these changes were not duplicated. The reasons for these discrepancies are possibly in (i) the method of comparing Purkinje cell linear density vs. stereologic quantitation of Purkinje neurons in the entire cerebellum, (ii) the region of cerebellum analyzed or (iii) the number of subjects analyzed (usually small), or (iv) the contribution of confounding factors, such as generalized degenerative CNS disorders (i.e., Alzheimer’s disease), drugs of abuse, seizures, or antipsychotic treatment.
In our cohort, on the basis of statistical analyses, we can also exclude that demographic factors or other confounding variables are the cause of the Purkinje neuron decrease in the cerebellum of SZ or BP disorder patients. Hence, we can provisionally assume that the reduction of Purkinje neuron density in the Purkinje cell layer of the anterior lobe of the cerebellum of SZ or BP disorder patients is independent from demographic factors and instead can be attributed to psychiatric pathophysiology. A similar decrease of Purkinje neurons observed in BP and SZ patients suggests that the two disorders may share a common underlying cerebellar deficit.
Decrease of GAD67 in the Cerebellum of SZ and BP Disorder Patients.
It is possible that the decrease of Purkinje neurons (cells that express GAD67) could be part of the reason for the down-regulation of GAD67 mRNA and protein we and others (3, 4) have reported in the cerebella of SZ and BP disorder patients. However, although the reduction of the Purkinje neurons is only 20%, the decrease of GAD67 mRNA and protein is approximately 50%. Hence, a GAD67 down-regulation caused by morphologically undetectable changes of Purkinje neurons or of other GABAergic interneurons (i.e., basket and Golgi cells) is a possibility.
We can infer that the down-regulation of GAD67 in Purkinje cells contributes to the dysfunction of cerebellar–dentate–prefrontal cortex circuitry (32, 33) in SZ, which could also be one reason for the psychiatric symptoms and neurologic abnormalities, such as the altered coordination of limbic movements (1, 26, 33), found in these patients.
Is the Reduction of Cerebellar Purkinje Neurons in SZ and BP Disorder Patients Related to a Reduction of Reelin?
An important and reproducible finding in SZ and BP disorder patients is the marked decrease of reelin mRNA and protein expression that we have measured in various brain areas, including the cerebellum (2–4, 7–9, 34).
It was suggested that reelin, in addition to its important role in the control of neuronal migration, also promotes neuronal maturation and function in postnatal brains (11, 35).
In support of this conclusion are experiments showing that adding recombinant reelin to hippocampal slices preparations enhanced NMDA-mediated CA1 responses and dramatically elevated the magnitude of long-term potentiation induction and maintenance (12). In cultured hippocampal neurons, reelin signaling is required for the normal development of dendritic structures and involves a signal transduction pathway that is dependent on the activation of VLDL/APO-E2 receptors (20). Furthermore, in postnatal organotypic cortical cultures, the application of reelin regulates dendritic maturation of cortical layer II/III pyramidal neurons. This mechanism presumably involves stimulation of integrin receptors preferentially expressed on the apical dendrites of cortical layer II/III pyramidal neurons (14).
A role for reelin–integrin receptor signaling in dendritic maturation and spine formation is supported by other studies (22), which reported that reelin applied to cortical synaptoneurosomal preparations increases biosynthesis not only of activity-regulated cytoskeletal protein (Arc) but also other rapidly translated proteins in a manner that is inhibited by echistatin, a potent integrin receptor antagonist ligand. Hence, reelin deficits in the brain of SZ patients may be indicative of an altered trophic reelin signaling that leads to a decreased synaptic function and possibly a defective Purkinje neuron migration to the Purkinje cell layer.
A previous study reported that HRM, which express half the reelin levels in the telencephalon and cerebellum of WTM (for review see ref. 36), exhibit a reduction of cerebellar Purkinje neurons (21).
In our study, we have confirmed the existence of a reduction of Purkinje neurons in the cerebellum of 3–9-month-old HRM. In a preliminary experiment, the reduction of Purkinje neurons in HRM remained unchanged in 22–26-month-old animals, even though these mice show a significant reduction of Purkinje cells due to aging. This experiment suggests that both reelin and unknown aging factors may contribute independently to the regulation of GABAergic function in the cerebellum.
In conclusion, the data from our cerebellar postmortem studies show a statistically significant loss of Purkinje neurons in the Purkinje cell layer of the anterior lobe of cerebellum in patients diagnosed with SZ and BP disorder. Although the reason for the decrease remains unknown, the reelin expression down-regulation in the cerebella of SZ and BP disorder patients and the reduction of Purkinje neurons in HRM, which are reelin haploinsufficient, suggest an important role for reelin in the regulation of Purkinje GABAergic neuron expression or positioning in the cerebellar Purkinje cell layer.
Autism is a mental disorder in which cerebellar global reelin levels are decreased (37). However, in the lateral cerebellar hemispheres of autistic patients, the number of Purkinje cells is not decreased (38). One possible reason for the apparent discrepancy between SZ and autistic patients is the fact that the correlation between reelin and Purkinje cell decreases may not be global but instead could be restricted to specific parts of the cerebellum.
The goal of our future studies is to elucidate whether the reelin deficit and reduction of Purkinje neurons reported in the anterior lobe of the cerebellum of SZ and BP patients can be extended to other cerebellar areas.
The decreased synthesis and release of reelin from cerebellar granule cells of HRM could serve as a model to understand the pathophysiologic mechanisms that regulate GABAergic neuron dysfunction in the cerebellum of SZ or BP disorder patients.
Materials and Methods
Tissue Collection and Histologic Methods.
Postmortem human cerebellum.
Blocks of cerebellar tissue from NPS, SZ, and BP disorder patients were obtained from the Harvard Brain Tissue Resource Center, McLean Hospital (Belmont, MA). The cerebella were roughly matched for age, PMI, and other demographic parameters (Table 1). The cerebellar cortices were cut sagittally in 0.5-cm blocks starting from the midline. For each sample, we received a section of cerebellar cortex lateral to the vermis and including the dentate nucleus. All blocks were received fixed in 4% paraformaldehyde (PFA) and then cryopreserved in 30% sucrose in PBS before they were sliced at the cryostat (Microm HM 550; Thermo Scientific). From each block, 10 sections of 12 μm each were microdissected. Purkinje neuron measurements were performed on every four sections stained with 1% cresyl violet.
Purkinje neuron counts.
Quantification was carried out using a Zeiss Axioscope 2 microscope connected to a computer-assisted imaging analyzer system (Axiovision). For each section, two cerebellar folias located closest to the white matter were selected for counting. Within each folium, two smaller (900 μm) subregions at each side of the folia were measured, thus eight subregions were counted for each subject. Nucleated Purkinje neurons were counted blind to conditions using a 20× objective and expressed as linear cell density (i.e., Purkinje neurons per millimeter of Purkinje cell layer). To assess reliability, the measurements were repeated in five control subjects by an independent tester. Agreement between the two testers was high (95% or better).
Reelin in situ hybridization.
To visualize the reelin mRNA signal, free-floating 18-μm sections were incubated for 40–48 h with a mixture of 50 pmol/mL of three antisense oligonucleotide probes complementary to bases 1729–11752, 5505–5528, and 10102–10125 of the human reelin DNA (accession no. NM_001379). The specificity of such probes is described in detail by Veldic et al. (7). The oligonucleotide 3′ terminals were labeled with a digoxigenin kit (Roche Diagnostics). In situ hybridization was performed as previously described (39). After hybridization reactions were completed, tissue sections were incubated for 72 h at 4 °C in 0.1 M PBS containing 1% normal goat serum and 0.1 μg/mL of a monoclonal antibody against digoxigenin (mouse antidigoxigenin monoclonal antiserum from Roche Diagnostics). This procedure was followed by (i) incubation with biotinylated goat anti-mouse antiserum (Vector Laboratories; 1:250) for 1 h, and (ii) a successive reaction with the avidin-biotin-peroxidase complex (Vector Laboratories) followed by 3-3`-diaminobenzidine tetrahydrochloride (Sigma-Aldrich) with 0.1% nickel ammonium sulfate (2–3 min), such that a gray-black shade in the precipitate was obtained.
To test the immunologic detection specificity of digoxigenin, oligonucleotides were omitted from the hybridization buffer, and no staining was observed. Oligoprobe specificity was tested by using digoxigenin-labeled scrambled oligonucleotide sequences. As expected, specific neuronal staining was not detected.
Reelin mRNA-positive neuron counts in the cerebellar granule cell layer.
Reelin mRNA-positive neurons identified by solid or punctate labeling were counted in six fields regularly distributed in the tissue sections in the middle of the cerebellar granule layer. In adjacent slides, Nissl-stained neuronal nuclei were also identified by their special shape and size. The irregularly shaped nuclei that are characteristic of glial cells were not recorded. Reelin mRNA- and Nissl-positive neurons were counted using a 100× oil immersion objective. The size of the counting frame (40 × 40 μm) was chosen to obtain cell counts with an error of less than 10% between repeated measurements.
Mouse cerebellum.
From an established breeding colony of B6C3FE mice, male WTM were bred with female HRM. The progeny were 50% HRM and 50% WTM. Genotyping was performed as previously described (15). Mice had access to food and water ad libitum and were housed on a 14 h/10 h light/dark cycle. Male HRM and WTM were studied between ages 3 and 9 months (adult) and between ages 22 and 25 months (old). To establish the number of cerebellar Purkinje neurons, mice were anesthetized with CO2 and perfused intracardially with prewarmed saline, followed by 30 mL of ice-cold 4% PFA in 0.1 M PBS (pH 7.0). Brains were dissected out and postfixed overnight at 4 °C in PFA, followed by 24-h cryopreservation in 30% sucrose in PBS.
Cerebellar blocks were cut sagittally, and 10-μm sections were Nissl-stained with 1% cresyl violet solution and observed under an Olympus microscope using 20× objective. Purkinje neurons were counted in every 40 sections of the cerebellar cortex, starting from the midline. Purkinje neuron identification includes the location in the Purkinje cell layer and the expression of a large soma with a nucleus. The total number of cerebellar Purkinje neurons was calculated by the area under the curve defined by connecting data points of each section and correcting the row values by the Hendry correction factor (40).
Statistical Analyses.
Analysis was performed with a commercial statistical package (SAS Institute) using mixed models of four replications of Purkinje neuron linear density counts, each nested in two regions (random effect), and fixed effects of SZ, BP, and NPS.
We also examined whether variables such as age, gender, PMI, hemisphere, brain pH, drug or alcohol abuse, suicide, age of onset of illness, cause of death, or types of medication had different effects among SZ and BP patients and NPS by adding them to the model as a fixed factor or covariate (PMI, brain PH, and age of onset of illness as continuous covariates; age was treated as continuous or categorized with three categories using 41 and 65 as cutoffs).
We arbitrarily chose a value of 4.2 Purkinje neurons/mm as the cutoff point for average Purkinje neuron linear density, so that each diagnostic group was categorized as two categories of <4.2 and ≥4.2. We then compared the frequencies of these two categories in SZ or BP subjects with NPS using χ2 or Fisher exact tests. Fisher exact test was used only when the expected number of subjects was fewer than five. The Fisher exact test was also used to compare the number of Purkinje neurons from WTM and HRM.
Acknowledgments
We thank Dr. Francine Benes (Harvard Medical School, Department of Psychiatry) and Dr. S. Hossein Fatemi (University of Minnesota, Division of Neuroscience Research in Psychiatry) for constructive criticism and suggestions in the preparation of the manuscript. This research was partially supported by National Institutes of Health Grants MH071667 (to E.C.) and MH070855 (to A.G.).
Footnotes
The authors declare no conflict of interest.
References
- 1.Picard H, Amado I, Mouchet-Mages S, Olié JP, Krebs MO. The role of the cerebellum in schizophrenia: An update of clinical, cognitive, and functional evidences. Schizophr Bull. 2008;34:155–172. doi: 10.1093/schbul/sbm049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Impagnatiello F, et al. A decrease of reelin expression as a putative vulnerability factor in schizophrenia. Proc Natl Acad Sci USA. 1998;95:15718–15723. doi: 10.1073/pnas.95.26.15718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guidotti A, et al. Decrease in reelin and glutamic acid decarboxylase67 (GAD67) expression in schizophrenia and bipolar disorder: A postmortem brain study. Arch Gen Psychiatry. 2000;57:1061–1069. doi: 10.1001/archpsyc.57.11.1061. [DOI] [PubMed] [Google Scholar]
- 4.Fatemi SH, et al. GABAergic dysfunction in schizophrenia and mood disorders as reflected by decreased levels of glutamic acid decarboxylase 65 and 67 kDa and Reelin proteins in cerebellum. Schizophr Res. 2005;72:109–122. doi: 10.1016/j.schres.2004.02.017. [DOI] [PubMed] [Google Scholar]
- 5.Eastwood SL, Law AJ, Everall IP, Harrison PJ. The axonal chemorepellant semaphorin 3A is increased in the cerebellum in schizophrenia and may contribute to its synaptic pathology. Mol Psychiatry. 2003;8:148–155. doi: 10.1038/sj.mp.4001233. [DOI] [PubMed] [Google Scholar]
- 6.Pesold C, et al. Reelin is preferentially expressed in neurons synthesizing gamma-aminobutyric acid in cortex and hippocampus of adult rats. Proc Natl Acad Sci USA. 1998;95:3221–3226. doi: 10.1073/pnas.95.6.3221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Veldic M, et al. DNA-methyltransferase 1 mRNA is selectively overexpressed in telencephalic GABAergic interneurons of schizophrenia brains. Proc Natl Acad Sci USA. 2004;101:348–353. doi: 10.1073/pnas.2637013100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Veldic M, Guidotti A, Maloku E, Davis JM, Costa E. In psychosis, cortical interneurons overexpress DNA-methyltransferase 1. Proc Natl Acad Sci USA. 2005;102:2152–2157. doi: 10.1073/pnas.0409665102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Veldic M, et al. Epigenetic mechanisms expressed in basal ganglia GABAergic neurons differentiate schizophrenia from bipolar disorder. Schizophr Res. 2007;91:51–61. doi: 10.1016/j.schres.2006.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lacor PN, et al. Reelin secretion from glutamatergic neurons in culture is independent from neurotransmitter regulation. Proc Natl Acad Sci USA. 2000;97:3556–3561. doi: 10.1073/pnas.050589597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Costa E, et al. Dendritic spine hypoplasticity and downregulation of reelin and GABAergic tone in schizophrenia vulnerability. Neurobiol Dis. 2001;8:723–742. doi: 10.1006/nbdi.2001.0436. [DOI] [PubMed] [Google Scholar]
- 12.Levenson JM, Qiu S, Weeber EJ. The role of reelin in adult synaptic function and the genetic and epigenetic regulation of the reelin gene. Biochim Biophys Acta. 2008;1779:422–431. doi: 10.1016/j.bbagrm.2008.01.001. [DOI] [PubMed] [Google Scholar]
- 13.Carboni G, et al. Enhanced dizocilpine efficacy in heterozygous reeler mice relates to GABA turnover downregulation. Neuropharmacology. 2004;46:1070–1081. doi: 10.1016/j.neuropharm.2004.02.001. [DOI] [PubMed] [Google Scholar]
- 14.Campo CG, Sinagra M, Verrier D, Manzoni OJ, Chavis P. Reelin secreted by GABAergic neurons regulates glutamate receptor homeostasis. PLoS One. 2009;4:e5505. doi: 10.1371/journal.pone.0005505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tueting P, et al. The phenotypic characteristics of heterozygous reeler mouse. Neuroreport. 1999;10:1329–1334. doi: 10.1097/00001756-199904260-00032. [DOI] [PubMed] [Google Scholar]
- 16.Qiu S, et al. Cognitive disruption and altered hippocampus synaptic function in Reelin haploinsufficient mice. Neurobiol Learn Mem. 2006;85:228–242. doi: 10.1016/j.nlm.2005.11.001. [DOI] [PubMed] [Google Scholar]
- 17.Weeber EJ, et al. Reelin and ApoE receptors cooperate to enhance hippocampal synaptic plasticity and learning. J Biol Chem. 2002;277:39944–39952. doi: 10.1074/jbc.M205147200. [DOI] [PubMed] [Google Scholar]
- 18.Pillai A, Mahadik SP. Increased truncated TrkB receptor expression and decreased BDNF/TrkB signaling in the frontal cortex of reeler mouse model of schizophrenia. Schizophr Res. 2008;100:325–333. doi: 10.1016/j.schres.2007.11.030. [DOI] [PubMed] [Google Scholar]
- 19.Liu WS, et al. Down-regulation of dendritic spine and glutamic acid decarboxylase 67 expressions in the reelin haploinsufficient heterozygous reeler mouse. Proc Natl Acad Sci USA. 2001;98:3477–3482. doi: 10.1073/pnas.051614698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Niu S, Renfro A, Quattrocchi CC, Sheldon M, D’Arcangelo G. Reelin promotes hippocampal dendrite development through the VLDLR/ApoER2-Dab1 pathway. Neuron. 2004;41:71–84. doi: 10.1016/s0896-6273(03)00819-5. [DOI] [PubMed] [Google Scholar]
- 21.Hadj-Sahraoui N, Frédéric F, Delhaye-Bouchaud N, Mariani J. Gender effect on Purkinje cell loss in the cerebellum of the heterozygous reeler mouse. J Neurogenet. 1996;11:45–58. doi: 10.3109/01677069609107062. [DOI] [PubMed] [Google Scholar]
- 22.Dong E, et al. A reelin-integrin receptor interaction regulates Arc mRNA translation in synaptoneurosomes. Proc Natl Acad Sci USA. 2003;100:5479–5484. doi: 10.1073/pnas.1031602100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Reyes MG, Gordon A. Cerebellar vermis in schizophrenia. Lancet. 1981;2:700–701. doi: 10.1016/s0140-6736(81)91039-4. [DOI] [PubMed] [Google Scholar]
- 24.Stevens JR. Neuropathology of schizophrenia. Arch Gen Psychiatry. 1982;39:1131–1139. doi: 10.1001/archpsyc.1982.04290100011003. [DOI] [PubMed] [Google Scholar]
- 25.Tran KD, Smutzer GS, Doty RL, Arnold SE. Reduced Purkinje cell size in the cerebellar vermis of elderly patients with schizophrenia. Am J Psychiatry. 1998;155:1288–1290. doi: 10.1176/ajp.155.9.1288. [DOI] [PubMed] [Google Scholar]
- 26.Weinberger DR, Kleinman JE, Luchins DJ, Bigelow LB, Wyatt RJ. Cerebellar pathology in schizophrenia: A controlled postmortem study. Am J Psychiatry. 1980;137:359–361. doi: 10.1176/ajp.137.3.359. [DOI] [PubMed] [Google Scholar]
- 27.Nasrallah HA. Right-hemisphere speech, callosal size, perinatal brain insult, and schizophrenia. Ann Neurol. 1989;26:290–291. doi: 10.1002/ana.410260222. [DOI] [PubMed] [Google Scholar]
- 28.Lippmann S, et al. Cerebellar vermis dimensions on computerized tomographic scans of schizophrenic and bipolar patients. Am J Psychiatry. 1982;139:667–668. doi: 10.1176/ajp.139.5.667. [DOI] [PubMed] [Google Scholar]
- 29.Dewan MJ, et al. Cerebellar morphology in chronic schizophrenic patients: A controlled computed tomography study. Psychiatry Res. 1983;10:97–103. doi: 10.1016/0165-1781(83)90108-7. [DOI] [PubMed] [Google Scholar]
- 30.Jeste DV, Barban L, Parisi J. Reduced Purkinje cell density in Huntington’s disease. Exp Neurol. 1984;85:78–86. doi: 10.1016/0014-4886(84)90162-6. [DOI] [PubMed] [Google Scholar]
- 31.Andersen BB, Pakkenberg B. Stereological quantitation in cerebella from people with schizophrenia. Br J Psychiatry. 2003;182:354–361. doi: 10.1192/bjp.182.4.354. [DOI] [PubMed] [Google Scholar]
- 32.Andersen BB, Korbo L, Pakkenberg B. A quantitative study of the human cerebellum with unbiased stereological techniques. J Comp Neurol. 1992;326:549–560. doi: 10.1002/cne.903260405. [DOI] [PubMed] [Google Scholar]
- 33.Katsetos CD, Hyde TM, Herman MM. Neuropathology of the cerebellum in schizophrenia—an update: 1996 and future directions. Biol Psychiatry. 1997;42:213–224. doi: 10.1016/S0006-3223(96)00313-7. [DOI] [PubMed] [Google Scholar]
- 34.Eastwood SL, Harrison PJ. Cellular basis of reduced cortical reelin expression in schizophrenia. Am J Psychiatry. 2006;163:540–542. doi: 10.1176/appi.ajp.163.3.540. [DOI] [PubMed] [Google Scholar]
- 35.D’Arcangelo G. The role of the Reelin pathway in cortical development. Symp Soc Exp Biol. 2001;53:59–73. [PubMed] [Google Scholar]
- 36.Tueting P, Doueiri MS, Guidotti A, Davis JM, Costa E. Reelin down-regulation in mice and psychosis endophenotypes. Neurosci Biobehav Rev. 2006;30:1065–1077. doi: 10.1016/j.neubiorev.2006.04.001. [DOI] [PubMed] [Google Scholar]
- 37.Fatemi SH, Stary JM, Halt AR, Realmuto GR. Dysregulation of Reelin and Bcl-2 proteins in autistic cerebellum. J Autism Dev Disord. 2001;31:529–535. doi: 10.1023/a:1013234708757. [DOI] [PubMed] [Google Scholar]
- 38.Fatemi SH, et al. Purkinje cell size is reduced in cerebellum of patients with autism. Cell Mol Neurobiol. 2002;22:171–175. doi: 10.1023/A:1019861721160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rodriguez MA, et al. In Patas monkey, glutamic acid decarboxylase-67 and reelin mRNA coexpression varies in a manner dependent on layers and cortical areas. J Comp Neurol. 2002;451:279–288. doi: 10.1002/cne.10341. [DOI] [PubMed] [Google Scholar]
- 40.Hendry IA. A method to correct adequately for the change in neuronal size when estimating neuronal numbers after nerve growth factor treatment. J Neurocytol. 1976;5:337–349. doi: 10.1007/BF01175119. [DOI] [PubMed] [Google Scholar]
- 41.Feighner JP, et al. Diagnostic criteria for use in psychiatric research. Arch Gen Psychiatry. 1972;26:57–63. doi: 10.1001/archpsyc.1972.01750190059011. [DOI] [PubMed] [Google Scholar]