Abstract
Itk and Txk/Rlk are Tec family kinases expressed in T cells. Itk is expressed in both TH1 and TH2 cells. By contrast, Txk is preferentially expressed in TH1 cells. Although Itk is required for TH2 responses in vivo and Txk is suggested to regulate IFNγ expression and TH1 responses, it remains unclear whether these kinases have distinct roles in TH cell differentiation/function. We demonstrate here that Txk null CD4+ T cells are capable of producing both TH1 and TH2 cytokines similar to those produced by WT CD4+ T cells. To further examine whether Itk and Txk play distinct roles in TH cell differentiation and function we examined Itk-null mice carrying a transgene that expresses Txk at levels similar to the expression of Itk in TH2 cells. Using two TH2 model systems: allergic asthma and Schistosome egg-induced lung granulomas, we found that the Txk transgene rescued TH2 cytokine production and all TH2 symptoms without notable enhancement of IFNγ expression. These results suggest that Txk is not a specific regulator of TH1 responses. Importantly, they suggest that Itk and Txk exert their effects on TH cell differentiation/function at the level of expression.
Introduction
Txk/Rlk (hereafter referred to as Txk) and Itk are distantly related members of the Tec family of tyrosine kinases that are involved in signaling downstream from the TCR. While Txk has a palmitoylation site instead of a PH domain that allows it to be constitutively associated with lipid raft membrane fractions, Itk requires the activation of PI3 kinase for recruitment to the membrane via its PH domain (1, 2). Mutations affecting Itk in mice lead to altered T cell development and mature T cell function with reduced TCR-induced proliferation and impaired IL-2 production in vitro ((3-6) see (7) for review)). One of the most dramatic phenotype of Itk−/− mice is their defect in TH2 responses in vivo. Itk−/− mice are incapable of developing allergic asthma and have decreased responses to challenge with a number of TH2-inducing parasites including the eggs of Schistosome mansoni or the worm Nippostrongyloides brasilienses (8-11). Indeed, in some cases, Itk-deficient mice have been found to mount TH1 responses to TH2-inducing pathogens. In contrast, overexpression of Txk has been associated with increased expression of IFN-γ, a TH1 cytokine (12-15). Txk has been found to bind directly to a sequence in the IFN-γ promoter, suggesting a direct role for Txk in driving IFN-γ transcription and TH1 responses (16). Together, these data suggested that Itk and Txk have distinct roles in TH2 and TH1 differentiation or function respectively.
Nonetheless, the exact mechanism by which the Tec kinases influence T helper cell differentiation remains controversial. While some data suggest that Itk induces TH2 differentiation by suppressing the expression of T-bet (17), other reports propose that Itk but not Txk directly interacts with and tyrosine phosphorylates T-bet, promoting its interaction with GATA3, which suppresses the latter’s activity (18). It has also been proposed that Itk may modulate TH2 differentiation by virtue of its expression: Itk is expressed at higher levels than Txk in naïve T cells and while both Itk and Txk are expressed in TH1 cells, Txk is downregulated in TH2 cells, leaving Itk as the major Tec kinase (17). Consistent with this idea, recent data argue that Itk is not required for TH2 differentiation per se, but rather is required for effector function of differentiated TH2 cells (19). These data suggest it may not be the intrinsic function of these kinases, but rather their patterns of expression that determines their roles in T helper cell differentiation and function.
To evaluate these questions, we examined whether forced over-expression of Txk in T cells could rescue TH2 responses in Itk−/− mice using transgenic mice that drive expression of Txk at levels similar to Itk in TH2 cells. If these kinases have distinct TH1 and TH2-inducing properties, one would predict that overexpression of Txk would preferentially drive TH1 responses and TH2 defects in Itk−/− mice may be exacerbated. However, if defects in TH2 responses in Itk−/− mice are secondary to the low levels of expression of Tec kinases in TH2 cells in the absence of Itk, expression of the Txk transgene may rescue these responses. Utilizing two systems: 1) a murine model of allergic asthma and 2) challenge with the eggs of Schistosome mansoni, a strong TH2 inducing parasite (8, 10), we demonstrate that Itk−/− mice expressing a Txk transgene can rescue TH2 responses, with no evidence of overexpression of TH1 cytokines. Our results thus strongly suggest that the effects of Itk and Txk on T helper cell function may result from the differential patterns of expression of these kinases.
Materials and Methods
Mice
Wild-type (WT), Txk−/− (20) Itk−/− (6) and Tg(CD2-Txk) (13), were used between 6-12 week of age and were backcrossed on the C57BL/6 background for at least 5 generations. Tg(CD2-Txk)Itk−/− mice were generated by breeding Itk−/− and Tg(CD2-Txk) mice. All experiments were approved by the Office of Research Protection’s Institutional Animal Care and Use Committee at Pennsylvania State University and the NIH.
Flow cytometry
Single cell preparations were made from thymi and spleens. Red blood cells were lysed from spleens using ammonium chloride lysis solution. Cells were preincubated with anti-CD16, then stained with combinations of: anti-CD8α (Clone 53-6.7, FITC or PE), CD62L (MEL-14, PE), CD4 (RM4-5, PerCP-Cy5.5), CD44 (IM7, APC), CD24 (M1/69, FITC) (eBioscience, San Diego, CA or BD Biosciences/Pharmingen, San Jose, CA).
Allergic asthma induction
Mice were sensitized with ovalbumin (Sigma-Aldrich) complexed to aluminum hydroxide (10 μg ovalbumin/1 mg alum; Pierce) intraperitoneally in a total volume of 200 μl on days 0 and 5. Mice were later challenged intranasally with ovalbumin from days 12 through 15 (at a concentration of 2 mg/ml, for a total of 40 μg total exposure). Development of allergic asthma was measured by analyzing AHR on day 16 using a custom made mechanical ventilator as previously described (9, 10, 21). Mice were then sacrificed, BALF obtained and lungs sectioned and stained using H&E or PAS as detailed (9, 10).
Schistosome egg injection and analysis of response
Mice were primed and challenged with S. mansoni eggs as described using frozen eggs for primary injections and fresh eggs for secondary injections 10 days later (8). Nine to 10 days after secondary challenge, lungs were collected and processed as described for analysis of granulomas (8).
Quantitative Real-time RT-PCR analysis
After analysis of AHR, RNA was extracted from the lungs of mice using Trizol reagent (Invitrogen Life Technologies, Carlsbad, CA). cDNA was generated with a kit from Amersham Biosciences (Piscataway, NJ). Quantitative RT-PCR was then performed for IL-4, IL-13, IFN-γ, CCL-7 and CCL-11, with GAPDH used as a housekeeping gene and the data expressed as 2−ΔΔCT (9). To quantify the expression level of Itk and Txk, Quantitative RT-PCR was performed on cDNA from native CD4+ T cells or CD4+ T cells differentiated to TH2 cells. Signals were compared to standards generated from plasmids carrying the cDNA for Itk and Txk.
Analysis of calcium responses
CD4+ T cells were purified from spleens by negative selection using MACS mouse CD4+ T cell isolation kit microbeads (Miltenyi, Auburn, CA), with the addition anti-NK1.1(BD Biosciences/Pharmingen, San Jose, CA). Cells were labeled with 5 μg/ml Fluo3 and 5 μg/ml Fura-red (Molecular Probes/Invitrogen, Carlsbad, CA). Cells were kept at room temperature, then stimulated at 37°C with 15 μg/ml biotinylated anti-CD3 (2C11) with the addition of 8 μg/ml streptavidin. Data were collected on a FacsCalibur for 10 minutes and analyzed using FlowJo software (TreeStar, Ashland, OR).
Analysis of lymphocyte proliferation and cytokine secretion in response to antigen challenge
Purified splenocytes obtained from ovalbumin challenged mice were cultured with 10 and 100 μg/ml of OVA (2 × 105 cells/well). After 72 hours of culture, cells were pulsed with [3H]-thymidine for 18 h and incorporated radioactivity determined. Cytokine secretion was analyzed by stimulating splenocytes at 2 × 106/ml with 100 μg/ml OVA for 96 h following which supernatants were harvested. Cytokine concentration in supernatants and BAL were determined using a Luminex system (Biorad, Austin, TX), with plates from Lincoplex (Millipore Systems, Billerica, MA). In mice exposed to S. mansoni eggs, cells from draining mediastinal lymph nodes and spleens were collected and restimulated with SEA as described (8). Three days later, cells were stimulated with PMA and Ionomycin in the presence of Golgi-stop (BD Biosciences/Pharmingen, San Jose, CA), stained for CD4 and intracellular IL-4, -5, -10 and IFN-γ using specific antibodies and analyzed by flow cytometry.
In vitro differentiation to TH1 and TH2 cells
T cells were purified by T cell isolation columns (R&D, Minneapolis, MN) and then sorted for CD4+CD62Lhi and CD44lo naïve cells. Cells were stimulated with 1 μg/ml anti-CD3 plus anti-CD28 in the presence of mitomycin-treated T-depleted splenocytes for 2-3 days, then restimulated with PMA and Ionomycin in the presence of golgi-plug, stained for intracellular levels of IFN-γ and IL-4 and analyzed by flow cytometry as above. TH0 conditions contained no extra cytokines, TH1 conditions included 40 ng/ml IL-12 and 10 μg/ml anti-IL-4; TH2 conditions included 40 ng/ml IL-4 and 10 μg/ml anti-IL-12 and anti-IFN-γ. Cytokines were obtained from Peprotech (Rocky Hill, NJ).
Data analysis
Statistical evaluation was conducted by using the Student’s t test with a probability value of p<0.05 considered statistically significant.
Results
Txk null T cells are not defective in TH1 or TH2 cytokine secretion
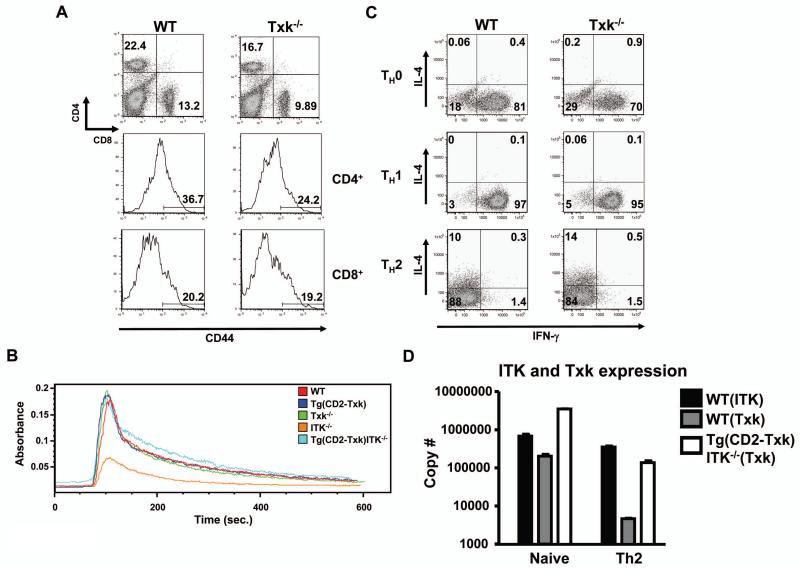
Txk has been suggested to specifically regulate the production of IFN-γ in TH1 cells. To determine if T cells lacking Txk have defects in this function, we analyzed T cells from Txk null mice (20). We first confirmed that Txk null mice had similar T cell subpopulations to WT mice (Fig. 1a (20)). Consistent with these observations, Txk−/− CD4+ T cells showed normal TCR-induced Ca2+ mobilization (Fig. 1b), supporting the conclusion that at this level of analysis, T cells from Txk null mice exhibit no significant abnormalities. We next analyzed naïve CD4+ T cells that were differentiated under TH1 or TH2 conditions (Fig. 1c). Txk null T cells were able express both TH1 and TH2 cytokines similar to WT T cells. Thus, although it has been proposed that Txk is a TH1-inducing kinase, these data support the conclusion that Txk is not required for the generation of IFN-γ in TH1 cells.
Figure 1. Txk null T cells do not have defects in TH1 or TH2 cytokine secretion.
(a) Splenocytes from WT or Txk null mice were analyzed for the indicated markers by flow cytometry. Top panels: whole splenocytes; Middle panels: Gated CD4+ T cells analyzed for CD44; Bottom panels: Gated CD8+ T cells analyzed for CD44. (b) CD4+ T cells from WT, Itk null, Txk null, Tg(CD2-Txk) or Tg(CD2-Txk)Itk−/− mice were analyzed for calcium response following stimulation with anti-CD3 antibodies as indicated. (c) Naïve CD4+ T cells from WT or Txk null mice were differentiated under the indicated conditions, then restimulated and analyzed for expression of the indicated cytokines by intracellular staining and flow cytometry. Data are gated on CD4+ T cells. (d) qRT-PCR of mRNA for Txk or Itk from fresh naïve CD4+ T cells from WT or Tg(CD2-Txk)Itk−/− mice. Alternatively, these cells were differentiated under TH2 conditions prior to qRT-PCR.
The CD2 promoter driven Txk transgene is expressed in TH2 cells at similar levels to endogenous Itk
Although the above results suggest that Txk is not required for IFN-γ expression, Txk is expressed at lower levels than Itk (Fig. 1d), and may have unique functions that may not be obvious due to its low level of expression. To evaluate whether Txk can compensate for Itk if expressed at higher levels, we utilized a transgenic mouse model in which Txk was overexpressed using the CD2 promoter (Tg(CD2-Txk)). When crossed onto an Itk null background (referred to here as Tg(CD2-Txk)Itk−/−), expression of Txk was able to rescue the known calcium signaling defect downstream of TCR stimulation that occurs in T cells lacking Itk (Fig. 1b), similar to what has been reported in the thymus (13). However, although expression of this Txk transgene has been shown to improve positive selection in Itk-deficient mice, Tg(CD2-Txk)Itk−/− mice still had low numbers of peripheral T cells, even lower than Itk-deficient mice (Supplemental Table 1).
To further evaluate the Txk transgene, we used qRT-PCR to evaluate the relative expression levels of Txk and Itk mRNA in freshly isolated CD4+ T cells and in cells that were differentiated under TH2-inducing conditions. Although the expression of Txk in freshly isolated CD4+ T cells was higher (17-fold) than endogenous Txk in WT mice, it was only 5-fold higher than the level of expression of Itk mRNA as measured by qRT-PCR (Fig. 1d). As previously reported, endogenous Txk expression in WT TH2 cells was dramatically reduced in TH2 cells (17). However, in TH2 cells from Tg(CD2-Txk)Itk−/− mice, Txk expression remained high, similar to levels of Itk in WT TH2 cells (Fig. 1d). Thus, in TH2 cells Txk expression was similar to Itk expression on a copy number basis. We therefore utilized this transgene on the Itk−/− background to evaluate the ability of Txk to complement Itk function in TH2 cells.
Rescue of allergic airway inflammation and airway hyperresponsiveness (AHR) in mice lacking Itk by expression of Txk transgene
To determine the effect of expression of Txk in Th2 cells, we analyzed the development of allergic asthma, a disease dependent on TH2 cells and cytokines. OVA immunized and challenged WT and Itk−/− mice, as well as mice expressing the Txk transgene on an Itk−/− background (Tg(CD2-Txk)Itk−/−) were evaluated for airway resistance in response to methacholine challenge as a measure of AHR. In these experiments, while WT mice developed significant airway resistance, Itk−/− mice responded poorly as previously reported (Fig. 2a) (9). However, Tg(CD2-Txk)Itk−/− transgenic mice exhibited significant levels of airway resistance, similar to the WT mice (Fig. 2a).
Figure 2. Rescue of AHR in Itk-null mice by expression of the related kinase Txk.
(a) WT, Itk-null or Tg(CD2-Txk)Itk−/− mice were immunized and challenged intranasally with OVA, followed by analysis of AHR by mechanical ventilation. Filled circles, WT; open circles Itk−/−; Filled triangles, Tg(CD2-Txk)Itk−/− mice. O/O indicates mice that have been immunized with OVA, then challenged intranasally with OVA. Differences are statistically significant between WT and Itk−/− or Tg(CD2-Txk)Itk−/− and Itk−/− (p<0.05) but not significant between WT and Tg(CD2-Txk)Itk−/− (n=5-7 mice per group, representative of three experiments). (b) Mice were immunized and challenged intranasally with OVA, followed by analysis of lung sections by H&E (top panels) or PAS staining (bottom panels), representative of three experiments.
We next analyzed airway inflammation and mucous production, factors that can contribute to the development of allergic asthma in this model. Histological evaluation of lung sections revealed that Tg(CD2-Txk)Itk−/− mice exhibit massive leukocyte infiltration in the lung, which was similar or higher than that observed in WT mice (Fig. 2b). In contrast, Itk−/− mice showed reduced leukocyte infiltration, as previously reported (9, 10). Increased thickening of the epithelial cell lining of the bronchioles and mucous production by airway goblet cells was also observed in Tg(CD2-Txk)Itk−/− mice, similar to that seen in WT mice (Fig. 2b).
Expression of Txk transgene in Itk-null mice enhances production of TH2 cytokines in response to allergic inflammation
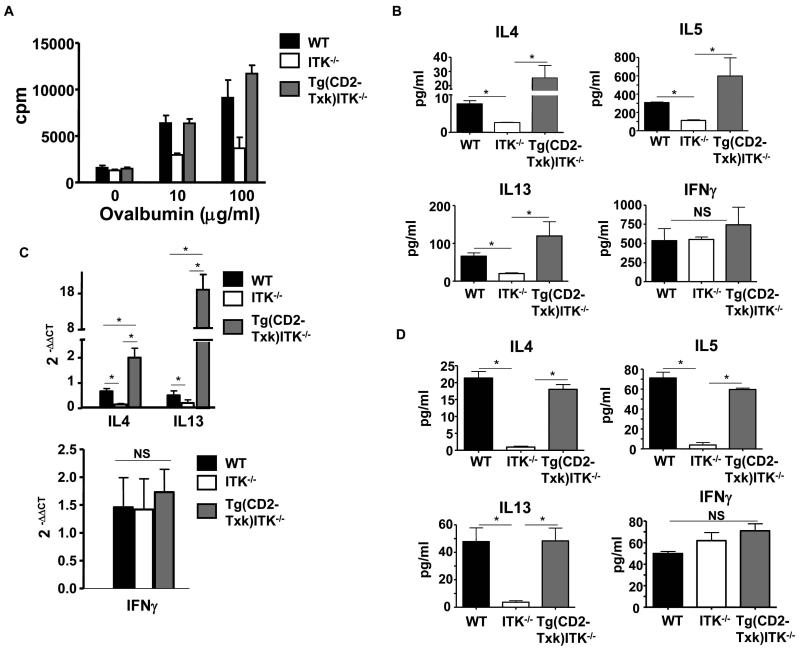
TH2 specific cytokines such as IL-4, IL-5 and IL-13 are involved in inducing allergic airway inflammation (22, 23). To examine whether expression of Txk transgene rescued production of TH2 cytokines, we first analyzed cytokine production from splenic T cells of mice immunized and challenged with OVA. Stimulation with OVA in vitro induced proliferation of T cells from WT, Tg(CD2-Txk)Itk−/− and Itk−/− OVA-challenged mice, although those from Itk−/− mice exhibited reduced proliferation in comparison to WT mice (Fig. 3a) (10). Splenocytes from Tg(CD2-Txk)Itk−/− mice, however, had proliferative responses equivalent to WT mice, and the Txk transgene rescued IL-4, IL-5 and IL-13 secretion from these cells in vitro (Fig. 3b). Since Txk is suggested to regulate the expression of TH1 cytokines such as IFN-γ, we also examined the expression of IFN-γ. Strikingly, Tg(CD2-Txk)Itk−/− mice did not secrete elevated levels of IFN-γ as would be expected if it specifically regulated IFNγ (Fig. 3b).
Figure 3. Txk-mediated rescue of cytokine production in vitro and in vivo.
(a) Splenocytes from WT, Itk-null or Tg(CD2-Txk)Itk−/− mice treated as in figure 2 were incubated in vitro with the indicated concentration of OVA, and analyzed for proliferative responses after 96 h of culture. Data are the mean ± SEM of triplicate cultures and are representative of three experiments, *p<0.05. (b) Splenocytes were treated as in (a) with 100 μg/ml OVA and supernatants analyzed for IL-4, -5 and -13 and IFN-γ after 72 h of culture. Data are the mean ± SEM of triplicate cultures and are representative of three experiments, *p<0.05, NS, Not significant. (c) Lungs from WT, Itk−/− or Tg(CD2-Txk)Itk−/− mice treated as in figure 2 were analyzed by qRT-PCR for IL-4, IL-13 and IFN-γ. n = 6, *p<0.05, NS, not statistically significant. (d) BAL from lungs of mice treated as in figure 2 were analyzed for IL-4, -5, -13 and IFNγ. n = 6, *p<0.05, NS, not statistically significant.
To further examine the expression level of TH2 cytokines in the lungs upon induction of allergic asthma, we measured the expression level of IL-4 and IL-13 mRNA in the lungs of OVA immunized and challenged WT, Itk−/− and Tg(CD2-Txk)Itk−/− mice using real-time quantitative RT-PCR. As shown in Figure 3c, there was enhanced expression of IL-4 and IL-13 mRNA in the Tg(CD2-Txk)Itk−/− mice in comparison to WT and Itk−/− mice. By contrast, the mRNA levels of IFN-γ were similar in all three strains of mice (Fig. 3c). These findings were further confirmed by the analysis of BAL fluid for the level of TH2 cytokines in these mice, indicating that expression of Txk rescued the TH2 mediated inflammation in the lungs of OVA challenged mice (Fig. 3d). Therefore the Txk transgene does not appear to lead to enhanced TH1 differentiation or IFN-γ production, nor does it block TH2 differentiation as would be observed if Txk were a TH1-inducing kinase (12-16). Higher levels of IL-13 mRNA but not protein were also observed in the Tg(CD2-Txk)Itk−/− mice in comparison to WT. These results could be due to IL-13 mRNA expression in other inflammatory cells such as eosinophils and mast cells, recruited to the lungs in response to the disease. Nonetheless, we did not observe higher levels of the IL-13 protein, suggesting this message may not be fully translated in these cells. Altogether, these data indicate that in the absence of Itk, Txk expression can lead to the generation of a TH2 response both systemically and in the lungs.
Rescue of CD4+ T cell recruitment in Itk-null mice expressing Txk transgene
Analysis of CD4+ T cell numbers in the lungs revealed that while WT mice could recruit these cells into the lung during airway inflammation, as previously reported, Itk-null mice could not (10, 24). However, Tg(CD2-Txk)Itk−/− mice had similar numbers of CD4+ T cells in the lungs compared to WT mice, indicating that expression of Txk was able to rescue migration and recruitment of T cells into the lung (Fig. 4). These results were not secondary to increased numbers of CD4+ T cells in Tg(CD2-Txk)Itk−/− mice as these mice have even lower numbers of mature CD4+ T cells as Itk−/− mice (Supplemental Table 1). These data thus confirm that in vivo, Txk can rescue specific functions of Itk that lead to the recruitment of leukocytes into the lungs during the development of allergic asthma.
Figure 4. Rescue of CD4+ T cell recruitment into the lungs of Itk-null mice by expression of the related kinase Txk.
BAL fluid from lungs from WT, Itk−/− or Tg(CD2-Txk)Itk−/− mice treated as in figure 1 were analyzed for the number of CD4+ T cells by flow cytometry. *p<0.05, n = 6, NS, not statistically significant.
The Txk transgene rescues TH2 responses to Schistosome mansoni eggs
To examine induction of TH2-mediated responses using a different model, we analyzed responses to i.v. injection of S. mansoni eggs. Injection of S. mansoni eggs i.v. into mice results in the formation of eosinophilic granulomas in the lung in a TH2 cell dependent manner. In Itk−/− mice, the size of these granulomas is reduced (Fig. 5a) (8). However, consistent with our observations in the OVA induced model, injection of S. mansoni eggs into Tg(CD2-Txk)Itk−/− mice resulted in granulomas of similar size to those seen in WT mice (Fig. 5a). In contrast, overexpression of Txk on a WT background also did not increase granuloma size (Fig. 5a). To evaluate TH2 cytokine production in this model, we examined cytokine production by intracellular staining of cells from the spleen and draining mediastinal nodes of mice injected with S. mansoni eggs. Analyses of splenic and lymph node T cells restimulated in vitro with Schistosome egg antigen (SEA) confirmed that Itk-null mice had fewer cells producing the TH2 cytokines IL-4, -5 and -10. However, Itk−/− mice carrying the Txk transgene had similar numbers of cells making these cytokines to that seen in WT mice, confirming that TH2 differentiation was rescued. Again, Tg(CD2-Txk)Itk−/− mice did not show elevated numbers of IFN-γ producing cells (Fig. 5b,c). These mice also did not show increased percentages of cells expressing TH2 cytokines compared to WT cells, demonstrating that the cells were not merely over-responsive due to the expression of the Txk transgene. Similarly, overexpression of Txk on a WT background did not increase the level of TH1 or TH2 cytokines (Fig. 5b,c), again arguing that expression of the Txk transgene did not merely result in increased activation of these cells. These data are in keeping with the finding that T cells from these mice do not exhibit higher levels of calcium influx in response to TCR stimulation (see Fig. 1). Thus, expression of Txk transgene can rescue patterns of TH2 cytokine production in response to multiple TH2 inducing agents.
Figure 5. Rescue of S. mansoni induced lung granulomas in Itk-null mice by Txk.
(a) WT, Itk-null, Tg(CD2-Txk) or Tg(CD2-Txk)Itk−/− mice were injected i.p., then challenged i.v. with S. mansoni eggs, followed by analysis of lung sections by H&E (top panel). Granuloma size was determined and plotted (bottom panel). Data are representative of two experiments examining 8-9 mice per genotype, *p<0.05. (b) Cells collected from the draining LN, or (c) spleens of mice treated as in Figure 5a were restimulated with SEA, fixed and analyzed for intracellular IL-4, -5, -10 and IFN-γ. Data are representative of two experiments each consisting 4-6 mice per genotype.
Discussion
The Tec kinases Itk and Txk are both expressed in T cells and regulate their development, activation and function (7). A role for Itk has been demonstrated for the production of Th2 cytokines, while roles for Txk are less clear (8-11, 17, 19). Prior work suggests that Txk regulates IFN-γ production and thus TH1 development, however a specific role of Txk in the regulation of TH1 specific cytokine production is still unresolved (12-16). Our data argue that Txk does not specifically drive expression of this TH1 cytokine. First, Txk null T cells do not exhibit defects in producing IFN-γ, arguing that TXK is not essential for IFN-γ production. More importantly, in the models examined here, overexpression of Txk did not induce elevated levels of IFN-γ and prevent the development of TH2 cell mediated disease. Instead, overexpression of Txk rescued production of TH2 type cytokines including IL-4, -5 and 13 in the lungs of Tg(CD2-Txk)Itk−/− mice in response to TH2-inducing agents. It should be noted that these observations do not rule out the possibility that Txk could indirectly regulate IFN-γ production via interaction with other factors that are specifically expressed in TH1 cells.
Txk is the most distantly related family member of the Tec family of tyrosine kinases. This protein has a N-terminal palmitoylation site instead of a PH domain found in Itk and other Tec kinases, allowing it to be anchored constitutively in the plasma membrane. Thus Txk, unlike Itk, does not require the activation of PI3 kinase in order to be recruited to the plasma membrane (1, 2). This would suggest specific and unique functions for Txk. However, we show that when expressed at similar levels to Itk, Txk can functionally replace Itk for the induction of predominant TH2 responses in vivo by enhancing the expression of TH2 specific cytokines. Our data provide strong evidence that Txk can function to rescue TH2 responses in the absence of Itk, including AHR, airway inflammation, granuloma formation, T cell recruitment and cytokine production in the lungs in vivo as well as TH2 cytokine production from T cells restimulated in vitro. Our data thus support the idea that these kinases have overlapping functions.
Although Txk is normally expressed at very low levels in TH2 cells, our observations with these transgenic mice nonetheless help provide a better understanding of the specific functions of Txk and Itk which would have been otherwise difficult to address. In addition, these observations provided greater insight about the relevance of selective expression of Txk and Itk in TH1 and TH2 cells respectively. Our work also confirms previous studies by Berg and colleagues, as well as Luban and colleagues that show that, Txk expression is specifically downregulated in TH2 cells (17, 25). In contrast, these groups observed similar levels of Itk expression in TH1 as well as TH2 cells. Thus negative regulation of Txk expression may occur in a TH2 specific manner, perhaps via negative regulation by TH2 specific transcription factors. Alternatively, Itk’s expression may be specifically maintained by TH2 specific transcription factors. In either case, this expression pattern could result in a critical dependence of TH2 cells on the function of Itk. Our work here lends support to this model since expression of Txk using the T cell specific CD2 promoter would allow for continuous expression of Txk, even in differentiating TH2 cells, and thus provide crucial Tec kinase signals needed for functional responses in the absence of Itk. Hence, our observations suggest that the selective expression of Txk and Itk in TH1 and TH2 cells respectively may provide the delicate balance of signals required for inducing or maintaining different types of T cell responses. It should be noted that although the CD2-Txk transgene has been reported to improve positive selection in Itk−/− thymocytes, we find that it does not rescue peripheral T cell numbers (perhaps because the low T cell numbers are secondary to earlier defects in Itk−/− thymocytes (26). These observations suggest that the rescue of TH2 responses are not merely the result of increased CD4+ T cell numbers, although we cannot rule out other effects on development (3, 5, 27, 28)). However, the normal levels of TCR-induced Ca2+ influx in Tg(CD2-Txk) and Tg(CD2-Txk)Itk−/− peripheral T cells also argues that our results are not the result of non-specific hyperactivation of cells expressing the Txk transgene.
In previous studies, we have found that while the absence of Itk leads to defective TH2 responses, absence of both Txk and Itk surprisingly leads to normal TH2 responses in Rlk−/−Itk−/− mice (8). Although these results may appear to contradict our current findings, it is possible that these findings result from the fact that CD4+ T cells lacking both Txk and Itk maintain high levels of GATA3 following stimulation compared to WT and Itk null mice. These results suggested that a defect in GATA3 downregulation may lead to a propensity to develop into TH2 cells in T cells from the Txk/Itk double knockout mice, perhaps because this was the only type of response that could occur. Our data presented here indicates that expression of Txk at similar levels as Itk can lead to normalized TH2 responses in Itk-deficient mice, and that potential redundancy in function between Txk and Itk may explain the TH2 specific response observed in Tg(CD2-Txk)Itk−/− mice.
Our data are also in support of recent data from Fowell and colleagues, who demonstrated that Itk deficient T cells can differentiate into TH2 cells, however, they cannot elaborate and secrete TH2 cytokines upon restimulation (19). The enforced expression of Txk in these TH2 cells allows for functional rescue of this event. Given previous results suggesting that rescue of calcium signaling can rescue TH2 cytokine production in Itk−/− CD4+ T cells (11, 19), it is likely that rescue of Ca2+ mobilization by expression of the Txk transgene contributes to our observations. Thus, Txk may be able to rescue TH2 responses by restoration of intracellular Ca2+ increases that are defective in Itk−/− T cells, perhaps by participating in the Slp76/GADS/LAT complex, which regulate PLCγ1 activation. Indeed, expression of the Txk transgene has also been shown to be able to rescue PLCγ1 tyrosine phosphorylation in Itk−/− double positive thymocytes (13).
Other cell types such as mast cells and eosinophils have also been suggested to play an important role in the development of TH2 specific responses (29-31). These cells are capable of producing TH2 cytokines and chemokines that can induce initial immune responses and mediate further activation and migration of TH2 cells to the lungs thereby exacerbating the immune responses (32). However, since the CD2 promoter cassette that drives Txk expression in these transgenic mice is primarily active in T cells, (including NKT cells), these findings suggest that the defect in Itk-null mice in developing effective TH2 responses in our models is unlikely to be due to the lack of expression of Itk in other cell types such as mast cells (33).
In recent years there has been a growing interest in the Tec kinases Itk and Txk as potential therapeutic targets for TH2 and TH1-mediated diseases, respectively. Our data here support the model where the Tec kinases, Txk and Itk regulate T helper cell mediated responses via their differential expression in TH1 and TH2 cells respectively and not due to intrinsic functional differences as previously suggested (12-16). Our data also suggest that the defective TH2 response in the absence of Itk is due to reduced Tec kinase signals, and that either Itk or Txk can fulfill this role if expressed at high enough levels. Overall, these findings provide novel insight into the role of Tec kinases Txk and Itk in the regulation of T helper cell differentiation/function and disease.
Supplementary Material
Acknowledgements
We thank members of the Center for Molecular Immunology & Infectious Disease at Penn State for helpful comments. E. Kunze, N. Bem, S. Magargee, Dr. D. Grove and M. Potter for technical assistance.
Abbreviations
- AHR
Airway Hyperresponsiveness
- BAL
Bronchoalveolar Lavage
- PH
Pleckstrin Homology
- PI3 Kinase
Phosphoinositide-3-kinase
- SEA
Schistosome Egg Antigen
- TCR
T cell Receptor
Footnotes
Supported by NIH grants AI051626 & AI065566, the AHA (AA) and by the intramural program of the National Institutes of Health, National Human Genome Research Institute (AMV, JLC, JGR, and PLS), the National Institute of Allergy and Infectious Disease (DJ and AS) and the National Institute of Child Health and Development (CS and PL).. NS thanks the American Academy of Allergy, Asthma and Immunology’s Strategic Training in Allergy Research (ST*AR) Award for support.
References cited
- 1.August A, Sadra A, Dupont B, Hanafusa H. Src-induced activation of inducible T cell kinase (ITK) requires phosphatidylinositol 3-kinase activity and the Pleckstrin homology domain of inducible T cell kinase. Proc Natl Acad Sci USA. 1997;94:11227–11232. doi: 10.1073/pnas.94.21.11227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Debnath J, Chamorro M, Czar M, Schaeffer E, Lenardo M, Varmus H, Schwartzberg P. rlk/TXK encodes two forms of a novel cysteine string tyrosine kinase activated by Src family kinases. Mol Cell Biol. 1999;19:1498–1507. doi: 10.1128/mcb.19.2.1498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Broussard C, Fleischecker C, Horai R, Chetana M, Venegas A, Sharp L, Hedrick S, Fowlkes B, Schwartzberg P. Altered development of CD8+ T cell lineages in mice deficient for the tec kinases Itk and Rlk. Immunity. 2006;25:93–104. doi: 10.1016/j.immuni.2006.05.011. [DOI] [PubMed] [Google Scholar]
- 4.Schaeffer EM, Broussard C, Debnath J, Anderson S, McVicar DW, Schwartzberg PL. Tec Family Kinases Modulate Thresholds for Thymocyte Development and Selection. J. Exp. Med. 2000;192:987–1000. doi: 10.1084/jem.192.7.987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Atherly L, Lucas J, Felices M, Yin C, Reiner S, Berg L. The Tec family tyrosine kinases Itk and Rlk regulate the development of conventional CD8+ T cells. Immunity. 2006;25:79–91. doi: 10.1016/j.immuni.2006.05.012. [DOI] [PubMed] [Google Scholar]
- 6.Liao X, Littman D. Altered T cell receptor signaling and disrupted T cell development in mice lacking Itk. Immunity. 1995;3:757–769. doi: 10.1016/1074-7613(95)90065-9. [DOI] [PubMed] [Google Scholar]
- 7.Berg L, Finkelstein L, Lucas J, Schwartzberg P. Tec family kinases in T lymphocyte development and function. Annu Rev Immunol. 2005;23:549–600. doi: 10.1146/annurev.immunol.22.012703.104743. [DOI] [PubMed] [Google Scholar]
- 8.Schaeffer E, Yap G, Lewis C, Czar M, McVicar D, Cheever A, Sher A, Schwartzberg P. Mutation of Tec family kinases alters T helper cell differentiation. Nat Immunol. 2001;2:1183–1188. doi: 10.1038/ni734. [DOI] [PubMed] [Google Scholar]
- 9.Ferrara T, Mueller C, Sahu N, Ben-Jebria A, August A. Reduced airway hyperresponsiveness and tracheal responses during allergic asthma in mice lacking tyrosine kinase inducible T-cell kinase. J Allergy Clin Immunol. 2006;117:780–786. doi: 10.1016/j.jaci.2005.12.1330. [DOI] [PubMed] [Google Scholar]
- 10.Mueller C, August A. Attenuation of Immunological Symptoms of Allergic Asthma in Mice Lacking the Tyrosine Kinase ITK. J Immunol. 2003;170:5056–5063. doi: 10.4049/jimmunol.170.10.5056. [DOI] [PubMed] [Google Scholar]
- 11.Fowell D, Shinkai K, Liao X, Beebe A, Coffman R, Littman D, Locksley R. Impaired NFATc Translocation and Failure of Th2 Development in Itk Deficient CD4 T Cells. Immunity. 1999;11:399–409. doi: 10.1016/s1074-7613(00)80115-6. [DOI] [PubMed] [Google Scholar]
- 12.Suzuki N, Nara K, Suzuki T. Skewed Th1 responses caused by excessive expression of Txk, a member of the Tec family of tyrosine kinases, in patients with Behcet’s disease. Clin Med Res. 2006;4:147–151. doi: 10.3121/cmr.4.2.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sommers C, Rabin R, Grinberg A, Tsay H, Farber J, Love P. A role for the Tec family tyrosine kinase Txk in T cell activation and thymocyte selection. J Exp Med. 1999;190:1427–1438. doi: 10.1084/jem.190.10.1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nagafuchi H, Takeno M, Yoshikawa H, Kurokawa M, Nara K, Takada E, Masuda C, Mizoguchi M, Suzuki N. Excessive expression of Txk, a member of the Tec family of tyrosine kinases, contributes to excessive Th1 cytokine production by T lymphocytes in patients with Behcet’s disease. Clin Exp Immunol. 2005;139:363–370. doi: 10.1111/j.1365-2249.2004.02688.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Takeno M, Yoshikawa H, Kurokawa M, Takeba Y, Kashiwakura J, Sakaguchi M, Yasueda H, Suzuki N. Th1-dominant shift of T cell cytokine production, and subsequent reduction of serum immunoglobulin E response by administration in vivo of plasmid expressing Txk/Rlk, a member of Tec family tyrosine kinases, in a mouse model. Clin Exp Allergy. 2004;34:965–970. doi: 10.1111/j.1365-2222.2004.01981.x. [DOI] [PubMed] [Google Scholar]
- 16.Takeba Y, Nagafuchi H, Takeno M, Kashiwakura J, Suzuki N. Txk, a member of nonreceptor tyrosine kinase of Tec family, acts as a Th1 cell-specific transcription factor and regulates IFN-gamma gene transcription. J Immunol. 2002;168:2365–2370. doi: 10.4049/jimmunol.168.5.2365. [DOI] [PubMed] [Google Scholar]
- 17.Miller A, Wilcox H, Lai Z, Berg L. Signaling through Itk Promotes T Helper 2 Differentiation via Negative Regulation of T-bet. Immunity. 2004;21:67–80. doi: 10.1016/j.immuni.2004.06.009. [DOI] [PubMed] [Google Scholar]
- 18.Hwang E, Szabo S, Schwartzberg P, Glimcher L. T helper cell fate specified by kinase-mediated interaction of T-bet with GATA-3. Science. 2005;307:430–433. doi: 10.1126/science.1103336. [DOI] [PubMed] [Google Scholar]
- 19.Au-Yeung B, Katzman S, Fowell D. Cutting edge: Itk-dependent signals required for CD4+ T cells to exert, but not gain, Th2 effector function. J Immunol. 2006;176:3895–3899. doi: 10.4049/jimmunol.176.7.3895. [DOI] [PubMed] [Google Scholar]
- 20.Schaeffer E, Debnath J, Yap G, McVicar D, Liao X, Littman D, Sher A, Varmus H, Lenardo M, Schwartzberg P. Requirement for Tec kinases Rlk and Itk in T cell receptor signaling and immunity. Science. 1999;284:638–641. doi: 10.1126/science.284.5414.638. [DOI] [PubMed] [Google Scholar]
- 21.Ewart S, Levitt R, Mitzner W. Respiratory system mechanics in mice measured by end-inflation occlusion. J Appl Physiol. 1995;79:560–566. doi: 10.1152/jappl.1995.79.2.560. [DOI] [PubMed] [Google Scholar]
- 22.Wills-Karp M, Luyimbazi J, Xu X, Schofield B, Neben T, Karp C, Donaldson D. Interleukin-13: central mediator of allergic asthma. Science. 1998;282:2258–2261. doi: 10.1126/science.282.5397.2258. [DOI] [PubMed] [Google Scholar]
- 23.Cohn L, Elias J, Chupp G. Asthma: mechanisms of disease persistence and progression. Annu Rev Immunol. 2004;22:789–815. doi: 10.1146/annurev.immunol.22.012703.104716. [DOI] [PubMed] [Google Scholar]
- 24.Fischer A, Mercer J, Iyer A, Ragin M, August A. Regulation of CXC chemokine receptor 4-mediated migration by the Tec family tyrosine kinase ITK. J Biol Chem. 2004;279:29816–29820. doi: 10.1074/jbc.M312848200. [DOI] [PubMed] [Google Scholar]
- 25.Colgan J, Asmal M, Neagu M, Yu B, Schneidkraut J, Lee Y, Sokolskaja E, Andreotti A, Luban J. Cyclophilin A regulates TCR signal strength in CD4+ T cells via a proline-directed conformational switch in Itk. Immunity. 2004;21:189–201. doi: 10.1016/j.immuni.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 26.Lucas J, Felices M, Evans J, Berg L. Subtle defects in pre-TCR signaling in the absence of the Tec kinase Itk. J Immunol. 2007;179:7561–7567. doi: 10.4049/jimmunol.179.11.7561. [DOI] [PubMed] [Google Scholar]
- 27.Hu J, August A. Naive and innate memory phenotype CD4+ T cells have different requirements for active Itk for their development. J Immunol. 2008;180:6544–6552. doi: 10.4049/jimmunol.180.10.6544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hu J, Sahu N, Walsh E, August A. Memory phenotype CD8(+) T cells with innate function selectively develop in the absence of active Itk. Eur J Immunol. 2007;37:2892–2899. doi: 10.1002/eji.200737311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Taube C, Wei X, Swasey C, Joetham A, Zarini S, Lively T, Takeda K, Loader J, Miyahara N, Kodama T, Shultz L, Donaldson D, Hamelmann E, Dakhama A, Gelfand E. Mast cells, Fc epsilon RI, and IL-13 are required for development of airway hyperresponsiveness after aerosolized allergen exposure in the absence of adjuvant. J Immunol. 2004;172:6398–6406. doi: 10.4049/jimmunol.172.10.6398. [DOI] [PubMed] [Google Scholar]
- 30.Lee J, Dimina D, Macias M, Ochkur S, McGarry M, O’Neill K, Protheroe C, Pero R, Nguyen T, Cormier S, Lenkiewicz E, Colbert D, Rinaldi L, Ackerman S, Irvin C, Lee N. Defining a Link with Asthma in Mice Congenitally Deficient in Eosinophils. Science. 2004;305:1773–1776. doi: 10.1126/science.1099472. [DOI] [PubMed] [Google Scholar]
- 31.Humbles A, Lloyd C, McMillan S, Friend D, Xanthou G, McKenna E, Ghiran S, Gerard N, Yu C, Orkin S, Gerard C. A Critical Role for Eosinophils in Allergic Airways Remodeling. Science. 2004;305:1776–1779. doi: 10.1126/science.1100283. [DOI] [PubMed] [Google Scholar]
- 32.Rothenberg M, Hogan S. The eosinophil. Annu Rev Immunol. 2006;24:147–174. doi: 10.1146/annurev.immunol.24.021605.090720. [DOI] [PubMed] [Google Scholar]
- 33.Zhumabekov T, Corbella P, Tolaini M, Kioussis D. Improved version of a human CD2 minigene based vector for T cell-specific expression in transgenic mice. J Immunol Methods. 1995;185:133–140. doi: 10.1016/0022-1759(95)00124-s. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.