Abstract
Rho GTPases are key regulators of actin dynamics. We report that the Rho GTPase TCL, which is closely related to Cdc42 and TC10, localizes to the plasma membrane and the early/sorting endosomes in HeLa cells, suggesting a role in the early endocytic pathway. Receptor-dependent internalization of transferrin (Tf) is unaffected by suppression of endogenous TCL by small interfering RNA treatment. However, Tf accumulates in Rab5-positive uncoated endocytic vesicles and fails to reach the early endosome antigen-1–positive early endosomal compartments and the pericentriolar recycling endosomes. Moreover, Tf release upon TCL knockdown is significantly slower. Conversely, in the presence of dominant active TCL, internalized Tf accumulates in early endosome antigen-1–positive early/sorting endosomes and not in perinuclear recycling endosomes. Tf recycles directly from the early/sorting endosomes and it is normally released by the cells. The same phenotype is generated by replacing the C terminus of dominant active Cdc42 and TC10 with that of TCL, indicating that all three proteins share downstream effector proteins. Thus, TCL is essential for clathrin-dependent endocytosed receptors to enter the early/sorting endosomes. Furthermore, the active GTPase favors direct recycling from early/sorting endosomes without accumulating in the perinuclear recycling endosomes.
INTRODUCTION
In addition to the prominent role of the GTPases of the Rab family as regulators of membrane traffic (Zerial and McBride, 2001), the actin and microtubule cytoskeleton plays a key role in vesicle movement and fusion through the action of the small GTPases from the Rho family (Etienne-Manneville and Hall, 2002). Rho proteins are molecular switches that are mostly known for their effects on actin cytoskeleton remodeling. The Rho family contains 21 members in mammals (Vignal et al., 2003), among which is TCL, the closest relative to TC10 and Cdc42 (Vignal et al., 2000). Like Cdc42, Rac1, and TC10, TCL can bind to proteins containing a Cdc42/Rac interactive binding (CRIB) domain. Overexpression of TCL induces cortical actin reorganization, as well as the formation of very dynamic ruffle-like structures associated with the formation of large cytoplasmic vesicles in REF52 fibroblast cells (Vignal et al., 2000). Although TCL and TC10 share common effectors, they have differential effects on actin cytoskeleton reorganization (Vignal et al., 2000). The divergence in their C-terminal domains suggests that the two proteins might be targeted to different intracellular membranes and thus have distinct functions.
Rho GTPases are involved in various cellular processes, including adhesion, cell polarization, motility and transformation, gene activation, and vesicular trafficking (Etienne-Manneville and Hall, 2002). Several Rho GTPases apparently regulate different aspects of intracellular membrane dynamics. On uptake of extracellular material, plasma membrane reorganization occurs. This involves the GTPases Rac1, RhoA, or Cdc42, during pinocytosis (Nobes and Marsh, 2000; Fiorentini et al., 2001) and phagocytosis (Ridley, 2001a,b). These Rho GTPases may also be involved in clathrin-mediated endocytosis (Lamaze et al., 1996; Ellis and Mellor, 2000; Rojas et al., 2001). Intracellular vesicle trafficking is also regulated by Rho GTPases. RhoG regulates lysosomal dynamics (Vignal et al., 2001), whereas Cdc42 controls transport from Golgi to endoplasmic reticulum (ER) (Luna et al., 2002). Several Rho GTPases also regulate endosomal vesicle trafficking: RhoD is involved in early/sorting endosome (EE/SE) movements (Murphy et al., 2001; Gasman et al., 2003), whereas RhoB seems to control transfer from the early to the late endosomal compartments (Gampel et al., 1999). In adipocytes, TC10 participates in the regulation of insulin-dependent transfer of the glucose transporter GLUT4 from its storage compartment to the plasma membrane. Notably, TC10 overexpression inhibits insulin-stimulated GLUT4 receptor translocation (Chiang et al., 2001). Transport of the GLUT4-containing vesicles involves actin cytoskeleton reorganization induced by TC10 activation (Kanzaki et al., 2001), possibly through N-WASP recruitment (Jiang et al., 2002). In contrast to TC10, TCL does not to affect GLUT4 translocation from its storage compartment to the plasma membrane in 3T3L1 adipocytes (Chiang et al., 2002).
Here, we investigate the specific cellular roles of TCL. We show that TCL localizes at the plasma membrane and in the EE/SE. Suppression of endogenous TCL by small interfering RNA (siRNA) perturbs the distribution of receptor-dependent internalized transferrin (Tf): Tf does not enter the early/sorting endosomes and remains in decoated endocytic vesicles. Furthermore, Tf release is slower when TCL is knocked down. On the other hand, a constitutively active mutant of TCL (TCLda) provokes sequestering of Tf in EE/SE. If Tf cannot reach the perinuclear recycling endosomes (REs), it is still able to recycle directly from the early/sorting endosomes. Finally, TC10da and Cdc42da targeted to the TCL compartment have the same inhibitory effect as TCLda. We conclude that TCL is a new Rho GTPase involved in clathrin-dependent endocytosis: on one hand, TCL is necessary for the transport of endocytosed receptors from the decoated endocytic vesicles to the early/sorting endosomes and normal recycling, whereas on the other, TCL inactivation is required for endocytosed receptors to reach perinuclear recycling endosomes.
MATERIALS AND METHODS
Antibodies and Plasmids
Antibodies raised against GST-TCL (Vignal et al., 2000) were affinity purified on an MBP-TCL resin. Affinity-purified anti-Rab11 antibodies were described previously (Wilcke et al., 2000). Other primary antibodies: anti-transferrin receptor (TfR) and anti-green fluorescent protein (GFP) (Zymed Laboratories, South San Francisco, CA), anti-early endosome antigen-1 (EEA1) (Santa Cruz Biotechnology, Santa Cruz, CA), anti-histone H3 (Upstate Biotechnology, Lake Placid, NY), anti-p115 and anti-p230 (Transduction Laboratories, Lexington, KY), anti-cathepsin D (Sigma-Aldrich, St. Louis, MO), and anti-Rab5 (BD Bioscience PharMingen, San Diego, CA). Anti-Lamp1 antibodies were a gift from Bruno Goud (Institut Curie, Paris, France), and anti-glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and anti-enhanced green fluorescent protein (EGFR) were gifts from Urszula Hibner (Institut deGénétique Moléculaire, Montpellier, France). Fluorescent Alexa 568- or Alexa 488-coupled anti-mouse, anti-rabbit, and anti-goat antibodies secondary antibodies, tetramethylrhodamine, and Alexa 647-conjugated Tf were from Molecular Probes (Eugene, OR) and horseradish peroxidase-conjugated secondary antibodies were from Amersham Biosciences (Piscataway, NJ).
Green fluorescent protein (GFP) and myc-tagged TCL, TC10, RhoG, and Cdc42 expression vectors were described previously (Gauthier-Rouviere et al., 1998; Vignal et al., 2000). Uncaxxed TCL was obtained by PvuII deletion in the cDNA. The various chimerical proteins were all engineered by polymerase chain reaction (PCR) and subcloned in pEGFP (BD Biosciences Clontech, Palo Alto, CA) according to a similar strategy. The resulting chimeric proteins are fused in the conserved ECSAL motif, which stands just upstream the variable basic rich region followed by the C-terminal CAAX box. For example, to generate the chimeric protein between TC10Q75L and TCL C terminus, an oligonucleotide coding the conserved motif ECSALTQ, matching half with TCL and half with TC10 nucleotide sequences, was combined with 5′-TC10 and 3′-TCL–specific border oligonucleotides to amplify two fragments: TC10-1 coding amino acids 1–176 of TC10 and TCL-2 coding amino acids 174–214 of TCL. Hybrid open reading frame was then generated by PCR by mixing the complementary fragments TC10-1 and TCL-2, and amplifying with 5′-TC10 and 3′-TCL border oligonucleotides produced TC10daCterTCL. PCR fragments were digested with EcoRI and XhoI and subcloned in pEGFP. TCLwt expression vector was obtained by inserting complete TCL cDNA (Vignal et al., 2000) in pIRES-EGFP bicistronic vector (BD Biosciences Clontech). GFP-Rab4 expression vector was a gift from Mary McCauffray (Cell and Molecular Biology Laboratory, Cork, Ireland), GFP-Rab5 expression vector was a gift from Michel Vidal (University of Montpellier II, Montpellier, France), GFP-Rab7 expression vector was a gift from Bo van Deurs (University of Copenhagen, Copenhagen, Denmark).
Short interfering RNA constructs were made in pSUPER polymerase III expression vector (Brummelkamp et al., 2002), a kind gift of R. Agami (The Netherland's Cancer Institute, Plesmanlaan, The Netherlands). To suppress endogenous TCL expression, oligonucleotide gatccccGGACTGCATGCCTCACGTGCCttcaagagaGGCACGTGAGGCATGCAGTCCtttttggaaaagct was inserted into pSUPER. Bold letters correspond to an inverted repeat of nucleotides 360–380 of TCL open reading frame, a region with no homology to TC10 (Vignal et al., 2000). A pSuper construct expressing a siRNA against the unrelated human protein interferon receptor 1 was used as a control (Uze et al., 1990).
Cell Culture and Immunocytochemistry
HeLa cells were grown in DMEM (Invitrogen, Carlsbad, CA) supplemented with 10% fetal calf serum in a 6% humidified CO2 incubator. Transfections were performed by LipofectAMINE or calcium phosphate precipitation methods as described previously (Vignal et al., 2000; Wilcke et al., 2000). For immunofluorescence, cells were fixed for 10 min in 3.7% formalin (in phosphate-buffered saline [PBS]) followed by a 15-min permeabilization in 0.1% saponin (in PBS containing 0.1% bovine serum albumin) and incubation in PBS containing 0.1% bovine serum albumin (BSA). Expression of GFP-tagged proteins was visualized directly. Other proteins were visualized by double labeling with primary antibodies for 60 min followed by incubation with Alexa 350-, 488-, or 546-conjugated anti-IgG for 40 min (Molecular Probes). Cells were washed in PBS and mounted in Mowviol (Aldrich Chemical, Milwaukee, WI). For wide-field and deconvolved images, cells were observed using a DMR B microscope (Leica Microsystems, Deerfield, IL) by using a 100× (numerical aperture 1.4) PLAPO lens. Images were captured with a MicroMax 1300 Y/HS (B/W) cooled (–10°C) charge-coupled device camera as 16-bit images, by using a MetaMorph (version 4.11) control program (Princeton Scientific Instruments, Monmouth Junction, NJ) run by a PC-compatible microcomputer. Image stacks were deconvolved using Huygens2.3.5p2a program (Scientific Volume Imaging SVI, Hilversum, The Netherlands). For confocal microscopy, images were acquired using a confocal microscope (TCS4D or SP2; Leica Microsystems). Individual wide-field, deconvolved, or confocal sections were further processed using Adobe Photoshop. Fluorescence quantifications were performed using MetaMorph (version 4.11) program (Princeton Scientific Instruments).
Subcellular Fractionation
Subcellular fractionations were performed as described previously (Mary et al., 2002). Briefly, transfected HeLa cells were lysed in cold hypotonic buffer containing 10 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM dithiothreitol, and 1 mM phenylmethylsulfonyl fluoride. Cell extracts were centrifuged (600 × g for 5 min at 4°C) to pellet nuclei and nuclei-associated structures, including the Golgi and endoplasmic reticulum membranes (N). Postnuclear supernatants were ultracentrifugated (100,000 × g for 45 min) to separate cytoplasmic membranes (M) and cytosolic proteins (C). Samples were fractionated on a 12% SDS-polyacrylamide gel and transferred to nitrocellulose membranes.
Percoll Gradients
Percoll gradients were performed as described previously (Meresse et al., 1995). Briefly, transfected HeLa cells were scrapped and pelleted for 5 min at 800 rpm, overlaid with 3 ml of homogenization buffer (250 mM sucrose, 10 mM triethanolamine, 10 mM acetic acid, 1 mM EDTA, pH 7.4, and 1 mM phenylmethylsulfonyl fluoride) and centrifuged for 5 min at 3000 rpm. Cells were resuspended in 1 ml of homogenization buffer and homogenized by 10 passages through a 22-gauge needle. After centrifugation for 10 min at 3000 rpm, the postnuclear supernatant was loaded at the top of a 9-ml cushion of 27% Percoll in homogenization buffer. Gradients were centrifuged in a 50Ti rotor for 65 min at 23,000 rpm, 4°C. Fractions of 0.5 ml were then collected from the top of the gradient. Each fraction was supplemented with 0.25% NP-40 and centrifuged for 25 min at 70,000 rpm in a TL100.2 rotor. Percoll-free supernatants were precipitated with 40% ammonium sulfate when appropriate and used for Western blots.
Quantification of mRNA by Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
HeLa cells cotransfected with pSuper constructs and pEGFP were sorted 24 h after transfection by using a FACSvantage (BD Biosciences, San Jose, CA) measuring GFP fluorescence in FL-1. Total RNAs were then extracted from sorted transfected cells and oligodT-primed cDNAs were prepared using Superscript II reverse transcriptase (Invitrogen). The levels of the various cDNAs were determined by quantitative PCR by using the SYBR Green I technology on a Light Cycler (Roche Diagnostics, Sommerville, NJ) with the Taq Platinium DNA polymerase (Invitrogen). The primer pairs used were as follows: 5′-GCTACGCCAACGACGCCTTC-3′ and 5′-TTGAGCTCGGGGACCCATTC-3′ for TCL and 5′-CTTCGACCACTACGCAGTCA-3′ and 5′-GTTCCGGTACCCACTCCTCT-3′ for TC10. The specificity of the primers was tested by DNA sequencing. All data are presented as a ratio to the GAPDH cDNA level (95% confidence limits).
Transferrin Internalization and Release
HeLa cells were depleted from endogenous Tf by incubation at 37°C for 1 h in internalization medium (DMEM, 10 mM HEPES, pH 7.4, and 0.1% BSA). For steady-state studies, cells were incubated for 45 min at 37°C with fluorescent labeled Tf to a final concentration of 5 μg/ml in internalization medium and then rinsed twice with ice-cold internalization medium before fixation in paraformaldehyde. For Tf chase experiments, cells were first incubated for 20 min at 19°C with fluorescent-labeled Tf as described above, rinsed twice with ice-cold internalization medium, and then incubated for 20 min at 37°C in internalization medium, rinsed, and fixed as described above. Tf uptake and release were performed essentially as described previously (Perez et al., 2002). Briefly, for Tf uptake cells expressing GFP, GFP-TCLda or TCL siRNA was incubated in recycling medium (DMEM, 10% fetal calf serum, 10 mM HEPES, pH 7.4, 0.1% BSA, and 5 μg/ml Alexa 647-labeled Tf) at 37°C. For Tf release, after 60 min of uptake, cells were incubated at 37°C in recycling medium (DMEM, 10% fetal calf serum, 10 mM HEPES, pH 7.4, 0.1% BSA, and 50 μg/ml unlabeled Tf). At desired time points, cells were fixed in 1% paraformaldehyde and fluorescence-activated cell sorting (FACS) analysis was performed using a FACScalibur (BD Biosciences), measuring GFP fluorescence in FL1 and Alexa 647 in FL-4. At last 103 cells were counted in each window.
RESULTS
TCL Is Inserted in Plasma and Intracellular Membranes of HeLa Cells
The expression and subcellular localization of TCL was studied using affinity-purified polyclonal antibodies to TCL (Vignal et al., 2000). On Western blots, the antibodies recognized myc-tagged wild-type TCL (mycTCLwt) expressed in HeLa cells, unlike the preimmune serum (Figure 1A, a). They immunoprecipitated endogenous TCL that migrated in SDS-PAGE as a 25-kDa band. This comigrated with the immunoprecipitated protein expressed from the full-length TCL cDNA inserted in a bicistronic vector (Figure 1A, b). Anti-TCL and anti-myc antibodies showed the same staining pattern in HeLa cells expressing myc-TCLwt. Thus, our anti-TCL antibodies detected overexpressed TCL by immunofluorescence (Figure 1B, a and b), whereas preimmune serum did not (Figure 1B, c and d). However, we were unable to detect the endogenous protein by immunofluorescence (our unpublished data), possibly due to the low amount of endogenous protein. Notably, the anti-TCL antibodies did not recognize overexpressed TC10 in Western blot and immunofluorescence experiments (our unpublished data).
Figure 1.
Analysis of the subcellular localization of TCL by cell fractionation. (A) Specificity of anti-TCL antibodies in Western blot and immunoprecipitation experiments. a, extracts of HeLa cells overexpressing myc-TCLwt were analyzed by Western blots with anti-myc (αmyc) and anti-TCL (αTCL) antibodies or anti-TCL preimmune serum (PI). b, extracts of HeLa cells untransfected (12.106 cells, lanes 1 and 3) or overexpressing untagged TCLwt (6 × 106 cells, lane 2) were immunoprecipitated with immune (αTCL) or preimmune (PI) serum and analyzed by Western blot by using αTCL antibodies. (B) Specificity of anti-TCL antibodies in indirect immunofluorescence. HeLa cells expressing myc-TCLwt were processed for immunofluorescence by using αTCL (a), αmyc (b and d) antibodies, or preimmune serum (c) followed by Alexa 488-coupled anti-rabbit (a and c) and Alexa 568-coupled anti-mouse (b and d) secondary antibodies. Bar, 10 μm. (C) TCL associates with cellular membranes through its CAAX box. Nuclei and associated membranes (N), cytoplasm (C) and plasma membrane and microsomes (M) fractions were prepared from 4 × 106 nontransfected HeLa cells (TCL) or 2 × 106 cells expressing either myc-tagged wild-type TCL (mycTCLwt), dominant active Q79L TCL mutant (mycTCLda), dominant negative T35N TCL mutant (mycTCLdn), or uncaaxed TCL mutant (mycTCLΔCAAX). Fractions were analyzed by immunoblotting by using αTCL (TCL lane) or αmyc (other TCL lanes) antibodies. Antibodies to Histone H3, GAPDH, and EGFR were used as controls for the fractionation. (D) TCL distribution in a Percoll gradient. Postnuclear supernatants from 15 × 106 untransfected HeLa cells (a) or 106 cells expressing either mycTCLwt (b) or mycTCLda (c) were fractionated on a 27% Percoll gradient. Fractions (1–18) were collected from the top of the gradient and analyzed by Western blotting for TCL, TfR, Rab5, and cathepsin D (CathD). The numbers refer to the gradient fractions.
To localize the protein intracellularly, HeLa cell extracts were separated in three fractions: nucleus and associated membranes (N), cytosol (C), and high-speed sedimenting membranes, corresponding to plasma membrane and microsomes (M) (Figure 1C). Endogenous TCL, as well as overexpressed wild-type, dominant active Q79L (TCLda) or inactive T35N (TCLdn) mutant proteins was enriched in the two membrane fractions (N and M) and absent from the cytosol (C) (Figure 1C). Deletion of the carboxy-terminal polybasic and CAAX box motives of TCL, which are required for membrane anchoring, resulted the protein relocalizing in the cytosolic fraction (Figure 1C, mycTCLΔCter lane).
Postnuclear supernatants, corresponding to the C and M fractions described above were then fractionated on a 27% Percoll gradient to separate lysosomes from the plasma membrane and the other vesicular compartments (Meresse et al., 1995). Endogenous TCL protein predominantly fractionated in the medium fractions of the gradient, corresponding to the vesicular compartments that contain TfR and Rab5 and in the upper fractions of the gradient corresponding to the plasma membrane. TCL was absent from the bottom lysosomal fractions containing cathepsin D (Figure 1D, a). Importantly, myc-TCLwt and myc-TCLda proteins showed similar fractionation patterns (Figure 1D, b and c, respectively), as did the overexpressed untagged protein (our unpublished data). These results show that endogenous TCL and expressed TCL mutants are inserted in cellular membranes through the C-terminal polybasic and CAAX box sequences. TCL is present at the plasma membrane and in various intracellular membrane compartments but not in lysosomes.
TCL Localizes at the Plasma Membrane and in Early Endocytic Compartments
TCL was localized more precisely using immunofluorescence and colocalization with endogenous makers or GFP-tagged Rab proteins. In HeLa cells expressing mycTCLwt, TCL immunoreactivity was detected at the plasma membrane (Figure 1B, a and b) and as a punctuate staining throughout the cytoplasm (Figure 2, b, d, f, h, and j). TCL significantly colocalized with EEA1 (Figure 2, a and b), a marker of the EE/SE compartment, and TfR (Figure 2, c and d), which cycles between the plasma membrane, EE/SE, and RE compartments. No colocalization was observed with the cis- and median-Golgi marker p115 (Figure 2, e–f), with the lysosomal protein Lamp1 (Figure 2, g and h) or with the trans-Golgi network marker p230 (Figure 2, i and j). The morphology of these compartments was not affected by mycTCLwt expression (our unpublished data). These results corroborate and expand the fractionation experiments, indicating that TCL localizes to the plasma membrane and compartments of the endocytic pathway.
Figure 2.
TCL colocalizes with early/sorting and recycling endosome markers. Double immunofluorescence was performed on HeLa cells expressing mycTCLwt by using anti-TCL (b, d, f, h, and j), anti-EEA1, an early/sorting endosome marker (a); anti-TfR, an early/sorting and recycling endosome marker (c); anti-p115, a cis- and median-Golgi marker (e); anti-Lamp1, a lysosome marker (g); and anti-p230, a trans-Golgi marker (i). Deconvolved optical section images (0.3 μm) are shown. The boxed areas were superimposed (insets), where the yellow color reveals colocalization representative of the whole cell. Bar, 10 μm.
Different Rab GTPases define distinct compartments within the endocytic pathway. Rab4 and Rab5 are in early/sorting endosomes, whereas Rab11 is found in recycling endosomes and Rab7 locates in the late endosomes (Zerial and McBride, 2001). GFP-tagged wild-type Rab GTPases have been widely used to define the various compartments of the endosomal pathway (Meresse et al., 1995; Bottger et al., 1996; Sonnichsen et al., 2000). When coexpressed with GFP-tagged Rab4, 5, and 7, myc-TCLwt shows substantial colocalization with Rab4 and Rab5 (Figure 3, a–d), but not with Rab7 (Figure 3, e and f). GFP-TCLwt only shows very limited colocalization with endogenous Rab11 (Figure 3, g and h). Dominant active TCL presented a similar distribution (our unpublished data). In HeLa cells, mycTCLdn occurred in very bright and scattered aggregates, suggesting the protein is degraded (our unpublished data). Therefore, we did not use this mutant for further functional analyses. In summary, TCL- and the Q79L-activated mutant protein associate predominantly with membranes of the EE/SE and the plasma membrane. It is absent from recycling and late endosomes, from the Golgi apparatus, and secretion vesicles.
Figure 3.
TCL is localized in early/sorting endosomes with Rab4 and Rab5. Immunofluorescence was performed on HeLa cells expressing mycTCLwt alone (g and h) or together with GFP-Rab4 (a and b), GFP-Rab5 (c and d), and GFP-Rab7 (e and f). Fixed cells were stained with anti-TCL (b, d, and f), anti-myc (h), and anti-Rab11 antibodies (g). The boxed areas were superimposed (insets), where the yellow color reveals colocalization representative of the whole cell. Bar, 10 μm.
Suppression of Endogenous TCL Expression Impairs Normal Functioning of the Early Endocytic Pathway
We next examined the effect of suppressing TCL expression on the endosomal pathway, by using an siRNA delivered by a pSuper polymerase III expression vector (Brummelkamp et al., 2002). TCL siRNA specifically inhibited the expression of GFP-tagged wild-type TCL, whereas the control siRNA had no effect (Figure 4A). To examine the effect of siRNA expression on endogenous TCL mRNA, HeLa cells were transfected with the pSuper constructs, sorted by FACS by using a GFP marker, and the amount of TCL mRNA was determined by quantitative RT-PCR. In cells expressing TCL siRNA, the level of TCL mRNA was ∼40% the amount in control pSuper transfected cells, whereas TC10 mRNA was not affected (Figure 4B). Trace amounts of TCL mRNA were detected 72 h after transfection, and the level was too low for reliable quantification (our unpublished data). The siRNA thus specifically interferes with endogenous TCL expression.
Figure 4.
Suppression of endogenous TCL perturbs the distribution of receptor-dependent internalized Tf. (A) TCL siRNA interferes with ectopic TCL protein expression. Expression of wild-type GFP-TCL was analyzed by Western blot in HeLa cells cotransfected with the empty pSuper or the vector expressing TCL or control siRNA as indicated. GAPDH was used to control of protein levels. (B) TCL siRNA causes down-regulation of TCL gene expression. HeLa cells were transfected with pSuper or the vector expressing TCL siRNA. Twenty-four hours later, cells were sorted and total RNA extracted and used for quantification by RT-PCR. The levels of TCL and TC10 mRNAs in TCL siRNA-expressing cells were expressed as percentage of the mRNA levels in cells transfected with the empty pSuper vector, which were arbitrary set to 100%. (C) TCL siRNA affects the localization of internalized transferrin. HeLa cells were cotransfected with GFP and siRNA expression vectors. Seventy-two hours later, they were incubated with rhodamine-labeled Tf for 45 min at 37°C. After fixation, fluorescent Tf was localized in cells expressing no siRNA (pSuper, a), TCL siRNA (b), or the control siRNA (c). In each panel, insets show the GFP fluorescence to identify transfected cells. The images shown are representative of >200 transfected cells. The stars indicate the same transfected cells in panels and insets. Bar, 10 μm. (D) TCL siRNA prevents Tf from reaching the early endosomal and recycling compartments. Immunofluorescence was performed on HeLa cells expressing TCL siRNA and incubated for 45 min at 37°C with rhodamine-labeled Tf. Fixed cells were stained with anti-AP2, anti-EEA1, anti-Rab11, and anti-Rab5 antibodies. Deconvolved superimposed images show no significant colocalization between internalized Tf (red in a–d) and EEA1 (green in a) or Rab11 (green in b), whereas Tf (d) is predominantly in Rab5-labeled structures (e) as revealed by the yellow color in merged images from d and e (f). The arrowheads indicate the same vesicles in d, e, and f. Bar, 10 μm. (E) Transferrin release. HeLa cells transfected with pEGFP and either pSuper expressing TCL siRNA (TCLsiRNA) or the empty pSuper vector (pSuper) were incubated with Tf coupled to Alexa 647 at 37°C. After 60 min, the mean Alexa647-Tf was measured. The inset shows the fluorescence in TCL siRNA-transfected cells and in nontransfected cells (NT). Labeled Tf was then chased with unlabeled Tf. The mean Alexa 647-Tf fluorescence remaining in the cells (percentage of remaining Tf) was measured after 0, 2.5, 5, 15, 30, and 90 min of chase in pSuper (circles) and TCL siRNA (closed diamonds)-transfected cells and in nontransfected cells (open diamonds); 100% corresponds to the amount of fluorescence measured after 60 min of internalization at 37°C and before the chase. The plots represent means ± SEM of three independent experiments of Tf release that counted at least 103 cells per time point.
We then performed steady-state Tf internalization for 45 min at 37°C in control and siRNA-treated cells. In nontransfected cells and in pSuper or in control siRNA transfected cells (Figure 4C, stars, a and c), Tf accumulated normally in the juxtanuclear recycling endosomes, with little punctuate staining throughout the cells. The latter corresponds to endocytic vesicles, early/sorting, and peripheral recycling endosomes. In contrast, cells expressing TCL siRNA presented a scattered distribution of Tf (Figure 4C, stars, b). This effect was due to the suppression of endogenous TCL, because coexpression of TCL siRNA and GFP-TCL bearing three silent mutations in the siRNA target sequence resulted in normal distribution of Tf (our unpublished data). Tf-containing vesicles did not colocalize with AP2, EEA1, or Rab11 (Figure 4D, a–c). Interestingly, in TCL siRNA-expressing cells, internalized Tf is predominantly in Rab5-positive vesicles (Figure 4D, d–f). This suggests that when TCL is absent, internalized Tf is sequestered in AP2-negative decoated endocytic vesicles without further transport to the EEA1-positive early/sorting and Rab11-positive recycling endosomes.
To analyze the effect of TCL suppression on transferrin recycling, we used FACS. HeLa cells were cotransfected with pEGFP and with either pSuper alone or pSuper expressing TCLsiRNA. Cells were depleted of endogenous Tf by serum starvation and then put in suspension and incubated with Alexa 647-conjugated Tf. After 60 min, Tf uptake was comparable in the TCL siRNA-expressing cells and in nontransfected (NT) cells (Figure 4E, inset). Labeled Tf was then removed and chased by unlabeled Tf for 90 min. Interestingly, during the first 20 min of chase, Tf release was significantly slower in TCLsiRNA-expressing cells compared with the nontransfected and to the control pSupertransfected cells (Figure E, closed diamonds, open diamonds, and circles, respectively). After 10 min, about one-half of the internalized Tf had recycled out of the nontransfected and the pSuper-transfected cells, whereas at that time >70% was still in TCLsiRNA-expressing cells. This did not reflect partial trapping of Tf in TCLsiRNA-expressing cells as after the 90 min of chase the amount of labeled Tf remaining was comparable in all cases. Thus, TCL is a new Rho GTPase essential for the normal functioning of the early endocytic pathway. Suppression of the endogenous protein leads to accumulation of receptor-bound internalized Tf in Rab5-positive decoated endocytic vesicles. TCL knock down also modifies the dynamics of Tf release.
Expression of Dominant Active TCL Favors the Recycling of Tf Directly from Early/Sorting Endosomes
Rho GTPases cycle between an inactive GDP-bound and an active GTP-bound form. We thus examined the effect of expressing a GTP-bound dominant active mutant (Q79L) of TCL on receptor-mediated Tf endocytosis. In most cells expressing GFP-TCLwt, Tf distribution seemed unaltered relative to control cells (Figure 5A, a and b). However, in cells expressing GFP-TCLda, Rhod-Tf presented a scattered distribution and did not accumulate in the juxtanuclear recycling endosomes (Figure 5A, c). Its distribution was normal in cells expressing dominant active Q75L TC10 mutant (TC10da; Figure 6A, a) or G12V Cdc42 mutant (Cdc42da; our unpublished data). Thus, TCLda specifically perturbs the distribution of endocytosed Tf.
Figure 5.
Dominant positive TCL accumulates internalized transferrin in early/sorting endosomes. (A) Dominant positive TCL perturbs the localization of internalized transferrin. HeLa cells expressing GFP-tagged TCLwt and TCLda were incubated with rhodamine-labeled Tf at 37°C. After fixation, fluorescent Tf was localized in untransfected cells (a) or in cells expressing GFP-TCLwt (b) and GFP-TCLda (c). In b and c, insets show the GFP fluorescence in the transfected cells. Bar, 10 μm. (B) Internalized Tf accumulates in early/sorting endosomes in TCLda-expressing cells. HeLa cells expressing GFP-TCLwt or GFP-TCLda were incubated for 20 min at 19°C in medium containing 5 μg/ml Tf coupled to Alexa 647. Labeled Tf was then chased for 20 min at 37°C. After fixation and permeabilization, cells were stained for Rab11 (a and d) or EEA1 (g). GFP-TCLwt, GFP-TCLda (a, d, and g, insets) and Alexa 647-Tf (b, e, and h) were visualized directly by confocal microscopy. The bottom row of panels shows merged images where the yellow color indicated Tf colocalization with Rab11 or EEA1. Bars, 10 μm. (C) Transferrin release. HeLa cells expressing GFP or GFP-TCLda and nontransfected HeLa cells (NT) were incubated with Tf coupled to Alexa 647 at 37°C. After 60 min, the mean Alexa 647-Tf was measured. The inset shows the ratio of the fluorescence in GFP or GFP-TCLda–expressing cells and nontransfected cells (GFP/NT and TCLda/NT, respectively). Labeled Tf was then chased with unlabeled Tf and the mean Alexa 647-Tf fluorescence remaining in the cells (percentage of remaining Tf) was measured after 0, 5, 15, 30, 60, and 90 min of chase in GFP-TCLda–expressing cells (circles, plain line), in GFP-expressing cell (diamonds, dotted line), and in nontransfected cells (squares, dotted line). 100% corresponds to the amount of fluorescence measured after 60 min of internalization at 37°C and before the chase. The plots represent means ± SEM of two independent experiments of Tf release counting at least 103 cells per time point.
Figure 6.
TCL function in the early endocytic pathway requires activation of effectors that can also bind Cdc42 and TC10. (A) HeLa cells expressing GFP-TC10da (a) or TC10daCterTCL (b) were incubated with rhodamine-labeled Tf, which was then visualized microscopically. The insets show the GFP fluorescence in the transfected cells. Images shown are representative of >200 transfected cells. Bar, 10 μm. (B) HeLa cells expressing the indicated GFP-tagged chimeric proteins were incubated with rhodamine-labeled Tf for 45 min at 37°C. After fixation, internalized Tf was visualized by immunofluorescence. Bar graphs show the percentage of cells expressing the indicated GFP fusion proteins that have scattered transferrin distribution (percentage of scattered Tf). The points represent the average and SEM of four independent experiments involving >600 cells. The frequencies of cells with scattered Tf did not differ between RhoGda and RhoGdaCterTCL (stars, Mann-Whitney U test, P > 0.05 in all pairwise comparisons). All other differences were statistically significant (Mann–Whitney, P = 0.0286 for each comparison).
To examine the effect of TCLda expression on Tf distribution within the endocytic pathway, HeLa cells transfected with GFP, GFP-TCLwt, or GFP-TCLda were allowed to internalize Alexa 647-labeled Tf at 19°C for 20 min to specifically load EE/SE and prevent its further transport to REs. After uptake at 19°C, internalized Tf accumulated normally in EE/SE, labeled with EEA1, in GFP-TCLda– and GFP-TCLwt–expressing cells as in GFP-expressing control cells. As in the case of TCL suppression, TCLda expression did not affect Tf internalization (Figure 5C, inset). Labeled Tf was then removed from the medium and replaced by unlabeled Tf for 20 min at 37°C to allow Tf to be transported to the perinuclear recycling endosomes and to recycle. Cells were then fixed, stained with anti-EEA1 or anti-Rab11 antibodies, and observed by confocal microscopy. After transfer at 37°C, Tf accumulated normally in the juxtanuclear RE in GFP (our unpublished data) and GFP-TCLwt–expressing cells (Figure 5B, a–c). In contrast, when GFP-TCLda was expressed, Tf was scattered throughout the cytoplasm, where it barely colocalized with REs labeled with Rab11 (Figure 5B, d–f). We also noticed a discrete effect of TCLda overexpression on Rab11-positive membranes, which seemed slightly more scattered throughout the cells than in TCLwt-overexpressing cells (Figure 5B, a and d). Nevertheless, Tf did accumulate exclusively in EEA1-labeled early/sorting endosome (Figure 5B, g–i). These results confirm that TCL is involved in the regulation of the early endocytic pathway and show that cycling of the GTPase between its active and inactive form is essential for normal trafficking of clathrin-dependent endocytosed receptors.
The accumulation of internalized Tf in early/sorting endosomes observed upon dominant active TCL expression may reflect preferential recycling of Tf directly from EE/SE to the plasma membrane without prior distribution in the juxtanuclear positive recycling endosomes, or a block of Tf in EE/SE without further recycling. To examine whether Tf was still able to recycle, we used FACS to analyze nontransfected cells and cells expressing GFP alone or GFP-TCLda that were loaded with Alexa 647-labeled transferrin for 60 min at 37°C. Tf uptake was comparable in all cases (Figure 5C, inset). Labeled Tf was then removed from the medium and chased by unlabeled Tf for 90 min to allow labeled Tf release. Tf release was similar in cells expressing either GFP-TCLda or GFP and in nontransfected cells (Figure 5C). Together, these results suggest that dominant active TCL expression does not affect Tf uptake and release from the cells, but it favors the preferential recycling of Tf from early/sorting endosomes to the plasma membrane without prior transit through the pericentriolar recycling endosomes.
TCL Function Requires Effector Proteins That Can Also Be Activated by TC10 and Cdc42
TC10, Cdc42, and TCL are able to bind the same effector proteins, in particular CRIB-domain containing proteins (Joberty et al., 1999; Vignal et al., 2000). We compared the effect of the expression of dominant active TCL, TC10, and Cdc42 on the localization of receptor-dependent internalized Tf. We analyzed steady-state Tf internalization in HeLa cells expressing the different mutant proteins. After fixation, we scored the number of transfected cells where Tf accumulated normally in the juxtanuclear recycling endosomes (see control and TCLwt, Figure 5A, a and b) and of transfected cells in which Rhod-Tf presented a scattered distribution as defined above in the case of TCLda expression (see TCLda, Figure 5A, c). Overexpression of dominant active TC10 (TC10da) and Cdc42 (Cdc42da) resulted in normal Tf accumulation in the juxtanuclear recycling compartment (shown for TC10da, Figure 6A, a) in ∼70% of the cells (Figure 6B). It was reported previously that the hypervariable region determines to which membrane compartment Rho proteins are targeted (Michaelson et al., 2001). To target TC10da and Cdc42da to the same compartments as TCL, we replaced the C-terminal basic regions and CAAX boxes of TC10da and Cdc42da with that of TCL. Remarkably, the resulting chimeric proteins (TC10daCterTCL and Cdc42daCterTCL) affected the distribution of internalized Tf (shown for TC10da, Figure 6A, compare a and b) in ∼70% of the cells, similarly to TCLda (Figure 6B). RhoG, a more divergent GTPase, is unable to bind CRIB domain-containing proteins (Gauthier-Rouviere et al., 1998). Interestingly, targeting dominant active RhoG (RhoGda) to the TCL compartments as described above (RhoGdaCterTCL) had little effect on Tf distribution (Figure 6B). Thus, TCL activity in the early endocytic pathway relies on its insertion in specific membrane compartments, as defined by its C-terminal domain, and involves binding to effector proteins that can also be activated by TC10 and Cdc42.
DISCUSSION
Several studies have highlighted the role of Rho GTPases in the control of intracellular membrane traffic. We identified TCL, a new member of the Rho family that is similar to TC10 and Cdc42 (Vignal et al., 2000). Here, we report that TCL localizes to the plasma membrane and early/sorting endosomes. When endogenous TCL is suppressed by siRNA, receptor-dependent internalized transferrin remains in decoated vesicles and fails to enter the early/sorting and recycling endosomes. Moreover, Tf release is significantly slower upon TCL knockdown. On the other hand, TCLda causes internalized Tf to accumulate in early/sorting endosomes, from which Tf recycles directly instead of being transported to the recycling endosomes. The effect of TCLda requires its insertion in membranes of the early endocytic pathway and relies on downstream effector proteins that can also be activated by TC10 and Cdc42.
TCL Controls the Early Endocytic Pathway
Actin dynamics plays a key role in early steps of endocytosis in Saccharomyces cerevisiae (Fujimoto et al., 2000), and increasing evidence is in favor of the involvement of actin filaments in these processes in mammalian cells (Schafer, 2002). Rho GTPases are major regulators of actin cytoskeleton dynamics and thus participate in the control of endocytosis (Ridley, 2001b; Qualmann and Mellor, 2003). Both actin- and microtubule-dependent events govern to the distribution of endocytosed proteins throughout the cell. The early steps of endocytosis involve actin reorganization, and actin polymerization would participate in short-range endocytic vesicle movements, whereas long-range endosome mobility toward perinuclear recycling endosomes would be mediated by microtubules (van Deurs et al., 1995; Merrifield et al., 1999; Leung et al., 2000; Murray et al., 2000; Taunton et al., 2000; Schafer, 2002).
Our present results show that the Rho GTPase TCL is localized to the plasma membrane and the early/sorting endosomes. TCL is involved in the control of the early endocytic pathway. Its activity is not required for endocytic vesicle formation: suppression of TCL expression by siRNA did not prevent receptor-dependent Tf internalization. Nevertheless, TCL is required for subsequent steps of endocytosis. In cells expressing TCL siRNA, internalized transferrin was sequestered in Rab5-positive decoated endocytic vesicles and it was not further distributed into the early/sorting and the recycling endosomes. Moreover, suppression of TCL delayed transferrin recycling to the plasma membrane. These results suggest that TCL is involved in endocytic vesicle movements, and that it is essential for clathrin-dependent endocytosed receptors to be transported to the early/sorting endosomes (Figure 7).
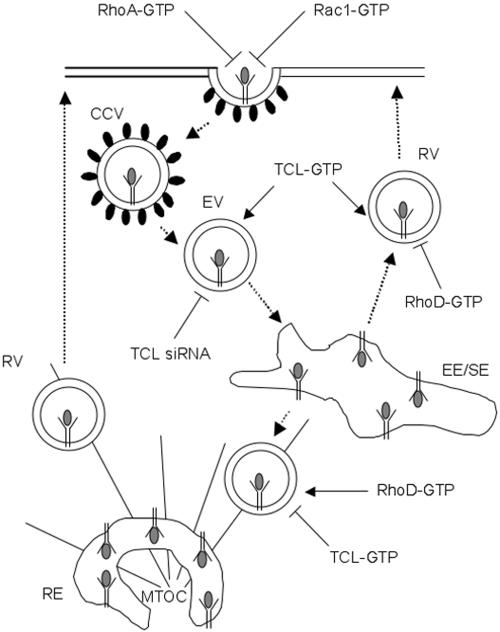
Figure 7.
Diagram summarizing the roles of TCL, RhoD, Rac1, and RhoA in clathrin-mediated endocytosis. TCL is required for receptor bound Tf in endocytic vesicles to reach early/sorting endosomes. Activated TCL favors Tf recycling directly from early/sorting endosomes and prevents it from accumulating in the pericentriolar recycling endosomes. Activated RhoA and Rac1 inhibit the internalization of transferring, whereas activated RhoD strongly reduces endosome motility (reviewed in Qualmann and Mellor, 2003). CCV, clathrin-coated vesicle; EV, endocytic vesicle; RV, recycling vesicle; MTOC, microtubule organizing center.
Expression of TCLda also allowed normal Tf internalization in HeLa cells, contrary to what was reported for Rac1 and RhoA (Lamaze et al., 1996). Nevertheless, TCLda prevented Tf transport to the juxtanuclear recycling endosomes. Instead, Tf remained scattered in the cells and accumulated in early/sorting endosomes. TCLda did not perturb subsequent Tf release, indicating that the early/sorting endosomes are still able to move within the cell cortex. After Tf internalization, the Tf receptor can recycle through two pathways: it can go back to the plasma membrane directly from the early/sorting endosomes, or it can initially be transported to the recycling endosomes (Sheff et al., 1999). Internalized Tf can recycle back to the plasma membrane even if the juxtanuclear recycling compartment is removed from the cells (Sheff et al., 2002). Consistent with the former, our results indicate that in intact cells, TCL activation would favor direct recycling of receptor bound Tf from the early/sorting endosomes. Recycling receptors clearly fail to enter a Rab11-positive compartment when active TCL is expressed. Membrane homeostasis between EE/SE and REs should be affected, because physiologically, the two membrane domains are actively exchanging material (Sonnichsen et al., 2000). This may explain why, although cargo molecules like Tf/TfR complexes are not reaching the Rab11-positive membranes, we still see an effect on RE cellular distribution in TCLda-expressing cells (Figure 5). The present results suggest that TCLda favors Tf direct recycling from EE/SE endosomes to the plasma membrane. The levels of active TCL may thereby influence the kinetics of individual receptor return to the plasma membrane after ligand internalization, by regulating the balance between the two recycling pathways. One is actin-dependent short range recycling from peripheral cytoplasmic compartments directly to the plasma membrane, and the other is microtubule-dependent long range recycling through the perinuclear recycling endosomes (Figure 7).
Specific Downstream Targets or Specific Localization of GTPase and Regulators?
TCL, TC10, and Cdc42 share extensive sequence similarities in their effector binding domains and they all bind to CRIB domain-containing proteins, such as PAK, WASP, BORG, and SPEC families, unlike the more divergent GTPase RhoG (Joberty et al., 1999; Pirone et al., 2000; Vignal et al., 2000; Chiang et al., 2002; our personal observations). GTP-bound TCL, Cdc42Hs, and TC10 can all activate N-WASP to catalyze Arp2/3-mediated actin polymerization in vitro (Abe et al., 2003). TCL is restricted to EE/SE and the plasma membrane. Consistent with its localization, we show that TCL is involved in early/sorting endosome trafficking, whereas it is not involved in GLUT4 secretion in response to insulin, a process that is controlled by TC10 (Chiang et al., 2002). Their specific roles do not seem to involve differential downstream effectors. When targeted to TCL compartments by C terminus swapping, TC10da and Cdc42da mimicked the effects of TCLda on internalized Tf distribution. Therefore, the three active GTPases can substitute for each other, most likely by interacting with the same downstream targets in vivo. In vitro, N-WASP that is activated through the binding of active TCL, Cdc42, or TC10 indistinguishably catalyzes Arp2/3-mediated actin polymerization (Abe et al., 2003). Vesicle movements can be driven by actin comet tail polymerization catalyzed by N-WASP (Merrifield et al., 1999; Taunton et al., 2000). N-WASP is involved in Cdc42-driven trafficking from Golgi to ER (Luna et al., 2002) and in TC10-dependent secretion vesicle transport (Jiang et al., 2002). N-WASP can also drive actin-dependent endosome movements (Taunton et al., 2000; Schafer, 2002). Our results suggest that TCL may have a similar function in driving actin-dependent endocytic vesicle movements toward early/sorting endosomes and to the plasma membrane. TCL suppression in HeLa cells affects transport of endocytic vesicles to EEA1-labeled EE/SE. This could reflect from impaired dynamics of the vesicles, due to the lack of actin polymerization at their surface in the absence of N-WASP recruitment by active TCL. Conversely, increasing the level of active TCL, which can recruit N-WASP at the vesicle surface (our personal observations), would maintain sustained actin polymerization at the surface of the vesicles. This would prevent their interaction with microtubules, thereby precluding their transport toward the juxtanuclear recycling compartment.
Exchange factors and/or GTPase-activating proteins most likely control the regulation of TCL activity, rather than GDP-dissociation inhibitors, which can extract GTPases from their target membranes. Fractionation analyses showed that the endogenous TCL is associated with membranes, and we could not detect the protein in the cytoplasm. Moreover, the potential GDI-RhoGTPase interaction site, as defined for Cdc42, contains identical substitutions in TCL and TC10, namely, Glu-Asp-Cys-Ser instead of Arg66-His103-His104-Arg186 in Cdc42. These substitutions rendered TC10 insensitive to the action of RhoGDI, unlike Cdc42, whose membrane association is affected by RhoGDI expression (Hoffman et al., 2000). An interesting potential activator of TCL is Intersectin, an important scaffolding protein of the early endocytic pathway. Intersectin has a Dbl homology domain able to activate Cdc42, and it regulates actin assembly via N-WASP and Arp2/3 (Hussain et al., 2001; Qualmann and Mellor, 2003). Given the role of Intersectin in the early endocytic pathway and the high degree of homology between TCL and Cdc42, it is tempting to speculate that Intersectin may activate TCL in vivo. In a cellular context, the effective binding of a particular target to a given Rho GTPase is difficult to estimate. Furthermore, it will be essential to know the subcellular distribution of the different partners, in particular GEFs and GAPs, to decipher the signaling cascades downstream of closely related GTPases such as TCL, TC10, and Cdc42.
Rho GTPases as Regulators of Vesicle Trafficking
Several Rho family proteins localize to vesicular structures and affect their intracellular transport. This was shown for RhoB and RhoD (Ellis and Mellor, 2000; Murphy et al., 2001; Gasman et al., 2003) and more recently for Cdc42 (Luna et al., 2002), TC10 (Chiang et al., 2001; Watson et al., 2001), RhoG (Vignal et al., 2001), and TCL (this report). The involvement of Rho GTPases in the regulation of intracellular membrane traffic now seems a general property of this family of proteins (Qualmann and Mellor, 2003). Recent studies suggest that their common function may be to regulate the transport of vesicles from one compartment to another, most likely through their action on cytoskeleton dynamics (Schafer, 2002). For instance, RhoD localizes to early/sorting endosomes and is involved in controlling the movement of early and late endosomes (Murphy et al., 2001; Gasman et al., 2003), whereas RhoG regulates microtubule-dependent lysosomal transport through its interaction with kinectin (Vignal et al., 2001). RhoB is associated with multivesicular bodies and is thought to be involved in regulating their trafficking toward the late endosomes (Ellis and Mellor, 2000). Although the precise molecular mechanisms linking Rho GTPases and vesicle dynamics are still unknown, actin assembly, driven by the Arp2/3 complex and the WASP family of proteins, seems to play an essential role in intracellular organelle movements (Taunton et al., 2000; Eitzen et al., 2002). This can be activated in vitro by the three Rho GTPases TCL, TC10, and Cdc42 (Abe et al., 2003). N-WASP and the Arp2/3 complex are involved both in Cdc42-regulated transport from the endoplasmic reticulum to the Golgi apparatus (Luna et al., 2002), and in TC10-dependent GLUT4 receptor transport stimulated by insulin (Jiang et al., 2002). Endocytic vesicle movements were also shown to involve actin filament polymerization at their surface through an N-WASP–dependent pathway (Merrifield et al., 1999; Taunton et al., 2000; Benesch et al., 2002), a mechanism that may also be regulated by TCL. Several Rho GTPases are involved in the regulation of different steps during clathrin-mediated endocytosis: RhoA and Rac1 participate in internalization (Lamaze et al., 1996), whereas RhoD (Murphy et al., 2001; Gasman et al., 2003) and TCL seem to control the movement of endocytic vesicles (summarized in Figure 7). The identification of their in vivo regulators and targets should shed light on their role in coordinating the complexity of intracellular membrane fluxes and membrane receptor trafficking, in conjunction with other small GTPases from the Rab and the Arf families (Takai et al., 2001).
Acknowledgments
We are grateful to Bruno Goud, Urszula Hibner, Mary McCauffray, Bo van Deurs, and Michel Vidal for the gift of antibodies and plasmids. We thank Robert Hipskin, Cécile Gauthier-Rouvičreand, and Pierre Roux for critical reading of the manuscript and fruitful discussion. We also want to thank François Rousset for statistic analyses, José Van der Heyden, Christophe Dupeyret, and Marion Blein for technical assistance and Pierre Travo, head of the Integrated Imaging Facility at the Centre de Recherches en Biochimie Macromoléculaire for technical assistance, constant interest, and support. This work was supported by The French “Ligue contre le Cancer” (“Equipe Labelisée”).
Article published online ahead of print. Mol. Biol. Cell 10.1091/mbc.E03–04–0254. Article and publication date are available at www.molbiolcell.org/cgi/doi/10.1091/mbc.E03-04-0254.
Abbreviations used: CRIB, Cdc42/Rac interaction binding; da, dominant active; dn, dominant negative; EE/SE, early/sorting endosomes; EEA1, early endosome antigen-1; EGFR, epidermal growth factor receptor; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; GFP, green fluorescent protein; RE, recycling endosome; Tf, transferrin; TfR, transferrin receptor; wt, wild-type.
References
- Abe, T., Kato, M., Miki, H., Takenawa, T., and Endo, T. (2003). Small GTPase Tc10 and its homologue RhoT induce N-WASP-mediated long process formation and neurite outgrowth. J. Cell Sci. 116, 155–168. [DOI] [PubMed] [Google Scholar]
- Benesch, S., Lommel, S., Steffen, A., Stradal, T.E., Scaplehorn, N., Way, M., Wehland, J., and Rottner, K. (2002). Phosphatidylinositol 4, 5-biphosphate (PIP2)-induced vesicle movement depends on N-WASP and involves Nck, WIP, and Grb2. J. Biol. Chem. 277, 37771–37776. [DOI] [PubMed] [Google Scholar]
- Bottger, G., Nagelkerken, B., and van der Sluijs, P. (1996). Rab4 and Rab7 define distinct nonoverlapping endosomal compartments. J. Biol. Chem. 271, 29191–29197. [DOI] [PubMed] [Google Scholar]
- Brummelkamp, T.R., Bernards, R., and Agami, R. (2002). A system for stable expression of short interfering RNAs in mammalian cells. Science 296, 550–553. [DOI] [PubMed] [Google Scholar]
- Chiang, S.H., Baumann, C.A., Kanzaki, M., Thurmond, D.C., Watson, R.T., Neudauer, C.L., Macara, I.G., Pessin, J.E., and Saltiel, A.R. (2001). Insulin-stimulated GLUT4 translocation requires the CAP-dependent activation of TC10. Nature 410, 944–948. [DOI] [PubMed] [Google Scholar]
- Chiang, S.H., Hou, J.C., Hwang, J., Pessin, J.E., and Saltiel, A.R. (2002). Cloning and functional characterization of related TC10 isoforms, a subfamily of Rho proteins involved in insulin-stimulated glucose transport. J. Biol. Chem. 277, 13067–13073. [DOI] [PubMed] [Google Scholar]
- Eitzen, G., Wang, L., Thorngren, N., and Wickner, W. (2002). Remodeling of organelle-bound actin is required for yeast vacuole fusion. J. Cell Biol. 158, 669–679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis, S., and Mellor, H. (2000). Regulation of endocytic traffic by Rho family GTPases. Trends Cell Biol. 10, 85–88. [DOI] [PubMed] [Google Scholar]
- Etienne-Manneville, S., and Hall, A. (2002). Rho GTPases in cell biology. Nature 420, 629–635. [DOI] [PubMed] [Google Scholar]
- Fiorentini, C., Falzano, L., Fabbri, A., Stringaro, A., Logozzi, M., Travaglione, S., Contamin, S., Arancia, G., Malorni, W., and Fais, S. (2001). Activation of rho GTPases by cytotoxic necrotizing factor 1 induces macropinocytosis and scavenging activity in epithelial cells. Mol. Biol. Cell 12, 2061–2073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujimoto, L.M., Roth, R., Heuser, J.E., and Schmid, S.L. (2000). Actin assembly plays a variable, but not obligatory role in receptor-mediated endocytosis in mammalian cells. Traffic 1, 161–171. [DOI] [PubMed] [Google Scholar]
- Gampel, A., Parker, P.J., and Mellor, H. (1999). Regulation of epidermal growth factor receptor traffic by the small GTPase rhoB. Curr. Biol. 9, 955–958. [DOI] [PubMed] [Google Scholar]
- Gasman, S., Kalaidzidis, Y., and Zerial, M. (2003). RhoD regulates endosome dynamics through Diaphanous-related Formin and Src tyrosine kinase. Nat. Cell Biol. 5, 195–204. [DOI] [PubMed] [Google Scholar]
- Gauthier-Rouviere, C., Vignal, E., Meriane, M., Roux, P., Montcourier, P., and Fort, P. (1998). RhoG GTPase controls a pathway that independently activates Rac1 and Cdc42Hs. Mol. Biol. Cell 9, 1379–1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman, G.R., Nassar, N., and Cerione, R.A. (2000). Structure of the Rho family GTP-binding protein Cdc42 in complex with the multifunctional regulator RhoGDI. Cell 100, 345–356. [DOI] [PubMed] [Google Scholar]
- Hussain, N.K., et al. (2001). Endocytic protein intersectin-l regulates actin assembly via Cdc42 and N-WASP. Nat. Cell Biol 3, 927–932. [DOI] [PubMed] [Google Scholar]
- Jiang, Z.Y., Chawla, A., Bose, A., Way, M., and Czech, M.P. (2002). A phosphatidylinositol 3-kinase-independent insulin signaling pathway to N-WASP/Arp2/3/F-actin required for GLUT4 glucose transporter recycling. J. Biol. Chem. 277, 509–515. [DOI] [PubMed] [Google Scholar]
- Joberty, G., Perlungher, R.R., and Macara, I.G. (1999). The Borgs, a new family of Cdc42 and TC10 GTPase-interacting proteins. Mol. Cell. Biol. 19, 6585–6597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanzaki, M., Watson, R.T., Khan, A.H., and Pessin, J.E. (2001). Insulin stimulates actin comet tails on intracellular GLUT4-containing compartments in differentiated 3T3L1 adipocytes. J. Biol. Chem. 276, 49331–49336. [DOI] [PubMed] [Google Scholar]
- Lamaze, C., Chuang, T.H., Terlecky, L.J., Bokoch, G.M., and Schmid, S.L. (1996). Regulation of receptor-mediated endocytosis by Rho and Rac. Nature 382, 177–179. [DOI] [PubMed] [Google Scholar]
- Leung, S.M., Ruiz, W.G., and Apodaca, G. (2000). Sorting of membrane and fluid at the apical pole of polarized Madin-Darby canine kidney cells. Mol. Biol. Cell 11, 2131–2150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luna, A., Matas, O.B., Martinez-Menarguez, J.A., Mato, E., Duran, J.M., Ballesta, J., Way, M., and Egea, G. (2002). Regulation of protein transport from the Golgi complex to the endoplasmic reticulum by CDC42 and N-WASP. Mol. Biol. Cell 13, 866–879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mary, S., Charrasse, S., Meriane, M., Comunale, F., Travo, P., Blangy, A., and Gauthier-Rouviere, C. (2002). Biogenesis of N-cadherin-dependent cell-cell contacts in living fibroblasts is a microtubule-dependent kinesin-driven mechanism. Mol. Biol. Cell 13, 285–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meresse, S., Gorvel, J.P., and Chavrier, P. (1995). The rab7 GTPase resides on a vesicular compartment connected to lysosomes. J. Cell Sci. 108, 3349–3358. [DOI] [PubMed] [Google Scholar]
- Merrifield, C.J., Moss, S.E., Ballestrem, C., Imhof, B.A., Giese, G., Wunderlich, I., and Almers, W. (1999). Endocytic vesicles move at the tips of actin tails in cultured mast cells. Nat. Cell Biol. 1, 72–74. [DOI] [PubMed] [Google Scholar]
- Michaelson, D., Silletti, J., Murphy, G., D'Eustachio, P., Rush, M., and Philips, M.R. (2001). Differential localization of Rho GTPases in live cells: regulation by hypervariable regions and RhoGDI binding. J. Cell Biol. 152, 111–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy, C., Saffrich, R., Olivo-Marin, J.C., Giner, A., Ansorge, W., Fotsis, T., and Zerial, M. (2001). Dual function of rhoD in vesicular movement and cell motility. Eur. J. Cell Biol. 80, 391–398. [DOI] [PubMed] [Google Scholar]
- Murray, J.W., Bananis, E., and Wolkoff, A.W. (2000). Reconstitution of ATP-dependent movement of endocytic vesicles along microtubules in vitro: an oscillatory bidirectional process. Mol. Biol. Cell 11, 419–433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nobes, C., and Marsh, M. (2000). Dendritic cells: new roles for Cdc42 and Rac in antigen uptake? Curr. Biol. 10, R739–R741. [DOI] [PubMed] [Google Scholar]
- Perez, F., Pernet-Gallay, K., Nizak, C., Goodson, H.V., Kreis, T.E., and Goud, B. (2002). CLIPR-59, a new trans-Golgi/TGN cytoplasmic linker protein belonging to the CLIP-170 family. J. Cell Biol. 156, 631–642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pirone, D.M., Fukuhara, S., Gutkind, J.S., and Burbelo, P.D. (2000). SPECs, small binding proteins for Cdc42. J. Biol. Chem. 275, 22650–22656. [DOI] [PubMed] [Google Scholar]
- Qualmann, B., and Mellor, H. (2003). Regulation of endocytic traffic by Rho GTPases. Biochem. J. 271 (Pt 2), 233–241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridley, A.J. (2001a). Rho proteins, PI 3-kinases, and monocyte/macrophage motility. FEBS Let.t 498, 168–171. [DOI] [PubMed] [Google Scholar]
- Ridley, A.J. (2001b). Rho proteins: linking signaling with membrane trafficking. Traffic 2, 303–310. [DOI] [PubMed] [Google Scholar]
- Rojas, R., Ruiz, W.G., Leung, S.M., Jou, T.S., and Apodaca, G. (2001). Cdc42-dependent modulation of tight junctions and membrane protein traffic in polarized Madin-Darby canine kidney cells. Mol. Biol. Cell 12, 2257–2274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafer, D.A. (2002). Coupling actin dynamics and membrane dynamics during endocytosis. Curr. Opin. Cell Biol. 14, 76–81. [DOI] [PubMed] [Google Scholar]
- Sheff, D.R., Daro, E.A., Hull, M., and Mellman, I. (1999). The receptor recycling pathway contains two distinct populations of early endosomes with different sorting functions. J. Cell Biol. 145, 123–139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheff, D., Pelletier, L., O'Connell, C.B., Warren, G., and Mellman, I. (2002). Transferrin receptor recycling in the absence of perinuclear recycling endosomes. J. Cell Biol. 156, 797–804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonnichsen, B., De Renzis, S., Nielsen, E., Rietdorf, J., and Zerial, M. (2000). Distinct membrane domains on endosomes in the recycling pathway visualized by multicolor imaging of Rab4, Rab5, and Rab11. J. Cell Biol. 149, 901–914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takai, Y., Sasaki, T., and Matozaki, T. (2001). Small GTP-binding proteins. Physiol. Rev. 81, 153–208. [DOI] [PubMed] [Google Scholar]
- Taunton, J., Rowning, B.A., Coughlin, M.L., Wu, M., Moon, R.T., Mitchison, T.J., and Larabell, C.A. (2000). Actin-dependent propulsion of endosomes and lysosomes by recruitment of N-WASP. J. Cell Biol. 148, 519–530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uze, G., Lutfalla, G., and Gresser, I. (1990). Genetic transfer of a functional human interferon alpha receptor into mouse cells: cloning and expression of its cDNA. Cell 60, 225–234. [DOI] [PubMed] [Google Scholar]
- van Deurs, B., Holm, P.K., Kayser, L., and Sandvig, K. (1995). Delivery to lysosomes in the human carcinoma cell line HEp-2 involves an actin filament-facilitated fusion between mature endosomes and preexisting lysosomes. Eur J. Cell Biol. 66, 309–323. [PubMed] [Google Scholar]
- Vignal, E., Blangy, A., and Fort, P. (2003). Signaling networks of Rho GTPases. In: Rho GTPases, ed. M. Symons, Austin, TX: R.G. Landes Co. 323, 253–262. [Google Scholar]
- Vignal, E., Blangy, A., Martin, M., Gauthier-Rouviere, C., and Fort, P. (2001). Kinectin is a key effector of RhoG microtubule-dependent cellular activity. Mol. Cell. Biol. 21, 8022–8034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vignal, E., De Toledo, M., Comunale, F., Ladopoulou, A., Gauthier-Rouviere, C., Blangy, A., and Fort, P. (2000). Characterization of TCL, a new GTPase of the rho family related to TC10 andCcdc42. J. Biol. Chem. 275, 36457–36464. [DOI] [PubMed] [Google Scholar]
- Watson, R.T., Shigematsu, S., Chiang, S.H., Mora, S., Kanzaki, M., Macara, I.G., Saltiel, A.R., and Pessin, J.E. (2001). Lipid raft microdomain compartmentalization of TC10 is required for insulin signaling and GLUT4 translocation. J. Cell Biol. 154, 829–840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilcke, M., Johannes, L., Galli, T., Mayau, V., Goud, B., and Salamero, J. (2000). Rab11 regulates the compartmentalization of early endosomes required for efficient transport from early endosomes to the trans-Golgi network. J. Cell Biol. 151, 1207–1220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zerial, M., and McBride, H. (2001). Rab proteins as membrane organizers. Nat. Rev. Mol. Cell. Biol. 2, 107–117. [DOI] [PubMed] [Google Scholar]