Abstract
This investigation tested the hypothesis that metabolic syndrome decreases the relative contribution of specific K+ channels to coronary reactive hyperemia. Ca2+-activated (BKCa), voltage-activated (KV), and ATP-dependent (KATP) K+ channels were investigated. Studies were conducted in anesthetized miniature Ossabaw swine fed a normal maintenance diet (11% kcal from fat) or an excess calorie atherogenic diet (43% kcal from fat, 2% cholesterol, 20% kcal from fructose) for 20 wk. The latter diet induces metabolic syndrome, increasing body weight, fasting glucose, total cholesterol, and triglyceride levels. Ischemic vasodilation was determined by the coronary flow response to a 15-s occlusion before and after cumulative administration of antagonists for BKCa (penitrem A; 10 μg/kg iv), KV (4-aminopyridine; 0.3 mg/kg iv) and KATP (glibenclamide; 1 mg/kg iv) channels. Coronary reactive hyperemia was diminished by metabolic syndrome as the repayment of flow debt was reduced ∼30% compared with lean swine. Inhibition of BKCa channels had no effect on reactive hyperemia in either lean or metabolic syndrome swine. Subsequent inhibition of KV channels significantly reduced the repayment of flow debt (∼25%) in both lean and metabolic syndrome swine. Additional blockade of KATP channels further diminished (∼45%) the repayment of flow debt in lean but not metabolic syndrome swine. These data indicate that the metabolic syndrome impairs coronary vasodilation in response to cardiac ischemia via reductions in the contribution of K+ channels to reactive hyperemia.
Keywords: coronary reactive hyperemia, calcium-activated potassium channels, voltage-activated potassium channels, adenosine triphosphate-dependent potassium channels, Ossabaw miniature swine, type 2 diabetes
coronary vasodilation in response to myocardial ischemia is an important mechanism maintaining adequate oxygen delivery to the myocardium to mitigate ischemic injury and infarction (43, 45). Although the exact mechanisms of ischemic dilation are not completely understood, earlier studies support that the putative vasodilator metabolites adenosine and nitric oxide contribute significantly to this response via activation of vascular smooth muscle K+ channels (12, 16). These channels determine vascular smooth muscle membrane potential and thereby regulate electromechanical control of vascular tone (30, 41). Furthermore, K+ channels mediate vasodilation in response to adenosine (3, 12, 16, 28, 34) and nitric oxide (12, 38). Previous investigations document a role for Ca2+-activated K+ (BKCa; see Ref. 42), voltage-activated K+ (KV; see Ref. 12), and ATP-dependent K+ (KATP; see Refs. 2 and 9) channels in coronary vasodilation in response to a brief episode of myocardial ischemia, i.e., reactive hyperemia. However, the relative contribution of these specific K+ channels to ischemic coronary vasodilation has not been delineated.
Earlier studies demonstrate that disease states such as obesity and the metabolic syndrome significantly impair control of coronary blood flow (8, 14, 32, 46, 47, 50). Recent evidence also indicates that peripheral (forearm, calf) vasodilation in response to ischemia is significantly impaired in obese and type 2 diabetic human patients (1, 11, 29). Although coronary flow reserve is reduced by obesity and metabolic syndrome (8, 10, 31, 33, 46), no study has examined the effects of metabolic syndrome on ischemic coronary vasodilation. Based on recent data indicating that metabolic syndrome impairs the function of vascular smooth muscle K+ channels (3, 4, 7, 13, 25, 40), we propose that metabolic syndrome attenuates coronary reactive hyperemia via alterations in the contribution of specific K+ channels to ischemic vasodilation. Examining the mechanisms by which metabolic syndrome affects the coronary response to myocardial ischemia stands to improve our understanding of the increased incidence of cardiac arrhythmias, infarction, and sudden cardiac death observed in obese patients with metabolic syndrome (20, 21, 24, 26, 27, 35, 44).
The goal of this investigation was to determine the relative contribution of K+ channels to coronary reactive hyperemia and the impact of metabolic syndrome. In particular, we tested the hypothesis that metabolic syndrome decreases in the relative contribution of specific K+ channels (BKCa, KV, and KATP) to coronary reactive hyperemia. To examine this hypothesis, experiments were conducted in anesthetized miniature Ossabaw swine fed a normal maintenance diet or an excess calorie atherogenic diet that induces many common features of metabolic syndrome, including obesity, insulin resistance, impaired glucose tolerance, and dyslipidemia (4, 6, 17, 48). The contribution of K+ channels to ischemic vasodilation was determined from the coronary microvascular response to a 15-s occlusion before and after cumulative administration of selective K+ channel antagonists penitrem A (BKCa), 4-aminopyridine (4-AP) (KV), and glibenclamide (KATP).
METHODS
Swine model of metabolic syndrome.
All experimental procedures and protocols used in this investigation were approved by an Institutional Animal Care and Use Committee at Indiana University School of Medicine in accordance with the Guide for the Care and Use of Laboratory Animals. Lean Ossabaw swine were fed ∼2,200 kcal/day of standard chow (5L80, Purina TestDiet) containing (kcal/100 kcal) 18 protein, 71 complex carbohydrates, and 11 fat. To induce metabolic syndrome, Ossabaw swine were fed ∼8,000 kcal/day of a diet containing (kcal/100 kcal) 17 protein, 20 complex carbohydrates, 20 fructose, and 43 fat. The fat was a mixture of lard, hydrogenated soybean oil, and hydrogenated coconut oil. This chow (5B4L, Purina TestDiet) was supplemented with 2.0% cholesterol and 0.7% sodium cholate by weight. Metabolic syndrome swine were fed this diet for 20 wk. Both male and female swine were used for these studies, and no significant effects of gender were noted in lean or metabolic syndrome swine.
Intravascular ultrasound.
Swine were fasted overnight before surgery and sedated with telazol (5 mg/kg sc) and xylazine (2.2 mg/kg sc). After endotracheal intubation, a surgical plane of anesthesia was maintained by mechanical ventilation with 1–3% isoflurane gas and supplemental O2. Utilizing sterile technique, a 7-Fr vascular introducer sheath (Boston Scientific) was inserted in the right femoral artery, and a guiding catheter (Amplatz L, sizes 0.75–2.0; Boston Scientific) was advanced to engage the left main coronary ostium. A 3.2-Fr, 30-MHz intravascular ultrasound (IVUS) catheter (Boston Scientific) was advanced over a guide wire and positioned in the left anterior descending coronary artery (LAD). Automated IVUS pullbacks were performed at 0.5 mm/s to obtain diameters. Video images were analyzed off-line (Sonos Intravascular Imaging System; Hewlett Packard) (18).
In vivo coronary blood flow studies.
Swine were anesthetized with telazol (5 mg/kg sc) and xylazine (2.2 mg/kg sc) and then intubated and ventilated with O2-supplemented air. Anesthesia was maintained with morphine sulfate (3 mg/kg im) and α-chloralose (100 mg/kg iv). Catheters were placed in the right femoral artery and vein as well as the left femoral artery. Blood pressure was measured from the right femoral artery catheter. Furthermore, arterial blood was drawn from this catheter to analyze blood gas parameters every 15–30 min. Ventilatory adjustments and/or sodium bicarbonate administration maintained arterial blood gases within normal physiological limits. A left thoracotomy was performed at the fifth intercostal space, the left lung was restrained in gauze, and the pericardium was opened. A proximal portion of the LAD was isolated, and a perivascular Transonics flow transducer (2.5 mm) was implanted to measure coronary blood flow; a snare was used to occlude the LAD.
Hemodynamic variables were allowed to stabilize 15–30 min before the reactive hyperemia protocol. Following baseline measurements, the LAD was occluded for 15 s and then released to measure reactive hyperemia. This reactive hyperemia protocol was performed in lean (n = 8) and metabolic syndrome (n = 8) swine. Subsequently, the relative contribution of K+ channels to ischemic coronary vasodilation in lean and metabolic syndrome swine was examined by the cumulative intravenous administration of specific K+ channel antagonists penitrem A (BKCa; 10 μg/kg), 4-AP (KV; 0.3 mg/kg), and glibenclamide (KATP; 1 mg/kg). LAD perfusion territory was estimated to be 30% of total heart weight, as previously described by Feigl et al. (19).
Statistical analyses.
Data are presented as means ± SE from n pigs. Hyperemic volume was calculated as area under the curve with Prism software (GraphPad). Statistical comparisons were made with t-tests or two-way repeated-measures ANOVA (factor A = diet; factor B = drug treatment) as appropriate. In all statistical tests, P < 0.05 was considered statistically significant. When significance was found with ANOVA, a Student-Newman-Keuls multiple-comparison test was used to identify differences.
RESULTS
Phenotype of Ossabaw swine.
Phenotypic characteristics of lean and metabolic syndrome swine are given in Table 1. As previously documented by our laboratory (4, 6, 17), we found that the high-calorie atherogenic diet significantly increased body weight, fasting glucose, total cholesterol, and triglyceride levels. These changes represent classic clinical features of the metabolic syndrome. In addition, these swine can be considered “diabetic,” since their plasma glucose concentration exceeded the 126 mg/dl threshold for clinical diagnosis of type 2 diabetes. Under anesthesia, no significant differences in blood pressure were noted between lean and metabolic syndrome swine (Table 2). IVUS revealed that coronary atherosclerosis in the LAD was increased from 7 ± 1% in lean swine to 21 ± 1% in metabolic syndrome swine (Table 1). Importantly, this degree of coronary luminal narrowing in swine with metabolic syndrome did not represent a flow-limiting stenosis, since the peak hyperemic response and volume of repayment were not different between lean and metabolic syndrome pigs (Table 2).
Table 1.
Phenotypic characteristics of lean and MetS Ossabaw swine
| Lean | MetS | |
|---|---|---|
| Body wt, kg | 61 ± 5 | 80 ± 7* |
| Heart wt, g | 176 ± 17 | 193 ± 8 |
| Plasma glucose, mg/dl | 60 ± 8 | 129 ± 7* |
| Total cholesterol, mg/dl | 61 ± 6 | 503 ± 41* |
| Triglycerides, mg/dl | 21 ± 2 | 48 ± 5* |
| Coronary stenosis, % | 7 ± 1 | 21 ± 1* |
Values are means ± SE for n = 7 lean and metabolic syndrome (MetS) swine.
P < 0.05 vs. lean.
Table 2.
Effect of K+ channel blockade on systemic hemodynamics and coronary blood flow
| Lean | MetS | |
|---|---|---|
| Arterial blood pressure, mmHg | ||
| Control | 90 ± 7 | 87 ± 7 |
| Penitrem A | 94 ± 8 | 89 ± 6 |
| Penitrem A +4-AP | 118 ± 8* | 104 ± 4* |
| Penitrem A +4-AP + glibenclamide | 129 ± 8* | 123 ± 4* |
| Heart rate, beats/min | ||
| Control | 71 ± 10 | 70 ± 7 |
| Penitrem A | 76 ± 13 | 70 ± 7 |
| Penitrem A +4-AP | 48 ± 6* | 64 ± 5 |
| Penitrem A +4-AP + glibenclamide | 48 ± 5* | 64 ± 3 |
| Coronary blood flow, ml·min−1·g−1 | ||
| Control | 0.49 ± 0.06 | 0.69 ± 0.07† |
| Penitrem A | 0.54 ± 0.08 | 0.71 ± 0.05 |
| Penitrem A +4-AP | 0.54 ± 0.06 | 0.81 ± 0.08† |
| Penitrem A +4-AP + glibenclamide | 0.49 ± 0.07 | 0.70 ± 0.05† |
| Peak coronary blood flow, ml·min−1·g−1 | ||
| Control | 2.5 ± 0.4 | 2.3 ± 0.3 |
| Penitrem A | 2.7 ± 0.3 | 2.6 ± 0.3 |
| Penitrem A +4-AP | 2.9 ± 0.4 | 3.1 ± 0.2* |
| Penitrem A +4-AP + glibenclamide | 2.6 ± 0.3 | 3.4 ± 0.2* |
| Coronary conductance, μl·min−1·g−1·mmHg−1 | ||
| Control | 6.1 ± 0.9 | 8.2 ± 1.2† |
| Penitrem A | 6.0 ± 1.1 | 9.1 ± 0.9† |
| Penitrem A +4-AP | 4.6 ± 0.7* | 6.9 ± 0.7† |
| Penitrem A +4-AP + glibenclamide | 3.9 ± 0.7*‡ | 5.4 ± 0.4*‡ |
| Peak coronary conductance, μl·min−1·g−1·mmHg−1 | ||
| Control | 28 ± 3 | 27 ± 2 |
| Penitrem A | 30 ± 2 | 31 ± 3 |
| Penitrem A +4-AP | 25 ± 3 | 28 ± 2 |
| Penitrem A +4-AP + glibenclamide | 20 ± 3*‡ | 26 ± 2 |
| Duration of hyperemia, s | ||
| Control | 52 ± 16 | 68 ± 10 |
| Penitrem A | 53 ± 9 | 63 ± 9 |
| Penitrem A +4-AP | 24 ± 9* | 35 ± 9* |
| Penitrem A +4-AP + glibenclamide | 26 ± 9* | 40 ± 9* |
| Flow volume repayment, ml | ||
| Control | 20 ± 4 | 33 ± 7 |
| Penitrem A | 22 ± 4 | 33 ± 6 |
| Penitrem A +4-AP | 18 ± 2 | 28 ± 3 |
| Penitrem A +4-AP + glibenclamide | 16 ± 3 | 31 ± 6 |
| Conductance volume repayment, μl·g−1·mmHg−1 | ||
| Control | 5.1 ± 1.3 | 5.3 ± 0.6 |
| Penitrem A | 5.3 ± 1.4 | 5.7 ± 0.7 |
| Penitrem A +4-AP | 3.3 ± 0.7* | 4.0 ± 0.5* |
| Penitrem A +4-AP + glibenclamide | 2.7 ± 0.6* | 3.6 ± 0.4* |
Values are means ± SE for n = 8 lean and MetS swine. 4-AP, 4-aminopyridine.
P < 0.05 vs. untreated-control same diet group,
lean same drug treatment,
and penitrem A + 4-AP, same diet group.
Effects of metabolic syndrome on coronary reactive hyperemia.
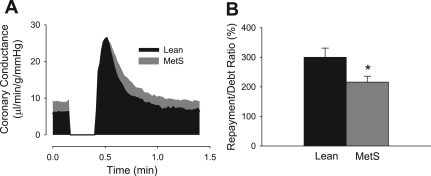
Arterial blood gas parameters were maintained within normal physiological limits throughout the experimental protocol in both lean and metabolic syndrome swine (pH = 7.42 ± 0.02; Pco2 = 42 ± 1 mmHg; Po2 = 151 ± 8 mmHg). Figure 1 demonstrates that metabolic syndrome diminished coronary reactive hyperemia in response to a 15-s total occlusion. This effect is directly evidenced by the significant decrease in the repayment of coronary flow debt (Fig. 1B). The ∼30% reduction in the debt-to-repayment ratio is specifically related to the significant increase in baseline coronary blood flow/conductance and thus greater oxygen “debt” incurred during the occlusion relative to the similar peak hyperemic response and volume repayment observed in metabolic syndrome vs. lean swine (Table 2).
Fig. 1.
Effect of metabolic syndrome (MetS) on coronary reactive hyperemia. A: group average traces of coronary reactive hyperemia in lean (n = 8) and metabolic syndrome (n = 8) swine are shown. Peak hyperemic conductance and the overall volume of repayment were not different between lean and metabolic syndrome swine. However, baseline coronary conductance was significantly elevated by metabolic syndrome; thus, the overall ischemic dilator response in proportion to the incurred oxygen debt (i.e., repayment of flow debt, B) was significantly diminished in metabolic syndrome swine. *P < 0.05 vs. lean.
Effect of K+ channel blockade on baseline hemodynamics and coronary blood flow.
The relative contribution of BKCa, KV, and KATP channels to ischemic coronary vasodilation was examined in lean and metabolic syndrome swine by the cumulative administration of specific K+ channel blockers. Table 2 shows the effect of intravenous infusion of these K+ channel blockers on baseline systemic hemodynamics and coronary blood flow. Inhibition of BKCa channels with penitrem A (10 μg/kg) did not significantly affect mean aortic pressure, heart rate, or coronary blood flow in lean or metabolic syndrome swine. Subsequent administration of the KV channel antagonist 4-AP (0.3 mg/kg) increased blood pressure in both groups of swine, resulting in a reflex-mediated decrease in heart rate. Successive addition of the KATP channel antagonist glibenclamide (1 mg/kg) further elevated blood pressure in both groups. Baseline coronary blood flow was not significantly altered by K+ channel inhibition in either group, since resting coronary blood flow was preserved through an increase in arterial pressure (Table 2). Importantly, when coronary blood flow was normalized to arterial pressure, significant effects of 4-AP (∼25% reduction in lean vs. ∼15% reduction in metabolic syndrome; P = 0.12) and glibenclamide (∼30% decrease in lean and metabolic syndrome; P = 0.39) on coronary vascular conductance were observed relative to untreated baseline conditions (Table 2). Thus, these large increases in pressure (which likely exceeded the autoregulatory range) significantly masked changes in coronary blood flow and the flow-volume of repayment induced by K+ channel inhibition (Table 2). Accordingly, coronary conductance data were utilized to assess the relative contribution of K+ channels to the coronary reactive hyperemic response in lean vs. metabolic syndrome swine.
Effect of metabolic syndrome on the role of K+ channels in coronary reactive hyperemia.
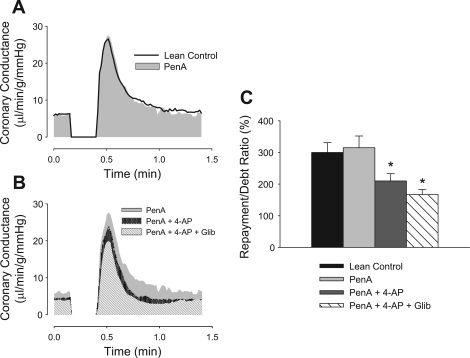
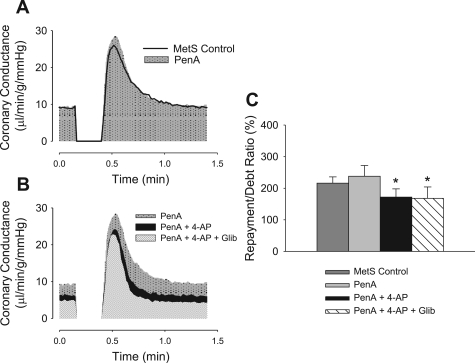
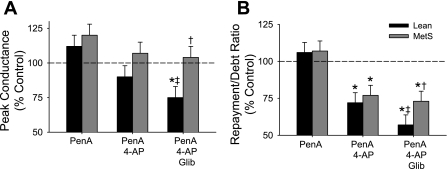
The effect of K+ channel inhibition on the coronary reactive hyperemia is shown in Fig. 2 for lean swine and in Fig. 3 for swine with metabolic syndrome. We found that blockade of BKCa channels with penitrem A had no effect on the reactive hyperemic response in either lean swine (Fig. 2) or swine with metabolic syndrome (Fig. 3). Consistent with our recent findings (12), subsequent inhibition of KV channels with 4-AP significantly decreased the hyperemic response in lean swine, as evidenced by the ∼50% reduction in the duration of hyperemia, the ∼30% reduction in the volume of repayment (Table 2), and the repayment of flow debt (Fig. 2). Addition of 4-AP also significantly reduced the duration of hyperemia, repayment volume (Table 2), and debt-to-repayment ratio by ∼20% in metabolic syndrome swine (Fig. 3). Subsequent inhibition of KATP channels with glibenclamide in lean swine markedly diminished peak hyperemic conductance (Table 2; Fig. 4) and further reduced the repayment of flow debt by an additional 15% (Fig. 2). Thus, combined inhibition of BKCa, KV, and KATP channels reduced the coronary reactive hyperemic response ∼45% in lean swine. In contrast, combined inhibition of K+ channels (addition of glibenclamide to penitrem A and 4-AP) in metabolic syndrome swine did not affect peak hyperemic conductance (Table 2; Fig. 4) or further diminish the debt-to-repayment ratio relative to penitrem A + 4-AP administration (Fig. 3). Figure 4 further illustrates the diminished contribution of K+ channels to coronary reactive hyperemia in metabolic syndrome vs. lean swine on both peak hyperemic conductance (Fig. 4A) and the debt-to-repayment ratio (Fig. 4B).
Fig. 2.
Effect of K+ channel inhibition on coronary reactive hyperemia in lean swine. Group average traces of coronary reactive hyperemia in lean swine (n = 8) are shown before and after cumulative administration of K+ channel blockers. The Ca2+-activated K+ (BKCa) channel antagonist penitrem A (PenA) had no effect on reactive hyperemia (A and C). Subsequent administration of the voltage-dependent-K+ (KV) channel antagonist 4-aminopyridine (4-AP) and the KATP channel antagonist glibenclamide (Glib) significantly reduced the reactive hyperemic response relative to untreated control conditions (B and C). *P < 0.05 vs. lean control.
Fig. 3.
Effect of K+ channel inhibition on coronary reactive hyperemia in metabolic syndrome swine. Group average traces of coronary reactive hyperemia in metabolic syndrome swine (n = 8) are shown before and after cumulative administration of K+ channel blockers. The BKCa channel antagonist penitrem A had no effect on reactive hyperemia (A and C). Subsequent administration of the KV channel antagonist 4-AP significantly reduced the reactive hyperemic response relative to untreated control conditions (B and C). Further inhibition of KATP channels with glibenclamide had no additional effect. *P < 0.05 vs. lean control.
Fig. 4.
Relative contribution of BKCa, KV, and ATP-dependent K+ channels to coronary reactive hyperemia in lean and metabolic syndrome swine. Inhibition of BKCa channels with penitrem A had no effect on peak coronary conductance (A) or the repayment of coronary flow debt (B). Subsequent administration of the KV channel antagonist 4-AP significantly reduced the repayment-to-debt ratio in both lean and metabolic syndrome swine. Further inhibition of KATP channels with glibenclamide reduced peak conductance and the repayment of flow debt in lean but not metabolic syndrome swine. P < 0.05 vs. untreated control same diet group (*), lean same drug treatment (†), and lean penitrem A + 4-AP (‡).
DISCUSSION
The present investigation was designed to elucidate the relative contribution of K+ channels to coronary reactive hyperemia in lean Ossabaw swine and Ossabaw swine with metabolic syndrome. In particular, we tested the hypothesis that metabolic syndrome decreases the relative contribution of specific K+ channels (BKCa, KV, and KATP) to coronary reactive hyperemia. The primary findings of this study are: 1) metabolic syndrome significantly attenuates coronary vasodilation in response cardiac ischemia; 2) BKCa channels do not play a role in coronary reactive hyperemia in lean or metabolic syndrome swine; 3) KV channels contribute to ischemic coronary vasodilation in lean and metabolic syndrome swine; 4) KATP channels contribute to coronary reactive hyperemia in lean but not metabolic syndrome swine; and 5) reduction of coronary reactive hyperemia following combined BKCa, KV, and KATP channel inhibition is markedly attenuated in metabolic syndrome (∼20%) vs. lean (∼45%) swine. Taken together, these data indicate that the metabolic syndrome impairs coronary vasodilation in response to cardiac ischemia via reductions in the contribution of K+ channels to reactive hyperemia.
Effect of metabolic syndrome on ischemic coronary vasodilation.
Our data demonstrate that the ability of the coronary circulation to dilate in response to a brief episode of myocardial ischemia is depressed by the metabolic syndrome (Fig. 1). This effect is directly evidenced by the significant reduction in the repayment-to-debt ratio in metabolic syndrome swine. Because the coronary reactive hyperemic response varies directly with the duration of the occlusion as well as the baseline flow rate, estimation of the repayment of flow debt is critical for the analysis of the hyperemic response (43). Thus, although peak hyperemic conductance and the volume of repayment were not different between lean and metabolic syndrome swine, baseline flow and conductance were significantly elevated by metabolic syndrome (Table 2). Therefore, the overall ischemic dilator response in proportion to the incurred oxygen debt was diminished in metabolic syndrome swine. The increase in baseline flow in this group of metabolic syndrome swine is puzzling but could be related to alterations in substrate utilization, internal work of the myocardium, and/or cardiac efficiency. Although these variables were not assessed in this group of animals, a recent study by our laboratory found that a trend for an increase in baseline flow in conscious, metabolic syndrome swine was associated with a reduction in average lactate uptake and cardiac index with no difference in myocardial oxygen consumption, i.e., altered substrate utilization and cardiac efficiency (5). It is important to recognize that the 21 ± 1% stenosis in metabolic syndrome swine did not limit flow during hyperemia, since the peak hyperemic response (i.e., the minimum vascular resistance) and volume of repayment were not different between lean and metabolic syndrome swine (Table 2). Our finding is consistent with earlier studies demonstrating decreases in coronary flow reserve (8, 10, 31, 33, 46) and impaired ischemic peripheral (forearm, calf) vasodilation (1, 11, 29) in obese human patients. In addition, these data extend our earlier observation that metabolic syndrome significantly impairs metabolic control of coronary blood flow at rest and during exercise-induced increases in MVo2 (47, 50). Taken together, these findings demonstrate that coronary microvascular dysfunction in metabolic syndrome impairs responses to both physiological (exercise) and pathophysiological (ischemia) stimuli.
Contribution of BKCa channels to ischemic coronary vasodilation.
We hypothesized that the metabolic syndrome decreases the relative contribution of specific K+ channels (BKCa, KV, and KATP) to coronary reactive hyperemia. This hypothesis was based on earlier studies documenting a role for BKCa (42), KV (12), and KATP (2, 9, 49) channels in ischemic vasodilation in normal or lean subjects as well as recent data indicating that metabolic syndrome impairs the function of K+ channels in vascular smooth muscle (4, 7, 13, 25, 40). However, to date, no study has examined the relative contribution of K+ channels to coronary reactive hyperemia in the context of metabolic syndrome. We found that BKCa channels do not contribute to the regulation of ischemic vasodilation in either lean or metabolic syndrome swine, since BKCa channel inhibition with penitrem A had no effect on peak hyperemic conductance, volume of repayment, or the repayment of flow debt (Fig. 2; Table 2). These data contrast with an earlier study by Node et al. (42) showing that inhibition of BKCa channels with iberiotoxin reduced both the peak coronary flow and the repayment of flow debt by ∼20% in response to a 20-s coronary artery occlusion in anesthetized dogs. Aside from differences in species, anesthesia, and BKCa channel antagonist used, the reason for these discrepant results is unclear. However, it is important to point out that we recently documented that the dose of penitrem A used in this study is effective, since it essentially abolished coronary vasodilation in response to the BKCa channel agonist NS-1619 in anesthetized, open-chest lean Ossabaw swine (4). Therefore, we are confident that BKCa channels play little, if any, role in coronary vasodilation at rest or in response to cardiac ischemia in lean or metabolic syndrome Ossabaw swine.
Effect of metabolic syndrome on the contribution of KV and KATP channels to coronary reactive hyperemia.
Consistent with a recent study from our laboratory in normal lean dogs (12), we found that KV channels play an important role in the regulation of baseline coronary tone and reactive hyperemia in both lean and metabolic syndrome swine. This was evidenced by a decrease in coronary conductance at rest as well as reductions in peak hyperemic conductance, the volume of repayment, and the repayment-to-debt ratio following administration of the KV channel antagonist 4-AP (Figs. 2 and 3; Table 2). Because inhibition of BKCa channels alone had no effect on key coronary response variables, we attribute these changes to 4-AP-sensitive KV channels. This assumption is supported by the previous findings of Dick et al. (12) that documented a very similar ∼35% reduction in the coronary reactive hyperemic response following inhibition of KV channels with 4-AP alone. Thus, it is unlikely that BKCa channels represent a compensatory mechanism activated secondary to KV channel inhibition. The similar decrease in coronary reactive hyperemia following inhibition of KV channels (Fig. 4B) is consistent with our recent whole cell patch-clamp recordings that found no difference in KV channel current in isolated coronary vascular smooth muscle cells from lean vs. metabolic syndrome Ossabaw swine (4). Our findings contrast with earlier studies which documented that hypercholesterolemia (22, 23) or hyperglycemia (36, 37) alone significantly impairs coronary KV channel current and contribution to arteriolar vasodilation and indicate that the entire metabolic syndrome milieu is critical in determining functional expression of ion channels, i.e., results cannot be overgeneralized from one disease state to another.
Our data also support a role for KATP channels in coronary reactive hyperemia in lean swine, since the addition of glibenclamide to penitrem A and 4-AP further diminished peak coronary conductance and the repayment of flow debt (Fig. 2; Table 2). Although we did not conduct studies with glibenclamide alone, it is critical to recognize that the additional ∼20% reduction in the repayment-to-debt ratio relative to penitrem A + 4-AP administration is consistent with the earlier study of Duncker et al. (15) that documented an ∼25% reduction in the volume of repayment following intracoronary administration of glibenclamide (10 μg·kg−1·min−1) in normal dogs. We fully acknowledge that a more prominent role for KATP channels may exist in the absence of combined BKCa and KV channel inhibition. However, these data importantly document that combined inhibition of BKCa, KV, and KATP channels in lean swine cuts the coronary reactive hyperemic response approximately in half (Fig. 2).
In contrast to our findings in lean swine, we found that subsequent inhibition of KATP channels did not affect the coronary reactive hyperemic response in swine with the metabolic syndrome (Fig. 3). Although baseline conductance was reduced to a similar extent (Table 2), both peak hyperemic conductance and the repayment of flow debt were further reduced following the subsequent administration of glibenclamide in lean but not metabolic syndrome swine (Fig. 4). This finding is supported by a recent study by our group that documented a significant reduction in the contribution of KATP channels to 2-chloroadenosine in isolated coronary arterioles from Ossabaw swine with metabolic syndrome (3). Importantly, reductions in KATP channel function have also been reported in the skeletal muscle microcirculation of obese Zucker rats (25) and in the coronary circulation of diabetic humans (39). Taken together, the present findings extend these earlier observations and indicate that the metabolic syndrome significantly attenuates the contribution of KATP channels to ischemic coronary vasodilation. However, whether this decrement is related to alterations in channel expression, activity, and/or vasoactive mediators that converge on these K+ channels merits future investigation.
Limitations of the study.
We acknowledge that the present experimental design in which K+ channel antagonists were added in a cumulative manner inherently limits the interpretation of the relative contribution of each K+ channel subtype to coronary ischemic vasodilation. However, as discussed above, this limitation is tempered by the similar quantitative effects of combined K+ channel blockade vs. earlier reports examining the effects of 4-AP (12) or glibenclamide (15) alone. Although we cannot rule out a greater role for BKCa channels if penitrem A was administered after 4-AP and/or glibenclamide, we are confident that BKCa channels are not required for coronary reactive hyperemia under “normal” conditions in either lean or metabolic syndrome swine. An additional limitation of this investigation was the significant increase in arterial pressure observed following systemic administration of K+ channel antagonists (Table 2). These increases in arterial pressure masked the effect of K+ channel blockers on baseline coronary blood flow and flow-volume of repayment, since normalization to pressure uncovered significant reductions in baseline conductance following 4-AP and glibenclamide administration. Interestingly, this is also true for the peak hyperemic coronary blood flow in lean swine. In contrast, the changes in pressure resulted in a significant elevation of the peak hyperemic coronary blood flow response in metabolic syndrome swine with successive inhibition of K+ channels. These data suggest that coronary autoregulatory capability may be altered in metabolic syndrome swine, at least to elevations in arterial pressure. The extent to which changes in coronary pressure-flow autoregulation affect ischemic coronary vasodilation in the setting of the metabolic syndrome merits further investigation.
In summary, data from this investigation offer novel insight into the relative contribution of BKCa, KV, and KATP channels to ischemic coronary vasodilation in lean and obese swine with the metabolic syndrome. Our findings confirm the significant contribution of KV and KATP channels to coronary reactive hyperemia in normal lean animals (12, 15) and importantly document that the metabolic syndrome impairs coronary vasodilation in response to cardiac ischemia. We conclude that this decrease in coronary reactive hyperemia is not related to decreased BKCa channel function, as recently documented by our laboratory (4, 40), but more directly to the diminished contribution of KATP channels to the dilator response. We propose that this decrease in the functional contribution of KATP channels to the control of coronary blood flow likely represents a critical mechanism underlying the increased incidence of cardiac arrhythmias, infarction, and sudden cardiac death in obese patients with the metabolic syndrome (20, 21, 24, 26, 27, 35, 44) that should be explored further.
GRANTS
This work was supported by American Heart Association Grant 0810048Z (L. Borbouse), National Institutes of Health Grants HL-092245 (J. D. Tune), RR-13223 (M. Sturek), HL-62552 (M. Sturek), and UL1 RR-025761 (Z. P. Neeb), and the Fortune-Fry Ultrasound Research Fund of the Department of Cellular and Integrative Physiology, Indiana Univeristy School of Medicine.
DISCLOSURES
No conflicts of interest are declared by the authors.
REFERENCES
- 1.Acree LS, Comp PC, Whitsett TL, Montgomery PS, Nickel KJ, Fjeldstad AS, Fjeldstad C, Gardner AW. The influence of obesity on calf blood flow and vascular reactivity in older adults (Abstract). Dyn Med 6: 4, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Banitt PF, Smits P, Williams SB, Ganz P, Creager MA. Activation of ATP-sensitive potassium channels contributes to reactive hyperemia in humans. Am J Physiol Heart Circ Physiol 271: H1594–H1598, 1996 [DOI] [PubMed] [Google Scholar]
- 3.Bender SB, Tune JD, Borbouse L, Long X, Sturek M, Laughlin MH. Altered mechanism of adenosine-induced coronary arteriolar dilation in early-stage metabolic syndrome. Exp Biol Med (Maywood) 234: 683–692, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Borbouse L, Dick GM, Asano S, Bender SB, Dincer UD, Payne GA, Neeb ZP, Bratz IN, Sturek M, Tune JD. Impaired function of coronary BKCa channels in metabolic syndrome. Am J Physiol Heart Circ Physiol 297: H1629–H1637, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Borbouse L, Dick GM, Payne GA, Payne BD, Svendsen MC, Neeb ZP, Alloosh M, Bratz IN, Sturek M, Tune JD. Contribution of BKCa channels to local metabolic coronary vasodilation: effects of metabolic syndrome. Am J Physiol Heart Circ Physiol doi:10.1152/ajpheart.00876.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bratz IN, Dick GM, Tune JD, Edwards JM, Neeb ZP, Dincer UD, Sturek M. Impaired capsaicin-induced relaxation of coronary arteries in a porcine model of the metabolic syndrome. Am J Physiol Heart Circ Physiol 294: H2489–H2496, 2008 [DOI] [PubMed] [Google Scholar]
- 7.Burnham MP, Johnson IT, Weston AH. Reduced Ca2+-dependent activation of large-conductance Ca2+-activated K+ channels from arteries of type 2 diabetic Zucker diabetic fatty rats. Am J Physiol Heart Circ Physiol 290: H1520–H1527, 2006 [DOI] [PubMed] [Google Scholar]
- 8.Camici PG, Marraccini P, Lorenzoni R, Buzzigoli G, Pecori N, Perissinotto A, Ferrannini E, L'Abbate A, Marzilli M. Coronary hemodynamics and myocardial metabolism in patients with syndrome X: response to pacing stress. J Am Coll Cardiol 17: 1461–1470, 1991 [DOI] [PubMed] [Google Scholar]
- 9.Capecchi PL, Guideri F, Colafati M, Acampa M, Cuomo A, Lazzerini PE, Pasini FL. Acute effects of glibenclamide on reactive hyperaemia in the lower limbs in humans. Clin Hemorheol Microcirc 27: 77–82, 2002 [PubMed] [Google Scholar]
- 10.Dagres N, Saller B, Haude M, Husing J, von BC, Schmermund A, Sack S, Baumgart D, Mann K, Erbel R. Insulin sensitivity and coronary vasoreactivity: insulin sensitivity relates to adenosine-stimulated coronary flow response in human subjects. Clin Endocrinol (Oxf) 61: 724–731, 2004 [DOI] [PubMed] [Google Scholar]
- 11.De FE, Cusi K, Ocampo G, Berria R, Buck S, Consoli A, Mandarino LJ. Exercise-induced improvement in vasodilatory function accompanies increased insulin sensitivity in obesity and type 2 diabetes mellitus. J Clin Endocrinol Metab 91: 4903–4910, 2006 [DOI] [PubMed] [Google Scholar]
- 12.Dick GM, Bratz IN, Borbouse L, Payne GA, Dincer UD, Knudson JD, Rogers PA, Tune JD. Voltage-dependent K+ channels regulate the duration of reactive hyperemia in the canine coronary circulation. Am J Physiol Heart Circ Physiol 294: H2371–H2381, 2008 [DOI] [PubMed] [Google Scholar]
- 13.Dimitropoulou C, Han G, Miller AW, Molero M, Fuchs LC, White RE, Carrier GO. Potassium (BKCa) currents are reduced in microvascular smooth muscle cells from insulin-resistant rats. Am J Physiol Heart Circ Physiol 282: H908–H917, 2002 [DOI] [PubMed] [Google Scholar]
- 14.Dincer UD, Araiza AG, Knudson JD, Molina PE, Tune JD. Sensitization of coronary alpha-adrenoceptor vasoconstriction in the prediabetic metabolic syndrome. Microcirculation 13: 587–595, 2006 [DOI] [PubMed] [Google Scholar]
- 15.Duncker DJ, van Zon NS, Altman JD, Pavek TJ, Bache RJ. Role of K+ATP channels in coronary vasodilation during exercise. Circulation 88: 1245–1253, 1993 [DOI] [PubMed] [Google Scholar]
- 16.Duncker DJ, van Zon NS, Ishibashi Y, Bache RJ. Role of K+ ATP channels and adenosine in the regulation of coronary blood flow during exercise with normal and restricted coronary blood flow. J Clin Invest 97: 996–1009, 1996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dyson MC, Alloosh M, Vuchetich JP, Mokelke EA, Sturek M. Components of metabolic syndrome and coronary artery disease in female Ossabaw swine fed excess atherogenic diet. Comp Med 56: 35–45, 2006 [PubMed] [Google Scholar]
- 18.Edwards JM, Alloosh MA, Long XL, Dick GM, Lloyd PG, Mokelke EA, Sturek M. Adenosine A1 receptors in neointimal hyperplasia and in-stent stenosis in Ossabaw miniature swine. Coron Artery Dis 19: 27–31, 2008 [DOI] [PubMed] [Google Scholar]
- 19.Feigl EO, Neat GW, Huang AH. Interrelations between coronary artery pressure, myocardial metabolism and coronary blood flow. J Mol Cell Cardiol 22: 375–390, 1990 [DOI] [PubMed] [Google Scholar]
- 20.Galassi A, Reynolds K, He J. Metabolic syndrome and risk of cardiovascular disease: a meta-analysis. Am J Med 119: 812–819, 2006 [DOI] [PubMed] [Google Scholar]
- 21.Grundy SM, Benjamin IJ, Burke GL, Chait A, Eckel RH, Howard BV, Mitch W, Smith SC, Jr, Sowers JR. Diabetes and cardiovascular disease: a statement for healthcare professionals from the American Heart Association. Circulation 100: 1134–1146, 1999 [DOI] [PubMed] [Google Scholar]
- 22.Heaps CL, Jeffery EC, Laine GA, Price EM, Bowles DK. Effects of exercise training and hypercholesterolemia on adenosine activation of voltage-dependent K+ channels in coronary arterioles. J Appl Physiol 105: 1761–1771, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Heaps CL, Tharp DL, Bowles DK. Hypercholesterolemia abolishes voltage-dependent K+ channel contribution to adenosine-mediated relaxation in porcine coronary arterioles. Am J Physiol Heart Circ Physiol 288: H568–H576, 2005 [DOI] [PubMed] [Google Scholar]
- 24.Henry P, Thomas F, Benetos A, Guize L. Impaired fasting glucose, blood pressure and cardiovascular disease mortality. Hypertension 40: 458–463, 2002 [DOI] [PubMed] [Google Scholar]
- 25.Hodnett BL, Xiang L, Dearman JA, Carter CB, Hester RL. K(ATP)-mediated vasodilation is impaired in obese Zucker rats. Microcirculation 15: 485–494, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hubert HB, Feinleib M, McNamara PM, Castelli WP. Obesity as an independent risk factor for cardiovascular disease: a 26-year follow-up of participants in the Framingham Heart Study. Circulation 67: 968–977, 1983 [DOI] [PubMed] [Google Scholar]
- 27.Hunt KJ, Resendez RG, Williams K, Haffner SM, Stern MP. National Cholesterol Education Program versus World Health Organization metabolic syndrome in relation to all-cause and cardiovascular mortality in the San Antonio Heart Study. Circulation 110: 1251–1257, 2004 [DOI] [PubMed] [Google Scholar]
- 28.Ishibashi Y, Duncker DJ, Zhang J, Bache RJ. ATP-sensitive K+ channels, adenosine, and nitric oxide-mediated mechanisms account for coronary vasodilation during exercise. Circ Res 82: 346–359, 1998 [DOI] [PubMed] [Google Scholar]
- 29.Ishibashi Y, Takahashi N, Shimada T, Sugamori T, Sakane T, Umeno T, Hirano Y, Oyake N, Murakami Y. Short duration of reactive hyperemia in the forearm of subjects with multiple cardiovascular risk factors. Circ J 70: 115–123, 2006 [DOI] [PubMed] [Google Scholar]
- 30.Jackson WF. Ion channels and vascular tone. Hypertension 35: 173–178, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kiviniemi TO, Snapir A, Saraste M, Toikka JO, Raitakari OT, Ahotupa M, Hartiala JJ, Scheinin M, Koskenvuo JW. Determinants of coronary flow velocity reserve in healthy young men. Am J Physiol Heart Circ Physiol 291: H564–H569, 2006 [DOI] [PubMed] [Google Scholar]
- 32.Knudson JD, Rogers PA, Dincer UD, Bratz IN, Araiza AG, Dick GM, Tune JD. Coronary vasomotor reactivity to endothelin-1 in the prediabetic metabolic syndrome. Microcirculation 13: 209–218, 2006 [DOI] [PubMed] [Google Scholar]
- 33.Kondo I, Mizushige K, Hirao K, Nozaki S, Tsuji T, Masugata H, Kohno M, Matsuo H. Ultrasonographic assessment of coronary flow reserve and abdominal fat in obesity. Ultrasound Med Biol 27: 1199–1205, 2001 [DOI] [PubMed] [Google Scholar]
- 34.Kuo L, Chancellor JD. Adenosine potentiates flow-induced dilation of coronary arterioles by activating KATP channels in endothelium. Am J Physiol Heart Circ Physiol 269: H541–H549, 1995 [DOI] [PubMed] [Google Scholar]
- 35.Lakka HM, Laaksonen DE, Lakka TA, Niskanen LK, Kumpusalo E, Tuomilehto J, Salonen JT. The metabolic syndrome and total and cardiovascular disease mortality in middle-aged men. J Am Med Assoc 288: 2709–2716, 2002 [DOI] [PubMed] [Google Scholar]
- 36.Li H, Chai Q, Gutterman DD, Liu Y. Elevated glucose impairs cAMP-mediated dilation by reducing Kv channel activity in rat small coronary smooth muscle cells. Am J Physiol Heart Circ Physiol 285: H1213–H1219, 2003 [DOI] [PubMed] [Google Scholar]
- 37.Liu Y, Terata K, Rusch NJ, Gutterman DD. High glucose impairs voltage-gated K(+) channel current in rat small coronary arteries. Circ Res 89: 146–152, 2001 [DOI] [PubMed] [Google Scholar]
- 38.Miura H, Liu Y, Gutterman DD. Human coronary arteriolar dilation to bradykinin depends on membrane hyperpolarization: contribution of nitric oxide and Ca2+-activated K+ channels. Circulation 99: 3132–3138, 1999 [DOI] [PubMed] [Google Scholar]
- 39.Miura H, Wachtel RE, Loberiza FR, Jr, Saito T, Miura M, Nicolosi AC, Gutterman DD. Diabetes mellitus impairs vasodilation to hypoxia in human coronary arterioles: reduced activity of ATP-sensitive potassium channels. Circ Res 92: 151–158, 2003 [DOI] [PubMed] [Google Scholar]
- 40.Mokelke EA, Dietz NJ, Eckman DM, Nelson MT, Sturek M. Diabetic dyslipidemia and exercise affect coronary tone and differential regulation of conduit and microvessel K+ current. Am J Physiol Heart Circ Physiol 288: H1233–H1241, 2005 [DOI] [PubMed] [Google Scholar]
- 41.Nelson MT, Patlak JB, Worley JF, Standen NB. Calcium channels, potassium channels, and voltage dependence of arterial smooth muscle tone. Am J Physiol Cell Physiol 259: C3–C18, 1990 [DOI] [PubMed] [Google Scholar]
- 42.Node K, Kitakaze M, Kosaka H, Minamino T, Hori M. Bradykinin mediation of Ca(2+)-activated K+ channels regulates coronary blood flow in ischemic myocardium. Circulation 95: 1560–1567, 1997 [DOI] [PubMed] [Google Scholar]
- 43.Olsson RA. Myocardial reactive hyperemia. Circ Res 37: 263–270, 1975 [DOI] [PubMed] [Google Scholar]
- 44.Ritchie SA, Connell JM. The link between abdominal obesity, metabolic syndrome and cardiovascular disease. Nutr Metab Cardiovasc Dis 17: 319–326, 2007 [DOI] [PubMed] [Google Scholar]
- 45.Ruiter JH, Spaan JA, Laird JD. Transient oxygen uptake during myocardial reactive hyperemia in the dog. Am J Physiol Heart Circ Physiol 235: H87–H94, 1978 [DOI] [PubMed] [Google Scholar]
- 46.Schindler TH, Cardenas J, Prior JO, Facta AD, Kreissl MC, Zhang XL, Sayre J, Dahlbom M, Licinio J, Schelbert HR. Relationship between increasing body weight, insulin resistance, inflammation, adipocytokine leptin, and coronary circulatory function. J Am Coll Cardiol 47: 1188–1195, 2006 [DOI] [PubMed] [Google Scholar]
- 47.Setty S, Sun W, Tune JD. Coronary blood flow regulation in the prediabetic metabolic syndrome. Basic Res Cardiol 98: 416–423, 2003 [DOI] [PubMed] [Google Scholar]
- 48.Sturek M, Alloosh M, Wenzel J, Byrd JP, Edwards JM, Lloyd PG, Tune JD, March KL, Miller MA, Mokelke EA, Brisbin IL. Ossabaw island miniature swine: cardiometabolic syndrome assessment. In: Swine in the Laboratory: Surgery, Anesthesia, Imaging and Experimental Techniques , edited by Swindle MM. Boca Raton, FL: CRC, 2007, p. 397–402 [Google Scholar]
- 49.Zatta AJ, Headrick JP. Mediators of coronary reactive hyperaemia in isolated mouse heart. Br J Pharmacol 144: 576–587, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang C, Knudson JD, Setty S, Araiza A, Dincer UD, Kuo L, Tune JD. Coronary arteriolar vasoconstriction to angiotensin II is augmented in prediabetic metabolic syndrome via activation of AT1 receptors. Am J Physiol Heart Circ Physiol 288: H2154–H2162, 2005. [DOI] [PubMed] [Google Scholar]