Abstract
There has recently been intense interest in the clinical measurement of tissue factor (TF)-positive microparticles (MPs) in clinical disease states. This interest has been driven by the demonstration of an putative role for circulating TF-positive MPs in animal models of thrombus propagation. Both immunological and functional assays for MP-TF have been described. While each approach has its own advantages and drawbacks, neither has yet been truly established as the ‘gold standard’. Heterogeneity of TF-bearing MPs, such as the variable co-expression of surface phosphatidylserine, may determine not only their procoagulant potential, but also additional properties including rate of clearance from the circulation.
Keywords: tissue factor, microparticle, assay, phosphatidylserine
TF-bearing microparticles; history and rationale for study
The landmark study of Giesen and colleagues is generally acknowledged as the first demonstration of the existence of circulating MPs containing functionally intact TF in the blood of healthy subjects (1). Until that time, the prevailing paradigm had been that constitutive TF expression in healthy individuals is confined to cells in the extravascular compartment, only coming into contact with factor VII/VIIa after vessel injury (2). The discovery that endotoxin induces TF expression in monocytes served to maintain the focus on intravascular cell-bound TF in certain disease states, such as sepsis (3). Although a few earlier studies had demonstrated the presence of detectable blood-borne TF activity in healthy subjects (4, 5), it was not specifically localized to the MP fraction, with the possible exception of the report that about one third of circulating ‘soluble’ TF antigen was present in a sedimentable fraction of plasma (6).
Elegant studies using intra-vital microscopy to track the origin and accumulation of endogenous TF on the luminal aspect of experimental thrombi in the microvasculature of small animal models soon followed (7). These studies appeared to establish an important role for circulating MP-borne, but not leucocyte-borne TF (8) in the genesis of fibrin formation in vivo. However, there remained some discrepancies in the results of studies designed to evaluate the cellular source (i.e. hematopoietic vs. vessel wall) of MP-TF during thrombus formation. When laser injury was used to initiate clot formation in the microvasculature, circulating TF appeared to be required for normal fibrin generation (9). However, in large vessel thrombosis models, such as in the carotid artery following photochemical injury, vessel wall TF appeared to be much more important than TF derived from hematopoietic sources (10). These inconsistencies have been interpreted to be a result of the particular experimental trigger used to initiate thrombus formation, specifically whether the triggering stimulus leads to endothelial denudation and exposure of the high concentrations of sub-endothelial TF (11). Arguably, venous thrombosis is a more relevant model in which to study the role of circulating TF; unlike many forms of arterial thrombosis, it is generally believed that vascular endothelium below the thrombus remains intact, thereby raising the question as to the source of TF required to initiate thrombosis. Small animal models have demonstrated the presence of abundant TF antigen embedded within thrombi and within adherent leucocytes, (12-14) which appears to correlate with reports in human thrombi (15). However, this evidence clearly does not establish that circulating MPs are the source of the accumulated TF, because antigenically-detectable intravascular TF may also include alternatively spliced TF (asTF), first described in vivo in 2006 (16). In this isoform, the transmembrane and cytoplasmic domains are replaced by a unique 40 amino acid C-terminal domain. Although this soluble form of TF probably does not retain any significant amount of procoagulant activity, it may be important in other biological settings, such as in the promotion of angiogenesis (17). Inhibition of TF expression or synthesis prevented extension of the fibrin clot in some models of venous thrombosis (13, 18). Mice expressing very low levels of hTF showed reduced thrombus size in the IVC ligation model, but no contribution from hematopoietic cell derived, TF-positive MPs could be demonstrated (10).
These animal models remain the best available evidence that circulating MP-TF is an important mediator -- and thus a potential biomarker -- of thrombotic risk in disease states. In addition, studies correlating the cellular source(s) of MP-TF detected in vivo with in vitro observations add weight to the rationale for studying the role of circulating MP-TF in disease states. Thus, the in vitro demonstration that cancer cells generate TF positive MPs (19) appears to have been confirmed by the finding of circulating MPs expressing tumor-specific antigens in the plasma of patients with cancer (20, 21). Similarly, the detection of circulating monocyte-derived MPs expressing TF following exposure to endotoxin in vivo (22) is consistent with earlier in vitro studies on LPS-stimulated monocytes (23).
In this narrative, I will briefly describe the approaches that have been developed to detect and quantify circulating MP-TF in humans, and discuss a few areas in which our existing knowledge base requires additional data.
Methods of MP-TF detection
Most commonly, MP-TF is assayed on circulating MPs in plasma, although other body fluids have also been shown to contain TF-bearing MPs that may have diagnostic and/or prognostic relevance (24). Not all methods of MP-TF detection require a MP isolation step, but at a minimum, there is a need to prepare cell-free samples from blood or the body fluid under study. Centrifugation speed, which must be sufficient to remove contaminating cells, but not the MPs of interest, is a critical variable. Other important pre-analytical variables include: 1] the venipuncture method by which the sample was obtained (since contamination by TF originating from skin and vessel wall cells can be significant, given the minute quantities of TF being measured); 2] the anticoagulant into which the samples are drawn (since calcium chelators may dissociate bound TFPI, among other effects); 3] and whether the samples are analyzed fresh or after freezing (since freeze-thawing may ‘decrypt’ TF expression in the membrane of MPs (25)).
When employed, the specific conditions used to isolate MPs from plasma also vary considerably among research laboratories (26). Few published studies have systematically evaluated the pre-analytical variables that may influence MP-TF detection (25), but it remains an important area for future standardization if MP-TF assays are ever to enter the clinical diagnosis arena.
Broadly speaking, MP-TF assays can be classified as either immunological or functional methods that we will briefly summarize.
A] Immunological
Immunological methods detect TF antigen on the surface of MPs using specific antibodies or other probes that have been labeled with a detector, such as a fluorescent tag. In flow cytometry, an operational definition is used to detect MPs. The usual criteria include events that; 1] are <1μm in size; 2] express phosphatidylserine (PS), which is usually detected by binding of labeled Annexin V or lactadherin; 3] express antigens that characterize the cell of origin; and 4] may be removed by high speed centrifugation (at least 18,000g for 30 minutes (27)). TF may be detected on MPs using specific labeled antibodies, but this approach clearly imparts no information about whether the TF is functionally active, and it is at best semi-quantitative. Flow cytometry also suffers from the fact that particles <0.5μm are smaller than the wavelength of the laser light used to detect them, and thus they may not be detectable. Therefore, there has been recent interest in other physical methods to detect MPs. These approaches have included dynamic light scattering, capillary electrophoresis with laser-induced fluorescence detection, impedance-based flow cytometry, and atomic force microscopy. In theory, all of these methods can be adapted to detect TF and other antigens on the MP surface, although these studies are largely still in their infancy.
B] Functional
Since the earliest demonstration of MP generation by platelets, it has been known that PS expression on MPs contributes to thrombin generation by facilitating assembly of the prothrombinase complex (28). Membrane PS exposure is also important in ensuring that the full procoagulant potential of MP-TF is expressed, and it is an important component of the ‘decryption’ process of TF (Figure 1a). TF-independent procoagulant activity of MPs in plasma may be measured using prothrombinase assays on MPs isolated from cell-free plasma by a capture technique (29) or by high speed centrifugation. Similarly, TF-dependent procoagulant activity may be assayed following isolation by capture (22) or high-speed centrifugation (30). Finally, global assays of thrombin generation such as calibrated automated thrombography have been adapted to measure endogenous MP-TF activity in platelet rich or platelet poor plasma (31).
Figure 1.
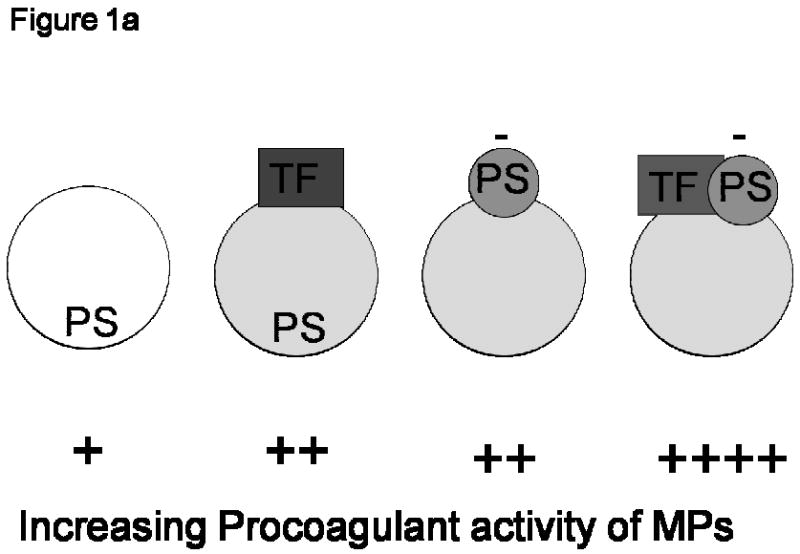
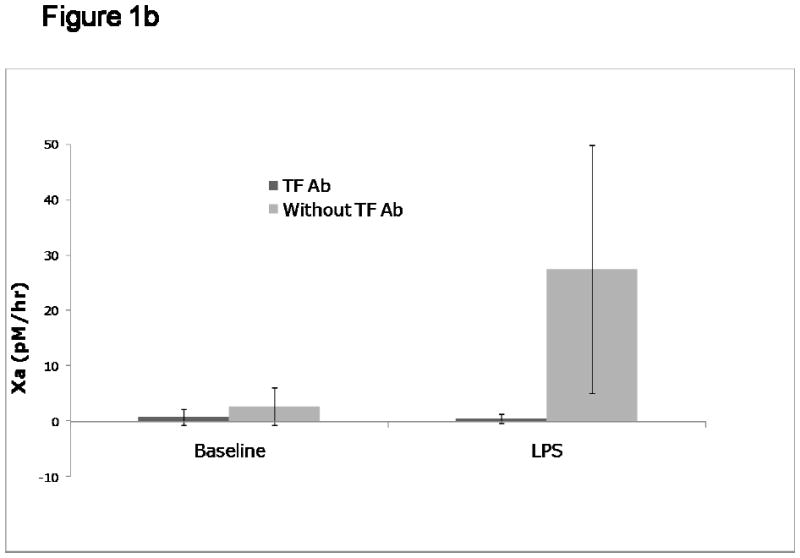
Figure 1a: Cartoon representing the hierarchical procoagulant activity of microparticles (MPs). Depending on their cellular origin, the procoagulant potential of circulating MPs may be either tissue factor (TF)-dependent and/or TF-independent. The MP on the left hand side that maintains PS on the inner leaflet, and does not express TF, would express very little (if any) procoagulant activity. The negative bars (-) adjacent to ‘PS’ indicate the surface expression of negatively charged PS. MPs expressing both TF and negatively charged surface phosphatidylserine (PS), as represented by the particle on the extreme right hand side, would theoretically contribute most to fibrin generation. Figure courtesy of Dr Nigel Mackman.
Figure 1b: TF-dependent vs. TF-independent procoagulant activity on circulating MPs. Platelet free plasma was obtained from human volunteers exposed to endotoxin, and assayed for MP-TF activity as previously described (22). The concentration of factor Xa generated following the addition of 5 nM factor VIIa and 150 nM factor X for 6 hours is shown on the y-axis. Both at baseline and at 4 hours after LPS administration, the Xa generated with and without the addition of an inhibitory polyclonal antibody to TF is shown.
Analytical variables to be considered include not only the sensitivity, but also the specificity of the TF assay. Specificity here is defined as the TF dependence of factor VIIa activation of factor X. In practice, this is usually determined by the addition of a specific blocking antibody to TF in parallel wells. Thus, for example, using the capture assay for MP-TF activity (22), about two thirds of the VIIa-dependent activation of factor X was inhibited by a blocking antibody to TF, whereas >95% of Xa generation was TF-dependent in MPs obtained from plasma following endotoxin exposure (Figure 1b).
Is phosphatidylserine exposure ubiquitous in MPs?
A number of in vitro studies on isolated or cultured cells have shown that MP formation is associated with loss of the membrane phospholipid asymmetry that prevails in healthy cells. Cellular activation or apoptosis induce calcium-dependent activation of scramblase and floppase, and inhibition of flippase activities, with the net result that phosphatidylserine (PS) is exposed on the outer leaflet of the MP membrane (32). Thus, to a large extent, PS exposure on the surface of MPs has become synonymous with the definition of a MP, and this property is exploited in many of the immunological and functional assays for MP-TF already described.
However, the processes of membrane PS exposure and MP generation can be dissociated in isolated cells (33). Furthermore, both in sickle cell disease and in the human endotoxemia model, we detected an increased number of PS-negative MPs (as judged by the absence of annexin V binding) in vivo (22, 34). Others have similarly reported the presence of PS-negative MPs in vivo (35). In vitro, we and others have shown that PS expression on platelet-derived MPs varies according to the stimulus used to generate them (36, 37). In cultured endothelial cells also, the PS content of MPs released into the supernatant may depend on whether they were produced by agonists that result in activation or apoptosis (38).
PS exposure is potentially important not only from the standpoint of the inherent MP procoagulant potential, but also with respect to MP clearance. Much has been learned about the importance of PS exposure on apoptotic cells or senescent red cells, where it appears to be a critical ligand that interacts with several receptors on phagocytic macrophages, resulting in removal of the apoptotic cells (39). Endogenous lactadherin may be an important ‘tag’, binding to PS on MPs and marking them for clearance (40). Theoretically therefore, PS-negative MPs may evade this fate, and could serve as a distinct long lasting ‘surveillance reservoir’ of MPs; TF on these MPs would become decrypted after binding to a PS-expressing membrane, such as the surface of an activated platelet (7, 41, 42).
What is needed to prove that MP-TF plays an important role in thrombus formation?
The concept that a clinically significant concentration of TF circulates in the plasma of healthy subjects has been questioned using sensitive and specific assays. These studies concluded that the concentration of functionally active TF in whole blood of healthy subjects is < 20 fM, which was the lower concentration limit for initiation of coagulation in contact pathway inhibited blood (43). However, other studies have demonstrated that while the addition of 100 fM TF to whole blood is insufficient to initiate coagulation under static conditions, it will promote fibrin formation under physiologic flow conditions (44). Undoubtedly, one of the difficulties in comparing studies is the lack of a TF standard to ensure that comparable concentrations of TF are being evaluated. As such, technical issues with the various assays, including the development of an internationally recognized TF standard, remain an important priority.
Although there are now many cross-sectional studies documenting increased concentrations of MP-TF in a variety of autoimmune, inflammatory and malignant disease states, there are limited data from prospective studies demonstrating that MP-TF levels can be considered a true marker of thrombotic risk. Thus far, it is unclear whether the lack of such data is the result of technical limitations or the lack of such a causative association.
Finally, although recent studies on circulating TF have mostly focused on MP-associated TF, less than 5% of the total circulating pool of TF is associated with MPs (45). Furthermore, high-risk situations for thrombosis -- such as the postoperative period following total knee replacement surgery -- are associated with a temporally relevant increase in mononuclear cell-associated TF activity (45). Therefore, clinical studies focusing on the causative relationship between MP-TF and thrombotic disease should not neglect a possible role for cell-associated TF.
Acknowledgments
The author is supported by NIH grant RO1 HL095096. He wishes to acknowledge the scientific collaboration of colleagues at the University of North Carolina, particularly Drs Nigel Mackman, Raj Kasthuri, Mac Monroe and Jen-Yea Chang, as well as the excellent technical assistance of Patrick Moody and Sam Glover.
Footnotes
Conflict of Interest Statement: The author has no conflicts of interest to declare
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Giesen PL, Rauch U, Bohrmann B, Kling D, Roque M, Fallon JT, et al. Blood-borne tissue factor: another view of thrombosis. Proc Natl Acad Sci U S A. 1999 Mar 2;96(5):2311–5. doi: 10.1073/pnas.96.5.2311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Semeraro N, Colucci M. Tissue factor in health and disease. Thromb Haemost. 1997 Jul;78(1):759–64. [PubMed] [Google Scholar]
- 3.Levi M, ten Cate H, van der Poll T, van Deventer SJ. Pathogenesis of disseminated intravascular coagulation in sepsis. JAMA. 1993 Aug 25;270(8):975–9. [PubMed] [Google Scholar]
- 4.Fukuda C, Iijima K, Nakamura K. Measuring tissue factor (factor III) activity in plasma. Clin Chem. 1989 Sep;35(9):1897–900. [PubMed] [Google Scholar]
- 5.Key NS, Slungaard A, Dandelet L, Nelson SC, Moertel C, Styles LA, et al. Whole blood tissue factor procoagulant activity is elevated in patients with sickle cell disease. Blood. 1998 Jun 1;91(11):4216–23. [PubMed] [Google Scholar]
- 6.Koyama T, Nishida K, Ohdama S, Sawada M, Murakami N, Hirosawa S, et al. Determination of plasma tissue factor antigen and its clinical significance. Br J Haematol. 1994 Jun;87(2):343–7. doi: 10.1111/j.1365-2141.1994.tb04919.x. [DOI] [PubMed] [Google Scholar]
- 7.Falati S, Gross P, Merrill-Skoloff G, Furie BC, Furie B. Real-time in vivo imaging of platelets, tissue factor and fibrin during arterial thrombus formation in the mouse. Nat Med. 2002 Oct;8(10):1175–81. doi: 10.1038/nm782. [DOI] [PubMed] [Google Scholar]
- 8.Gross PL, Furie BC, Merrill-Skoloff G, Chou J, Furie B. Leukocyte-versus microparticle-mediated tissue factor transfer during arteriolar thrombus development. J Leukoc Biol. 2005 Dec;78(6):1318–26. doi: 10.1189/jlb.0405193. [DOI] [PubMed] [Google Scholar]
- 9.Chou J, Mackman N, Merrill-Skoloff G, Pedersen B, Furie BC, Furie B. Hematopoietic cell-derived microparticle tissue factor contributes to fibrin formation during thrombus propagation. Blood. 2004 Nov 15;104(10):3190–7. doi: 10.1182/blood-2004-03-0935. [DOI] [PubMed] [Google Scholar]
- 10.Day SM, Reeve JL, Pedersen B, Farris DM, Myers DD, Im M, et al. Macrovascular thrombosis is driven by tissue factor derived primarily from the blood vessel wall. Blood. 2005 Jan 1;105(1):192–8. doi: 10.1182/blood-2004-06-2225. [DOI] [PubMed] [Google Scholar]
- 11.Furie B, Furie BC. Mechanisms of thrombus formation. N Engl J Med. 2008 Aug 28;359(9):938–49. doi: 10.1056/NEJMra0801082. [DOI] [PubMed] [Google Scholar]
- 12.Zhou J, May L, Liao P, Gross PL, Weitz JI. Inferior vena cava ligation rapidly induces tissue factor expression and venous thrombosis in rats. Arterioscler Thromb Vasc Biol. 2009 Jun;29(6):863–9. doi: 10.1161/ATVBAHA.109.185678. [DOI] [PubMed] [Google Scholar]
- 13.Li YD, Ye BQ, Zheng SX, Wang JT, Wang JG, Chen M, et al. NF-kappaB transcription factor p50 critically regulates tissue factor in deep vein thrombosis. J Biol Chem. 2009 Feb 13;284(7):4473–83. doi: 10.1074/jbc.M806010200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hoffman M, Whinna HC, Monroe DM. Circulating tissue factor accumulates in thrombi, but not in hemostatic plugs. J Thromb Haemost. 2006 Sep;4(9):2092–3. doi: 10.1111/j.1538-7836.2006.02085.x. [DOI] [PubMed] [Google Scholar]
- 15.Himber J, Kling D, Fallon JT, Nemerson Y, Riederer MA. In situ localization of tissue factor in human thrombi. Blood. 2002 Jun 1;99(11):4249–50. doi: 10.1182/blood-2002-02-0557. [DOI] [PubMed] [Google Scholar]
- 16.Bogdanov VY, Kirk RI, Miller C, Hathcock JJ, Vele S, Gazdoiu M, et al. Identification and characterization of murine alternatively spliced tissue factor. J Thromb Haemost. 2006 Jan;4(1):158–67. doi: 10.1111/j.1538-7836.2005.01680.x. [DOI] [PubMed] [Google Scholar]
- 17.van den Berg YW, van den Hengel LG, Myers HR, Ayachi O, Jordanova E, Ruf W, et al. Alternatively spliced tissue factor induces angiogenesis through integrin ligation. Proc Natl Acad Sci U S A. 2009 Nov 17;106(46):19497–502. doi: 10.1073/pnas.0905325106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Himber J, Wohlgensinger C, Roux S, Damico LA, Fallon JT, Kirchhofer D, et al. Inhibition of tissue factor limits the growth of venous thrombus in the rabbit. J Thromb Haemost. 2003 May;1(5):889–95. doi: 10.1046/j.1538-7836.2003.00110.x. [DOI] [PubMed] [Google Scholar]
- 19.Dvorak HF, Van DeWater L, Bitzer AM, Dvorak AM, Anderson D, Harvey VS, et al. Procoagulant activity associated with plasma membrane vesicles shed by cultured tumor cells. Cancer Res. 1983 Sep;43(9):4434–42. [PubMed] [Google Scholar]
- 20.Tesselaar ME, Romijn FP, Van Der Linden IK, Prins FA, Bertina RM, Osanto S. Microparticle-associated tissue factor activity: a link between cancer and thrombosis? J Thromb Haemost. 2007 Mar;5(3):520–7. doi: 10.1111/j.1538-7836.2007.02369.x. [DOI] [PubMed] [Google Scholar]
- 21.Skog J, Wurdinger T, van Rijn S, Meijer DH, Gainche L, Sena-Esteves M, et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol. 2008 Dec;10(12):1470–6. doi: 10.1038/ncb1800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Aras O, Shet A, Bach RR, Hysjulien JL, Slungaard A, Hebbel RP, et al. Induction of microparticle- and cell-associated intravascular tissue factor in human endotoxemia. Blood. 2004 Jun 15;103(12):4545–53. doi: 10.1182/blood-2003-03-0713. [DOI] [PubMed] [Google Scholar]
- 23.Satta N, Toti F, Feugeas O, Bohbot A, Dachary-Prigent J, Eschwege V, et al. Monocyte vesiculation is a possible mechanism for dissemination of membrane-associated procoagulant activities and adhesion molecules after stimulation by lipopolysaccharide. J Immunol. 1994 Oct 1;153(7):3245–55. [PubMed] [Google Scholar]
- 24.Bastarache JA, Fremont RD, Kropski JA, Bossert FR, Ware LB. Procoagulant alveolar microparticles in the lungs of patients with acute respiratory distress syndrome. Am J Physiol Lung Cell Mol Physiol. 2009 Dec;297(6):L1035–41. doi: 10.1152/ajplung.00214.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lechner D, Weltermann A. Circulating tissue factor-exposing microparticles. Thromb Res. 2008;122 1:S47–54. doi: 10.1016/S0049-3848(08)70019-7. [DOI] [PubMed] [Google Scholar]
- 26.Jy W, Horstman LL, Jimenez JJ, Ahn YS, Biro E, Nieuwland R, et al. Measuring circulating cell-derived microparticles. J Thromb Haemost. 2004 Oct;2(10):1842–51. doi: 10.1111/j.1538-7836.2004.00936.x. [DOI] [PubMed] [Google Scholar]
- 27.Dignat-George F, Freyssinet JM, Key NS. Centrifugation is a crucial step impacting microparticle measurement. Platelets. 2009 May;20(3):225–6. doi: 10.1080/09537100902795500. author reply 7-8. [DOI] [PubMed] [Google Scholar]
- 28.Sandberg H, Bode AP, Dombrose FA, Hoechli M, Lentz BR. Expression of coagulant activity in human platelets: release of membranous vesicles providing platelet factor 1 and platelet factor 3. Thromb Res. 1985 Jul 1;39(1):63–79. doi: 10.1016/0049-3848(85)90122-7. [DOI] [PubMed] [Google Scholar]
- 29.Aupeix K, Hugel B, Martin T, Bischoff P, Lill H, Pasquali JL, et al. The significance of shed membrane particles during programmed cell death in vitro, and in vivo, in HIV-1 infection. J Clin Invest. 1997 Apr 1;99(7):1546–54. doi: 10.1172/JCI119317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang JG, Manly D, Kirchhofer D, Pawlinski R, Mackman N. Levels of microparticle tissue factor activity correlate with coagulation activation in endotoxemic mice. J Thromb Haemost. 2009 Jul;7(7):1092–8. doi: 10.1111/j.1538-7836.2009.03448.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ollivier V, Wang J, Manly D, Machlus KR, Wolberg AS, Jandrot-Perrus M, et al. Detection of endogenous tissue factor levels in plasma using the calibrated automated thrombogram assay. Thromb Res. 2009 Apr 3; doi: 10.1016/j.thromres.2009.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bevers EM, Comfurius P, Dekkers DW, Harmsma M, Zwaal RF. Transmembrane phospholipid distribution in blood cells: control mechanisms and pathophysiological significance. Biol Chem. 1998 Aug-Sep;379(8-9):973–86. [PubMed] [Google Scholar]
- 33.Siljander P, Farndale RW, Feijge MA, Comfurius P, Kos S, Bevers EM, et al. Platelet adhesion enhances the glycoprotein VI-dependent procoagulant response: Involvement of p38 MAP kinase and calpain. Arterioscler Thromb Vasc Biol. 2001 Apr;21(4):618–27. doi: 10.1161/01.atv.21.4.618. [DOI] [PubMed] [Google Scholar]
- 34.Shet AS, Aras O, Gupta K, Hass MJ, Rausch DJ, Saba N, et al. Sickle blood contains tissue factor-positive microparticles derived from endothelial cells and monocytes. Blood. 2003 Oct 1;102(7):2678–83. doi: 10.1182/blood-2003-03-0693. [DOI] [PubMed] [Google Scholar]
- 35.Amabile N, Guerin AP, Leroyer A, Mallat Z, Nguyen C, Boddaert J, et al. Circulating endothelial microparticles are associated with vascular dysfunction in patients with end-stage renal failure. J Am Soc Nephrol. 2005 Nov;16(11):3381–8. doi: 10.1681/ASN.2005050535. [DOI] [PubMed] [Google Scholar]
- 36.Perez-Pujol S, Marker PH, Key NS. Platelet microparticles are heterogeneous and highly dependent on the activation mechanism: studies using a new digital flow cytometer. Cytometry A. 2007 Jan;71(1):38–45. doi: 10.1002/cyto.a.20354. [DOI] [PubMed] [Google Scholar]
- 37.Rukoyatkina N, Begonja AJ, Geiger J, Eigenthaler M, Walter U, Gambaryan S. Phosphatidylserine surface expression and integrin alpha IIb beta 3 activity on thrombin/convulxin stimulated platelets/particles of different sizes. Br J Haematol. 2009 Feb;144(4):591–602. doi: 10.1111/j.1365-2141.2008.07506.x. [DOI] [PubMed] [Google Scholar]
- 38.Jimenez JJ, Jy W, Mauro LM, Soderland C, Horstman LL, Ahn YS. Endothelial cells release phenotypically and quantitatively distinct microparticles in activation and apoptosis. Thromb Res. 2003 Feb 15;109(4):175–80. doi: 10.1016/s0049-3848(03)00064-1. [DOI] [PubMed] [Google Scholar]
- 39.Ravichandran KS, Lorenz U. Engulfment of apoptotic cells: signals for a good meal. Nat Rev Immunol. 2007 Dec;7(12):964–74. doi: 10.1038/nri2214. [DOI] [PubMed] [Google Scholar]
- 40.Dasgupta SK, Abdel-Monem H, Niravath P, Le A, Bellera RV, Langlois K, et al. Lactadherin and clearance of platelet-derived microvesicles. Blood. 2009 Feb 5;113(6):1332–9. doi: 10.1182/blood-2008-07-167148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rauch U, Bonderman D, Bohrmann B, Badimon JJ, Himber J, Riederer MA, et al. Transfer of tissue factor from leukocytes to platelets is mediated by CD15 and tissue factor. Blood. 2000 Jul 1;96(1):170–5. [PubMed] [Google Scholar]
- 42.Del Conde I, Shrimpton CN, Thiagarajan P, Lopez JA. Tissue-factor-bearing microvesicles arise from lipid rafts and fuse with activated platelets to initiate coagulation. Blood. 2005 Sep 1;106(5):1604–11. doi: 10.1182/blood-2004-03-1095. [DOI] [PubMed] [Google Scholar]
- 43.Butenas S, Bouchard BA, Brummel-Ziedins KE, Parhami-Seren B, Mann KG. Tissue factor activity in whole blood. Blood. 2005 Apr 1;105(7):2764–70. doi: 10.1182/blood-2004-09-3567. [DOI] [PubMed] [Google Scholar]
- 44.Okorie UM, Denney WS, Chatterjee MS, Neeves KB, Diamond SL. Determination of surface tissue factor thresholds that trigger coagulation at venous and arterial shear rates: amplification of 100 fM circulating tissue factor requires flow. Blood. 2008 Apr 1;111(7):3507–13. doi: 10.1182/blood-2007-08-106229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Johnson GJ, Leis LA, Bach RR. Tissue factor activity of blood mononuclear cells is increased after total knee arthroplasty. Thromb Haemost. 2009 Oct;102(4):728–34. doi: 10.1160/TH09-04-0261. [DOI] [PubMed] [Google Scholar]


