Abstract
Background
We previously reported a limb-salvage technique by treating tumor-bearing bone with liquid nitrogen. We also reported systemic antitumor immunity was enhanced by cryotreatment in a murine osteosarcoma (LM8) model. We therefore combined the cryotreatment of tumor with dendritic cells to promote tumor-specific immune responses.
Questions/purposes
We determined whether our technique could enhance systemic immune response and inhibit metastatic tumor growth in a murine osteosarcoma model.
Materials and Methods
To evaluate activation of the immune response, we prepared six groups of C3H mice (80 mice total): (1) excision only, (2) dendritic cells without reimplantation of the cryotreated primary tumor, (3) reimplantation of the cryotreated primary tumor alone, (4) dendritic cells combined with reimplantation of the cryotreated primary tumor, (5) dendritic cells exposed to cryotreated tumor lysates without reimplantation of the cryotreated primary tumor, and (6) dendritic cells exposed to cryotreated tumor lysates with reimplantation of the cryotreated primary tumor. We then compared and verified the activation state of each group’s antitumor immunity.
Results
Mice that received dendritic cells exposed to cryotreated tumor lysates with reimplantation of the cryotreated primary tumor group had high serum interferon γ, reduced pulmonary metastases, and increased numbers of CD8(+) T lymphocytes in the metastatic areas.
Conclusions
Combining tumor cryotreatment with dendritic cells enhanced systemic immune responses and inhibited metastatic tumor growth.
Clinical Relevance
We suggest immunotherapy could be developed further to improve the treatment of osteosarcoma.
Introduction
The standard treatment of osteosarcoma consists of preoperative chemotherapy, surgical tumor excision, and postoperative chemotherapy. Limb-saving surgery is feasible in most cases. Advances in osteosarcoma treatment have now achieved a 5-year survival rate of 60% to 90% for patients, and limb function after reconstruction continues to improve with time [3, 16, 30, 46, 47, 49].
Tsuchiya et al. developed a new approach using frozen autografts [48] to improve reconstruction after osteosarcoma resection. The tumor is resected with an adequate margin, and the resected specimen is immersed in liquid nitrogen for 20 minutes to kill all tumor cells. After thawing, the specimen is returned to the original place with appropriate internal fixation to reconstruct the defect. Compared with heat-treated bones [8, 14], bone genetic proteins and native biomechanical structures are preserved after cryotreatment [53]. In one report limb function using the technique of Tsuchiya et al. was rated as excellent in 71.4% of patients, and good in 10.7%, as assessed by the functional evaluation system of Enneking [11]. Two studies suggest the approach enhanced bone formation when compared histologically with pasteurized bone and irradiated bone [43, 48]. Another advantage in reimplanting cryotreated tumor tissue is its effect on the immune system [50]: tumor tissue after cryoablation in situ provokes an immune reaction in patients with breast and prostate cancer [6, 8, 39]. Brewer et al. reported metastatic tumors sometimes disappear or shrink after in situ cryoablation of the primary tumor with liquid nitrogen [4]. The structure of tumor antigens is retained in frozen tumor, and leukocytes probably can recognize these antigens. Similar antitumor effects can be expected from our reconstructive procedure of reimplanting tumor-bearing bone after cryotreatment with liquid nitrogen.
Nishida et al. observed an inadequate antitumor effect after reimplantation of frozen tumor tissue alone [35]. However, the antitumor effect was enhanced by promoting nonspecific immune activation by intraperitoneal injection of OK-432, a substance extracted from alpha-Streptococcus pyogenes. This approach promotes inflammation and activation of dendritic cells (DCs) that initiate the specific antitumor effect [19]. This type of immunotherapy reportedly is effective for breast and prostate cancers [6, 8, 39]. Many groups have reported successful immunotherapy for osteosarcoma [5, 15, 18, 20, 22, 24, 25, 33, 34, 36, 42, 51, 52]. However, the ability to control metastatic lesions and local recurrence does not appear to be superior to other adjuvant treatments [2, 7, 13, 23, 29].
We therefore wondered whether combining cryotreatment and immunotherapy might enhance tumor response. We specifically determined whether: (1) antitumor immunity could be enhanced through activation and transfer of DCs combined with reimplantation of the cryotreated primary tumor, and (2) metastatic lesions could be prevented owing to the involvement of T lymphocytes in a murine osteosarcoma model (LM8).
Materials and Methods
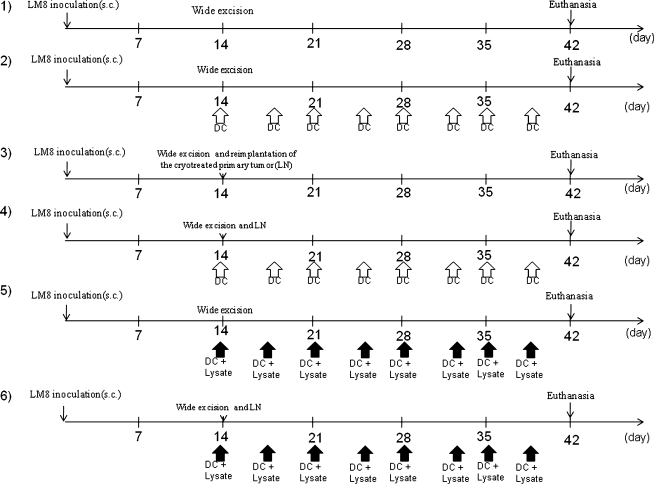
Using a reported method to induce osteosarcoma [1, 35], we hypodermically implanted 1 × 106 LM8 cells (a murine osteosarcoma cell line) into the subcutaneous gluteal region of 80 female C3H mice, 6 to 8 weeks old. Tumors developed in all animals. Two weeks after inoculation, we surgically excised the tumors and cryotreated them with liquid nitrogen. We established the following six groups (Fig. 1): (1) the tumor was excised with wide margins 14 days after inoculation (n = 15); (2) the tumor was excised with wide margins 14 days after inoculation and bone marrow-derived DCs then were injected into the contralateral subcutaneous gluteal region without reimplantation of the cryotreated primary tumor twice a week (n = 15); (3) the tumor was excised with wide margins 14 days after inoculation and reimplanted after cryotreatment with liquid nitrogen into the contralateral gluteal region to evaluate for local recurrence from frozen tumor tissue (n = 15); (4) the tumor was excised 14 days after inoculation and reimplanted after cryotreatment into the contralateral gluteal region to evaluate for local recurrence, and DCs then were injected twice a week into this secondary site (n = 15); (5) the tumor was excised with wide margins 14 days after inoculation and DCs exposed to cryotreated tumor lysates were injected twice a week into the contralateral gluteal region without reimplantation of the cryotreated primary tumor (n = 15); and (6) the tumor was excised with wide margins 14 days after inoculation and reimplanted after the treatment with liquid nitrogen into the contralateral gluteal region to evaluate for local recurrence (same as Group 3) with the addition of DCs exposed to cryotreated tumor lysates injected twice a week (n = 15). We harvested tumor from 30 mice, and then the tumor was treated with liquid nitrogen to create the lysates. We presumed a systemic immune response would be induced by injecting DCs around the frozen tumor tissue. We microscopically determined the presence of metastases in the lungs 2 weeks after the tumor inoculation. We had previously confirmed the presence of pulmonary metastases in an additional 20 mice in a preliminary experiment in advance. We also confirmed that there were no viable cells after cryotreatment using liquid nitrogen, in agreement with a previous study [35]. We observed no recurrence of the tumor at the primary site of inoculation after excision. All experiments were performed under the guidelines for animal experiments as stipulated by the Kanazawa University Graduate School of Medical Science [37].
Fig. 1.
A diagram of the experimental protocol and treatment schedule is shown. Two weeks after tumor inoculation, tumors were treated by one of the following methods: (1) excision only (n = 15); (2) DCs without reimplantation of the cryotreated primary tumor (n = 15); (3) reimplantation of the cryotreated primary tumor (n = 15); (4) DCs pulsed with cryotreated tumor lysates and reimplantation of the cryotreated primary tumor (n = 15); (5) DCs pulsed with cryotreated tumor lysates without reimplantation of the cryotreated primary tumor (n = 15); or (6) DCs pulsed with cryotreated tumor and reimplantation of the cryotreated primary tumor (LN) (n = 15). The mice were euthanized and evaluated 6 weeks after tumor inoculation. sc = subcutaneous.
LM8 cells, derived from Dunn osteosarcoma, were provided by the Riken BioResource Center (Saitama, Japan). The cells were maintained in complete medium consisting of RPMI 1640 supplemented with 10% heat-inactivated fetal bovine serum, 100 μg streptomycin per mL, and 100 units penicillin per mL and were cultured at 37°C in 5% CO2. To establish local implantation of the tumor and subsequent lung metastasis, the LM8 cells (1 × 106) were suspended in 0.2 mL phosphate-buffered saline (PBS) and subcutaneously inoculated into the right gluteal region of the mice. All animals had macroscopically and microscopically confirmed lung metastases within 4 weeks [1].
C3H mice were purchased from Sankyo Labo Inc (Toyama, Japan) and housed in a specific pathogen-free animal facility in our laboratory. We were not able to accurately determine the survival time of each group because the guidelines for animal experiments concerning pain required euthanasia in distressed animals.
Liquid nitrogen (−196°C) was used for cryotreatment. Tumor tissue was collected on gauze and soaked in liquid nitrogen for 20 minutes for en bloc tumor tissue freezing. The tumor was prethawed at room temperature (20°C) for 15 minutes and then thawed in distilled water (20°C) for 15 minutes. The liquid nitrogen-treated tumor tissue was transplanted subcutaneously in the left gluteal region of the same mouse.
Because the mice were genetically identical, the structure of the major histocompatibility complex (MHC) Class I molecules was such that the T cells would be able to recognize the MHC Class I with antigens on the antigen-presenting cells (APCs) [17, 27]. Bone marrow-derived DCs were generated as described by Lutz and Rössner [28] with minor modifications. Briefly, erythrocyte-depleted mouse bone marrow cells obtained from flushed marrow cavities (1 × 106 cells/mL) were cultured in complete medium with 20 ng/mL recombinant mouse GMCSF (PeproTech EC Ltd, London, UK) in 10-cm tissue culture dishes at 37°C in an atmosphere containing 50 mL CO2 per L. On Days 3 and 6, half of the medium was added to the same volume of fresh complete medium and used to replenish the original plates. The freeze-thawed tumor lysate was added to the DC cultures on Day 6 at a ratio of five DC equivalents to one tumor cell (ie, 5:1) and incubated at 37°C in an atmosphere containing 50 mL CO2 per L. After 24 hours of incubation, nonadherent cells including DCs were harvested by gentle pipetting.
For fluorescence activated cell sorting (FACS) analysis, DCs were counted with a FACSCalibur™ Flow Cytometer (Becton-Dickinson, San Jose, CA) and stained with fluorochrome-conjugated antibodies (BD Pharmingen, Tokyo, Japan) for the following markers: cluster of differentiation (CD)11c, CD80, CD86, I-Ad, and CD40. CD11c was used as a marker for all DCs regardless of the degree of maturation, whereas CD80, CD86, I-Ad, and CD40 are markers for DCs. Data analysis was performed with CELLQuest™ software (Becton-Dickinson). The corresponding labeled isotype antibodies served as controls. DCs used for vaccination were washed twice, enumerated, and resuspended in PBS at 1 × 106/mL.
We inoculated LM8 cells (5 × 106) in a mouse to make the tumor lysate. After 4 weeks, we resected the tumor mass and soaked the entire tumor in liquid nitrogen to kill the tumor cells. We mixed cryonecrotic tissue with DCs at Culture Day 6, after the tumor was defrosted, and the homogenate was prepared using PBS. The homogenate was passed through a 0.2-μm filter to remove bacteria and tissues and mixed with the DCs for 24 hours.
After intraperitoneal injection of 5 mL sodium pentobarbital (Somnopentyl®; Kyontsu Seiyaku, Tokyo, Japan), mice were euthanized by cervical dislocation and their blood was collected. Murine interferon (IFN)-γ and interleukin (IL)-4 release were measured by ELISA using Quantikine® (R & D Systems, Minneapolis, MN) according to the manufacturer’s protocol using an Easy Reader EAR340 microtest plate reader (SLT-Labinstruments, Salzburg, Austria).
We estimated the area of the pulmonary metastatic lesion on 50 serial histologic sections of each lung by manually drawing orthogonal lines delimiting the edges of the pulmonary metastatic lesion and selected the widest part of the specimen. The area was determined by multiplying the maximum orthogonal dimensions using ImageJ software (NIH, Bethesda, MD; http://rsb.info.nih.gov/ij/). We compared the mean areas between the six groups.
For immunohistochemistry, lung specimens were fixed in 20% formalin and embedded in paraffin. For each case, we examined all the blocks of lung tissues of formalin-fixed, paraffin-embedded tumor tissue. All specimens were decalcified, although we found the decalcification step did not influence the immunohistochemistry for any of the stains. Five sections for each mouse were cut 4-μm thick. Each section was cut at the maximum diameter. CD8(+) T lymphocytes and natural killer (NK) cells in the pulmonary metastatic lesion were quantified by measuring the immunohistochemistry-positive cells per unit area in each group. Rehydrated tissue sections were incubated with rat monoclonal antibody raised against CD8(+) T lymphocytes of mouse origin (Santa Cruz Biotechnology, Santa Cruz, CA) and rat monoclonal antibody raised against NK cells of mouse origin (Abcam Plc, Cambridge, UK). The two antibodies were diluted 1:50 with PBS. Color reactions were performed at room temperature for 15 minutes and cover slips were mounted with glycerol and gelatin.
We determined differences in serum IFN-γ, serum IL-4, pulmonary metastatic area, and number of CD8(+) lymphocytes and NK cells in the metastatic area among the six groups using a nonrepeated-measures ANOVA and the Scheffe test. All analyses were conducted with SPSS® 11.0 software (SPSS Japan Inc, Tokyo, Japan).
Results
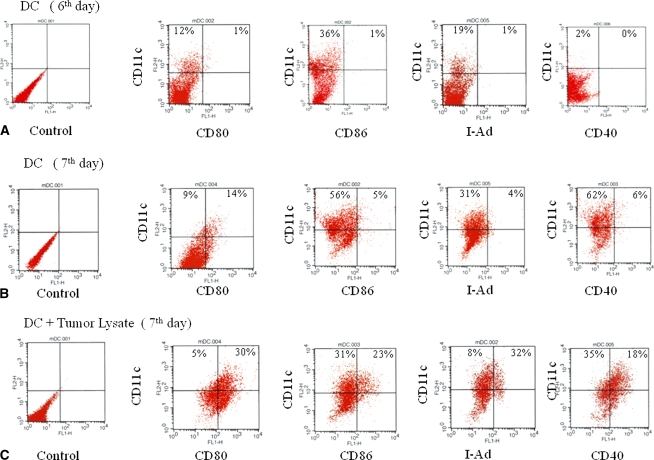
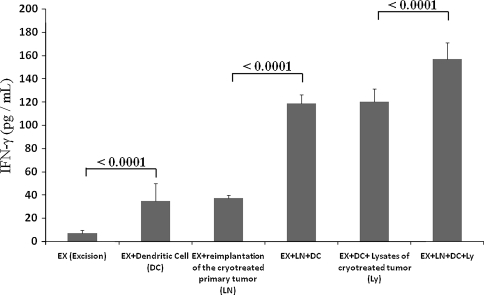
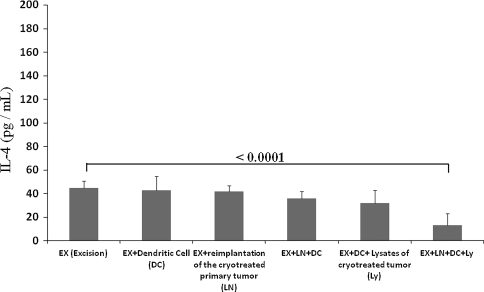
We activated antitumor immunity by combining DCs exposed to lysates of cryotreated tumor and reimplantation of the cryotreated primary tumor. On Culture Day 7, the ratio of mature DCs to immature DCs was increased compared with the ratio at Culture Day 6 (Fig. 2; immature DCs, upper left; mature DCs, upper right). Moreover, this increase was more apparent in groups incubated with tumor lysate. Serum IFN-γ levels were greater (p < 0.0001) in the mice that received DCs combined with reimplantation of the cryotreated primary tumor (119.0 ± 7.61 pg/mL) than in the cryotreated primary tumor alone group (37.33 ± 2.58 pg/mL). Moreover, the group that received tumor lysate-exposed DCs combined with reimplantation of the cryotreated primary tumor (157.33 ± 14 pg/mL) had a greater (p < 0.0001) IFN-γ level than the group that received only tumor lysate-exposed DCs without reimplantation of the cryotreated primary tumor (120.27 ± 11.29 pg/mL) (Fig. 3). Serum IL-4 was lower (p < 0.0001) in the mice that received DCs exposed to the lysates of cryotreated tumor and reimplantation of the cryotreated primary tumor group (13.33 ± 9.75 pg/mL) than in the excision-only group (45.06 ± 5.71 pg/mL) (Fig. 4).
Fig. 2A–C.
DC activation status was examined using flow cytometry. DCs at Culture Day 7 (Group B) were more mature than DCs at Culture Day 6 (Group A). On Culture Day 7, DC maturity was greatest in the groups receiving lysate-primed DCs (Group C) than in those not receiving lysate-primed DCs (Group B).
Fig. 3.
A graph of the serum IFN-γ levels in the six treatment groups is shown. The samples were collected 28 days after the reimplantation surgery and/or DC adoptive transfer. Mice that received DCs exposed to the lysates of cryotreated tumor and reimplantation of the cryotreated primary tumor group showed a highest IFN-γ level. Error bars represent SD.
Fig. 4.
A graph of the serum IL-4 in the six treatment groups is shown. Sera were collected 28 days after the reimplantation surgery and/or DC adoptive transfer. DCs exposed to the lysates of cryotreated tumor and reimplantation of the cryotreated primary tumor group showed a lower level than any of the other groups. Error bars represent SD.
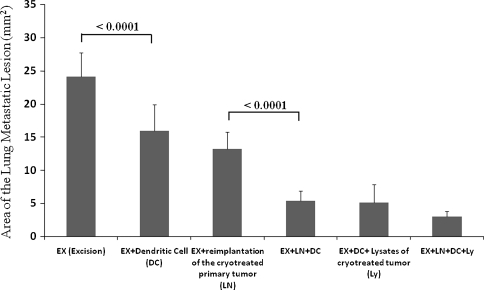
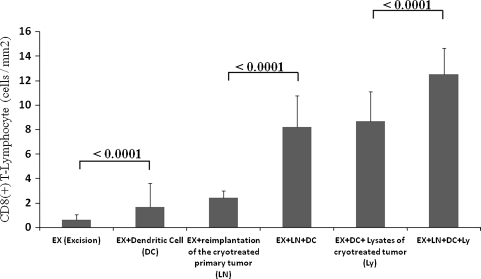
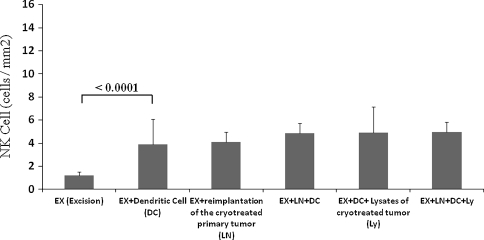
The enhanced immune response by T lymphocytes reduced metastatic lesions. Reduction of the metastatic area was greater (p < 0.0001) in the group that received DCs without reimplantation of the cryotreated primary tumor (15.99 ± 3.93 mm2) than in the excision-only group (24.12 ± 3.60 mm2). The reduction of the metastatic area was greater (p < 0.0001) in the DCs combined with reimplantation of the cryotreated primary tumor group (5.39 ± 1.49 mm2) than in the reimplantation of the cryotreated primary tumor alone group (13.22 ± 2.59 mm2) (Fig. 5). CD8(+) T lymphocytes gathered in the pulmonary metastatic area in DC-treated groups, however, NK cells were not recruited to the metastatic area in the DC-treated groups compared with the nonDC-treated groups (Fig. 6). The number of CD8(+) T lymphocytes per unit area was greater (p < 0.0001) in the DCs combined with reimplantation of the cryotreated primary tumor group (8.33 ± 2.57 cells/mm2) than in the reimplantation of the cryotreated primary tumor alone group (2.44 ± 0.53 cells/mm2). Mice that received DCs exposed to the lysates of cryotreated tumor and reimplantation of the cryotreated primary tumor (12.79 ± 2.14 cells/mm2) showed higher (p < 0.0001) levels than the group that received DCs exposed to the lysates of cryotreated tumor without reimplantation of the cryotreated primary tumor (8.71 ± 2.39 cells/mm2) (Fig. 7). The number of NK cells per unit area was greater (p < 0.0001) in the group that received DCs exposed to the lysates of cryotreated tumor without reimplantation of the cryotreated primary tumor (3.90 ± 2.17 cells/mm2) than in the excision-only group (1.20 ± 0.30 cells/mm2) (Fig. 8). The CD8(+)T lymphocyte, CD4(+) T lymphocyte, and DC infiltrations in reimplanted tumors were similar to those seen with pulmonary metastases (data not shown).
Fig. 5.
Reduction of the metastatic area in the six treatment groups is shown. The samples were gathered 28 days after the reimplantation surgery and/or DC adoptive transfer. Error bars represent SD.
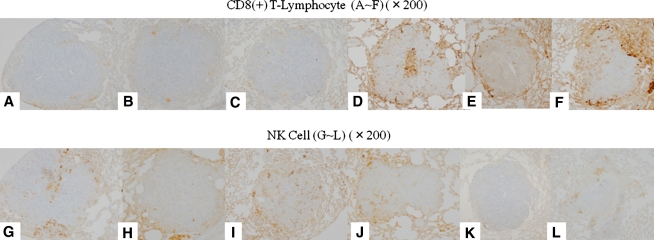
Fig. 6A–L.
To evaluate CD8(+) T lymphocytes and NK cells in pulmonary metastasis, immunostaining was performed: (A) CD8(+) T lymphocytes in Group 1, (B) CD8(+) T lymphocytes in Group 2, (C) CD8(+) T lymphocytes in Group 3, (D) CD8(+) T lymphocytes in Group 4, (E) CD8(+) T lymphocytes in Group 5, (F) CD8(+) T lymphocytes in Group 6, (G) NK cells in Group 1, (H) NK cells in Group 2, (I) NK cells in Group 3, (J) NK cells in Group 4, (K) NK cells in Group 5, and (L) NK cells in Group 6. CD8(+) T lymphocytes gathered in Groups D,E, and F. However, they did not gather in Groups A, B, and C. However, NK cells were recruited only in Groups A, B, and C. (Original magnification, ×200).
Fig. 7.
The numbers of CD8(+) T lymphocytes per unit area in the six treatment groups are shown. The samples were gathered 28 days after the reimplantation surgery and/or DC adoptive transfer. DCs exposed to the lysates of cryotreated tumor and reimplantation of the cryotreated primary tumor group showed a higher level than any other groups. Error bars represent SD.
Fig. 8.
The numbers of NK cells per unit area in the six treatment groups are shown. The samples were gathered 28 days after the reimplantation surgery and/or DC adoptive transfer. Error bars represent SD.
Discussion
Various immunotherapies for osteosarcoma have been tried. As standard treatments for osteosarcoma are ineffectual for many patients, new treatments need to be developed. In the 1970s, immunotherapy for osteosarcoma was reported by Southam et al. [42], Neff and Enneking [34], and Campbell et al. [5]. In the 1980s, new methods such as the use of interferons and Bacille de Calmette et Guérin were reported [22, 24, 36]. Another approach used antiidiotypic antibodies using T cells and liposome encapsulation [18, 51, 52]. Current methods of immunotherapy for osteosarcoma include peptide therapy or gene transfer therapy combined with hyperthermia therapy [10, 15, 21, 25, 33]. We asked whether (1) antitumor immunity could be achieved through activation of DCs combined with reimplantation of the cryotreated primary tumor and (2) if metastatic lesions would be prevented owing to enhanced T lymphocyte involvement.
We acknowledge limitations in this study. First, we used mice with an identical genetic makeup. The structure of the MHC Class I molecules was similar and the T cells could recognize the MHC Class I. However, we needed to use DCs from a different (albeit genetically identical) mouse to accomplish our adoptive transfer experiments. We minimized the potential for an immune response to nonself antigens by using genetically identical tumor tissue and mice. It would be necessary to use DCs derived from the same individual in clinical application, but this could not be achieved in our mouse model. In humans, however, monocytes are separated from the patient’s own peripheral blood and DCs can be induced from these monocytes. Second, we could not completely replicate the clinical approach used in humans in our mouse model. In clinical cases frozen bone always is returned to the same site. However, it was impossible to replicate this in our experimental mouse model in which transplanted tumor cells were removed from the tibia and then returned to the same place after cryotreatment. In a preliminary experiment we attempted to do just that and these 20 mice could not move and died of starvation. We therefore used the contralateral gluteal region to check for local recurrence after tumor excision or recurrence from frozen tissue.
Antitumor immunity appeared to be activated through DCs combined with reimplantation of the cryotreated primary tumor or by exposing the transferred DC to lysates of cryotreated tumor. The use of lymphokine-activated killer (LAK) therapy has been used with other types of tumors [26]. However, T lymphocytes, which are the effectors, do not accumulate inside osteosarcoma tumors as expected. Autoclaving supplemented by DCs is thought to enhance the antitumor effect, but hyperthermia causes proteins to denature, and activation of the antitumor effect is often insufficient [37]. Several studies [12, 31, 41] report peptide vaccine therapy, but many patients apparently develop immunotolerance [45]. Thus, immunotherapy for malignant tumor achieved by these various methods has not been established definitively although investigations continue to try to overcome the major hurdles associated with immunotherapy (Table 1). We emphasize the immune response is activated by cryotreatment but not by heat-treated tissue. Our method differs from those described by others [7, 9, 10, 14]. In some regards DCs are believed to be the principal APCs for initiating immune responses in vivo [32]. In comparison with other traditional adjunct therapeutic options for cancer, such as radiation therapy and chemotherapy, immunotherapy provides a more targeted treatment to the cancer, with potentially fewer detrimental effects on noncancerous cells [30, 40]. DCs without sufficient cancer antigens may not have the ability to kill tumor cells and present the antigen to T lymphocytes by themselves. Our data suggest the antitumor effect in the group that received DCs without reimplantation of cryotreated primary tumor was almost the same as that in the reimplantation of cryotreated primary tumor alone group. The data further suggest the effects increased only when exposing the DCs to tumor lysates in the absence of cryonecrotic primary tumors. However, combining reimplantation of cryotreated primary tumor and DCs exposed to cryotreated tumor lysates produced synergistic effects. Using reimplantation of cryotreated primary tumor is more appropriate for clinical applications. We therefore believe an efficient immune response will be activated when DCs recognize tumor antigens appropriately. CD8(+) T cells act as an effector by the Th1 route, and this is promoted mainly by IFN-γ and IL-12 [38]. However, IL-4 [21], IL-6, and IL-10 strengthen humoral immunity. Levels of IFN-γ, IL-2, and IL-12 generally increase when cell-mediated immunity is activated, and IL-4, IL-6, and IL-10 increase when humoral immunity is activated. These cytokines act in opposition to maintain an immune balance.
Table 1.
Immunotherapeutic trials of malignant tumors
| Tumor | Immune intervention | Route | Immunologic response | Comments | References |
|---|---|---|---|---|---|
| Osteosarcoma | BCG | SC | NC | No consistent clinical effect | [22, 24] |
| Osteosarcoma | Interferon α | SC, IV | PR–NC | Osteosarcoma-associated antigens have potential for targeted immunotherapy | [36] |
| Unknown | LAK | IV | NC | T lymphocytes were unable to penetrate the tumor | [26] |
| Osteosarcoma | Antiidiotypic antibodies | IV | NC | It may be possible to circumvent this heterogeneity by activation of tissue macrophages to the tumoricidal state | [18, 51, 52] |
| Breast cancer, osteosarcoma | Peptide therapy combined with hyperthermia therapy | SC, IV | NC | It may be a potential agent for use in immunotherapy | [15, 20] |
| Osteosarcoma | Gene transfer therapy combined with hyperthermia therapy | IV | NC | IL-23 seems to be a less effective immunotherapeutic for adjuvant treatment of osteosarcomas | [25, 33] |
| Unknown | Peptide vaccine therapy | SC | NC–PD | Many patients have peptide-induced tolerance develop | [45] |
| Osteosarcoma | Cryoimmunology and DCs | SC | PR | Combining cryotreatment with DCs resulted in enhanced antitumor effects | Our data |
BCG = Bacille de Calmette et Guérin; SC = subcutaneous; NC = no change; IV = intravenous; PR = partial response; LAK = lymphokine-activated killer; IL = interleukin; PD = progressive disease; DCs = dendritic cells.
Our data suggest enhanced T lymphocyte recruitment and function reduce metastatic lesions in a murine osteosarcoma model. Immunoreactivity increased slightly in mice that received DCs exposed to lysates of cryotreated tumor combined with reimplantation of the cryotreated primary tumor. NK cells attack the tumor independently of APCs. NK cells attack cells that downregulate MHC Class I expression or have a stressed appearance [44]. We observed a reduced tumor burden in the groups that received transplanted DCs, which correlated with recruitment of CD8 lymphocytes to the tumor site as observed with immunohistochemistry.
Returning the frozen bone after liquid nitrogen treatment to its original place can be readily used in the clinic. After the first cryotreatment, it is possible to perform the treatment again using cultured DCs if a patient’s tumor cells have been preserved. This approach therefore still can be used even after other methods (such as chemotherapy, radiation therapy, or surgery) no longer are reasonable. Combining DCs pulsed with lysates of cryotreated tumor and reimplantation of the cryotreated primary tumor enhanced antitumor effects. We believe the approach may be a useful alternative for patients with osteosarcoma when other treatment options including chemotherapy, radiotherapy, and surgical treatment have been ineffective.
Acknowledgments
We thank Katsuro Tomita, Akihiko Takeuchi, Shuichi Kaneko, and Yohei Marukawa for supervision in this study.
Footnotes
Each author certifies that he or she has no commercial associations that might pose a conflict of interest in connection with the submitted article.
Each author certifies that his or her institution has approved the animal protocol for this investigation, and that all investigations were conducted in conformity with ethical principles of research.
This work was performed at the Department of Orthopaedic Surgery, Graduate School of Medical Science, Kanazawa University, and the Department of Orthopaedic Surgery, Faculty of Medicine, Oita University.
References
- 1.Asai T, Ueda T, Itoh K, Yoshioka K, Aoki Y, Mori S, Yoshikawa H. Establishment and characterization of a murine osteosarcoma cell line (LM8) with high metastatic potential to the lung. Int J Cancer. 1998;76:418–422. doi: 10.1002/(SICI)1097-0215(19980504)76:3<418::AID-IJC21>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 2.Bacci G, Lari S. Adjuvant and neoadjuvant chemotherapy in osteosarcoma. Chir Organi Mov. 2001;86:253–268. [PubMed] [Google Scholar]
- 3.Bielack SS, Kempf-Bielack B, Delling G, Exner GU, Flege S, Helmke K, Kotz R, Salzer-Kuntschik M, Werner M, Winkelmann W, Zoubek A, Jürgens H, Winkler K. Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1, 702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol. 2002;20:776–790. doi: 10.1200/JCO.20.3.776. [DOI] [PubMed] [Google Scholar]
- 4.Brewer WH, Austin RS, Capps GW, Neifeld JP. Intraoperative monitoring and postoperative imaging of hepatic cryosurgery. Semin Surg Oncol. 1998;14:129–155. doi: 10.1002/(SICI)1098-2388(199803)14:2<129::AID-SSU6>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 5.Campbell CJ, Cohen J, Enneking WF. Editorial: New therapies for osteogenic sarcoma. J Bone Joint Surg Am. 1975;57:143–144. [PubMed] [Google Scholar]
- 6.Chin JL, Lim D, Abdelhady M. Review of primary and salvage cryo-ablation for prostate cancer. Cancer Control. 2007;14:231–237. doi: 10.1177/107327480701400305. [DOI] [PubMed] [Google Scholar]
- 7.DeLaney TF, Park L, Goldberg SI, Hug EB, Liebsch NJ, Munzenrider JE, Suit HD. Radiotherapy for local control of osteosarcoma. Int J Radiat Oncol Biol Phys. 2005;61:492–498. doi: 10.1016/j.ijrobp.2004.05.051. [DOI] [PubMed] [Google Scholar]
- 8.Moraes AM, Pavarin LB, Herreros F, Aguiar Michelman F, Velho PE, Souza EM. Cryosurgical treatment of lentigo maligna. J Dtsch Dermatol Ges. 2007;5:477–480. doi: 10.1111/j.1610-0387.2007.06331.x. [DOI] [PubMed] [Google Scholar]
- 9.Dinçbaş FO, Koca S, Mandel NM, Hiz M, Dervişoğlu S, Seçmezacar H, Oksüz DC, Ceylaner B, Uzel B. The role of preoperative radiotherapy in nonmetastatic high-grade osteosarcoma of the extremities for limb-sparing surgery. Int J Radiat Oncol Biol Phys. 2005;62:820–828. doi: 10.1016/j.ijrobp.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 10.Duparc J, Massin P, Bocquet L, Benfrech E, Cavagna R. [Autoclaved tumoral autografts: apropos of 12 cases, 6 of which highly malignant] [in French] Rev Chir Orthop Reparatrice Appar Mot. 1993;79:261–271. [PubMed] [Google Scholar]
- 11.Enneking WF. A system for functional evaluation of the surgical management of musculoskeletal tumors. In: Enneking WF, editor. Limb Salvage in Musculoskeletal Oncology. New York, NY: Churchill-Livingstone; 1987. pp. 5–16. [Google Scholar]
- 12.Enomoto Y, Bharti A, Khaleque AA, Song B, Liu C, Apostolopoulos V, Xing PX, Calderwood SK, Gong J. Enhanced immunogenicity of heat shock protein 70 peptide complexes from dendritic cell-tumor fusion cells. J Immunol. 2006;177:5946–5955. doi: 10.4049/jimmunol.177.9.5946. [DOI] [PubMed] [Google Scholar]
- 13.Fagioli F, Biasin E, Mereuta OM, Muraro M, Luksch R, Ferrari S, Aglietta M, Madon E. Poor prognosis osteosarcoma: new therapeutic approach. Bone Marrow Transplant. 2008;41(suppl 2):S131–S134. doi: 10.1038/bmt.2008.71. [DOI] [PubMed] [Google Scholar]
- 14.Harrington KD. The use of hemipelvic allografts or autoclaved grafts for reconstruction after wide resections of malignant tumors of the pelvis. J Bone Joint Surg Am. 1992;74:331–341. [PubMed] [Google Scholar]
- 15.Herbert LM, Grosso JF, Dorsey M, Jr, Fu T, Keydar I, Cejas MA, Wreschner DH, Smorodinski N, Lopez DM. A unique mucin immunoenhancing peptide with antitumor properties. Cancer Res. 2004;64:8077–8084. doi: 10.1158/0008-5472.CAN-04-0853. [DOI] [PubMed] [Google Scholar]
- 16.Hugate RR, Wilkins RM, Kelly CM, Madsen W, Hinshaw I, Camozzi AB. Intraarterial chemotherapy for extremity osteosarcoma and MFH in adults. Clin Orthop Relat Res. 2008;466:1292–1301. doi: 10.1007/s11999-008-0252-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Inaba K, Inaba M, Romani N, Aya H, Deguchi M, Ikehara S, Muramatsu S, Steinman RM. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J Exp Med. 1992;176:1693–1702. doi: 10.1084/jem.176.6.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Killion JJ, Fidler IJ. Systemic targeting of liposome-encapsulated immunomodulators to macrophages for treatment of cancer metastasis. Immunomethods. 1994;4:273–279. doi: 10.1006/immu.1994.1029. [DOI] [PubMed] [Google Scholar]
- 19.Koido S, Hara E, Homma S, Torii A, Mitsunaga M, Yanagisawa S, Toyama Y, Kawahara H, Watanabe M, Yoshida S, Kobayashi S, Yanaga K, Fujise K, Tajiri H. Streptococcal preparation OK-432 promotes fusion efficiency and enhances induction of antigen-specific CTL by fusions of dendritic cells and colorectal cancer cells. J Immunol. 2007;178:613–622. doi: 10.4049/jimmunol.178.1.613. [DOI] [PubMed] [Google Scholar]
- 20.Kubista B, Trieb K, Blahovec H, Kotz R, Micksche M. Hyperthermia increases the susceptibility of chondro- and osteosarcoma cells to natural killer cell-mediated lysis. Anticancer Res. 2002;22:789–792. [PubMed] [Google Scholar]
- 21.Kumaratilake LM, Ferrante A. IL-4 inhibits macrophage-mediated killing of Plasmodium falciparum in vitro: a possible parasite-immune evasion mechanism. J Immunol. 1992;149:194–199. [PubMed] [Google Scholar]
- 22.Larsson SE, Lorentzon R, Boquist L. Immunotherapy with irradiated tumour cells and BCG in experimental osteosarcoma. Acta Orthop Scand. 1981;52:469–474. doi: 10.3109/17453678108992134. [DOI] [PubMed] [Google Scholar]
- 23.Lee JW, Kim H, Kang HJ, Kim HS, Park SH, Kim IO, Ahn HS, Shin HY. Clinical characteristics and treatment results of pediatric osteosarcoma: the role of high dose chemotherapy with autologous stem cell transplantation. Cancer Res Treat. 2008;40:172–177. doi: 10.4143/crt.2008.40.4.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Leventhal BG. Immunotherapy of sarcomas. Natl Cancer Inst Monogr. 1981;56:183–187. [PubMed] [Google Scholar]
- 25.Liebau C, Roesel C, Schmidt S, Karreman C, Prisack JB, Bojar H, Merk H, Wolfram N, Baltzer AW. Immunotherapy by gene transfer with plasmids encoding IL-12/IL-18 is superior to IL-23/IL-18 gene transfer in a rat osteosarcoma model. Anticancer Res. 2004;24:2861–2867. [PubMed] [Google Scholar]
- 26.Lotze MT, Line BR, Mathisen DJ, Rosenberg SA. The in vivo distribution of autologous human and murine lymphoid cells grown in T cell growth factor (TCGF): implications for the adoptive immunotherapy of tumors. J Immunol. 1980;125:1487–1493. [PubMed] [Google Scholar]
- 27.Lutz MB, Kukutsch N, Ogilvie AL, Rössner S, Koch F, Romani N, Schuler G. An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. J Immunol Methods. 1999;223:77–92. doi: 10.1016/S0022-1759(98)00204-X. [DOI] [PubMed] [Google Scholar]
- 28.Lutz MB, Rössner S. Factors influencing the generation of murine dendritic cells from bone marrow: the special role of fetal calf serum. Immunobiology. 2007;212:855–862. doi: 10.1016/j.imbio.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 29.Machak GN, Tkachev SI, Solovyev YN, Sinyukov PA, Ivanov SM, Kochergina NV, Ryjkov AD, Tepliakov VV, Bokhian BY, Glebovskaya VV. Neoadjuvant chemotherapy and local radiotherapy for high-grade osteosarcoma of the extremities. Mayo Clin Proc. 2003;78:147–155. doi: 10.4065/78.2.147. [DOI] [PubMed] [Google Scholar]
- 30.Meyers PA, Schwartz CL, Krailo M, Kleinerman ES, Betcher D, Bernstein ML, Conrad E, Ferguson W, Gebhardt M, Goorin AM, Harris MB, Healey J, Huvos A, Link M, Montebello J, Nadel H, Nieder M, Sato J, Siegal G, Weiner M, Wells R, Wold L, Womer R, Grier H. Osteosarcoma: a randomized, prospective trial of the addition of ifosfamide and/or muramyl tripeptide to cisplatin, doxorubicin, and high-dose methotrexate. J Clin Oncol. 2005;23:2004–2011. doi: 10.1200/JCO.2005.06.031. [DOI] [PubMed] [Google Scholar]
- 31.Monzavi-Karbassi B, Hennings LJ, Artaud C, Liu T, Jousheghany F, Pashov A, Murali R, Hutchins LF, Kieber-Emmons T. Preclinical studies of carbohydrate mimetic peptide vaccines for breast cancer and melanoma. Vaccine. 2007;25:3022–3031. doi: 10.1016/j.vaccine.2007.01.072. [DOI] [PubMed] [Google Scholar]
- 32.Morikawa Y, Tohya K, Ishida H, Matsuura N, Kakudo K. Different migration patterns of antigen-presenting cells correlate with Th1/Th2-type responses in mice. Immunology. 1995;85:575–581. [PMC free article] [PubMed] [Google Scholar]
- 33.Nakashima Y, Deie M, Yanada S, Sharman P, Ochi M. Magnetically labeled human natural killer cells, accumulated in vitro by an external magnetic force, are effective against HOS osteosarcoma cells. Int J Oncol. 2005;27:965–971. [PubMed] [Google Scholar]
- 34.Neff JR, Enneking WF. Adoptive immunotherapy in primary osteosarcoma: an interim report. J Bone Joint Surg Am. 1975;57:145–148. [PubMed] [Google Scholar]
- 35.Nishida H, Tsuchiya H, Tomita K. Re-implantation of destructive tumour tissue treated by liquid nitrogen cryotreatment induces anti-tumour activity against murine osteosarcoma. J Bone Joint Surg Br. 2008;90:1249–1255. doi: 10.1302/0301-620X.90B9.20671. [DOI] [PubMed] [Google Scholar]
- 36.Pelham JM, Gray JD, Flannery GR, Pimm MV, Baldwin RW. Interferon-alpha conjugation to human osteogenic sarcoma monoclonal antibody 791T/36. Cancer Immunol Immunother. 1983;15:210–216. doi: 10.1007/BF00199167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Research Promotion Bureau, Life Sciences Divisions. Fundamental guidelines for proper conduct of animal experiments and related activities in academic research institutions under the jurisdiction of the Ministry of Education, Culture, Sports, Science and Technology. Ministry of Education, Culture, Sports, Science and Technology, Notice No. 71. Available at: http://www.lifescience.mext.go.jp/policies/pdf/an_material011.pdf. Accessed October 29, 2007.
- 38.Romieu R, Baratin M, Kayibanda M, Guillet JG, Viguier M. IFN-gamma-secreting Th cells regulate both the frequency and avidity of epitope-specific CD8 + T lymphocytes induced by peptide immunization: an ex vivo analysis. Int Immunol. 1998;10:1273–1279. doi: 10.1093/intimm/10.9.1273. [DOI] [PubMed] [Google Scholar]
- 39.Sabel MS, Kaufman CS, Whitworth P, Chang H, Stocks LH, Simmons R, Schultz M. Cryoablation of early-stage breast cancer: work-in-progress report of a multi-institutional trial. Ann Surg Oncol. 2004;11:542–549. doi: 10.1245/ASO.2004.08.003. [DOI] [PubMed] [Google Scholar]
- 40.Schendel DJ, Gansbacher B, Oberneder R, Kriegmair M, Hofstetter A, Riethmüller G, Segurado OG. Tumor-specific lysis of human renal cell carcinomas by tumor-infiltrating lymphocytes. I. HLA-A2-restricted recognition of autologous and allogeneic tumor lines. J Immunol. 1993;151:4209–4220. [PubMed] [Google Scholar]
- 41.Slingluff CL, Jr, Chianese-Bullock KA, Bullock TN, Grosh WW, Mullins DW, Nichols L, Olson W, Petroni G, Smolkin M, Engelhard VH. Immunity to melanoma antigens: from self-tolerance to immunotherapy. Adv Immunol. 2006;90:243–295. doi: 10.1016/S0065-2776(06)90007-8. [DOI] [PubMed] [Google Scholar]
- 42.Southam CM, Marcove R, Shanks E. Clinical trials of autogenous tumor vaccine for treatment of osteogenic sarcoma. Proceedings of the Seventh National Cancer Conference. Philadelphia, PA: JB Lippincott; 1973:91. [PubMed]
- 43.Tanzawa Y, Tsuchiya H, Yamamoto N, Sakayama K, Minato H, Tomita K. Histological examination of frozen autograft treated by liquid nitrogen removed 6 years after implantation. J Orthop Sci. 2008;13:259–264. doi: 10.1007/s00776-007-1222-2. [DOI] [PubMed] [Google Scholar]
- 44.Terunuma H, Deng X, Dewan Z, Fujimoto S, Yamamoto N. Potential role of NK cells in the induction of immune responses: implications for NK cell-based immunotherapy for cancers and viral infections. Int Rev Immunol. 2008;27:93–110. doi: 10.1080/08830180801911743. [DOI] [PubMed] [Google Scholar]
- 45.Toes RE, Blom RJ, Offringa R, Kast WM, Melief CJ. Enhanced tumor outgrowth after peptide vaccination: functional deletion of tumor-specific CTL induced by peptide vaccination can lead to the inability to reject tumors. J Immunol. 1996;156:3911–3918. [PubMed] [Google Scholar]
- 46.Tsuchiya H, Tomita K, Mori Y, Asada N, Morinaga T, Kitano S, Yamamoto N. Caffeine-assisted chemotherapy and minimized tumor excision for nonmetastatic osteosarcoma. Anticancer Res. 1998;18:657–666. [PubMed] [Google Scholar]
- 47.Tsuchiya H, Tomita K, Mori Y, Asada N, Yamamoto N. Marginal excision for osteosarcoma with caffeine assisted chemotherapy. Clin Orthop Relat Res. 1999;358:27–35. doi: 10.1097/00003086-199901000-00005. [DOI] [PubMed] [Google Scholar]
- 48.Tsuchiya H, Wan SL, Sakayama K, Yamamoto N, Nishida H, Tomita K. Reconstruction using an autograft containing tumour treated by liquid nitrogen. J Bone Joint Surg Br. 2005;87:218–225. doi: 10.1302/0301-620x.87b2.15325. [DOI] [PubMed] [Google Scholar]
- 49.Tsuchiya H, Yasutake H, Yokogawa A, Baba H, Ueda Y, Tomita K. Effect of chemotherapy combined with caffeine for osteosarcoma. J Cancer Res Clin Oncol. 1992;118:567–569. doi: 10.1007/BF01211797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Urano M, Tanaka C, Sugiyama Y, Miya K, Saji S. Antitumor effects of residual tumor after cryoablation: the combined effect of residual tumor and a protein-bound polysaccharide on multiple liver metastases in a murine model. Cryobiology. 2003;46:238–245. doi: 10.1016/S0011-2240(03)00039-7. [DOI] [PubMed] [Google Scholar]
- 51.Visonneau S, Cesano A, Jeglum KA, Santoli D. Adjuvant treatment of canine osteosarcoma with the human cytotoxic T-cell line TALL-104. Clin Cancer Res. 1999;5:1868–1875. [PubMed] [Google Scholar]
- 52.Warren RQ, Tsang KY. Induction of immunity to a human osteosarcoma-associated antigen in mice using anti-idiotypic antibodies. Clin Immunol Immunopathol. 1990;56:334–343. doi: 10.1016/0090-1229(90)90154-I. [DOI] [PubMed] [Google Scholar]
- 53.Yamamoto N, Tsuchiya H, Tomita K. Effects of liquid nitrogen treatment on the proliferation of osteosarcoma and the biomechanical properties of normal bone. J Orthop Sci. 2003;8:374–380. doi: 10.1007/s10776-002-0626-3. [DOI] [PubMed] [Google Scholar]