Abstract
Aim
This review posits that fatty acid amide hydrolase (FAAH) inhibition has therapeutic potential against neuropathological states including traumatic brain injury, Alzheimer's, Huntington's, and Parkinson's diseases, and stroke.
Main Methods
This proposition is supported by data from numerous in vitro and in vivo experiments establishing metabolic and pharmacological contexts for the neuroprotective role of the endogenous cannabinoid (“endocannbinoid”) system and selective FAAH inhibitors.
Key Findings
The systems biology of endocannabinoid signaling involves two main cannabinoid receptors, the principal endocannabinoid lipid mediators N-arachidonoylethanolamine (“anandamide”) (AEA) and 2-arachidonoyl glycerol (2-AG), related metabolites, and the proteins involved in endocannabinoid biosynthesis, biotransformation, and transit. The endocannabinoid system is capable of activating distinct signaling pathways on-demand in response to pathogenic events or stimuli, thereby enhancing cell survival and promoting tissue repair. Accumulating data suggest that endocannabinoid system modulation at discrete targets is a promising pharmacotherapeutic strategy for treating various medical conditions. In particular, neuronal injury activates cannabinoid signaling in the central nervous system as an intrinsic neuroprotective response. Indirect potentiation of this salutary response through pharmacological inhibition of FAAH, an endocannabinoid-deactivating enzyme, and consequent activation of signaling pathways downstream from cannabinoid receptors, have been shown to promote neuronal maintenance and function.
Significance
This therapeutic modality has the potential to offer site- and event-specific therapeutic relief in those tissues where endocannabinoids are being produced as part of a physiological protective mechanism. In contrast, direct application of cannabinoid receptor agonists to the central nervous system may activate CB receptors indiscriminately and invite unwanted psychotrophic effects.
Keywords: Alzheimer's disease, anandamide, brain trauma, cannabinoid, CB1 receptor, endocannabinoid, FAAH, Huntington's disease, neurodegeneration, neuroinflammation, Parkinson's disease, stroke
Introduction
Demonstration that cannabinoid (CB) constituents of the Cannabis plant, including Δ9-tetrahydrocannabinol (THC), exert their effects by engaging and activating discrete cellular CB receptors prompted the search in animals for naturally produced CB-receptor agonist ligands. This quest led to the discovery of a ubiquitous mammalian signaling system in which endogenous cannabinoids (endocannabinoids) synthesized by the body act as signaling lipids play varied homeostatic and regulatory roles (Mackie 2006; Pacher et al. 2006; Pertwee 2008). Experimental and clinical data have unequivocally demonstrated that one of the most important functions of the endocannabinoid signaling system is tissue protection against pathological insult or injury-- a function that has opened several attractive therapeutic modalities for pharmacological endocannabinoid-system modulation (Chang et al. 2006; Mackie 2008; Pacher and Haskó 2008; Vemuri et al. 2008; Janero and Makriyannis, 2009a,b). In particular, great advances have been made toward targeted pharmacotherapeutic manipulation of endocannabinoid signaling for neuroprotection. One well-studied endocannabinoid in particular, N-arachidonoylethanolamine (or “anandamide”) (AEA), has been linked to the neuroprotective property of CB-receptor transmission (Table 1) (see reviews: Bahr et al. 2006; Pavlopoulos et al. 2006; Janero and Makriyannis 2007; Micale et al. 2007). Distributed throughout the brain as an integral membrane protein, the enzyme fatty acid amide hydrolase (FAAH) is primarily responsible for inactivating AEA and quenching AEA-induced biological responses (Basavarajappa 2007; Vandevoorde and Lambert 2007; Di Marzo 2008; Ahn et al. 2008; Fezza et al. 2008). Whereas THC and other direct CB-receptor agonists can negatively influence psychomotor, cognitive, and appetitive behaviors (Freund 2003; Iversen 2003; Di Marzo and Matias 2005), selective FAAH inhibitors appear to offer site- and event-specific therapeutic relief in those tissues where endocannabinoids are being produced as part of a physiological protective mechanism (Vemuri et al. 2008; Janero and Makriyannis, 2009a). By virtue of this pharmacological mode of action, FAAH inhibitors are advantageously poised to exploit the neuroprotective nature of endocannabinoid signaling without risk of eliciting adverse psychotropic (or other) effects associated with chronic application of cannabinoid receptor agonists.
Table 1.
Pathogenic events that increase anandamide levels.
| Pathogenic insult | Alteration to the endocannabinoid system |
|---|---|
| traumatic brain injury | increases in anandamide |
| NMDA-induced excitotoxicity | increases in anandamide |
| kainic acid-induced seizures | increases in anandamide |
| 6-Hydroxydopamine toxicity | increases in anandamide |
| model of multiple sclerosis | increases in anandamide |
The foregoing implies that endocannabinoid signaling may be enhanced indirectly to therapeutic levels through FAAH inhibition, making FAAH an attractive pharmacotherapeutic target and selective FAAH inhibitors attractive drug candidates for various neurological and neurodegenerative/neuroinflammatory disorders (Table 2). This proposition will be examined herein from the perspectives of endocannabinoid metabolism and endocannabinoid-system neuropharmacology.
Table 2.
Medicinal applications for controlled FAAH inhibitors.
| Potential indications | Example FAAH inhibitors |
|---|---|
| exposure to excitotoxic neurotoxins | AM374, URB597 |
| stroke/ischemia | URB597 |
| epileptic seizures | AM374 |
| multiple sclerosis | AM374 |
| spinal cord injury | |
| traumatic brain injury | |
| addiction |
I. Endocannabinoid Metabolic Pathways
A growing number of endocannabinoid amide and ester long-chain fatty-acid derivatives are either known agonists or candidate ligands for one or both principal CB receptor subtypes, CB1 and CB2 (Mackie 2008; Pei et al. 2008; Pertwee 2008; Wood et al. 2008). It should also be noted that there have been other studies identifying potential cannabinoid receptors including a species named GPR55 (Begg et al. 2005; Pertwee 2007; Lauckner et al. 2008). The two signaling lipids first identified as endocannabinoids, AEA and 2-AG, remain nonetheless the most intensively studied and best characterized CB-receptor agonists (Bisogno 2008). AEA is a partial CB1-receptor activator with modest affinity [Ki = 61 nM (rat) and 240 nM (human)] and a relatively weak CB2-receptor ligand (Ki = 440-1930 nM for rodent and human CB2 receptors) with low overall efficacy. 2-AG is a full agonist at the CB1 and CB2 receptors, albeit with lower affinity (Ki = 472 and 1400 nM, respectively) and greater efficacy relative to AEA (Vemuri et al. 2008; Janero and Makriyannis, 2009a). Their distinctive biochemical and pharmacological properties notwithstanding, homeostatic regulation of AEA and 2-AG signaling depends critically upon controlled endocannabinoid generation and biotransformation/inactivation. The balanced actions of endocannabinoid synthesizing and metabolizing enzymes help localize and direct the intrinsically broad influence these lipid mediators may exert (Alexander and Kendall 2007). It is not surprising, therefore, that spatiotemporal changes in the expression/activity of endocannabinoid biosynthetic and inactivating enzymes are associated with both normal physiological processes as well as pathological states in humans (Wang et al. 2007; Ludányi et al. 2008). As distinct from most water-soluble neurotransmitters that are mobilized from membrane-delimited storage vesicles in bioactive form, AEA and 2-AG are synthesized on-demand in response to (patho)physiological stimuli (Figure 1), and their signaling functions are efficiently terminated by cellular uptake through a presently ill-defined transporter system and the rapid, enzyme-catalyzed hydrolytic inactivation (Figure 2) (Di Marzo 2008; Ahn et al. 2008). Reflecting the importance of AEA and 2-AG to intercellular communication within the central nervous system (CNS), investigations into the metabolism of these endocannabinoids have made extensive use of brain tissue and neuronal cells (Ahn et al. 2008; Lovinger 2008). For this reason, a synopsis of key metabolic pathways implicated in the regulation of endocannabinoid biosignaling primarily in the CNS is presented here. Recent reviews may be consulted for additional primary literature as well as details on the molecular and structural properties of various endocannabinoid-metabolizing enzymes, their physiological chemistry, and their potential as therapeutic targets (Bahr et al. 2006; Basavarajappa 2007; Vandevoorde and Lambert 2007; Saario and Laitinen 2007; Schneider et al. 2007; Ahn et al. 2008; Fezza et al. 2008; Vemuri et al. 2008; Janero and Makriyannis 2009a,b; Kano et al. 2009).
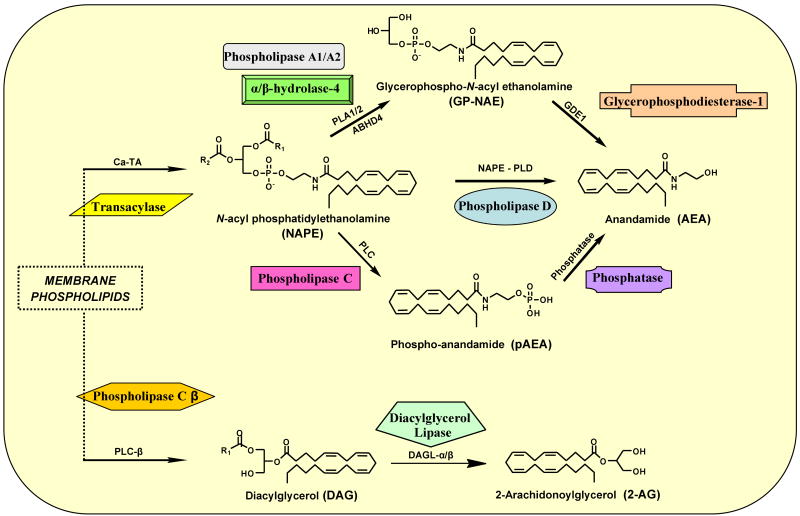
Figure 1.
Diagrammatic representation of the major metabolic pathways implicated in the biosynthesis of the principal endocannabinoids, anandamide and 2-arachidonoyl glycerol.
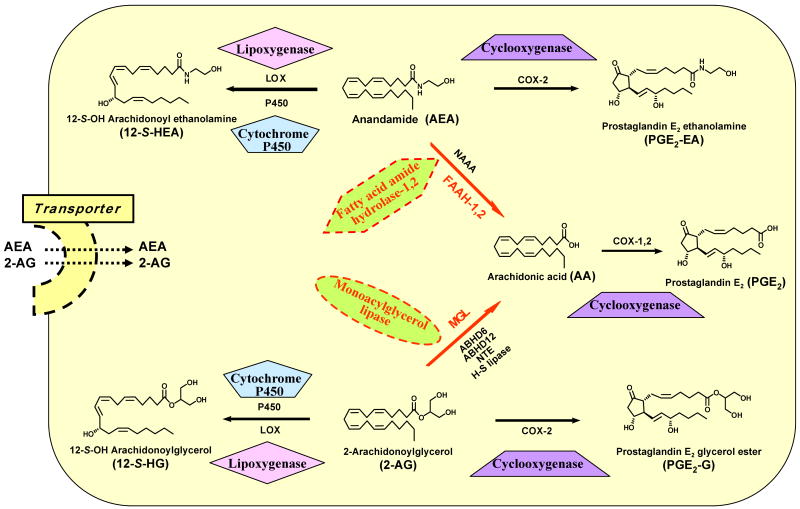
Figure 2.
Diagrammatic representation of the major hydrolytic inactivation and oxidative biotransformation pathways implicated in anandamide and 2-arachidonoyl glycerol catabolism.
A. Endocannabinoid Biosynthesis
Enzymatic synthesis of both AEA and 2-AG draws upon pools of membrane phospholipids such as phosphatidylethanolamine (PE), phosphatidylcholine (PC), and phosphatidylinositol 4,5-bisphosphate (PtdIns(4,5)P2) (Figure 1) (Ahn et al. 2008; Lovinger 2008) (Figure 1). The pathways by which N-acyl ethanolamines (NAEs) such as AEA are synthesized had been studied well before AEA was identified as an endocannabinoid. Early data suggested a two-step pathway involving the sequential action of a calcium-dependent transacylase (Ca-TA) that transfers a fatty-acyl chain from a membrane phospholipid molecule onto the primary amine of membrane PE to generate N-acyl phosphatidylethanolamine (NAPE) and a NAPE-selective phospholipase D (NAPE-PLD) that hydrolyzes NAPE to NAEs such as AEA (Natarajan et al. 1983, 1984; Cadas et al. 1997). Two NAPE-PLD-independent pathways have also been implicated in NAE/AEA formation. One involves the hydrolysis of NAPE (and, perhaps, lyso-NAPE) to a glycerophospho(GP)-NAE intermediate by phospholipase A1/A2 (PLA1/2) or-- perhaps more importantly in the CNS-- by the serine hydrolase α/β-hydrolase-4 (ABHD4). GP-NAE ester cleavage by glycerophosphodiesterase-1 (GDE1) then generates NAE/AEA (Simon and Cravatt 2006, 2008). Another candidate NAPE-PLD-independent pathway for AEA biosynthesis involves the conversion of NAPE to phospho-AEA (pAEA) by phospholipase C (PLC) followed by the phosphatase-mediated hydrolysis of pAEA (Liu et al. 2006, 2008). Formation of polyunsaturated NAEs such as AEA appears to rely more upon NAPE-PLD-independent routes, whereas synthesis of long-chain saturated or monounsaturated NAEs proceeds mainly through NAPE-PLD (Ahn et al. 2008). All of these candidate pathways for NAE/AEA formation, however, likely have NAPEs as key lipid precursors.
As is the case for AEA, multiple, incompletely characterized pathways for 2-AG biosynthesis by neurons have been proposed, each of which involves sequential enzyme-catalyzed transformations (Figure 1). The first and best-studied pathway is implicated in 2-AG formation elicited by intracellular calcium elevation or by Gαq/11-coupled receptor activation. Both of these signals stimulate membrane-bound phospholipase C β (PLC-β) to hydrolyze the minor membrane phospholipid PtdIns(4,5)P2, generating inositol 1,4,5-triphosphate (InsP3) and diacylglycerol (DAG). Two developmentally-regulated serine hydrolases, the sn-1-selective DAG lipases (DAGLs) DAGL-α and -β, then catalyze the final step of 2-AG formation from DAG (Figure 1) (Jung et al. 2005, 2007; Vandevoorde and Lambert 2007; Ahn et al. 2008). Although both DAGL-α and -β are highly enriched in brain, the latter is expressed primarily in early development, whereas DAGL-α expression is sustained throughout adulthood and shifts from a predominantly pre- to post-synaptic locus during neuronal maturation (Bisogno et al. 2003). Another presumptive pathway for 2-AG formation involves the synergistic actions of calcium elevation and receptor activation leading to DAG hydrolysis by DAGL-α/β as its final step (Hashimotodani et al. 2007, 2008).
Most enzymes implicated in AEA or 2-AG biosynthesis presently lack sufficiently selective genetic or pharmacological tools (e.g., potent small-molecule inhibitors) for experimental provocation to quantify the relative contribution of each endocannabinoid biosynthetic pathway discussed. Among the recognized endocannabinoid biosynthesizing enzymes, DAGL is the focus of much current research. The strategic positioning of DAGL in multiple 2-AG biosynthetic pathways suggests that selective DAGL inhibition might offer an attractive therapeutic modality for medical conditions to which heightened 2-AG signaling through the cannabinoid 1 (CB1) receptor may contribute, such as obesity, metabolic syndrome, and substance abuse disorders (Vemuri et al. 2008; Ortar et al. 2008; Hoover et al. 2008; Janero and Makriyannis 2009a,b).
B. Endocannabinoid Inactivation and Biotransformation
In marked contrast to the multiplicity and partial redundancy of putative pathways for AEA and 2-AG biosynthesis, each of these two principal endocannabinoids is inactivated in the nervous system primarily by a specific, relatively well-characterized hydrolase that generates the biologically important polyunsaturated fatty acid, arachidonic acid (AA) (Figure 2). Fatty acid amide hydrolase (FAAH) is the principal enzyme responsible for terminating AEA signaling and is widely expressed throughout the CNS as an integral membrane protein (Basavarajappa 2007; Vandevoorde and Lambert 2007; Di Marzo 2008; Ahn et al. 2008; Fezza et al. 2008). Another amidase-signature serine hydrolase with FAAH activity shares limited sequence identity with the original, so-called FAAH-1 enzyme and has been designated as FAAH-2 (Wei et al. 2006). FAAH-1 and -2 display several distinct biochemical, phylogenic, and tissue-distribution characteristics. In the CNS, FAAH-1 is the more abundant FAAH and it hydrolyzes NAEs (including AEA) at greater rates than FAAH-2, which is enriched in select peripheral tissues but is absent in lower mammals, including rodents (Wei et al. 2006). FAAH-1 and -2 also display quantitatively distinct inhibitor sensitivities (Wei et al. 2006). An NAE-hydrolyzing acid amidase (NAAA) identified in immune system cells and select peripheral tissues has been cloned (Tsuboi et al. 2007a,b). NAAA is not a serine hydrolase and is also quite different from FAAH in its intracellular (lysosomal) localization, acidic pH optimum, and lack of preference for AEA as substrate. The contribution of NAAA to AEA inactivation in vivo is unknown.
Genetic polymorphisms in FAAH that disturb endocannabinoid biosignaling are associated with increased risk for substance abuse (Flanagan et al. 2006), gastrointestinal dysfunction (Storr et al. 2008a), and overweight/obesity issues (Aberle et al. 2008) in certain human subpopulations. Because of the important roles of AEA in neuronal plasticity, memory, and protection against neurological insult, selective FAAH inhibitors are a prime translational focus as potential drugs for important medical problems including pain management, psychobehavioral states (anxiety, depression), inflammation, excitotoxic events (i.e., stroke, seizures), and age-related neurodegenerative disorders including Huntington's, Parkinson's, and Alzheimer's diseases (Bahr et al. 2006; Karanian et al. 2005b, 2007; Centonze et al. 2007; Scherma et al. 2008; Storr et al. 2008b; Naidu et al. 2009) (vide infra). Targeted FAAH inhibition (i.e., blocking AEA inactivation) is considered an attractive therapeutic approach to enhance indirectly cannabinergic signaling, for it would take place preferentially in regions where endocannabinoid synthesis and release are ongoing as a salutary, tissue-protective response. The site- and event-specific character of the pharmacological inhibition of endocannabinoid deactivating enzymes such as FAAH may offer increased selectivity with less risk of the undesirable side effects that have been observed with CB-receptor agonists capable of activating all accessible receptors indiscriminately (Vemuri et al. 2008; Janero and Makriyannis 2009).
Although FAAH hydrolyzes both AEA and 2-AG at similar rates in vitro (Goparaju et al. 1998), multiple lines of genetic, biochemical, and pharmacological evidence suggest that enzyme(s) other than FAAH regulate 2-AG inactivation in vivo. In particular, a soluble serine hydrolase, monoacylglycerol lipase (MGL), is considered the principal enzyme responsible for terminating 2-AG signaling in the CNS by catalyzing 2-AG's conversion to AA (Figure 2) (Vandevoorde and Lambert 2007; Saario and Laitinen 2007; Ahn et al. 2008; Zvonok et al. 2008a and 2008b). Four other hydrolases are capable of generating AA from 2-AG: α/β-hydrolase-6 (ABHD6), α/β-hydrolase-12 (ABHD4), neuropathy-target esterase (NTE), and hormone-sensitive lipase (H-S lipase) (Figure 2) (Belfrage et al. 1977; van Tienhoven et al. 2002; Blankman et al. 2007). Some 85% of total brain 2-AG hydrolysis has been assigned to MGL, the remaining ∼ 15% catalyzed mostly by ABHD6 and ABHD12 (Blankman et al. 2007). The distinct subcellular distributions of MGL, ABHD6, and ABHD12 suggest that they may modulate specific CNS 2-AG pools (Ahn et al. 2008).
Enzymatic conversion of AEA and 2-AG to AA constitutes the hydrolytic deactivation mode of endocannabinoid metabolism. AEA and 2-AG can also serve as indirect and direct sources of substrates for oxidative transformation to various lipid derivatives (Figure 2). The AA product of both FAAH and MGL catalysis is a second-messenger molecule whose positional oxygenation, epoxidation, or hydroxylation by cyclooxygenase (COX), lipoxygenase (LOX), epoxygenase, or hydroxylase enzymes can generate an array of fatty acid derivatives including prostaglandin, prostacyclin, thromboxane, and leukotriene eicosanoids and hydroxyeicosatetraenoic and epoxyeicosatrienoic acids (Khanapure et al. 2007). Many of these AA derivatives have important physiological functions: e.g., prostaglandin E2 (PGE2) is a potent modulator of vascular tone and mediates diverse immune and inflammatory responses (Khanapure et al. 2007). Both AEA and 2-AG can be oxygenated directly by COX to generate prostaglandin ethanolamides (“prostamides”) [e.g., prostaglandin E2 ethanolamine (PGE2-EA)] and glyceryl prostaglandins [e.g., prostaglandin E2 glycerol ester (PGE2-G)], respectively (Figure 2). COX-1 is much less effective than COX-2 in catalyzing oxidative endocannabinoid metabolism (Woodward et al. 2008). Likewise, AEA and 2-AG can serve as LOX and cytochrome P450 substrates, leading to an array of oxygenated compounds including hydroxyeicosatetraenoic acid ethanolamides [e.g., 12-S-OH arachidonoyl ethanolamine (12-S-HEA)], hydroxyeicosatetraenoic acid glycerol esters [e.g., 12-S-OH arachidonoylglycerol (12-S-HG)] (Figure 2), epoxyeicosatetraenoic acid ethanolamides, and epoxyeicosatetraenoic acid glycerol esters (Prusakiewicz et al. 2007; Chen et al. 2008). The production and physiological activity of most products of direct AEA and 2-AG oxidative metabolism in vivo largely remain to be established (Guindon and Hohmann 2008). In this regard, it has recently been observed that COX-2 converts 2-AG into PGE2-G, which induces hyperalgesia in the rat (Shu-Jung et al. 2008), and that fat ingestion elicits NAPE biosynthesis to suppress food intake (Gillum et al. 2008). The finding that AEA and 2-AG induce COX-2 expression and stimulate prostaglandin production in human gestational tissue (Mitchell et al. 2008) is suggestive of physiologically significant cross-talk between endocannabinoids and enzymes regulating their metabolism. This suggestion is reinforced by the ability of AEA to inhibit 2-AG biosynthesis and activity in mouse-brain striatum by interfering with DAGL-α catalysis through activation of a nonselective cation channel, transient receptor potential vanilloid 1 (TRPV1) (Maccarrone et al. 2008).
II. Neuroprotection Through Endocannabinoid Modulation
Responses to CB receptor activation include opening of potassium channels, inhibition of calcium currents, and stimulation of various protein kinases (Deadwyler et al. 1995; Gomez Del Pulgar et al. 2000; Galve-Roperh et al. 2002; Molina-Holgado et al. 2005; Karanian et al. 2005b, 2007). Some of the many such signaling elements activated by endocannabinoids play important roles in neuronal maintenance (Bahr et al. 2006; Galve-Roperh et al. 2008). CB receptor transmission elicits modulatory effects on calcium channels, resulting in reduced neurotransmitter (e.g., GABA, glutamate) release (Hajos et al. 2000; Kreitzer and Regehr 2001; Ohno-Shosaku et al. 2001; Wilson et al. 2001). One particular mitogen-activated protein kinase, extracellular signal-regulated kinase (ERK), is involved in cannabinergic signaling, as are focal adhesion kinase (FAK) and phosphatidylinositol 3′-kinase (PI3K). These signaling elements appear to play key roles in the neuroprotective nature of the endocannabinoid system, and the associated signaling pathways are disrupted by blocking CB receptor activation (Wallace et al. 2003; Khaspekov et al. 2004; Karanian et al. 2005a,b). Compromise of basal cross-talk among pathways involving ERK, FAK, and growth-factor receptors during endocannabinergic blockade elicits a corresponding increase in neuronal pathogenic susceptibility and a decrease in synaptic maintenance. Several reports further indicate that genetic ablation of CB receptors and their associated downstream signaling increases susceptibility to seizure induction, traumatic and ischemic brain injury, and neuronal inflammatory damage (Parmentier-Batteur et al. 2002; Marsicano et al. 2003; Jackson et al. 2005; Panikashvili et al. 2005). Conversely, promoting CB receptor responses through the action of exogenous or endogenous ligands can generate enhanced levels of signaling via ERK, FAK, and other pathways. This makes FAAH and other endocannabinoid deactivation mechanisms ideal targets for neuroprotective modulation of the cannabinergic system. Selective FAAH inhibitors and FAAH knockout studies have described the outcome of elevated endogenous signaling through CB1 receptors (Cravatt et al. 2001; Kathuria et al. 2003; Karanian et al. 2005b, 2007). Dual modulation of endocannabinoid action was achieved by combining a FAAH inhibitor with another blocker of endocannabinoid deactivation (anandamide transport inhibitor), resulting in pronounced increases in FAK and ERK responses, to levels similar to those triggered by CB1-receptor agonists. The effects of exogenously applied AEA are also potentiated by FAAH inhibition (Gifford et al. 1999; Arizzi et al. 2004). Together, these studies indicate that positive modulation of endocannabinoid responses can harness endogenous signaling networks that support neuroprotection.
A. Neuroprotective Effects of FAAH Inhibitors
In the CNS, endocannabinoids are produced by neurons on demand (Figure 1), and their efficient catalytic inactivation (Figure 2) helps ensure that they act locally near their site of synthesis. Endocannabinoids are synthesized and released in response to neuronal injury, and the cannabinergic action of the released CB-receptor ligands can be enhanced by attenuating FAAH activity. Since AEA is effectively inactivated by the endocannabinoid-hydrolyzing enzyme FAAH (vide supra), inhibiting FAAH would increase the availability of released AEA to activate local CB receptors to elevate the initial neuroprotective response to a therapeutic level. The significance of FAAH inhibition as a means of enhancing cannabinergic signaling has been demonstrated through both knockout strategies as well as pharmacological FAAH blockade by synthetic inhibitors (Cravatt et al. 2001; Karanian et al. 2005a; Moreira et al. 2008). Not surprisingly, potential therapeutic indications to which this strategy could be applied for therapeutic purposes are expanding and include management of multiple sclerosis (Benito et al. 2007), spinal cord injury (Garcia-Ovejero et al. 2009), ischemic events (Nucci et al. 2007), and other disorders, particularly those involving the CNS (Table 2).
Endocannabinoid production and signaling through CB receptors elicit neuronal-cell survival responses that could play a salutary role against diverse neuropathologies. Table 3 lists the growing number of neuroprotective effects associated with FAAH inhibition. For example, the FAAH inhibitor URB597 attenuated hippocampal neuronal hyperactivity (Coomber et al. 2008), and systemic administration of URB597 minimized the retinal damage observed in ischemic-reperfusion studies (Nucci et al. 2007). Another FAAH inhibitor, palmitylsulfonyl fluoride (AM374), enhanced the effects of AEA (Gifford et al. 1999; Arizzi et al. 2004) and promoted endogenous CB-related signaling in brain tissue (Karanian et al. 2005b). Pharmacological modulation of endocannabinoid deactivation to promote signaling resulted in excitotoxic protection in both hippocampal slice and animal models. The neuroprotection was demonstrated cellularly and functionally, i.e., by a reduction in neuronal loss in the hippocampus, enhanced synaptic integrity, reduced behavioral abnormalities, and improved memory performance (Karanian et al. 2005b). These neuroprotective effects were completely abrogated by a selective CB1-receptor antagonist, thus indicating that CB1-receptor transmission was responsible for the neuroprotection.
Table 3.
Effects of FAAH inhibitors
|
Another example of neuroprotective FAAH modulation is provided by data from the kainate rat model. As an excitotoxin, kainic acid (KA) induces seizures and damage to the hippocampus, a brain region involved in higher-order brain functions. Uncontrolled seizures produce excitotoxic brain damage reminiscent of that in many disease states. Recurring seizures can result from epilepsy, brain injury, and genetic conditions. Among the medical complications associated with substance abuse/drug addiction, excitotoxic brain injury can be caused by alcohol (Wilkins et al., 2006), diverse amphetamine derivatives (Itzhak and Ali, 2006), and cocaine (Kosten et al., 1994; Mets et al., 1998; Arai et al., 2003; Matsumoto et al., 2004; Witkins et al., 2008). The selective FAAH inhibitor AM374 was shown to enhance AEA levels and decrease seizure severity in KA-treated rats (Karanian et al. 2007). This study also documented a dose-dependent and accelerated rate of seizure recovery with AM374 treatment, which protected against excitotoxic cytoskeletal damage and synaptic decay. The controlled modulation of cannabinergic signals also protected against the cellular and functional deficits in the kainate rats. These salutary responses were prevented by the CB1-receptor antagonist AM251, indicating that the neuroprotective action of FAAH inhibition was mediated through endocannabinoid transmission. This FAAH-inhibitor study further implicated the activated MAPK/ERK pathway in the reduction of neuronal damage. Another selective FAAH inhibitor, URB597, has been found to attenuate the damaging effects of KA-induced neuronal activity (Coomber et al. 2008). Together, these studies show dose-dependent congruence in vivo among: 1) reduced seizure severity, 2) enhanced synaptic integrity, and 3) improved behavioral performance. The endocannabinoid enhancement attenuated seizure severity, perhaps by reducing intracellular calcium via cannabinergic actions on voltage-gated calcium channels through inhibition of adenylyl cyclase (Deadwyler et al., 1993; Shen and Thayer, 1996, 1998; Mu et al., 1999) or by eliciting nonspecific and synapse-specific depression of excitatory circuits (Shen et al., 1996; Kim and Thayer, 2000; Gerdeman and Lovinger, 2001; Singla et al., 2007). Clinical studies indicate that epileptic seizures in humans can result from perturbation of the endocannabinoid system, including the down-regulation of CB receptors located in hippocampal glutamatergic terminals (Ludanyi et al., 2008). In sum, manipulation of the endocannabinoid system, a key regulator of synaptic transmission in the brain, through FAAH inhibition is a potentially attractive approach for treating seizures of diverse etiology.
The neuroprotective action of FAAH inhibitors is reminiscent of the compensatory protective response in which AEA levels are elevated 2- to 13-fold after KA-induced seizures (Marsicano et al., 2003), ischemia (Amantea et al., 2007), concussive head trauma (Hansen et al., 2001), and neurotoxin exposure (Hansen et al., 2001; Maccarrone et al., 2003). In humans experiencing stroke-related excitotoxic insult, AEA release during the injury process has been observed as a compensatory protective response to potentiate intrinsic survival signaling (Schäbitz et al., 2002). Cannabinergic modulation of network excitability may also have the potential to offset the neuronal over-activation produced by drugs of abuse that can involve various transmitters including dopamine, serotonin, norepinephrine, GABA, and glutamate. Note that inhibitors of. The strategy of developing FAAH inhibitors as neuroprotective drugs gains attractiveness from findings that FAAH inhibitors are devoid of cataleptic effects (Beltramo et al., 2000; Arizzi et al., 2004; Karanian et al., 2007) and do not carry an abuse potential themselves (Justinova et al., 2008). These properties suggest that FAAH inhibitors could represent novel, safe drugs for treating brain damage from epileptic seizures, traumatic injury, and drugs of abuse.
Other medical indications where FAAH inhibition may be an effective pharmacotherapeutic strategy include multiple sclerosis and neurodegenerative/neuroinflammatory diseases such as Alzheimer's, Huntington's, and Parkinson's diseases (Benito et al. 2003; Maccarrone et al. 2003; Ramirez et al. 2005; Micale et al. 2007; Bisogno and Di Marzo 2008). Recent data presented in abstract indicate that the content of AEA and a prime AEA precursor, N-arachidonoyl phosphatidylethanolamine (Figure 1), were markedly reduced in the temporal and frontal cortices of Alzheimer's patients, whereas 2-AG and 2-AG-related related lipids were largely unchanged (Jung et al. 2008). Interestingly, AEA levels in different brain regions of the Alzheimer's patients correlated with region-specific cognitive test scores, implicating the importance of endocannabinoid signaling for cognition. In another clinical Alzheimer's study, AEA was shown to prevent toxicity of the human amyloid-β peptide (Milton 2002), which is believed responsible for the neurodegenerative changes in the Alzheimer's brain. In an animal model of multiple sclerosis, AM374 significantly reduced a hallmark symptom, spasticity, a therapeutic response similar to that elicited by treatment with potent cannabinergic agonists (Baker et al. 2001).
Further support for beneficial effects of FAAH inhibition in multiple sclerosis was recently demonstrated in a chronic encephalitis model with the observation that that FAAH knockout mice exhibited a more substantial remission compared to wild-type mice (Webb et al. 2008). The reduced endocannabinoid signaling associated with Huntington's disease could be reversed through the blockade of FAAH activity (see review: Micale et al. 2007). FAAH inhibition by methylarachidonoyl fluorophosphonate (MAFP) also restored normal glutamatergic activity in an animal model of Parkinson's disease (Maccarrone et al. 2003). These experimental and clinical data suggest that FAAH inhibition will gain importance as a potential therapeutic modality for age-related neurodegenerative diseases.
III. Conclusion
The growing understanding of the biosynthetic and inactivation pathways that help regulate endocannabinoid signaling and the successful biochemical description and functional annotation of several molecular constituents of the endocannabinoid metabolome have suggested new treatment approaches for several important disease states ill-satisfied by currently available drugs. Therapeutic exploitation of “on-demand,” tissue-protective endocannabinoid responses is particularly attractive with respect to many neuropathological conditions. Blocking the inactivation of the endogenous, tissue-protective CB-receptor ligands promotes beneficial neuromodulatory and neuroprotective events (e.g., reduced transmitter release) as well as various downstream signaling pathways. Enhancing AEA levels by inhibiting a critical deactivation step-- i.e., the hydrolytic enzyme FAAH-- is a particularly noteworthy example of exploiting the protective nature of the endocannabinoid system. Selective inhibitors of FAAH boost the endogenous tissue-protective responses in a site- and event-specific manner, thereby avoiding the risk of unwanted psychotropic effects that may result from the prolonged systemic application of CB-receptor agonists. FAAH inhibition is especially effective at attenuating excitotoxic progression in the brain and protecting against synaptic compromise, neuronal death, and the behavioral symptoms of excitotoxic brain damage. Future advances in identifying critical targets of endocannabinoid metabolism and metabolic intermediates whose pharmacological modulation can enhance endocannabinoid signaling to therapeutic levels will undoubtedly open new drug-discovery avenues. Accumulated data already encourage continued preclinical profiling of newer-generation FAAH inhibitors with the aim of promoting lead development candidates into the clinic for first-in-man proof-of-principal studies (Janero and Makriyannis, 2009a).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Aberle J, Flitsch J, Beck NA, Mann O, Busch P, Peitsmeier P, Bell FU. Genetic variation may influence obesity only under conditions of diet: Analysis of three candidate genes. Molecular Genetics and Metabolism. 2008;95:188–191. doi: 10.1016/j.ymgme.2008.07.008. [DOI] [PubMed] [Google Scholar]
- Ahn K, McKinney MK, Cravatt BF. Enzymatic pathways that regulate endocannabinoid signaling in the nervous system. Chemical Reviews. 2008;108:1687–1707. doi: 10.1021/cr0782067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander SPH, Kendall DA. The complications of promiscuity: Endocannabinoid action and metabolism. British Journal of Pharmacology. 2007;152:602–623. doi: 10.1038/sj.bjp.0707456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amantea D, Spagnuolo P, Bari M, Fezza F, Mazzei C, Tassorelli C, Morrone LA, Corasaniti MT, Maccarrone M, Bagetta G. Modulation of the endocannabinoid system by focal brain ischemia in the rat is involved in neuroprotection afforded by 17beta-estradiol. The FEBS Journal. 2007;274:4464–4775. doi: 10.1111/j.1742-4658.2007.05975.x. [DOI] [PubMed] [Google Scholar]
- Arai S, Morita K, Kitayama S, Kumagai K, Kumagai M, Kihira K, Dohi T. Chronic inhibition of the norepinephrine transporter in the brain participates in seizure sensitization to cocaine and local anesthetics. Brain Research. 2003;1:83–90. doi: 10.1016/s0006-8993(02)04068-4. [DOI] [PubMed] [Google Scholar]
- Arizzi MN, Cervone KM, Aberman JE, Betz A, Liu Q, Lin S, Makriyannis A, Salamone JD. Behavioral effects of inhibition of cannabinoid metabolism: The amidase inhibitor AM374 enhances the suppression of lever pressing produced by exogenously administered anandamide. Life Sciences. 2004;74:1001–1011. doi: 10.1016/j.lfs.2003.07.024. [DOI] [PubMed] [Google Scholar]
- Bahr BA, Karanian DA, Makanji SS, Makriyannis A. Targeting the endocannabinoid system in treating brain disorders. Expert Opinion on Investigational Drugs. 2006;15:351–365. doi: 10.1517/13543784.15.4.351. [DOI] [PubMed] [Google Scholar]
- Baker D, Pryce G, Croxford JL, Brown P, Pertwee RG, Makriyannis A, Khanolkar A, Layward L, Fezza F, Bisogno T, Di Marzo V. Endocannabinoids control spasticity in a multiple sclerosis model. FASEB Journal. 2001;15:300–302. doi: 10.1096/fj.00-0399fje. [DOI] [PubMed] [Google Scholar]
- Basavarajappa BS. Critical enzymes involved in endocannabinoid metabolism. Protein and Peptide Letters. 2007;14:237–246. doi: 10.2174/092986607780090829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Begg M, Pacher P, Bátkai S, Osei-Hyiaman D, Offertáler L, Mo FM, Liu J, Kunos G. Evidence for novel cannabinoid receptors. Pharmacology and Therapeutics. 2005;106:133–145. doi: 10.1016/j.pharmthera.2004.11.005. [DOI] [PubMed] [Google Scholar]
- Belfrage P, Gergil B, Strålfors P, Tornqvist H. Hormone-sensitive lipase of rat adipose tissue: Identification and some properties of the enzyme protein. FEBS Letters. 1977;75:259–264. doi: 10.1016/0014-5793(77)80099-9. [DOI] [PubMed] [Google Scholar]
- Beltramo M, Stella N, Calignano A, Lin SY, Makriyannis A, Piomelli D. Functional role of high-affinity anandamide transport, as revealed by selective inhibition. Science. 1997;277:1094–1097. doi: 10.1126/science.277.5329.1094. [DOI] [PubMed] [Google Scholar]
- Beltramo M, de Fonseca FR, Navarro M, Calignano A, Gorriti MA, Grammatikopoulos G, Sadile AG, Giuffrida A, Piomelli D. Reversal of dopamine D(2) receptor responses by an anandamide transport inhibitor. Journal of Neuroscience. 2000;20:3401–3407. doi: 10.1523/JNEUROSCI.20-09-03401.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benito C, Núñez E, Tolón RM, Carrier EJ, Rábano A, Hillard CJ, Romero J. Cannabinoid CB2 receptors and fatty acid amide hydrolase are selectively overexpressed in neuritic plaque-associated glia in Alzheimer's disease brains. Journal of Neuroscience. 2003;23:11136–11141. doi: 10.1523/JNEUROSCI.23-35-11136.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benito C, Kim WK, Chavarría I, Hillard CJ, Mackie K, Tolón RM, Williams K, Romero J. A glial endogenous cannabinoid system is upregulated in the brains of macaques with simian immunodeficiency virus-induced encephalitis. Journal of Neuroscience. 2005;25:2530–2536. doi: 10.1523/JNEUROSCI.3923-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benito C, Romero JP, Tolón RM, Clemente D, Docagne F, Hillard CJ, Guaza C, Romero J. Cannabinoid CB1 and CB2 receptors and fatty acid amide hydrolase are specific markers of plaque cell subtypes in human multiple sclerosis. Journal of Neuroscience. 2007;27:2396–2402. doi: 10.1523/JNEUROSCI.4814-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisogno T, Berrendero F, Ambrosino G, Cebeira M, Ramos JA, Fernandez-Ruiz JJ, Di Marzo V. Brain regional distribution of endocannabinoids: Implications for their biosynthesis and biological function. Biochemical and Biophysical Research Communications. 1999;256:377–380. doi: 10.1006/bbrc.1999.0254. [DOI] [PubMed] [Google Scholar]
- Bisogno T, Howell F, Williams G, Minmassi A, Cascio MG, Ligresti A, Matias I, Schiano-Moriello A, Paul P, Williams EJ, Gangadharan U, Hobbs C, Di Marzo V, Doherty P. Cloning of the first sn-1-DAG lipases points to the spatial and temporal regulation of endocannabinoid signaling in the brain. The Journal of Cell Biology. 2003;163:463–468. doi: 10.1083/jcb.200305129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisogno T. Endogenous cannabinoids: Structure and metabolism. Journal of Neuroendocrinology. 2008;20:1–9. doi: 10.1111/j.1365-2826.2008.01676.x. [DOI] [PubMed] [Google Scholar]
- Bisogno T, Di Marzo V. The role of the endocannabinoid system in Alzheimer's disease: Facts and hypotheses. Current Pharmaceutical Design. 2008;14:2299–3305. doi: 10.2174/138161208785740027. [DOI] [PubMed] [Google Scholar]
- Blankman JL, Simon GM, Cravatt BF. A comprehensive profile of brain enzymes that hydrolyze the endocannabinoid 2-arachidonoylglycerol. Chemistry and Biology. 2007;14:1347–1356. doi: 10.1016/j.chembiol.2007.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cadas H, di Tomaso E, Piomelli D. Occurrence and biosynthesis of endogenous cannabinoid precursor, N-arachidonoyl phosphatidylethanolamine, in rat brain. Journal of Neuroscience. 1997;17:1226–1242. doi: 10.1523/JNEUROSCI.17-04-01226.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Centonze D, Finazzi-Agrò A, Bernardi G, Maccarrone M. The endocannabinoid system in targeting inflammatory neurodegenerative diseases. Trends in Pharmacological Sciences. 2007;28:180–187. doi: 10.1016/j.tips.2007.02.004. [DOI] [PubMed] [Google Scholar]
- Chang L, Luo L, Palmer JA, Sutton S, Wilson SJ, Barbier AJ, Breitenbucher JG, Chaplan SR, Webb M. Inhibition of fatty acid amide hydrolase produces analgesia by multiple mechanisms. British Journal of Pharmacology. 2007;148:102–113. doi: 10.1038/sj.bjp.0706699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen JK, Chen J, Imig JD, Wei S, Hachey DL, Guthi JS, Falck JR, Capdevila JH, Harrris RC. Identification of novel endogenous cytochrome p450 arachidonate metabolites with high affinity for cannaibnoid receptors. The Journal of Biological Chemistry. 2008;283:24514–24524. doi: 10.1074/jbc.M709873200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coomber B, O'Donoghue MF, Mason R. Inhibition of endocannabinoid metabolism attenuates enhanced hippocampal neuronal activity induced by kainic acid. Synapse. 2008;62:746–755. doi: 10.1002/syn.20547. [DOI] [PubMed] [Google Scholar]
- Cravatt BF, Demarest K, Patricelli MP, Bracey MH, Giang DK, Martin BR, Lichtman AH. Supersensitivity to anandamide and enhanced endogenous cannabinoid signaling in mice lacking fatty acid amide hydrolase. Proceedings of the National Academy of Sciences of the United States of America. 2001;98:9371–9376. doi: 10.1073/pnas.161191698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deadwyler SA, Hampson RE, Bennett BA, Edwards TA, Mu J, Pacheco MA, Ward SJ, Childers SR. Cannabinoids modulate potassium current in cultured hippocampal neurons. Recept Chan. 1993;1:121–134. [PubMed] [Google Scholar]
- Deadwyler SA, Hampson RE, Mu J, Whyte A, Childers S. Cannabinoids modulate voltage sensitive potassium A-current in hippocampal neurons via a cAMP-dependent process. The Journal of Pharmacology and Experimental Therapeutics. 1995;273:734–743. [PubMed] [Google Scholar]
- Devane WA, Hanus L, Breuer A, Pertwee RG, Stevenson LA, Griffin G, Gibson D, Mandelbaum A, Etinger A, Mechoulam R. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science. 1992;258:1946–1949. doi: 10.1126/science.1470919. [DOI] [PubMed] [Google Scholar]
- Di Marzo V, Fontana A, Cadas H, Schinelli S, Cimino G, Schwartz JC, Piomelli D. Formation and inactivation of endogenous cannabinoid anandamide in central neurons. Nature. 1994;372:686–691. doi: 10.1038/372686a0. [DOI] [PubMed] [Google Scholar]
- Di Marzo V, Matias I. Endocannabinoid control of food intake and energy balance. Nature Neuroscience. 2005;8:585–589. doi: 10.1038/nn1457. [DOI] [PubMed] [Google Scholar]
- Di Marzo V. Endocannabinoids: Synthesis and degradation. Reviews of Physiology, Biochemistry and Pharmacology. 2008;160:1–24. doi: 10.1007/112_0505. [DOI] [PubMed] [Google Scholar]
- Fegley D, Kathuria S, Mercier R, Li C, Goutopoulos A, Makriyannis A, Piomelli D. Anandamide transport is independent of fatty-acid amide hydrolase activity and is blocked by the hydrolysis-resistant inhibitor AM1172. Proceedings of the National Academy of Sciences of the United States of America. 2004;101:8756–8761. doi: 10.1073/pnas.0400997101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Felder CC, Joyce KE, Briley EM, Mansouri J, Mackie K, Blond O, Lai Y, Ma AL, Mitchell RL. Comparison of the pharmacology and signal transduction of the human cannabinoid CB1 and CB2 receptors. Molecular Pharmacology. 1995;48:443–450. [PubMed] [Google Scholar]
- Fezza F, De Simone C, Amadio D, Maccarrone M. Fatty acid amide hydrolase: A gate-keeper of the endocannabinoid system. Sub-cellular Biochemistry. 2008;49:101–132. doi: 10.1007/978-1-4020-8831-5_4. [DOI] [PubMed] [Google Scholar]
- Flanagan JM, Gerber AL, Cadet JL, Beutler E, Sipe JC. The fatty acid amide hydrolase 385 A/A (P129T) variant: Haplotype analysis of an ancient missense mutation and validation of risk for drug addiction. Human Genetics. 2006;120:581–588. doi: 10.1007/s00439-006-0250-x. [DOI] [PubMed] [Google Scholar]
- Freund TF. Interneuron diversity series: Rhythm and mood in perisomatic inhibition. Trends in Neurosciences. 2003;26:489–495. doi: 10.1016/S0166-2236(03)00227-3. [DOI] [PubMed] [Google Scholar]
- Galve-Roperh I, Rueda D, Gómez del Pulgar T, Velasco G, Guzmán M. Mechanism of extracellular signal-regulated kinase activation by the CB(1) cannabinoid receptor. Molecular Pharmacology. 2002;62:1385–1392. doi: 10.1124/mol.62.6.1385. [DOI] [PubMed] [Google Scholar]
- Galve-Roperh I, Aguado T, Palazuelos J, Guzmán M. Mechanisms of control of neuron survival by the endocannabinoid system. Current Pharmaceutical Design. 2008;14:2279–2288. doi: 10.2174/138161208785740117. [DOI] [PubMed] [Google Scholar]
- Garcia-Ovejero D, Arevalo-Martin A, Petrosino S, Docagne F, Hagen C, Bisogno T, Watanabe M, Guaza C, Di Marzo V, Molina-Holgado E. The endocannabinoid system is modulated in response to spinal cord injury in rats. Neurobiology of Disease. 2009;33:57–71. doi: 10.1016/j.nbd.2008.09.015. [DOI] [PubMed] [Google Scholar]
- Gerdeman G, Lovinger DM. CB1 cannabinoid receptor inhibits synaptic release of glutamate in rat dorsolateral striatum. Journal of Neurophysiology. 2001;85:468–471. doi: 10.1152/jn.2001.85.1.468. [DOI] [PubMed] [Google Scholar]
- Gifford AN, Bruneus M, Lin S, Goutopoulos A, Makriyannis A, Volkow ND, Gatley SJ. Potentiation of the action of anandamide on hippocampal slices by the fatty acid amide hydrolase inhibitor, palmitylsulphonyl fluoride (AM 374) European Journal of Pharmacology. 1999;383:9–14. doi: 10.1016/s0014-2999(99)00609-3. [DOI] [PubMed] [Google Scholar]
- Gillum MP, Zhang D, Zhang XM, Erion DM, Jamison RA, Choi C, Dong J, Shanabrough M, Duenas HR, Frederick DW, Hsiao JJ, Horvath TL, Lo CM, Tso P, Cline GW, Shulman GI. N-acylphosphatidylethanolamine, a gout-derived circulating factor induced by fat ingestion, inhibits food intake. Cell. 2008;135:813–824. doi: 10.1016/j.cell.2008.10.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gómez del Pulgar T, Velasco G, Guzmán M. The CB1 cannabinoid receptor is coupled to the activation of protein kinase B/Akt. The Biochemical Journal. 2000;347:369–373. doi: 10.1042/0264-6021:3470369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goparaju SK, Udea N, Yamaguchi H, Yamamoto S. Anandamide aminohydrolase reacting with 2-arachidonoylglycerol, another cannabinoid receptor ligand. FEBS Letters. 1998;422:69–73. doi: 10.1016/s0014-5793(97)01603-7. [DOI] [PubMed] [Google Scholar]
- Griffin G, Wray EJ, Tao Q, McAllister SD, Rorrer WK, Aung MM, Martin BR, Abood ME. Evaluation of the cannabinoid CB2 receptor-selective antagonist, SR144528: Further evidence for cannabinoid CB2 receptor absence in the rat central nervous system. European Journal of Pharmacology. 1999;377:117–125. doi: 10.1016/s0014-2999(99)00402-1. [DOI] [PubMed] [Google Scholar]
- Guindon J, Hohmann AG. A physiological role for endocannabinoid-derived products of cyclooxygenase-2-mediated oxidative metabolism. British Journal of Pharmacology. 2008;153:1341–1343. doi: 10.1038/bjp.2008.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hájos N, Katona I, Naiem SS, Mackie K, Ledent C, Mody I, Freund TF. Cannabinoids inhibit hippocampal GABAergic transmission and network oscillations. The European Journal of Neuroscience. 2000;12:3239–3249. doi: 10.1046/j.1460-9568.2000.00217.x. [DOI] [PubMed] [Google Scholar]
- Hansen HH, Schmid PC, Bittigau P, Lastres-Becker I, Berrendero F, Manzanares J, Ikonomidou C, Schmid HH, Fernandez-Ruiz JJ, Hansen HS. Anandamide, but not 2 arachidonoylglycerol, accumulates during in vivo neurodegeneration. Journal of Neurochemistry. 2001;78:1415–1427. doi: 10.1046/j.1471-4159.2001.00542.x. [DOI] [PubMed] [Google Scholar]
- Hashimotodani Y, Ohno-Shosaku T, Watanabe M, Kano M. Roles of phospholipase Cβ and NMDA receptor in activity-dependent endocannabinoid release. The Journal of Physiology. 2007;584:373–380. doi: 10.1113/jphysiol.2007.137497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashimotodani Y, Ohno-Shosaku T, Maejima T, Fukami K, Kano M. Pharmacological evidence for the involvement of diacylglycerol lipase in depolarization-induced endocannabinoid release. Neuropharmacology. 2008;54:58–67. doi: 10.1016/j.neuropharm.2007.06.002. [DOI] [PubMed] [Google Scholar]
- Hillard CJ. Biochemistry and pharmacology of the endocannabinoids arachidonylethanolamide and 2-arachidonylglycerol. Prostaglandins and Other Lipid Mediators. 2000;61:3–18. doi: 10.1016/s0090-6980(00)00051-4. [DOI] [PubMed] [Google Scholar]
- Hoover HS, Blankman JL, Niessen S, Cravatt BF. Selectivity of inhibitors of endocannabinoid biosynthesis evaluated by activity-based protein profiling. Bioorganic and Medicinal Chemistry Letters. 2008;18:5838–5841. doi: 10.1016/j.bmcl.2008.06.091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itzhak Y, Ali SF. Role of nitrergic system in behavioral and neurotoxic effects of amphetamine analogs. Pharmacology and Therapeutics. 2006;109:246–262. doi: 10.1016/j.pharmthera.2005.08.004. [DOI] [PubMed] [Google Scholar]
- Iversen L. Cannabis and the brain. Brain. 2003;126:1252–1270. doi: 10.1093/brain/awg143. [DOI] [PubMed] [Google Scholar]
- Jackson SJ, Pryce G, Diemel LT, Cuzner ML, Baker D. Cannabinoid-receptor 1 null mice are susceptible to neurofilament damage and caspase 3 activation. Neuroscience. 2005;134:261–268. doi: 10.1016/j.neuroscience.2005.02.045. [DOI] [PubMed] [Google Scholar]
- Janero DR, Makriyannis A. Targeted modulators of the endogenous cannabinoid system: Future medications to treat addiction disorders and obesity. Current Psychiatry Reports. 2007;9:365–373. doi: 10.1007/s11920-007-0047-1. [DOI] [PubMed] [Google Scholar]
- Janero DR, Makriyannis A. International Review of Psychiatry. Abingdon, England: 2009a. Pharmacotherapeutic modulation of the endocannabinoid signaling system in psychiatric disorders: Drug-discovery strategies. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janero DR, Makriyannis A. Cannabinoid receptor antagonists: Pharmacological opportunities, clinical experience, and translational prognosis. Expert Opinion on Emerging Drugs. 2009b;14:43–65. doi: 10.1517/14728210902736568. [DOI] [PubMed] [Google Scholar]
- Jung KM, Mangieri R, Stapleton C, Fegley D, Wallace M, Mackie K, Piomelli D. Stimulation of endocannabinoid formation in brain slice cultures through activation of group 1 metabotropic glutamate receptors. Molecular Pharmacology. 2005;68:1196–1202. doi: 10.1124/mol.105.013961. [DOI] [PubMed] [Google Scholar]
- Jung KM, Astarita G, Zhu C, Wallace M, Mackie K, Piomelli D. A key role for diacylglycerol lipase-α in metabotropic glutamate receptor-dependent endocannabinoid mobilization. Molecular Pharmacology. 2007;72:612–621. doi: 10.1124/mol.107.037796. [DOI] [PubMed] [Google Scholar]
- Jung KM, Astarita G, Yasar S, Head E, Cotman CW, Piomelli D. Deficits in anandamide signaling underlie cognitive dysfunction in Alzheimer's Disease. Society for Neuroscience Abstracts. 2008;626(1) [Google Scholar]
- Justinova Z, Mangieri RA, Bortolato M, Chefer SI, Mukhin AG, Clapper JR, King AR, Redhi GH, Yasar S, Piomelli D, Goldberg SR. Fatty acid amide hydrolase inhibition heightens anandamide signaling without producing reinforcing effects in primates. Biological Psychiatry. 2008;64:930–937. doi: 10.1016/j.biopsych.2008.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kano M, Ohno-Shoakau T, Hashimotodani Y, Uchigashima M, Watanabe M. Endocannabinoid-mediated control of synaptic transmission. Physiological Reviews. 2009;89:309–380. doi: 10.1152/physrev.00019.2008. [DOI] [PubMed] [Google Scholar]
- Karanian DA, Brown QB, Makriyannis A, Bahr BA. Blocking cannabinoid activation of FAK and ERK1/2 compromises synaptic integrity in hippocampus. European Journal of Pharmacology. 2005a;508:47–56. doi: 10.1016/j.ejphar.2004.12.009. [DOI] [PubMed] [Google Scholar]
- Karanian DA, Brown QB, Makriyannis A, Kosten TA, Bahr BA. Dual modulation of endocannabinoid transport and fatty acid amide hydrolase protects against excitotoxicity. Journal of Neuroscience. 2005b;25:7813–7820. doi: 10.1523/JNEUROSCI.2347-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karanian DA, Karim SL, Wood JT, Williams JS, Lin S, Makriyannis A, Bahr BA. Endocannabinoid enhancement protects against kainic acid-induced seizures and associated brain damage. Journal of Pharmacology and Experimental Therapeutics. 2007;322:1059–1066. doi: 10.1124/jpet.107.120147. [DOI] [PubMed] [Google Scholar]
- Kathuria S, Gaetani S, Fegley D, Valiño F, Duranti A, Tontini A, Mor M, Tarzia G, La Rana G, Calignano A, Giustino A, Tattoli M, Palmery M, Cuomo V, Piomelli D. Modulation of anxiety through blockade of anandamide hydrolysis. Nature Medicine. 2003;9:76–81. doi: 10.1038/nm803. [DOI] [PubMed] [Google Scholar]
- Khanapure SP, Garvey DS, Janero DR, Letts LG. Eicosanoids in inflammation: Biosynthesis, pharmacology, and therapeutic frontiers. Current Topics in Medicinal Chemistry. 2007;7:311–340. doi: 10.2174/156802607779941314. [DOI] [PubMed] [Google Scholar]
- Khaspekov LG, Brenz Verca MS, Frumkina LE, Hermann H, Marsicano G, Lutz B. Involvement of brain-derived neurotrophic factor in cannabinoid receptor-dependent protection against excitotoxicity. The European Journal of Neuroscience. 2004;19:1691–1698. doi: 10.1111/j.1460-9568.2004.03285.x. [DOI] [PubMed] [Google Scholar]
- Kim DJ, Thayer SA. Activation of CB1 cannabinoid receptors inhibits neurotransmitter release from identified synaptic sites in rat hippocampal cultures. Brain Research. 2000;852:398–405. doi: 10.1016/s0006-8993(99)02210-6. [DOI] [PubMed] [Google Scholar]
- Kosten TA, Miserendino MJ, Chi S, Nestler EJ. Fischer and Lewis rat strains show differential cocaine effects in conditioned place preference and behavioral sensitization but not in locomotor activity or conditioned taste aversion. The Journal of Pharmacology and Experimental Therapeutics. 1994;269:137–144. [PubMed] [Google Scholar]
- Kreitzer AC, Regehr WG. Retrograde inhibition of presynaptic calcium influx by endogenous cannabinoids at excitatory synapses onto Purkinje cells. Neuron. 2001;29:717–727. doi: 10.1016/s0896-6273(01)00246-x. [DOI] [PubMed] [Google Scholar]
- Kreutz S, Koch M, Böttger C, Ghadban C, Korf HW, Dehghani F. 2-Arachidonoylglycerol elicits neuroprotective effects on excitotoxically lesioned dentate gyrus granule cells via abnormal-cannabidiol-sensitive receptors on microglial cells. Glia. 2009;57:286–294. doi: 10.1002/glia.20756. [DOI] [PubMed] [Google Scholar]
- Lauckner JE, Jensen JB, Chen HY, Lu HC, Hille B, Mackie K. GPR55 is a cannabinoid receptor that increases intracellular calcium and inhibits M current. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:2699–2704. doi: 10.1073/pnas.0711278105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Wang L, Harvey-White J, Osei-Hyiaman D, Razdan RK, Gong Q, Chan AC, Zhou Z, Huang BX, Kim HY, Kunos G. A biosynthetic pathway for anandamide. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:13345–13350. doi: 10.1073/pnas.0601832103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Wang L, Harvey-White J, Huang BX, Kim HY, Luquet S, Palmiter RD, Krystal G, Rai R, Mahadevan A, Razdan RK, Kunos G. Multiple pathways involved in the biosynthesis of anandamide. Neuropharmacology. 2008;54:1–7. doi: 10.1016/j.neuropharm.2007.05.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovinger DM. Presynaptic modulation by endocannabinoids. Handbook of Experimental Pharmacology. 2008;184:435–477. doi: 10.1007/978-3-540-74805-2_14. [DOI] [PubMed] [Google Scholar]
- Ludányi A, Eross L, Czirják S, Vajda J, Halász P, Watanabe M, Palkovits M, Maglóczky Z, Freund TF, Katona I. Downregulation of the CB1 cannabinoid receptor and related molecular elements of the endocannabinoid system in epileptic human hippocampus. Journal of Neuroscience. 2008;28:2976–2990. doi: 10.1523/JNEUROSCI.4465-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maccarrone M, Gubellini P, Bari M, Picconi B, Battista N, Centonze D, Bernardi G, Finazzi-Agrò A, Calabresi P. Levodopa treatment reverses endocannabinoid system abnormalities in experimental parkinsonism. Journal of Neurochemistry. 2003;85:1018–1025. doi: 10.1046/j.1471-4159.2003.01759.x. [DOI] [PubMed] [Google Scholar]
- Maccarrone M, Rossi S, Bari M, De Chiara V, Fezza F, Musella A, Gasperi V, Prosperetti C, Bernardi G, Finazzi-Agrò A, Cravatt BF, Centonze D. Anandamide inhibits metabolism and physiological actions of 2-arachidonoylglycerol in the striatum. Nature Neuroscience. 2008;11:152–159. doi: 10.1038/nn2042. [DOI] [PubMed] [Google Scholar]
- Mackie K. Cannabinoid receptors as therapeutic targets. Annual Review of Pharmacology and Toxicology. 2006;46:101–122. doi: 10.1146/annurev.pharmtox.46.120604.141254. [DOI] [PubMed] [Google Scholar]
- Mackie K. Cannabinoid receptors: What they are and what they do. Journal of Neuroendocrinology. 2008;20:10–14. doi: 10.1111/j.1365-2826.2008.01671.x. [DOI] [PubMed] [Google Scholar]
- Marsicano G, Goodenough S, Monory K, Hermann H, Eder M, Cannich A, Azad SC, Cascio MG, Gutiérrez SO, van der Stelt M, López-Rodriguez ML, Casanova E, Schütz G, Zieglgänsberger W, Di Marzo V, Behl C, Lutz B. CB1 cannabinoid receptors and on-demand defense against excitotoxicity. Science. 2003;302:84–88. doi: 10.1126/science.1088208. [DOI] [PubMed] [Google Scholar]
- Matsuda LA, Lolait SJ, Brownstein MJ, Young AC, Bonner TI. Structure of a cannabinoid receptor and functional expression of cloned cDNA. Nature. 1990;346:561–564. doi: 10.1038/346561a0. [DOI] [PubMed] [Google Scholar]
- Matsumoto RR, Gilmore DL, Pouw B, Bowen WD, Williams W, Kausar A, Coop A. Novel analogs of the sigma receptor ligand BD1008 attenuate cocaine-induced toxicity in mice. European Journal of Pharmacology. 2004;492:21–26. doi: 10.1016/j.ejphar.2004.03.037. [DOI] [PubMed] [Google Scholar]
- Mechoulam R, Ben-Shabat S, Hanus L, Ligumsky M, Kaminski NE, Schatz AR, Gopher A, Almog S, Martin BR, Compton DR, et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochemical Pharmacology. 1995;50:83–90. doi: 10.1016/0006-2952(95)00109-d. [DOI] [PubMed] [Google Scholar]
- Mechoulam R, Fride E, Di Marzo V. Endocannabinoids. European Journal of Pharmacology. 1998;359:1–18. doi: 10.1016/s0014-2999(98)00649-9. [DOI] [PubMed] [Google Scholar]
- Mets B, Winger G, Cabrera C, Seo S, Jamdar S, Yang G, Zhao K, Briscoe RJ, Almonte R, Woods JH, Landry DW. A catalytic antibody against cocaine prevents cocaine's reinforcing and toxic effects in rats. Proceedings of the National Academy of Sciences of the United States of America. 1998;95:10176–10181. doi: 10.1073/pnas.95.17.10176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Micale V, Mazzola C, Drago F. Endocannabinoids and neurodegenerative diseases. Pharmacological Research. 2007;56:382–392. doi: 10.1016/j.phrs.2007.09.008. [DOI] [PubMed] [Google Scholar]
- Milton NG. Anandamide and noladin ether prevent neurotoxicity of the human amyloid- beta peptide. Neuroscience Letters. 2002;332:127–130. doi: 10.1016/s0304-3940(02)00936-9. [DOI] [PubMed] [Google Scholar]
- Mitchell MD, Sato TA, Wang A, Keelan JA, Ponnampalam AP, Glass M. Cannabinoids stimulate prostaglandin production by human gestational tissues through a tissue- and CB1-receptor-specific mechanism. American Journal of Physiology, Endocrinology and Metabolism. 2008;294:352–356. doi: 10.1152/ajpendo.00495.2007. [DOI] [PubMed] [Google Scholar]
- Molina-Holgado F, Pinteaux E, Heenan L, Moore JD, Rothwell NJ, Gibson RM. Neuroprotective effects of the synthetic cannabinoid HU-210 in primary cortical neurons are mediated by phosphatidylinositol 3-kinase/AKT signaling. Molecular and Cellular Neuroscience. 2005;28:189–194. doi: 10.1016/j.mcn.2004.09.004. [DOI] [PubMed] [Google Scholar]
- Moreira FA, Kaiser N, Monory K, Lutz B. Reduced anxiety-like behaviour induced by genetic and pharmacological inhibition of the endocannabinoid-degrading enzyme fatty acid amide hydrolase (FAAH) is mediated by CB1 receptors. Neuropharmacology. 2008;54:141–150. doi: 10.1016/j.neuropharm.2007.07.005. [DOI] [PubMed] [Google Scholar]
- Mu J, Zhuang SY, Kirby MT, Hampson RE, Deadwyler SA. Cannabinoid receptors differentially modulate potassium A and D currents in hippocampal neurons in culture. The Journal of Pharmacology and Experimental Therapeutics. 1999;291:893–902. [PubMed] [Google Scholar]
- Munro S, Thomas KL, Abu-Shaar M. Molecular characterization of a peripheral receptor for cannabinoids. Nature. 1993;365:61–65. doi: 10.1038/365061a0. [DOI] [PubMed] [Google Scholar]
- Naidu PS, Booker L, Cravatt BF, Lichtman AH. Synergy between enzyme inhibitors of fatty acid amide hydrolase and cyclooxygenase in visceral nociception. The Journal of Pharmacology and Experimental Therapeutics. 2009 doi: 10.1124/jpet.108.143487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Natarajan V, Schmid PC, Reddy PV, Zuzarte-Augustin ML, Schmid HH. Biosynthesis of N-acylethanolamine phospholipids by dog brain preparations. Journal of Neurochemistry. 1983;41:1303–1312. doi: 10.1111/j.1471-4159.1983.tb00825.x. [DOI] [PubMed] [Google Scholar]
- Natarajan V, Schmid PC, Reddy PV, Schmid HH. Catabolism of N-acylethanolamine phospholipids by dog brain preparations. Journal of Neurochemistry. 1984;41:1303–1312. doi: 10.1111/j.1471-4159.1983.tb00825.x. [DOI] [PubMed] [Google Scholar]
- Nucci C, Gasperi V, Tartaglione R, Cerulli A, Terrinoni A, Bari M, De Simone C, Agrò AF, Morrone LA, Corasaniti MT, Bagetta G, Maccarrone M. Involvement of the endocannabinoid system in retinal damage after high intraocular pressure-induced ischemia in rats. Investigative Ophthalmology and Visual Science. 2007;48:2997–3004. doi: 10.1167/iovs.06-1355. [DOI] [PubMed] [Google Scholar]
- Ohno-Shosaku T, Maejima T, Kano M. Endogenous cannabinoids mediate retrograde signals from depolarized postsynaptic neurons to presynaptic terminals. Neuron. 2001;29:729–738. doi: 10.1016/s0896-6273(01)00247-1. [DOI] [PubMed] [Google Scholar]
- Ortar G, Bisogno T, Ligresti A, Morera E, Nalli M, Di Marzo V. Tetrahydrolipstatin analogues as modulators of endocannabinoid 2-arachidonoylglycerol metabolism. Journal of Medicinal Chemistry. 2008;51:6970–6979. doi: 10.1021/jm800978m. [DOI] [PubMed] [Google Scholar]
- Pacher P, Bátkai S, Kunos G. The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacological Reviews. 2006;58:389–462. doi: 10.1124/pr.58.3.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pacher P, Haskò G. Endocannabinoids and cannabinoid receptors in ischaemia-reperfusion injury and preconditioning. British Journal of Pharmacology. 2008;153:252–262. doi: 10.1038/sj.bjp.0707582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panikashvili D, Mechoulam R, Beni SM, Alexandrovich A, Shohami E. CB1 cannabinoid receptors are involved in neuroprotection via NF-kappa B inhibition. Journal of Cerebral Blood Flow and Metabolism. 2005;25:477–484. doi: 10.1038/sj.jcbfm.9600047. [DOI] [PubMed] [Google Scholar]
- Parmentier-Batteur S, Jin K, Mao XO, Xie L, Greenberg DA. Increased severity of stroke in CB1 cannabinoid receptor knock-out mice. Journal of Neuroscience. 2002;22:9771–9775. doi: 10.1523/JNEUROSCI.22-22-09771.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavlopoulos S, Thakur GA, Nikas SP, Makriyannis A. Cannabinoid receptors as therapeutic targets. Current Pharmaceutical Design. 2006;12:1751–1769. doi: 10.2174/138161206776873743. [DOI] [PubMed] [Google Scholar]
- Pei Y, Mercier RW, Anday JK, Thakur GA, Zvonok AM, Hurst D, Reggio PH, Janero DR, Makriyannis AM. Ligand-binding architecture of human CB2 cannabinoid receptor: Evidence for receptor subtype-specific binding motif and modeling GPCR activation. Chemistry & Biology. 2008;15:1207–1219. doi: 10.1016/j.chembiol.2008.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pertwee RG. GPR55: A new member of the cannabinoid receptor clan? British Journal of Pharmacology. 2007;152:984–986. doi: 10.1038/sj.bjp.0707464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pertwee RG. Ligands that target cannabinoid receptors: From THC to anandamide and beyond. Addiction Biology. 2008;13:147–159. doi: 10.1111/j.1369-1600.2008.00108.x. [DOI] [PubMed] [Google Scholar]
- Piomelli D, Beltramo M, Glasnapp S, Lin SY, Goutopoulos A, Xie XQ, Makriyannis A. Structural determinants for recognition and translocation by the anandamide transporter. Proceedings of the National Academy of Sciences of the United States of America. 1999;96:5802–5807. doi: 10.1073/pnas.96.10.5802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prusakiewicz JJ, Turman MV, Vila A, Ball HL, Al-Mestarihi AH, Di Marzo V, Marnett LJ. Oxidative metabolism of lipoamino acids and vanilloids by lipoxygenases and cyclooxygenases. Archives of Biochemistry and Biophysics. 2007;464:260–268. doi: 10.1016/j.abb.2007.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramírez BG, Blázquez C, Gómez del Pulgar T, Guzmán M, de Ceballos ML. Prevention of Alzheimer's disease pathology by cannabinoids: Neuroprotection mediated by blockade of microglial activation. Journal of Neuroscience. 2005;25:1904–1913. doi: 10.1523/JNEUROSCI.4540-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saario SM, Laitinen JT. Monoglyceride lipase as an enzyme hydrolyzing 2-arachidonoylglycerol. Chemistry and Biodiversity. 2007;4:1903–1913. doi: 10.1002/cbdv.200790158. [DOI] [PubMed] [Google Scholar]
- Schäbitz WR, Giuffrida A, Berger C, Aschoff A, Schwaninger M, Schwab S, Piomelli D. Release of fatty acid amides in a patient with hemispheric stroke: A microdialysis study. Stroke. 2002;33:2112–2114. doi: 10.1161/01.str.0000023491.63693.18. [DOI] [PubMed] [Google Scholar]
- Scherma M, Medalie J, Fratta W, Vadivel SK, Makriyannis A, Piomelli D, Mikics E, Haller J, Yasar S, Tanda G, Goldberg SR. The endogenous cannabinoid anandamide has effects on motivation and anxiety that are revealed by fatty acid amide hydrolase (FAAH) inhibition. Neuropharmacology. 2008;54:129–140. doi: 10.1016/j.neuropharm.2007.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider C, Pratt DA, Porter NA, Brash AR. Control of oxygenation in lipoxygenase and cyclooxygenase catalysis. Chemistry and Biology. 2007;14:473–488. doi: 10.1016/j.chembiol.2007.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schomacher M, Müller HD, Sommer C, Schwab S, Schäbitz WR. Endocannabinoids mediate neuroprotection after transient focal cerebral ischemia. Brain Research. 2008;1240:213–220. doi: 10.1016/j.brainres.2008.09.019. [DOI] [PubMed] [Google Scholar]
- Shen M, Piser TM, Seybold VS, Thayer SA. Cannabinoid receptor agonists inhibit glutamatergic synaptic transmission in rat hippocampal cultures. Journal of Neuroscience. 1996;16:4322–4334. doi: 10.1523/JNEUROSCI.16-14-04322.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen M, Thayer SA. The cannabinoid agonist Win55,212-2 inhibits calcium channels by receptor-mediated and direct pathways in cultured rat hippocampal neurons. Brain Research. 1998;783:77–84. doi: 10.1016/s0006-8993(97)01195-5. [DOI] [PubMed] [Google Scholar]
- Shu-Jung S, Bradshaw HB, Chen JS, Tan B, Walker JM. Prostaglandin E2 glycerol ester, an endogenous COX-2 metabolite of 2-arachidonoylglycerol, induces hyperalgesia and modulates NFκB activity. British Journal of Pharmacology. 2008;153:1538–1549. doi: 10.1038/bjp.2008.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon GM, Cravatt BF. Endocannabinoid biosynthesis proceeding through glycerophospho-N-acyl ethanolamine and a role for α/β-hydrolase in this pathway. The Journal of Biological Chemistry. 2006;281:26465–26472. doi: 10.1074/jbc.M604660200. [DOI] [PubMed] [Google Scholar]
- Simon GM, Cravatt BF. Anandamide biosynthesis catalyzed by the phosphodiesterase GDE1 and detection of glycerophospho-N-acyl ethanolamine precursors in mouse brain. The Journal of Biological Chemistry. 2008;283:9341–9349. doi: 10.1074/jbc.M707807200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singla S, Kreitzer AC, Malenka RC. Mechanisms for synapse specificity during striatal long-term depression. Journal of Neuroscience. 2007;27:5260–5264. doi: 10.1523/JNEUROSCI.0018-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Storr M, Emmerdinger D, Diegelmann J, Yüce B, Pfenning S, Ochsenkün T, Göke B, Brand S. The role of fatty acid hydrolase (FAAH) gene variants in inflammatory bowel disease. Alimentary Pharmacology and Therapeutics. 2008a;29:542–551. doi: 10.1111/j.1365-2036.2008.03910.x. [DOI] [PubMed] [Google Scholar]
- Storr MA, Keenan CM, Emmerdinger D, Zhang H, Yüce B, Sibaev A, Massa F, Buckley NE, Lutz B, Göke B, Brand S, Patel KD, Sharkey KA. Targeting endocannabinoid degradation products against experimental colitis in mice: Involvement of CB1 and CB2 receptors. Journal of Molecular Medicine. 2008b;86:925–936. doi: 10.1007/s00109-008-0359-6. [DOI] [PubMed] [Google Scholar]
- Sugiura T, Kondo S, Sukagawa A, Tonegawa T, Nakane S, Yamashita A, Ishima Y, Waku K. Transacylase-mediated and phosphodiesterase-mediated synthesis of N-arachidonoylethanolamine, an endogenous cannabinoid-receptor ligand, in rat brain microsomes. Comparison with synthesis from free arachidonic acid and ethanolamine. European Journal of Biochemistry. 1996;240:53–62. doi: 10.1111/j.1432-1033.1996.0053h.x. [DOI] [PubMed] [Google Scholar]
- Tsuboi K, Zhao LY, Okamoto Y, Araki N, Ueno M, Sakamoto H, Udea N. Predominanat expression of lysosomal N-acylethanolamine-hydrolyzing acid amidase in macrophages revealed by immunochemical studies. Biochimica et Biophysica Acta. 2007a;1771:623–632. doi: 10.1016/j.bbalip.2007.03.005. [DOI] [PubMed] [Google Scholar]
- Tsuboi K, Takezaki N, Udea N. The N-acylethanolamine-hydrolyzing acid amidase (NAAA) Chemistry and Biodiversity. 2007b;4:1914–1925. doi: 10.1002/cbdv.200790159. [DOI] [PubMed] [Google Scholar]
- Vandevoorde S, Lambert DM. The multiple pathways of endocannabinoid metabolism: A zoom out. Chemistry and Biodiversity. 2007;4:1858–1881. doi: 10.1002/cbdv.200790156. [DOI] [PubMed] [Google Scholar]
- Van Sickle MD, Duncan M, Kingsley PJ, Mouihate A, Urbani P, Mackie K, Stella N, Makriyannis A, Piomelli D, Davison JS, Marnett LJ, Di Marzo V, Pittman QJ, Patel KD, Sharkey KA. Identification and functional characterization of brainstem cannabinoid CB2 receptors. Science. 2005;310:329–332. doi: 10.1126/science.1115740. [DOI] [PubMed] [Google Scholar]
- van Tienhoven M, Atkins J, Li Y, Glynn P. Human neuropathy target esterase catalyzes hydrolysis of membrane lipids. The Journal of Biological Chemistry. 2002;277:20942–20948. doi: 10.1074/jbc.M200330200. [DOI] [PubMed] [Google Scholar]
- Vemuri VK, Janero DR, Makriyannis A. Pharmacotherapeutic targeting of the endocannabinoid signaling system: Drugs for obesity and the metabolic syndrome. Physiology and Behavior. 2008;93:671–686. doi: 10.1016/j.physbeh.2007.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace MJ, Blair RE, Falenski KW, Martin BR, DeLorenzo RJ. The endogenous cannabinoid system regulates seizure frequency and duration in a model of temporal lobe epilepsy. The Journal of Pharmacology and Experimental Therapeutics. 2003;307:129–137. doi: 10.1124/jpet.103.051920. [DOI] [PubMed] [Google Scholar]
- Wang H, Xie H, Sun X, Kingsley PJ, Marnett LJ, Cravatt BF, Dey SK. Differential regulation of endocannabinoid synthesis and degradation in the uterus during embryo implantation. Prostaglandins and Other Lipid Mediators. 2007;83:62–74. doi: 10.1016/j.prostaglandins.2006.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webb M, Luo L, Ma JY, Tham CS. Genetic deletion of fatty acid amide hydrolase results in improved long-term outcome in chronic autoimmune encephalitis. Neuroscience Letters. 2008;439:106–110. doi: 10.1016/j.neulet.2008.04.090. [DOI] [PubMed] [Google Scholar]
- Wei BO, Mikkelsen TS, McKinney MK, Lander ES, Cravatt BF. A second fatty acid amide hydrolase with variable distribution among placental mammals. The Journal of Biological Chemistry. 2006;281:36569–36578. doi: 10.1074/jbc.M606646200. [DOI] [PubMed] [Google Scholar]
- Wilkins LH, Jr, Prendergast MA, Blanchard J, Holley RC, Chambers ER, Littleton JM. Potential value of changes in cell markers in organotypic hippocampal cultures associated with chronic EtOH exposure and withdrawal: Comparison with NMDA-induced changes. Alcoholism, Clinical and Experimental Research. 2006;30:1768–1780. doi: 10.1111/j.1530-0277.2006.00210.x. [DOI] [PubMed] [Google Scholar]
- Wilson RI, Kunos G, Nicoll RA. Presynaptic specificity of endocannabinoid signaling in the hippocampus. Neuron. 2001;31:453–462. doi: 10.1016/s0896-6273(01)00372-5. [DOI] [PubMed] [Google Scholar]
- Wood JT, Williams JS, Pandarinathan L, Courville A, Keplinger MR, Janero DR, Vouros P, Makriyannis A, Lammi-Keefe CJ. Comprehensive profiling of the human circulating endocannabinoid metabolome: Clinical sampling and sample storage parameters. Clinical Chemistry and Laboratory Medicine. 2008;46:1289–1295. doi: 10.1515/CCLM.2008.242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodward DF, Liang Y, Krauss AH. Prostamides (prostaglandin-ethanolamides) and their pharmacology. British Journal of Pharmacology. 2008;153:410–419. doi: 10.1038/sj.bjp.0707434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zvonok N, Pandarinathan L, Williams J, Johnston M, Karageorgos I, Janero DR, Krishnan SC, Makriyannis A. Covalent inhibitors of human monoacylglycerol lipase: Ligand-assisted characterization of the catalytic site by mass spectrometry and mutational analysis. Chemistry & Biology. 2008a;15:854–862. doi: 10.1016/j.chembiol.2008.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zvonok N, Williams J, Johnston M, Pandarinathan L, Janero DR, Li L, Krishnan SC, Makriyannis A. Full mass spectrometric characterization of human monoacylglycerol lipase generated by large-scale expression and single-step purification. Journal of Proteome Research. 2008;7:2158–2164. doi: 10.1021/pr700839z. [DOI] [PMC free article] [PubMed] [Google Scholar]