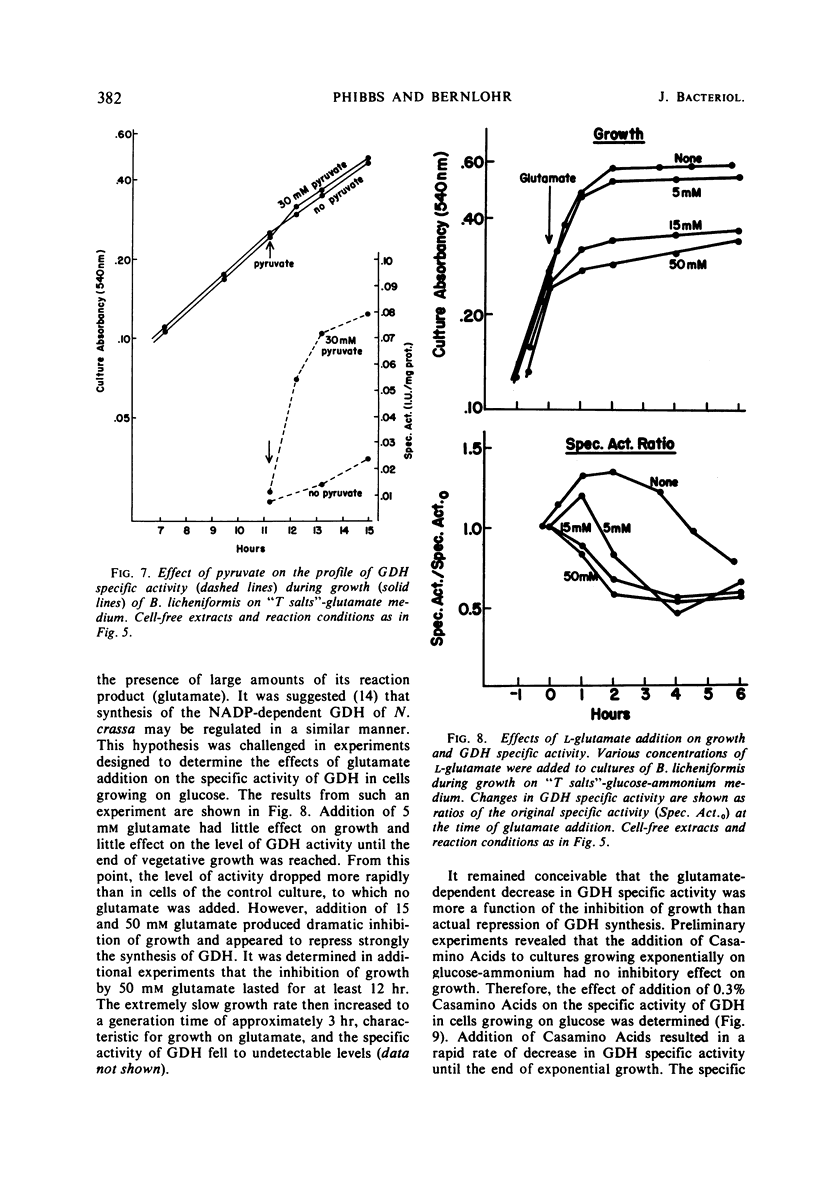
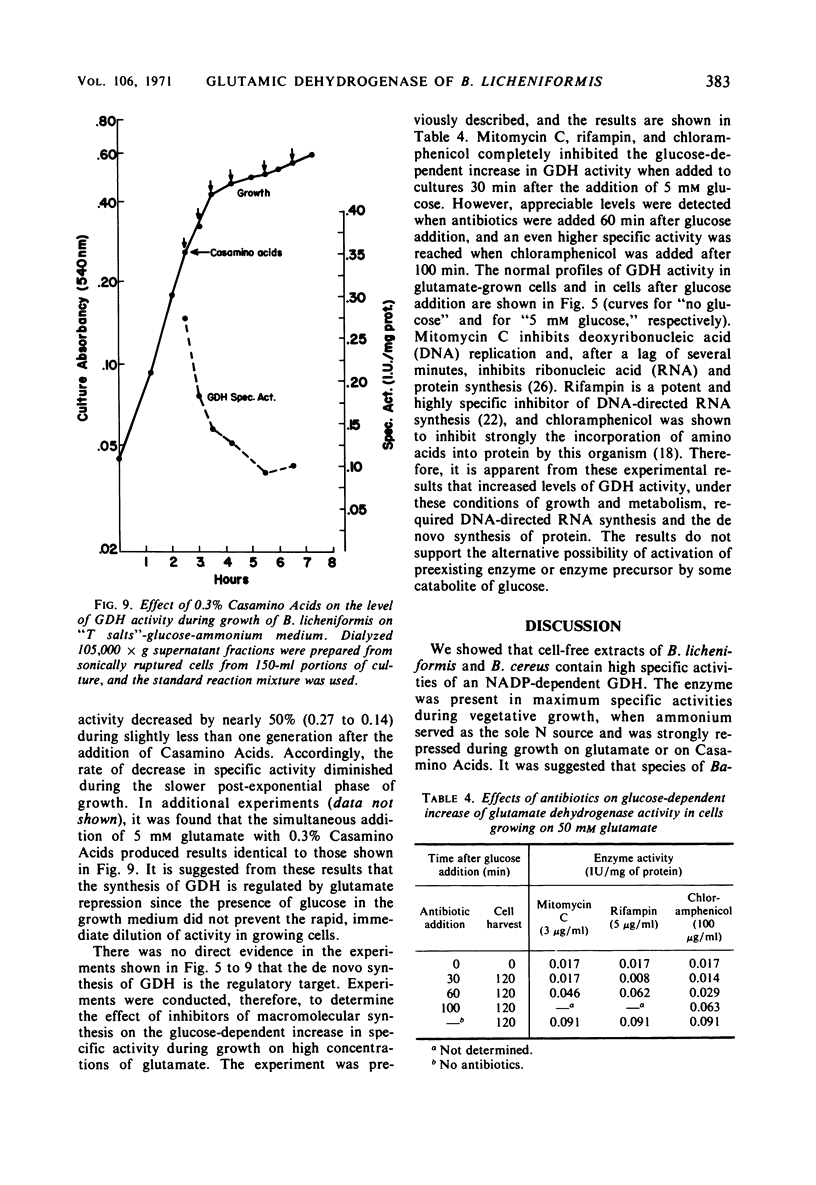
Abstract
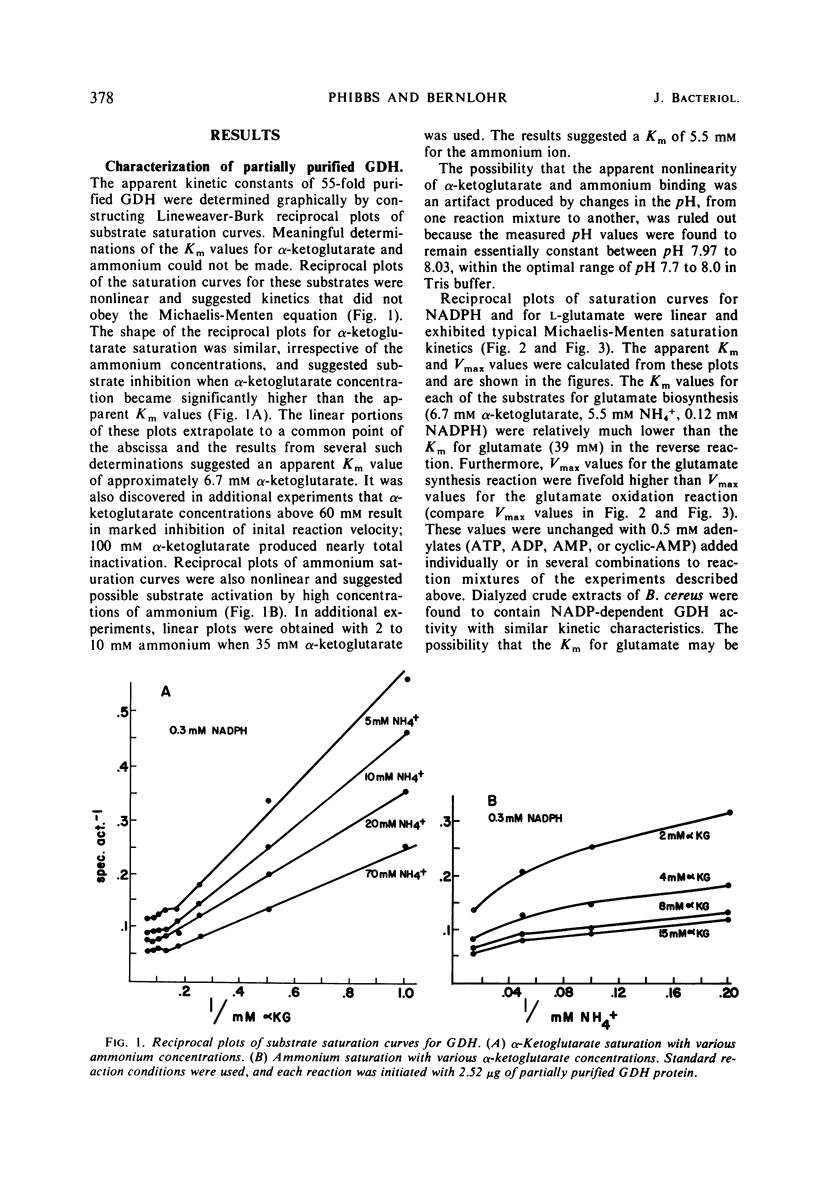
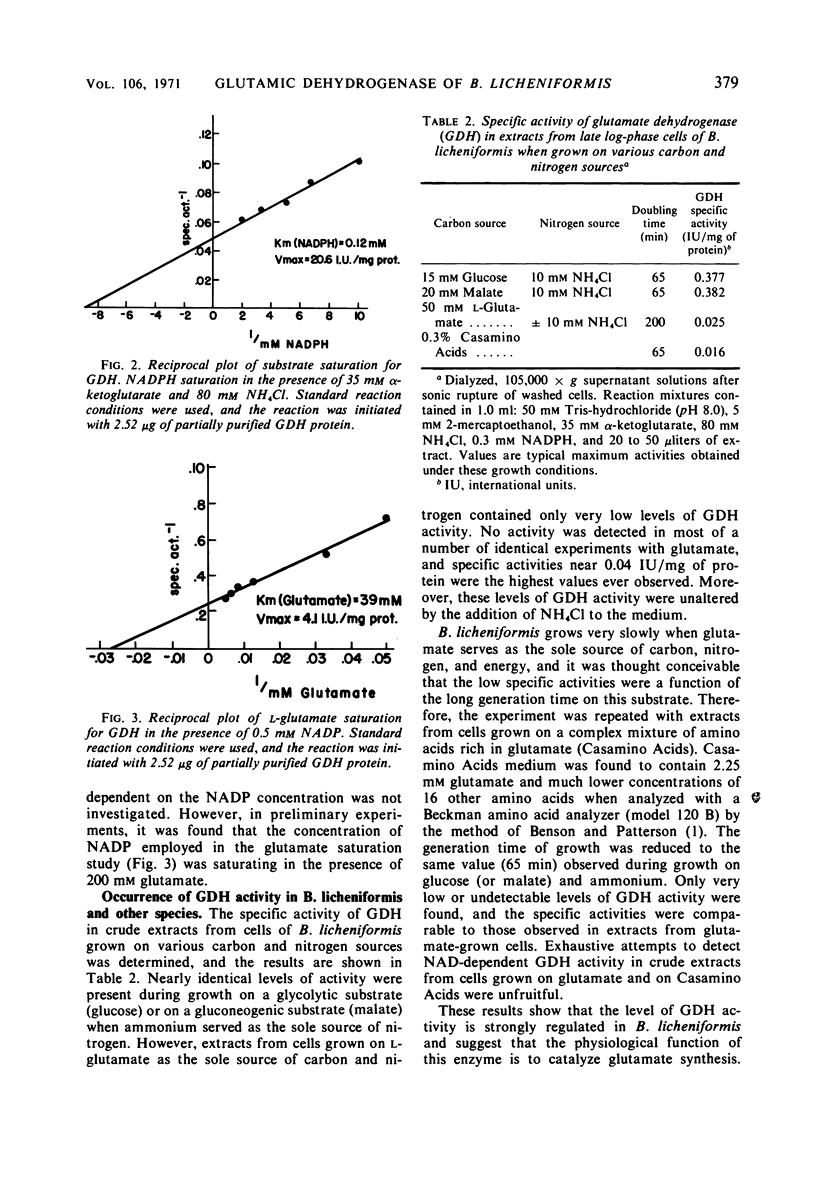
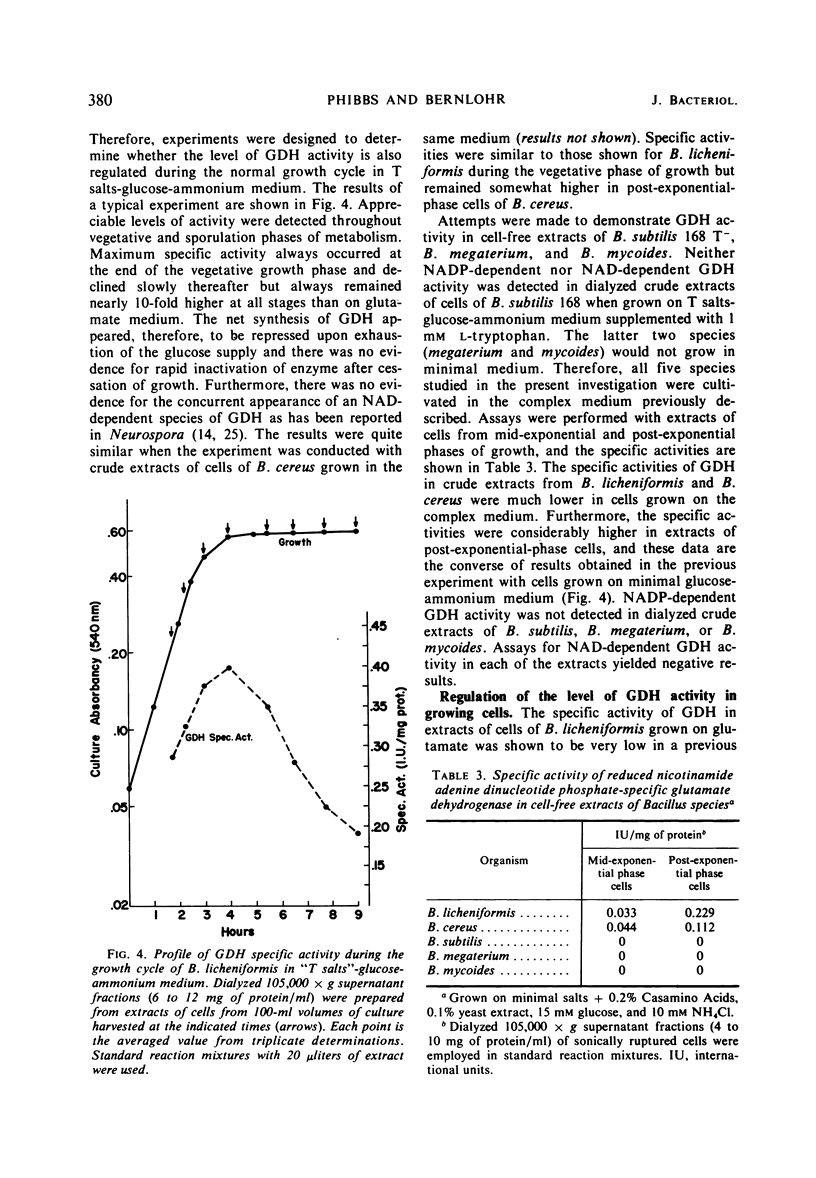
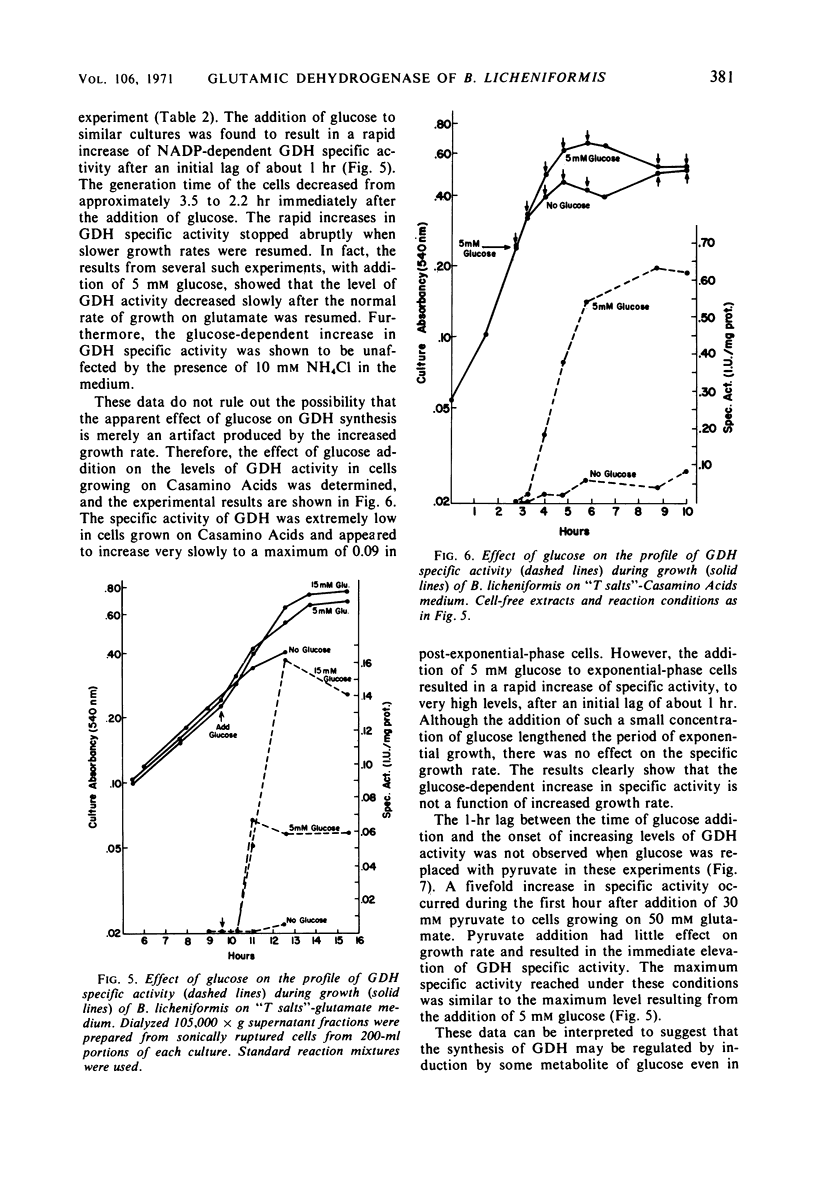
Cell-free extracts of Bacillus licheniformis and B. cereus were found to contain high specific activities of nicotinamide adenine dinucleotide phosphate (NADP)-dependent-l-glutamate dehydrogenase [EC 1.4.1.4; l-glutamate: NADP oxidoreductase (deaminating)]. Maximum specific activities were found in extracts of cells during the late exponential phase of growth when ammonium ion served as the sole source of nitrogen. Extremely low specific activities were detected throughout the growth cycle when l-glutamate or Casamino Acids served as the source of carbon and nitrogen. The enzyme was purified 55-fold from crude extracts of B. licheniformis, and apparent kinetic constants were determined. Sigmoidal saturation kinetics were not observed, and various adenylates had no effect on the enzyme. Repression of enzyme synthesis during growth on l-glutamate or Casamino Acids was partially overcome by additions of glucose or pyruvate, and this apparent derepression was totally abolished by inhibitors of ribonucleic acid and protein synthesis. Similarly, additions of l-glutamate or Casamino Acids to cells growing on glucose-ammonium ion resulted in strong repression of enzyme synthesis. It is suggested that the enzyme serves an anabolic role in metabolism. Nicotinamide adenine dinucleotide-dependent glutamate dehydrogenase activity was not detected in five species of Bacillus, irrespective of nutritional conditions or of the physiological age of cells.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BERNLOHR R. W. POSTLOGARITHMIC PHASE METABOLISM OF SPORULATING MICROORGANISMS. I. PROTEASE OF BACILLUS LICHENIFORMIS. J Biol Chem. 1964 Feb;239:538–543. [PubMed] [Google Scholar]
- Bernlohr R. W. Changes in amino acid permeation during sporulation. J Bacteriol. 1967 Mar;93(3):1031–1044. doi: 10.1128/jb.93.3.1031-1044.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borris D. P., Aronson J. N. Relationship of L-alanine and L-glutamate dehydrogenases of Bacillus thuringienses. Biochim Biophys Acta. 1969;191(3):716–718. doi: 10.1016/0005-2744(69)90366-0. [DOI] [PubMed] [Google Scholar]
- Dennen D. W., Niederpruem D. J. Regulation of glutamate dehydrogenases during morphogenesis of Schizophyllum commune. J Bacteriol. 1967 Mar;93(3):904–913. doi: 10.1128/jb.93.3.904-913.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fortnagel P., Freese E. Analysis of sporulation mutants. II. Mutants blocked in the citric acid cycle. J Bacteriol. 1968 Apr;95(4):1431–1438. doi: 10.1128/jb.95.4.1431-1438.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HALPERN Y. S., UMBARGER H. E. Conversion of ammonia to amino groups in Escherichia coli. J Bacteriol. 1960 Sep;80:285–288. doi: 10.1128/jb.80.3.285-288.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HANSON R. S., SRINIVASAN V. R., HALVORSON H. O. BIOCHEMISTRY OF SPORULATION. II. ENZYMATIC CHANGES DURING SPORULATION OF BACILLUS CEREUS. J Bacteriol. 1963 Jul;86:45–50. doi: 10.1128/jb.86.1.45-50.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HIERHOLZER G., HOLZER H. REPRESSION DER SYNTHESE VON DPN-ABHAENGIGER GLUTAMINSAEUREDEHYDROGENASE IN SACCHAROMYCES CEREVISIAE DURCH AMMONIUMIONEN. Biochem Z. 1963 Dec 3;339:175–185. [PubMed] [Google Scholar]
- HONG M. M., SHEN S. C., BRAUNSTEIN A. E. Distribution of L-alanine dehydrogenase and L-glutamate dehydrogenase in Bacilli. Biochim Biophys Acta. 1959 Nov;36:288–289. doi: 10.1016/0006-3002(59)90111-8. [DOI] [PubMed] [Google Scholar]
- Hanson R. S., Cox D. P. Effect of different nutritional conditions on the synthesis of tricarboxylic acid cycle enzymes. J Bacteriol. 1967 Jun;93(6):1777–1787. doi: 10.1128/jb.93.6.1777-1787.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joseph A. A., Wixon R. L. Ammonia incorporation in Hydrogenomonas eutropha. Biochim Biophys Acta. 1970 Feb 24;201(2):295–299. doi: 10.1016/0304-4165(70)90303-x. [DOI] [PubMed] [Google Scholar]
- KATO K., KOIKE S., YAMADA K., YAMADA H., TANAKA S. Di- and triphosphopyridine nucleotide linked glutamic dehydrogenases of Piricularia oryzae and their behaviors in glutamate media. Arch Biochem Biophys. 1962 Aug;98:346–347. doi: 10.1016/0003-9861(62)90195-9. [DOI] [PubMed] [Google Scholar]
- Kapoor M., Grover A. K. Catabolite-controlled regulation of glutamate dehydrogenases of Neurospora crassa. Can J Microbiol. 1970 Jan;16(1):33–40. doi: 10.1139/m70-006. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laishley E. J., Bernlohr R. W. Catabolite repression of "three sporulation enzymes" during growth of Bacillus licheniformis. Biochem Biophys Res Commun. 1966 Jul 6;24(1):85–90. doi: 10.1016/0006-291x(66)90414-1. [DOI] [PubMed] [Google Scholar]
- Laishley E. J., Bernlohr R. W. Regulation of arginine and proline catabolism in Bacillus licheniformis. J Bacteriol. 1968 Aug;96(2):322–329. doi: 10.1128/jb.96.2.322-329.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leitzmann C., Bernlohr R. W. Threonine dehydratase of Bacillus licheniformis. II. Regulation during development. Biochim Biophys Acta. 1968 Feb 5;151(2):461–472. doi: 10.1016/0005-2744(68)90114-9. [DOI] [PubMed] [Google Scholar]
- LéJohn H. B., McCrea B. E. Evidence for two species of glutamate dehydrogenases in Thiobacillus novellus. J Bacteriol. 1968 Jan;95(1):87–94. doi: 10.1128/jb.95.1.87-94.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MCCORMICK N. G., HALVORSON H. O. PURIFICATION AND PROPERTIES OF L-ALANINE DEHYDROGENASE FROM VEGETATIVE CELLS OF BACILLUS CEREUS. J Bacteriol. 1964 Jan;87:68–74. doi: 10.1128/jb.87.1.68-74.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pateman J. A. Regulation of synthesis of glutamate dehydrogenase and glutamine synthetase in micro-organisms. Biochem J. 1969 Dec;115(4):769–775. doi: 10.1042/bj1150769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SANWAL B. D., LATA M. Concurrent regulation of glutamic acid dehydrogenases of Neurospora. Arch Biochem Biophys. 1962 Jun;97:582–588. doi: 10.1016/0003-9861(62)90127-3. [DOI] [PubMed] [Google Scholar]
- Suzuki H., Kilgore W. W. Effects of mitomycin C on macromolecular synthesis in Escherichia coli. J Bacteriol. 1967 Feb;93(2):675–682. doi: 10.1128/jb.93.2.675-682.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varricchio F. Control of glutamate dehydrogenase synthesis in Escherichia coli. Biochim Biophys Acta. 1969 May 6;177(3):560–564. doi: 10.1016/0304-4165(69)90319-5. [DOI] [PubMed] [Google Scholar]
- WIAME J. M., COLLETTE J., BOURGEOIS S. Etude de l'assimilation de l'ammonium à l'aide de variants de Bacillus subtilis. Arch Int Physiol Biochim. 1955 Jun;63(2):271–271. doi: 10.3109/13813455509150435. [DOI] [PubMed] [Google Scholar]
- Winnacker E. L., Barker H. A. Purification and properties of a NAD-dependent glutamate dehydrogenase from Clostridium SB4. Biochim Biophys Acta. 1970 Aug 15;212(2):225–242. doi: 10.1016/0005-2744(70)90203-2. [DOI] [PubMed] [Google Scholar]
- di Mauro E., Synder L., Marino P., Lamberti A., Coppo A., Tocchini-Valentini G. P. Rifampicin sensitivity of the components of DNA-dependent RNA polymerase. Nature. 1969 May 10;222(5193):533–537. doi: 10.1038/222533a0. [DOI] [PubMed] [Google Scholar]



