Abstract
Failure to recognize that many standard control rats and mice used in biomedical research are sedentary, obese, glucose intolerant, and on a trajectory to premature death may confound data interpretation and outcomes of human studies. Fundamental aspects of cellular physiology, vulnerability to oxidative stress, inflammation, and associated diseases are among the many biological processes affected by dietary energy intake and exercise. Although overfed sedentary rodents may be reasonable models for the study of obesity in humans, treatments shown to be efficacious in these animal models may prove ineffective or exhibit novel side effects in active, normal-weight subjects.
Keywords: cancer, clinical trials, exercise, insulin resistance, obesity
Mice and rats are the most widely used animal models in biomedical research. When housed under standard laboratory conditions these rodents are sedentary, have continuous access to food, and have virtually no environmental stimulation. Animals maintained in this manner are widely used as “standard” controls in basic and translational biomedical research studies, including preclinical drug testing. Compared to those that are fed less, exercise more, and have a stimulating environment, animals maintained under the usual standard laboratory conditions are relatively overweight, insulin resistant, hypertensive, and are likely to experience premature death (1–5). Indeed, simply reducing daily food intake 20–40% below the ad libitum amount, or providing food intermittently, rather than continuously, has been shown to significantly reduce the risk of developing diseases such as cancer, type 2 diabetes, and renal failure and can extend lifespan by up to 40% in rats and mice (3, 6, 7). Currently, one of the greatest health concerns is the rise in obesity and its associated pathologies, such as metabolic syndrome, diabetes, cardiovascular disease, and cancer. In the United States, more than 30% of the adult population is obese, and some studies estimate that by 2030, more than 366 million people worldwide will develop type 2 diabetes, with obesity being an important factor responsible for this increase (8, 9). Although dietary energy restriction can increase the maximum lifespan of laboratory animals and is therefore hailed as an “antiaging” intervention (10), its major effect is to increase the average lifespan by preventing or delaying the development of various diseases that are the primary cause of death in overweight rodents (11).
The use of overweight and unstimulated animals as standard controls may bias the measured experimental outcomes. We therefore suggest that new guidelines with regards to laboratory animal husbandry should be developed and implemented that ensure that control animals are fed in portions rather than ad libitum and are provided environmental stimulation. At the very least, institutional animal care and use committees should make investigators aware of the fact that their control animals are overfed and relatively sedentary. In the present article, we provide an overview and discuss how experimental outcomes and data interpretation may be altered depending on the type of control animal that is used, i.e., healthy nonobese “lean” control compared to an overweight unhealthy standard control.
Health of Laboratory Animals Is Poor by Human Standards
Laboratory mice and rats are typically housed either singly or in groups of 2–5 per cage with 1–3 square feet of floor space covered with bedding. The animals have continuous access to food, which typically consists of dry pelleted food provided in an overhead bin. Under these conditions the animals gain weight progressively during their adult life; some strains of laboratory rats achieve body weights upwards of 1 kg and fat mass that accounts for 30–50% of the body weight (12). Regular exercise (voluntary exercise on an in-cage running wheel, for example) and/or dietary energy restriction have been shown to cause significant reductions in body weight and body fat mass (∼20–40%) (12, 13). In addition to being overweight, laboratory rodents maintained under standard laboratory conditions exhibit a physiological profile consistent with increased disease susceptibility, compared to animals maintained on lower energy diets and/or animals with higher physical activity levels (see Table 1 for overview). Thus, relative to their leaner counterparts on reduced energy diets, typical overweight ad libitum-fed rodents exhibit elevated levels of energy regulatory hormones and factors such as glucose, insulin, triglycerides, low-density lipoprotein (LDL) cholesterol and leptin, and decreased levels of adiponectin and ghrelin (3, 14). Additionally, the general cardiovascular health of laboratory rats has been shown to be improved when their food intake is reduced; i.e., their plasma lipid profiles are improved, blood pressure and resting heart rate are reduced, and the ability of their cardiovascular system to recover from stress is enhanced (15, 16). Consistent with an adverse effect of the standard housing conditions on the overall health and well-being of laboratory animals, it has been demonstrated that wild mice eat less and live longer than domesticated laboratory mice (17).
Table 1.
Comparisons of physiological and metabolic factors in rats maintained under standard housing conditions (overfed and sedentary) and more natural conditions (reduced energy intake or running wheel exercise)
| Housing conditions | |||
| Factor | Standard | Diet restriction | Exercise |
| Body weight | 600–700 g | 350–500 g | 500–600 g |
| Total body fat | 25–40% | 5–20% | 10–20% |
| Mean blood pressure | 110–130 mm Hg | 80–90 mm Hg | 115–125 mm Hg |
| Resting heart rate | 350–400 bpm | 250–300 bpm | 280–300 bpm |
| Plasma glucose | 150–160 mg/dL | 110–130 mg/dL | 125–135 mg/dL |
| Plasma insulin | 125–140 nmol/L | 70–80 nmol/L | NA |
| Plasma leptin | 8–12 ng/mL | 2–6 ng/mL | NA |
| Plasma adiponectin | 9–11 ng/mL | 14–16 ng/mL | NA |
| Total cholesterol | 140–170 mg/dL | 70–100 mg/dL | 130–140 mg/dL |
| LDL cholesterol | 15–25 mg/dL | 10–15 mg/dL | NA |
| TNFα* | 6–7 pg/mL | 3–4 pg/mL | NA |
| IL-6* | 6.5–7.5 pg/mL | 4.5–5.5 pg/mL | NA |
Studies in which the dietary energy intake of rats or mice was reduced have clearly demonstrated that the standard ad libitum feeding paradigm fosters poor health outcomes and premature death. In one study, the average lifespan of rats was increased from 2.4 years for those housed under standard conditions to 4 years for those maintained on a reduced energy diet; the longest-lived control ad libitum rat lived for 2.9 years, whereas the oldest energy-restricted rat lived for 4.6 years (18). The activity levels of rodents that eat a reduced calorie diet have been shown to be increased compared to their ad libitum-fed counterparts (15), which may contribute to their increased lean/fat mass ratio. The insulin sensitivity of muscle and liver cells increases when dietary energy intake is decreased or animals are provided with the opportunity to exercise, indicating that animals housed under standard laboratory conditions are relatively insulin resistant (15, 19). In humans, such a metabolic profile is a harbinger of future dysfunction and disease in multiple organ systems (20). Not only are ad libitum-fed animals less healthy than those that eat less and exercise more, they also have reduced cognitive function. Thus, the cognitive abilities of rats maintained on restricted feeding schedules (15) or provided access to a running wheel (21) are superior to rats maintained under standard housing conditions.
Insulin Resistance and Diabetes
Insulin resistance is defined by impaired sensitivity to insulin of its main target organs, adipose tissue, liver, and muscle. Insulin regulates glucose uptake and circulating free fatty acid (FFA) concentrations. Insulin decreases lipolysis and FFA efflux from adipocytes, inhibits gluconeogenesis in liver cells by reducing key enzyme activities, and induces glucose uptake in skeletal muscle cells by stimulating the translocation of the GLUT4 glucose transporter to the plasma membrane. Obesity, and particularly central obesity, is the most common risk factor for the development of type 2 diabetes and other features of the metabolic syndrome such as dyslipidemia and hypertension. Numerous studies have demonstrated that dietary energy restriction promotes euglycemia and increases insulin sensitivity (22, 23). One longitudinal study of rats showed that dietary energy restriction decreases the mean 24-h plasma glucose levels by ≈15 mg/dL and the circulating insulin levels by ≈50% (24). It has been proposed that the significant effects of dietary energy restriction on circulating levels of glycemic hormones could play a role in the beneficial life-extending actions of dietary energy restriction. However, as control laboratory rats and mice tend to become overweight and insulin resistant under standard housing conditions, the question arises: Is the lifespan of the restricted animals extended only because it has been compared to the lifespan of the relatively unhealthy standard control animals?
Immune Function and Inflammation
There are strong functional connections between the metabolic and immune systems (25–27). Metabolic disruption and obesity involve overactivation of an inflammatory process in metabolically active sites such as adipose tissue, liver, and immune cells. The consequence of this effect is a potent increase in circulating levels of proinflammatory cytokines and other inflammatory markers (28). Activation of the immune system in response to obesity is mediated by several specific and conserved signaling pathways, with Jun N-terminal kinase (JNK) and IκB kinase β/nuclear factor κ-light-chain-enhancer of activated B (IKKβ/NFκB) pathways being the most well studied (29). It is known that the immune activation of these signaling pathways can modify insulin signaling and result in the development of insulin resistance, which exacerbates systemic physiological failure. In obese patients as well as standard control experimental animals, a chronic low-grade inflammation occurs which is characterized by increased plasma levels of C-reactive protein, inflammatory cytokines such as TNF-α, IL-6, MCP-1, IL-8, and multifunctional proteins such as leptin (30) and osteopontin (31). Adipose tissue produces large quantities of inflammatory cytokines and chemokines (collectively called adipokines) and adipose tissue is one of the main contributors to elevated systemic TNF-α concentrations in obesity (32). Long-term obesity-induced systemic metabolic disruption also generates a chronic stressful state response (33) that can suppress humoral immunity. This stress can be causally linked to obesity through increased levels of plasma glucocorticoids, which can stimulate the development and differentiation of preadipocytes (34). The biological activity of the stress-responsive glucocorticoids is tightly controlled by the expression of the enzyme 11β-hydroxysteroid dehydrogenase (11β-HSD) (35). Protection from diet-induced obesity and insulin resistance was found in mice lacking 11β-HSD1 (36), whereas mice with adipose tissue-specific overexpression of 11β-HSD1 developed an obese phenotype when fed a high calorie diet (37). Studies in obese humans resulted in similar findings (38, 39) and, most importantly, suggested that the ability to regulate the activity of 11β-HSD is lost in type 2 diabetic patients and is compromised in nondiabetic, obese individuals. Proinflammatory mediators, (TNF-α, IL-6, MCP-1, and IL-8) intracellular processes (stress response) and signaling pathways (JNK and NFκB activation) have all been linked to obesity and inflammation in experimental animals, demonstrating the importance of consideration of the metabolic-immune status of the control animal. In humans, several functional similarities with the animal findings have been found (40, 41). However, there are disparities between animal and human therapeutic responsiveness, which may be due to the use of the standard animal control. Obesity-related immunotherapeutics based on mouse studies, such as neutralization of TNF-α, have not transferred successfully to humans. Obese humans administered TNF-α monoclonal antibodies do not show similar responses to those of mice with respect to a decrease in body weight and the reversal of insulin resistance (42–44). As the standard control animal may possess altered immune responses, activation of adipokines, and stress status, it is likely that this may impact the apparent efficacy of lifestyle or pharmacotherapeutic interventions for disorders that are influenced by inflammatory and immune activity.
Cancer
The standard living conditions of laboratory animals might be considered a good reflection of an increasingly large proportion of populations in industrialized countries who are sedentary and overfed. With respect to the induction of tumors in experimental animal conditions, standard controls develop more spontaneous tumors than do those that eat less (45, 46). Many different types of tumors grow more rapidly in animals fed ad libitum compared to those on reduced energy diets (47–49). In addition, some carcinogens are less effective in inducing cancers in animals maintained on reduced energy diets, compared to overweight animals on the standard ad libitum diet (50, 51). Certain aspects of the mechanisms of carcinogenesis, tumor growth, and metastasis may be different in obese mice compared to more slender mice that eat less. Indeed, the effects of energy intake and exercise on carcinogenesis may result from changes in the expression of p450 enzymes that metabolize carcinogens, or in the amounts of oxidative DNA damage and apoptosis (50–53).
Although the risk of several types of cancers is increased in individuals who are overweight and sedentary, many cancers can strike otherwise healthy people at any age. It is therefore important to know whether potential cancer therapies that are efficacious in reducing tumor growth in overweight and unstimulated animals are equally effective in more physically fit animals. There have been more than 500 phase II clinical trials in cancer patients involving drugs that fall into only a few general types with regard to their mechanisms of action, including cytotoxic, antiproliferative, and antiangiogenic agents (54). The majority of these drugs, although exhibiting efficacy in animal studies are often ineffective in the human clinical trials. It is reasonable to consider that some of these drugs might have failed because they were evaluated in preclinical rodent models in which the subjects are uniformly in a metabolically morbid state. For example, angiogenesis may play a much more prominent role in the growth of tumors in obese individuals (55), and so antiangiogenic drugs may have relatively little effect on the same type of tumor in a slender and fit individual. In addition, numerous hormones and growth factors whose levels are affected by energy intake (testosterone, estrogen, leptin, IGF1, VEGF, etc.) also affect the growth of cancers (45, 46, 49–58). A given cancer drug might therefore be more (or less) effective in overweight individuals compared to their slender counterparts.
Neurological Disorders
What are the arguments, both pro and con, of standard ad libitum animal models for major neurodegenerative disorders such as Alzheimer’s, Parkinson’s and Huntington’s diseases, and stroke? The most widely used models of Alzheimer’s disease are transgenic mice that express a mutant form of human amyloid precursor protein alone, or in combination with mutant presenilin-1 and tau (59). Such “Alzheimer’s mice” develop progressive accumulation of amyloid β-peptide, and associated dysfunction of synapses, in brain regions that play a role in regulating learning and memory processes. The progression of the latter pathological processes is retarded by dietary energy restriction (60) and environmental enrichment (61), and is hastened by diabetes (62). Similarly, dietary energy restriction (63, 64) and exercise (65, 66) have been reported to delay disease onset and slow progression in mouse models of Parkinson’s and Huntington’s diseases. A potential advantage of the standard ad libitum laboratory housing conditions is that the disease process is accelerated, and so experimental interventions can be tested more rapidly. A possible disadvantage is that drugs that show good efficacy in the overweight animal models may be less effective or ineffective in active normal weight animals. For example, because levels of oxidative stress are elevated in brain cells of sedentary obese animals, antioxidant therapies may be very effective in animals housed under standard laboratory conditions, but not in more healthy animals that eat less food and exercise more (67, 68). The failure of several different drugs to improve outcome in human stroke patients, despite their clear effectiveness in animal models of stroke, might be due to the fact that the animal models involved young sedentary overfed rats and mice (69, 70). Indeed, neurons in the brains of overfed animals are more vulnerable to ischemic stroke than are neurons in the brains of their more healthy counterparts (64). Thus, drugs such as glutamate receptor antagonists (71) and antiinflammatory agents (72) may be effective in animal models, but ineffective in human clinical trials, because they modify processes associated with a sedentary gluttonous lifestyle instead of, or in addition to, stroke-specific processes.
A Comparison of Various Genomic and Physiological Parameters in an ad Libitum Overfed Standard Control Laboratory Rat Model to a Healthy Body Weight Control
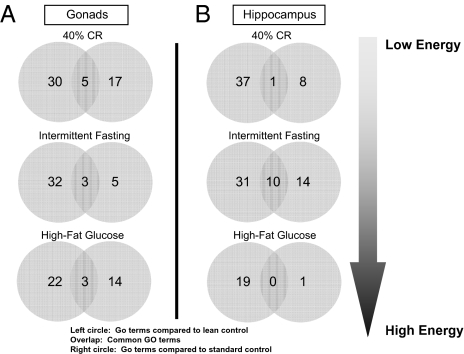
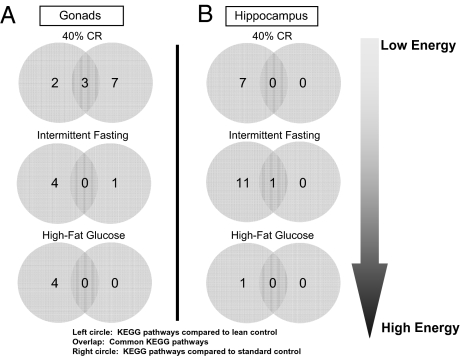
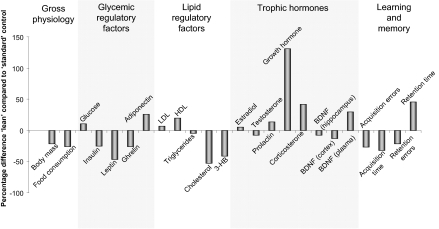
Using a standardized animal model, the Sprague–Dawley rat, we compared how usage of an overweight standard control or a lean control can affect various output data. In this experiment, multiple forms of animal data were recorded from Sprague–Dawley rats subjected to different amounts of energy intake and food availability (40% calorie restriction, alternate day fasting and a high fat/glucose diet) for a period of 4 months (from 4 to 8 months of age). The output variables measured, using comparison with standard or lean controls, included transcriptional regulation in multiple tissues including gonads (73) and hippocampus (74), whole body weight and food intake, plasma hormones, and performance in cognitive tests (15). In this scenario, we compared output data indices first to the standard overweight ad libitum control and then to a lean “mild calorie-restricted” control (20% calorie restriction compared to ad libitum control). First, we studied the differences in global transcription in two different tissues of female rats (similar results were also obtained for males). When comparing the genes significantly modulated by the implemented experimental diets to the two controls (standard or lean) it is clear from Fig. 1 that there is minimal similarity in the identity of the significantly regulated genes in both tissues for all of the experimental paradigms. Therefore, using either an ad libitum standard control or a lean control leads to distinct transcriptional outputs in both the tissues. Because physiological processes are regulated by the concerted actions of multiple genes in related groups, it may be possible that even though the gene identities appear different on the basis of which control animal is used, their predictive functional output may still be the same, albeit mediated via different genes. To assess the potential functional signaling output of these genes, we assessed their functional clustering using both gene ontology (GO: http://www.geneontology.org/) and KEGG (Kyoto Encyclopedia of Genes and Genomes: http://www.genome.jp/kegg/) pathway analysis. Using functional clustering of the output genes with GO terms, it is again clear in Fig. 2 that in each of the experimental cases the predicted significant GO term functional clusters are very different, depending on which control animals are used. In a similar manner to Figs. 1 and 2, using an even higher level of functional sophistication, i.e., signaling pathway analysis, it is clear in Fig. 3 that when one predicts the signaling behavior of the significantly regulated gene sets in the two tissues that there is very little similarity in the predicted physiological activity when using standard versus lean control animals. Therefore, if one were to attempt to assess the role of gene transcription between various tissues it seems that divergent data could be obtained depending on which control was employed. As many scientists often subject experimental animals to a battery of physiological or behavioral tests, we also compared the differences in the data collected for several physiological variables depending on which control animals were used. Finally, we compared the percentage difference in the respective output index values for body mass, food consumption, plasma hormones, and maze-solving performance when using either lean or standard controls (Fig. 4). If the output index value was greater using the lean control than the output index value using the standard control, then a positive percentage difference was calculated. Negative percentage differences resulted when the output value for lean controls was less than the value for standard controls. It is clear that many of the measured indices (chemical, behavioral, and cognitive) show large percentage differences in their values when the two types of control animals are used, especially with respect to plasma hormones (leptin and growth hormone) and blood-borne energy-regulatory factors such as cholesterol and 3-hydroxybutyrate (Fig. 4). As the relative health status of lean versus standard control animals is divergent, it is not surprising that there are profound differences in experimental outcome, but it is surprising how dramatically distinct some experimental results can be (e.g., transcriptional responses). Therefore it is important for the extrapolation of data from animals to humans that the physiological health of the control animals be considered because important drug effects or physiological actions may be completely missed or wildly exaggerated.
Fig. 1.
Differences and commonalities in gonadal and hippocampal gene expression for Sprague–Dawley rats maintained on low and high energy diets, compared with both healthy “lean” controls and standard ad libitum–fed overweight controls. (A and B) Common and unique significantly altered genes in gonadal tissue (ovaries; A) and hippocampal tissue (B) from rats on 40% caloric restriction, alternate day fasting, and a high fat/glucose diet, compared with standard ad libitum overweight controls and healthy lean controls (20% caloric restriction).
Fig. 2.
Differences and commonalities in gonadal and hippocampal gene ontology (GO) functional groups for Sprague–Dawley rats maintained on low and high energy diets, compared with both healthy “lean” controls and standard ad libitum–fed overweight controls. (A) Common and unique significantly altered GO functional groups in ovaries (A) and hippocampus (B) from rats on 40% caloric restriction, intermittent fasting, and a high fat/glucose diet, compared with standard ad libitum overweight controls and healthy lean controls (20% caloric restriction).
Fig. 3.
Differences and commonalities in gonadal and hippocampal KEGG functional pathways for Sprague–Dawley rats maintained on low and high energy diets, compared with both healthy controls and standard ad libitum–fed overweight controls. (A) Common and unique significantly altered KEGG functional pathways in ovaries (A) and hippocampus (B) from rats on 40% caloric restriction, intermittent fasting, and a high fat/glucose diet, compared with standard ad libitum overweight controls and healthy lean controls (20% caloric restriction).
Fig. 4.
Overview of various physiological differences between standard ad libitum–fed control animals and moderately fed lean animals. The percentage difference for numerous variables (gross physiology, glycemic regulatory factors, lipid regulatory factors, trophic hormones, and learning and memory) for “lean” control Sprague–Dawley rats (20% caloric restriction) compared to “standard” control Sprague–Dawley rats fed ad libitum. LDL, low density lipoprotein; HDL, high density lipoprotein; BDNF, brain-derived neurotrophic factor; and 3-HB, 3-β-hydroxybutyrate.
Research on Standard ad Libitum Overfed Laboratory Animals May Misinform the Design and Outcome of Human Studies
When maintained under the standard laboratory housing conditions of continuous food availability and minimal opportunity for exercise, laboratory animals exhibit increased vulnerability to cancers and neurodegenerative disorders. For example, in a mouse model of prostate cancer, reducing energy intake results in slower growth of the prostate tumors (47), and intermittent caloric restriction reduces the incidence of mammary tumors in a mouse model of breast cancer (75). In a mouse brain tumor model, dietary energy restriction reduces the growth of blood vessels in the tumors and slows the growth of the tumors (48). Compared to animals that eat more sparingly or exercise regularly, animals that overeat and live a sedentary lifestyle exhibit accelerated dysfunction and degeneration of neurons in the brain in experimental models of Alzheimer’s disease (60, 61, 76), Parkinson's disease (63, 77) and stroke (78, 79). Moreover, cognitive function, synaptic plasticity, and neurogenesis (the production of new nerve cells from stem cells) are enhanced by exercise and dietary restriction and are compromised by a sedentary lifestyle (80–83). The adverse effects of a sedentary lifestyle on the brain results from an impaired ability to respond adaptively to stress as indicated by reduced production of neurotrophic factors, protein chaperones, and antioxidant proteins (56, 78, 81).
For many decades, countless studies have investigated the potential health benefits and mechanisms of action of dietary energy restriction regimens. Many studies have shown that caloric restriction can significantly extend lifespan in a variety of species, ranging from flies and worms to mice and rats (84–86). Dietary energy restriction is thought to prolong lifespan by impinging upon fundamental metabolic and cellular signaling pathways including insulin-like signaling, FoxO transcription factors, sirtuins, and peroxisome proliferator-activated receptor. These pathways stimulate the production of various protein chaperones, neurotrophic factors, and antioxidant enzymes, all of which help cells cope with stress and resist disease (see refs. 83, 87 for review). However, other studies have demonstrated that dietary energy restriction has no or even harmful effects on lifespan in different animal models (17, 84, 88–90). Further research is needed, using a standardized approach, to determine the pros and cons of long- and short-term dietary energy restriction. Moreover, it was recently reported that in recombinant inbred mice, there are striking variations in lifespan in response to food restriction, due to differences in genetic background (84). These findings suggest that the role of genetic background in the choice of experimental rodent models would also be important to consider, as different genetic backgrounds are likely to affect experimental outcomes.
Another factor to consider besides body weight, metabolic health, housing conditions, and genetic background of laboratory rodents is whether the rodent model in itself could have experimental drawbacks. Rodents are typically used for experimental research for their ease of use, relatively low cost, rapid breeding, high offspring numbers, and easy maintenance (91). In addition, mice and humans have considerable genome similarities, and more than 90% of the respective genomes can be grouped in terms of corresponding regions (92). However, it is well established that mice and humans have several physiological differences that may render the use of mouse models for the investigation of human physiology and pathophysiology imperfect (91). For example, there are significant differences between rodents and humans in olfactory function (93), reproductive function (94), and digestive function (95).
Two examples of animal models for which the standard housing conditions can be considered healthier than the standard conditions for rodents are canines and nonhuman primates. In both cases the animals are fed portioned meals two or three times daily and may exercise in large cages or open arenas; accordingly, they maintain lower levels of body fat compared to laboratory rats and mice. In contrast to rodents whose lifespans are shortened by the usual housing conditions, the lifespans of dogs (10–20 years for beagles) and monkeys (25–40 years for rhesus macaques) are believed similar to their wild counterparts (96, 97). On the other hand, some organ systems of dogs and monkeys are more sensitive than their rodent counterparts to adverse effects of excessive energy intake, with the cardiovascular system being one clear example, where rodents are resistant to atherosclerosis. This issue of human disease-relevant physiology has been, in part, circumvented in mice by generating transgenic animals that express mutated human genes that cause inherited diseases (cancers, diabetes, Alzheimer’s disease, Parkinson’s disease, and many others).
We suggest that all investigators who employ rodent models in their research should consider how housing conditions, particularly dietary energy intake and level of exercise, might affect the responses of the animals to experimental manipulations, as well as the outcomes measured and their interpretation. Both qualitative and quantitative features of many physiological processes are subject to modification by energy intake and exercise, with recent global gene expression studies (98, 99) indicating that the variables shown in Table 1 represent only the tip of the iceberg of a myriad of signaling and metabolic pathways influenced by energy intake and expenditure.
Conclusions
The beneficial effects of some drugs in animal models might result from their effects on processes associated with an unhealthy lifestyle (increased oxidative stress, inflammation, insulin resistance, etc.) rather than a specific effect of the drug on the disease process. This is a critical issue that should be addressed in future studies in many different branches of biomedical research. Ideally, the efficacy of interventions should be established in animals housed in both the usual overfed, sedentary conditions and more healthy conditions of reduced energy intake and increased exercise, i.e., lean control animals. The standard overfed sedentary control animal is a good model for an increasing fraction of human subjects who are overweight and sedentary, but may be inadequate for preclinical studies relevant to normal weight active humans. Many patients suffering from cancer, vascular disorders, and neurodegenerative disease may in many other ways be relatively fit and cognitively stimulated. Treatments for the latter patients should be tested in healthy animal models. It is not a major expense or hardship to reduce the food intake of laboratory rats and mice and raise them in cages with running wheels. In addition to better informing the development of therapeutic interventions, comparisons between sedentary and fit animals will undoubtedly reveal novel mechanisms by which diet and exercise affect basic biological processes and disease pathogenesis.
Acknowledgments
This work was supported by the Intramural Research Program of the National Institutes of Health, National Institute on Aging.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
References
- 1.Goodrick CL. Effects of long-term voluntary wheel exercise on male and female Wistar rats. I. Longevity, body weight, and metabolic rate. Gerontology. 1980;26:22–33. doi: 10.1159/000212390. [DOI] [PubMed] [Google Scholar]
- 2.Holloszy JO. Exercise increases average longevity of female rats despite increased food intake and no growth retardation. J Gerontol. 1993;48:B97–B100. doi: 10.1093/geronj/48.3.b97. [DOI] [PubMed] [Google Scholar]
- 3.Weindruch R. The retardation of aging by caloric restriction: Studies in rodents and primates. Toxicol Pathol. 1996;24:742–745. doi: 10.1177/019262339602400618. [DOI] [PubMed] [Google Scholar]
- 4.Alessio HM, et al. Exercise improves biomarkers of health and stress in animals fed ad libitum. Physiol Behav. 2005;84:65–72. doi: 10.1016/j.physbeh.2004.10.010. [DOI] [PubMed] [Google Scholar]
- 5.Mattson MP, Wan R. Beneficial effects of intermittent fasting and caloric restriction on the cardiovascular and cerebrovascular systems. J Nutr Biochem. 2005;16:129–137. doi: 10.1016/j.jnutbio.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 6.Heilbronn LK, Ravussin E. Calorie restriction and aging: review of the literature and implications for studies in humans. Am J Clin Nutr. 2003;78:361–369. doi: 10.1093/ajcn/78.3.361. [DOI] [PubMed] [Google Scholar]
- 7.Hursting SD, Lavigne JA, Berrigan D, Perkins SN, Barrett JC. Calorie restriction, aging, and cancer prevention: Mechanisms of action and applicability to humans. Annu Rev Med. 2003;54:131–152. doi: 10.1146/annurev.med.54.101601.152156. [DOI] [PubMed] [Google Scholar]
- 8.Flegal KM, Carroll MD, Ogden CL, Curtin LR. Prevalence and trends in obesity among US adults, 1999-2008. JAMA. 2010;303:235–241. doi: 10.1001/jama.2009.2014. [DOI] [PubMed] [Google Scholar]
- 9.Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047–1053. doi: 10.2337/diacare.27.5.1047. [DOI] [PubMed] [Google Scholar]
- 10.Masoro EJ. Caloric restriction-induced life extension of rats and mice: A critique of proposed mechanisms. Biochim Biophys Acta. 2009;1790:1040–1048. doi: 10.1016/j.bbagen.2009.02.011. [DOI] [PubMed] [Google Scholar]
- 11.Dirx MJ, Zeegers MP, Dagnelie PC, van den Bogaard T, van den Brandt PA. Energy restriction and the risk of spontaneous mammary tumors in mice: A meta-analysis. Int J Cancer. 2003;106:766–770. doi: 10.1002/ijc.11277. [DOI] [PubMed] [Google Scholar]
- 12.Oscai LB, Holloszy JO. Effects of weight changes produced by exercise, food restriction, or overeating on body composition. J Clin Invest. 1969;48:2124–2128. doi: 10.1172/JCI106179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brochmann EJ, Duarte ME, Zaidi HA, Murray SS. Effects of dietary restriction on total body, femoral, and vertebral bone in SENCAR, C57BL/6, and DBA/2 mice. Metabolism. 2003;52:1265–1273. doi: 10.1016/s0026-0495(03)00194-x. [DOI] [PubMed] [Google Scholar]
- 14.Barzilai N, Banerjee S, Hawkins M, Chen W, Rossetti L. Caloric restriction reverses hepatic insulin resistance in aging rats by decreasing visceral fat. J Clin Invest. 1998;101:1353–1361. doi: 10.1172/JCI485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Martin B, et al. Sex-dependent metabolic, neuro-endocrine, and cognitive responses to dietary energy restriction and excess. Endocrinology. 2007;148:4318–4333. doi: 10.1210/en.2007-0161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wan R, Camandola S, Mattson MP. Intermittent food deprivation improves cardiovascular and neuroendocrine responses to stress in rats. J Nutr. 2003;133:1921–1929. doi: 10.1093/jn/133.6.1921. [DOI] [PubMed] [Google Scholar]
- 17.Harper JM, Leathers CW, Austad SN. Does caloric restriction extend life in wild mice? Aging Cell. 2006;5:441–449. doi: 10.1111/j.1474-9726.2006.00236.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Weindruch R, Sohal RS. Seminars in medicine of the Beth Israel Deaconess Medical Center. Caloric intake and aging. N Engl J Med. 1997;337:986–994. doi: 10.1056/NEJM199710023371407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.James DE, Kraegen EW, Chisholm DJ. Effect of exercise training on whole-body insulin sensitivity and responsiveness. J Appl Physiol. 1984;56:1217–1222. doi: 10.1152/jappl.1984.56.5.1217. [DOI] [PubMed] [Google Scholar]
- 20.Bonora E. The metabolic syndrome and cardio-vascular disease. Ann Med. 2006;38:64–80. doi: 10.1080/07853890500401234. [DOI] [PubMed] [Google Scholar]
- 21.Aguiar AS, Jr, et al. Physical exercise improves motor and short-term social memory deficits in reserpinized rats. Brain Res Bull. 2009;79:452–457. doi: 10.1016/j.brainresbull.2009.05.005. [DOI] [PubMed] [Google Scholar]
- 22.Wang ZQ, Bell-Farrow AD, Sonntag W, Cefalu WT. Effect of age and caloric restriction on insulin receptor binding and glucose transporter levels in aging rats. Exp Gerontol. 1997;32:671–684. doi: 10.1016/s0531-5565(97)00054-5. [DOI] [PubMed] [Google Scholar]
- 23.Wing RR, et al. Caloric restriction per se is a significant factor in improvements in glycemic control and insulin sensitivity during weight loss in obese NIDDM patients. Diabetes Care. 1994;17:30–36. doi: 10.2337/diacare.17.1.30. [DOI] [PubMed] [Google Scholar]
- 24.Masoro EJ. Retardation of aging processes by food restriction: An experimental tool. Am J Clin Nutr. 1992;55(6, Suppl):1250S–1252S. doi: 10.1093/ajcn/55.6.1250S. [DOI] [PubMed] [Google Scholar]
- 25.Chandra RK. Immune response in overnutrition. Cancer Res. 1981;41:3795–3796. [PubMed] [Google Scholar]
- 26.Nieman DC, et al. Immune response to obesity and moderate weight loss. Int J Obes Relat Metab Disord. 1996;20:353–360. [PubMed] [Google Scholar]
- 27.Womack J, et al. Obesity and immune cell counts in women. Metabolism. 2007;56:998–1004. doi: 10.1016/j.metabol.2007.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shoelson SE, Lee J, Goldfine AB. Inflammation and insulin resistance. J Clin Invest. 2006;116:1793–1801. doi: 10.1172/JCI29069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yuan M, et al. Reversal of obesity- and diet-induced insulin resistance with salicylates or targeted disruption of Ikkbeta. Science. 2001;293:1673–1677. doi: 10.1126/science.1061620. [DOI] [PubMed] [Google Scholar]
- 30.Friedman JM, Halaas JL. Leptin and the regulation of body weight in mammals. Nature. 1998;395:763–770. doi: 10.1038/27376. [DOI] [PubMed] [Google Scholar]
- 31.Kiefer FW, et al. Osteopontin expression in human and murine obesity: Extensive local up-regulation in adipose tissue but minimal systemic alterations. Endocrinology. 2008;149:1350–1357. doi: 10.1210/en.2007-1312. [DOI] [PubMed] [Google Scholar]
- 32.Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor-alpha: Direct role in obesity-linked insulin resistance. Science. 1993;259:87–91. doi: 10.1126/science.7678183. [DOI] [PubMed] [Google Scholar]
- 33.Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444:860–867. doi: 10.1038/nature05485. [DOI] [PubMed] [Google Scholar]
- 34.Purnell JQ, et al. Enhanced cortisol production rates, free cortisol, and 11beta-HSD-1 expression correlate with visceral fat and insulin resistance in men: Effect of weight loss. Am J Physiol Endocrinol Metab. 2009;296:E351–E357. doi: 10.1152/ajpendo.90769.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Seckl JR, Walker BR. Minireview: 11beta-hydroxysteroid dehydrogenase type 1- a tissue-specific amplifier of glucocorticoid action. Endocrinology. 2001;142:1371–1376. doi: 10.1210/endo.142.4.8114. [DOI] [PubMed] [Google Scholar]
- 36.Morton NM, et al. Improved lipid and lipoprotein profile, hepatic insulin sensitivity, and glucose tolerance in 11beta-hydroxysteroid dehydrogenase type 1 null mice. J Biol Chem. 2001;276:41293–41300. doi: 10.1074/jbc.M103676200. [DOI] [PubMed] [Google Scholar]
- 37.Masuzaki H, et al. A transgenic model of visceral obesity and the metabolic syndrome. Science. 2001;294:2166–2170. doi: 10.1126/science.1066285. [DOI] [PubMed] [Google Scholar]
- 38.Engeli S, et al. Regulation of 11beta-HSD genes in human adipose tissue: Influence of central obesity and weight loss. Obes Res. 2004;12:9–17. doi: 10.1038/oby.2004.3. [DOI] [PubMed] [Google Scholar]
- 39.Valsamakis G, et al. 11beta-hydroxysteroid dehydrogenase type 1 activity in lean and obese males with type 2 diabetes mellitus. J Clin Endocrinol Metab. 2004;89:4755–4761. doi: 10.1210/jc.2003-032240. [DOI] [PubMed] [Google Scholar]
- 40.Arkan MC, et al. IKK-beta links inflammation to obesity-induced insulin resistance. Nat Med. 2005;11:191–198. doi: 10.1038/nm1185. [DOI] [PubMed] [Google Scholar]
- 41.Solinas G, et al. JNK1 in hematopoietically derived cells contributes to diet-induced inflammation and insulin resistance without affecting obesity. Cell Metab. 2007;6:386–397. doi: 10.1016/j.cmet.2007.09.011. [DOI] [PubMed] [Google Scholar]
- 42.Karalis KP. Mechanisms of obesity and related pathology: Linking immune responses to metabolic stress. FEBS J. 2009;276:5747–5754. doi: 10.1111/j.1742-4658.2009.07304.x. [DOI] [PubMed] [Google Scholar]
- 43.Lo J, et al. Effects of TNF-alpha neutralization on adipocytokines and skeletal muscle adiposity in the metabolic syndrome. Am J Physiol Endocrinol Metab. 2007;293:E102–E109. doi: 10.1152/ajpendo.00089.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bernstein LE, Berry J, Kim S, Canavan B, Grinspoon SK. Effects of etanercept in patients with the metabolic syndrome. Arch Intern Med. 2006;166:902–908. doi: 10.1001/archinte.166.8.902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ross MH, Bras G. Lasting influence of early caloric restriction on prevalence of neoplasms in the rat. J Natl Cancer Inst. 1971;47:1095–1113. [PubMed] [Google Scholar]
- 46.Albanes D. Total calories, body weight, and tumor incidence in mice. Cancer Res. 1987;47:1987–1992. [PubMed] [Google Scholar]
- 47.Bonorden MJ, et al. Cross-sectional analysis of intermittent versus chronic caloric restriction in the TRAMP mouse. Prostate. 2009;69:317–326. doi: 10.1002/pros.20878. [DOI] [PubMed] [Google Scholar]
- 48.Mukherjee P, El-Abbadi MM, Kasperzyk JL, Ranes MK, Seyfried TN. Dietary restriction reduces angiogenesis and growth in an orthotopic mouse brain tumour model. Br J Cancer. 2002;86:1615–1621. doi: 10.1038/sj.bjc.6600298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bunk B, Zhu P, Klinga K, Berger MR, Schmähl D. Influence of reducing luxury calories in the treatment of experimental mammary carcinoma. Br J Cancer. 1992;65:845–851. doi: 10.1038/bjc.1992.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hikita H, Vaughan J, Babcock K, Pitot HC. Short-term fasting and the reversal of the stage of promotion in rat hepatocarcinogenesis: Role of cell replication, apoptosis, and gene expression. Toxicol Sci. 1999;52:2S17–2S23. doi: 10.1093/toxsci/52.2.17. [DOI] [PubMed] [Google Scholar]
- 51.Qu B, Halliwell B, Ong CN, Lee BL, Li QT. Caloric restriction prevents oxidative damage induced by the carcinogen clofibrate in mouse liver. FEBS Lett. 2000;473:85–88. doi: 10.1016/s0014-5793(00)01506-4. [DOI] [PubMed] [Google Scholar]
- 52.Hart RW, et al. Modulation of chemical toxicity by modification of caloric intake. Adv Exp Med Biol. 1992;322:73–81. doi: 10.1007/978-1-4684-7953-9_7. [DOI] [PubMed] [Google Scholar]
- 53.Manjgaladze M, et al. Effects of caloric restriction on rodent drug and carcinogen metabolizing enzymes: Implications for mutagenesis and cancer. Mutat Res. 1993;295:201–222. doi: 10.1016/0921-8734(93)90021-t. [DOI] [PubMed] [Google Scholar]
- 54.Lee JJ, Feng L. Randomized phase II designs in cancer clinical trials: Current status and future directions. J Clin Oncol. 2005;23:4450–4457. doi: 10.1200/JCO.2005.03.197. [DOI] [PubMed] [Google Scholar]
- 55.Thompson HJ, et al. Effect of dietary energy restriction on vascular density during mammary carcinogenesis. Cancer Res. 2004;64:5643–5650. doi: 10.1158/0008-5472.CAN-04-0787. [DOI] [PubMed] [Google Scholar]
- 56.Rose DP, Komninou D, Stephenson GD. Obesity, adipocytokines, and insulin resistance in breast cancer. Obes Rev. 2004;5:153–165. doi: 10.1111/j.1467-789X.2004.00142.x. [DOI] [PubMed] [Google Scholar]
- 57.O’Malley RL, Taneja SS. Obesity and prostate cancer. Can J Urol. 2006;13:2S11–2S17. [PubMed] [Google Scholar]
- 58.Birmingham JM, Busik JV, Hansen-Smith FM, Fenton JI. Novel mechanism for obesity-induced colon cancer progression. Carcinogenesis. 2009;30:690–697. doi: 10.1093/carcin/bgp041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Götz J, Ittner LM. Animal models of Alzheimer's disease and frontotemporal dementia. Nat Rev Neurosci. 2008;9:532–544. doi: 10.1038/nrn2420. [DOI] [PubMed] [Google Scholar]
- 60.Halagappa VK, et al. Intermittent fasting and caloric restriction ameliorate age-related behavioral deficits in the triple-transgenic mouse model of Alzheimer's disease. Neurobiol Dis. 2007;26:212–220. doi: 10.1016/j.nbd.2006.12.019. [DOI] [PubMed] [Google Scholar]
- 61.Lazarov O, et al. Environmental enrichment reduces Abeta levels and amyloid deposition in transgenic mice. Cell. 2005;120:701–713. doi: 10.1016/j.cell.2005.01.015. [DOI] [PubMed] [Google Scholar]
- 62.Cao D, Lu H, Lewis TL, Li L. Intake of sucrose-sweetened water induces insulin resistance and exacerbates memory deficits and amyloidosis in a transgenic mouse model of Alzheimer disease. J Biol Chem. 2007;282:36275–36282. doi: 10.1074/jbc.M703561200. [DOI] [PubMed] [Google Scholar]
- 63.Duan W, Mattson MP. Dietary restriction and 2-deoxyglucose administration improve behavioral outcome and reduce degeneration of dopaminergic neurons in models of Parkinson's disease. J Neurosci Res. 1999;57:195–206. doi: 10.1002/(SICI)1097-4547(19990715)57:2<195::AID-JNR5>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- 64.Yu ZF, Mattson MP. Dietary restriction and 2-deoxyglucose administration reduce focal ischemic brain damage and improve behavioral outcome: evidence for a preconditioning mechanism. J Neurosci Res. 1999;57:830–839. [PubMed] [Google Scholar]
- 65.Pothakos K, Kurz MJ, Lau YS. Restorative effect of endurance exercise on behavioral deficits in the chronic mouse model of Parkinson's disease with severe neurodegeneration. BMC Neurosci. 2009;10:6. doi: 10.1186/1471-2202-10-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pang TY, Stam NC, Nithianantharajah J, Howard ML, Hannan AJ. Differential effects of voluntary physical exercise on behavioral and brain-derived neuro-trophic factor expression deficits in Huntington's disease transgenic mice. Neuroscience. 2006;141:569–584. doi: 10.1016/j.neuroscience.2006.04.013. [DOI] [PubMed] [Google Scholar]
- 67.Lim GP, et al. The curry spice curcumin reduces oxidative damage and amyloid pathology in an Alzheimer transgenic mouse. J Neurosci. 2001;21:8370–8377. doi: 10.1523/JNEUROSCI.21-21-08370.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Quinn JF, et al. Chronic dietary alpha-lipoic acid reduces deficits in hippocampal memory of aged Tg2576 mice. Neurobiol Aging. 2007;28:213–225. doi: 10.1016/j.neurobiolaging.2005.12.014. [DOI] [PubMed] [Google Scholar]
- 69.Cheng YD, Al-Khoury L, Zivin JA. Neuroprotection for ischemic stroke: Two decades of success and failure. NeuroRx. 2004;1:36–45. doi: 10.1602/neurorx.1.1.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lodder J. Neuroprotection in stroke: Analysis of failure, and alternative strategies. Neurosci Res Commun. 2000;26:173–179. [Google Scholar]
- 71.Hoyte L, Barber PA, Buchan AM, Hill MD. The rise and fall of NMDA antagonists for ischemic stroke. Curr Mol Med. 2004;4:131–136. doi: 10.2174/1566524043479248. [DOI] [PubMed] [Google Scholar]
- 72.Jordán J, Segura T, Brea D, Galindo MF, Castillo J. Inflammation as therapeutic objective in stroke. Curr Pharm Des. 2008;14:3549–3564. doi: 10.2174/138161208786848766. [DOI] [PubMed] [Google Scholar]
- 73.Martin B, et al. Gonadal transcriptome alterations in response to dietary energy intake: Sensing the reproductive environment. PLoS One. 2009;4:e4146. doi: 10.1371/journal.pone.0004146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Martin B, et al. Conserved and differential effects of dietary energy intake on the hippocampal transcriptomes of females and males. PLoS One. 2008;3:e2398. doi: 10.1371/journal.pone.0002398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Cleary MP, et al. Weight-cycling decreases incidence and increases latency of mammary tumors to a greater extent than does chronic caloric restriction in mouse mammary tumor virus-transforming growth factor-alpha female mice. Cancer Epidemiol Biomarkers Prev. 2002;11:836–843. [PubMed] [Google Scholar]
- 76.Adlard PA, Perreau VM, Pop V, Cotman CW. Voluntary exercise decreases amyloid load in a transgenic model of Alzheimer's disease. J Neurosci. 2005;25:4217–4221. doi: 10.1523/JNEUROSCI.0496-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Smith AD, Zigmond MJ. Can the brain be protected through exercise? Lessons from an animal model of parkinsonism. Exp Neurol. 2003;184:31–39. doi: 10.1016/j.expneurol.2003.08.017. [DOI] [PubMed] [Google Scholar]
- 78.Arumugam TV, et al. Age and energy intake interact to modify cell stress pathways and stroke outcome. Ann Neurol. 2010 doi: 10.1002/ana.21798. 10.1002/ana.21798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ploughman M, Attwood Z, White N, Doré JJ, Corbett D. Endurance exercise facilitates relearning of forelimb motor skill after focal ischemia. Eur J Neurosci. 2007;25:3453–3460. doi: 10.1111/j.1460-9568.2007.05591.x. [DOI] [PubMed] [Google Scholar]
- 80.Stranahan AM, et al. Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons. Nat Neurosci. 2008;11:309–317. doi: 10.1038/nn2055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Stranahan AM, et al. Voluntary exercise and caloric restriction enhance hippocampal dendritic spine density and BDNF levels in diabetic mice. Hippocampus. 2009;19:951–961. doi: 10.1002/hipo.20577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.van Praag H. Exercise and the brain: Something to chew on. Trends Neurosci. 2009;32:283–290. doi: 10.1016/j.tins.2008.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Martin B, Mattson MP, Maudsley S. Caloric restriction and intermittent fasting: Two potential diets for successful brain aging. Ageing Res Rev. 2006;5:332–353. doi: 10.1016/j.arr.2006.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Liao CY, Rikke BA, Johnson TE, Diaz V, Nelson JF. Genetic variation in the murine lifespan response to dietary restriction: From life extension to life shortening. Aging Cell. 2010;9:92–95. doi: 10.1111/j.1474-9726.2009.00533.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.McCay CM, Crowell MF, Maynard LA. The effect of retarded growth upon the length of life and upon the ultimate body size. J Nutr. 1935;10:63–79. [PubMed] [Google Scholar]
- 86.Masoro EJ. Overview of caloric restriction and ageing. Mech Ageing Dev. 2005;126:913–922. doi: 10.1016/j.mad.2005.03.012. [DOI] [PubMed] [Google Scholar]
- 87.Martin B, et al. Caloric restriction: Impact upon pituitary function and reproduction. Ageing Res Rev. 2008;7:209–224. doi: 10.1016/j.arr.2008.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Turturro A, et al. Growth curves and survival characteristics of the animals used in the Biomarkers of Aging Program. J Gerontol A Biol Sci Med Sci. 1999;54:B492–B501. doi: 10.1093/gerona/54.11.b492. [DOI] [PubMed] [Google Scholar]
- 89.Forster MJ, Morris P, Sohal RS. Genotype and age influence the effect of caloric intake on mortality in mice. FASEB J. 2003;17:690–692. doi: 10.1096/fj.02-0533fje. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Harrison DE, Archer JR. Genetic differences in effects of food restriction on aging in mice. J Nutr. 1987;117:376–382. doi: 10.1093/jn/117.2.376. [DOI] [PubMed] [Google Scholar]
- 91.Sundberg JP, King LE., Jr Mouse models for the study of human hair loss. Dermatol Clin. 1996;14:619–632. doi: 10.1016/s0733-8635(05)70389-2. [DOI] [PubMed] [Google Scholar]
- 92.Waterston RH, et al. Mouse Genome Sequencing Consortium. Initial sequencing and comparative analysis of the mouse genome. Nature. 2002;420:520–562. doi: 10.1038/nature01262. [DOI] [PubMed] [Google Scholar]
- 93.Godfrey PA, Malnic B, Buck LB. The mouse olfactory receptor gene family. Proc Natl Acad Sci USA. 2004;101:2156–2161. doi: 10.1073/pnas.0308051100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Finn CA. Why do women menstruate? Historical and evolutionary review. Eur J Obstet Gynecol Reprod Biol. 1996;70:3–8. doi: 10.1016/s0301-2115(96)02565-1. [DOI] [PubMed] [Google Scholar]
- 95.Yamamoto K, Matsunaga S, Matsui M, Takeda N, Yamatodani A. Pica in mice as a new model for the study of emesis. Methods Find Exp Clin Pharmacol. 2002;24:135–138. doi: 10.1358/mf.2002.24.3.802297. [DOI] [PubMed] [Google Scholar]
- 96.Cotman CW, Head E. The canine (dog) model of human aging and disease: Dietary, environmental and immunotherapy approaches. J Alzheimers Dis. 2008;15:685–707. doi: 10.3233/jad-2008-15413. [DOI] [PubMed] [Google Scholar]
- 97.Mattison JA, Roth GS, Lane MA, Ingram DK. Dietary restriction in aging nonhuman primates. Interdiscip Top Gerontol. 2007;35:137–158. doi: 10.1159/000096560. [DOI] [PubMed] [Google Scholar]
- 98.Zahn JM, et al. AGEMAP: A gene expression database for aging in mice. PLoS Genet. 2007;3:e201. doi: 10.1371/journal.pgen.0030201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Xu X, et al. Gene expression atlas of the mouse central nervous system: Impact and interactions of age, energy intake and gender. Genome Biol. 2007;8:R234. doi: 10.1186/gb-2007-8-11-r234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Pitts GC. Body composition in the rat: Interactions of exercise, age, sex, and diet. Am J Physiol. 1984;246:R495–R501. doi: 10.1152/ajpregu.1984.246.4.R495. [DOI] [PubMed] [Google Scholar]
- 101.Seymour EM, Parikh RV, Singer AAM, Bolling SF. Moderate calorie restriction improves cardiac remodeling and diastolic dysfunction in the Dahl-SS rat. J Mol Cell Cardiol. 2006;41:661–668. doi: 10.1016/j.yjmcc.2006.07.012. [DOI] [PubMed] [Google Scholar]
- 102.Mueller PJ, Hasser EM. Putative role of the NTS in alterations in neural control of the circulation following exercise training in rats. Am J Physiol Regul Integr Comp Physiol. 2006;290:R383–R392. doi: 10.1152/ajpregu.00455.2005. [DOI] [PubMed] [Google Scholar]