Abstract
Background
Distinct virulence factors of H. pylori have been described: the vaculating cytotoxin (vacA), the cytotoxin associated gene (cagA), the induced by contact with epithelium factor Antigen (iceA gene) and the outer membrane protein oipA. In Tunisia, there are no data regarding the pattern of H. pylori genotypes; therefore, this prospective and multicentre study was the first to be done in Tunisia and aimed to investigate the prevalence of the vacA, cagA, iceA and oipA genotypes of H. pylori isolates from Tunisian patients with peptic ulceration, gastric cancer, MALT lymphoma and gastritis.
Methods
H. pylori was cultured from endoscopic biopsies obtained from 281 Tunisian patients. The vacA alleles, cagA, iceA and oipA genotypes were determined by PCR.
Results
The vacA s1m1, s1m2 and s2m2 were respectively found in 10.7%, 12.5% and 45.6% of strains. The s2m1 genotype was not detected in our study. The cagA was found in 61.6% of isolates. The iceA1 and the iceA2 genotypes were respectively isolated in 60.2% and in 16% of strains. The oipA genotype was detected in 90.8% of strains. Considering the vacA and iceA genotypes, the presence of multiple H. pylori strains in a single biopsy specimen was found respectively in 31.4% and 23.8%. The comparison between strains isolated from antrum and fundus showed that Tunisian patients were infected with two or more strains of different cagA, vacA, iceA and oipA genotypes and the discordance was respectively in 9.6%, 4.6%, 8.9% and 8.5% of strains.
Conclusion
Our results showed that in 46% (131 strains among 281), the H. pylori strains were highly virulent in relation of the three or four virulent factors they could carry. These finding were described before in the literature. Tunisian patients were colonized by one or multiple strains of H. pylori in the same time in relation of presence of vacA m1/m2 and iceA1/iceA2 in the same biopsy. The discordance between strains isolated from antrum and fundus was high, and it is in favour of multicolonization.
Background
Helicobacter pylori (H. pylori) is one of the most common bacterial pathogens of humans and has a worldwide distribution. Infection by H. pylori is associated with the development of chronic gastritis, gastric or duodenal ulcer, gastric cancer and MALT-lymphoma [1]. Different virulence genes have been described in H. pylori infection such as cagA, vacA, iceA and oipA genes. The cytotoxin-associated gene (cagA) is frequently associated with cytotoxin production and the induction of interleukin 8 (IL8) by gastric epithelial cells [2]. Several studies have suggested that cagA is a useful marker for the most virulent strains that are associated with peptic ulcer, atrophic gastritis and adenocarcinoma [3-5]. The cag pathogenicity island (PaI) encodes a type IV secretory system and delivers CagA into the host cytosol where becomes phosphorylated on tyrosine residue. Phosphorylated CagA interacts with the phosphatase SHP-2 causing dephosphorylation of cortactin and cytoskeletal rearrangements forming the "hummingbird" phenotype [6]. The vacuolating cytotoxin gene (vacA) is an important virulence factor encoding the vacuolating toxin and it is present in all strains [7,8]. There is a considerable variation in vacuolating activities among strains [9-11] due to the sequence heterogeneity at the middle (m) and the signal (s) regions within the vacA gene. The middle region has m1 and m2 allelic types and the signal region located at the 5' end of the gene has an s1 (s1a, s1b and s1c) or an s2 allele. Strains carrying the s1ml mosaic combination of the gene vacA exhibit higher levels of cytotoxic activity than s1m2 strains, while s2m2 strains do not secrete the vacuolating cytotoxin [12]. Another virulence gene designated iceA (for induced by contact with epithelium) has been recently described. The iceA gene has two main allelic variants iceA1 and iceA2 [13] but the function of these variants is not yet clear [14-18]. iceA1 is upregulated upon contact of H. pylori with the gastric epithelium and has been regarded as a marker for peptic ulcer disease [19]. Recently, a novel putative virulence factor has been identified, the oipA (outer inflammatory protein) gene, which encodes one of the outer membrane proteins and is an inflammation-related gene located approximately 100 kb from the cag PAI on the H. pylori chromosome [20]. oipA induces IL-8 secretion by epithelial cells and active OipA protein production may be "on" or "off" depending on the number of CT dinucleotide repeats in the signal sequence of the oipA gene (HP0638) [21].
In Tunisia, there are no data regarding the pattern of H. pylori genotypes in patients; therefore, our prospective and multicentre study was the first to be done in Tunisia and to our knowledge the first one in North Africa. It aimed to investigate the prevalence of the vacA, cagA, iceA and oipA genotypes of H. pylori isolates from Tunisian patients.
Materials and methods
Materials
Biopsy samples were obtained over a 2 years-period (March 2005 to August 2007) from patients referred for endoscopy at 6 centers of gastroenterology in Tunisia. 281 patients, who had H. pylori positive cultures, were enrolled in this study. The mean age of the patients was 39.9 (range, 2 to 88) and 54.4% were women. Regarding macroscopic aspect of the mucosa and histological routine results, patients were distributed into gastritis in 195 cases (69.3%), peptic ulceration in 78 cases (27.7%) (duodenal ulcer in 67 cases, gastric ulcer in 11 cases), MALT lymphoma of the stomach in one case (0.3%) and gastric cancer in 7 cases (2.5%). One endoscopic biopsy specimen taken from the antrum and one taken from the fundus were immediately frozen and kept separately at -80°C. In addition, one biopsy specimen from the antrum and one from the fundus were used for the culture. In our study, we also obtained colonies isolated separately from two biopsy sites (antrum and fundus) to determine if one patient can be colonized with one or more strains of H. pylori. All subjects were answering to questionnaire related to age, sex, gastric or duodenal peptic ulcer diseases upon endoscopy.
Culturing of H. pylori and DNA extraction
The biopsy specimens were cut into small pieces, homogenized in a Petri dish with a sterile scalpel and were smeared on the surface of Columbia agar plates containing 10% horse blood and Skirrow (Oxoid, England) supplement. Incubation was performed in microaerophilic conditions at 37°C for a maximum of 6 days. The H. pylori colonies were smooth, translucent and small (2 mm of diameter). In our study, the antral and fundic biopsy specimens were studied separately. Colonies that exhibited the described characteristic morphologies were identified as H. pylori if they were Gram negative and shaped bacilli, and urease, catalase and oxidase positive. From the primary growth, seven or eight colonies were pooled together, and genomic DNA was extracted with the QIAamp DNA mini kit (Qiagen, Germany) according to the manufacturer's instructions. The isolated DNA was eluted in 200 μl of 1× TE buffer (10 mM Tris-HCl, 1 mM EDTA [pH 8.0]) and stored at -20°C until use.
H. pylori genotyping
After DNA extraction, polymerase chain reactions (PCR) were performed in a volume of 50 μl containing 1 μM of each primers, 1 μL of genomic DNA (approximately 200 ng), 1 mM of dNTPs mix (invitrogen), 2 mM of Mgcl2, and 0.05 U/μL Taq DNA polymerase (invitrogen). PCR amplifications were performed in an automated thermal cycler (Biometra Co., Germany). Table 1 summarized the primer sequences and the expected size of PCR products. The following cycle conditions were used: for vacA: 35 cycles of 1 min at 94°C, 1 min at 53°C, and 1 min at 72°C; for cagA: 1 min at 94°C, 1 min at 56°C, and 1 min at 72°C; for iceA: 1 min at 94°C, 1 min at 56°C, and 1 min at 72°C, and for oipA: 1 min at 94°C, 1 min at 56°C and 1 min at 72°C. All runs included one negative DNA control consisting of PCR-grade water and two or more positive controls (26695, J99, SS1, Tx30, 88-23 and 84-183). The amplified PCR products were resolved in 1.5% agarose gels stained with ethidium bromide and visualized under a short wave length ultraviolet light source.
Table 1.
PCR primers for amplification of cagA, vacA, iceA and oipA sequences
| Region | Primer | Primer sequence (5'-3') | Size of PCR product (bp) | References |
|---|---|---|---|---|
| cagA | F1 | GATAACAGCCAAGCTTTTGAGG | 349 | [36] |
| B1 | CTGCAAAAGATTGTTTGGCAGA | |||
| vacA | ||||
| m1 | VA3-F | GGTCAAAATGCGGTCATGG | 290 | |
| VA3-R | CCATTGGTACCTGTAGAAAC | |||
| m2 | VA4-F | GGAGCCCCAGGAAACATTG | 352 | |
| VA4-R | CATAACTAGCGCCTTGCAC | |||
| s1/s2 | VA1-F | ATGGAAATACAACAAACACAC | 259/286 | [12] |
| VA1-R | CTGCTTGAATGCGCCAAAC | |||
| s1b | SS3-R | AGCGCCATACCGCAAGAG | 187 | |
| VA1-R | CTGCTTGAATGCGCCAAAC | |||
| s1a | SS1-R | GTCAGCATCACACCGCAAC | 190 | |
| VA1-R | CTGCTTGAATGCGCCAAAC | |||
| s2 | SS2-R | GCTAACACGCCAAATGATGC | 199 | |
| VA1-R | CTGCTTGAATGCGCCAAAC | |||
| iceA1 | iceA1-F | GTGTTTTTAACCAAAGTATC | 247 | [37] |
| iceA1-R | CTATAGCCASTYTCTTTGCA | |||
| iceA2 | iceA2-F | GTTGGGTATATCACAATTTAT | 229/334 | [37] |
| iceA2-R | TTRCCCTATTTTCTAGTAGGT | |||
| oipA | HPO638F | GTTTTTGATGCATGGGATTT | 401 | [38] |
| HPO638R | GTGCATCTCTTATGGCTTT |
Statistical analysis
Data were analyzed using X2 test. A p value of < 0.05 was considered to be statistically significant.
Results
cagA genotyping
The 349-bp PCR product indicating the presence of the cagA gene was obtained with 173 isolates (61.6%) and 108 (38.4%) were negative. We showed cagA genotype discordance between antrum and fundus in 27 isolates (9.6%) (Table 2).
Table 2.
Prevalence of H. pylori genotypes detected in strains
| Genotype | Prevalence |
|---|---|
| cagA | |
| cagA + | 173 (61.6%) |
| cagA - | 108 (38.4%) |
| d(A/F)* | 27(9.6%) |
| vacA | |
| m1s1a | 5 (1.8%) |
| m1s1b | 25 (8.9%) |
| m2s1a | 10 (3.5%) |
| m2s1b | 25 (8.9%) |
| m2s2 | 128 (45.6%) |
| m1m2 | 88 (31.4%) |
| m1s2 | 0 (0%) |
| d(A/F)* | 13 (4.6%) |
| iceA | |
| iceA1 | 169 (60.2%) |
| iceA2 | 45(16%) |
| iceA1/iceA2 | 67(23.8%) |
| d(A/F)* | 25(8.9%) |
| oipA | |
| oipA + | 255 (90.8%) |
| oipA - | 26 (9.2%) |
| d(A/F)* | 24(8.5%) |
d(A/F)*: discordance between strains isolated from the antrum and the fundus in the same patient
vacA genotyping
The vacA s- and m- region genotype were determined in all strains studied. In the m-region, 88 strains contained both m1 and m2 alleles. In the strains containing one single vacA m allele, the m1 allele was found in 30 isolates (10.7%) and m2 in 163 one (58%).
For the s-region, in strains where a single vacA s allele was found, the majority 128 (45.6%) contained the s2 allele. In 65 isolates contained s1 allele (23.2%), 50 (77%) were subtype s1b while only 15 (23%) were subtype s1a.
Considering strains with only one single vacA genotype, and taking vacA s- and m-region together, three genotypes were found: s1/m1 (10.7%), s1/m2 (12.5%) and s2m2 (45.6%). The s2m1 genotype was not found in our study.
The discordance between strains taken from antrum and those from fundus was found in 13 cases (4.6%) (Table 2).
iceA genotyping
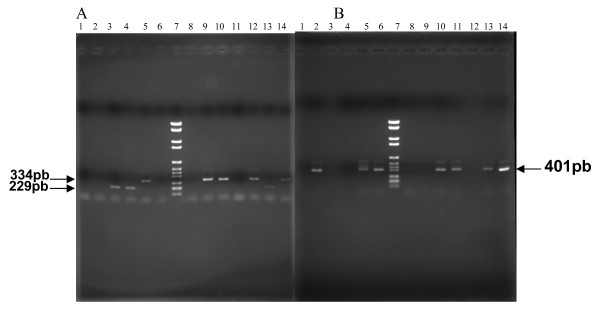
Overall, iceA1 was detected in 169 strains (60.2%) of all 281 isolates and iceA2 was found in 45 strains (16%). In the present study, the iceA2 amplification yielded both the 229 bp and 334 bp fragments (figure 1), this difference in the fragment size is due to the presence of a 105 bp in - frame amplicon present in the 334 bp fragment that is absent in the 229 bp fragment [22]. Sixty-seven strains (23.8%) were positive for both iceA1 and iceA2, and discordance between isolates from antrum and fundus was seen in 25 strains (8.9%) (Table 2).
Figure 1.
Genotyping of iceA2 and oipA genes by PCR. A: example of iceA2 genotyping. Lane 1, negative control without DNA, Lanes 2, 6, 8 and11, iceA2 negative H. pylori strain, Lanes 3, 4 and 13, iceA2 positive H. pylori strain (229-bp), Lanes, 5, 9, 10, 12 and 14, iceA2 positive strain (334-bp), Lane 7,100-bp DNA marker. B: example of oipA genotyping. Lane 1, negative control without DNA, Lanes 2, 5, 6, 10, 11, 13 and 14, oipA positive strains, Lane 7, 100-bp DNA marker, Lanes 3, 4, 8, 9 and 12, oipA negative strains.
oipA genotyping
The oipA genotype was detected in 255 (90.8%) from 281 strains, and only 26 strains (9.2%) were oipA negative. The discordance between strains obtained from antrum and fundus was determined in 24 strains (8.5%) (Table 2). The 401-bp PCR product indicating the presence of the oipA gene was shown in figure 1.
Combined cagA, vacA, iceA and oipA genotypes
Based on analysis of the cagA gene (positive and negative), the vacA s-region (s1 and s2), the iceA allelic type (iceA1 and iceA2) and the oipA gene (positive and negative), ten different genotypic combinations were recognized (Table 3). The prevalence of each of these genotypes among the 131 strains with a single combined genotype was shown in table 3. The most prevalent genotypes were s2/cagA+/iceA1/oipA+ (27.5%), s2/cagA-/iceA1/oipA+ (23.6%), s2/cagA+/iceA2/oipA+ (11.4%) and s1/cagA+/iceA1/oipA+ (9.9%). Among the strains isolated from 24 ulcer patients (gastric and duodenal ulcer) with a single genotype, 8 strains (33.3%) contained the vacA s1/cagA+/iceA1 genotype while only one (4.1%) contained vacA s1/cagA-/iceA2. If we consider only vacA and cagA genotypes, 14 strains (58.3%) isolated from 24 ulcer patients had genotype vacA s1/cagA+, and none (0%) had vacA s2/cagA-. If we consider only vacA and iceA, 18 strains (75%) contained genotype vacA s1/iceA1, and none (0%) had vacA s2/iceA2. When we take only vacA and oipA genotypes, 21 strains (87.5%) had genotype vacA s1/oipA+ and none (0%) had vacA s2/oipA-. Comparing with strains isolated from patients having gastritis, we found that the prevalence of genotypes vacA s1/cagA+, vacA s1/iceA1 and vacA s1/oipA+ was 11.2%, 10.3%, and 16.8%, respectively. The genotype vacA s1/cagA+/iceA1/oipA+ was found in 13 (9.9%) of 131 isolates, and 8 (61.5%) of these are associated with a clinical diagnosis of peptic ulcer disease, whereas genotypes harbouring the s2 allele of vacA gene were predominant in the strains isolated from patients with gastritis.
Table 3.
Combined vacA, cagA, iceA and oipA genotypes
| Combination | DU* | GU* | G* | Total |
|---|---|---|---|---|
| s1/cagA+/iceA1/oipA+ | 4(20%) | 4(100%) | 7(6.5%) | 13(9.9%) |
| s1/cagA+/iceA2/oipA+ | 4(20%) | 0(0%) | 4(3.7%) | 8(6.1%) |
| s1/cagA-/iceA1/oipA+ | 8(40%) | 0(0%) | 3(2.8%) | 11(8.4%) |
| s1/cagA-/iceA2/oipA+ | 1(5%) | 0(0%) | 4(3.7%) | 5(3.8%) |
| s1/cagA+/iceA1/oipA- | 2(10%) | 0(0%) | 1(0.9%) | 3(2.3%) |
| s2/cagA+/iceA1/oipA+ | 1(5%) | 0(0%) | 35(32.7%) | 36(27.5%) |
| s2/cagA+/iceA2/oipA+ | 0(0%) | 0(0%) | 15(14%) | 15(11.4%) |
| s2/cagA-/iceA1/oipA+ | 0(0%) | 0(0%) | 31(29%) | 31(23.6%) |
| s2/cagA-/iceA2/oipA+ | 0(0%) | 0(0%) | 4(3.7%) | 4(3%) |
| s2/cagA-/iceA2/oipA- | 0(0%) | 0(0%) | 5(4.6%) | 5(3.8%) |
| Total | 20 | 4 | 107 | 131 |
DU*: duodenal ulcer; GU*: gastric ulcer; G*: gastritis
In our study, first, we have studied the relationship between virulence genes (vacA, cagA, iceA, oipA) and the different gastroduodenal diseases, between the two groups (patients with peptic ulceration/patients with gastritis). When considering the vacA and oipA genes, the difference was statistically significant between the two groups (p < 0.001), but, when considering the cagA and iceA genes, this relation is not statistically significant (p > 0.05) (Tables 4). Second, we were interesting in studying the relationship between: a/the age of patients and virulence factors (Table 5) and b/the sex and virulence factors (Table 6). Our study showed that, among age, the difference was statistically significant between adults and children with the cagA and iceA genes (p < 0.05), but not significant when considering the vacA and oipA genes. When about sex, we have demonstrated that the difference between men and women was statistically significant among vacA, oipA and iceA genes (p < 0.05), which was not statistically significant with the cagA gene.
Table 4.
cagA, vacA, iceA and oipA status of H. pylori strains with different gastric diseases: a comparison between patients with peptic ulceration, and with gastritis
| PU* | G* | p | |
|---|---|---|---|
| cagA+ | 53 (67.9%) | 114 (58.4%) | > 0.05 |
| oipA+ | 63 (80.7%) | 186 (95.3%) | < 0.001 |
| iceA1 | 51 (65.3%) | 113 (57.9%) | > 0.05 |
| iceA2 | 12 (15.3%) | 33 (16.9%) | |
| iceA1/A2 | 15 (19.2%) | 49 (25.1%) | |
| m1s1 | 12 (15.3%) | 18 (9.2%) | < 0.001 |
| m2s1 | 17 (21.8%) | 17 (8.7%) | |
| m1m2 | 49 (62.8%) | 32 (16.4%) | |
| m2s2 | 0 (0%) | 128 (65.6%) |
PU*: peptic ulceration; G*: gastritis
Table 5.
Relation between the age of patients and virulence factors:
| Adults | Children | p | |
|---|---|---|---|
| cagA + | 155 | 18 | < 0.001 |
| oipA+ | 209 | 46 | > 0.05 |
| iceA1 | 137 | 32 | 0.05 |
| iceA2 | 43 | 2 | |
| iceA1/iceA2 | 53 | 14 | |
| m1s1 | 26 | 4 | > 0.05 |
| m2s1 | 31 | 4 | |
| m1m2 | 72 | 16 | |
| m2s2 | 104 | 24 |
Table 6.
Relation between the sex of patients and virulence factors:
| Male | Female | p | |
|---|---|---|---|
| cagA + | 84 | 89 | > 0.05 |
| oipA+ | 111 | 144 | < 0.05 |
| iceA1 | 78 | 91 | |
| iceA2 | 17 | 28 | 0.05 |
| iceA1/iceA2 | 40 | 27 | |
| m1s1 | 19 | 11 | |
| m2s1 | 19 | 16 | < 0.001 |
| m1/m2 | 49 | 39 | |
| m2s2 | 40 | 88 |
Discussion
The predominant genotype in strains that were positive for H. pylori by PCR was the oipA (90.8%), followed by the cagA gene (61.6%), and the iceA1 gene (60.2%), while the iceA2 was amplified only in 45 strains (16%). Our results were in agreement with other studies conducted in Europe, Central and South America, and East Asia where a higher prevalence (67% or more) of the cagA genotype was reported [22]. In Turkey [23], Korea and Japan, the prevalence of the iceA1 genotype in patient with duodenal ulcer were respectively 68.8%, 69.8% and 62.5% which is similar to our study, whereas iceA2 was prevalent in the USA and Columbia [24]. The already mentioned similitude can be defined by the epidemiological resemblance in these countries.
For the vacA genotype, and when considering a single combined genotype, our results showed that the vacA s2 allele was predominant (45.6%) followed by the vacA s1 allele (23.2%). A study in Kuwait reported that vacA s1 and s2 types were detected in approximately equal numbers in biopsies obtained from patients of Middle-Eastern origin, while North Africans were predominantly infected with the s2 type [25].
The prevalence of cagA+ strains in Tunisian peptic ulcer and those with gastritis was similar to that shown in the study from South Africa [26].
In a previous report by van Doorn et al (19), an association was found between the iceA1 allele and peptic ulcer disease. Other studies from Asia were suggesting that vacA, cagA and iceA genotypes were not associated with peptic ulcer disease [27]. These findings may reflect important geographic differences between H. pylori strains and patients. As reported by van Doorn et al, H. pylori genotypes are not uniformly distributed over the world [22]. When considering the vacA and oipA genes, our results showed that there is a significant difference between the two studied groups (p < 0.001), but, cagA and iceA genes were not associated with gastroduodenal diseases in our study (p > 0.05). The oipA (HpO638) outer membrane protein expression was linked to severe inflammation and the induction of IL-8 secretion. In one Turkish study, oipA (HpO638) gene was highly associated with peptic ulcer disease (92.9%) than with gastritis (80.9%) [28]. In other studies, no difference in prevalence of cagA genotypes was found between peptic ulcers and other gastroduodenal diseases [29]. Yamaoka et al [24] reported that there was no association between the iceA, vacA or cagA status and clinical outcome in studied patients.
In our study, the oipA gene was found in 100% of gastric ulcer. Several studies failed to show a relationship between the gene status and clinical symptoms in several patient populations, this might be due to that patient selection is extremely important and the study group should be sufficiently large and diverse with respect to genotypes and clinical symptoms.
As for age, we have demonstrated that the difference between children and adults was statistically significant about the cagA and iceA genes, but not with the vacA and oipA genes. Regarding the distribution of gene virulent factors according to sex, no difference seen in term of cagA gene, but the difference was statistically significant among the vacA, iceA and oipA genes. In other reported studies, there were no significant differences in frequencies of H. pylori virulence-associated genes, between children and adults, and the strains from men exhibited genotypes similar to those in women [30]. Laila FN et al [31] have concluded in their study that the presence of certain genotypes was not significantly associated with the age or gender of the patient.
The detection of multiple genotypes implies the presence of multiple strains in a clinical sample. Considering vacA and iceA genes in our study, the presence of multiple H. pylori strains in a single biopsy specimen was found respectively in 31.4% and 23.8%. Then, when comparing the genetic differences in isolates of H. pylori from the antrum and the fundus, we determined that patients were infected with two or more strains of different cagA, vacA, iceA and oipA, and the percentage of discordance was respectively in 9.6%, 4.6%, 8.9% and 8.5%. Consequently, if multiple genotypes are found, this is a strong indication for the presence of multiple strains. It may be speculated that more than one strain may be acquired in childhood, especially in countries with a very high prevalence of H. pylori, but, it is not known whether multiple strains colonize simultaneously (coinfection) or at different time points (superinfection), and several studies demonstrated that coinfection or superinfection are common [32,33]. The co-existence of more than one strain in the same individual may reflect the capacity of H. pylori to evolve genetic variations during the long-term colonization from childhood [34], and the dynamics of co-colonization by multiple strains has been studied in animal models [35]. Since the prevalence of multiple strains colonization is clinically important in our study, there is a clear need to be considered when planning therapeutic strategies to avoid the risk of the emergence of such strains.
The present study showed that patients with peptic ulcer disease and those with gastritis were almost infected similarly by multiple strains of H. pylori. This underlines that the positive associations between H. pylori genotypes and peptic ulcer disease do not imply that patients without ulcer disease cannot be infected with high-risk H. pylori genotypes.
The combination of the distinct vacA, cagA, iceA and oipA genotypes illustrated the mosaic composition of the H. pylori genome. Strains typed as vacA s2/cagA+/iceA+/oipA+ were more prevalent than those typed as vacA s1/cagA+/iceA+/oipA+ and which are associated to severe pathologies.
In conclusion, we have examined the prevalence of vacA, cagA, iceA and oipA genotypes of H. pylori strains clinically isolated in Tunisia. No significant correlation was found between the expression of cagA and iceA genes and the two groups of studied patients, but the difference was statistically significant with the vacA and oipA genes. As for age, the difference between children and adults was statistically significant with the cagA and iceA genes, but not with oipA and vacA genes. Regarding the distribution of virulence genes to sex, the difference was statistically significant among oipA, iceA and vacA genes, but not with cagA gene.
In Tunisia, it is so possible that the high prevalence of infection with virulent factors contributes to the characteristics of H. pylori infection, but not used to discern the risk of developing serious gastroduodenal diseases in the host. Our results showed that Tunisian patients were colonized by one or more strains of H. pylori in relation of presence of vacA m1/m2 and iceA1/iceA2 in the same biopsy, than, the discordance between strains isolated from antrum and fundus was higher, and it is in favour of multicolonization.
Competing interests
The authors declare that they have no competing interests.
This prospective multicentre study was performed in Microbiology laboratory-Rabta University Hospital, Tunis, Tunisia in research unit UR04SP08.
Authors' contributions
BMK was responsible for the collection of biopsies, H. pylori conventional culture, had carried out the molecular genetic studies, performed the statistical analysis, drafted the manuscript and participated in the design of the study. ZM and MA were participated in the design of the study. FA, BMN, NT, MA and ST were helped to the constitution of H. pylori strains collection. FC had conceived of the study, participated in its design and coordination and helped to draft the manuscript. Finally, BC had helped to molecular genetic studies and to draft the manuscript.
All authors have read and approved the final manuscript.
Contributor Information
Khansa Ben Mansour, Email: bmkhansa@gmail.com.
Chédlia Fendri, Email: fendri.chedlia@planet.tn.
Meriem Zribi, Email: m.zribi@tunet.tn.
Afef Masmoudi, Email: afefmasmoudi@planet.tn.
Mounir Labbene, Email: labbenem@yahoo.fr.
Azza Fillali, Email: filaliza2@gmail.com.
Nabil Ben Mami, Email: nabyl.benmami@rns.tn.
Taoufik Najjar, Email: taoufik.najjar@rns.tn.
Ahmed Meherzi, Email: ahmed.meherzi.@rns.tn.
Tahar Sfar, Email: sfar.medtahar@planet.tn.
Christophe Burucoa, Email: c.burucoa@chu-poitiers.fr.
References
- Blaser MJ. Ecology of Helicobacter pylori in the human stomach. J Clin Invest. 1997;100:759–762. doi: 10.1172/JCI119588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenks PJ, Mégraud F, Labigne A. Clinical outcome after infection with Helicobacter pylori does not appear to be reliably predicted by the presence of any of the genes of the cag pathogenicity island. Gut. 1998;43:752–758. doi: 10.1136/gut.43.6.752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figura N, Guglielmetti P, Rossolini A. Cytotoxin production by Campylobacter pylori strains isolated from patients with peptic ulcers and from patients with chronic gastritis only. J Clin Microbiol. 1989;27:225–226. doi: 10.1128/jcm.27.1.225-226.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crabtree JE, Figura N, Taylor JD. Expression of 120 kilodalton protein and cytotoxicity in Helicobacter pylori. J Clin Pathol. 1992;45:733–734. doi: 10.1136/jcp.45.8.733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaser MJ, Perez-Perez GI, Cover TL. Infection with Helicobacter pylori strains possessing CagA is associated with an increased risk of developing adenocarcinoma of stomach. Cancer Res. 1995;55:2111–2115. [PubMed] [Google Scholar]
- Segal ED, Falkow S, Tompkins LS. Helicobacter pylori attachment to gastric cells induces cytoskeletal rearrangements and tyrosine phosphorylation of host cell proteins. Proc Natl Acad Sci USA. 1996;93:1259–1264. doi: 10.1073/pnas.93.3.1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atherton JC. The clinical relevance of strain types of Helicobacter pylori. Gut. 1997;40:701–704. doi: 10.1136/gut.40.6.701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cover TL. The vacuolating cytotoxin of Helicobacter pylori. Mol Microbiol. 1996;20:241–246. doi: 10.1111/j.1365-2958.1996.tb02612.x. [DOI] [PubMed] [Google Scholar]
- Cover TL, Blaser MJ. Purification and characterization of the vacuolating toxin from Helicobacter pylori. J Biol Chem. 1992;267:10570–10575. [PubMed] [Google Scholar]
- de Bernard M, Arico B, Panini E, Rizzuto R, Grandi G, Rappuoli R, Montecucco C. Helicobacter pylori toxin VacA induces vacuole formation by acting in the cell cytosol. Mol Microbiol. 1997;26:665–674. doi: 10.1046/j.1365-2958.1997.5881952.x. [DOI] [PubMed] [Google Scholar]
- Leunk RD. Production of a cytotoxin by Helicobacter pylori. Rev Infect Dis. 1991;13(Suppl.8):S686–S689. doi: 10.1093/clinids/13.supplement_8.s686. [DOI] [PubMed] [Google Scholar]
- Atherton J, Cao P, Peek R, Tumurru M, Blaser M, Cover TL. Mosaicism in vacuolating cytotoxin alleles of Helicobacter pylori. Association of peptic vacA types with cytotoxin production and peptic ulceration. J Biol Chim. 1995;270:17771–17777. doi: 10.1074/jbc.270.30.17771. [DOI] [PubMed] [Google Scholar]
- Peek RM, Thompson SA, Atherton JC, Blaser MG, Miller GG. Expression of a novel ulcer-associated H. pylori gene, iceA, following adherence to gastric epithelial cells. Gastroenterology. 1996;110(Suppl):A225. [PubMed] [Google Scholar]
- Forsyth MH, Atherton JC, Blaser MJ, Cover TL. Heterogeneity in levels of vacuolating cytotoxin gene (vacA)transcriptionamong Helicobacter pylori strains. Infect Immun. 1998;66:3088–3094. doi: 10.1128/iai.66.7.3088-3094.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox JG, Dewhirst FE, Fraser GJ, Paster BJ, Shames B, Murphy JC. Intracellular Campylobacter-like organism from ferrets and hamsters with proliferative bowel disease is a Desulfovibrio sp. J Clin Microbiol. 1994;32:1229–1237. doi: 10.1128/jcm.32.5.1229-1237.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Go MF, Cissell L, Graham DY. Failure to confirm association of vacA gene mosaicism with duodenal ulcer disease. Scand J Gastroenterol. 1998;33:132–136. doi: 10.1080/00365529850166842. [DOI] [PubMed] [Google Scholar]
- Gzyl A, Berg DE, Dzierzanowska D. Epidemiology of cagA/vacA genes in H. pylori isolated from children and adults in Poland. J Physiol Pharmacol. 1997;48:333–343. [PubMed] [Google Scholar]
- Jorgensen M, Daskalopoulos G, Warburton V, Mitchell HM, Hazell SL. Multiple strain colonization and metronidazole resistance in Helicobacter pylori-infected patients: identification from sequential and multiple biopsy specimens. J Infect Dis. 1996;174:631–635. doi: 10.1093/infdis/174.3.631. [DOI] [PubMed] [Google Scholar]
- van Doorn LJ, Figueiredo C, Sanna R. Clinical relevance of the cagA, vacA, and iceA status of Helicobacter pylori. Gastroenterology. 1998;115:58–66. doi: 10.1016/S0016-5085(98)70365-8. [DOI] [PubMed] [Google Scholar]
- Yamaoka Y, Kwon DH, Graham DY. A M(r) 34,000 proinflammatory outer membrane protein (oipA) of Helicobacter pylori. Proc Natl Acad Sci USA. 2000;97:7533–7538. doi: 10.1073/pnas.130079797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farhana K, Abid MH, Irshad A, Sriramula S, Manjulata DS, Ahmed AM, Rajender K, Aleem AK, Leonardo AS, Niyaz A. Comparative genomics of Helicobacter pylori isolates recovered from ulcer disease patients in England. BMC Microbiol. 2005;5:32–42. doi: 10.1186/1471-2180-5-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Doorn LJ, Figueiredo C, Mégraud F, Pena S, Midolo P, Queroz DM, Carnero F, Pegado MD, Sanna R. Geographic distribution of vacA allelic types of Helicobacter pylori. Gastroenterology. 1999;116:823–830. doi: 10.1016/S0016-5085(99)70065-X. [DOI] [PubMed] [Google Scholar]
- Vildan C, Mustafa Y, Nadir Y, Sevil Z, Nedim K, Ilknur K, Huseyin B. H pylori iceA alleles are disease-specific virulence factors. World J Gastroenterol. 2007;13:2581–2585. doi: 10.3748/wjg.v13.i18.2581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaoka Y, Kodama T, Gutierrez O, Kim JG, Kashima K, Graham DY. Relationship between Helicobacter pylori iceA, cagA and vacA status and clinical outcome: studies in four different countries. J Clin Microbiol. 1999;37:2274–2279. doi: 10.1128/jcm.37.7.2274-2279.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al Qabandi A, Mustafa AS, Siddique I, Khajah AK, Madda JP. Distribution of vacA and cagA genotypes of Helicobacter pylori in Kuwait. Acta Trop. 2005;93:283–288. doi: 10.1016/j.actatropica.2005.01.004. [DOI] [PubMed] [Google Scholar]
- Kidd M, Lastovica AJ, Atherton JC, Louw JA. Heterogeneity in the Helicobacter pylori vacA and cagA genes: association with gastroduodenal disease in South Africa? Gut. 1999;45:499–502. doi: 10.1136/gut.45.4.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng PY, Hua J, Yeoh KG, Ho B. Association of peptic ulcer with increased expression of Lewis antigens but not cagA, iceA, and vacA in Helicobacter pylori isolates in an Asian population. Gut. 2000;47:18–22. doi: 10.1136/gut.47.1.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salih BA, Abasiyanik MF, Ahmed N. A preliminary study on the genetic profile of cag pathogenicity-island and other virulent gene loci of Helicobacter pylori strains from Turkey. Infect Genet Evol. 2007;7:509–512. doi: 10.1016/j.meegid.2007.03.002. [DOI] [PubMed] [Google Scholar]
- Anderson H, Loivukene K, Sillakivi T, Maaroos HI, Ustav M, Peetsalu A, Mikelsaar M. Association of cagA and vacA genotypes of Helicobacter pylori with gastric diseases in Estonia. J Clin Microbiol. 2002;40:298–300. doi: 10.1128/JCM.40.1.298-300.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyudmila B, Rumiana M, Daniel Y, Maya M, Katusha I, Stephan P, Galina G, Ivan M. High prevalence of virulent Helicobacter pylori strains is symptomatic Bulgarian patients. Diag Microbiol and Infect Dis. 2009;64:374–380. doi: 10.1016/j.diagmicrobio.2009.04.005. [DOI] [PubMed] [Google Scholar]
- Laila FN, Ismail M, Kamal BH, Marwa I. Helicobacter pylori genotypes identified in gastric biopsy specimens from Jordanian patients. BMC Gastroenterol. 2006;6:1–6. doi: 10.1186/1471-230X-6-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Doorn LJ, Figueiredo C, Rossau R, Jannes G, van Asbroek M, Sousa JC, Carneiro F, Quint WG. Typing of Helicobacter pylori vacA gene and detection of cagA gene by PCR and reverse hybridization. J Clin Microbiol. 1998;36:1271–1276. doi: 10.1128/jcm.36.5.1271-1276.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Valencia G, Atherton JC, Munoz O, Dehesa M, la Garza AM, Torres J. Helicobacter pylori vacA and cagA genotypes in Mexican adults and children. J Infect Dis. 2000;182:1450–1454. doi: 10.1086/315864. [DOI] [PubMed] [Google Scholar]
- Kersulyte D, Chalkauskas H, Berg DE. Emergence of recombinant strains of Helicobacter pylori during human infection. Mol Microbiol. 1999;31:31–43. doi: 10.1046/j.1365-2958.1999.01140.x. [DOI] [PubMed] [Google Scholar]
- Dubois A, Berg DE, Incecik ET, Fiala N, Heman-Ackah LM, Del Valle J, Yang M, Wirth HP, Perez-Perez GI, Blaser MJ. Host specificity of Helicobacter pylori strains and host responses in experimentally challenged nonhuman primates. Gastroenterology. 1999;116:90–96. doi: 10.1016/S0016-5085(99)70232-5. [DOI] [PubMed] [Google Scholar]
- Tumurru MK, Cover TL, Blaser MJ. Cloning and expression of a high-molecular mass major antigen of Helicobacter pylori: evidence of linkage to cytotoxin production. Infect Immun. 1993;61:1799–1809. doi: 10.1128/iai.61.5.1799-1809.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peek RM, Thompson SA, Donahue JP, Tham KT, Atherton JC, Blaser MJ, Miller GG. Adherence to gastric epithelial cells induces expression of a Helicobacter pylori gene, iceA, that is associated with clinical outcome. Proc Assoc Am Physicians. 1998;110:531–544. [PubMed] [Google Scholar]
- Versalovic J, Koeuth T, Lupski JR. Distribution of repetitive DNA sequences in Eubacteria and application to fingerprinting of bacterial genomes. Nucleic Acids Res. 1991;19:6823–6831. doi: 10.1093/nar/19.24.6823. [DOI] [PMC free article] [PubMed] [Google Scholar]