Abstract
Organic selenium compounds are known to prevent certain cancers although mechanisms may be complex. A widely-held view is that selenium compounds can induce apoptosis in cancer cells, or more precisely, in aberrant cells that are undergoing clonal evolution somewhere along the carcinogenesis process. There are at least 20 different selenium compounds, inorganic as well as organic, that have been used in various published studies. Extrapolation between studies should therefore be undertaken with caution. Similarly, it will be important to ascertain the physiological relevance of the selenium concentrations used in some studies. While cancer prevention by selenium is well-established, recently, organic selenium in the form of pure seleno-L-methionine (SeMet) has been used in combination with cancer chemotherapy drugs. SeMet can induce a DNA repair response in some cell types including bone marrow. Cancer cells generally lack a SeMet-inducible DNA repair response. Thus, SeMet appears to selectively regulate a DNA repair pathway and thereby potentially alter responses to cancer chemotherapy drugs. The specific pathway implicated, nucleotide excision DNA repair (NER) is required for repair of cisplatin or carboplatin DNA damage relevant to chemotherapy. Moreover, some studies have implicated NER as a factor in carcinogenesis processes. Thus, the capacity of SeMet to selectively regulate NER may prove useful in both therapeutic and preventive contexts.
Keywords: Carboplatin, Cisplatin, DNA-repair, Selenium
Much confusion arises when discussing selenium compounds owing to the diverse chemical forms that have been studied. Generally, selenium compounds fall into two categories, organic and inorganic forms. SeMet is a prototype organic form that can be utilized as a seleno-amino-acid after being converted to selenocysteine. A fraction of SeMet that is not converted to selenocysteine is metabolized to methylselenol, probably also a relevant to cancer prevention and treatment. The fraction of SeMet that is metabolized to methylselenol varies by tissue as the rate-limiting enzyme exhibits tissue-specific expression and/or enzyme activity. Quite distinct from SeMet is inorganic selenium, exemplified by sodium selenite. Sodium selenite is itself a DNA damaging agent that causes double-strand breaks similar to ionizing radiation (1). As such, sodium selenite is toxic to animals. Therefore, the first order of business is not to confound the two classes of selenium compounds. Methylselenol may be considered as yet a third class, exemplified by methyl-seleninic acid or MSA (2).
In the U.S. selenium deficiency is rare and serum selenium concentrations in the 1 μM range are not uncommon (3). Dietary selenium, mostly in the form of SeMet, is of sufficient abundance in the U.S. diet. Nonetheless, supplementation of dietary selenium with 50-200 μg selenium per day can delay or prevent prostate cancers (4-6). The over-the-counter selenium supplements sold in drug stores are however usually not in the form of pure SeMet. To underscore the safety of SeMet in pure form, SeMet is now being used in human patients undergoing chemotherapy (7-9). Oral administration of SeMet of 3200 μg/day has been achieved in these patients, resulting in a serum selenium concentration of over 15 μM (5-9). The 15 μM threshold was based on studies in mice (10). However, the long-term effects of such high serum selenium are not known. The idea is to give SeMet concurrent with chemotherapy, then discontinue SeMet after the rounds of chemotherapy are complete.
Agents that enhance the selectivity of cancer chemotherapy have been eagerly sought. Typically, dose-limiting toxicity to bone marrow and other tissues is an impediment to successful chemotherapy. Numerous approaches have included agents that protect bone marrow, or agents that sensitize cancer cells. An agent that did both, protect bone marrow and sensitize cancer cells would be highly valuable. Studies in mice have shown that SeMet holds promise as such an agent. First, it is well-tolerated and does not cause toxicity by itself. It is now a subject of human clinical trials (9). Second, in mice, the maximum tolerated dose (MTD) of chemotherapy drugs was doubled and even tripled when combined with SeMet (10). Third, distinct anticancer effects are attributed to SeMet either alone or in combination with chemotherapy drugs in both human and mouse studies (7-10).
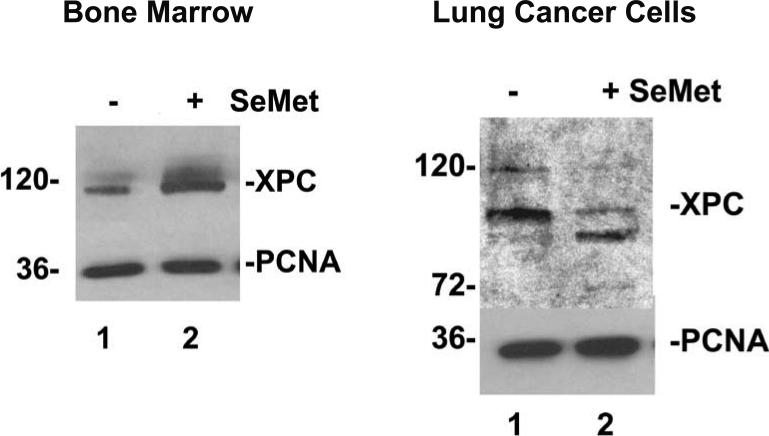
Most cancer chemotherapy drugs are DNA-damaging agents. Therefore, the ability to modify or modulate DNA repair may be an important and novel adjunct to chemotherapy. To be effective, SeMet must be given one week prior to chemotherapy and daily concurrent with chemotherapy (7-10). For this reason we have emphasized the SeMet pre-treatment step (7-10). We have shown that SeMet, during the pre-treatment step, regulates the cellular concentration of XPC protein, the rate-limiting protein for nucleotide excision DNA repair (NER). XPC is required for DNA damage recognition and repair of 1,2 and 1,3 platinum diadducts caused by cisplatin or carboplatin. Importantly, XPC is increased in bone marrow but decreased in several cancer cell lines tested (11; and unpublished findings). NER corresponds to XPC protein expression (11; and Figure 1).
Figure 1. SeMet treatment, 15 μM, 15 hrs, leads to XPC protein stabilization or induction in mouse bone marrow (left) but not in H1299 human lung cancer cells (right).
In fact, SeMet may destabilize XPC in some cancer cell types. The idea is that SeMet pre-treatment “sets the stage” for DNA-damaging treatments by altering XPC concentrations and hence the relative NER rates in normal cells and in cancer cells. Bone marrow was protected from carboplatin while cancer cells were sensitive to carboplatin. Adapted from JL Fischer et al, references 11 and 12.
One factor that regulates XPC is the p53 tumor suppressor, a transcription factor that regulates over 100 genes (12-14). At least in some cell types, NER corresponds to p53 functional status (15-17). This is important because the majority of cancer cells are defective in p53, hence defective in NER. While p53 mutations occurring in human cancers are generally regarded as a bad thing, it is possible that the NER defect in at least some p53-mutant cancers could be exploited as a target. This idea has been around for some time (18) but receives only occasional attention from insightful individuals (19). It is likely that XPC is regulated by additional mechanisms however. More studies are needed in order to exploit XPC as a target in cancer therapy, most relevant to platinum compounds (Figure 2).
Figure 2. Diagram of selective DNA repair response evoked by SeMet.
One factor is p53 status, which is functional in normal cells, but mutant or nonfunctional in the majority of cancer cells. XPC is regulated by p53.
The ability of SeMet to regulate XPC and NER could be relevant to cancer prevention as well as cancer chemotherapy. Typically, NER is associated with UV-radiation as the prototype DNA-damaging agent, which leads to thoughts of skin cancer prevention. However, selenium did not appear to prevent skin cancer in humans (4). On the other hand, NER is a versatile DNA repair pathway and UV-radiation is not the only relevant DNA-damaging agent. Mice lacking Xpc genes develop spontaneous lung cancers, suggesting a role for XPC and NER in lung carcinogenesis even in the absence of exogenous DNA-damaging agents (20). Clinical data implicates NER in prostate carcinogenesis (21). At first, one might ask, where is the DNA damage? However an endogenous lesion, the cyclodeoxyadenine adduct, arises from oxidative stress and requires NER for its removal (22). The endogenous cyclodeoxyadenine adduct may thus be a good candidate whereby NER could prevent carcinogenesis. Would enhancing NER by SeMet increase the rate of removal of the cyclodeoxyadenine lesion? It has not been tested.
Many studies of cancer prevention have focused on apoptotic removal of aberrant cells, meaning those cells that have progressed to a certain degree along a carcinogenesis pathway. In this sense, there is a fine line between what constitutes cancer “prevention” and what constitutes cancer “therapy” because in either scenario, the goal is to kill pre-cancerous or cancerous cells. Such approaches fit well with the “clonal evolution” model of carcinogenesis, in which successive mutations and chromosomal aberrations propel cells toward cancer. However, the key word here is “clonal”. What about carcinogenic mechanisms that occur across a population of cells, termed “field carcinogenesis”? (23-25). DNA repair may constitute an early mechanism to prevent field carcinogenesis especially in situations where high levels of DNA damage are being incurred. Lung epithelium in smokers comes to mind. As such, DNA repair may contribute to the cancer preventive properties of selenium compounds.
Acknowledgement
Supported by NIH 5R01 HL086978 to M.L.S.
Footnotes
Conflicts of Interest
No potential conflicts of interest to disclose.
References
- 1.Letavayová L, Vlasáková D, Vlcková V, Brozmanová J, Chovanec M. Rad52 has a role in the repair of sodium selenite-induced DNA damage in Saccharomyces cerevisiae. Mutat Res. 2008;652:198–203. doi: 10.1016/j.mrgentox.2008.03.001. [DOI] [PubMed] [Google Scholar]
- 2.Hu H, Jiang C, Ip C, Rustum YM, Lu J. Methylseleninic acid potentiates apoptosis induced by chemotherapeutic drugs in androgen-independent prostate cancer cells. Clin Cancer Res. 2005;11:2379–88. doi: 10.1158/1078-0432.CCR-04-2084. [DOI] [PubMed] [Google Scholar]
- 3.Meuillet E, Stratton S, Prasad-Cherukuri D, et al. Chemoprevention of prostate cancer with selenium: an update on current clinical trials and preclinical findings. J. Cell Biochem. 2004;91:443–58. doi: 10.1002/jcb.10728. [DOI] [PubMed] [Google Scholar]
- 4.Clark LC, Combs GF, Jr, Turnbull BW, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA. 1996;276:1957–63. [PubMed] [Google Scholar]
- 5.Reid MC, Stratton MS, Lillico AJ, et al. A report of high-dose selenium supplementation: response and toxicities. J. Trace Elem Biol. 2004;18:69–74. doi: 10.1016/j.jtemb.2004.03.004. [DOI] [PubMed] [Google Scholar]
- 6.Reid MC, Duffield-Lillico AJ, Slate E, et al. The nutritional prevention of cancer: 400 mcg per day selenium treatment. Nutr Cancer. 2008;60:155–63. doi: 10.1080/01635580701684856. [DOI] [PubMed] [Google Scholar]
- 7.Fakih M, Cao S, Durrani FA, Rustum YM. Selenium protects against toxicity induced by anticancer drugs and augments antitumor activity: a highly selective, new, and novel approach to the treatment of solid tumors. Clin Colorectal Cancer. 2005;5:132–35. doi: 10.3816/ccc.2005.n.026. [DOI] [PubMed] [Google Scholar]
- 8.Fakih MG, Pendyala L, Smith PF, et al. A phase I and pharmacokinetic study of fixed-dose selenomethionine and irinotecan in solid tumors. Clin Cancer Res. 2006;12:1237–44. doi: 10.1158/1078-0432.CCR-05-2004. [DOI] [PubMed] [Google Scholar]
- 9.Fakih MG, Pendyala L, Brady W, et al. A Phase I and pharmacokinetic study of selenomethionine in combination with a fixed dose of irinotecan in solid tumors. Cancer Chemother Pharmacol. 2008;62:499–508. doi: 10.1007/s00280-007-0631-4. [DOI] [PubMed] [Google Scholar]
- 10.Cao S, Durrani FA, Rustum YM. Selective modulation of the therapeutic efficacy of anticancer drugs by selenium containing compounds against human tumor xenografts. Clin Cancer Res. 2004;10:2561–9. doi: 10.1158/1078-0432.ccr-03-0268. [DOI] [PubMed] [Google Scholar]
- 11.Fischer JL, Mihelc E, Pollok KE, Smith ML. Chemotherapeutic selectivity conferred by selenium: a role for p53-dependent DNA repair. Mol Cancer Ther. 2007;6:355–61. doi: 10.1158/1535-7163.MCT-06-0472. [DOI] [PubMed] [Google Scholar]
- 12.Fischer JL, Kumar MA, Day TW, et al. The Xpc gene markedly affects cell survival in mouse bone marrow. Mutagenesis. 2009;24:309–16. doi: 10.1093/mutage/gep011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Adimoolam S, Ford JM. p53 and DNA damage-inducible expression of the xeroderma pigmentosum group C gene. Proc Natl Acad Sci U S A. 2002;99:12985–90. doi: 10.1073/pnas.202485699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ford JM. Regulation of DNA damage recognition and nucleotide excision repair: another role for p53. Mutat Res. 2005;577:195–202. doi: 10.1016/j.mrfmmm.2005.04.005. [DOI] [PubMed] [Google Scholar]
- 15.Smith ML, Chen IT, Zhan Q, O'Connor PM, Fornace AJ., Jr Involvement of the p53 tumor suppressor in repair of u.v.-type DNA damage. Oncogene. 1995;10:1053–9. [PubMed] [Google Scholar]
- 16.Smith ML, Fornace AJ., Jr p53-mediated protective responses to UV irradiation. Proc Natl Acad Sci U S A. 1997;94:12255–7. doi: 10.1073/pnas.94.23.12255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Smith ML, Ford JM, Hollander MC, et al. p53-mediated DNA repair responses to UV radiation: studies of mouse cells lacking p53, p21, and/or gadd45 genes. Mol Cell Biol. 2000;20:3705–14. doi: 10.1128/mcb.20.10.3705-3714.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fan S, Smith ML, Rivet DJ, et al. Disruption of p53 sensitizes breast cancer MCF7 cells to cisplatin and pentoxifylline. Cancer Res. 1995;55:1649–54. [PubMed] [Google Scholar]
- 19.Gudkov A, Komarova EA. Dangerous habits of a security guard: the two faces of p53 as a drug target. Human Mol Genet. 2007;16:67–72. doi: 10.1093/hmg/ddm052. [DOI] [PubMed] [Google Scholar]
- 20.Hollander MC, Philburn RT, Patterson AD, et al. Deletion of XPC leads to lung tumors in mice and is associated with early events in human lung carcinogenesis. Proc Natl Acad Sci U S A. 2005;102:13200–5. doi: 10.1073/pnas.0503133102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hu JJ, Hall MC, Grossman L, et al. Deficient nucleotide excision repair capacity enhances human prostate cancer risk. Cancer Res. 2004;64:1197–1201. doi: 10.1158/0008-5472.can-03-2670. [DOI] [PubMed] [Google Scholar]
- 22.Brooks PJ, Wise DS, Berry DA, et al. The oxidative DNA lesion 8,5'-(S)-cyclo-2'-deoxyadenosine is repaired by the nucleotide excision repair pathway and blocks gene expression in mammalian cells. J Biol Chem. 2000;275:22355–62. doi: 10.1074/jbc.M002259200. [DOI] [PubMed] [Google Scholar]
- 23.Van Rees BP, Cleton-Jansen AM, Cense HA, et al. Molecular evidence of field cancerization in a patient with 7 tumors of the aerodigestive tract. Hum Pathol. 2000;31:269–71. doi: 10.1016/s0046-8177(00)80234-8. [DOI] [PubMed] [Google Scholar]
- 24.Kammori M, Poon SS, Nakamura, et al. Squamous cell carcinomas of the esophagus arise from a telomere-shortened epithelial field. Int J Mol Med. 2007;20:793–9. [PubMed] [Google Scholar]
- 25.McDonald SA, Greaves LC, Gutierrez-Gonzales L, et al. Mechanisms of field cancerization in the human stomach: the expansion and spread of mutated gastric stem cells. Gastroenterology. 2008;134:500–10. doi: 10.1053/j.gastro.2007.11.035. [DOI] [PubMed] [Google Scholar]