Abstract
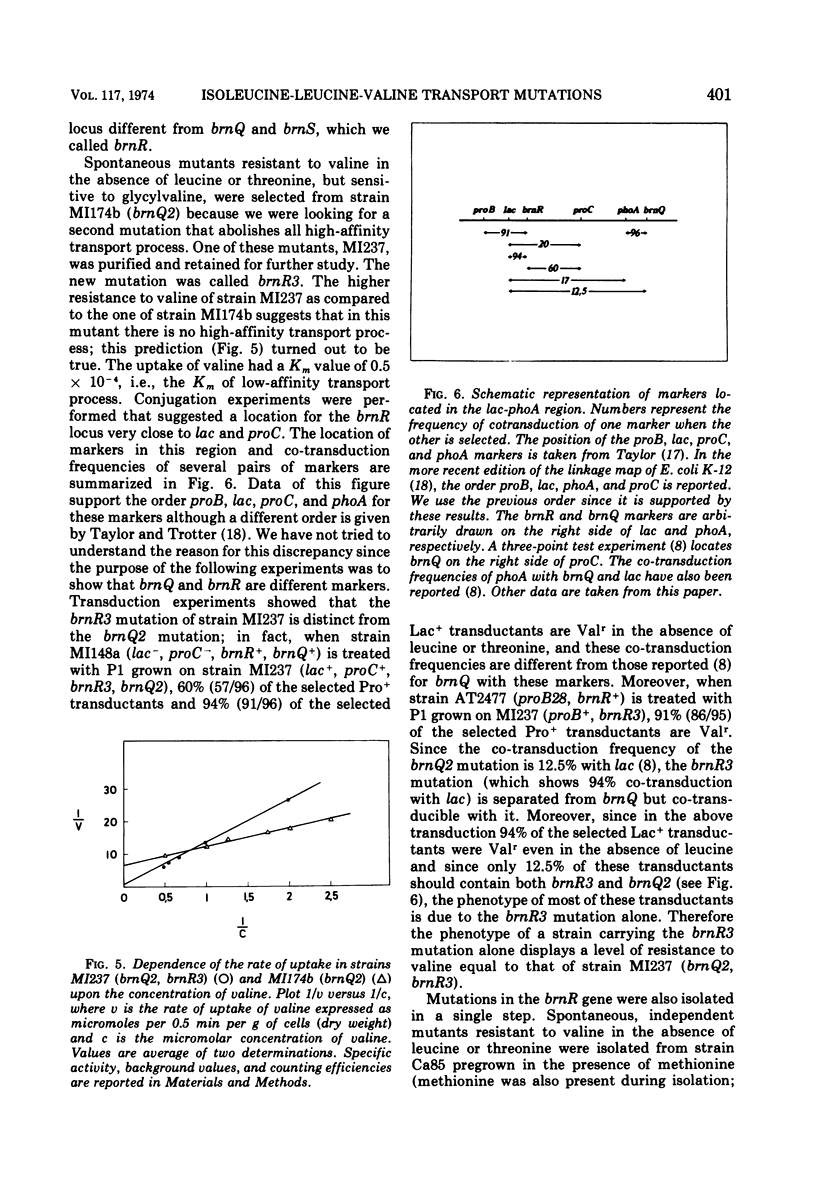
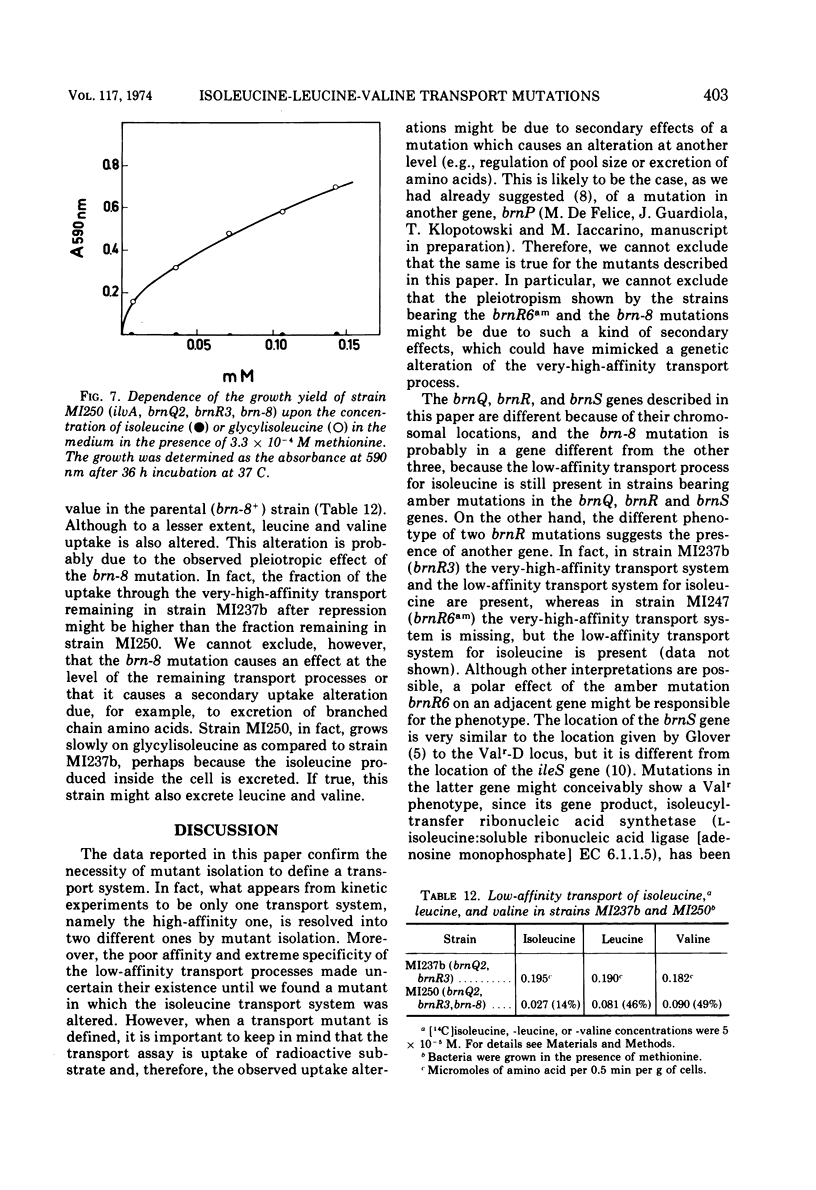
Uptake of isoleucine, leucine, and valine in Escherichia coli K-12 is due to several transport processes for which kinetic evidence has been reported elsewhere. A very-high-affinity transport process, a high-affinity transport process, and three different low-affinity transport processes were described. In this paper the existence of these transport processes is confirmed by the isolation and preliminary characterization of mutants altered in one or more of them. The very-high-affinity transport process is missing either in strains carrying the brnR6am mutation or in strains carrying the brn-8 mutation. This appears to be a pleiotropic effect since other transport systems are also missing. Mutant analysis shows that more than one transport system with high affinity is present. One of them, high-affinity 1, which needs the activity of a protein produced by the brnQ gene, transports isoleucine, leucine, and valine and is unaffected by threonine. The other, high-affinity 2, which needs the activity of a protein produced by the brnS gene, transports isoleucine, leucine, and valine; this uptake is inhibited by threonine which probably is a substrate. Another protein, produced by the brnR gene, is required for uptake through both high-affinity 1 and high-affinity 2 transport systems. The two systems therefore appear to work in parallel, brnR being a branching point. The brnQ gene is located close to phoA at 9.5 min on the chromosome of E. coli, the brnR gene is located close to lac at 9.0 min, and the brnS gene is close to pdxA at 1 min. A mutant lacking the low-affinity transport system for isoleucine was isolated from a strain in which the high-affinity system was missing because of a brnR mutation. This strain also required isoleucine for growth because of an ilvA mutation. The mutant lacking the low-affinity transport system was unable to grow on isoleucine but could grow on glycylisoleucine. This mutant had lost the low-affinity transport for isoleucine, whereas those for leucine and valine were unaffected. A pleiotropic consequence of this mutation (brn-8) was a complete absence of the very-high-affinity transport system due either to the alteration of a common gene product or to any kind of secondary interference which inhibits it. Mutants altered in isoleucine-leucine-valine transport were isolated by taking advantage of the inhibition that valine exerts on the K-12 strain of E. coli. Mutants resistant both to valine inhibition (Valr) and to glycylvaline inhibition are regulatory mutants. Valr mutants that are sensitive to glycylvaline inhibition are transport mutants. When the very-high-affinity transport process is repressed (for example by methionine) the frequency of transport mutants among Valr mutants is higher, and it is even higher if the high-affinity transport process is partially inhibited by leucine.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ames G. F., Lever J. Components of histidine transport: histidine-binding proteins and hisP protein. Proc Natl Acad Sci U S A. 1970 Aug;66(4):1096–1103. doi: 10.1073/pnas.66.4.1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andoh T., Ozeki H. Suppressor gene Su3+ of E. coli, a structural gene for tyrosine TRNA. Proc Natl Acad Sci U S A. 1968 Mar;59(3):792–799. doi: 10.1073/pnas.59.3.792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Felice M., Guardiola J., Lamberti A., Iaccarino M. Escherichia coli K-12 mutants altered in the transport systems for oligo- and dipeptides. J Bacteriol. 1973 Nov;116(2):751–756. doi: 10.1128/jb.116.2.751-756.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furlong C. E., Weiner J. H. Purification of a leucine-specific binding protein from Escherichia coli. Biochem Biophys Res Commun. 1970 Mar 27;38(6):1076–1083. doi: 10.1016/0006-291x(70)90349-9. [DOI] [PubMed] [Google Scholar]
- GORINI L., KAUFMAN H. Selecting bacterial mutants by the penicillin method. Science. 1960 Feb 26;131(3400):604–605. doi: 10.1126/science.131.3400.604. [DOI] [PubMed] [Google Scholar]
- Guardiola J., De Felice M., Klopotowski T., Iaccarino M. Multiplicity of isoleucine, leucine, and valine transport systems in Escherichia coli K-12. J Bacteriol. 1974 Feb;117(2):382–392. doi: 10.1128/jb.117.2.382-392.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guardiola J., Iaccarino M. Escherichia coli K-12 mutants altered in the transport of branched-chain amino acids. J Bacteriol. 1971 Dec;108(3):1034–1044. doi: 10.1128/jb.108.3.1034-1044.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hill C. W., Foulds J., Soll L., Berg P. Instability of a missense suppressor resulting from a duplication of genetic material. J Mol Biol. 1969 Feb 14;39(3):563–581. doi: 10.1016/0022-2836(69)90146-6. [DOI] [PubMed] [Google Scholar]
- Iaccarino M., Berg P. Isoleucine auxotrophy as a consequence of a mutationally altered isoleucyl-transfer ribonucleic acid synthetase. J Bacteriol. 1971 Feb;105(2):527–537. doi: 10.1128/jb.105.2.527-537.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LEAVITT R. I., UMBARGER H. E. Isoleucine and valine metabolism in Escherichia coli. XI. Valine inhibition of the growth of Escherichia coli strain K-12. J Bacteriol. 1962 Mar;83:624–630. doi: 10.1128/jb.83.3.624-630.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LENNOX E. S. Transduction of linked genetic characters of the host by bacteriophage P1. Virology. 1955 Jul;1(2):190–206. doi: 10.1016/0042-6822(55)90016-7. [DOI] [PubMed] [Google Scholar]
- Penrose W. R., Nichoalds G. E., Piperno J. R., Oxender D. L. Purification and properties of a leucine-binding protein from Escherichia coli. J Biol Chem. 1968 Nov 25;243(22):5921–5928. [PubMed] [Google Scholar]
- Rosner J. L. Formation, induction, and curing of bacteriophage P1 lysogens. Virology. 1972 Jun;48(3):679–689. doi: 10.1016/0042-6822(72)90152-3. [DOI] [PubMed] [Google Scholar]
- Strigini P., Gorini L. Ribosomal mutations affecting efficiency of amber suppression. J Mol Biol. 1970 Feb 14;47(3):517–530. doi: 10.1016/0022-2836(70)90319-0. [DOI] [PubMed] [Google Scholar]
- Taylor A. L. Current linkage map of Escherichia coli. Bacteriol Rev. 1970 Jun;34(2):155–175. doi: 10.1128/br.34.2.155-175.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor A. L., Trotter C. D. Linkage map of Escherichia coli strain K-12. Bacteriol Rev. 1972 Dec;36(4):504–524. doi: 10.1128/br.36.4.504-524.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Treiber G., Iaccarino M. Biochemical characterization of a mutant isoleucyl-transfer ribonucleic acid synthetase from Escherichia coli K-12. J Bacteriol. 1971 Sep;107(3):828–832. doi: 10.1128/jb.107.3.828-832.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]



