Abstract
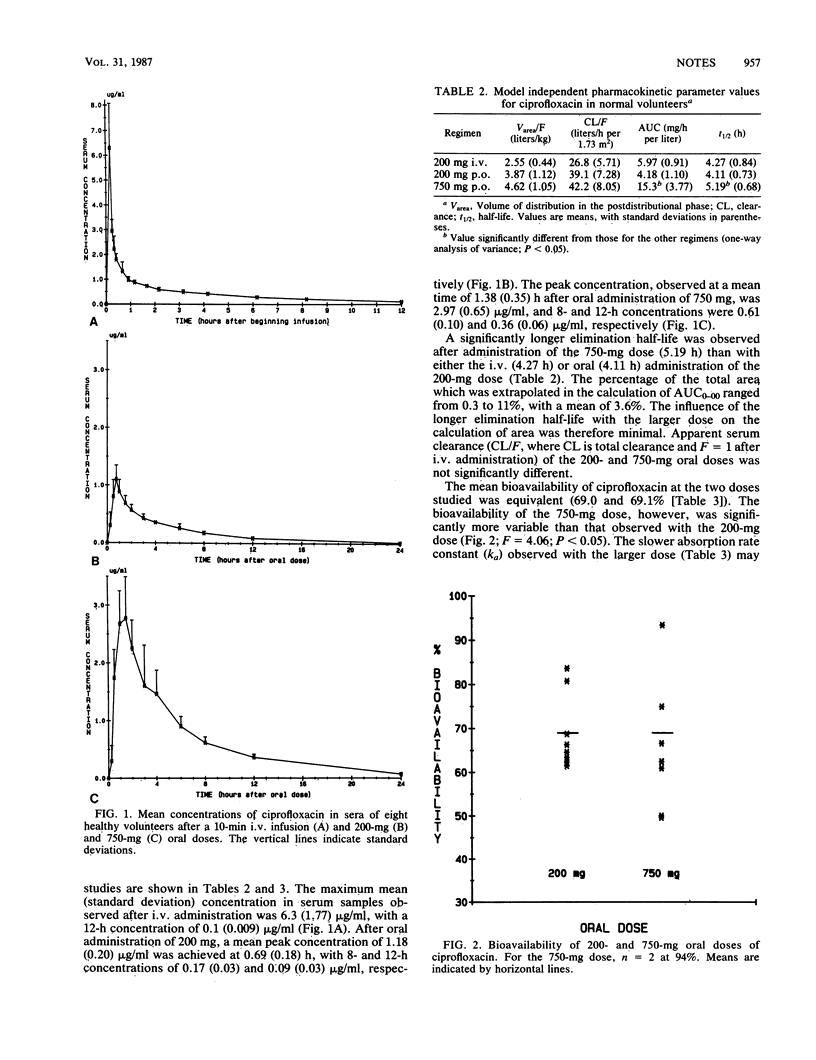
We evaluated the bioavailability of ciprofloxacin at two dose sizes in eight healthy volunteers. Each volunteer was given 200 mg of ciprofloxacin both orally and intravenously in a randomized crossover fashion and 750 mg orally. Bioavailability at the two doses was similar: 69 and 69.1% for the 200- and 750-mg doses, respectively. However, the bioavailability observed with the 750-mg dose was significantly more variable than that observed with the 200-mg dose. Between 375 and 700 mg of ciprofloxacin reached the systemic circulation after administration of the 750-mg dose, with no evidence of adverse reactions.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- D'Argenio D. Z., Schumitzky A. A program package for simulation and parameter estimation in pharmacokinetic systems. Comput Programs Biomed. 1979 Mar;9(2):115–134. doi: 10.1016/0010-468x(79)90025-4. [DOI] [PubMed] [Google Scholar]
- Drusano G. L., Standiford H. C., Plaisance K., Forrest A., Leslie J., Caldwell J. Absolute oral bioavailability of ciprofloxacin. Antimicrob Agents Chemother. 1986 Sep;30(3):444–446. doi: 10.1128/aac.30.3.444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolfson J. S., Hooper D. C. The fluoroquinolones: structures, mechanisms of action and resistance, and spectra of activity in vitro. Antimicrob Agents Chemother. 1985 Oct;28(4):581–586. doi: 10.1128/aac.28.4.581. [DOI] [PMC free article] [PubMed] [Google Scholar]


