Abstract
Background
Isolated non-compaction cardiomyopathy (NCCM) was first described in 1984. This disorder, a primary genetic cardiomyopathy, is now attracting increased attention.
Method
The current state of the epidemiology, pathogenesis, pathophysiology, clinical features, diagnosis, treatment, and prognosis of NCCM are discussed on the basis of a review of selected literature as well as the authors’ personal experience.
Results
The pathogenesis of NCCM is thought to involve a genetically determined disturbance of the myocardial compaction process during fetal endomyocardial morphogenesis. It is not accompanied by any other cardiac anomalies. Echocardiography is the diagnostic method of choice. The diagnosis is based on the following echocardiographic criteria: the presence of at least 4 prominent trabeculations and deep intertrabecular recesses, blood flow from the ventricular cavity into the intertrabecular recesses, and a typical bilaminar structure of the affected portion of the left ventricular myocardium. NCCM can also be diagnosed with magnetic resonance imaging of the heart. The clinical severity of NCCM is variable; its manifestations include heart failure, thromboembolic events, and arrhythmias. The treatment is symptom-based. Patients with symptomatic NCCM have a poor prognosis.
Conclusion
NCCM is a type of cardiomyopathy that was first described 25 years ago. Its molecular genetic basis is not yet fully clear, and the same is true of its diagnosis, treatment, and prognosis. Further study of these matters is needed.
Isolated non-compaction cardiomyopathy (NCCM) and its typical echocardiographic appearance were first described in 1984 by Engberding and Bender (1). Before then, this type of myocardial abnormality had been recognized only in cases of congenital heart defects with an intact ventricular septum and atresia of the semilunar valves (2–4). Engberding and Bender themselves called the disorder “persistence of isolated myocardial sinusoids,” attributing it to an abnormal lack of regression of sinusoids during cardiac embryogenesis. In 1990, Chin et al. (5) suggested that it be renamed “isolated non-compaction of the left ventricular myocardium” on the basis of an improved understanding of developmental physiology. This designation implies a disturbance of the compaction process normally occurring as part of the embryonal morphogenesis of the myocardium.
NCCM is a heart-muscle disorder that is still little known among physicians. The purpose of this article is to provide an overview of the current knowledge of the epidemiology, pathogenesis, pathophysiology, clinical manifestations, diagnostic evaluation, treatment, and prognosis of this type of cardiomyopathy. For this purpose, we will present a review of the scientific literature on the subject from 1984 to 2009, as well as the few relevant recommendations of the cardiological societies regarding NCCM.
Clinical features
NCCM can arise in either children or adults. The age of onset of its manifestations is highly variable (Tables 1 and 2). Rarely, the disorder is not diagnosed until old age; in one reported case, a 94-year-old man was found to have NCCM after presenting to medical attention with a transient ischemic attack (TIA) (6).
Table 1. Clinical findings in adult patients with isolated non-compaction cardiomyopathy.
| Adult patients | |||||
| Oechslin et al. (15) | Sengupta et al. (16) | Murphy et al. (17) | Lofiego et al. (e52) | Stöllberger et al. (22) | |
| Number of patients | 34 | 32 | 45 | 65 | 86 |
| Age (median) at diagnosis | 40 years | 49 years | 37 years (mean) | 47 years (mean) | 52 years |
| Percent male | 74% | 53% | 62% | 37% | 76% |
| Familial clustering | 18% | 15% | |||
| Length of follow-up | up to 11 years | up to 15 years | up to 8 years | ||
| Clinical manifestations | |||||
| Heart failure | 68% | 62,5% | 62% | 61% | 70% |
| Arterial embolic event | 21% | 4% | |||
| Pulmonary embolism | 9% | ||||
| Ventricular thrombus | 9% | 6% | 1% | ||
| Dysmorphic facies | 0% | ||||
| Neuromuscular disorder | 9% | 38% | |||
| Death | 35% | 2% | 22% | ||
| Heart transplantation | 12% | 1% | |||
| ECG | |||||
| A-V block | 56% | 29% | 32% | 26% | |
| Wolff-Parkinson-White syndrome | 0% | 2% | |||
| Ventricular tachycardia | 41% | 20% | |||
| Left ventricular hypertrophy | |||||
Table 2. Clinical findings in pediatric patients with isolated non-compaction cardiomyopathy.
| Pediatric patients | ||||
| Chin et al. (5) | Ichida et al. (10) | Alehan et al. (e53) | Wald et al. (e54) | |
| Number of patients | 8 | 27 | 9 | 22 |
| Age (median) at diagnosis | 7 years | 5 years | 9 years | 3.9 years |
| Percent male | 63% | 56% | 89% | 40% |
| Familial clustering | 50% | 44% | 0% | 18% |
| Length of follow-up | up to 5 years | up to 17 years | up to 5 years | up to 16 years |
| Clinical manifestations | ||||
| Heart failure | 63% | 30% | 55% | 54% |
| Arterial embolic event | 38% | 0% | 0% | 0% |
| Pulmonary embolism | 0% | 7% | 0% | 0% |
| Ventricular thrombus | 25% | 0% | 0% | 0% |
| Dysmorphic facies | 38% | 33% | 10% | |
| Neuromuscular disorder | 0% | 5% | ||
| Death | 38% | 7% | 22% | 14% |
| Heart transplantation | 0% | 4% | 0% | 9% |
| ECG | ||||
| A-V block | 25% | 15% | 0% | 5% |
| Wolff-Parkinson-White syndrome | 13% | 15% | 0% | 5% |
| Ventricular tachydardia | 38% | 0% | 0% | 15% |
| Left ventricular hypertrophy | 4% | 66% | 0% | |
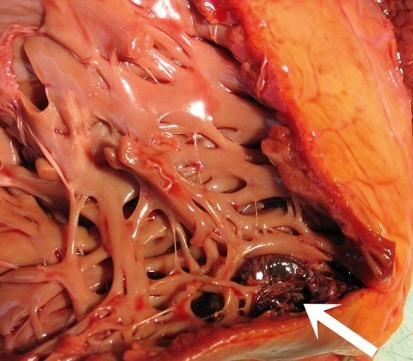
The main clinical feature of NCCM is heart failure, with varying manifestations depending on the severity of the disease (7). Thromboembolic events are particularly common in persons who have both NCCM and atrial fibrillation (8, 9). Intraventricular thrombi are also found in some cases. These are due to an impairment of left-ventricular function, with stasis of blood in the deep intertrabecular recesses (figure 1).
Figure 1.
Dissected autopsy specimen: the heart of a patient with isolated non-compaction cardiomyopathy. There is extensive trabecula formation in the left ventricular wall. The arrow points to a thrombus in the trabecular meshwork
The ECG shows non-specific changes and multiple types of arrhythmia: In particular, there are non-specific changes of the ST segments and the T waves, bundle branch block of various types, and atrial fibrillation as well as ventricular tachyarrhythmias, which can be either self-limited or persistent. Wolff-Parkinson-White (WPW) syndrome has been described mostly among affected children (8, 10, 11). Other types of supraventricular tachycardia (AVNRT) have also been reported (12).
The cardiac laboratory parameters are usually normal in patients with NCCM. In one study, elevated troponin-T levels were seen in patients with NCCM who had accompanying neuromuscular disturbances and a poor clinical outcome (13).
Examination by a neurologist revealed neuromuscular disturbances in 82% of a series of 49 patients with NCCM (14), but no neurological abnormalities were reported in other studies (15–17).
An acquired form of NCCM was described in a single patient who suffered from Duchenne-type muscular dystrophy (18).
Epidemiology
In a study of children with primary cardiomyopathy of all types, NCCM was present in 9.2%. It was thus the third most common type of primary cardiomyopathy, after dilated cardiomyopathy (DCM) and hypertrophic cardiomyopathy (HCM) (19).
The prevalence of this disease in adults remains unclear. In observational studies, NCCM has been found in 0.014% to 0.26% of all adults referred to an echocardiography laboratory (15, 20). The incidence of NCCM in the general population has been estimated at 0.05% to 0.25% per year (21, 22). The diagnosis is presumably often missed, because the disease is still not as well known as it should be among physicians at large.
Cardiac embryology and pathogenesis
The development of the heart is a complex process, regulated at the molecular level by special signaling molecules and tissue-specific transcription factors (23, 24).
Studies have shown that de novo addition of myocardial cells occurs at multiple times during embryogenesis, first from the “primary cardiac field” (myocardial precursor cells in the anterolateral region of the embryo) and later from the “secondary (or anterior) cardiac field” (mesodermal cells in the region of the distal outflow tract) (25, e1). The myocardium develops from two different layers, a trabecular layer and a compact layer. The endocardium forms the cellular substrate of the trabecular layer, while the compact layer develops subepicardially (e2). Before the coronary vessels develop, the embryonal myocardium consists of a “spongy” meshwork of trabecular myocardial fibers and intertrabecular recesses that communicate with the cavum of the ventricle to receive their blood supply (e2– e4). Then, in the 5th to the 8th week of embryonal development of the human myocardium, the ventricular myocardium gradually becomes compacted, and the larger intertrabecular spaces are transformed into capillaries, while the smaller ones disappear. This process of compaction proceeds from the epicardium to the endocardium, and from the base of the heart to its apex. The epicardially lying coronary vessels develop in tandem (e2– e4). An abnormal arrest of this complex embryonal process of endomyocardial morphogenesis is assumed to be the pathogenetic mechanism that underlies NCCM (7, e4, e5).
Other putative pathogenetic mechanisms for NCCM have been proposed in other publications, including myocardial dissection, myocardial tearing due to dilatation, metabolic defects, and compensatory hypervascularization (14).
Genetics
NCCM can occur sporadically, but familial clustering has also been described in up to 44% of cases (10, e6, e7). In one case study, the familial occurrence of NCCM was found to be associated with a mutation in the G4.5 gene on chromosome Xq28, which encodes tafazzin, an enzyme participating in cardiolipin metabolism (8). Further details, and a list of the genetic mutations that have been described in NCCM, can be found in the supplement to this article (see e-Supplement).
Pathology
Pathoanatomical studies of NCCM reveal a marked trabecular meshwork with many intertrabecular recesses in the involved mural segments of the ventricular myocardium (figure 1) (e17– e19).
The intertrabecular recesses are lined with endothelium (5), end blindly in the external compact layer, and have no connection to the coronary circulation (e17). In normal development, myocardial compaction occurs to a greater extent in the left ventricular than in the right ventricular myocardium; it can thus be difficult to distinguish a normal variant of the right ventricular myocardium from pathological non-compaction (15, e20). This explains the recommendation that the expression “areas of non-compaction” should not be used at all to describe findings in the right ventricular wall (e20).
Prominent trabecula were found in the left ventricle at autopsy in up to 70% of a group of subjects who had not suffered from heart disease during life (e21), but more than three trabecula were found in only 4%. On the basis of these data, Stöllberger et al. defined pathological left ventricular trabeculation as the echocardiographically demonstrable presence of more than three trabecula apical to the papillary muscles (e22).
Pathophysiology
Coronary angiography reveals no abnormalities in patients with NCCM (1, 5, e23), but positron emission tomography (PET) shows a diminished reserve of coronary blood flow in the compact and non-compact myocardial segments of the left ventricle, presumably because of impaired microcirculation. Similar findings are obtained with single photon emission computerized tomography (SPECT) (e24). Impaired microcirculation can lead to impaired left ventricular contraction and can account for the histologically demonstrable subendocardial fibrosis (11, e20, e23, e25). Furthermore, marked trabeculation can impair the diastolic function of the left ventricle as well, with abnormal relaxation and restricted filling (e3). These systolic and diastolic disturbances of left ventricular function, if severe enough, can lead to the clinical manifestations of heart failure that are seen in many patients with NCCM.
Diagnostic evaluation
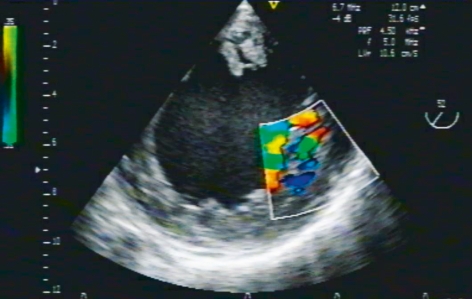
Echocardiography is the diagnostic test of choice for NCCM (Figure 2 and 3) (1, 5, 7, 16, e18, e20, e26). The diagnostic criteria of Jenni and Stöllberger are reproduced in Box 1. (9, 14). At present, the diagnosis of NCCM is probably often missed; in one report, there was a delay of three and a half years from the onset of symptoms until the correct diagnosis was made (e19). On the other hand, there is a risk of a false-positive diagnosis of NCCM as well. To prevent this, non-compact myocardial areas should be carefully distinguished ultrasonographically from aberrant chordae tendineae, papillary muscles, and muscular bands. Transesophageal echocardiography can be helpful for this purpose.
Figure 2.
TEE demonstration of the short axis of the left ventricle, visualized by transgastric insonation. Color Doppler flow ultrasonography reveals blood flow between the extensive trabecula in this patient with isolated NCCM
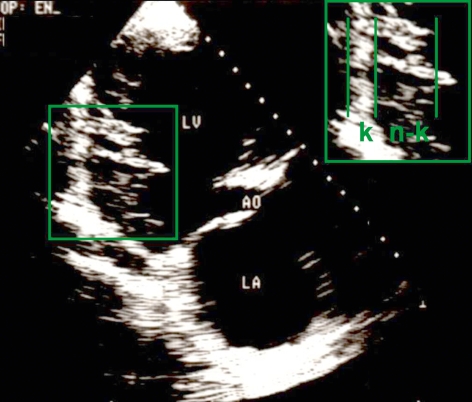
Figure 3.
Transthoracic echocardiographic view of the long axis of the heart in a patient with isolated NCCM, revealing the compact and non-compact layers of the left ventricle. c, compact layer; nc, non-compact layer
Box 1. Echocardiographic criteria for the diagnosis of isolated non-compaction cardiomyopathy.
There are at least four prominent trabecula and deep intertrabecular recesses.
Blood flow between the cavum of the left ventricle and the recesses is demonstrable with color Doppler ultrasonography (figure 2) or through the use of ultrasonographic contrast medium.
The non-compact mural segments have a typical bilaminar structure, and the non-compact subendocardial layer is at least twice as thick as the compact subepicardial layer in systole (figure 3). Non-compaction is seen mainly at the cardiac apex and in the inferior, central, and lateral portions of the left ventricular wall.
No other cardiac abnormalities are present.
Newer echocardiographic techniques can also aid in the diagnostic evaluation of certain cases. For example, real-time three-dimensional echocardiography seems to facilitate the identification of trabecula (e27, e28). In one case of NCCM, it was possible to demonstrate ventricular dyssynchronization through the use of tissue Doppler and the “strain rate” (i.e., the difference in velocity between two neighboring points on the myocardium) (e29). Speckle tracking (a procedure for the recognition of characteristic patterns in the texture of the myocardium) has been used to show that the apical and basal segments of the left ventricle rotate in the same direction in patients with NCCM, rather than in opposite directions, as in persons without cardiac disease (e30). This finding is of major significance for left ventricular function.
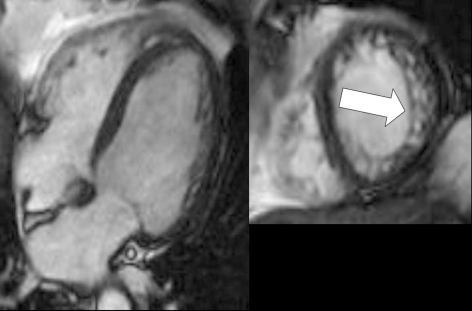
Cardiac magnetic resonance imaging (MRI) is a further method that can be used to diagnose NCCM accurately (figure 4), although artefacts can be produced by breathing or by cardiac arrhythmia. In cardiac MRI, a diastolic quotient of more than 2.3 between the non-compact and compact layers is required for the diagnosis of a bilaminar myocardial wall structure (e31). As physicians become more familiar with NCCM, cardiac MRI is likely to be used more frequently to establish the diagnosis (e23, e32– e34). Initial studies have been performed on the quantification of trabeculation with cardiac MRI and its correlation with the clinical severity of the disease (e35).
Figure 4.
Magnetic resonance imaging of the heart (cardiac MRI) in a man with isolated non-compaction cardiomyopathy. Cine-SSFP images taken along the long and short axes are shown (left and right, respectively); arrow: increased trabeculation
Multi-slice computerized tomography (CT) can also be used to identify non-compact areas of the left ventricle, but there are as yet no generally accepted criteria for this purpose (e36).
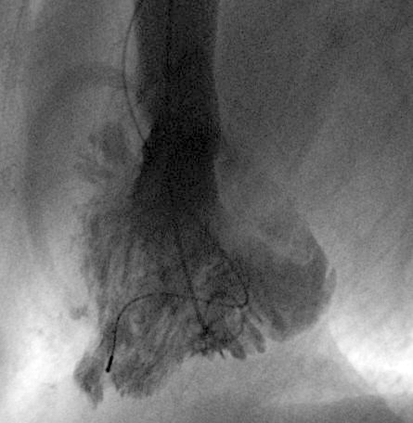
For adults suspected of having NCCM who also have reduced left ventricular function, an invasive diagnostic evaluation is usually indicated, so that coronary heart disease or other causes of left ventricular dysfunction can be ruled out. In such cases, NCCM can be diagnosed with left ventriculography (figure 5) (1, 7).
Figure 5.
Left ventriculogram of a woman with isolated non-compaction cardiomyopathy, showing excessive trabeculation.
Differential diagnosis
NCCM is not uncommonly misdiagnosed as apical HCM. The correct diagnosis can be achieved in such cases by application of the typical echocardiographic criteria for NCCM. In cases of advanced NCCM, the distinction between this entity and DCM is of particular importance. A comparison of the echocardiographic findings of 32 patients with NCCM and 20 with DCM is shown in Table 3 (16).
The other entities listed here in the differential diagnosis of NCCM are derived from a review that analyzed published cases of patients whose NCCM was not correctly diagnosed on the initial echocardiographic study (e37). The findings are displayed in Box 2.
Box 2. The differential diagnosis of isolated non-compaction cardiomyopathy (e37).
Hypertrophic cardiomyopathy
Localized left-ventricular hypertrophy
Dilated cardiomyopathy
Endocardial fibroelastosis
Myo-, pericarditis
Restrictive cardiomyopathy
Left-ventricular thrombi
Aberrant chordae tendineae
Intramyocardial hematoma/abscess
Cardiac metastases
Treatment
The main emphasis of the clinical treatment of patients with NCCM is on the treatment of heart failure and arrhythmias and the prevention of thromboembolic events. The treatment of heart failure in patients with NCCM follows the general guidelines for heart failure treatment (7, e38). In cases with left bundle branch block and severe left ventricular dysfunction, resynchronization treatment may be used. Heart transplantation has been performed successfully for end-stage heart failure (11, e39, e40).
For patients with persistent ventricular tachycardia or patients who have survived an episode of cardiac arrest, the implantation of an implantable cardiac defibrillator (ICD) is indicated as secondary prophylaxis. The American Heart Association’s new guidelines on ICD implantation permit the primary prophylactic implantation of an ICD in patients with NCCM (level IIb evidence) (e41).
In patients with WPW syndrome or other types of supraventricular tachycardia, the indication for an ablative procedure should be assessed (12, e42).
Long-term oral anticoagulation is indicated in particular for patients with atrial fibrillation, impaired left ventricular function, or demonstrated intracardiac thrombi (15, 17, e5). In a study of NCCM patients among whom a large percentage (60%) were anticoagulated, the frequency of thromboembolic complications was only 4% (17), while the reported event rates in other studies were as high as 38% (5, 15).
Because the disease can run in families, the affected patients’ relatives should undergo screening (14, 16, 17). It is advisable for patients to be examined by a neurologist in order to rule out any accompanying neuromuscular disturbances (10, 14).
Prognosis
The prognosis of patients with NCCM is determined by the extent and degree of progression of heart failure, by the severity of any accompanying arrhythmia, and by the occurrence of thromboembolic events. Oechslin et al. followed 34 patients with NCCM over a mean period of 44 months; over this time, 53% developed heart failure, 41% developed a ventricular tachycardia, and 24% had thromboembolic events (table 1) (15). Six patients suffered a sudden cardiac death, and four died of heart failure. Four underwent cardiac transplantation, and four further patients received an ICD. In studies of children with NCCM, only 30% had clinical manifestations of heart failure at the time of diagnosis, but 90% developed heart failure over the ensuing ten years (10). Recently published data from the NCCM registry of the Italian Society of Echocardiography suggest that the prognosis of patients with symptomatic NCCM is adverse, while the prognosis of patients with asymptomatic disease is favorable (e43).
Patients with the following risk factors have been found to have an especially poor prognosis: enlarged end-diastolic left ventricular diameter when first measured, NYHA class III or IV heart failure, permanent atrial fibrillation, and a bundle branch block on ECG (9, e44).
The findings published to date with regard to arrhythmogenic complications do not yield a uniform picture (15, 17, e45). It therefore seems inappropriate at present to issue a general recommendation for the primary prophylactic implantation of an ICD in patients with NCCM.
It remains to be seen whether, in the future, genetic analysis will play a role in prognostication for individual patients.
Murphy et al. found that cardiological follow-up examinations conducted at least once every six months led to a markedly better prognosis among their patients than in other studies (table 1) (17).
An aggressive treatment strategy is recommended for high-risk patients, including ICD implantation and early listing for heart transplantation where appropriate (15, e46), but asymptomatic patients with NCCM and patients who have neither a cardiac arrhythmia nor any left ventricular dysfunction do not require any treatment at all. In such cases, the best management strategy according to the current state of medical knowledge is that patients should be informed about the presence of the disease and about the symptoms that might arise in future, reassured about the generally favorable prognosis, and told of the importance of annual cardiological follow-up. Symptomatic and high-risk patients should have cardiological follow-up examinations at least twice per year.
Prospects
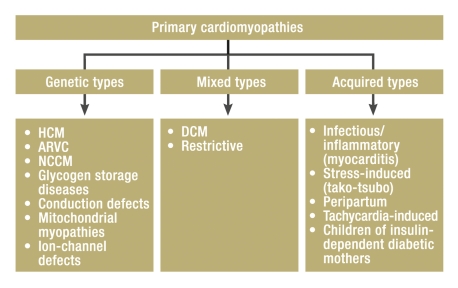
The WHO, in 1995, designated NCCM as an unclassified cardiomyopathy (e47), but the American Heart Association recently recommended classifying it as a primary genetic cardiomyopathy (figure 6) (e48). Most of the cases of NCCM that have been published to date were in single case reports or in case series involving relatively few patients, so that no generally applicable recommendations on the management of this disease can yet be issued. In order to acquire data from a larger patient collective, the German Arbeitsgemeinschaft Leitende Kardiologische Krankenhausärzte (ALKK) has initiated an NCCM registry. The registry accepts patients both retro- and prospectively; a number of centers are already participating (Appendix, and further centers are welcome to join. Case report forms can be downloaded from www.alkk.org or obtained directly from the first author at med1@klinikum.wolfs burg.de. Unclear case histories and discussion cases can also be sent in for consultative assessment.
Figure 6.
Classification of primary cardiomyopathies by the American Heart Association (e48); HCM, hypertrophic cardiomyopathy; DCM, dilated cardiomyopathy; ARVC, arrhythmogenic right-ventricular cardiomyopathy; NCCM, isolated non-compaction cardiomyopathy
Appendix.
The following centers, listed in alphabetical order, currently participate actively in the NCCM registry of the ALKK: Charité Berlin Mitte (P.D. Dr. Borges, Dr. Gromann), Deutsches Herzzentrum Berlin (Prof. Dr. Fleck, Dr. Li), Klinikum Braunschweig (Prof. Dr. Heintzen, Dr. Gradaus), Kardiologisch-Angiologisches Zentrum Frankfurt (PD. Dr. Schmermund), Praxis Dr. Krug, Frankfurt, Universitätsklinik Greifswald (Prof. Dr. Felix, Dr. Hummel), Kreiskrankenhaus Helmstedt (Dr. Nguyen), Klinikum Idar-Oberstein (Prof. Dr. Klein, Dr. Toepel), Klinikum Ingolstadt (Prof. Dr. Pfafferott, Dr. Schöpflin), St. Vinzenz Hospital Cologne (PD. Dr. Fehske), Praxis Dr. Fritsch, Cologne, Klinikum Ludwigsburg (Prof. Dr. Liebau, Dr. Nothnagel), Sana Kliniken Lübeck (Dr. Schneider), Universitätsklinik Münster (Prof. Dr. Breithardt, Dr. Zellerhoff), Klinikum Rotenburg/Wümme (Dr. Andresen), Robert-Bosch-Krankenhaus Stuttgart (Prof. Dr. Sechtem, Dr. Ong), Kranken anstalt Rudolfstiftung Vienna (Prof. Dr. Stöllberger), Klinikum Wolfsburg (Prof. Dr. Engberding, Dr. Gerecke).
Overview
NCCM is a type of heart muscle disease that is still not widely known. The current state of scientific knowledge suggests that it is due to a disturbance of embryonal endomyocardial morphogenesis. It is a genetically heterogeneous disease that can run in families. The American Heart Association classifies it as a primary genetic cardiomyopathy.
NCCM is preferably diagnosed by echocardiography or by cardiac MRI. Its main clinical manifestations are heart failure, thromboembolic events, and arrhythmias.
The prognosis of advanced NCCM is adverse but might possibly be improved by early diagnosis, screening tests in affected families, and better management, including closely spaced follow-up examinations and an aggressive treatment strategy for patients at risk.
It is hoped that the data gathered by the NCCM registry of the ALKK will play an important role in improving the diagnostic evaluation, risk stratification, and management of this disease.
Table 3. Comparison of echocardiographic findings in patients with isolated non-compaction cardiomyopathy (NCCM) and dilated cardiomyopathy (DCM) (from 16).
| NCCM | DCM | |
| Left atrium (diameter) | normal | ↑ |
| End-diastolic LV volume | (↑) | ↑↑↑ |
| End-systolic LV volume | ↑↑ | ↑↑↑ |
| Ejection fraction (%) | ↓ ↓ (↓ ) | ↓ ↓ ↓ |
| Impaired relaxation in diastole | in 50% of cases | in 30% of cases |
Key Messages.
Non-compaction cardiomyopathy is classified as primary genetic cardiomyopathy and is still rarely considered in the differential diagnosis of chronic heart failure.
Its clinical manifestations vary in severity and can include heart failure, thromboembolic events, and arrhythmias.
The diagnosis is usually made by echocardiography or cardiac MRI.
Depending on the severity of the disease, it is treated with the usual treatments of heart failure, anticoagulation, and anti-arrhythmic treatment, including the implantation of an ICD in high-risk patients.
Symptomatic patients have an adverse prognosis and should undergo cardiological follow-up at least once every six months.
eTable. Genetic mutations in patients with isolated non-compaction cardiomyopathy.
| Up to 44% familial clustering, with genetically heterogeneous findings | |
| Affected chromosomes: autosomal dominant and X-linked inheritance | Reference no. |
| Various mutations in the G 4.5 gene for tafazzin (on Xq28) | (8) |
| alpha-dystrobrevin (DTNA) LIM-domain binding proteins: LDB3, cypher/ZASP, lamin A/C | (e8, e9) |
| Sarcomere proteins | |
| – beta-myosin (MYH7) | (e7, e49, e50) |
| – alpha-cardiac actin (ACTC) | (e7, e51) |
| – Cardiac troponin T (TNNT2) | (e7) |
eBox.
The affected region in the G4.5 gene on chromosome Xq 28 is also affected in a number of muscle diseases, including Barth syndrome, Emery-Dreifuss muscular dystrophy, and myotubular myopathy. Mutations of other genes, particularly the sarcomere genes, e.g., DTNA (for alpha-dystrobrevin), FKBP-12, LDB3/Cypher/ZASP, CSX, MLP/SOX6, and the E101K-mutation in the alpha-cardiac actin (ACTC) gene have also been described in NCCM (e8, e9) (etable). In a study of 48 patients with NCCM, only one patient was found to have a G4.5 mutation, and none of the other genetic mutations mentioned above could be demonstrated (e10).
Adults with autosomal dominant NCCM have been found to harbor a mutation at a new genetic locus on chromosome 11p15 (e11). A study of six sarcomere protein genes in 63 adults with NCCM revealed 11 heterogeneous mutations of the beta-myosin heavy chain gene (MYH7), the alpha-cardiac actin (ACTC) gene, and the gene for cardiac troponin T (TNNT2). Of the 9 different mutations that were found, seven were in MYH7, and one each was in ACTC and TNNT2. Six of the 11 patients with mutated sarcomere genes were found to have other family members with the disease, and, in some cases, an autosomal dominant inheritance pattern was found (e7). Other studies have revealed marked variability within affected families (e12).
Thus, the currently available data show a heterogeneous molecular genetic basis for NCCM (e13). The molecular genetic basis of cardiomyopathies in general will have to be understood in more detail before NCCM can be genetically classified and distinguished from other types of cardiomyopathy (10, e4, e9, e14, e15). Animal models will play an important role in this process. Recently, for example, cardiospecific ablation of the cytoskeletal protein cypher was found to be related to the development of a severe dilated cardiomyopathy in a murine model (e16).
Acknowledgments
We thank Dr. med. A. Reinecke-Lüthge, Head of the Pathology Department at the Klinikum Wolfsburg, for the dissected pathological specimen.
Translated from the original German by Ethan Taub, M.D.
Footnotes
Conflict of interest statement
The authors declare that they have no conflict of interest as defined by the guidelines of the International Committee of Medical Journal Editors.
References
- 1.Engberding R, Bender F. Identification of a rare congenital anomaly of the myocardium by two-dimensional echocardiography: persistence of isolated myocardial sinusoids. Am J Cardiol. 1984;53:1733–1734. doi: 10.1016/0002-9149(84)90618-0. [DOI] [PubMed] [Google Scholar]
- 2.Bellet S, Gouley BA. Congenital heart disease with multiple cardiac anomalies. Report of a case showing aortic atresia, fibrous scar in myocardium and embryonal sinusoidal remains. Am J med Sci. 1932;183:458–465. [Google Scholar]
- 3.Dusek J, Ostádal B, Duskova M. Postnatal persistence of spongy myocardium with embryonic blood supply. Arch Pathol. 1975;99:312–317. [PubMed] [Google Scholar]
- 4.Lauer RM, Fink HP, Petry EL, Dunn MI, Diehl AM. Angiographic demonstration of intramyocardial sinusoids in pulmonary-valve-atresia with intact ventricular septum and hypoplastic right ventricle. N Engl J Med. 1964;271:68–72. doi: 10.1056/NEJM196407092710203. [DOI] [PubMed] [Google Scholar]
- 5.Chin TK, Perloff JK, Williams RG, Jue K, Mohrmann R. Isolated noncompaction of left ventricular myocardium. A study of eight cases. Circulation. 1990;82:507–513. doi: 10.1161/01.cir.82.2.507. [DOI] [PubMed] [Google Scholar]
- 6.Sato Y, Matsumoto N, Matsuo S, et al. Isolated noncompaction of the ventricular myocardium in a 94-year-old patient: Depiction at echocardiography and magnetic resonance imaging. Intern J Cardiol. 2007;119:e32–e34. doi: 10.1016/j.ijcard.2007.01.101. [DOI] [PubMed] [Google Scholar]
- 7.Engberding R, Yelbuz MT, Breithardt G. Isolated noncompaction of the left ventricular myocardium. A review of the literature two de-cades after the initial case description. Clin Res Cardiol. 2007;96:481–488. doi: 10.1007/s00392-007-0528-6. [DOI] [PubMed] [Google Scholar]
- 8.Bleyl SB, Mumford BR, Brown-Harrison M-C, et al. Xq28-linked noncompaction of the left ventricular myocardium: prenatal diagnosis and pathologic analysis of affected individuals. Am J Med Genet. 1997;72:257–265. [PubMed] [Google Scholar]
- 9.Jenni R, Oechslin EN, van de Loo B. Isolated ventricular non-compaction of the myocardium in adults. Heart. 2006;93:11–15. doi: 10.1136/hrt.2005.082271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ichida F, Tsubata S, Bowles KR, et al. Novel gene mutations in patients with left ventricular noncompaction or Barth syndrome. Circulation. 2001;103:1256–1263. doi: 10.1161/01.cir.103.9.1256. [DOI] [PubMed] [Google Scholar]
- 11.Jenni R, Wyss CA, Oechslin EN, Kaufmann PA. Isolated ventricular noncompaction is associated with coronary microcirculatory dysfunction. J Am Coll Cardiol. 2002;39:450–454. doi: 10.1016/s0735-1097(01)01765-x. [DOI] [PubMed] [Google Scholar]
- 12.Enriques SG, Entem FR, Cobo M, Olalla JJ. Uncommon etiology of syncope in a patient with isolated ventricular noncompaction. PACE. 2007;30:577–579. doi: 10.1111/j.1540-8159.2007.00713.x. [DOI] [PubMed] [Google Scholar]
- 13.Finsterer J, Stöllberger C, Krugluger W. Positive troponin-T in noncompation is associated with neuromuscular disorders and poor outcome. Clin Res Cardiol. 2006;95:1–5. doi: 10.1007/s00392-006-0467-7. [DOI] [PubMed] [Google Scholar]
- 14.Stöllberger C, Finsterer J, Blazek G. Left ventricular hypertrabeculation/noncompaction and association with additional cardiac abnormalities and neuromuscular disorders. Am J Cardiol. 2002;90:899–902. doi: 10.1016/s0002-9149(02)02723-6. [DOI] [PubMed] [Google Scholar]
- 15.Oechslin EN, Attenhofer Jost CH, Rojas JR, Kaufmann PA, Jenni R. Long-term follow-up of 34 adults with isolated left ventricular noncompaction: a distinct cardiomyopathy with poor prognosis. J Am Coll Cardiol. 2000;36:493–500. doi: 10.1016/s0735-1097(00)00755-5. [DOI] [PubMed] [Google Scholar]
- 16.Sengupta PP, Mohan JC, Mehta V, et al. Comparison of echocardiographic features of noncompaction of the left ventricle in adults versus idiopathic dilated cardiomyopathy in adults. Am J Cardiol. 2004;94:389–391. doi: 10.1016/j.amjcard.2004.04.046. [DOI] [PubMed] [Google Scholar]
- 17.Murphy RT, Thaman R, Blanes JG, et al. Natural history and familial characteristics of isolated left ventricular non-compaction. Eur Heart J. 2005;26:187–192. doi: 10.1093/eurheartj/ehi025. [DOI] [PubMed] [Google Scholar]
- 18.Finsterer J, Stöllberger C, Gaismayer K, Janssen B. Acquired noncompaction in Duchenne muscular dystrophy. Intern J Cardiol. 2006;106:420–421. doi: 10.1016/j.ijcard.2005.01.018. [DOI] [PubMed] [Google Scholar]
- 19.Andrews RE, Fenton MJ, Ridout DA, Burch M. British Congenital Cardiac Association. New-onset heart failure due to heart muscle disease in childhood: a prospective study in the United Kingdom and Ireland. Circulation. 2008;117:79–84. doi: 10.1161/CIRCULATIONAHA.106.671735. [DOI] [PubMed] [Google Scholar]
- 20.Sandhu R, Finkelhor RS, Gunawardena DR, Bahler RC. Prevalence and characteristics of left ventricular noncompaction in a community hospital cohort of patients with systolic dysfunction. Echocardiography. 2008;25:8–12. doi: 10.1111/j.1540-8175.2007.00560.x. [DOI] [PubMed] [Google Scholar]
- 21.Espinola-Zavaleta N, Soto ME, Castellanos LM, Játiva-Chávez S, Keirns C. Non-compacted cardiomyopathyy: clinical-echocardio-graphic study. Cardiovasc Ultrasound. 2006;4:35–45. doi: 10.1186/1476-7120-4-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stöllberger C, Winkler-Dworak M, Blazek G, Finsterer J. Prognosis of left ventricular hypertrabeculation/concompaction is dependent on cardiac and neuromuscular comorbidity. Int J Cardiol. 2007;121:189–193. doi: 10.1016/j.ijcard.2006.11.007. [DOI] [PubMed] [Google Scholar]
- 23.Harvey RP. Patterning the vertebrate heart. Nat Rev Genet. 2002;7:544–556. doi: 10.1038/nrg843. [DOI] [PubMed] [Google Scholar]
- 24.Srivastava D, Olson EN. A genetic blueprint for cardiac development. Nature. 2000;407:221–226. doi: 10.1038/35025190. [DOI] [PubMed] [Google Scholar]
- 25.Buckingham M, Meilhac S, Zaffran S. Building the mammalian heart from two sources of myocardial cells. Nat Rev Genet. 2005;6:826–835. doi: 10.1038/nrg1710. [DOI] [PubMed] [Google Scholar]
- e1.Eisenberg LM, Markwald RR. Cellular recruitment and the devel-opment of the myocardium. Dev Biol. 2004;274:225–232. doi: 10.1016/j.ydbio.2004.07.023. [DOI] [PubMed] [Google Scholar]
- e2.Bernanke DH, Velkey JM. Development of the coronary blood supply: changing concepts and current ideas. Anat Rec. 2002;269:198–208. doi: 10.1002/ar.10139. [DOI] [PubMed] [Google Scholar]
- e3.Agmon Y, Connolly HM, Olson LJ, Khandheria BK, Seward JB. Noncompaction of the ventricular myocardium. J Am Soc -Echocardiogr. 1999;12:859–863. doi: 10.1016/s0894-7317(99)70192-6. [DOI] [PubMed] [Google Scholar]
- e4.Freedom RM, Yoo SJ, Perrin D, Taylor G, Petersen S, Anderson RH. The morphological spectrum of ventricular noncompaction. Cardiol Young. 2005;15:345–364. doi: 10.1017/S1047951105000752. [DOI] [PubMed] [Google Scholar]
- e5.Weiford BC, Subbarao VD, Mulhern KM. Noncompaction of the ventricular myocardium Circulation. 2004;109:2965–2971 . doi: 10.1161/01.CIR.0000132478.60674.D0. [DOI] [PubMed] [Google Scholar]
- e6.Oechslin EN, Attenhofer Jost CH, Rojas JR, Kaufmann PA, Jenni R. Long-term follow-up of 34 adults with isolated left ventricular noncompaction: a distinct cardiomyopathy with poor prognosis. J Am Coll Cardiol. 2000;36:493–500. doi: 10.1016/s0735-1097(00)00755-5. [DOI] [PubMed] [Google Scholar]
- e7.Klaassen S, Probst S, Oechslin E, et al. Mutations in sarcomere protein genes in left ventricular noncompaction. Circulation. 2008;117:2893–2901. doi: 10.1161/CIRCULATIONAHA.107.746164. [DOI] [PubMed] [Google Scholar]
- e8.Chrissoheris MP, Ronan A, Vivas Y, Marieb M, Protopapas Z. Isolat-ed action of the ventricular myocardium: contemporary diagnosis and management. Clin Cardiol. 2007;30:156–160. doi: 10.1002/clc.20004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e9.Xing Y, Ichida F, Matsuoka T, et al. Genetic analysis in patients with left ventricular noncompaction and evidence for genetic heterogeneity. Mol Genet Metab. 2006;88:71–77. doi: 10.1016/j.ymgme.2005.11.009. [DOI] [PubMed] [Google Scholar]
- e10.Kenton AB, Sanchez X, Coveler KJ, et al. Isolated left ventricular noncompaction is rarely caused by mutations in G4. 5, alpha–dystrobrevin and FK Binding Protein-12. Mol Genet Metab. 2004;82:162–166. doi: 10.1016/j.ymgme.2004.02.009. [DOI] [PubMed] [Google Scholar]
- e11.Sasse-Klaassen S, Probst S, Gerull B, et al. Novel gene locus for autosomal dominant left ventricular noncompaction maps to chromosome 11p15. Circulation. 2004;109:2720–2723. doi: 10.1161/01.CIR.0000131865.21260.56. [DOI] [PubMed] [Google Scholar]
- e12.Johnson MT, Zhang S, Gilkeson R, et al. Intrafamilial variability of noncompaction of the ventricular myocardium. Am Heart J. 2006;151:e7–e14. doi: 10.1016/j.ahj.2006.01.021. [DOI] [PubMed] [Google Scholar]
- e13.Digilio MC, Marino B, Bevilacqua M, Musolino AM, Giannotti A, Dallapiccola B. Genetic heterogeneity of isolated noncompaction of the left ventricular myocardium. Am J Med Genet. 1999;85:90–91. doi: 10.1002/(sici)1096-8628(19990702)85:1<90::aid-ajmg19>3.0.co;2-u. [DOI] [PubMed] [Google Scholar]
- e14.Dellafave L, McNally EM. Sarcomere mutations in cardiomyopathy, noncompaction and the developing heart. Circulation. 2008;117:2847–2849. doi: 10.1161/CIRCULATIONAHA.108.781518. [DOI] [PubMed] [Google Scholar]
- e15.Brandner K, Mick DU, Frazier AE, Taylor RD, Meisinger C, Rehling P. Taz1, an outer mitochondrial membrane protein, affects stability and assembly of inner membrane protein complexes: implications for Barth Syndrome. Mol Biol Cell. 2005;16:5202–5214. doi: 10.1091/mbc.E05-03-0256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e16.Zheng M, Cheng H, Li X, Zhang J, Cui L, Ouyang K, Han L, Zhao T, Gu Y, Dalton ND, Bang ML, Peterson KL, Chen J. Cardiac-specific ablation of Cypher leads to a severe form of dilated cardiomyopathy with premature death. Human Molecular Genet. 2009;18:701–713. doi: 10.1093/hmg/ddn400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e17.Allenby PA, Gould NS, Schwartz MF, Chiemmongkoltip P. Dysplastic cardiac development presenting as cardiomyopathy. Arch Pathol Lab Med. 1988;112:1255–1258. [PubMed] [Google Scholar]
- e18.Jenni R, Goebel N, Tartini R, Schneider J, Arbenz U, Oelz O. Persisting myocardial sinusoids of both ventricles as an isolated anom-aly: echocardiographic, angiographic, and pathologic anatomical findings. Cardiovasc Intervent Radiol. 1986;9:127–131. doi: 10.1007/BF02577920. [DOI] [PubMed] [Google Scholar]
- e19.Ritter M, Oechslin E, Sütsch G, Attenhofer C, Schneider J, Jenni R. Isolated noncompaction of the myocardium in adults. Mayo Clin Proc. 1997;72:26–31. doi: 10.4065/72.1.26. [DOI] [PubMed] [Google Scholar]
- e20.Jenni R, Oechslin E, Schneider J, Attenhofer J, Kaufmann PA. Echocardiographic and pathoanatomical characteristics of isolat-ed left ventricular non-compaction: a step towards classification as a distinct cardiomyopathy. Heart. 2001;86:666–671. doi: 10.1136/heart.86.6.666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e21.Boyd MT, Seward JB, Tajik AJ, Edwards WD. Frequency and location of prominent left ventricular trabeculations at autopsy in 474 normal human hearts: implications for evaluation of mural thrombi by two-dimensional echocardiography. J Am Coll Cardiol. 1987;9:323–326. doi: 10.1016/s0735-1097(87)80383-2. [DOI] [PubMed] [Google Scholar]
- e22.Stöllberger C, Finsterer J. Trabeculation and left ventricular hypertrabeculation/noncompaction. J Am Soc Echocardiogr. 2004;17:1120–1121. doi: 10.1016/j.echo.2004.06.009. [DOI] [PubMed] [Google Scholar]
- e23.Junga G, Kneifel S, Von Smekal A, Steinert H, Bauersfeld U. Myocardial ischaemia in children with isolated ventricular non-compaction. Eur Heart J. 1999;20:910–916. doi: 10.1053/euhj.1998.1398. [DOI] [PubMed] [Google Scholar]
- e24.Sato Y, Matsumoto N, Matsuo S, et al. Myocardial perfusion abnormality and necrosis in a patient with isolated noncompaction of the ventricular myocardium: evaluation by myocardial perfusion SPECT and magnetic resonance imaging. Intern J Cardiol. 2007;120:e24–e26. doi: 10.1016/j.ijcard.2007.04.049. [DOI] [PubMed] [Google Scholar]
- e25.Soler R, Rodriguez E, Monserrat L, Alvarez N. MRI of subendocardial perfusion deficits in isolated left ventricular noncompaction. J Comput Assist Tomogr. 2002;26:373–375. doi: 10.1097/00004728-200205000-00011. [DOI] [PubMed] [Google Scholar]
- e26.Chow CM, Lim KD, Wu L, Leong-Poi H. Isolated left ventricular noncompaction enhanced by echocontrast agent. Circulation. 2007;116:e90–e91. doi: 10.1161/CIRCULATIONAHA.107.699314. [DOI] [PubMed] [Google Scholar]
- e27.Baker GH, Pereira NL, Hlavacek AM, Chessa K, Shirali G. Transthoracic real-time three-dimensional echocardiography in the -diagnosis and description of noncompaction of ventricular myocardium. Echocardiography. 2006;23:490–494. doi: 10.1111/j.1540-8175.2006.00246.x. [DOI] [PubMed] [Google Scholar]
- e28.Rajdev S, Singh A, Nanda NC, Baysan O, Hsiung MC. Comparison of two- and three-dimensional transthoracic echocardiography in the assessment of trabeculations and trabecular mass in left ventricular noncompaction. Echocardiography. 2007;24:760–767. doi: 10.1111/j.1540-8175.2007.00504.x. [DOI] [PubMed] [Google Scholar]
- e29.Williams RI, Masani ND, Buchalter MB, Fraser AG. Abnormal myocardial strain rate in noncompaction of the left ventricle. J Am Soc Echocardiogr. 2003;16:293–296. doi: 10.1067/mje.2003.47. [DOI] [PubMed] [Google Scholar]
- e30.Van Dalen BM, Caliskan K, Soliman OII, Nemes A, Vletter WB, ten Cate FJ, Geleijnse ML. Left ventricular solid body rotation in non-compaction cardiomyopathy: A potential new objective and quantitative functional diagnostic criterion? Eur J Heart Failure. 2008;10:1088–1893. doi: 10.1016/j.ejheart.2008.08.006. [DOI] [PubMed] [Google Scholar]
- e31.Petersen SE, Selvanayagam JB, Wiesmann F, Robson MD, Francis JM, Anderson RH, Watkins H, Neubauer S. Left ventricular non-compaction. Insights from cardiovascular magnetic resonance imaging. J Am Coll Cardiol. 2005;46:101–105. doi: 10.1016/j.jacc.2005.03.045. [DOI] [PubMed] [Google Scholar]
- e32.Daimon Y, Watanabe S, Takeda S, Hijikata Y, Komuro I. Two–layered appearance of noncompaction of the ventricular myocardium on magnetic resonance imaging. Circ J. 2002;66:619–621. doi: 10.1253/circj.66.619. [DOI] [PubMed] [Google Scholar]
- e33.Hamamichi Y, Ichida F, Hashimoto I, et al. Isolated noncompaction of the ventricular myocardium: ultrafast computed tomography and magnetic resonance imaging. Int J Cardiovasc Imaging. 2001;17:305–314. doi: 10.1023/a:1011658926555. [DOI] [PubMed] [Google Scholar]
- e34.Alsaileek AA, Syed I, Seward JB. Myocardial fibrosis of left ventricle: magnetic resonance imaging in noncompaction. J Magn Reson Imaging. 2008;2:621–624. doi: 10.1002/jmri.21068. [DOI] [PubMed] [Google Scholar]
- e35.Dodd JD, Holmvang G, Hoffmann U, et al. Quantification of left ventricular noncompaction and trabecular delayed hyperenhancement with cardiac MRI: Correlation with clinical severity. AJR. 2007;189:974–980. doi: 10.2214/AJR.07.2364. [DOI] [PubMed] [Google Scholar]
- e36.Mohrs OK, Magedanz A, Schlosser T. Images in cardiology: noncompaction of the left ventricular myocardium detected by 64-slice multidetector computed tomography. Clin Cardiol. 2007;30 doi: 10.1002/clc.20036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e37.Stöllberger C, Finsterer J. Pitfalls in the diagnosis of left ventricular hypertrabeculation/non-compaction. Postgrad Med J. 2006;82:679–683. doi: 10.1136/pgmj.2006.046169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e38.Toyono M, Kondo C, Nakajima Y, Nakazawa M, Momma K, Kusakabe K. Effects of carvedilol on left ventricular function, mass, and scintigraphic findings in isolated left ventricular non-compaction. Heart. 2001;86 doi: 10.1136/heart.86.1.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e39.Conraads V, Paelinck B, Vorlat A, Goethals M, Jacobs W, Vrints C. Isolated non-compaction of the left ventricle: a rare indication for transplantation. J Heart Lung Transplant. 2001;20:904–907. doi: 10.1016/s1053-2498(01)00264-9. [DOI] [PubMed] [Google Scholar]
- e40.Strüber M, Meyer AL, Malehsa D, Kugler C, Simn AR, Haverich A. The Current Status of Heart Transplantation and the Development of Artificial Heart Systems. [Situation der Herztransplantation und Weiterentwicklung von Kunstherzen] Dtsch Arztebl Int. 2009;106(28-29):471–477. doi: 10.3238/arztebl.2009.0471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e41.Epstein AE, DiMarco JP, Ellenbogen KA, et al. ACC/AHA/HRS 2008 Guidelines for device-based therapy of cardiac rhythm -abnormalities. J Am Coll Cardiol. 2008;51:e1–e62. doi: 10.1016/j.hrthm.2008.04.014. [DOI] [PubMed] [Google Scholar]
- e42.Fichet J, Legras A, Bernard A, Babuty D. Aborted sudden cardiac death revealing isolated noncompaction of the left ventricle in a patient with Wolff-Parkinson-White Syndrome. PACE. 2007;30:444–447. doi: 10.1111/j.1540-8159.2007.00690.x. [DOI] [PubMed] [Google Scholar]
- e43.Corrado G, Fazio G, Zachara E, et al. Natural history of isolated noncompaction of the ventricular myocardium in adults. Data from the Società Italiana di Ecografia Cardiovascolare (SIEC) Registry. Circulation. 2008;118 [Google Scholar]
- e44.Stöllberger C, Blazek G, Winkler-Dworak M, Finsterer J. Atrial fibrillation in left ventricular noncompaction with and without neuromuscular disorders is associated with a poor prognosis. Int J Cardiol. 2008 doi: 10.1016/j.ijcard.2007.11.099. epub 4 Feb. [DOI] [PubMed] [Google Scholar]
- e45.Fazio G, Corrado G, Zachara E, et al. Ventricular tachycardia in non-compaction of left ventricle: Is this a frequent complication? PACE. 2007;30:544–546. doi: 10.1111/j.1540-8159.2007.00706.x. [DOI] [PubMed] [Google Scholar]
- e46.Sato Y, Matsumoto N, Takahashi G, et al. Cardioverter defibrillator implantation in an adult with isolated noncompaction of the ventricular myocardium. Intern J Cardiol. 2006;110:417–419. doi: 10.1016/j.ijcard.2005.08.056. [DOI] [PubMed] [Google Scholar]
- e47.Richardson P, Mc Kenna, Bristow M, et al. Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the Definition and Classification of Cardiomyopathies. Circulation. 1996;93:841–842. doi: 10.1161/01.cir.93.5.841. [DOI] [PubMed] [Google Scholar]
- e48.Maron BJ, Towbin JA, Thiene G, et al. Contemporary definitions and classifications of the cardiomyopathies: an American Heart Association Scientific Statement form the Council on Clinical Cardiology, Heart Failure and transplantation Committee: Quality of Care and Outcomes Research and functional genomics and translational biology interdisciplinary working groups; and the Council on Epidemiology and Prevention. Circulation. 2006;113:1807–1816. doi: 10.1161/CIRCULATIONAHA.106.174287. [DOI] [PubMed] [Google Scholar]
- e49.Budde BS, Binner P, Waldmüller S, et al. Noncompation of the ventricular myocardium is associated with a de novo mutation in the β-myosin heavy chain gene. PLoS ONE. 2007;2 doi: 10.1371/journal.pone.0001362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e50.Hoedemaekers YM, Caliskan K, Majoor-Krakauer D, et al. -Cardiac β-myosin heavy chain defects in two families with non-compaction cardiomyopathy: linking non-compaction to hypertrophic, -restrictive, and dilated cardiomyopathies. Eur Heart J. 2007;28:2732–2737. doi: 10.1093/eurheartj/ehm429. [DOI] [PubMed] [Google Scholar]
- e51.Monserrat L, Hermida-Prieto M, Fernandez X, et al. Mutation in the alpha-cardiac actin gene associated with apical hypertrophic cardiomyopathy, left ventricular non-compaction, and septal defects. Eur Heart J. 2007;28:1953–1961. doi: 10.1093/eurheartj/ehm239. [DOI] [PubMed] [Google Scholar]
- e52.Lofiego C, Biagini E, Ferlito M, et al. Paradoxical contributions of non-compacted and compacted segments to global left ventricular dysfunction in isolated left ventricular noncompaction. Am J Cardiol. 2006;97:738–741. doi: 10.1016/j.amjcard.2005.09.109. [DOI] [PubMed] [Google Scholar]
- e53.Alehan D. Clinical features of isolated left ventricular noncompaction in children. Int J Cardiol. 2004;97:233–237. doi: 10.1016/j.ijcard.2003.09.004. [DOI] [PubMed] [Google Scholar]
- e54.Wald R, Veldtman G, Golding F, Kirsh J, McCrindle B, Benson L. Determinants of outcome in isolated ventricular noncompaction in childhood. Am J Cardiol. 2004;94:1581–1584. doi: 10.1016/j.amjcard.2004.08.047. [DOI] [PubMed] [Google Scholar]