Abstract
In addition to its function as an excitatory neurotransmitter, glutamate plays a major role as an osmolyte within the central nervous system (CNS). Accordingly, mechanisms that regulate glutamate release and uptake are of physiological importance not only during conditions in which cell volume remains constant but also when cells are subjected to hypoosmotic stress. In the present study, the ability of muscarinic cholinergic receptors (mAChRs) to regulate the uptake of glutamate (monitored as d-aspartate) into human SH-SY5Y neuroblastoma cells under isotonic or hypotonic conditions has been examined. In isotonic media, agonist activation of mAChRs resulted in a significant increase (250–300% of control) in the uptake of d-aspartate and, concurrently, a cellular redistribution of the excitatory amino acid transporter 3 (EAAT3) to the plasma membrane. mAChR-mediated increases in d-aspartate uptake were potently blocked by the EAAT3 inhibitor l-β-threo-benzyl-aspartate. In hypotonic media, the ability of mAChR activation to facilitate d-aspartate uptake was significantly attenuated (40–50%), and the cellular distribution of EAAT3 was disrupted. Reduction of mAChR-stimulated d-aspartate uptake under hypoosmotic conditions could be fully reversed upon re-exposure of the cells to isotonic media. Under both isotonic and hypotonic conditions, mAChR-mediated increases in d-aspartate uptake depended on cytoskeletal integrity, protein kinase C and phosphatidylinositol 3-kinase activities, and the availability of intracellular Ca2+. In contrast, dependence on extracellular Ca2+ was observed only under isotonic conditions. The results suggest that, although the uptake of d-aspartate into SH-SY5Y cells is enhanced after mAChR activation, this process is markedly attenuated by hypoosmolarity.
The maintenance of cell volume is an ancient homeostatic mechanism that is of prime importance to the central nervous system (CNS) because of the restrictions imposed by the skull and the need to maintain correct spatial relationships between cells to avoid changes in neuronal excitability (Sykova, 2004). Cell swelling can occur under physiological conditions (after intense neuronal discharge) or result from pathological causes, such as a reduction in plasma osmolarity (hypoosmotic swelling). Although the renal system normally maintains plasma osmolarity within a very narrow range, this regulation can be compromised under pathological conditions, the most prevalent of which is hyponatremia, the most commonly diagnosed electrolyte disorder in clinical practice. Hyponatremia is defined as a reduction in the serum Na+ concentration from 145 to ≤136 mEq/l with severe cases resulting in up to 25% reductions in the concentration of serum Na+. Changes in brain cell volume (<5%) can have profound effects within the CNS and lead to neurological symptoms such as nausea, headache, coma, and, ultimately, respiratory arrest (Bhardwaj, 2006; Lien and Shapiro, 2007). Under hypoosmotic conditions, cells initially swell and then correct their volume through the release of both inorganic and organic osmolytes, a process that results in the exit of obligated water. Glutamate, taurine, and myoinositol represent the major organic osmolytes that, together with the inorganic osmolytes (K+ and Cl−), account for the bulk of the osmolytes used by neural cells. In response to a reduction in osmolarity most cells appear to extrude organic osmolytes and Cl− through a common volume-sensitive organic osmolyte and anion channel, whereas the release of K+ occurs via one or more K+ channels. Although volume control has previously been considered largely to be an intrinsic property of the cell, there is now abundant evidence to suggest that volume-dependent osmolyte release from a variety of neural and non-neural preparations can be enhanced after the activation of a wide spectrum of G protein-coupled receptors (GPCRs) (for reviews see Franco et al., 2008; Vazquez-Juarez et al., 2008; Fisher et al., 2008). Receptor activation appears to not only enhance the magnitude of osmolyte release but also to lower the osmotic threshold at which release occurs, thereby facilitating the ability of cells to respond to small, physiologically relevant reductions in osmolarity.
Although the net loss of an osmolyte from a cell maintained under hypoosmotic conditions depends on both its release and subsequent influx, the possibility that activation of GPCRs may regulate not only the efflux, but also the reuptake of osmolytes, has received little attention. In this context, we have recently demonstrated that the activation of muscarinic cholinergic receptors (mAChRs) present on human SH-SY5Y neuroblastoma cells results in not only a facilitation of the volume-dependent efflux of both taurine and K+ but also a regulation of the reuptake of both osmolytes. However, whereas mAChR activation results in an increase in the uptake of K+, and thereby its retention in cells, a decrease in the influx of taurine is observed, an effect that promotes the loss of this organic osmolyte from cells (Foster et al., 2008, 2009). Although an increase in the extracellular concentration of a relatively inert compound such as taurine might have minimal cellular consequences, a similar increase in the concentration of extracellular glutamate, a quantitatively important organic osmolyte in the CNS (Fisher et al., 2008), would be potentially deleterious because of its excitotoxic effects. Accordingly, in the present study we have evaluated the ability of osmolarity to regulate the uptake of glutamate into SH-SY5Y neuroblastoma, a model neuronal cell line. In contrast to our previous results obtained for taurine influx, we find that under isotonic conditions glutamate uptake (monitored as d-aspartate) is significantly enhanced after mAChR activation. This effect appears to be mediated primarily by a cellular redistribution to the plasma membrane of excitatory amino acid transporter 3 (EAAT3), a neuronal-specific transporter. The increase in d-aspartate uptake depended on the availability of extracellular and intracellular Ca2+, protein kinase C (PKC) and phosphatidylinositol 3-kinase (PI3K) activities, and the integrity of the cytoskeleton. Incubation of the cells under hypoosmotic conditions significantly attenuated mAChR-mediated d-aspartate uptake, an effect that could be reversed upon re-exposure of the cells to an isotonic medium. Although receptor-mediated increases in d-aspartate uptake monitored under hypoosmotic conditions also depended on the activities of PKC and PI3K and cytoskeletal integrity, there was little or no dependence on extracellular Ca2+. The results suggest that hypoosmolarity significantly attenuates the ability of mAChRs to facilitate glutamate accumulation in SH-SY5Y cells and provides further evidence for a role for GPCR activation in the regulation of osmolyte influx.
Materials and Methods
Materials.
d-[2,3-3H]Aspartic acid (23.0 Ci/mmol) and myo-[2-3H]inositol (15.0 Ci/mmol) were obtained from GE Healthcare (Chalfont St. Giles, UK). [3H]Quinuclidinyl benzilate (QNB; 50.5 Ci/mmol) was obtained from PerkinElmer Life and Analytical Sciences (Waltham, MA). Oxotremorine-M (Oxo-M), sphingosine 1-phosphate (S1P), thrombin, atropine, d-aspartic acid, wortmannin, colchicine, and l-β-threo-benzyl-aspartate (LβBA) were purchased from Sigma-Aldrich (St. Louis, MO). dl-threo-β-Benzyloxyaspartic acid (TBOA), dihydrokainic acid (DHK), and l-(−)-threo-3-hydroxyaspartic acid (L3HA) were obtained from Tocris Bioscience (Ellisville, MO). Thapsigargin, ionomycin, phorbol-12-myristate-13 acetate (PMA), cytochalasin D, and bisindolylmaleimide (BIM) 1 were obtained from Calbiochem (San Diego, CA). Lysophosphatidic acid (LPA) was purchased from Avanti Polar Lipids (Alabaster, AL). Dulbecco's modified Eagle's medium (DMEM; low glucose) and 50× penicillin/streptomycin were obtained from Invitrogen (Carlsbad, CA). Fetal calf serum was obtained from Lonza Walkersville Inc. (Walkersville, MD). Tissue culture supplies were obtained from Corning Inc. (Corning, NY), Starstedt (Newton, NC), and BD BioSciences (San Jose, CA). UniverSol was obtained from MP Biomedicals (Solon, OH).
Antibodies.
Rabbit anti-rat EAAC1 affinity pure IgG (EAAC11-A, recognizes the human homolog, EAAT3) was obtained from Alpha Diagnostic International (San Antonio, TX) and used at a 1:1000 dilution. Goat anti-early endosomal antigen (anti-EEA1) affinity-purified IgG (used at a 1:1000 dilution), goat anti-actin horseradish peroxidase (HRP)-conjugated polyclonal IgG (used at a 1:10,000 dilution), goat anti-rabbit HRP-conjugated IgG (used at a 1:10,000 dilution), and donkey anti-goat HRP-conjugated IgG (used at a 1:12,500 dilution) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). The following primary antibodies were not able to detect EAAT1 or EAAT2 in either SH-SY5Y cell lysate or subcellular fractions: guinea pig anti-EAAT1 (Millipore Corporation, Billerica, MA), guinea pig anti-EAAT2 (Millipore Corporation), goat anti-EAAT1 (Santa Cruz Biotechnology), and rabbit anti-EAAT1 (Santa Cruz Biotechnology).
Cell Culture Conditions.
Human SH-SY5Y neuroblastoma cells (passages 70–89) or C6 glioma cells (passages 46–48) were grown in tissue culture flasks (75 cm2/250 ml) in 20 ml of DMEM supplemented with 10% (v/v) fetal calf serum and 1% penicillin/streptomycin with the osmolarity of the media adjusted to 290 mOsM. Cells were grown at 37°C in a humidified atmosphere containing 10% CO2. The medium was aspirated, and cells were detached from the flask with a trypsin-versene mixture (Lonza Walkersville Inc.). Cells were then resuspended in DMEM/10% fetal calf serum with penicillin/streptomycin (290 mOsM) and subcultured into 35-mm, six-well culture plates at a density of 200 to 300,000 cells/well for 4 to 5 days. Cells that had reached 70 to 90% confluence with a protein content of ∼0.25 mg of protein/well were routinely used.
Measurement of d-Aspartate Uptake.
SH-SY5Y cells were washed once with 2 ml of isotonic buffer A [116 mM NaCl, 5.6 mM KCl, 2.2 mM CaCl2, 3.6 mM NaHCO3, 1 mM MgCl2, 30 mM HEPES (pH 7.4), and 1 mg/ml d-glucose, ∼290 mOsM] before assay. Cells were routinely incubated (unless stated otherwise) in 5 ml of hypotonic buffer A (230 mOsM; 90 mM NaCl) or isotonic buffer A (290 mOsM; adjusted by the addition of 60 mM sucrose to hypotonic buffer A to maintain a constant NaCl concentration) containing 10 μM d-[3H]aspartate (0.3 μCi/ml; used as a marker for l-glutamate) in the presence or absence of agonist at 37°C. The 5-ml assay volume (50 nmol of added d-aspartate) was chosen to minimize any dilution of specific activity caused by the release of intracellular glutamate during the uptake assay under hypotonic conditions according to the following calculations. In SH-SY5Y cells, the concentration of l-glutamate is 100 nmol/mg of protein (see Results). During a 10-min incubation period in the presence of Oxo-M under conditions of a 20% reduction in osmolarity, 5 to 10% of d-aspartate is released (Heacock et al., 2004). Assuming that d-aspartate release reflects that of l-glutamate, it can be calculated that ∼2 nmol of l-glutamate would be released, which would contribute <5% to the extracellular pool.
At the times indicated, the extracellular medium was aspirated, and cells were immediately washed with 2 ml of isotonic buffer A before lysis in 2 ml of 0.1 M NaOH. Aliquots of lysate (1 ml) were removed, and radioactivity was determined after the addition of 7.5 ml of UniverSol scintillation fluid and 50 μl of glacial acetic acid (to quench chemiluminescence). In all measurements, radioactivity associated with the cell monolayer at the zero time point was subtracted from the observed value. Protein contents of cell lysates were determined with a bicinchoninic acid protein assay reagent kit (Thermo-Fisher Scientific, Rockford, IL). Rates of d-aspartate uptake into SH-SY5Y cells were calculated as pmol/mg protein/min. Throughout the study, “basal” uptake of d-aspartate is defined as that which occurs at a specified osmolarity in the absence of an agonist. Because measurement of d-aspartate uptake in the presence of Oxo-M represents the sum of both the basal- and agonist-stimulated components, percentage changes in the rates of Oxo-M-stimulated d-aspartate uptake reported in this study are calculated on the basis of the net changes in the agonist-mediated component of uptake, i.e., d-aspartate uptake observed in the presence of Oxo-M minus the basal uptake observed under the same experimental conditions.
Glutamate Mass Measurements.
SH-SY5Y cells were washed once with 2 ml of isotonic buffer A and detached by the addition of 1 ml of Puck's D1 solution (Heacock et al., 2004). Six wells of detached cells were pooled and centrifuged (5 min at 1750g), and the resulting supernatant was aspirated. Cell pellets were lysed by the addition of 1 ml of boiling deionized water for 10 min with intermittent sonication. Intracellular glutamate was measured via a spectrophotometric method (Lund and Bergmeyer, 1986) using a glutamate assay kit (Sigma-Aldrich). Results are expressed as nmol endogenous glutamate/mg protein.
Measurement of Phosphoinositide Turnover.
To monitor phosphoinositide turnover, SH-SY5Y cells that had been prelabeled with 10 μCi/ml [3H]inositol for 96 h were washed with isotonic buffer A and then incubated in buffer of the osmolarity indicated containing 5 mM LiCl in the presence or absence of 100 μM Oxo-M. The accumulation of radiolabeled inositol phosphates present in the trichloroacetic acid cell lysates was determined as described previously (Thompson and Fisher, 1990).
Subcellular Fractionation.
A modified procedure of the method of Stadel et al. (1983) was used essentially as reported previously (Sorensen et al., 1997). Twenty flasks of SY5Y cells (five flasks/treatment) were washed with 5 ml of isotonic buffer A at 37°C before treatment in either hypotonic (230 mOsM; 90 mM NaCl) or isotonic buffer A (290 mOsM; rendered isotonic by the addition of 60 mM sucrose to hypotonic buffer) in the absence or presence of Oxo-M for 10 min. Reactions were terminated by aspiration of the media and the addition of 5 ml of ice-cold isotonic buffer A to the flasks that were maintained on ice. The buffer was then aspirated, and cells were detached by the addition of 5 ml of ice-cold Puck's D1 Solution (Heacock et al., 2004). Cells were then centrifuged at 1750g for 10 min at 4°C, and the supernatants were aspirated. Cell pellets were lysed by gentle homogenization in 10 ml of TE buffer (10 mM Tris-HCl, pH 7.4 and 2 mM EDTA) containing complete protease inhibitors (Roche Diagnostics, Indianapolis, IN). Cell lysates were then centrifuged at 1000g for 10 min at 4°C to obtain a crude nuclear fraction (N1). The N1 fraction was resuspended in 10 ml of TE buffer containing protease inhibitors, rehomogenized, and centrifuged at 1000g for 10 min at 4°C. Combined supernatants were then centrifuged at 27,000g for 20 min, and the resulting pellet was resuspended in 5 ml of TE buffer plus protease inhibitors to obtain a crude plasma membrane (P1) fraction. Supernatants were then centrifuged at 200,000g for 90 min to yield “light” membrane (V1) and high-speed supernatant (S2) fractions. All subcellular fractions were resuspended in 0.5 ml of KGEH buffer (139 mM potassium glutamate, 4 mM MgCl2, 10 mM EGTA, and 30 mM HEPES, pH 7.4) before analysis.
Western Blot Analysis.
Aliquots (30 μg of protein) of subcellular fractions were mixed with Laemmli SDS-sample buffer and resolved by SDS-polyacrylamide gel electrophoresis on a 10% Tris-HCl polyacrylamide gel (Bio-Rad, Hercules, CA). Proteins were transferred to a nitrocellulose membrane (0.45 μm Nitrobind; GE Osmonics Inc., Minnetonka, MN), and nonspecific binding sites were blocked by incubation in Tris-buffered saline (pH 7.4) containing 0.1% Tween (TBS-T) with 2.5% nonfat milk and 2.5% bovine serum albumin for 1 h at 22°C. Primary antibodies were diluted in blocking solution and incubated with membranes for 12 h at 4°C. Excess primary antibody was removed by washing the membranes 3 × 10 min in TBS-T. The blots were then incubated in the appropriate HRP-conjugated secondary antibody (diluted as specified) in blocking solution for 1 h at 22°C and subsequently washed three more times for 10 min in TBS-T. Immunoreactive proteins were detected with ECL (Western Lightening; PerkinElmer Life and Analytical Sciences), and images were captured with a EpiChemi3 darkroom (UVP, Inc., Upland, CA).
Radioligand Binding.
Subcellular fractions of SH-SY5Y cells were resuspended in KGEH buffer (139 mM potassium glutamate, 4 mM MgCl2, 10 mM EGTA, and 30 mM HEPES, pH 7.4) and incubated in a 2-ml volume with 0.5 nM [3H]QNB at 37°C for 90 min. Nonspecific binding was determined as that unaffected by inclusion of 12.5 μM atropine. Reactions were terminated by rapid filtration through Whatman (Piscataway, NJ) GF/B filters, and radioactivity was determined after the addition of 7.5 ml of UniverSol scintillation fluid. Specific [3H]QNB binding was calculated as fmol bound/mg protein.
Data Analysis.
All experiments shown were performed in duplicate or triplicate and repeated at least three times. Values quoted are given as means ± S.E.M. for the number of independent experiments indicated. Quantification of Western blot bands was performed with Scion Image software (Scion Corporation, Frederick, MD). A paired two-tailed Student's t test was used to evaluate differences between two experimental groups (level of significance, p < 0.05). Repeated measures analysis of variance (ANOVA) was routinely followed by a Bonferroni multiple comparisons test for statistical significance of differences between multiple groups. A Dunnett's multiple comparisons test was used when the only analysis of interest was the comparison of multiple groups to one control value. EC50 values were obtained with Prism 5.01 (GraphPad Software Inc., San Diego, CA). Kinetic analysis of d-aspartate uptake was fit to the sum of carrier-mediated transport and diffusional components by using a nonlinear least-squares approximation, as published previously (Foster et al., 2009), according to the following equation (eq. 1):
Kinetic values quoted are the best-fit values ± S.E. as derived by using Prism 5.01 (Graphpad Software Inc.). In this equation, V is the observed rate of d-aspartate uptake (nmol/mg protein/min), Vmax is the maximum rate of uptake, Km is the aspartate affinity binding constant (μM) for the saturable component of uptake, kdiff is the rate constant for the nonsaturable, diffusion-mediated influx (ml/mg protein/min), and S is the concentration of extracellular d-aspartate (μM). From this equation, the relative contributions of both the saturable, carrier-mediated component and the nonsaturable, diffusional component can be determined.
Results
Uptake of d-Aspartate into SH-SY5Y Neuroblastoma Cells Is Facilitated by Muscarinic Receptor Activation and Attenuated by Hypoosmolarity.
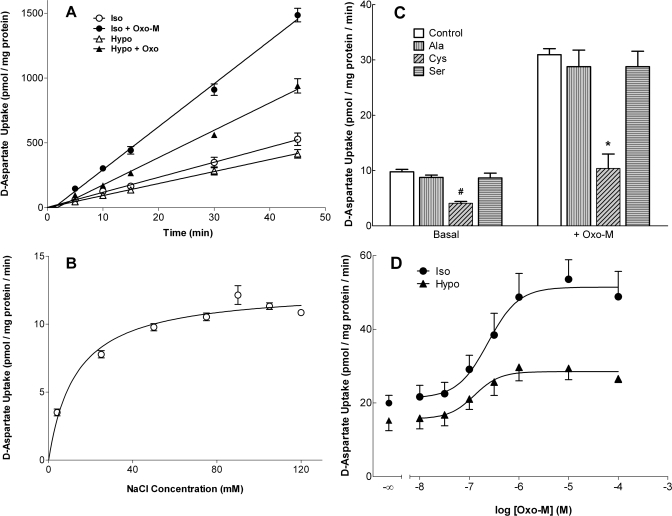
When SH-SY5Y cells were incubated at 37°C in isotonic buffer (290 mOsM) that contained 10 μM d-aspartate, the uptake of d-aspartate (used as a marker for l-glutamate) proceeded linearly for at least 45 min at a rate of 12 ± 1 pmol aspartate/mg protein/min (Fig. 1A). d-Aspartate uptake depended on the presence of NaCl (>85% inhibition in the absence of added NaCl; Fig. 1B) and was inhibited by 97 ± 2% when incubations were conducted at 4°C (n = 3). Analysis of SH-SY5Y cells indicated an endogenous glutamate concentration of 100 ± 13 nmol/mg protein (n = 5). Given that the intracellular water space in SH-SY5Y cells is 8.5 μl/mg protein (Foster et al., 2008), an intracellular concentration of glutamate of ∼12 mM can be calculated, a value similar to that observed for whole brain (McIlwain and Bachelard, 1971). Inclusion of the muscarinic cholinergic agonist Oxo-M (100 μM) significantly increased the rate of d-aspartate uptake to 275% of control (33 ± 1 pmol aspartate/mg protein/min) compared with that observed under basal isotonic conditions (p < 0.05 by ordinary ANOVA with a post hoc Bonferroni test; Fig. 1A). Inclusion of 10 μM atropine fully prevented the ability of Oxo-M to stimulate d-aspartate uptake (data not shown). Both basal- and Oxo-M-stimulated d-aspartate uptake were inhibited (∼60%) by inclusion of 100 μM l-cysteine, an amino acid that is cotransported by EAATs, in particular, EAAT3 (Zerangue and Kavanaugh, 1996; Aoyama et al., 2006). In contrast, inclusion of either l-alanine or l-serine, two neutral amino acids, was without effect (Fig. 1C). The rates of both basal- and Oxo-M-stimulated d-aspartate uptake were attenuated by a modest (21%) reduction in osmolarity of the incubation medium (Fig. 1A). Hypotonicity reduced the rate of Oxo-M-stimulated d-aspartate uptake by ∼40 to 50% but had no effect on the agonist EC50 values (0.24 and 0.13 μM under isotonic and hypotonic conditions, respectively; Fig. 1D). Hypotonicity consistently resulted in ∼20% reduction in the basal rate of d-aspartate uptake during the course of this study (14 ± 1 versus 11 ± 1 pmol/mg protein/min under isotonic and hypotonic conditions, respectively, n = 36, p < 0.001). The corresponding rates for d-aspartate uptake in the presence of Oxo-M under isotonic and hypotonic conditions were 40 ± 2 and 24 ± 1 pmol/mg protein (n = 36, p < 0.001). The possibility that reversal of EAAT contributes to the inhibition of d-aspartate uptake observed under hypoosmotic conditions can be excluded because inclusion of 200 μM TBOA, a nontransportable inhibitor of EAATs, had no effect on either the basal- or Oxo-M-stimulated d-aspartate efflux from SH-SY5Y cells (data not shown).
Fig. 1.
Characteristics of basal- and Oxo-M-stimulated d-aspartate uptake in SH-SY5Y cells under isotonic and hypotonic conditions. A, time course. Cells were incubated in 5 ml of isotonic buffer (Iso; 290 mOsM) in the absence (○) or presence (●) of 100 μM Oxo-M or hypotonic buffer (Hypo; 230 mOsM corresponding to a 21% reduction in OsM) with (▴) or without (▵) addition of Oxo-M. All incubations contained 0.3 μCi/ml [3H]aspartate and 10 μM unlabeled aspartate; reactions were terminated at the times indicated, and [3H]aspartate uptake was monitored. Results are expressed as d-aspartate uptake (pmol/mg protein) and are the means ± S.E.M. of three independent experiments, each performed in duplicate. Where error bars are absent the S.E.M. fell within the symbol. Rates of d-aspartate influx were calculated from linear regression analyses of the data. B, NaCl dependence. Cells were incubated for 45 min in the presence of NaCl at the concentrations indicated, and basal uptake of d-aspartate was monitored. Where NaCl is reduced, sucrose was substituted to maintain isotonicity. Results are expressed as d-aspartate uptake (pmol/mg protein/min) and are the means ± S.E.M. for three independent experiments. C, cells were incubated under isotonic conditions for 45 min in the absence or presence of Oxo-M (100 μM) with 100 μM concentrations of l-cysteine (l-Cys), l-serine (l-Ser), or l-alanine (l-Ala). Results are expressed as d-aspartate uptake (pmol/mg protein/min) and are means ± S.E.M. for three independent experiments. p < 0.05 different from d-aspartate uptake measured under control conditions in the absence (#) or presence (*) of Oxo-M (by repeated measures ANOVA with a post hoc Bonferroni multiple comparisons test). D, dose-response relationship for Oxo-M-mediated stimulation of d-aspartate uptake under isotonic (●) or hypotonic (▴) conditions (290 and 230 mOsM, respectively). Reactions were terminated after 10 min, and d-aspartate uptake was monitored. Results are expressed as d-aspartate uptake (pmol/mg protein/min) and are the means ± S.E.M. for three independent experiments.
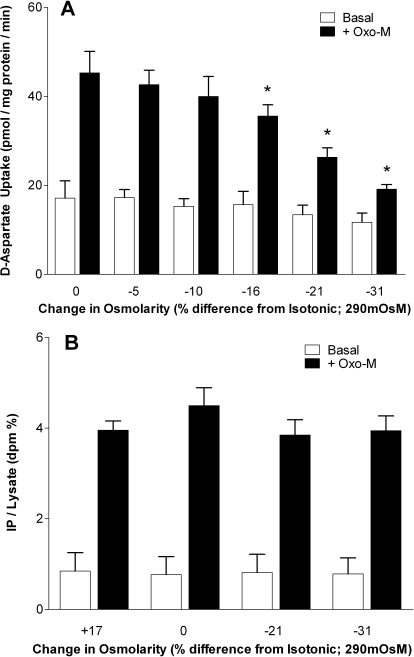
The sensitivity of d-aspartate uptake to changes in osmolarity (340–200 mOsM) was monitored under conditions of a constant NaCl concentration (Fig. 2A). Both basal- and Oxo-M-stimulated uptake were attenuated as osmolarity was reduced. Although the basal uptake was not significantly reduced from the value obtained under isotonic conditions in this limited series of experiments, a statistically significant attenuation (∼40%) of Oxo-M-stimulated uptake was observed after a reduction in osmolarity of 16%, whereas a 74% loss of d-aspartate uptake was observed when the osmolarity was reduced by 31%. In all subsequent experiments, an osmolarity of 230 mOsM (a 21% reduction from isotonic) was routinely used. To examine the possibility that these reductions in mAChR-mediated d-aspartate uptake observed under hypoosmotic conditions were secondary to an attenuation of cell signaling events, both basal- and mAChR-stimulated phosphoinositide turnover were monitored over a wide range of osmolarities. Neither hypertonic nor hypotonic conditions influenced the ability of Oxo-M to enhance phosphoinositide turnover, a result that indicates that mAChR-mediated signaling events are not directly regulated by osmolarity (Fig. 2B).
Fig. 2.
Effect of osmolarity on d-aspartate uptake and mAChR signaling. A, cells were incubated in buffers of the osmolarity indicated (isotonic buffer = 290 mOsM) in either the absence (open bars) or presence (closed bars) of 100 μM Oxo-M. The osmolarity of the buffers was adjusted under conditions of a fixed NaCl concentration (90 mM) by the addition of sucrose. Reactions were terminated after 10 min, and d-aspartate uptake was monitored. Results are expressed as d-aspartate uptake (pmol/mg protein/min) and are the means ± S.E.M. of four independent experiments, each performed in duplicate. *, p < 0.05 different from d-aspartate uptake measured under isotonic conditions (290 mOsm) in the presence of Oxo-M (by repeated measures ANOVA followed by a Bonferroni multiple comparisons test). B, cells that had been prelabeled for 48 h with [3H]inositol were treated for 10 min in buffers of the osmolarity indicated in either the absence (open bars) or presence (closed bars) of 100 μM Oxo-M. Reactions were terminated by the addition of trichloroacetic acid, and the accumulation of radiolabeled inositol phosphates (IP) was monitored as an index of stimulated phosphoinositide turnover. Results are expressed as inositol phosphate (IP) released/total soluble radioactivity in cell lysates (IP/lysate; dpm %) and are the means ± S.E.M. for three independent experiments, each performed in triplicate.
Osmosensitive Reductions in Oxo-M-Stimulated d-Aspartate Uptake Are Readily Reversible.
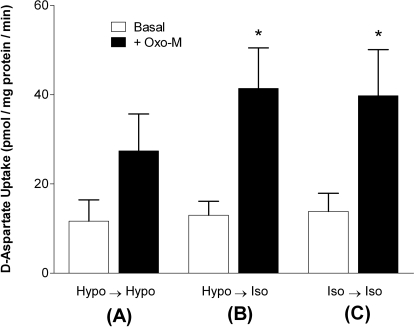
To address the possibility that exposure of the cells to hypotonic media resulted in deleterious effects, we determined whether the osmolarity-dependent reductions in agonist-stimulated d-aspartate uptake could be reversed. Cells were first preincubated for 10 min in either isotonic or hypotonic buffer in the absence or presence of 100 μM Oxo-M. Cells were then rapidly washed with isotonic buffer, and d-aspartate uptake was monitored for 20 min under either isotonic or hypotonic conditions in the presence or absence of the agonist (Fig. 3). Cells that had been preincubated in hypotonic buffer and then subsequently exposed to isotonic buffer (condition B) exhibited rates of d-aspartate uptake that were comparable with cells that had been exposed to isotonic conditions during both the preincubation and incubation phases (condition C). In contrast, cells that were maintained under hypotonic conditions during both preincubation and incubation phases (condition A) exhibited rates of d-aspartate uptake that were 40% lower than those in cells incubated under conditions B or C. These results suggest that tonicity-induced changes in d-aspartate uptake are readily reversible.
Fig. 3.
Reversibility of osmolarity-mediated inhibition of d-aspartate uptake. Cells were first preincubated for 10 min in either hypotonic buffer (Hypo; 230 mOsM, conditions A and B) or isotonic buffer (Iso; 290 mOsM, condition C) in the presence or absence of 100 μM Oxo-M. Cells were then washed twice with 2 ml of buffer (osmolarity of wash buffer matched to that of the pretreatment buffer) and incubated with 10 μM [3H]d-aspartate for an additional 20 min in 5 ml of either hypotonic (condition A) or isotonic buffers (conditions B and C) in the presence (closed bars) or absence (open bars) of Oxo-M (100 μM). Results are expressed as d-aspartate uptake (pmol/mg protein/min) and are the means ± S.E.M. of four independent experiments, each performed in triplicate. *, p < 0.05 different from d-aspartate uptake measured under condition A in the presence of Oxo-M (by repeated measures ANOVA followed by a Bonferroni multiple comparisons test).
Substrate Concentration Dependence of d-Aspartate Uptake.
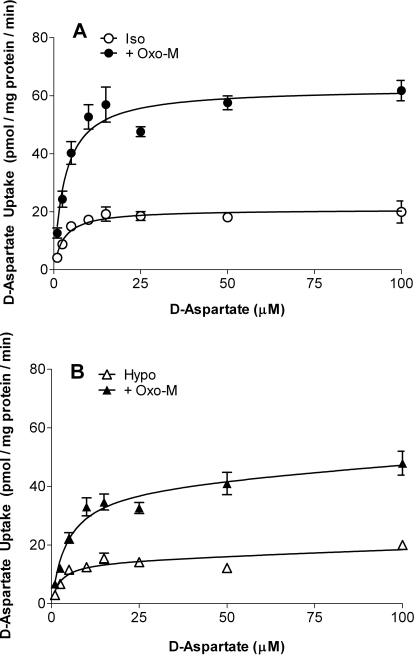
To determine whether changes in d-aspartate uptake observed under isotonic or hypotonic conditions reflected changes in the Vmax and/or Km values for d-aspartate uptake, the rate of d-aspartate uptake was monitored in the presence of increasing substrate concentrations. Rectangular hyperbolic dose-response plots were obtained for d-aspartate uptake under both isotonic and hypotonic conditions (Fig. 4). The diffusional (nonsaturable) component of d-aspartate influx was negligible under isotonic or hypotonic conditions in either the absence or presence of Oxo-M (< 3.5% of total). The Km values for d-aspartate uptake (2.4–3.2 μM) were similar under isotonic and hypotonic conditions in either the presence or absence of Oxo-M. However, under both isotonic and hypotonic conditions, the inclusion of Oxo-M significantly increased the Vmax values for d-aspartate uptake. Thus under isotonic conditions, the Vmax increased from 21 ± 1 to 63 ± 5 pmol/mg protein/min, whereas the comparable values obtained under hypotonic conditions were 15 ± 2 and 41 ± 5 pmol/mg protein/min (p < 0.05 by ordinary ANOVA). These results suggest that the increase in d-aspartate uptake observed in the presence of Oxo-M and the reduction in uptake mediated by hypoosmolarity occur primarily via changes in the Vmax for uptake.
Fig. 4.
Substrate concentration dependence of d-aspartate uptake. SH-SY5Y cells were incubated in either isotonic (Iso; 290 mOsM) (A) or hypotonic (Hypo; 230 mOsM) (B) buffer in the absence (open symbols) or presence (closed symbols) of 100 μM Oxo-M at the d-aspartate concentrations indicated. Reactions were terminated after 10 min, and d-aspartate uptake was determined. Results are the means ± S.E.M. of four independent experiments, each performed in duplicate. Where error bars are absent, the S.E.M. fell within the symbol. Kinetic parameters were obtained from eq. 1, as described under Materials and Methods.
Agonist-Mediated Increases in d-Aspartate Uptake Are Receptor-Specific in SH-SY5Y Cells.
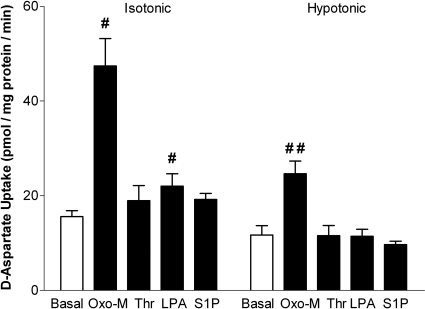
In addition to the mAChR, several GPCRs, including the protease-activated receptor, S1P receptor, and LPA receptor, have previously been demonstrated to regulate both the uptake and efflux of organic and inorganic osmolytes in SH-SY5Y cells (Foster et al., 2008, 2009). To determine whether these receptors also facilitate d-aspartate uptake, SH-SY5Y cells were exposed to isotonic or hypotonic buffer in the absence or presence of 100 μM Oxo-M, 1.25 nM thrombin (a protease-activated receptor agonist), 5 μM S1P, or 10 μM LPA (Fig. 5). The addition of thrombin or S1P had little or no effect under either isotonic or hypotonic conditions, whereas inclusion of LPA resulted in a small, but statistically significant, increase in d-aspartate uptake under isotonic, but not hypotonic, conditions (Fig. 5; n = 4, p < 0.05 by repeated measures ANOVA). In a separate series of experiments, the addition of 1 nM endothelin under isotonic conditions resulted in a statistically significant increase in the uptake of d-aspartate that was 36 ± 3% of that observed in the presence of Oxo-M (n = 4, p < 0.05).
Fig. 5.
Agonist-mediated increases in d-aspartate uptake are receptor-specific. Cells were incubated under either isotonic (290 mOsM) or hypotonic (230 mOsM) conditions in the absence or presence of 100 μM Oxo-M, 1.25 nM thrombin (Thr), 5 μM S1P, or 10 μM LPA. Reactions were terminated after 10 min, and d-aspartate uptake was monitored. Results are the means ± S.E.M. of four independent experiments, each performed in duplicate. p < 0.05 different from d-aspartate uptake observed under isotonic (#) or hypotonic (##) basal conditions (by repeated measures ANOVA followed by a Bonferroni multiple comparisons test).
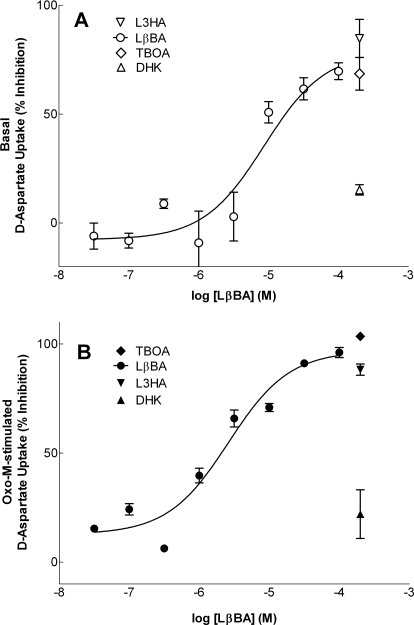
Pharmacological Characterization of Basal- and Oxo-M-Stimulated d-Aspartate Uptake.
Three distinct subtypes of EAATs (EAAT1–EAAT3) have been reported to be present in SH-SY5Y cells (Sala et al., 2005). To examine the subtypes of transporters responsible for d-aspartate uptake under basal- or agonist-stimulated conditions, the following pharmacological inhibitors were used: TBOA, a nontransportable inhibitor of EAAT1-5 (200 μM); L3HA, a transportable inhibitor of EAAT1-4 (200 μM); DHK, a nontransportable inhibitor of EAAT2 (200 μM); or LβBA, a nontransportable inhibitor of EAAT3 (and to a lesser extent, EAAT1; 0.3–100 μM). Addition of DHK had little effect on either basal (Fig. 6A) or Oxo-M-stimulated (Fig. 6B) d-aspartate uptake, indicating that EAAT2 is unlikely to play a significant role in either of these processes. Inclusion of either TBOA or L3HA essentially completely abolished Oxo-M-stimulated d-aspartate uptake (∼95%) and severely attenuated basal uptake (∼75%), indicating that the uptake is mediated via EAAT1 and/or EAAT3. To potentially differentiate between the involvement of EAAT1 and EAAT3, a dose-inhibition curve was constructed for the EAAT3-selective inhibitor LβBA. This agent exhibits a 6-fold selectivity for EAAT3 over EAAT1 (Ki values of 2 and 12 μM, respectively; Esslinger et al., 2005). d-Aspartate uptake under both basal- and Oxo-M-stimulated conditions was potently inhibited by LβBA (IC50 values = 8.6 and 2.6 μM, respectively) with corresponding Ki values calculated from the Cheng–Prusoff equation (Cheng and Prusoff, 1973) of 1.8 and 0.6 μM. Taken collectively, these results suggest that EAAT3 plays a major role in the basal-and Oxo-M-stimulated uptake of d-aspartate into SH-SY5Y cells.
Fig. 6.
Effect of EAAT inhibitors on basal- and Oxo-M-stimulated d-aspartate uptake. Cells were incubated in isotonic buffer (290 mOsM) in the absence (A) or presence (B) of 100 μM Oxo-M with or without the EAAT inhibitors TBOA, LβBA, L3HA, or DHK at the concentrations indicated. Reactions were terminated after 10 min, and results are presented as percentage inhibition of basal d-aspartate uptake (A) or percentage inhibition of Oxo-M-stimulated d-aspartate uptake (B) and are the means ± S.E.M. of three to six independent experiments, each performed in duplicate. LβBA inhibited basal d-aspartate uptake with an IC50 of 8.6 μM and attenuated Oxo-M-stimulated d-aspartate uptake with an IC50 of 2.6 μM.
Endothelin-1-Mediated Stimulation of d-Aspartate Uptake in C6 Glioma Cells Is Also Osmosensitive.
Previous reports have demonstrated that activation of endothelin-1 receptors in C6 glioma cells increases high-affinity d-aspartate uptake, a process mediated by the EAAT3 transporter (Najimi et al., 2005). To determine whether d-aspartate uptake in C6 glioma cells is also sensitive to changes in osmolarity, uptake of d-aspartate was measured under either isotonic or hypotonic conditions in the presence or absence of 1 nM endothelin. The rate of d-aspartate uptake under basal isotonic conditions (298 ± 7 pmol/mg protein/min, n = 3) was ∼20 fold greater than that observed in SH-SY5Y cells. Addition of ET-1 under isotonic conditions caused a significant (65%) stimulation of d-aspartate uptake (492 ± 33 pmol/mg protein/min; n = 3, p < 0.05 by repeated measures ANOVA), as reported previously. Under hypotonic conditions a small, yet significant, decrease in basal uptake was observed (∼14% reduction compared with basal isotonic conditions). Although the addition of ET-1 under hypotonic conditions increased d-aspartate uptake, the degree of this stimulation was significantly inhibited (∼45%) compared with ET-1-mediated uptake under isotonic conditons (n = 3, p < 0.05 by repeated measures ANOVA).
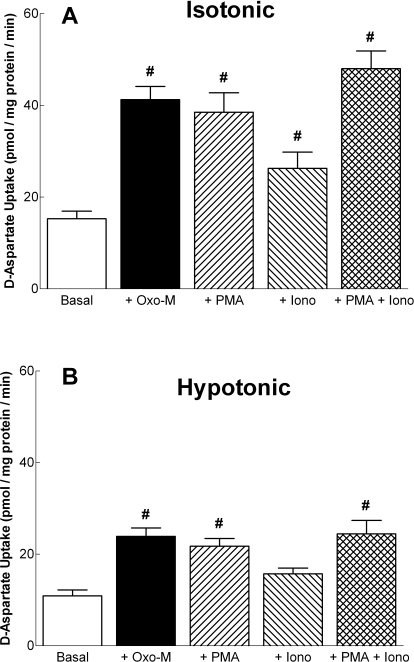
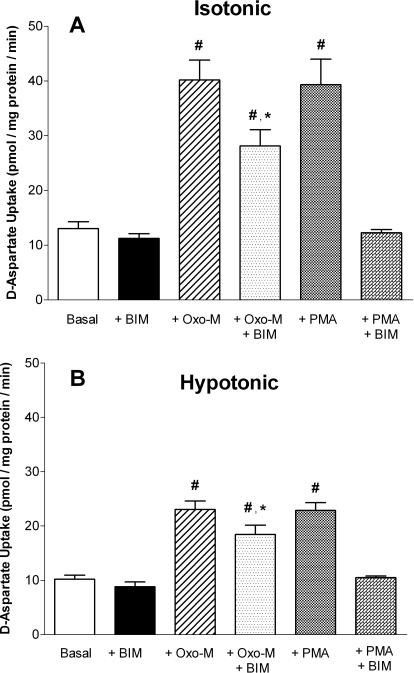
Oxo-M-Mediated Stimulation of d-Aspartate Uptake in SH-SY5Y Cells Occurs via PKC- and Ca2+-Dependent Mechanisms.
Agonist occupancy of mAChRs in SH-SY5Y cells is known to lead to the activation of PKC and mobilization of intracellular Ca2+ (Cioffi and Fisher, 1990). To evaluate the role for PKC and Ca2+ in Oxo-M-stimulated d-aspartate uptake, SH-SY5Y cells were incubated under isotonic or hypotonic conditions in the absence or presence of 100 μM Oxo-M or optimal concentrations of either PMA (100 nM) or ionomycin (1 μM). Addition of PMA significantly increased d-aspartate uptake under both isotonic and hypotonic conditions (p < 0.05 different from control basal by repeated measures ANOVA). Addition of ionomycin also resulted in increased d-aspartate uptake under isotonic conditions, but not under hypoosmotic conditions (Fig. 7B). In the presence of both PMA and ionomycin, the rate of d-aspartate uptake under isotonic or hypotonic conditions was comparable with that observed in the presence of Oxo-M (Fig. 7). To further examine a role for PKC in the mediation of Oxo-M-stimulated d-aspartate uptake, cells were preincubated for 10 min with either vehicle (DMSO) or 2.5 μM BIM (a broad spectrum PKC inhibitor) before the addition of either Oxo-M or PMA. Although BIM had no effect on basal d-aspartate uptake, it fully blocked the ability of PMA to stimulate uptake but only partially attenuated (30–45%) the ability of Oxo-M to enhance d-aspartate uptake, under either isotonic or hypotonic conditions (Fig. 8; p < 0.05 different from control + Oxo-M by repeated measures ANOVA). In contrast, the Ca2+ dependence of mAChR-stimulated d-aspartate uptake differed under isotonic and hypotonic conditions. Thus, under isotonic conditions, removal of extracellular Ca2+ resulted in a 60% inhibition of mAChR-mediated d-aspartate uptake (Fig. 9A), whereas little or no effect was observed under hypotonic conditions (Fig. 9B). Depletion of intracellular stores of Ca2+ with thapsigargin resulted in a small inhibitory effect on mAChR-mediated d-aspartate uptake under both isotonic and hypoosmotic conditions (Fig. 9, A and B). The combination of inhibition of PKC with BIM, along with depletion of intracellular and extracellular Ca2+, essentially abolished the ability of mAChR activation to stimulate d-aspartate uptake under both isotonic and hypoosmotic conditions (Fig. 9, C and D).
Fig. 7.
Agents that either activate PKC or increase intracellular Ca2+ concentrations mimic the ability of Oxo-M to induce d-aspartate uptake. d-Aspartate uptake was monitored in SH-SY5Y cells after 10 min of incubation under isotonic (290 mOsM) (A) or hypotonic (230 mOsM) (B) conditions in the absence or presence of 100 μM Oxo-M, 100 nM PMA, or 1 μM ionomycin (Iono). Results shown are the means ± S.E.M. of three to four independent experiments, each performed in duplicate. #, p < 0.05 different from d-aspartate uptake observed under basal conditions (by repeated measures ANOVA followed by Dunnett's multiple comparisons test).
Fig. 8.
Inhibition of PKC attenuates both Oxo-M- and PMA-stimulated d-aspartate uptake. Cells were incubated under either isotonic (290 mOsM) (A) or hypotonic (230 mOsM) (B) conditions in the absence or presence of 100 μM Oxo-M, 100 nM PMA, or 2.5 μM BIM, as indicated. All cells treated with BIM received a 10-min pretreatment in isotonic (290 mOsM) buffer containing 2.5 μM BIM. Reactions were terminated after 10 min, and results are expressed as d-aspartate uptake (pmol/min/mg protein) and are the means ± S.E.M. of three independent experiments, each performed in duplicate. p < 0.05 different from d-aspartate uptake observed under basal (#) or Oxo-M-treated (*) conditions (by repeated measures ANOVA followed by a Bonferroni multiple comparisons test).
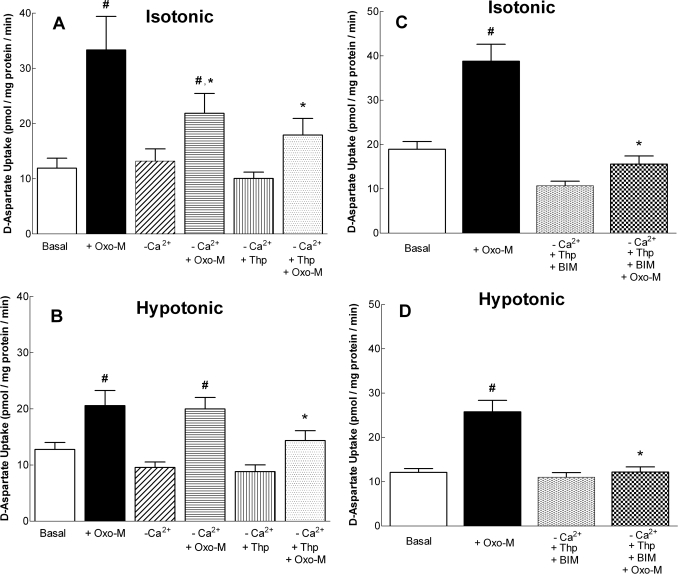
Fig. 9.
The role of extracellular and intracellular Ca2+ in Oxo-M-stimulated d-aspartate uptake. A and B, cells were incubated under either isotonic (290 mOsM) (A) or hypotonic (230 mOsM) (B) conditions in the absence (−Ca2+; Ca2+ was omitted from buffer and 50 μM EGTA was added) or the presence of extracellular Ca2+ with or without the addition of 100 μM Oxo-M. Some cells were preincubated for 15 min in isotonic buffer containing 1 μM thapsigargin (Thp) to deplete intracellular pools of Ca2+ before measurement of d-aspartate uptake in either the absence or presence of Oxo-M. Reactions were terminated after 10 min, and d-aspartate uptake was determined. C and D, cells were preincubated for 15 min with 2.5 μM BIM and 1 μM Thp (−Ca2+) and then incubated under isotonic or hypotonic conditions in the absence or presence of Oxo-M. Results shown are the means ± S.E.M. of four independent experiments, each performed in duplicate. p < 0.05 different from d-aspartate uptake observed either under basal conditions (#) or in the presence of Oxo-M alone (*) (by repeated measures ANOVA followed by a Bonferroni multiple comparisons test).
mAChR-Stimulated d-Aspartate Uptake in SH-SY5Y Cells Depends on PI3K Activity and an Intact Cytoskeleton.
There have been conflicting reports about the involvement of PI3K and cytoskeleton in the regulation of d-aspartate uptake (Najimi et al., 2002; Bianchi et al., 2006). Thus, the role, if any, played by these components in mAChR-mediated d-aspartate was examined. Inclusion of 100 nM wortmannin reduced the basal uptake of d-aspartate under both isotonic and hypotonic conditions and significantly attenuated (35–40%) the ability of Oxo-M to facilitate uptake (p < 0.05 by paired Student's t test; Fig. 10). Disruption of the actin cytoskeleton by pretreatment of the cells with 1 μM cytochalasin D (Cyto D), or alternatively, disruption of microtubule polymerization with 10 μM colchicine, had no effect on basal d-aspartate uptake but significantly attenuated Oxo-M-stimulated d-aspartate uptake (approximately 35–55%) under isotonic conditions when added individually or in combination (Fig. 11A; n = 4, p < 0.05 different versus control + Oxo-M by repeated measures ANOVA). Similar results were obtained under hypotonic conditions with the exception that basal d-aspartate uptake was attenuated in the combined presence of cytochalasin and colchicine. A combination of hypotonicity and disruption of the actin cytoskeleton and microtubules led to a >75% inhibition of Oxo-M-stimulated d-aspartate uptake in comparison with that observed under isotonic conditions alone.
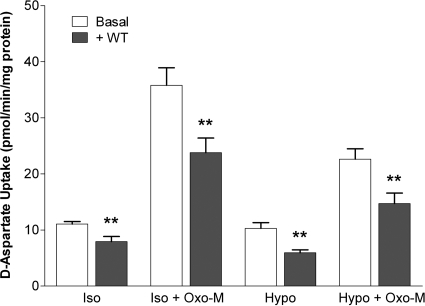
Fig. 10.
Inhibition of PI3K with wortmannin inhibits both basal- and Oxo-M-stimulated d-aspartate uptake. Cells were incubated in either isotonic (Iso; 290 mOsM) or hypotonic (Hypo; 230 mOsM) buffer in the absence or presence of 100 μM Oxo-M and 100 nM wortmannin (WT). d-Aspartate uptake was monitored after 10 min. Results shown are the means ± S.E.M. of four independent experiments, each performed in duplicate. **, p < 0.05 different from d-aspartate uptake observed in the absence of WT (by paired Student's t test).
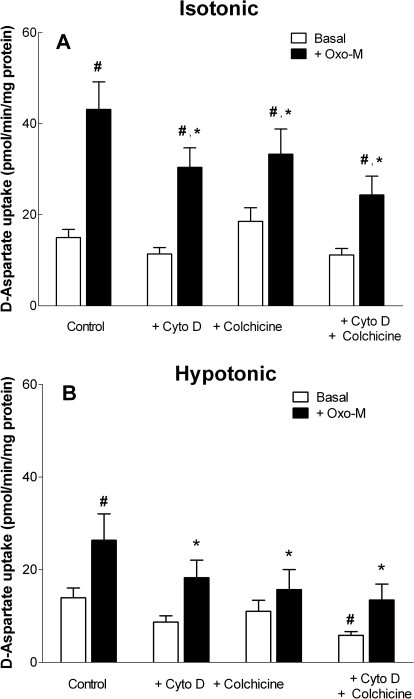
Fig. 11.
Disruption of the cytoskeleton attenuates Oxo-M-stimulated d-aspartate uptake. Cells were incubated under either isotonic (290 mOsM) (A) or hypotonic (230 mOsM) (B) conditions in the presence or absence of 100 μM Oxo-M, 1 μM Cyto D, or 10 μM colchicine for 10 min, and d-aspartate uptake was determined. All cells treated with Cyto D and/or colchicine were exposed to the drugs for 30 min in isotonic buffer before measurement of d-aspartate uptake. Results shown are the means ± S.E.M. of three independent experiments, each performed in duplicate. p < 0.05 different from d-aspartate uptake observed under control conditions in the absence (#) or presence (*) of Oxo-M (by repeated measures ANOVA followed by a Bonferroni multiple comparisons test).
Oxo-M-Mediated Subcellular Redistribution of EAAT3.
In quiescent cells, EAAT3 is localized predominantly to an intracellular location (Beart and O'Shea, 2007). With a cell-surface biotinylation approach, activation of neurotensin, platelet-derived growth factor, and endothelin receptors has previously been demonstrated to result in the appearance of EAAT3 at the plasma membrane (Najimi et al., 2002, 2005; Fournier et al., 2004). Given these previous findings and the pharmacological data that indicated a primary role for EAAT3 in Oxo-M-stimulated d-aspartate uptake (Fig. 6), the effect of Oxo-M and hypotonicity on the subcellular localization of EAAT3 was examined by using a previously characterized fractionation protocol for this cell line (Sorensen et al., 1997). (Efforts to use a biotinylation technique were unsuccessful because of the relatively low expression of EAAT3 in the SH-SY5Y cell line.) Cells were treated under isotonic or hypotonic conditions ± Oxo-M and then subjected to subcellular fractionation to obtain a crude plasma membrane fraction (P1: that is enriched in [3H]ouabain binding, a marker for Na+ + K+-ATPase activity) and an endosomal fraction (V1: that is enriched in NADPH cytochrome c reductase activity and endocytosed mAChRs). There were no significant differences in the immunoreactivities of actin or EEA1 recovered in the P1 and V1 fractions, respectively, across the experimental conditions examined. Thus, these values were routinely used as loading controls. Under basal isotonic conditions, EAAT3 immunoreactivity (detected as a major band at 66 kDa on Western blots) was approximately four times greater in the V1 fraction than in the P1 fraction. However, the addition of Oxo-M increased the amount of EAAT3 immunoreactivity associated with the P1 fraction by ∼2-fold (different from control cells treated, p < 0.05 by repeated measures ANOVA). A concurrent reduction in the amount of EAAT3 detected in the V1 fraction (∼25%) was observed upon addition of Oxo-M under isotonic conditions, although this value did not reach statistical significance. Unexpectedly, a similar enrichment of EAAT3 immunoreactivity in the P1 fraction (relative to basal isotonic conditions) occurred under basal hypotonic conditions and no further increase was observed in the presence of Oxo-M (Fig. 12B). No immunoreactivity associated with either EAAT1 or EAAT2 could be reliably detected in these subcellular fractions or cell lysates. To determine whether exposure of SH-SY5Y cells to a hypoosmotic medium resulted in a global disruption of trafficking events, the ability of Oxo-M to induce the redistribution of mAChRs from the plasma membrane to endosomal fractions was monitored. Exposure of the cells to Oxo-M for 10 min resulted in an increase in [3H]QNB binding (∼200% of control) in the V1 fractions that was similar in magnitude under both isotonic and hypotonic conditions (Fig. 12C).
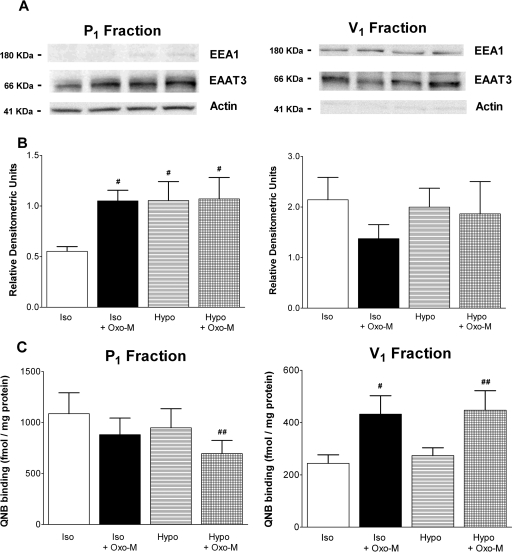
Fig. 12.
Western blot analysis of EAAT3 in subcellular fractions derived from SH-SY5Y cells incubated in the absence or presence of Oxo-M. Cells were treated for 10 min in the presence of either isotonic (Iso; 290 mOsM) or hypotonic (Hypo; 230 mOsM) buffer in the absence or presence of 100 μM Oxo-M. Cells were then lysed and fractionated as described under Materials and Methods. A, equivalent aliquots (30 μg of protein) of subcellular fractions P1 (crude plasma membrane fraction) and V1 (“light” membrane fraction) were electrophoresed and transferred to nitrocellulose membranes. Membranes were then blotted for EAAT3, actin, and early endosomal antigen 1 (EAA1) as detailed under Materials and Methods. Blots shown are representative of those obtained in five independent experiments. B, densitometric analysis of Western blots. Results shown are the density of each fraction divided by the appropriate loading control (actin for P1; EAA1 for V1; means ± S.E.M. of five independent experiments). #, p < 0.05 different from density observed under basal isotonic conditions (by repeated measures ANOVA followed by a Bonferroni multiple comparisons test). C, [3H]QNB binding in P1 and V1 fractions derived from SH-SY5Y cells treated under isotonic or hypotonic conditions in the absence or presence of Oxo-M. Results shown are the means ± S.E.M. of five independent experiments and are expressed as fmol bound/mg of protein. p < 0.05 different from receptor density observed in the absence of Oxo-M under isotonic (#) or hypotonic (##) conditions (by repeated measures ANOVA followed by a Bonferoni multiple comparisons test).
Discussion
In addition to its role as a major neurotransmitter, glutamate functions as a quantitatively important organic osmolyte within the CNS. Thus, mechanisms that regulate its concentration within the extracellular space are of physiological importance under both isotonic and hypoosmotic conditions. In the present study we have demonstrated that, under isotonic conditions, agonist occupancy of mAChRs leads to a rapid and sustained increase in glutamate uptake (monitored as d-aspartate) into SH-SY5Y neuroblastoma cells. Activation of LPA and endothelin receptors also increased d-aspartate uptake, but to a lesser extent. In C6 glioma cells, activation of several phosphoinositide-linked receptors has previously been demonstrated to facilitate d-aspartate uptake (Najimi et al., 2002, 2005; Fournier et al., 2004). In this context, it may be relevant that of the GPCRs tested in SH-SY5Y neuroblastoma only the mAChR couples to phosphoinositide hydrolysis in this cell line (Heacock et al., 2006).
d-Aspartate uptake into SH-SY5Y cells was mediated via a high-affinity, NaCl-dependent, saturable transport system that could be blocked by LβBA, a potent inhibitor of the neuron-specific glutamate transporter EAAT3. Although three subtypes of the glutamate transporter (EAAT1–EAAT3) have been reported to be present in SH-SY5Y cells (Sala et al., 2005), only immunoreactivity to the EAAT3 subtype was detectable in cell lysates in the present study. This observation, along with the potent inhibition of d-aspartate uptake by LβBA and the ability of cysteine to readily compete with d-aspartate for transport (a characteristic of the EAAT3 subtype; Zerangue and Kavanaugh, 1996), strongly suggests that the primary mediator of d-aspartate uptake in SH-SY5Y cells is EAAT3. In quiescent SH-SY5Y cells, the EAAT3 transporter is concentrated in an intracellular compartment, as reported previously for other cell types (Fournier et al., 2004; Beart and O'Shea, 2007). However, upon mAChR activation in SH-SY5Y cells, a redistribution of the transporter to the plasma membrane occurs (Fig. 12, A and B). It seems likely that a major component of Oxo-M-mediated increase in d-aspartate uptake in SH-SY5Y cells reflects this appearance of EAAT3 at the plasma membrane, although the possibility that receptor activation increases the intrinsic activity of the transporter cannot be excluded.
In SH-SY5Y cells, the activation of mAChRs elicits a number of biochemical consequences, including the activation of kinases, mobilization of intracellular and extracellular Ca2+, and the rearrangement of the cytoskeleton (Cioffi and Fisher, 1990; Linseman et al., 1999, 2000). The present results suggest that the activation of multiple signaling pathways is required for mAChR-mediated regulation of d-aspartate transport. For example, a role for PKC is indicated from the ability of BIM, a PKC inhibitor, to partially block Oxo-M-stimulated d-aspartate uptake (Fig. 8). Our results differ from those obtained for C6 glioma cells in which little or no effect of PKC inhibitors on receptor-simulated EAAT3 activity was observed (Sims et al., 2000; Najimi et al., 2002, 2005). Based on the ability of wortmannin to inhibit both basal- and mAChR-mediated d-aspartate transport, PI3K also appears to play a role in the regulation of EAAT3 activity in SH-SY5Y cells (Fig. 10), an observation consistent with some, but not all, reports of receptor-stimulated EAAT3 trafficking and activity (Sims et al., 2000; Najimi et al., 2002, 2005; Krizman-Genda et al., 2005). One aspect of receptor-mediated EAAT3 regulation that has previously received little attention is that of its dependence on Ca2+ availability. The present data strongly suggest that the mAChR-mediated influx of extracellular Ca2+ is a major determinant of the magnitude of mAChR-mediated increases in d-aspartate uptake. Influx of extracellular Ca2+ may serve to activate downstream effectors such as Ca2+/calmodulin-dependent protein kinase and/or the Ca2+-sensitive forms of PKC. The importance of both PKC and Ca2+ in regulation of EAAT3 activity in SH-SY5Y cells is evident from the observation that Ca2+ depletion, in conjunction with PKC inhibition, essentially abolishes mAChR-stimulated d-aspartate uptake (Fig. 9). These findings are consistent with results recently obtained for C6 glioma cells in which a sustained entry of Ca2+, elicited independently of receptor activation, resulted in a PKC-dependent increase in both the cell-surface expression and activity of EAAT3 (Murphy et al., 2009). Optimal d-aspartate transport in SH-SY5Y cells under both basal- and Oxo-M-stimulated conditions also requires the maintenance of cytoskeletal integrity (Fig. 11). Thus, it is possible that these signaling pathways (Ca2+, PKC, and PI3K) may act in concert to regulate cytoskeletal-dependent trafficking of EAAT3. Consistent with this possibility, a role for PKC, PI3K, and the actin cytoskeleton has been proposed for dopamine receptor-mediated translocation of Na+-K+-ATPase from endosomes to the plasma membrane (Bertorello and Sznajder, 2005).
Previous studies have indicated that during episodes of hypoosmotic stress organic osmolyte homeostasis is regulated not only via osmolyte efflux but also at the level of osmolyte reuptake (Olson and Martinho, 2006; Foster et al., 2009). Consistent with this possibility, a major finding from the present study is that mAChR-mediated uptake of d-aspartate (and to a lesser extent, basal uptake) is significantly attenuated when SH-SY5Y cells are subjected to hypoosmotic stress. The observation that ET-1-mediated increases in d-apsartate uptake are similarly attenuated by hypoosmolarity in C6 glioma cells indicates that osmotic regulation of glutamate transport may be a general, rather than cell-specific, phenomenon. The reductions in d-aspartate uptake in SH-SY5Y cells could be fully reversed upon re-exposure of the cells to an isotonic medium and were elicited by relatively modest reductions in osmolarity (≥16%). Inhibition of d-aspartate uptake under hypotonic conditions reflected a reduction in Vmax for transport, whereas the Km value was unaltered. The uptake of d-aspartate exhibited similar characteristics in terms of agonist concentration dependence, requirements for PKC and PI3K activity, and the need for an intact cytoskeleton under both isotonic and hypotonic conditions. However, two distinct osmosensitive features of d-aspartate uptake are apparent. The first is that under hypoosmotic conditions d-aspartate uptake is largely independent of extracellular Ca2+. Thus the addition of the Ca2+ ionophore, ionomycin, did not significantly increase uptake under hypotonic conditions, whereas a robust increase in d-aspartate uptake was apparent under isotonic conditions (Fig. 7). Similarly, removal of extracellular Ca2+ had little or no effect on mAChR-stimulated d-aspartate uptake under hypoosmotic conditions, whereas a marked inhibition was observed under isotonic conditions (Fig. 9, A and B). The mechanism whereby the Ca2+-dependent component of mAChR-stimulated d-aspartate uptake is lost under conditions of hypoosmolarity remains to be determined. However, this inability of Ca2+ to regulate d-aspartate uptake reflects the osmotic disruption of an event distal to mAChR activation because hypoosmolarity did not attenuate stimulated phosphoinositide hydrolysis, an event proximal to mAChR activation and dependent on a sustained influx of extracellular Ca2+ (Fig. 2B; Fisher et al., 1989). The second feature of EAAT3 regulation that differs under hypoosmotic conditions is that of its trafficking. Under hypoosmotic conditions, EAAT3 transporters accumulate at the plasma membrane, with little or no regulation of trafficking after mAChR activation (Fig. 12, A and B). These observations were unexpected given 1) that the rate of basal d-aspartate uptake is attenuated by a reduction in osmolarity and 2) that, although reduced in magnitude, activation of mAChRs still elicits an increase in uptake under hypoosmotic conditions. One possible explanation for the divergence between transporter trafficking and activity under hypotonic conditions is that EAAT3 that accumulates in the plasma membrane is either not correctly inserted and/or has a diminished intrinsic activity. Alternatively, hypotonicity may induce a translocation of the transporter into a vesicular fraction that associates with, but is not integral to, the plasma membrane. However, altered EAAT3 trafficking does not appear to reflect a global disruption of membrane trafficking events in SH-SY5Y cells because Oxo-M-mediated internalization of mAChRs was unaffected by changes in osmolarity (Fig. 12C).
The observed osmosensitive regulation of EAAT3-mediated glutamate uptake in vitro could potentially have physiological consequences in the intact brain. In this context, evidence for cholinergic regulation of glutamate concentrations in brain has been reported (Higley et al., 2009). mAChR activation may allow EAAT3-containing neurons to maintain or reduce local extracellular glutamate concentrations during small reductions in osmolarity, whereas depletion of intracellular glutamate would occur during more pronounced reductions in osmolarity. In addition, EAAT3 has been demonstrated to mediate cysteine uptake (Aoyama et al., 2006), the rate-limiting step in glutathione synthesis. Glutathione is depleted in hyponatremic brains (Clark et al., 1996), an observation that could potentially be explained by osmosensitive inhibition of EAAT3 activity. However, the physiological relevance of tonicity-mediated changes in basal or mAChR-mediated glutamate uptake remains to be directly evaluated in vivo.
In summary, the present results indicate that the activation of mAChRs results in a pronounced stimulation of EAAT3 transporter-mediated d-aspartate uptake into SH-SY5Y cells, under conditions of either isotonicity or mild hypoosmolarity. In contrast, mAChR activation results in an inhibition of the reuptake of the more benign osmolyte, taurine (Foster et al., 2009). As osmolarity is further decreased, the ability of mAChR activation to facilitate the uptake of d-aspartate, although still evident, is progressively attenuated, thereby providing additional evidence that, in response to hypoosmotic stress, cells preferentially release organic osmolytes while retaining their complement of inorganic osmolytes (Foster et al., 2008).
Acknowledgments
We thank Drs. Margaret Gnegy and Jeffrey Martens for helpful comments and technical assistance.
This work was supported by the National Institutes of Health [Grant GM007767] (to D.J.F.).
Article, publication date, and citation information can be found at http://jpet.aspetjournals.org.
doi:10.1124/jpet.109.164277.
- DMEM
- Dulbecco's modified Eagle's medium
- GPCR
- G protein-coupled receptor
- mAChR
- muscarinic cholinergic receptor
- S1P
- sphingosine 1-phosphate
- PKC
- protein kinase C
- Oxo-M
- oxotremorine-M
- LPA
- lysophosphatidic acid
- ANOVA
- analysis of variance
- CNS
- central nervous system
- PI3K
- phosphatidylinositol 3-kinase
- EAAT
- excitatory amino acid transporter
- QNB
- quinuclidinyl benzilate
- LβBA
- l-β-threo-benzyl-aspartate
- DHK
- dihydrokainic acid
- TBOA
- dl-threo-β-benzyloxyaspartic acid
- L3HA
- l-(−)-threo-3-hydroxyaspartic acid
- EEA1
- early endosomal antigen 1
- BIM
- bisindolylmaleimide
- PMA
- phorbol 12-myristate 13-acetate
- HRP
- horseradish peroxidase
- TE
- Tris-EDTA
- TBS-T
- Tris-buffered saline (pH 7.4) containing 0.1% Tween
- Cyto D
- cytochalasin D.
References
- Aoyama K, Suh SW, Hamby AM, Liu J, Chan WY, Chen Y, Swanson RA. (2006) Neuronal glutathione deficiency and age-dependent neurodegeneration in the EAAC1-deficient mouse. Nat Neurosci 9:119–126 [DOI] [PubMed] [Google Scholar]
- Beart PM, O'Shea RD. (2007) Transporters for l-glutamate: an update on their molecular pharmacology and pathological involvement. Br J Pharmacol 150:5–17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertorello AM, Sznajder JI. (2005) The dopamine paradox in lung and kidney epithelia: sharing the same target but operating different signaling networks. Am J Respir Cell Mol Biol 33:432–437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhardwaj A. (2006) Neurological impact of vasopressin dysregulation and hyponatremia. Ann Neurol 59:229–236 [DOI] [PubMed] [Google Scholar]
- Bianchi MG, Rotoli BM, Dall'Asta V, Gazzola GC, Gatti R, Bussolati O. (2006) PKC-dependent stimulation of EAAT3 glutamate transporter does not require the integrity of actin cytoskeleton. Neurochem Int 48:341–349 [DOI] [PubMed] [Google Scholar]
- Cheng Y, Prusoff WH. (1973) Relationship between the inhibition constant (KI) and the concentration of inhibitor which causes 50 percent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol 22:3099–3108 [DOI] [PubMed] [Google Scholar]
- Cioffi CL, Fisher SK. (1990) Reduction of muscarinic receptor density and of guanine nucleotide-stimulated phosphoinositide hydrolysis in human SH-SY5Y neuroblastoma cells following long-term treatment with 12-O-tetradecanoylphorbol 13-acetate or mezerein. J Neurochem 54:1725–1734 [DOI] [PubMed] [Google Scholar]
- Clark EC, Thomas D, Baer J, Sterns RH. (1996) Depletion of glutathione from brain cells in hyponatremia. Kidney Int 49:470–476 [DOI] [PubMed] [Google Scholar]
- Esslinger CS, Agarwal S, Gerdes J, Wilson PA, Davis ES, Awes AN, O'Brien E, Mavencamp T, Koch HP, Poulsen DJ, et al. (2005) The substituted aspartate analogue l-β-threo-benzyl-aspartate preferentially inhibits the neuronal excitatory amino acid transporter EAAT3. Neuropharmacology 49:850–861 [DOI] [PubMed] [Google Scholar]
- Fisher SK, Cheema TA, Foster DJ, Heacock AM. (2008) Volume-dependent osmolyte efflux from neural tissues: regulation by G protein-coupled receptors. J Neurochem 106:1998–2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher SK, Domask LM, Roland RM. (1989) Muscarinic receptor regulation of cytoplasmic Ca2+ concentrations in human SK-N-SH neuroblastoma cells: Ca2+ requirements for phospholipase C activation. Mol Pharmacol 35:195–204 [PubMed] [Google Scholar]
- Foster DJ, Heacock AM, Keep RF, Fisher SK. (2008) Activation of muscarinic cholinergic receptors on human SH-SY5Y neuroblastoma cells enhances both the influx and efflux of K+ under conditions of hypoosmolarity. J Pharmacol Exp Ther 325:457–465 [DOI] [PubMed] [Google Scholar]
- Foster DJ, Vitvitsky VM, Banerjee R, Heacock AM, Fisher SK. (2009) Muscarinic receptor regulation of osmosensitive taurine transport in human SH-SY5Y neuroblastoma cells. J Neurochem 108:437–449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fournier KM, Gonzalez MI, Robinson MB. (2004) Rapid trafficking of the neuronal glutamate transporter, EAAC1: evidence for distinct trafficking pathways differentially regulated by protein kinase C and platelet-derived growth factor. J Biol Chem 279:34505–34513 [DOI] [PubMed] [Google Scholar]
- Franco R, Panayiotidis MI, de la Paz LD. (2008) Autocrine signaling involved in cell volume regulation: the role of released transmitters and plasma membrane receptors. J Cell Physiol 216:14–28 [DOI] [PubMed] [Google Scholar]
- Heacock AM, Dodd MS, Fisher SK. (2006) Regulation of volume-sensitive osmolyte efflux from human SH-SY5Y neuroblastoma cells following activation of lysophospholipid receptors. J Pharmacol Exp Ther 317:685–693 [DOI] [PubMed] [Google Scholar]
- Heacock AM, Kerley D, Gurda GT, VanTroostenberghe AT, Fisher SK. (2004) Potentiation of the osmosensitive release of taurine and d-aspartate from SH-SY5Y neuroblastoma cells after activation of M3 muscarinic cholinergic receptors. J Pharmacol Exp Ther 311:1097–1104 [DOI] [PubMed] [Google Scholar]
- Higley MJ, Soler-Llavina GJ, Sabatini BL. (2009) Cholinergic modulation of multivesicular release regulates striatal synaptic potency and integration. Nat Neurosci 12:1121–1128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krizman-Genda E, Gonzalez MI, Zelenaia O, Robinson MB. (2005) Evidence that Akt mediates platelet-derived growth factor-dependent increases in activity and surface expression of the neuronal glutamate transporter, EAAC1. Neuropharmacology 49:872–882 [DOI] [PubMed] [Google Scholar]
- Lien YH, Shapiro JI. (2007) Hyponatremia: clinical diagnosis and management. Am J Med 120:653–658 [DOI] [PubMed] [Google Scholar]
- Linseman DA, Hofmann F, Fisher SK. (2000) A role for the small molecular weight GTPases, Rho and Cdc42, in muscarinic receptor signaling to focal adhesion kinase. J Neurochem 74:2010–2020 [DOI] [PubMed] [Google Scholar]
- Linseman DA, Sorensen SD, Fisher SK. (1999) Attenuation of focal adhesion kinase signaling following depletion of agonist-sensitive pools of phosphatidylinositol 4,5-bisphosphate. J Neurochem 73:1933–1944 [PubMed] [Google Scholar]
- Lund P, Bergmeyer HU. (1986) l-Glutamine and l-glutamate: UV method with glutaminase and glutamate dehydrogenase, in Methods of Enzymatic Analysis (Bergmeyer HU. ed) pp 357–363, VCH, Verlagsgesellschaft, Weinheim, Germany: [Google Scholar]
- McIlwain H, Bachelard H. (1971) Biochemistry and the Central Nervous System, Churchill Livingstone, Edinburgh, United Kingdom: [Google Scholar]
- Murphy A, Vines A, McBean GJ. (2009) Stimulation of EAAC1 in C6 glioma cells by store-operated calcium influx. Biochim Biophys Acta 1788:551–558 [DOI] [PubMed] [Google Scholar]
- Najimi M, Maloteaux JM, Hermans E. (2002) Cytoskeleton-related trafficking of the EAAC1 glutamate transporter after activation of the G(q/11)-coupled neurotensin receptor NTS1. FEBS Lett 523:224–228 [DOI] [PubMed] [Google Scholar]
- Najimi M, Maloteaux JM, Hermans E. (2005) Pertussis toxin-sensitive modulation of glutamate transport by endothelin-1 type A receptors in glioma cells. Biochim Biophys Acta 1668:195–202 [DOI] [PubMed] [Google Scholar]
- Olson JE, Martinho E. (2006) Regulation of taurine transport in rat hippocampal neurons by hypoosmotic swelling. J Neurochem 96:1375–1389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sala G, Beretta S, Ceresa C, Mattavelli L, Zoia C, Tremolizzo L, Ferri A, Carri MT, Ferrarese C. (2005) Impairment of glutamate transport and increased vulnerability to oxidative stress in neuroblastoma SH-SY5Y cells expressing a Cu,Zn superoxide dismutase typical of familial amyotrophic lateral sclerosis. Neurochem Int 46:227–234 [DOI] [PubMed] [Google Scholar]
- Sims KD, Straff DJ, Robinson MB. (2000) Platelet-derived growth factor rapidly increases activity and cell surface expression of the EAAC1 subtype of glutamate transporter through activation of phosphatidylinositol 3-kinase. J Biol Chem 275:5228–5237 [DOI] [PubMed] [Google Scholar]
- Sorensen SD, McEwen EL, Linseman DA, Fisher SK. (1997) Agonist-induced endocytosis of muscarinic cholinergic receptors: relationship to stimulated phosphoinositide turnover. J Neurochem 68:1473–1483 [DOI] [PubMed] [Google Scholar]
- Stadel JM, Strulovici B, Nambi P, Lavin TN, Briggs MM, Caron MG, Lefkowitz RJ. (1983) Desensitization of the β-adrenergic receptor of frog erythrocytes. Recovery and characterization of the down-regulated receptors in sequestered vesicles. J Biol Chem 258:3032–3038 [PubMed] [Google Scholar]
- Sykova E. (2004) Extrasynaptic volume transmission and diffusion parameters of the extracellular space. Neuroscience 129:861–876 [DOI] [PubMed] [Google Scholar]
- Thompson AK, Fisher SK. (1990) Relationship between agonist-induced muscarinic receptor loss and desensitization of stimulated phosphoinositide turnover in two neuroblastomas: methodological considerations. J Pharmacol Exp Ther 252:744–752 [PubMed] [Google Scholar]
- Vazquez-Juarez E, Ramos-Mandujano G, Hernandez-Benitez R, Pasantes-Morales H. (2008) On the role of G protein-coupled receptors in cell volume regulation. Cell Physiol Biochem 21:1–14 [DOI] [PubMed] [Google Scholar]
- Zerangue N, Kavanaugh MP. (1996) Interaction of l-cysteine with a human excitatory amino acid transporter. J Physiol 493:419–423 [DOI] [PMC free article] [PubMed] [Google Scholar]