Abstract
Adrenocorticotropic hormone and angiotensin II stimulate cortisol secretion from bovine adrenal zona fasciculata cells by the activation of adenylate cyclase and phospholipase C-coupled receptors. Curcumin (1– 20 µM), a compound found in the spice turmeric, inhibited cortisol secretion stimulated by ACTH, AngII, and 8CPT-cAMP. Curcumin also suppressed ACTH-stimulated increases in mRNAs coding for steroid acute regulatory protein and CYP11a1 steroid hydroxylase. In whole cell patch clamp recordings from AZF cells, curcumin at slightly higher concentrations also inhibited Cav3.2 current. These results identify curcumin as an effective inhibitor of ACTH- and AngII-stimulated cortisol secretion. The inhibition of Cav3.2 current by curcumin may contribute to its suppression of secretion.
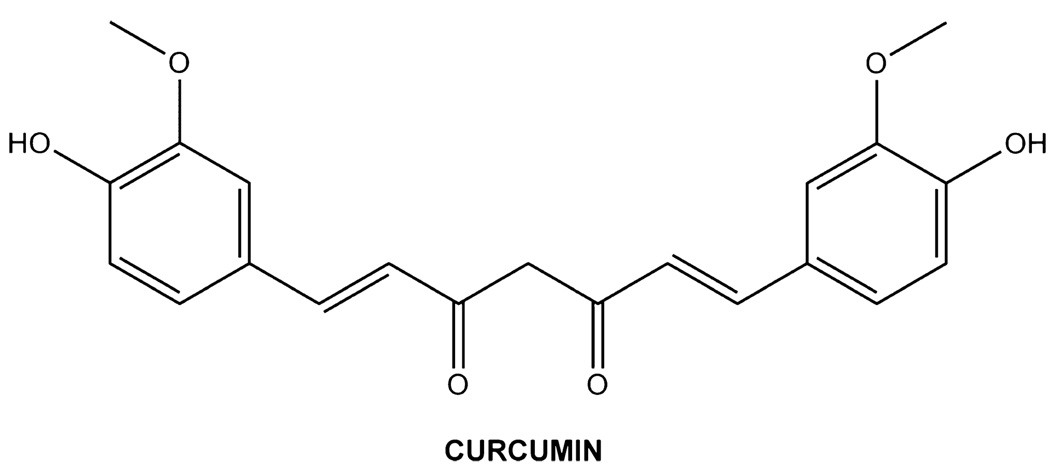
Curcumin (diferulomethane, Figure 1), a polyphenolic compound found in the curry spice turmeric, is obtained from the roots of the Curcuma longa plant.1 In addition to its use as a spice, this phytochemical has been used for centuries in traditional Indian medicine to treat a host of diseases and conditions ranging from allergies and arthritis to Alzheimer’s disease, diabetes, congestive heart failure, and various malignancies.1–4 In this regard, studies have demonstrated that curcumin exhibits antioxidant, antifungal, and antitumor activities.2,5 Currently, curcumin is being tested in ongoing clinical trials to assess its effectiveness in the treatment of cystic fibrosis, psoriasis, Alzheimer’s disease, cancers of the colon and pancreas, and multiple myeloma.5
Figure 1.
Chemical structure of curcumin.
The molecular mechanisms that mediate the therapeutic actions of curcumin are poorly understood. Curcumin regulates a variety of intracellular signaling pathways that ultimately modulate the expression or activity of transcription factors, enzymes, growth factors, and cytokines.1,2,5
Curcumin has also been shown to directly modulate the activity of several types of ion channels. It increases the activity of the cystic fibrosis transmembrane conductance (CFTR) Cl− channel and inhibits the inositol 1,4,5-triphosphate receptor Ca2+ channel and a voltage-gated Kv1.4 K+ channel.6–8
In a recent patch clamp study on bovine adrenal zona fasciculata (AZF) cells, we found that curcumin potently inhibited the activity of bTREK-1 background K+ channels with an IC50 of 0.93 µM.9 bTREK-1 channels set the resting potential of bovine adrenal zona fasciculata (AZF) cells.10,11 Inhibition of these leak-type channels by activation of adrenocorticotropic hormone (ACTH) or angiotensin II (AngII) receptors leads to membrane depolarization, activation of Cav3.2 T-type Ca2+ channels, and cortisol secretion.10,12 Accordingly, we discovered that curcumin induces a concentration-dependent increase in cortisol secretion by AZF cells.9
In the present study, we found that in contrast to its direct stimulatory effect on cortisol production, curcumin inhibited the large increases in cortisol production that are stimulated by ACTH and AngII. Curcumin also inhibited Cav3.2 Ca2+ channels in AZF cells with similar potency.
Curcumin Inhibits ACTH-and cAMP-Stimulated Cortisol Secretion
In the absence of stimulation, cultured bovine AZF cells secrete small quantities of cortisol. ACTH stimulates corticosteroid synthesis through two temporally distinct cAMP-dependent processes. The rapid phase occurs within minutes and is independent of gene transcription. At times up to 1 h, ACTH (2nM) induces no measurable increase in the steroid hydroxylase CYP11a1. ACTH produces no measurable increase in StAR mRNA for at least 0.5 h (data not shown). However, the delayed increase in cortisol synthesis is mediated through the enhanced transcription of genes coding for StAR and the steroidogenic enzymes that mediate the stepwise conversion of cholesterol to cortisol.13,14
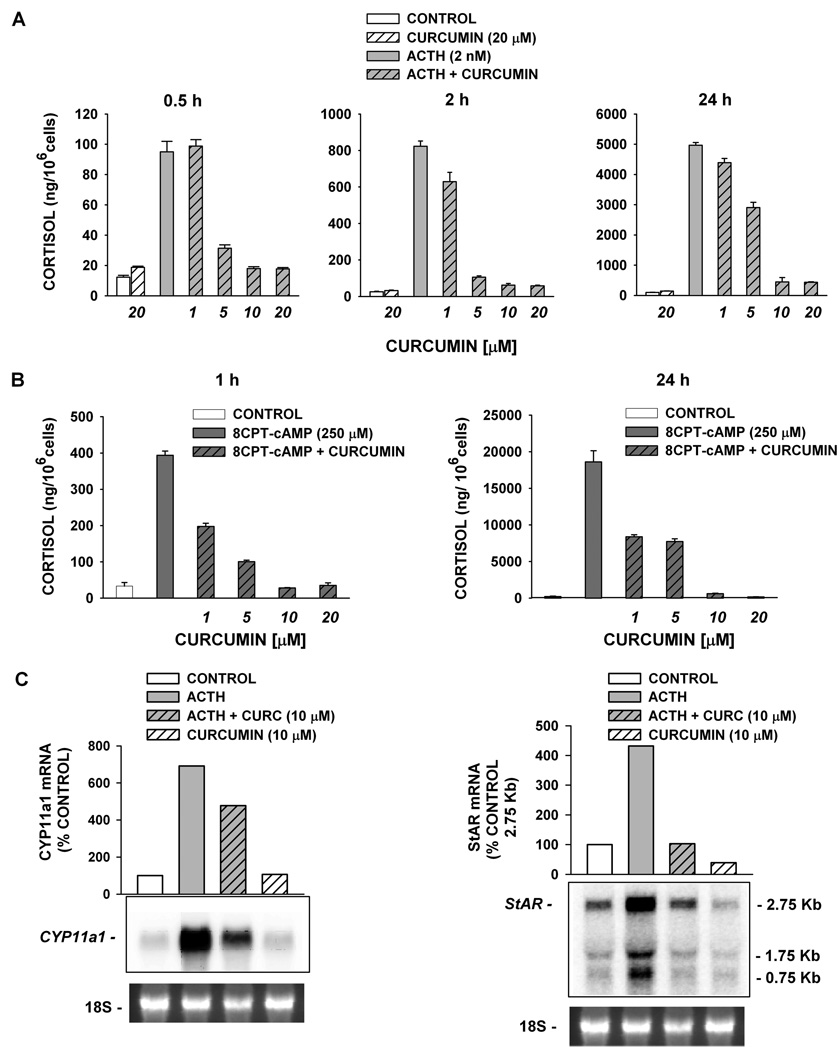
In the experiment illustrated in Figure 2A, ACTH (2 nM) stimulated 8-, 32-, and 50-fold increases in cortisol secretion after 0.5, 2, and 24 h, respectively. By comparison, curcumin (20 µM) alone stimulated only 2–3-fold increases in cortisol production at each time point. In contrast, curcumin inhibited both the rapid and delayed ACTH-stimulated increases in cortisol synthesis. Inhibition by curcumin was measured at concentrations ranging from 1 to 20 µM; 10 µM curcumin inhibited ACTH-stimulated cortisol production measured at 0.5, 2, and 24 h by 81.1 ± 1.3%, 92.4 ± 1.1%, and 91.0 ± 2.9%, respectively.
Figure 2.
Curcumin inhibits ACTH- and 8CPT-cAMP-mediated effects on AZF cells. (A) Bovine AZF cells were treated either without (control, white bar) or with curcumin (20 µM, striped bar), ACTH (gray bar), or ACTH plus curcumin (1–20 µM, gray striped bars) for 0.5, 2, and 24 h, as indicated. (B) Cells were treated either without (control, white bar) or with 8CPT-cAMP alone (250 µM, dark gray bar) or 8CPT-cAMP plus curcumin (1–20 µM, dark gray striped bars). Media was collected after 2 h (left panel) or 24 h (right panel) and cortisol concentration determined by EIA as described in the Experimental Section. Cortisol values expressed are the mean ± SEM of duplicates from duplicate samples. (C) Total AZF cell RNA was isolated after 24 h, electrophoresed, blotted, and probed as described in the Experimental Section. Each lane contained 10 µg of total RNA. Membranes were probed with specific probe for bovine CYP11a1 (left) or bovine StAR (right). Cells were incubated either without (control, white bar), with ACTH alone (2 nM, gray bar), or ACTH (2 nM) plus curcumin (10 µM, gray striped bar), or curcumin alone (white striped bar), as indicated. mRNA is expressed as % control value determined using Kodak 1D 3.6 Image Analysis software. Representative blots from ≥2 different experiments are shown. 18S rRNA bands from gel is shown as evidence of even loading.
The inhibition of ACTH-stimulated secretion by curcumin could occur at a number of sites in the complex signaling pathway linking the ACTH receptor activation to cortisol synthesis. If curcumin interfered only with ACTH-stimulated cAMP synthesis, then it should not suppress cAMP-stimulated cortisol secretion. However, curcumin also inhibited both the rapid and delayed increases in cortisol synthesis induced by a membrane-permeable cAMP analogue, 8CPT-cAMP (Figure 2B). Again, inhibition was observed at concentrations from 1 to 20 µM. At a concentration of 10 µM, curcumin inhibited cortisol production almost completely (92.9 ± 0.2% and 96.8 ± 0.3% at 1 and 24 h, respectively).
Curcumin Inhibits ACTH-Stimulated Increases in the Expression of CYP11a1 and StAR mRNA
Delayed increases in cortisol synthesis induced by ACTH or cAMP require enhanced expression of genes coding for steroidogenic proteins.13,14 Steroid acute regulatory (StAR) and CYP11a1 proteins catalyze the rate-limiting steps in cortisol synthesis. StAR transfers cholesterol from the outer to the inner mitochondrial membrane.15 Cyp11a1 is a mitochondrial enzyme that catalyzes the synthesis of pregnenolone from cholesterol.16 At concentrations that inhibit cortisol secretion, curcumin inhibited ACTH-induced expression of both StAR and CYP11A1 mRNAs. In the experiment illustrated in Figure 2C, curcumin (10 µM) inhibited ACTH-induced CYP11a1 and StAR mRNA expression by 59% and 99%, respectively.
Curcumin Inhibits AngII-Stimulated Cortisol Secretion
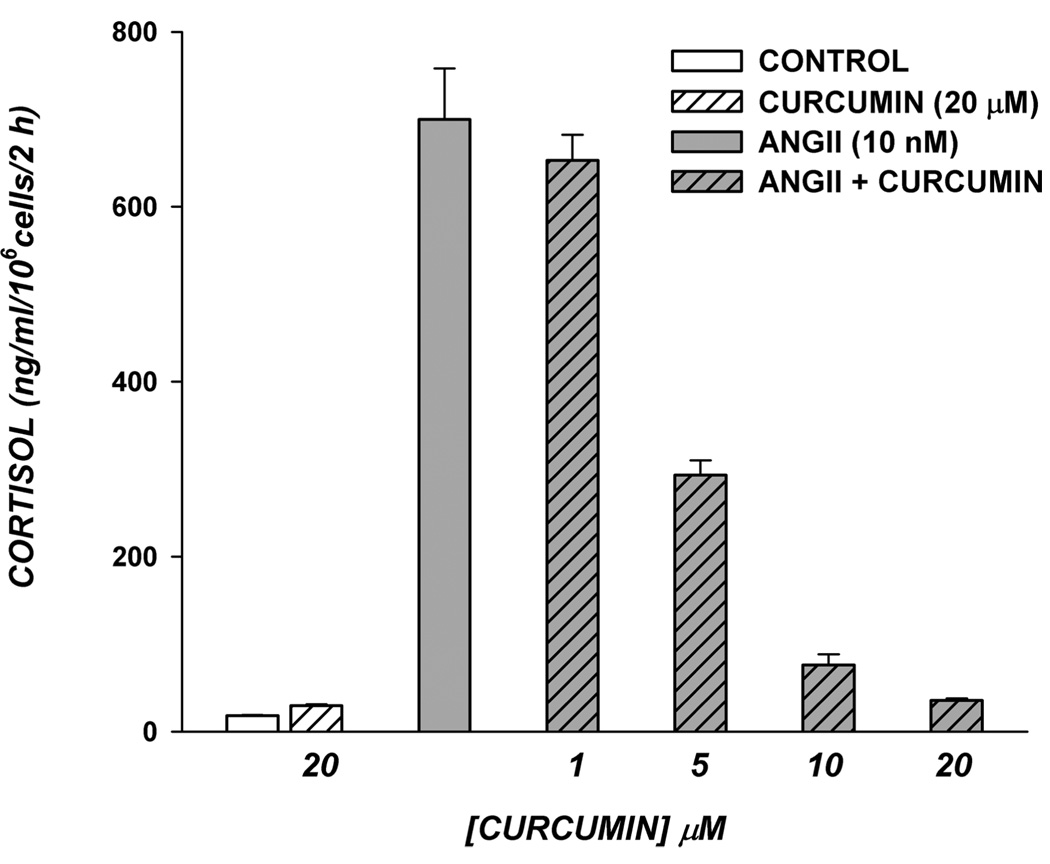
AngII rapidly stimulates cortisol secretion through activation of a PLC-coupled AT1 receptor.17 Curcumin inhibited AngII-stimulated cortisol secretion with a potency nearly identical to that observed for ACTH- and cAMP-stimulated secretion. As illustrated in Figure 3, the large 24-fold increase in cortisol synthesis produced in response to a 2 h exposure to AngII (10 nM) was nearly completely inhibited by 20 µM curcumin (94.5 ± 0.3%).
Figure 3.
Curcumin blocks AngII-mediated increase in cortisol secretion in AZF cells. Bovine AZF cells were plated as described in the Experimental Section. After 24 h, medium was aspirated and replaced with the same medium either without (control) or with curcumin (20 µM), AngII, or AngII plus curcumin (1–20 µM) as indicated. Media was collected after 2 h and cortisol concentration determined by EIA as described in the Experimental Section. Cortisol values expressed are the mean ± SEM of duplicates from duplicate samples.
Curcumin Inhibits Cav3.2 Ca2+ Channels in AZF Cells
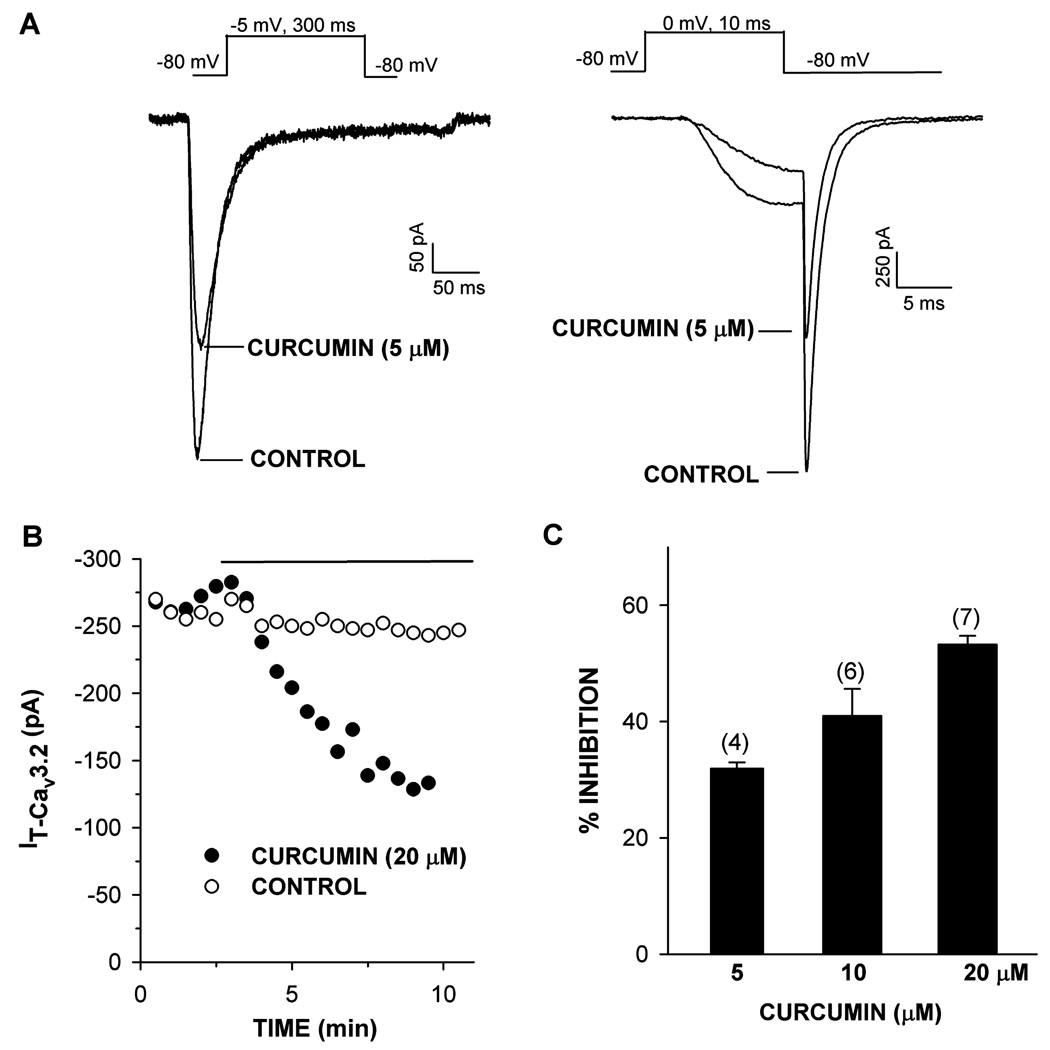
Whole cell patch clamp recording allows single cells to be voltage-clamped and specific ion currents to be measured with a single glass microelectrode. Bovine AZF cells express low voltage-activated Cav3.2 channels.18 ACTH- and AngII-stimulated cortisol secretion are inhibited by drugs that block these T-type Ca2+ channels.12 Experiments were done to determine whether curcumin might inhibit Cav3.2 channels. In whole cell patch clamp recordings, curcumin inhibited Cav3.2 channels at concentrations similar to, although slightly higher than, those that inhibited ACTH- and AngII-stimulated cortisol synthesis (Figure 4). At concentrations of 5, 10, and 20 µM, curcumin inhibited Cav3.2 by 31.9 ± 1.1% (n = 4), 41.0 ± 4.6% (n = 6), and 53.3. ± 1.5% (n = 7), respectively. Inhibition of Cav3.2 current by this lipid-soluble agent was poorly reversible.
Figure 4.
Effect of curcumin on Cav3.2 T-type Ca2+ current in bovine AZF cells. Ca2+ currents were activated by long (300 ms) or short (10 ms) voltage steps applied at 30 s intervals from a holding potential of −80 mV. After recording current in standard saline, cells were superfused with saline containing curcumin (5–20 µM). (A) Ca2+ current traces activated by long (left) and short (right) voltage steps before (control) and after superfusing curcumin (5 µM), as indicated. (B) Plot of T-type Ca2+ current amplitude against time. Cav3.2 current amplitudes are plotted against time for representative control and curcumin (20 µM) superfused cell. Open circles indicate control traces recorded without curcumin. Closed circles indicate traces from curcumin-treated cell where drug was superfused as indicated by solid line. (C) Summary of experiments in which AZF cells were superfused with 5, 10, or 20 µM curcumin (black bars) and inhibition of T-type Ca2+ current was measured. Values are mean ± SEM of indicated number of determinations.
In this study, we discovered that curcumin inhibits both ACTH-and AngII-stimulated cortisol secretion and Cav3.2 current in bovine AZF cells. These findings suggest that curcumin may inhibit secretion stimulated by these two hormones through a common mechanism. This conclusion is consistent with the finding that both ACTH- and AngII-stimulated cortisol secretion depend, at least in part, on Ca2+ entry through Cav3.2 channels as previously reported.12,19 However, since the rapid and delayed effects of these two peptides are not mediated exclusively by Ca2+, it is unlikely that the inhibition of T-type Ca2+ channels is solely responsible for curcumin’s effect on cortisol secretion.20,21 In this regard, cAMP and Ca2+ are dual messengers regulating cortisol synthesis by activating several protein kinases, including PKA and protein kinase C (PKC).21–23 In bovine adrenal cortical cells, the PKC inhibitor calphostin inhibits ACTH-stimulated cortisol secretion.23 Similarly, we found that the PKA inhibitor H-89 inhibited 2 nM ACTH-stimulated cortisol secretion by AZF cells measured at 3 and 24 h by 89% and 92%, respectively (unpublished observations). Curcumin has been reported to inhibit forms of both of these protein kinases.24,25 Thus, it is likely that curcumin inhibits ACTH- and AngII-stimulated cortisol secretion through effects on multiple proteins, including enzymes and ion channels.
With respect to cAMP, the finding that curcumin inhibited both ACTH- and 8CPT-cAMP-stimulated increases in cortisol synthesis with similar potency and effectiveness indicated that this agent did not interfere with ACTH binding to the MC2R receptor or the subsequent activation of adenylate cyclase by Gs. Thus, whatever the mechanism, curcumin inhibition of ACTH-stimulated cortisol secretion occurs at points distal to the synthesis of cAMP. Accordingly, ACTH inhibits bTREK-1 K+ channels and depolarizes bovine AZF cells by multiple cAMP-dependent mechanisms.26,27
The finding that curcumin suppresses both the rapid and delayed effects of ACTH and cAMP on cortisol secretion with similar potency is consistent with the idea that inhibition of a common signaling pathway is involved. Accordingly, both the rapid and delayed actions of ACTH on corticosteroid synthesis are mediated by cAMP through PKA and perhaps other protein kinases.14,28 By inhibiting the function of protein kinases including PKA, curcumin could inhibit either the function or expression of rate-limiting steroidogenic proteins, including StAR and CYP11a1. The marked reduction of both ACTH- and cAMP-stimulated increases in StAR and CYP11a1 mRNAs indicates that curcumin suppresses delayed increases in cortisol synthesis by inhibiting the transcription of these genes or the stability of the corresponding mRNAs.
The actions of curcumin on cortisol secretion are complex. While curcumin alone stimulates a concentration-dependent increase in cortisol synthesis, it inhibits both ACTH- and AngII-stimulated cortisol secretion with similar potency. The increases in cortisol synthesis induced by curcumin are only a few percent of those induced by ACTH or AngII.9 Therefore, it is unlikely that curcumin and ACTH stimulate cortisol secretion by similar mechanisms. The curcumin-mediated inhibition of the large quantity of cortisol produced in response to ACTH or AngII appears to be superimposed on the relatively small, stimulatory effects of curcumin alone on cortisol secretion.
The biochemical pathways that link ACTH and AngII receptors to rapid and delayed increases in cortisol synthesis involve activation, inhibition, and induction of proteins and enzymes located in the cell membrane, microsomes, and mitochondria.13,14,16 Because of complexity of the steroidogenic pathways and the multiplicity of signaling pathways modulated by curcumin, it will be difficult to identify the molecular mechanisms that underlie its complex effects on cortisol synthesis.
In addition to curcumin, a second polyphenol, cinnamyl 1–3,4-dihydroxy-α-cyanocinnamate (CDC), has been shown to produce effects on cortisol secretion similar to curcumin.29 Because the actions of polyphenols are complex, we have no proof that these two agents modulate basal or ACTH-stimulated cortisol secretion by similar mechanisms. In fact, curcumin and CDC produce opposing effects on bTREK-1 K+ channel activity in bovine AZF cells.29 In this regard, curcumin has been shown to modulate the activity of at least five different ion channels, including those permeable to Ca2+, K+, and Cl−.6–9 The potent inhibition of bTREK-1 K+ channels could underlie its potential therapeutic value in the treatment of prostate cancer.30,31 It will be important to identify other ion channels whose activity is regulated by curcumin, either directly or through actions on channel-associated signaling pathways.
The full value of curcumin as a therapeutic agent has not yet been realized. In two remarkable studies, curcumin recently has been shown to protect against, and even reverse, murine cardiac hypertrophy and heart failure.3,4 Perhaps it could also be useful in treating disorders of corticosteroid secretion, including Cushing’s or Addison’s disease. Regardless, in view of curcumin’s increasing value as a pharmaceutical agent, it will be important to fully characterize its action on corticosteroid secretion, including cortisol and aldosterone.
Experimental Section
General Experimental Procedures
DMEM/F12, antibiotics, and fetal bovine sera (FBS) were obtained from Invitrogen (Carlsbad, CA). Ultrahyb was purchased from Ambion (Austin, TX), and Prime-It II kit for random priming was from Stratagene (La Jolla, CA). Curcumin was purchased from Biomol (Plymouth Meeting, PA). Phosphate-buffered saline (PBS), bovine plasma fibronectin, tocopherol, selenite, ascorbic acid, 8CPT-cAMP, ACTH (1–24), and angiotensin II (AngII) were obtained from Sigma (St. Louis. MO). Cortisol EIA kit was from Diagnostic Systems Laboratories (Webster, TX).
Isolation and Culture of AZF Cells
Bovine adrenal glands were obtained from steers (age 2–3 yr) at a local slaughterhouse. Isolated AZF cells were obtained and prepared as previously described.32 After isolation, cells were either resuspended in DMEM/F12 (1:1) with 10% FBS, 100 U/mL penicillin, 0.1 mg/mL streptomycin, and the antioxidants 1 µM tocopherol, 20 nM selenite, and 100 µM ascorbic acid (DMEM/F12+) and plated for immediate use, or resuspended in FBS/5% DMSO, divided into 1 mL aliquots, and stored in liquid nitrogen for future use. To ensure cell attachment, dishes were treated with fibronectin (10 µg/mL) at 37 °C for 30 min, then rinsed with warm, sterile PBS immediately before adding cells. Cells were maintained at 37 °C in a humidified atmosphere of 95% air–5% CO2. For experiments where cortisol and RNA were assayed, cells were plated in DMEM/F12+ for 24 h, after which media was changed to serum-free DMEM/F12 containing 50 µg/mL BSA, 100 µM ascorbic acid, 1 µM tocopherol, 25 nM insulin, and 10 µg/mL transferrin.
Cortisol Assay
Media from experiments was either assayed immediately after collection or frozen (−20 °C) until all samples were available. Cortisol secretion by AZF cells was measured in duplicate from duplicate experimental media samples using a Cortisol EIA (DSL-10–200) from Diagnostic Systems Laboratories (Webster, TX), according to the manufacturer’s directions. If necessary, media samples were diluted using DMEM/F12+.
Northern Blot and Measurement of mRNA
Northern blot analysis involves the transfer of electrophoretically separated RNA molecules from a gel onto a positively charged nylon membrane, which is then blocked to prevent nonspecific binding and immersed in a radiolabeled probe that hybridizes to an RNA of interest to reveal its presence. RNA isolation and northern blot procedures used in this study have been described previously.33 Briefly, (5–8) × 106 AZF cells were plated on 60 mm fibronectin-treated dishes in DMEM/F12+. After 24 h, the serum-supplemented media were removed and replaced with either serum-free DMEM/F12 containing 50 µg/mL BSA, 100 µM ascorbic acid, 1 µM tocopherol, 25 nM insulin, and 10 µg/mL transferrin or the same media containing ACTH (1–24), 8CPT-cAMP (250 µM), or AngII (10 nM) either with or without curcumin (1–20 µM), as required. Samples containing ACTH, 8CPT-cAMP, or AngII plus curcumin were preincubated for 10 min with curcumin only before addition of secretagogue. At the end of the incubation period, total RNA was extracted using RNeasy columns (Qiagen, Valencia, CA), electrophoresed on a denaturing gel, and transferred to nylon transfer membrane (GeneScreen Plus, PerkinElmer Life Sciences, Waltham, MA). Probes were labeled with [α-32P]dCTP by random primer labeling (Prime-It II, Stratagene, La Jolla, CA). Probes were generated by RT-PCR using AMV reverse transcriptase (Promega, Madison WI), specific primers, and total RNA isolated from bovine AZF cells as described above. Specific probes generated were as follows: StAR, bases 104–1047 of NM_174189; CYP11a1, bases 679–1816 of NM_176644. Northern autoradiograms were imaged using a Typhoon 9200 variable-mode imager (GE Healthcare Life Sciences, Piscataway, NJ) and quantitated using Kodak 1D 3.6 Image Analysis software (Eastman Kodak Co., Rochester, NY).
T-Type Ca2+ Channel Recording
Patch clamp recordings of T-type Ca2+ currents from bovine AZF cells were made in the whole-cell configuration as previously described.18 The standard pipet solution was 120 mM CsCl, 1 mM CaCl2, 2 mM MgCl2, 11 mM 1,2-bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid, 10 mM HEPES, and 1 mM MgATP with pH titrated to 7.2 with CsOH. The external solution consisted of 117 mM tetraethylammonium chloride, 5 mM CsCl, 20 mM CaCl2, 2 mM MgCl2, 5 mM HEPES, and 5 mM glucose, with pH adjusted to 7.4 with tetraethylammonium hydroxide. All solutions were filtered through 0.22 µM cellulose acetate filters.
Recording Conditions and Electronics
AZF cells were used for patch clamp experiments 2–12 h after plating. Typically, cells with diameters <15 µm and capacitances of 10–15 pF were selected. Coverslips were transferred from 35 mm culture dishes to the recording chamber (volume: 1.5 mL) that was continuously perfused by gravity at a rate of 3–5 mL/min. For whole cell recordings, patch electrodes with resistances of 1.0–2.0 MΩ were fabricated from Corning 0010 glass (World Precision Instruments, Sarasota, FL). These electrodes routinely yielded access resistances of 1.5–4.0 MΩ and voltage-clamp time constants of <100 µs. Ca2+ currents were recorded at room temperature (22–25 °C) according to the procedure of Hamill et al.34 using a List EPC-7 patch clamp amplifier.
Pulse generation and data acquisition were done using a personal computer and PCLAMP software with Digidata 1200 interface (Axon Instruments, Inc., Burlingame, CA). Currents were digitized at 2–10 kHz after filtering with an 8-pole Bessel filter (Frequency Devices, Haverhill, MA). Linear leak and capacity currents were subtracted from current records using summed scaled hyperpolarizing steps of 1/4 pulse amplitude. Data were analyzed using PCLAMP (CLAMPFIT 9.2) and SigmaPlot (version 10.0) software. Curcumin was applied by bath perfusion, controlled manually by a six-way rotary valve.
Acknowledgment
This work was supported by Award Number R01-DK47875 (to J.J.E.) from the National Institute of Diabetes and Digestive and Kidney Diseases. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Diabetes and Digestive and Kidney Diseases or the National Institutes of Health.
References and Notes
- 1.Singh S. Cell. 2007;130:765–768. doi: 10.1016/j.cell.2007.08.024. [DOI] [PubMed] [Google Scholar]
- 2.Aggarwal BB, Sundaram C, Malani N, Ichikawa H. Adv. Exp. Med. Biol. 2007;595:1–75. doi: 10.1007/978-0-387-46401-5_1. [DOI] [PubMed] [Google Scholar]
- 3.Morimoto T, Sunagawa Y, Kawamura T, Takaya T, Wada H, Nagasawa A, Komeda M, Fujita M, Shimatsu A, Kita T, Hasegawa K. J. Clin. Invest. 2008;118:868–878. doi: 10.1172/JCI33160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li HL, Liu C, de Couto G, Ouzounian M, Sun M, Wang AB, Huang Y, He CW, Shi Y, Chen X, Nghiem MP, Liu Y, Chen M, Dawood F, Fukuoka M, Maekawa Y, Zhang L, Leask A, Ghosh AK, Kirshenbaum LA, Liu PP. J. Clin. Invest. 2008;118:879–893. doi: 10.1172/JCI32865. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 5.Corson TW, Crews CM. Cell. 2007;130:769–774. doi: 10.1016/j.cell.2007.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Berger AL, Randak CO, Ostedgaard LS, Karp PH, Vermeer DW, Welsh MJ. J. Biol. Chem. 2005;280:5221–5226. doi: 10.1074/jbc.M412972200. [DOI] [PubMed] [Google Scholar]
- 7.Dyer JL, Khan SZ, Bilmen JG, Hawtin SR, Wheatley M, Javed MU, Michelangeli F. Cell Calcium. 2002;31:45–52. doi: 10.1054/ceca.2001.0259. [DOI] [PubMed] [Google Scholar]
- 8.Liu H, Danthi SJ, Enyeart JJ. Biochem. Biophys. Res. Commun. 2006;344:1161–1165. doi: 10.1016/j.bbrc.2006.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Enyeart JA, Liu HL, Enyeart JJ. Biochem. Biophys. Res. Commun. 2008 [Google Scholar]
- 10.Mlinar B, Biagi BA, Enyeart JJ. J. Biol. Chem. 1993;268:8640–8644. No.12. [PubMed] [Google Scholar]
- 11.Enyeart JJ, Xu L, Danthi S, Enyeart JA. J. Biol. Chem. 2002;277:49186–49199. doi: 10.1074/jbc.M207233200. [DOI] [PubMed] [Google Scholar]
- 12.Enyeart JJ, Mlinar B, Enyeart JA. Mol. Endocrinol. 1993;7:1031–1040. doi: 10.1210/mend.7.8.8232302. [DOI] [PubMed] [Google Scholar]
- 13.Simpson ER, Waterman MR. Annu. Rev. Physiol. 1988;50:427–440. doi: 10.1146/annurev.ph.50.030188.002235. [DOI] [PubMed] [Google Scholar]
- 14.Parker KL, Schimmer BP. Vit. Horm. 1995;51:339–370. doi: 10.1016/s0083-6729(08)61044-4. [DOI] [PubMed] [Google Scholar]
- 15.Stocco DM. Annu. Rev. Physiol. 2001;63:213. doi: 10.1146/annurev.physiol.63.1.193. [DOI] [PubMed] [Google Scholar]
- 16.Payne AH, Hales DB. Endocr. Rev. 2004;25:947–970. doi: 10.1210/er.2003-0030. [DOI] [PubMed] [Google Scholar]
- 17.Hunyady L, Catt KJ. Mol. Endocrinol. 2005;20:953–970. doi: 10.1210/me.2004-0536. [DOI] [PubMed] [Google Scholar]
- 18.Mlinar B, Biagi BA, Enyeart JJ. J. Gen. Physiol. 1993;102:217–237. doi: 10.1085/jgp.102.2.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yamazaki T, Kimoto T, Higuchi K, Ohta Y, Kawato S, Kominami S. Endocrinology. 1998;139:4765–4771. doi: 10.1210/endo.139.12.6338. [DOI] [PubMed] [Google Scholar]
- 20.Waterman MR, Simpson ER. Rec. Progr. Horm. Res. 1988;45:532–566. doi: 10.1016/b978-0-12-571145-6.50016-9. [DOI] [PubMed] [Google Scholar]
- 21.Waterman MR. J. Biol. Chem. 1994;269:27783–27786. [PubMed] [Google Scholar]
- 22.Enyeart JJ. Vit. Horm. 2005;70:265–279. doi: 10.1016/S0083-6729(05)70008-X. [DOI] [PubMed] [Google Scholar]
- 23.Omura M, Suematsu S, Nishikawa T. Endocr. J. 2007;54:585–592. doi: 10.1507/endocrj.k07-015. [DOI] [PubMed] [Google Scholar]
- 24.Hasmeda M, Polya GM. Phytochemistry. 1996;42:599–605. doi: 10.1016/0031-9422(96)00091-x. [DOI] [PubMed] [Google Scholar]
- 25.Mahmmoud YA. Br. J. Pharmacol. 2007;150:200–208. doi: 10.1038/sj.bjp.0706970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Enyeart JJ, Mlinar B, Enyeart JA. J. Gen. Physiol. 1996;108:251–264. doi: 10.1085/jgp.108.4.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Liu H, Enyeart JA, Enyeart JJ. J. Gen. Physiol. 2008;132:279–294. doi: 10.1085/jgp.200810003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gyles SL, Burns CJ, Whitehouse BJ, Sugden D, Marsh PJ, Persaud SJ, Jones PM. J. Biol. Chem. 2001;276:34888–34895. doi: 10.1074/jbc.M102063200. [DOI] [PubMed] [Google Scholar]
- 29.Danthi S, Enyeart JA, Enyeart JJ. Mol. Pharmacol. 2004;65:1–12. doi: 10.1124/mol.65.3.599. [DOI] [PubMed] [Google Scholar]
- 30.Dorai T, Gehani N, Katz A. Prostate Cancer Prostatic Dis. 2000;3:84–93. doi: 10.1038/sj.pcan.4500399. [DOI] [PubMed] [Google Scholar]
- 31.Voloshyna I, Besana A, Castillo M, Matos T, Weinstein IB, Mansukhani M, Robinson RB, Cordon-Cardo C, Feinmark SJ. Cancer Res. 2008;68:1197–1203. doi: 10.1158/0008-5472.CAN-07-5163. [DOI] [PubMed] [Google Scholar]
- 32.Enyeart JJ, Gomora JC, Xu L, Enyeart JA. J. Gen. Physiol. 1997;110:679–692. doi: 10.1085/jgp.110.6.679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Enyeart JA, Danthi SJ, Enyeart JJ. Mol. Pharmacol. 2003;64:132–142. doi: 10.1124/mol.64.1.132. [DOI] [PubMed] [Google Scholar]
- 34.Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Pflügers Arch. 1981;391:85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]