Abstract
Resveratrol is a polyphenol that acts on multiple molecular targets important for cell differentiation and activation. Dendritic cells (DCs) are a functionally diverse cell type and represent the most potent antigen-presenting cells of the immune system. In this study, we investigated resveratrol-induced effects on DCs during their differentiation and maturation. Our results show that resveratrol induces DC-associated tolerance, particularly when applied during DC differentiation. Costimulatory molecules CD40, CD80 and CD86 were down-regulated, as was the expression of major histocompatibility complex (MHC) class II molecules. Surface expression of inhibitory immunoglobulin-like transcript 3 (ILT3) and ILT4 molecules was induced, while human leucocyte antigen (HLA)-G expression was not affected. Resveratrol-treated DCs lost the ability to produce interleukin (IL)-12p70 after activation, but had an increased ability to produce IL-10. Such DCs were poor stimulators of allogeneic T cells and had lowered ability to induce CD4+ T-cell migration. Furthermore, treated cells were able to generate allogeneic IL-10-secreting T cells, but were not competent in inducing FoxP3 expression These tolerogenic effects are probably associated with the effect of resveratrol on multiple molecular targets through which it interferes with DC differentiation and nuclear factor (NF)-κB translocation. Our data provide new insights into the molecular and functional mechanisms of the tolerogenic effects that resveratrol exerts on DCs.
Keywords: immune modulation, nuclear factor (NF)-кB, regulatory T cells, resveratrol, tolerance
Introduction
Resveratrol, a 3,5,4′-trihydroxystilbene, is a polyphenol present in relatively large amounts in red wine and grapes (Vitis vinifera) and in several other plants. With the discovery of its potential cardioprotective effects,1 there has been an exponential increase in the number of publications reporting various beneficial effects of resveratrol. Many have presented its cancer chemopreventive properties in various experimental models both in vivo and in vitro.2–5 Furthermore, numerous studies have shown the positive effects of resveratrol in ameliorating inflammatory conditions such as colonic inflammation,6 lipopolysaccharide (LPS)-induced airway inflammation7 and osteoarthritis,8 and even in preventing allograft rejection.9,10 Resveratrol has been shown to inhibit activation of the transcription factor nuclear factor (NF)-κB induced by interleukin (IL)-1β11 and by tumour necrosis factor (TNF)-α, and also to inhibit activation of activator protein (AP)-1, mitogen-activated protein kinase kinase and c-Jun N-terminal kinase.12
Dendritic cells (DCs) constitute a highly specialized antigen-presenting cell system which is critical for initiating effective CD4+ T-cell responses. There have been numerous demonstrations of the functional, phenotypic and morphological heterogeneity of DCs. DCs were found to modulate T-cell responses, not only in terms of immunogenicity but also, to a great extent, in terms of the induction of immune tolerance.13–16 After DCs have captured antigen and are activated by maturation stimuli such as lipopolysaccharide (LPS) or inflammatory cytokines, they migrate into secondary lymph nodes and on their way mature into potent initiators of immunity. However, under certain environmental conditions, such as adequate amounts of the anti-inflammatory cytokine IL-1017 or transforming growth factor (TGF)-β,18 contact with regulatory T cells19 or treatment with certain immunosuppressive drugs,20 the DCs can become tolerogenic and resistant to the normal maturation programme that would result in highly mature DCs capable of polarizing strong T helper type 1 (Th1) responses. High expression of surface inhibitory molecules, such as immunoglobulin-like transcript (ILT)-3 and ILT-4, is considered to be a biomarker for tolerogenic DCs (TDCs19,21). In addition, TDCs are capable of producing significant amounts of immunosuppressive IL-10 and do not produce proinflammatory cytokines IL-12p70 and TNF-α. Several drugs and endogenous ligands have been shown to induce tolerogenic properties in DCs by blocking their maturation by various means, mostly involving inhibition of NF-κB.20 Fewer agents have been shown to act on DCs during their differentiation from monocytes. Corticosteroids22 and 1α,25-dihydroxy-vitamin D323 are reported to block DC differentiation at the monocyte level, promoting the expression of the monocyte marker CD14.
In the present study, we analysed the immunomodulatory effects of resveratrol on human monocyte-derived DCs during their main biological stages of differentiation and maturation, with the aim of generating activation-resistant or, alternatively, activated TDCs. Our results demonstrate that resveratrol treatment during the differentiation stage is of crucial importance in obtaining DCs with strong regulatory potential. Such cells are resistant to LPS-induced activation towards Th1 polarizing DC type but acquire the potential to induce allogeneic IL-10-secreting CD4+ T cells.
Materials and methods
Cell preparation and culture
Buffy coats from the venous blood of normal healthy volunteers were obtained from the Blood Transfusion Centre of Slovenia, according to institutional guidelines. Peripheral blood mononuclear cells (PBMCs) were isolated using Lympholyte®-H (Cedarlane Laboratories, Ontario, Canada). The cells were washed twice with Dulbecco’s phosphate-buffered saline (DPBS), counted, and used as a source for immunomagnetic isolation of CD14-positive cells (CD14 Microbeads; Miltenyi Biotec GmbH, Bergisch Gladbach, Germany). CD14-positive cells (purity of CD14+ cells was always > 95%, as determined by flow cytometry) were cultured in RPMI-1640 (Cambrex, Charles City, IA) medium, supplemented with 10% fetal bovine serum (FBS), gentamicin (50 μg/ml; Gibco, Paisley, UK), 800 U/ml of recombinant human granulocyte–macrophage colony-stimulating factor (rhGM-CSF) and 1000 U/ml of recombinant human IL-4 (rhIL-4) (both Peprotech EC, London, UK). On day 2, half of the medium was exchanged with the addition of starting quantities of rhGM-CSF and rhIL-4. After 5 days, non-adherent immature DCs were harvested and characterized by flow cytometry as CD1ahi, CD80−, CD83−, CD86low and HLA-DRlow. For maturation, DCs were activated with 20 ng/ml of LPS plus 800 U/ml of rhGM-CSF for 48 hr. DCs were treated with resveratrol (Cayman Chemicals, Ann Arbor, MI) either on days 0 and 5, to determine its effects on DCs already at the stage of differentiation from monocytes (termed t = 0 treatment), or on day 5 alone (termed t = 5 treatment), to determine its effects on the maturation process alone. Resveratrol was used at concentrations ranging from 3 to 50 μm, where no effect on cell death or survival was noted (determined using trypan blue). For all functional experiments, DCs were washed extensively (three times) to prevent transfer of residual resveratrol to further cultures.
T cells were purified from human buffy coats. Whole CD4+ T cells were obtained by positive selection using CD4 microbeads (Miltenyi Biotec GmbH). The purity of CD4+ cells was always > 95% as determined by flow cytometry. Naive CD4+ CD45RA+ cells were isolated using the naïve CD4+ T-cell isolation kit from Miltenyi Biotec, following the manufacturer’s protocol strictly. The purity of naive CD4+ T cells was always > 98%.
Allogeneic T-cell proliferation
Dendritic cells obtained after 7 days of culture (immature, mature or differentially treated with resveratrol, as described above) were washed twice in DPBS and incubated with mitomycin C (Sigma-Aldrich, St Louis, MO) to block their proliferation. When using immature DCs, after day 5 the cells were cultured for two more days using 800 U/ml of rhGM-CSF and 1000 U/ml of rhIL-4. Purified whole CD4+ T cells were used as responders. The assays were carried out in 96-well plates, with a total volume of 200 μl per well. CD4+ responder cells were added at 1 × 105 per well, and stimulator DCs at 1 × 104 per well. On day 4 of culture the wells were pulsed with 1 μCi/well [3H]thymidine (Perkin Elmer, Boston, MA) and proliferation was measured by [3H]thymidine incorporation after 18 hr by liquid scintillation counting.
Flow cytometry analysis
The levels of membrane markers were determined by flow cytometry using fluorescently labelled antibodies. Non-adherent cells were harvested on day 5 or 7 and collected by centrifugation. Antibody was added and the cells incubated at room temperature for 15 min in the dark. They were then washed twice with DPBS and re-suspended in 2% paraformaldehyde. The following monoclonal antibodies were used: fluorescein isothiocyanate (FITC)-labelled anti-CD1a, anti-CD14, anti-CD40, anti-CD80, anti-CD4, anti-CCR7, anti-DC-specific ICAM-3 grabbing non-integrin (DC-SIGN) (CD209) and anti-HLA-G (all from Biolegend, San Diego, CA); FITC-labelled anti-CD83 (IQ Products, Groningen, the Netherlands); FITC-labelled anti-CD86 (DakoCytomation, Glostrup, Denmark); FITC-labelled anti-ILT4 and phycoerythrin (PE)-labelled anti-ILT3 (both R&D Systems, Minneapolis, MN); and PE-labelled anti-HLA-DR (Exalpha Biologicals Inc., Shirley, MA). To determine the percentage of CD4+ CD25+ FoxP3+ T cells, we used Biolegend’s Human Treg Flow™ Kit containing FoxP3-Alexa Fluor 488, CD4 PE-Cy5 and CD25 PE antibodies. Cells were permeabilized with Biolegend’s Fix/Perm and Perm buffers. For isotype control, an FITC-IgG1 and PE-IgG2a cocktail was used (Biolegend). Results are expressed as mean fluorescence intensity (MFI) values after subtracting the MFI obtained with the control antibody.
Measurement of cytokine production
Dendritic cells differentiated and subsequently matured (with the addition of 20 ng/ml LPS) in the presence or absence of different concentrations of resveratrol. Cell-free culture supernatants were collected 48 hr after cell activation. Cytokines were measured using a sandwich enzyme-linked immunosorbent assay (ELISA). The levels of each cytokine were determined using Biolegend’s ELISA MAX™ Deluxe Sets for IL-12p70 and IL-10. All assays were set up in duplicate and performed in accordance with the manufacturer’s protocols. The lower detection limits were 4 pg/ml for IL-12p70 and 3 pg/ml for IL-10.
For mixed lymphocyte reactions (MLRs) of resveratrol-treated DCs or control DCs with allogeneic naïve CD4+ T cells, after 7 days, responding CD4+ T cells were washed and stimulated for 24 hr using ionomycin and phorbol myristate acetate. After 24 hr supernatants were examined by ELISA for the presence of IL-10.
Intracellular cytokine staining
Cytokine secretion profiles for naïve CD4+ T cells at the single-cell level were obtained by staining the cells intracellularly for various cytokines and carrying out an analysis by flow cytometry. Freshly isolated, naïve CD4+ T cells were co-cultured with immature, LPS-activated or resveratrol-treated DCs for 14 days. On day 7 of co-culture, half of the medium was replaced and 20 U/ml of rhIL-2 (Perprotech EC) was added. After 14 days, the cells were collected, washed twice with DPBS and rested overnight in complete medium. Cytokine secretion was then induced by the addition of 50 ng/ml phorbol myristate acetate (PMA) and 500 ng/ml ionomycin. After 4 hr, brefeldin A was added to the medium at a concentration of 10 μg/ml for another 4 hr. At the end of stimulation, cells were washed twice and fixed with 4% paraformaldehyde, and then washed and permeabilized with 0·1% Triton X-100 solution for 10 min. The permeabilized cells were incubated in phosphate-buffered saline (PBS) containing 3% bovine serum albumin (BSA) for 30 min to prevent non-specific binding. They were stained intracellularly using FITC-labelled anti-interferon (IFN)-γ and PE-labelled anti-IL-10 (both from Invitrogen, Carlsbad, CA) and analysed by flow cytometry.
Confocal immunofluorescence microscopy
For NF-κB activation assay, resveratrol-treated or control immature DCs were stimulated with 20 ng/ml LPS for 1·5 hr, and then centrifuged (Cytofuge; IRIS Inc., Westwood, MA) for 6 min at 55 g onto glass coverslips, fixed with 4% paraformaldehyde for 45 min and permeabilized with 0·1% Triton X-100 in PBS, pH 7·4, for 10 min. Non-specific staining was blocked with 3% BSA in PBS, pH 7·4, for 1 hr. NF-κB p65 was labelled with Alexa Fluor 488 conjugated antibody (F-6; Santa Cruz Biotechnology, Santa Cruz, CA) for 1·5 hr in blocking buffer and washed with PBS. The ProLong Antifade kit (Molecular Probes, Carlsbad, CA) was used to mount coverslips on glass slides. Circular regions of interest (area 10 μm2) were selected in the nucleus and the cytoplasm, and activation of NF-κB determined as the ratio of mean fluorescence intensities of the nucleus and cytoplasm.
Fluorescence microscopy was performed using a Carl Zeiss LSM 510 confocal microscope (Jena, Germany). Alexa Fluor 488 or phalloidin tetramethylrhodamine B isothiocyanate conjugate were excited with an argon (488 nm) or helium/neon (543 nm) laser and emission was filtered using narrow-band 505–530-nm and long pass (LP) 560-nm filters, respectively. Images were analysed using Carl Zeiss LSM image software 3.0.
Results
DCs treated with resveratrol show down-regulation of positive costimulatory molecules, reduced NF-κB translocation and reduced DC differentiation markers
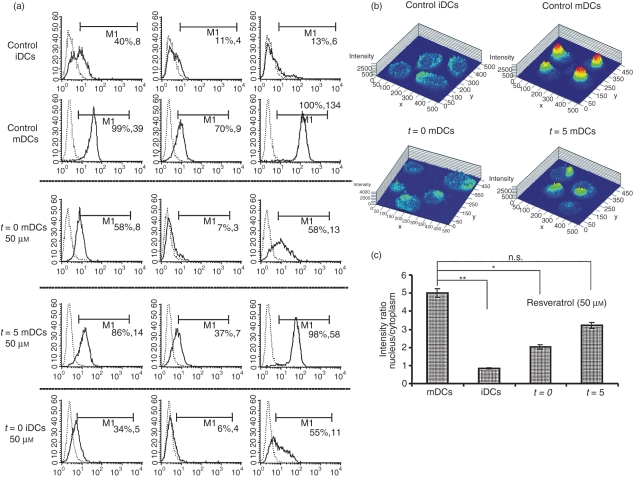
Resveratrol was added to DC cultures at a concentration of 10 or 50 μm, either at the start of DC differentiation on day 0 and again on day 5 (t = 0 DCs) along with the addition of LPS, or only on day 5, when the cells were incubated for 45 min before LPS was added (t = 5 DCs). Resveratrol dose-dependently (data not shown) and strongly down-regulated the expression of costimulatory molecules CD40, CD80 and CD86, compared with control mature DCs (mDCs) (Fig. 1a). This down-regulation was much more apparent when resveratrol was present during the stage of differentiation from monocytes from the start of DC cultures. Treatment of DCs with resveratrol from t = 0 down-regulated the expression of CD40, CD80 and CD86 by 2-, 5- and 10-fold, respectively. For t = 5 DCs, the suppression of positive costimulatory molecules was much less evident, being about 1·6-, 2·8- and 2·3-fold for CD40, CD80 and CD86, respectively.
Figure 1.
Resveratrol down-regulates the expression of costimulatory molecules and reduces nuclear factor (NF)-κB translocation. Dendritic cells (DCs) were prepared as described in the Materials and methods. Resveratrol was added to DC cultures at the start of differentiation from monocytes until the end of maturation (t = 0), or DCs were incubated with resveratrol 45 min prior to maturation (t = 5). (a) Flow cytometric analysis of CD83 and costimulatory molecules either on day 5, when DCs were immature [immature DCs (iDCs)], or on day 7, 48 hr after the addition of lipopolysaccharide (LPS) [mature DCs (mDCs)]. The dashed line represents isotypic controls and the solid line represents the depicted surface marker. The first number in brackets shows the percentage of positively stained cells, and the second shows the mean fluorescence intensity (MFI) values. Results are from one representative experiment out of four performed. (b) NF-κB translocation was assessed by confocal microscopy as described in the Materials and methods. A clear reduction in nuclear translocation of NF-κB can be seen with resveratrol-treated DCs compared with mature controls. (c) Activation of NF-κB was determined as the ratio of the MFIs of nucleus and cytoplasm. *, P ≤ 0·05; **, P ≤ 0·01; n.s., not significant.
In context of differentiation markers, resveratrol treatment from t = 0 caused a 3-fold down-regulation of CD1a on immature DCs (iDCs) (Fig. 2a). Resveratrol failed to retain up-regulation of CD14 (which is strongly expressed on monocytes) on either iDCs or mDCs (data not shown). When resveratrol was used at 50 μm, treated DCs did not up-regulate CD83, even 48 hr after stimulation with LPS. This inability to up-regulate CD83 was only evident for cells that were differentiated in the presence of resveratrol (Fig. 1a). Surface expression of HLA-DR molecules was down-regulated in DCs treated with resveratrol (data not shown).
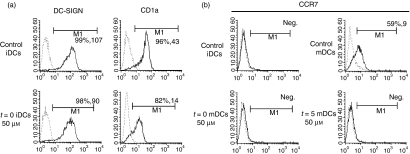
Figure 2.
Resveratrol prevents the expression of lipopolysaccharide (LPS)-induced CCR7 and down-regulates CD1a expression, while retaining the expression of DC-SIGN (CD209). Dendritic cells (DCs) were differentiated from human peripheral monocytes using granulocyte–macrophage colony-stimulating factor (GM-CSF) and interleukin (IL)-4 for 5 days and further stimulated to mature with LPS. In some cases, DCs were treated with 50 μm resveratrol (at this concentration the greatest effects were documented in the context of costimulatory, inhibitory and differentiation markers) either throughout the differentiation and maturation stage (t = 0) or just 45 min prior to maturation (t = 5). Cells were stained on day 5 [immature DCs (iDCs)] and day 7 [mature DCs (mDCs)] and analysed by flow cytometry. The dashed line represents isotypic controls and the solid line represents the depicted surface marker. (a) Surface expression of CD1a and DC-SIGN on control iDCs and resveratrol-treated differentiated iDCs is shown. (b) Expression of CCR7 on resveratrol-treated DCs at either t = 0 or t = 5 in comparison to control iDCs and mDCs. The first number in brackets shows the percentage of positively stained cells, and the second number shows the mean fluorescence intensity (MFI) values. One representative experiment out of three is shown.
We also determined whether resveratrol is capable of influencing the expression of the unique DC marker DC-SIGN. The addition of resveratrol to the culture medium at the start of differentiation (t = 0) affected DC-SIGN expression on iDCs only slightly (Fig. 2a). Interestingly, treatment of DCs with resveratrol, either at t = 0 or just prior to maturation with LPS, resulted in the retention of higher levels of DC-SIGN on the treated DCs than on non-treated controls stimulated to mature with LPS (data not shown).
The ability of resveratrol-treated DCs to translocate NF-κB after activation was assessed. Resveratrol treatment of DCs 45 min prior to LPS stimulation greatly reduced nuclear translocation of NF-κB p65. Furthermore, the ability of resveratrol to inhibit NF-κB was much greater in cells that were treated continuously during differentiation from day 0. The average ratios of nuclear to cytoplasmic staining of NF-κB p65 were 5·56 for control mDCs, 0·84 for control iDCs, 2·02 for t = 0/50 μmmDCs, and 3·21 for t = 5/50 μm mDCs (Fig 1b,c).
Resveratrol up-regulates inhibitory molecules ILT3 and ILT4, but does not induce expression of inhibitory HLA-G
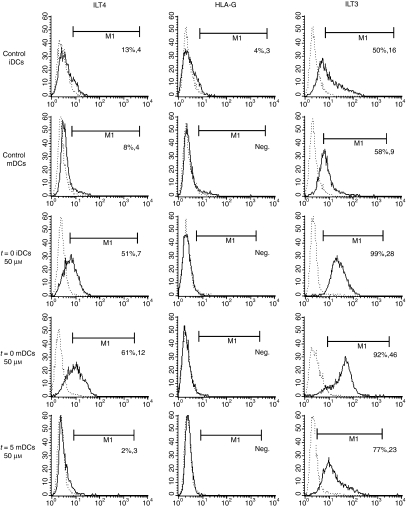
The immunoglobulin-like transcripts ILT3 and ILT4 are among the few well-established markers of TDCs. HLA-G can also be expressed by antigen-presenting cells (APCs) and has been shown to modulate T-cell proliferation through interaction with ILT4 and ILT2.24 We measured the expression of both ILT3 and ILT4, along with expression of HLA-G on treated DCs and controls. DCs were treated with resveratrol, either at t = 0 or t = 5. Compared with the down-regulation of costimulatory molecules, a quantitatively similar but inverse pattern was observed with ILT3 and ILT4. Resveratrol induced extensive up-regulation of ILT3 when present during the differentiation of DCs (Fig. 3). Expression of ILT3 was 5-fold higher than in control mDCs for t = 0-treated DCs. The expression of ILT4 on APCs is usually low or absent, but can be induced under certain conditions.25 Resveratrol-treated DCs on day 5 prior to stimulation were not able to up-regulate ILT4, so could not control iDCs or control mDCs. However, when resveratrol was present throughout the differentiation of DCs, a marked induction of ILT4 expression was seen (Fig. 3). Interestingly, resveratrol treatment did not result in any up-regulation of HLA-G, on either immature or LPS-stimulated DCs, using either the t = 0 or the t = 5 treatment regimen.
Figure 3.
Resveratrol treatment induces expression of inhibitory molecules immunoglobulin-like transcript 3 (ILT3) and ILT4 on dendritic cells (DCs), while it does not affect the expression of human leucocyte antigen (HLA)-G. DCs were prepared from human monocytes as described in the Materials and methods. Resveratrol was used in some cases to treat DC cultures either from the start of differentiation and during maturation (t = 0) or just 45 min prior to maturation (t = 5) at concentrations of 10 and 50 μm, as depicted in the charts. Flow cytometric analysis was performed on cells stained with fluoroscently labelled anti-ILT3, anti-ILT4 and anti-HLA-G either on day 5 [immature DCs (iDCs)] or on day 7, 48 hr after lipopolysaccharide (LPS) activation (t = 5). Solid lines represent the surface marker in question, and dashed lines are isotypic controls. The first number in brackets is the percentage of positively stained cells, and the second number represents the mean fluorescence intensity (MFI) value. One representative experiment out of four performed is shown.
Resveratrol-treated DCs are weak inducers of allogeneic T-cell proliferation
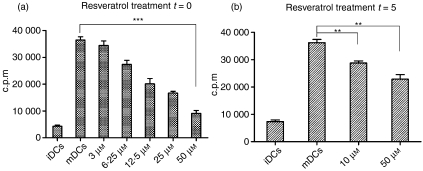
Mature DCs possess a remarkable capacity to induce proliferation in responding T cells. When resveratrol was used to treat DC cultures, there was a dose-dependent decrease in the ability of the treated DCs to stimulate allogeneic whole CD4+ T cells compared with controls (Fig. 4a,b). A suppressive effect of resveratrol on DC functionality was evident and, to a great extent, was exerted during their differentiation from monocytes. DCs treated from t = 0 were severely impaired in the attainment of their immunostimulatory capacity after LPS-induced activation (Fig. 4a). When 50 μm resveratrol was used to treat DCs, such cells had a 4-fold reduced immunostimulatory capacity (36410 ± 1224 and 9108 ± 1047 counts per minute (c.p.m.) for control mDCs and t = 0/50 μm DCs, respectively; P < 0·0001, n = 3). When t = 5 DCs were used, their ability to develop immunostimulatory capacity was lowered, but remained relatively high compared with DCs treated from the start of differentiation (36210 ± 1173 and 22850 ± 1666 c.p.m. for control mDCs and t = 5/50 μm DCs, respectively; P = 0·0028, n = 3), being just 1·5-fold lower than that of control mDCs (Fig. 4b).
Figure 4.
Resveratrol-treated dendritic cells (DCs) are poor stimulators of allogeneic CD4+ T cells. DCs were prepared as described in the Materials and methods and in some cultures resveratrol was added at various concentrations (from 3 to 50 μm), depending on the experiment. Treated DCs and controls were used to stimulate allogeneic T cells for 4 days, when [3H]thymidine was added for 18 hr, and proliferation was measured by liquid scintillation counting on day 5. (a) Resveratrol can greatly suppress the ability of lipopolysaccharide (LPS)-activated DCs to stimulate CD4+ T cells in mixed lymphocyte reactions (MLRs) when added at t = 0, as described above. At 50 μm concentrations, DCs were approximately four times less capable of inducing proliferation in responding T cells compared with controls (P < 0·0001). (b) Control DCs and DCs treated just prior to maturation were used as stimulators. Resveratrol suppressed the stimulatory capacity of DCs but only by about 40% in comparison to cases where it was already present during DC differentiation. Data represent the mean ± standard deviation of three independent experiments. *, P ≤ 0·05; **, P ≤ 0·01; ***, P ≤ 0·001 (Student’s t-test for independent samples).
DCs treated with resveratrol completely lose the ability to produce proinflammatory IL-12p70 and display increased IL-10 production upon stimulation with LPS
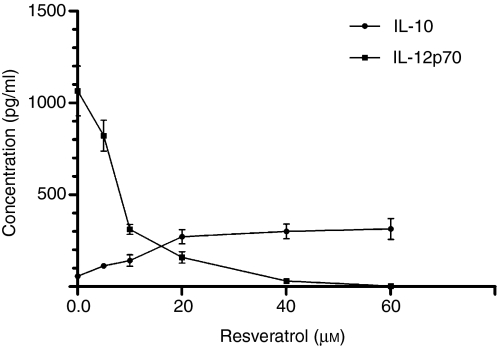
As DCs that differentiated and matured in the presence of resveratrol displayed several tolerogenic characteristics in the context of phenotype and functionality, it was important to determine the nature of their cytokine production profile. In order to determine how resveratrol treatment influences the ability of DCs to produce either proinflammatory IL-12p70 or immunosuppressive IL-10, DCs were differentiated in the presence or absence of various concentrations of resveratrol. After 5 days, DCs were stimulated to mature with LPS for a further 2 days, using the same resveratrol concentrations as during differentiation, and supernatants were analysed for the presence of IL-12p70 and IL-10. While control iDCs did not produce either IL-12p70 or IL-10 after 48 hr (data not shown), LPS-stimulated cells produced large quantities of IL-12p70 (1016·43 ± 86·31 pg/ml; n = 2), but only minor amounts of IL-10 (56·37 ± 12·27; n = 2) (Fig. 5). Resveratrol greatly reduced the ability of DCs to produce IL-12p70 after LPS stimulation, even at low doses. DCs treated with 50 μm resveratrol produced no detectable amounts of IL-12p70. However, resveratrol affected DCs by significantly increasing their IL-10 production (280·42 ± 23·00 pg/ml; n = 2). Figure 5 shows dose-dependent inhibition by resveratrol of the ability of DCs to produce IL-12p70, while at the same time greatly enhancing their capacity to produce IL-10.
Figure 5.
Levels of interleukin (IL)-12p70 and IL-10 were determined in supernatants of resveratrol-treated dendritic cells (DCs) and controls after 48 hr of lipopolysaccharide (LPS) stimulation. DCs were prepared as described in the Materials and methods and treated with various doses of resveratrol (from 3 to 50 μm) from t = 0. Even at low doses, resveratrol markedly inhibited the ability of DCs to produce IL-12p70, and completely inhibited LPS-induced IL-12p70 production at 50 μm. Simultaneously with dose-dependent inhibition of IL-12p70, treated DCs dose-dependently produced more IL-10. Data presented are the mean ± standard deviation of three independent experiments.
Resveratrol generates tolerogenic DCs capable of inducing allogeneic IL-10-secreting CD4+ T cells but not CD25+ FoxP3+ T cells
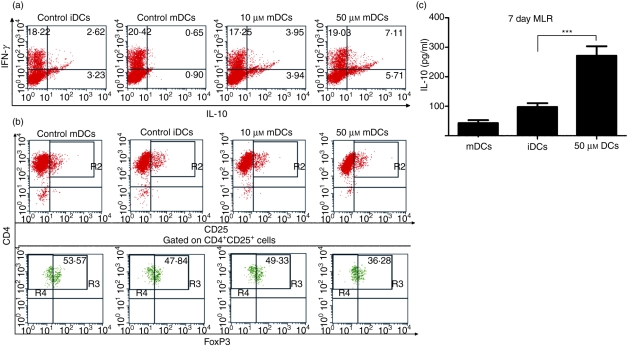
Dendritic cells are known for their functional plasticity in inducing various outcomes of immune responses. Treated DCs and untreated controls were used as stimulators in 14-day mixed co-cultures with naïve CD4+ T cells. Cytokine profiles of alloreactive T cells at the end of stimulation were analysed by intracellular cytokine staining. Mature DCs induced strong differentiation of naïve CD4+ T cells into Th1 cells that mainly produced IFN-γ and almost no IL-10 (Fig. 6a). Immature DCs were able to generate a high percentage of IL-10-secreting T cells, along with fewer cells producing IFN-γ. The use of resveratrol-treated DCs as stimulators produced T-cell populations with even more cells capable of producing IL-10 than use of iDCs (Fig. 6a). In addition to intracellular cytokine staining, IL-10 was measured by ELISAs in supernatants after 7 days of stimulation with either control or treated DCs (Fig. 6b). DCs treated with resveratrol were capable of inducing T cells that produced significantly greater amounts of IL-10 than those induced by iDCs, even after a short (7-day) period of stimulation.
Figure 6.
Resveratrol treatment generates dendritic cells (DCs) capable of inducing allogeneic interleukin (IL)-10-secreting CD4+ T cells, but not FoxP3+ T cells. DCs were prepared as described in the Materials and methods and then left untreated or treated with various doses of resveratrol during their differentiation and maturation stages. (a) We analysed FoxP3 expression in CD4 T cells by flow cytometry staining. After 5 days, cells were collected and stained for CD4 and CD25 and intracellularly for expression of FoxP3. Numbers in quadrants represent the percentage of positive cells (Fig. 6a,b). (b) DCs were used in allogeneic mixed cultures with CD4+ CD45RA+ naïve T cells for 14 days, with the addition of 20 U/ml of IL-2 on day 7. After 14 days, cells positive for interferon (IFN)-γ and IL-10 were determined by intracellular staining with anti-IFN-γ and anti-IL-10 and analysed by flow cytometry. (c) In parallel experiments, to determine earlier cytokine profiles and to quantitatively analyse cytokine production, CD4+ cells were collected after 7 days and stimulated for 24 hr with ionomycin and phorbol myristate acetate (PMA). After this time, supernatants were analysed for IL-10. DCs treated with 50 μm resveratrol induced T cells producing significantly greater quantities of IL-10 than controls. ***, P ≤ 0·001.
The ability of resveratrol-treated DCs to induce regulatory T cells expressing the transcription factor FoxP3 was assessed. Treated and untreated DCs were co-cultured with alloreactive naïve CD4+ T cells for 5 days. At the end of stimulation, the cells were stained for surface markers CD4 and CD25, as well as intracellularly for FoxP3 (Fig. 6c). mDCs induced the largest numbers of cells expressing FoxP3. Immature and resveratrol-treated DCs induced fewer FoxP3-expressing cells. Interestingly, DCs treated with 50 μm resveratrol were the least capable of inducing FoxP3 expression.
Discussion
In the present study, the tolerogenicity induced in DCs by resveratrol is clearly shown to be the result not merely of the blocking of maturation, but of the redirection of the differentiation of DCs from monocytes, which in turn leads to alternative activation by maturation signals. This tolerogenic effect was much greater when DCs had already differentiated in the presence of resveratrol. DCs treated from t = 0 expressed much less CD40, CD80 and CD86 than DCs treated prior to activation (Fig. 1a). Additionally, the expression of the CD83 marker, which is a hallmark of DC maturation, was completely inhibited only with t = 0 treated DCs. Immature DCs are CD1a++ and CD14−, and corticosteroids22 and 1α,25-dihydroxy-vitaminD3,23 two drugs known to affect DC differentiation, both retain CD14 expression on DCs. Resveratrol treatment from t = 0 reduced the level of CD1a on iDCs, although CD14 expression was not retained and was present on only a small number of cells (7% of mDCs; 19% of iDCs). As monocytes are precursors of both macrophages and DCs, the latter indicates that a shift of differentiation towards macrophages does not occur. To further confirm that treated monocytes actually differentiate into DCs, we assessed the expression of DC-SIGN (Fig. 2a), a C-type lectin selectively expressed by DCs. Resveratrol did not interrupt the expression of DC-SIGN during DC differentiation. Interestingly, although at first resveratrol treatment appeared to delay DC maturation and preserve the state of immaturity, the endocytic capacity of treated DCs was very low and not at all different from that of fully mature DCs (data not shown). This suggested that resveratrol, rather than a blocking of maturation, causes a divergence in the differentiation and maturation process that results in alternatively activated DCs or TDCs.
Surface molecules ILT3 and ILT4 transduce inhibitory signals via their cytoplasmic immunoreceptor tyrosine-based inhibitory motifs (ITIMs), and their expression is repeatedly associated with TDCs.21 When DCs were treated with resveratrol, their maturation with LPS resulted in extensive up-regulation of both ILT3 and ILT4. The extent of change in expression was much more apparent when resveratrol was present during differentiation (Fig. 3). Furthermore, ILT4 expression was generally negative for control iDCs and mDCs and did not occur on t = 5 treated mDCs, while DCs treated at t = 0 showed marked induction of ILT4. Interestingly, resveratrol failed to induce any up-regulation of HLA-G (Fig. 3).
The inhibitory effect of resveratrol on the expression of major histocompatibility complex (MHC) class II proteins was significant; however, the effect on HLA-DR expression was similar for the t = 0 and t = 5 treatments, in contrast to the much greater effects found with the t = 0 treatment for costimulatory and inhibitory molecules (data not shown). The reason for this could be that expression of costimulatory and ILT3 and ILT4 molecules is strongly associated with NF-κB activity,19 and, although NF-κB is known to have a direct role in HLA-DR expression,26 LPS-induced MHC class II expression was recently shown to be dependent mainly on AP-1.27 TNF-induced AP-1 activity is known to be inhibited by resveratrol treatment.12 NF-κB, in contrast to AP-1 is proven to be important already in the differentiation process of DCs. Thus, if the expression of HLA-DR in DCs is indeed dependent mainly on AP-1, this would explain why resveratrol's effect on HLA-DR expression is similar for both treatment regimens. .
Cytoskeletal changes accompanying DC maturation are shown through the formation of dendrites and loss of podosomes. Podosome and dendrite formation is regulated by CDc42 RhoGTPase28 and resveratrol has been shown to inhibit CDc42 at concentrations similar to those used here.29 Resveratrol treatment had a large negative impact on the number of dendrites formed on DCs after LPS stimulation (data not shown).
When DCs mature, they reduce expression of their tissue-homing receptors and up-regulate CCR7, which directs them to nearby lymph nodes. We analysed the expression of CCR7 on DCs treated and untreated with resveratrol. DCs stimulated with LPS after resveratrol treatment, either at t = 0 or at t = 5, completely failed to up-regulate CCR7. To assess whether resveratrol can influence production of T-cell-attracting factors by DCs, we performed chemotaxis assays using supernatants of treated or untreated DCs. DCs treated with resveratrol attracted significantly fewer CD4+ T cells than control mDCs or even iDCs (data not shown).
Secretion of bioactive IL-12p70 by DCs is the key to inducing a strong Th1 polarized immune response, and most immunosuppressive drugs that affect DC maturation have been reported to suppress production of IL-12.20 In contrast, DCs that produce significant levels of IL-10 with low or zero production of IL-12p70 have been reported to induce regulatory T cells.15 When treated with various concentrations of resveratrol, DCs dose-dependently produced less IL-12p70 after LPS activation. Additionally, the DCs became more proficient at producing IL-10, especially at higher drug concentrations. Thus, our results clearly demonstrate that resveratrol can create DCs with a potentially tolerogenic cytokine profile.
To determine whether resveratrol generates TDCs capable of inducing regulatory T cells, we allowed DCs to differentiate and mature in the presence of resveratrol and used them to stimulate allogeneic naïve CD4+ T cells. As iDCs can generate IL-10-producing T cells if multiple stimulations are used over a long cultivation time, we performed our co-cultures using a single stimulation for 2 weeks only. In this time, resveratrol-treated DCs were able to induce twice as many IL-10-secreting CD4+ T cells as iDCs (Fig. 6b). These results were confirmed using a different method and an even shorter cultivation time, by ELISAs of MLR supernatants after 1 week of stimulation, which showed significantly higher levels of IL-10 than controls (Fig. 6c).
NF-κB is a target for many pharmacological immunosuppressants. We studied nuclear translocation of NF-κB in controls and DCs treated at t = 0 or t = 5. Interestingly, the difference in NF-κB inhibition between the t = 0 and t = 5 treatments was very similar to the inhibition and induction patterns seen for costimulatory and inhibitory molecules, respectively. NF-κB translocation to the nucleus of resveratrol-treated cells was significantly lower for both t = 0 and t = 5 treatment regimens. There was, however, clearly less NF-κB staining in the nucleus for t = 0 than for t = 5 treated cells (Fig. 1b,c). NF-κB activity is stimulated by various signals which lead to activation of the kinases IkappaB kinase (IKK), which then release the p50 and p65 NF-κB subunits into the nucleus.30 Phosphorylation of p65 NF-κB, independent of pathways that lead to IκB degradation, is also important for its activity, and previous studies have shown that it can be controlled by a number of factors such as protein kinase C (PKC)31 and TNF receptor-associated factor (TRAF) family member associated NF kappa B activator (TANK) binding kinase 1 (TBK1).32 Phosphorylation of the p50 subunit, however, can be phosphorylated by IL-1-stimulated phosphoinositol-3-kinase (PI3K).33 In recent years, a number of factors regulating NF-κB activity were also shown to be crucial for DC differentiation. Specific inhibition of PKC has been shown to block cytokine-mediated DC differentiation from primary human monocytes as well as CD34+ hematopoietic cells (HPC).34 Activation of PKC triggers downstream targets NF-κB and mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinases (ERK) pathways, both of which have been implicated in DC differentiation.35 Furthermore, both PI3K/AKT (protein kinase B) signalling and ERK/mitogen-activated protein kinase kinase (MEK or MAP2K) signalling were found to be critical for proper differentiation of monocytes into DCs.36 Resveratrol affects all the above-mentioned targets, namely PKC,37 PI3K,38 AKT, ERK,39 TBK140 and NF-κB alone via IKK.41
In summary, our results provide a possible explanation that the combined effects of resveratrol on skewed DC differentiation, inhibition of crucial signalling pathways involved in NF-κB regulation independent of the IκB degradation pathway, together with inhibition of Toll-like receptor (TLR)-mediated NF-κB activation result in more extended tolerogenic effect than it would be seen merely from blockade of maturation. However, most protocols describing TDC generation are based on interfering with the maturation stage of DCs. Use of more specific inhibitors to alter important biological checkpoints in DC differentiation could suggest new strategies for the generation of stable TDCs and presents numerous subjects for future studies.
Acknowledgments
This work was supported by a project grant (no. L1-6295-3011-06) and by a scholarship (no. 1000-05-310105) both from the Slovenian Research Agency.
Disclosures
The authors have no financial conflict of interest.
References
- 1.Hung LM, Chen JK, Huang SS, Lee RS, Su MJ. Cardioprotective effect of resveratrol, a natural antioxidant derived from grapes. Cardiovasc Res. 2000;47:549–55. doi: 10.1016/s0008-6363(00)00102-4. [DOI] [PubMed] [Google Scholar]
- 2.Lu R, Serrero G. Resveratrol, a natural product derived from grape, exhibits antiestrogenic activity and inhibits the growth of human breast cancer cells. J Cell Physiol. 1999;179:297–304. doi: 10.1002/(SICI)1097-4652(199906)179:3<297::AID-JCP7>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- 3.Mitchell SH, Zhu W, Young CY. Resveratrol inhibits the expression and function of the androgen receptor in LNCaP prostate cancer cells. Cancer Res. 1999;59:5892–5. [PubMed] [Google Scholar]
- 4.Hwang JT, Kwak DW, Lin SK, Kim HM, Kim YM, Park OJ. Resveratrol induces apoptosis in chemoresistant cancer cells via modulation of AMPK signaling pathway. Ann N Y Acad Sci. 2007;1095:441–8. doi: 10.1196/annals.1397.047. [DOI] [PubMed] [Google Scholar]
- 5.Narayanan NK, Nargi D, Randolph C, Narayanan BA. Liposome encapsulation of curcumin and resveratrol in combination reduces prostate cancer incidence in PTEN knockout mice. Int J Cancer. 2009;125:1–8. doi: 10.1002/ijc.24336. [DOI] [PubMed] [Google Scholar]
- 6.Martin AR, Villegas I, La Casa C, de la Lastra CA. Resveratrol, a polyphenol found in grapes, suppresses oxidative damage and stimulates apoptosis during early colonic inflammation in rats. Biochem Pharmacol. 2004;67:1399–410. doi: 10.1016/j.bcp.2003.12.024. [DOI] [PubMed] [Google Scholar]
- 7.Birrell MA, McCluskie K, Wong S, Donnelly LE, Barnes PJ, Belvisi MG. Resveratrol, an extract of red wine, inhibits lipopolysaccharide induced airway neutrophilia and inflammatory mediators through an NF-kappaB-independent mechanism. FASEB J. 2005;19:840–1. doi: 10.1096/fj.04-2691fje. [DOI] [PubMed] [Google Scholar]
- 8.Elmali N, Esenkaya I, Harma A, Ertem K, Turkoz Y, Mizrak B. Effect of resveratrol in experimental osteoarthritis in rabbits. Inflamm Res. 2005;54:158–62. doi: 10.1007/s00011-004-1341-6. [DOI] [PubMed] [Google Scholar]
- 9.Wu SL, Yu L, Meng KW, Ma ZH, Pan CE. Resveratrol prolongs allograft survival after liver transplantation in rats. World J Gastroenterol. 2005;11:4745–9. doi: 10.3748/wjg.v11.i30.4745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu SL, Pan CE, Yu L, Meng KW. Immunosuppression by combined use of cyclosporine and resveratrol in a rat liver transplantation model. Transplant Proc. 2005;37:2354–9. doi: 10.1016/j.transproceed.2005.03.112. [DOI] [PubMed] [Google Scholar]
- 11.Estrov Z, Shishodia S, Faderl S, Harris D, Van Q, Kantarjian HM, Talpaz M, Aggarwal BB. Resveratrol blocks interleukin-1beta-induced activation of the nuclear transcription factor NF-kappaB, inhibits proliferation, causes S-phase arrest, and induces apoptosis of acute myeloid leukemia cells. Blood. 2003;102:987–95. doi: 10.1182/blood-2002-11-3550. [DOI] [PubMed] [Google Scholar]
- 12.Manna SK, Mukhopadhyay A, Aggarwal BB. Resveratrol suppresses TNF-induced activation of nuclear transcription factors NF-kappa B, activator protein-1, and apoptosis: potential role of reactive oxygen intermediates and lipid peroxidation. J Immunol. 2000;164:6509–19. doi: 10.4049/jimmunol.164.12.6509. [DOI] [PubMed] [Google Scholar]
- 13.Finkelman FD, Lees A, Birnbaum R, Gause WC, Morris SC. Dendritic cells can present antigen in vivo in a tolerogenic or immunogenic fashion. J Immunol. 1996;157:1406–14. [PubMed] [Google Scholar]
- 14.Lutz MB, Schuler G. Immature, semi-mature and fully mature dendritic cells: which signals induce tolerance or immunity? Trends Immunol. 2002;23:445–9. doi: 10.1016/s1471-4906(02)02281-0. [DOI] [PubMed] [Google Scholar]
- 15.Rutella S, Danese S, Leone G. Tolerogenic dendritic cells: cytokine modulation comes of age. Blood. 2006;108:1435–40. doi: 10.1182/blood-2006-03-006403. [DOI] [PubMed] [Google Scholar]
- 16.Sato K, Yamashita N, Baba M, Matsuyama T. Modified myeloid dendritic cells act as regulatory dendritic cells to induce anergic and regulatory T cells. Blood. 2003;101:3581–9. doi: 10.1182/blood-2002-09-2712. [DOI] [PubMed] [Google Scholar]
- 17.Steinbrink K, Wolfl M, Jonuleit H, Knop J, Enk AH. Induction of tolerance by IL-10-treated dendritic cells. J Immunol. 1997;159:4772–80. [PubMed] [Google Scholar]
- 18.Faunce DE, Terajewicz A, Stein-Streilein J. Cutting edge: in vitro-generated tolerogenic APC induce CD8+ T regulatory cells that can suppress ongoing experimental autoimmune encephalomyelitis. J Immunol. 2004;172:1991–5. doi: 10.4049/jimmunol.172.4.1991. [DOI] [PubMed] [Google Scholar]
- 19.Chang CC, Ciubotariu R, Manavalan JS, et al. Tolerization of dendritic cells by T(S) cells: the crucial role of inhibitory receptors ILT3 and ILT4. Nat Immunol. 2002;3:237–43. doi: 10.1038/ni760. [DOI] [PubMed] [Google Scholar]
- 20.Hackstein H, Thomson AW. Dendritic cells: emerging pharmacological targets of immunosuppressive drugs. Nat Rev Immunol. 2004;4:24–34. doi: 10.1038/nri1256. [DOI] [PubMed] [Google Scholar]
- 21.Wu J, Horuzsko A. Expression and function of immunoglobulin-like transcripts on tolerogenic dendritic cells. Hum Immunol. 2009;70:353–6. doi: 10.1016/j.humimm.2009.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Piemonti L, Monti P, Allavena P, Sironi M, Soldini L, Leone BE, Socci C, Di Carlo V. Glucocorticoids affect human dendritic cell differentiation and maturation. J Immunol. 1999;162:6473–81. [PubMed] [Google Scholar]
- 23.Berer A, Stockl J, Majdic O, Wagner T, Kollars M, Lechner K, Geissler K, Oehler L. 1,25-Dihydroxyvitamin D(3) inhibits dendritic cell differentiation and maturation in vitro. Exp Hematol. 2000;28:575–83. doi: 10.1016/s0301-472x(00)00143-0. [DOI] [PubMed] [Google Scholar]
- 24.Naji A, Durrbach A, Carosella ED, Rouas-Freiss N. Soluble HLA-G and HLA-G1 expressing antigen-presenting cells inhibit T-cell alloproliferation through ILT-2/ILT-4/FasL-mediated pathways. Hum Immunol. 2007;68:233–9. doi: 10.1016/j.humimm.2006.10.017. [DOI] [PubMed] [Google Scholar]
- 25.Vlad G, Piazza F, Colovai A, Cortesini R, Della Pietra F, Suciu-Foca N, Manavalan JS. Interleukin-10 induces the upregulation of the inhibitory receptor ILT4 in monocytes from HIV positive individuals. Hum Immunol. 2003;64:483–9. doi: 10.1016/s0198-8859(03)00040-5. [DOI] [PubMed] [Google Scholar]
- 26.Lee KW, Lee Y, Kim DS, Kwon HJ. Direct role of NF-kappaB activation in Toll-like receptor-triggered HLA-DRA expression. Eur J Immunol. 2006;36:1254–66. [Google Scholar]
- 27.Casals C, Barrachina M, Serra M, Lloberas J, Celada A. Lipopolysaccharide up-regulates MHC class II expression on dendritic cells through an AP-1 enhancer without affecting the levels of CIITA. J Immunol. 2007;178:6307–15. doi: 10.4049/jimmunol.178.10.6307. [DOI] [PubMed] [Google Scholar]
- 28.Burns S, Thrasher AJ, Blundell MP, Machesky L, Jones GE. Configuration of human dendritic cell cytoskeleton by Rho GTPases, the WAS protein, and differentiation. Blood. 2001;98:1142–9. doi: 10.1182/blood.v98.4.1142. [DOI] [PubMed] [Google Scholar]
- 29.Azios NG, Krishnamoorthy L, Harris M, Cubano LA, Cammer M, Dharmawardhane SF. Estrogen and resveratrol regulate Rac and Cdc42 signaling to the actin cytoskeleton of metastatic breast cancer cells. Neoplasia. 2007;9:147–58. doi: 10.1593/neo.06778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vallabhapurapu S, Karin M. Regulation and function of NF-kappaB transcription factors in the immune system. Annu Rev Immunol. 2009;27:693–733. doi: 10.1146/annurev.immunol.021908.132641. [DOI] [PubMed] [Google Scholar]
- 31.Leitges M, Sanz L, Martin P, et al. Targeted disruption of the zetaPKC gene results in the impairment of the NF-kappaB pathway. Mol Cell. 2001;8:771–80. doi: 10.1016/s1097-2765(01)00361-6. [DOI] [PubMed] [Google Scholar]
- 32.Bonnard M, Mirtsos C, Suzuki S, et al. Deficiency of T2K leads to apoptotic liver degeneration and impaired NF-kappaB-dependent gene transcription. EMBO J. 2000;19:4976–85. doi: 10.1093/emboj/19.18.4976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Koul D, Yao Y, Abbruzzese JL, Yung WK, Reddy SA. Tumor suppressor MMAC/PTEN inhibits cytokine-induced NFkappaB activation without interfering with the IkappaB degradation pathway. J Biol Chem. 2001;276:11402–8. doi: 10.1074/jbc.M007806200. [DOI] [PubMed] [Google Scholar]
- 34.Cejas PJ, Carlson LM, Zhang J, et al. Protein kinase C betaII plays an essential role in dendritic cell differentiation and autoregulates its own expression. J Biol Chem. 2005;280:28412–23. doi: 10.1074/jbc.M500345200. [DOI] [PubMed] [Google Scholar]
- 35.Xie J, Wang Y, Freeman ME, 3rd, Barlogie B, Yi Q. Beta 2-microglobulin as a negative regulator of the immune system: high concentrations of the protein inhibit in vitro generation of functional dendritic cells. Blood. 2003;101:4005–12. doi: 10.1182/blood-2002-11-3368. [DOI] [PubMed] [Google Scholar]
- 36.Xie J, Qian J, Yang J, Wang S, Freeman ME, 3rd, Yi Q. Critical roles of Raf/MEK/ERK and PI3K/AKT signaling and inactivation of p38 MAP kinase in the differentiation and survival of monocyte-derived immature dendritic cells. Exp Hematol. 2005;33:564–72. doi: 10.1016/j.exphem.2005.03.001. [DOI] [PubMed] [Google Scholar]
- 37.Stewart JR, Ward NE, Ioannides CG, O’Brian CA. Resveratrol preferentially inhibits protein kinase C-catalyzed phosphorylation of a cofactor-independent, arginine-rich protein substrate by a novel mechanism. Biochemistry. 1999;38:13244–51. doi: 10.1021/bi990875u. [DOI] [PubMed] [Google Scholar]
- 38.Frojdo S, Cozzone D, Vidal H, Pirola L. Resveratrol is a class IA phosphoinositide 3-kinase inhibitor. Biochem J. 2007;406:511–8. doi: 10.1042/BJ20070236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Venkatachalam K, Mummidi S, Cortez DM, Prabhu SD, Valente AJ, Chandrasekar B. Resveratrol inhibits high glucose-induced PI3K/Akt/ERK-dependent interleukin-17 expression in primary mouse cardiac fibroblasts. Am J Physiol Heart Circ Physiol. 2008;294:H2078–87. doi: 10.1152/ajpheart.01363.2007. [DOI] [PubMed] [Google Scholar]
- 40.Youn HS, Lee JY, Fitzgerald KA, Young HA, Akira S, Hwang DH. Specific inhibition of MyD88-independent signaling pathways of TLR3 and TLR4 by resveratrol: molecular targets are TBK1 and RIP1 in TRIF complex. J Immunol. 2005;175:3339–46. doi: 10.4049/jimmunol.175.5.3339. [DOI] [PubMed] [Google Scholar]
- 41.Kundu JK, Shin YK, Kim SH, Surh YJ. Resveratrol inhibits phorbol ester-induced expression of COX-2 and activation of NF-kappaB in mouse skin by blocking IkappaB kinase activity. Carcinogenesis. 2006;27:1465–74. doi: 10.1093/carcin/bgi349. [DOI] [PubMed] [Google Scholar]