Abstract
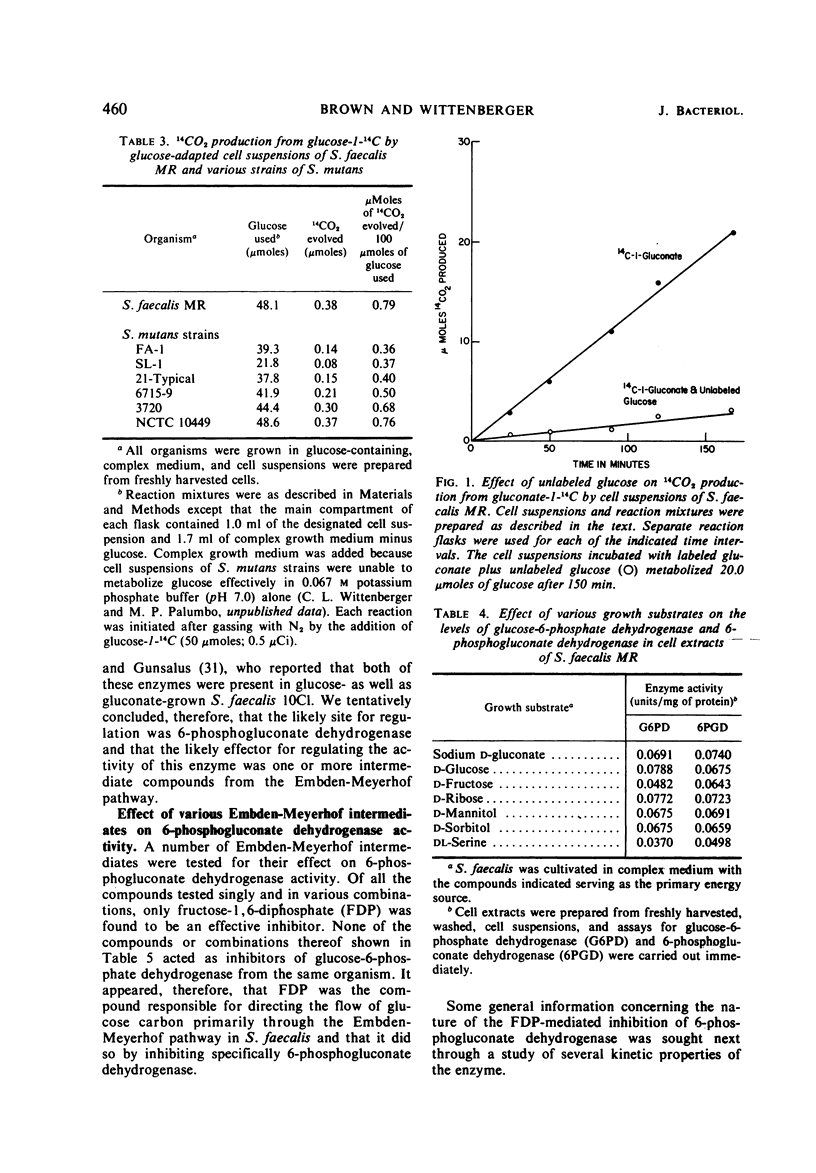
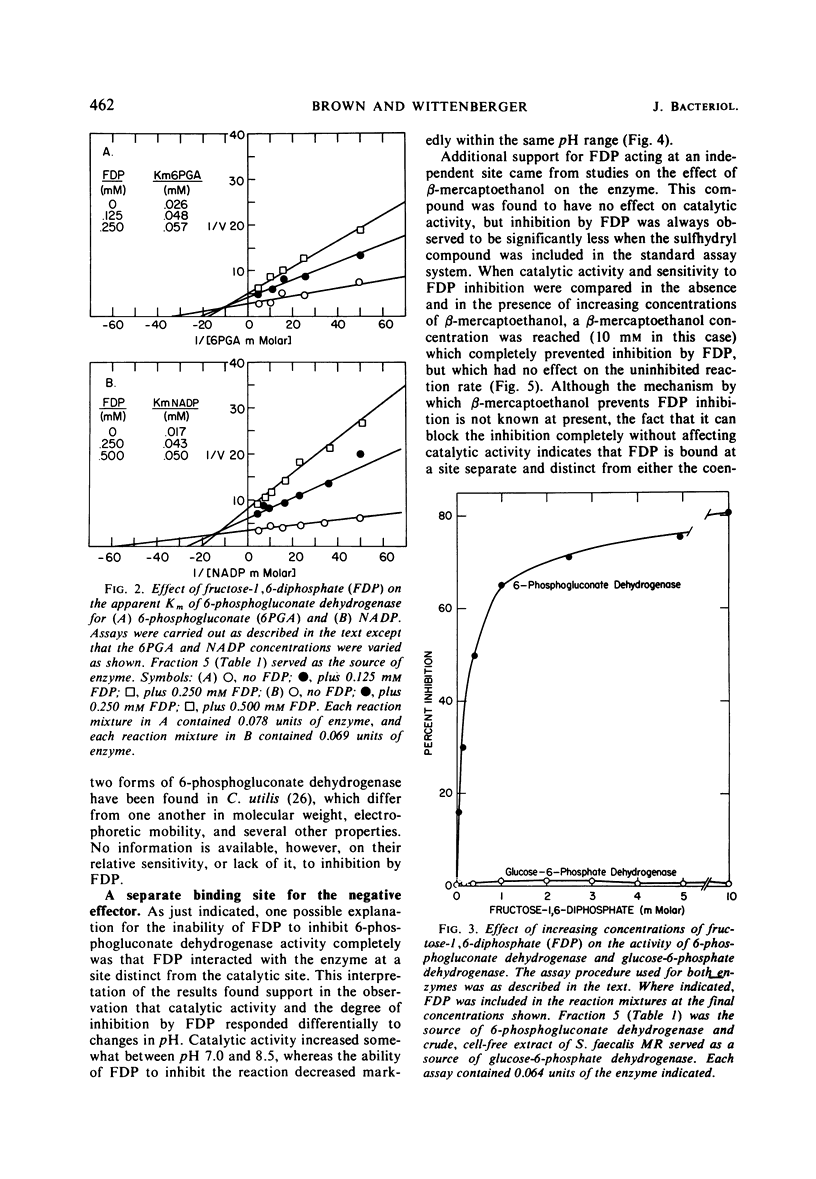
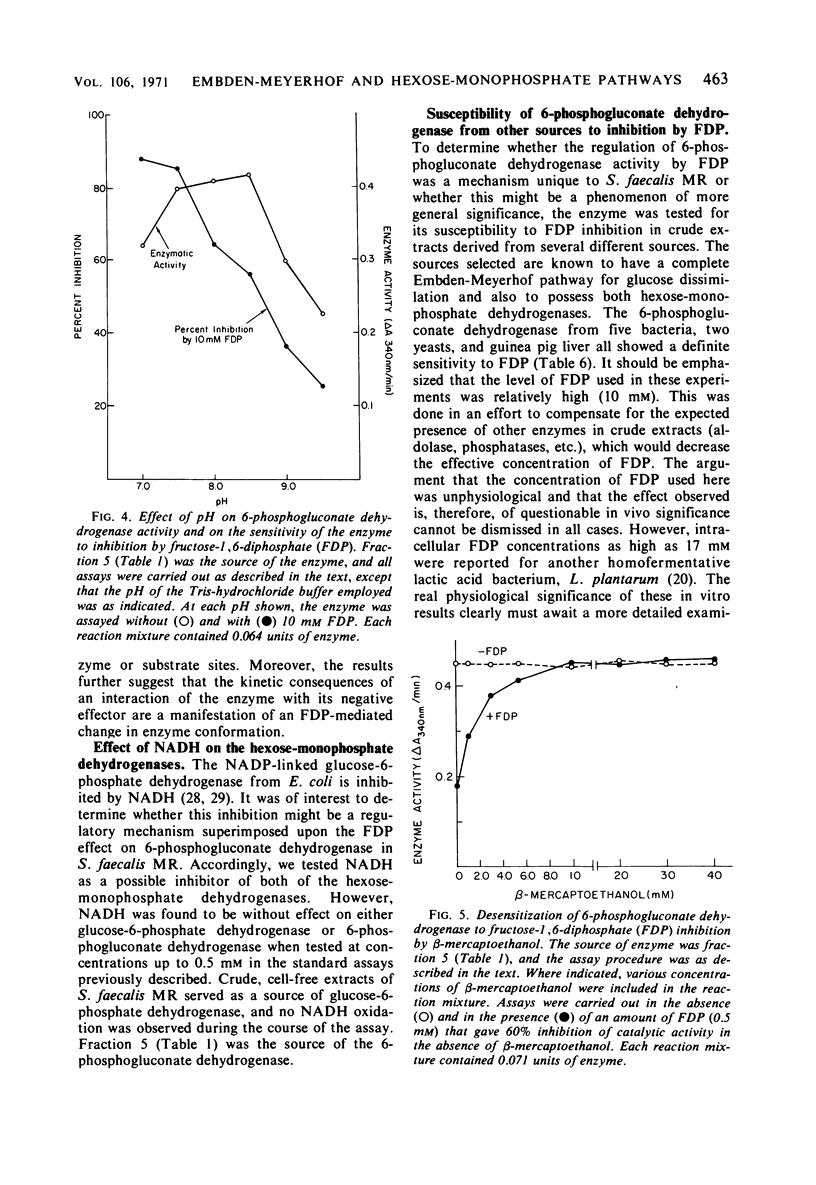
Glucose-adapted Streptococcus faecalis produced little if any 14CO2 from glucose-1-14C, although high levels of glucose-6-phosphate dehydrogenase (EC 1.1.1.49) and 6-phosphogluconate dehydrogenase (EC 1.1.1.44) were detected in cell-free extracts. Metabolism of glucose through the oxidative portion of the hexose-monophosphate pathway was shown to be regulated in this organism by the specific inhibitory interaction of the Embden-Meyerhof intermediate, fructose-1, 6-diphosphate (FDP), with 6-phosphogluconate dehydrogenase. Glucose-6-phosphate dehydrogenase activity was unaffected by FDP. The S. faecalis 6-phosphogluconate dehydrogenase was partially purified from crude extracts by standard fractionation procedures and certain kinetic parameters of the FDP-mediated inhibition were investigated. The negative effector was shown to cause a decrease in Vmax and an increase in the apparent Km for both 6-phosphogluconate and nicotinamide adenine dinucleotide phosphate (NADP). These effects were apparently a consequence of the ligand interacting with the enzyme at a site distinct from either the substrate or the coenzyme sites. Among the evidence supporting this was the fact that β-mercaptoethanol blocked completely FDP inhibition, but had no effect on catalytic activity. The possibility that the regulation of 6-phosphogluconate dehydrogenase activity by FDP might be of some general significance was suggested by the observation that this enzyme from several other sources was also sensitive to FDP.
Full text
PDF








Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Avigad G. Inhibition of glucose 6-phosphate dehydrogenase by adenosine 5'-triphosphate. Proc Natl Acad Sci U S A. 1966 Nov;56(5):1543–1547. doi: 10.1073/pnas.56.5.1543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EAGON R. G., WANG C. H. Dissimilation of glucose and gluconic acid by Pseudomonas natriegens. J Bacteriol. 1962 Apr;83:879–886. doi: 10.1128/jb.83.4.879-886.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FRAENKEL D. G., HORECKER B. L. PATHWAYS OF D-GLUCOSE METABOLISM IN SALMONELLA TYPHINMURIUM. A STUDY OF A MUTANT LACKING PHOSPHOGLUCOSE ISOMERASE. J Biol Chem. 1964 Sep;239:2765–2771. [PubMed] [Google Scholar]
- Frieden C. Kinetic aspects of regulation of metabolic processes. The hysteretic enzyme concept. J Biol Chem. 1970 Nov 10;245(21):5788–5799. [PubMed] [Google Scholar]
- GIBBS M., SOKATCH J. T., GUNSALUS I. C. Product labeling of glucose-1-C14 fermentation by homofermentative and heterofermentative lactic acid bacteria. J Bacteriol. 1955 Nov;70(5):572–576. doi: 10.1128/jb.70.5.572-576.1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GLASER L., BROWN D. H. Purification and properties of d-glucose-6-phosphate dehydrogenase. J Biol Chem. 1955 Sep;216(1):67–79. [PubMed] [Google Scholar]
- Guggenheim B. Streptococci of dental plaques. Caries Res. 1968;2(2):147–163. doi: 10.1159/000259553. [DOI] [PubMed] [Google Scholar]
- Horne R. N., Anderson W. B., Nordlie R. C. Glucose dehydrogenase activity of yeast glucose 6-phosphate dehydrogenase. Inhibition by adenosine 5'-triphosphate and other nucleoside 5'-triphosphates and diphosphates. Biochemistry. 1970 Feb 3;9(3):610–616. doi: 10.1021/bi00805a023. [DOI] [PubMed] [Google Scholar]
- KAHANA S. E., LOWRY O. H., SCHULZ D. W., PASSONNEAU J. V., CRAWFORD E. J. The kinetics of phosphoglucoisomerase. J Biol Chem. 1960 Aug;235:2178–2184. [PubMed] [Google Scholar]
- LOWRY O. H., PASSONNEAU J. V. A COMPARISON OF THE KINETIC PROPERTIES OF PHOSPHOFRUCTOKINASE FROM BACTERIAL, PLANT AND ANIMAL SOURCES. Naunyn Schmiedebergs Arch Exp Pathol Pharmakol. 1964 May 11;248:185–194. doi: 10.1007/BF00246673. [DOI] [PubMed] [Google Scholar]
- Lessie T., Neidhardt F. C. Adenosine triphosphate-linked control of Pseudomonas aeruginosa glucose-6-phosphate dehydrogenase. J Bacteriol. 1967 Apr;93(4):1337–1345. doi: 10.1128/jb.93.4.1337-1345.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- London J., Meyer E. Y. Malate utilization by a group D Streptococcus: regulation of malic enzyme synthesis by an inducible malate permease. J Bacteriol. 1970 Apr;102(1):130–137. doi: 10.1128/jb.102.1.130-137.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MIZUSHIMA S., KITAHARA K. QUANTITATIVE STUDIES ON GLYCOLYTIC ENZYMES IN LACTOBACILLUS PLANTARUM. II. INTRACELLULAR CONCENTRATIONS OF GLYCOLYTIC INTERMEDIATES IN GLUCOSE-METABOLIZING WASHED CELLS. J Bacteriol. 1964 Jun;87:1429–1435. doi: 10.1128/jb.87.6.1429-1435.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Model P., Rittenberg D. Measurement of the activity of the hexose monophosphate pathway of glucose metabolism with the use of [18O]glucose. Variations in its activity in Escherichia coli with growth conditions. Biochemistry. 1967 Jan;6(1):69–80. doi: 10.1021/bi00853a013. [DOI] [PubMed] [Google Scholar]
- PARR C. W. Inhibition of phosphoglucose isomerase. Nature. 1956 Dec 22;178(4547):1401–1401. doi: 10.1038/1781401a0. [DOI] [PubMed] [Google Scholar]
- PLATT T. B., FOSTER E. M. Products of glucose metabolism by homofermentative streptococci under anaerobic conditions. J Bacteriol. 1958 Apr;75(4):453–459. doi: 10.1128/jb.75.4.453-459.1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rippa M., Signorini M., Pontremoli S. Purification and properties of two forms of 6-phosphogluconate dehydrogenase from Candida utilis. Eur J Biochem. 1967 Apr;1(2):170–178. doi: 10.1007/978-3-662-25813-2_26. [DOI] [PubMed] [Google Scholar]
- SOKATCH J. T., GUNSALUS I. C. Aldonic acid metabolism. I. Pathway of carbon in an inducible gluconate fermentation by Streptococcus faecalis. J Bacteriol. 1957 Apr;73(4):452–460. doi: 10.1128/jb.73.4.452-460.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanwal B. D. Allosteric controls of amphilbolic pathways in bacteria. Bacteriol Rev. 1970 Mar;34(1):20–39. doi: 10.1128/br.34.1.20-39.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanwal B. D. Regulatory mechanisms involving nicotinamide adenine nucleotides as allosteric effectors. 3. Control of glucose 6-phosphate dehydrogenase. J Biol Chem. 1970 Apr 10;245(7):1626–1631. [PubMed] [Google Scholar]
- Schindler J., Schlegel H. G. Regulation der Glucose-6-phosphat-Dehydrogenase aus verschiedenen Bakterienarten durch ATP. Arch Mikrobiol. 1969;66(1):69–78. [PubMed] [Google Scholar]
- Stadtman E. R. Allosteric regulation of enzyme activity. Adv Enzymol Relat Areas Mol Biol. 1966;28:41–154. doi: 10.1002/9780470122730.ch2. [DOI] [PubMed] [Google Scholar]
- VENKATARAMAN R., RACKER E. Mechanism of action of transaldolase. I. Crystalization and properties of yeast enzyme. J Biol Chem. 1961 Jul;236:1876–1882. [PubMed] [Google Scholar]
- VINUELA E., SALAS M. L., SOLS A. End-product inhibition of yeast phosphofructokinase by ATP. Biochem Biophys Res Commun. 1963 Jul 18;12:140–145. doi: 10.1016/0006-291x(63)90250-x. [DOI] [PubMed] [Google Scholar]
- WOLIN M. J. FRUCTOSE-1,6-DIPHOSPHATE REQUIREMENT OF STREPTOCOCCAL LACTIC DEHYDROGENASES. Science. 1964 Nov 6;146(3645):775–777. doi: 10.1126/science.146.3645.775. [DOI] [PubMed] [Google Scholar]
- Wittenberger C. L., Angelo N. Purificationa and properties of a fructose-1,6-diphosphate-activated lactate dehydrogenase from Streptococcus faecalis. J Bacteriol. 1970 Mar;101(3):717–724. doi: 10.1128/jb.101.3.717-724.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]



