Abstract
Recent epidemiological evidence points to a link between a variety of negative health outcomes (e.g. metabolic syndrome, diabetes and cardiovascular disease) and the consumption of both calorically sweetened beverages and beverages sweetened with high-intensity, non-caloric sweeteners. Research on the possibility that non-nutritive sweeteners promote food intake, body weight gain, and metabolic disorders has been hindered by the lack of a physiologically-relevant model that describes the mechanistic basis for these outcomes. We have suggested that based on Pavlovian conditioning principles, consumption of non-nutritive sweeteners could result in sweet tastes no longer serving as consistent predictors of nutritive postingestive consequences. This dissociation between the sweet taste cues and the caloric consequences could lead to a decrease in the ability of sweet tastes to evoke physiological responses that serve to regulate energy balance. Using a rodent model, we have found that intake of foods or fluids containing non-nutritive sweeteners was accompanied by increased food intake, body weight gain, accumulation of body fat, and weaker caloric compensation, compared to consumption of foods and fluids containing glucose. Our research also provided evidence consistent with the hypothesis that these effects of consuming saccharin may be associated with a decrement in the ability of sweet taste to evoke thermic responses, and perhaps other physiological, cephalic phase, reflexes that are thought to help maintain energy balance.
A variety of recent epidemiological evidence points to a link between the consumption of sweetened beverages and incidence of obesity and other disorders associated with positive energy balance. For example, although not all studies have agreed, in recent years a number of reports have revealed that intake of sugar-sweetened beverages (SSBs; primarily soda and juices) is strongly and positively correlated with elevated body weight and also with increased incidence of the metabolic syndrome which includes Type II diabetes, hypertension, and cardiovascular disease 1-5; see also Hu, this volume.
Because SSBs are calorically-dense, it may not be surprising that increased consumption is associated with positive energy balance. Based on this fact, one approach to combating the current obesity crisis is to reduce access to SSBs. In the U.S., a much publicized application of this strategy has been a program to remove SSBs from primary and secondary school vending machines, replacing them with diet sodas and other non-nutritively sweetened or unsweetened beverages (see, for example, Storey, this volume). However, some question whether or not this program can reduce SSB intake enough to have a significant impact on childhood, and subsequently, adult obesity (e.g. Popkin, this volume).
As a means of weight control, the substitution of non-nutritively sweetened beverages for SSBs may also be questioned on other grounds. Several studies that have linked intake of SSBs to increased incidence of obesity and the metabolic syndrome also report a similar, and sometimes stronger, relationship when intake of diet soda is correlated with the same weight and health outcomes. For example, using both cross-sectional and longitudinal data from the Framingham Heart Study, Dhingra et al.2 reported positive relationships between consumption of diet soda and the prevalence of the metabolic syndrome that were larger than that obtained with consumption of regular soda. Similar results have also been reported as part of independent studies by Lutsey et al. 6 and Nettleton et al. 7. Furthermore, Fowler et al. 8 reported that for humans who were normal weight or nonobese (BMI < 30) at baseline, intake of >21 non-nutritively sweetened beverages per week (diet sodas and artificially sweetened coffee and tea) was associated with about double the risk of obesity compared to nonusers at follow-up seven to eight years later.
Of course, these epidemiological data are limited with respect to what they can say about how intake of non-nutritive sweeteners may be related causally to obesity and the metabolic syndrome. The mere finding that consumption of non-nutritively sweetened foods and beverages is positively correlated with weight gain is necessary, but clearly not sufficient, to confirm the hypothesis that the use of non-nutritive sweeteners causes increased weight gain. Nor can such information differentiate this interpretation from the alternative view that weight gain is the impetus for increased use of non-nutritive sweeteners. Indeed, there may be no causal relationship in either direction (i.e., other factors could cause both increased use of non-nutritive sweeteners and increased body weight gain). Furthermore, even if body weight gain does cause increased use of non-nutritive sweeteners, the question remains whether consuming these substances helps or hinders weight management in humans9) The available data relevant to this question are controversial 10, and the debate is sometimes heated. For example, holding everything else constant, substituting non-nutritive for high-calorie sweeteners obviously reduces the energy content of food and beverages. But some data from humans suggest that these kinds of substitutions may not reduce, and may even increase, total energy intake by stimulating appetite 11.
Research on the possibility that non-nutritive sweeteners promote food intake, body weight gain, and metabolic disorders has been hindered by the lack of a physiologically-relevant model that describes the mechanistic basis for these outcomes. The purpose of the present paper is to consider a theoretical account that may fill this gap in understanding and to review recent experimental findings from our laboratories that have been generated by this model.
A Pavlovian Perspective on Energy Regulation
Perhaps the most fundamental and well-established concept in psychology is that organisms learn about events that signal important biological outcomes. Pavlov's 12 finding that dogs would come to salivate to the sound of a metronome that preceded the delivery of meat powder provided the first experimental demonstration of this signaling relationship. Sensitivity to such simple Pavlovian contingencies is ubiquitous across species from the simple sea slug (e.g., Aplysia Californicus13) to humans, and can be demonstrated with a diverse array of stimuli and with a wide variety of response systems (see 14).
Among the most robust forms of Pavlovian conditioning are those that involve the establishment of taste stimuli (e.g., sweet, bitter) as signals for postingestive (e.g., nutritive, gastric malaise) consequences and that measure intake as the behavioral index of this learning (15,16). Similar to ideas originally proposed by Pavlov, current models of the physiological controls of energy regulation suggest that Pavlovian conditioning also enables tastes and other orosensory cues to evoke not only food intake, but a variety of hormonal, neural, and metabolic conditioned responses that prepare for and promote the efficient utilization of energy 17 One implication of these models is that circumstances that degrade the ability of tastes to predict the occurrence of caloric outcomes will also weaken the evocation of these physiological or “cephalic phase” responses with the effect that energy regulation would become less effective (18-21).
In nature, and throughout most of our evolutionary history, sweetness has been a reliable predictor of the energy content of food. Accordingly, within the Pavlovian framework outlined above, sweet taste should be a potent elicitor of the cephalic phase responses that promote energy regulation. The available data provide strong confirmation of this suggestion (22). However, we have suggested that beginning in the 1960's with the mass introduction of non-nutritive sweeteners into the food environment, the relationship between sweet taste and the caloric density of food may have changed (e.g., 18-21). That is, for consumers of non-nutritive sweeteners, sweet tastes are no longer always accompanied by energetic outcomes. Based on Pavlovian conditioning principles, because sweet tastes are no longer consistent predictors of strong nutritive postingestive consequences, the ability of sweet taste to evoke cephalic phase responses would be degraded, with the result being less effective energy regulation and increased caloric intake when normal sweet (and high calorie) foods are consumed.
An Animal Model of Energy Dysregulation
Pavlov was awarded the Nobel Prize in Medicine in 1904 for his research with dogs on digestive physiology. He then spent almost all of the remainder of his career investigating the implications of that work for the understanding of simple learning process, also using primarily non-human animal models. Pavlov's findings in both digestive physiology and in learning have been robustly confirmed in other species, including rats and humans, and have been the basis of important theoretical accounts of both types of processes (17,23). The research described next is based on the integration of principles from physiology and learning, and uses the control afforded by an animal model (the rat) to study how intake of non-nutritive sweeteners affects energy and body weight regulation.
Our research has investigated the effects of degrading the ability of sweet tastes to predict increments in the postingestive nutritive consequences of eating on several behavioral and physiological indices of energy regulation. We reasoned that if sweet tastes are normally valid predictors of increased caloric outcomes, then exposing rats to sweet taste that is not associated with these increased calories should degrade this predictive relationship and impair energy intake and body weight regulation. Some of our work has studied the effects of giving different groups of rats semi-solid dietary supplements (plain yogurt) that have either non-nutritive or nutritive sweeteners (glucose) added. We will begin by describing these findings before turning to studies that compared the effects on intake and body weight of consuming fluids that were mixed with nutritive or non-nutritive sweeteners.
Sweet taste-caloric relations: semi-solid foods
Body weight gain, adiposity, and food intake
In our basic design, in addition to ad libitum lab chow and water, we gave adult male rats access to both plain and sweetened lowfat, yogurt in addition to their regular lab chow and water. One group (Predictive) received 30 g of plain unsweetened yogurt (0.6 kcal/g) on some days and 30 g of yogurt sweetened with 20% glucose (1.2 kcal/g) on other days. Thus, for this group, sweetened diet predicted more calories than unsweetened diet. The second group (Non-Predictive) received plain unsweetened yogurt on some days and yogurt sweetened with 0.3% saccharin on the other days. Thus, for this group, sweetened yogurt did not predict more calories than unsweetened diet. With this arrangement, although the rats could differentiate between the sweetened and unsweetened forms of the yogurt, they were not given the opportunity to directly compare the taste or palatability of the caloric sweetener to the non-caloric sweetener. All rats (n's = 8-9 per group) were given 30 g of sweetened or unsweetened yogurt daily 6 days per week (3 days sweetened and 3 days unsweetened) in random order for 5 weeks in addition to ad lib chow and water. At the end of 5 weeks under these training conditions, rats in the Non-Predictive group, for which sweet taste did not signal an increase in the energy density of the yogurt, gained significantly more weight (left panel of Figure 1) and had significantly higher body adiposity (right panel of Figure 1) compared to the rats in the Predictive group for which sweet taste provided a valid signal for calories.
Figure 1.
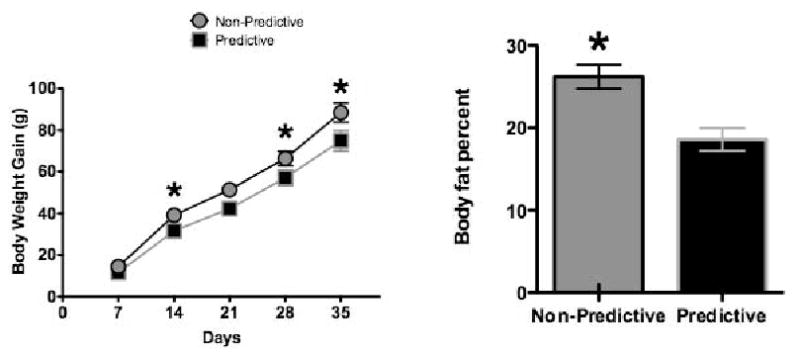
Body weight gain (left) and adiposity (right) were significantly greater in rats (n's = 8 – 9 per group) given access to saccharin-sweetened yogurt diet supplements in which sweet taste did not predict increased calories (Non-Predictive group) compared to animals given glucose-sweetened yogurt diet supplements (Predictive group) in which sweet taste did reliably predict increased calories. Adapted from 21. Used with permission.
* p < 0.05 compared to Predictive
Weekly total caloric intake (yogurt plus lab chow) intake over the same 5-week period is shown for the Non-predictive and Predictive groups. Figure 2 shows that total caloric intake (yogurt plus chow) tended to be higher in the Non-Predictive group. However, this difference did not achieve statistical significance.
Figure 2.
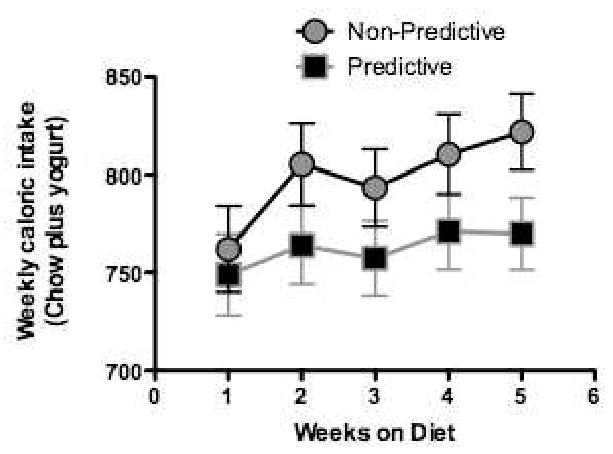
Total caloric intake tended to be greater in rats given access to saccharin-sweetened yogurt diet supplements in which sweet taste did not predict increased calories (Non-Predictive group) compared to animals given glucose-sweetened yogurt diet supplements (Predictive group) in which sweet taste did reliably predict increased calories (n's = 8 – 9 per group). Adapted from 21. Used with permission.
Caloric compensation
Additional findings show that consumption of saccharin-sweetened yogurt can promote excessive intake relative to yogurt sweetened with glucose by reducing caloric compensation. Caloric compensation is exhibited when animals adjust for calories consumed on one occasion by reducing their caloric intake at subsequent opportunities to eat (24-27). Therefore, weakened caloric compensation could result in excess energy intake and ultimately increased weight gain. We gave adult male Sprague-Dawley rats (n's = 8 – 9 per group) 30 g of low-fat, plain yogurt (Dannon) daily for 14 days in addition to ad-libitum lab chow and water. Similar to the design described above, rats in the Glucose group received plain, unsweetened yogurt for 7 of the 14 days and yogurt sweetened with 20% glucose for 7 of the 14 days. In contrast, rats in the Saccharin group received plain, unsweetened yogurt for 7 days and yogurt sweetened with 0.3% saccharin for 7 days. Thus, as above rats in the Glucose group were trained under conditions in which sweet taste was a highly valid predictor of increased calories, whereas for rats in the Saccharin group, sweet taste did not predict more calories. After this training, both groups were food-deprived overnight before being tested for caloric compensation using a novel, sweet tasting food. One test day, the rats were given a premeal of 5 g of Chocolate Ensure Plus thickened with guar gum (1.4 kcal/g) for 30 min and they were given no premeal on another test day, in counterbalanced order. On each test day, all rats were then given free access to lab chow and the amount eaten was recorded after 1, 2, and 4 hrs.
The results of the caloric compensation test showed that for the Glucose group (Figure 3a) caloric compensation was virtually complete in that the total amount of calories consumed (premeal and lab chow combined) was almost the same when the rats were tested following the premeal compared to the no premeal condition. To achieve this outcome, the rats in the Glucose Group had to reduce their intake of lab chow by an amount equivalent to the caloric content of the Ensure premeal. In contrast, much weaker caloric compensation was shown by the rats in the Saccharin group. As can be seen in Figure 3b, for this group, total energy intake was significantly greater following the Ensure premeal compared to the no-premeal condition. This outcome indicates that rats in the Saccharin group were less able to reduce their intake of lab chow to compensate for calories contained in the premeal compared to rats in the Glucose group. If continued over the long-term, reduced caloric compensation could contribute to excess body weight gain. Within our theoretical framework, weaker caloric compensation by the Saccharin group was expected to the extent that the control of energy intake relies, at least in part, on the ability of sweet taste to signal the caloric content of the Ensure premeal. This signaling relationship would be degraded by prior exposure to sweet but non-nutritive saccharin, but not by prior exposure to sweet and high-calorie glucose.
Figure 3.
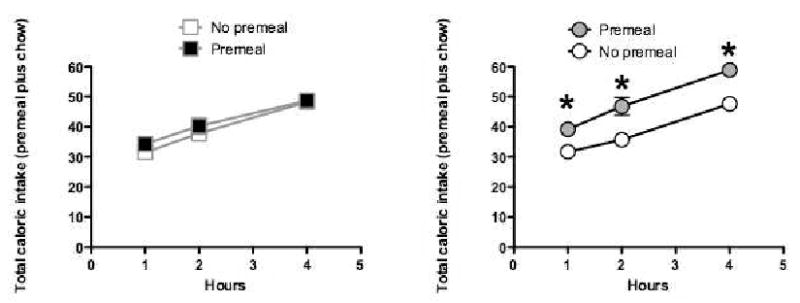
Rats given experience with Predictive experience with sweet tastes and calories (left) showed strong caloric compensation for calories provided in a novel, sweet tasting premeal. Total caloric intake was not significantly different on days when a premeal was provided compared to days on which a premeal was not provided. In contrast, rats given experience with Non-Predictive relations between sweet taste and calories (right) failed to compensate for calories provided in a novel sweet premeal (n's = 8 – 9 per group). Adapted from 21. Used with permission.
* p< 0.05 compared to No Premeal
Generality and persistence of the phenomenon
Additional research showed that the effects of degrading sweet taste-calorie relations on body weight gain are not specific to saccharin but are also obtained when 0.3% Acesulfame Potassium (AceK), a non-nutritive sweetener with a chemical structure that is distinct from saccharin. AceK has been employed in a variety of sweetened beverages including Diet Coke with Splenda, Diet Pepsi Lime and Diet Pepsi Vanilla, Minute Maid Light, and Dasani and Aquafina sweetened waters. In one study19, three groups of adult male rats were given 23-hr access to 30 g of low-fat, unflavored yogurt each day in addition to ad lib chow for 14 days; on seven days, plain yogurt was provided while sweetened yogurt was provided on the other seven days with order of presentation quasi-randomized. As in our previous studies, the yogurt was sweetened with 20% glucose for one group and was sweetened with 0.3% saccharin for a second group. Extending our earlier research, body weight gain for these groups was compared to a third group that received yogurt sweetened with 0.3% AceK. Figure 4 shows that while body weight gain over the 14 days of training differed little for rats given saccharin-sweetened relative to those given AceK-sweetened yogurt, both of these groups gained significantly more weight than the group given yogurt sweetened with glucose. A similar pattern of results with the same statistical outcome was obtained in another study (see Experiment 2, 19) that compared body weight gain for rats given only one hour access to yogurt sweetened with saccharin, AceK, or glucose (data not shown).
Figure 4.
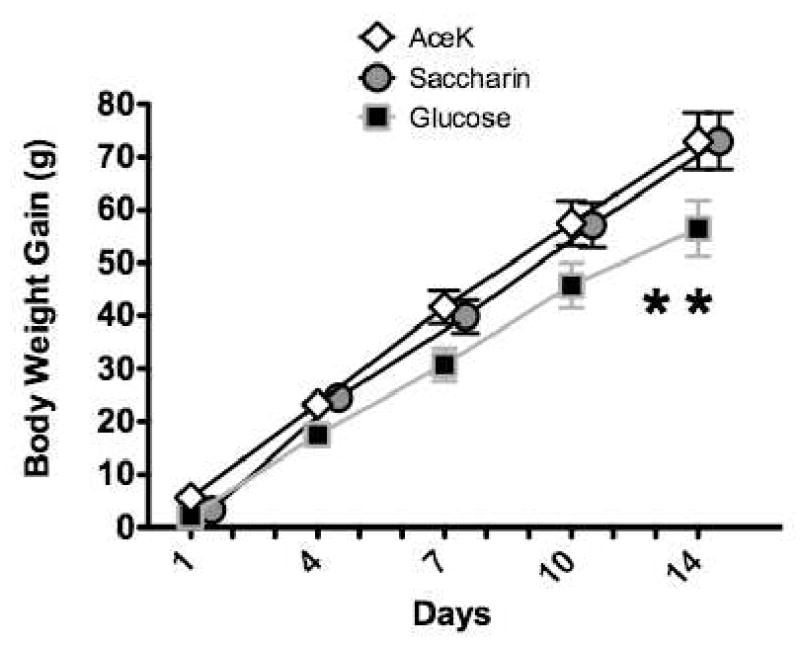
Body weight gain was significantly greater in rats given yogurts sweetened with saccharin or AceK compared to rats given yogurts sweetened with glucose (n's = 8 per group). Adapted from 19. Used with permission.
* p < 0.05 compared to AceK and Saccharin
We also compared the persistence of body weight differences following the discontinuation of the yogurt supplements sweetened with saccharin or glucose. Following 14 days of training with daily one hour access to 20 g of plain or sweetened yogurt, presentation of the yogurt supplements was discontinued and body weight gain was measured for an additional two weeks. Replicating our previous findings, body weight gain during the period when the yogurt supplements were presented was significantly greater for rats that ate saccharin-sweetened compared to glucose-sweetened yogurt (Figure 5A). In addition, body weight gain after yogurt presentation was terminated did not differ among these groups (Figure 5B) indicating that body weight differences that emerged during prior training with yogurt persisted over the course of the subsequent two-week period without yogurt.
Figure 5.
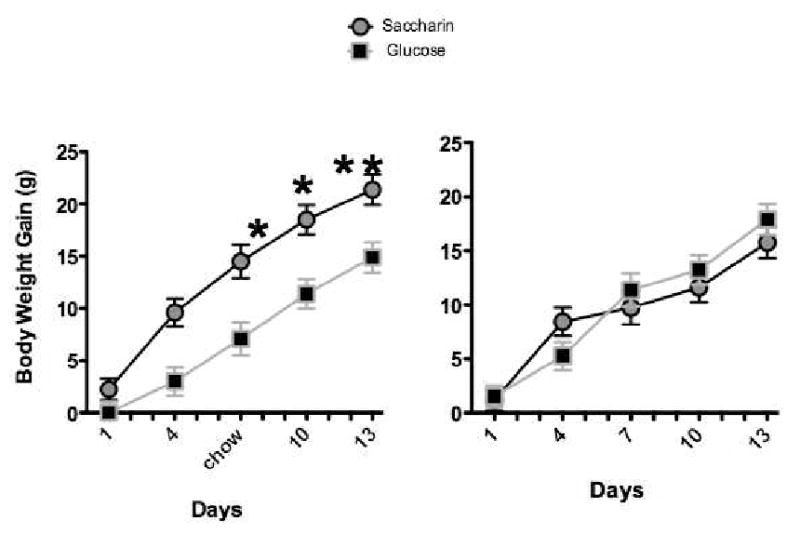
Body weight gain was significantly greater during 2 weeks exposure (left) to saccharin-sweetened yogurt compared to glucose-sweetened yogurt. Following discontinuation of yogurt access (right), animals in the saccharin-sweetened group failed to compensate for the prior excess weight gain (n=13 per group). Adapted from 19. Used with permission.
* p < 0.05 compared to Glucose
Reversibility of the effects of non-nutritive sweeteners on weight gain
We also assessed the effects of reversing the predictive and non-predictive relationships on weight gain and energy intake. Specifically, using a factorial design, we compared the effects on body weight gain of shifting adult, male, rats trained with saccharin-sweetened diets to diets sweetened with glucose (saccharin-glucose) and vice-versa (glucose-saccharin) and compared these groups to unshifted (saccharin-saccharin and glucose-glucose) controls. In addition, the sweeteners were presented mixed not only with plain yogurt, but in separate groups, mixed with refried beans (pilot work established that rats would readily consumed both sweetened and unsweetened refried beans). For all rats, training occurred with one base diet (yogurt or beans) and testing occurred with the other. Because the base diets were shifted from training to test for all groups, differences in body weight for the shifted versus unshifted sweetener conditions could not be attributable changes in the base diet. The training and test phases were each two weeks in duration, and daily 23-hr access to 30 g of the base diet, plain on half the days and sweetened on the other half, were presented in quasi-randomized order.
The results of the training and test phases are presented, collapsed across the yogurt and refried bean base diets, in Figures 6 and 7, respectively. Confirming our previous findings, the results of training (Figure 6) showed that weight gain was significantly greater for rats that consumed saccharin-sweetened compared to glucose-sweetened base diets. Furthermore, shifting from a base diet sweetened with glucose to one sweetened with saccharin was followed by a significant increase in weight gain relative to the unshifted glucose-glucose control. In fact, weight gain for the glucose-saccharin shifted group achieved the level of the unshifted saccharin-saccharin control group by the end of the testing. In contrast, shifting from a saccharin- to a glucose-sweetened base diet (Figure 7) produced little change in body weight gain relative to the unshifted saccharin-saccharin control. Thus, the results of this experiment showed not only that the effects on body weight of exposure to a non-predictive sweet-calorie relationship (as a result of training with saccharin) are persistent, but also that they are difficult to reverse when a predictive sweet-calorie relation is established by training with glucose.
Figure 6.
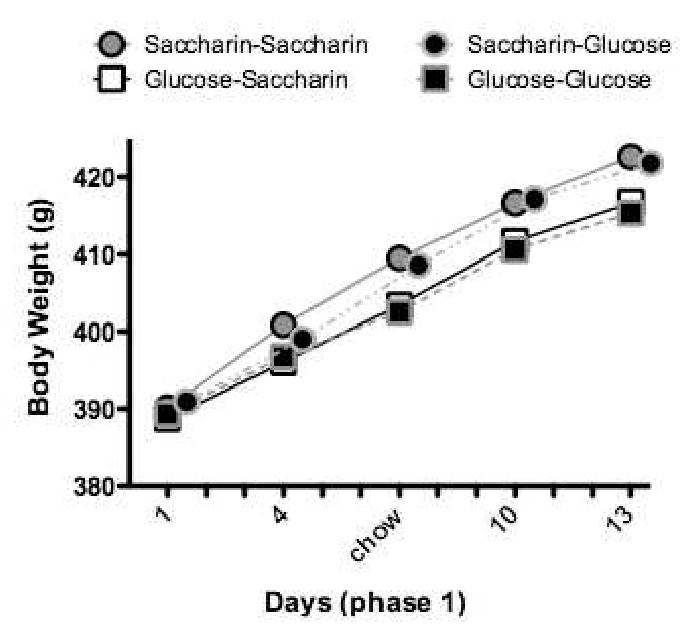
During exposure to yogurt or refried beans diets sweetened with saccharin, body weight gain was significantly greater compared to the same base diets sweetened with glucose (n's = 12-14 per group). Adapted from Adapted from 19. Used with permission.
Figure 7.
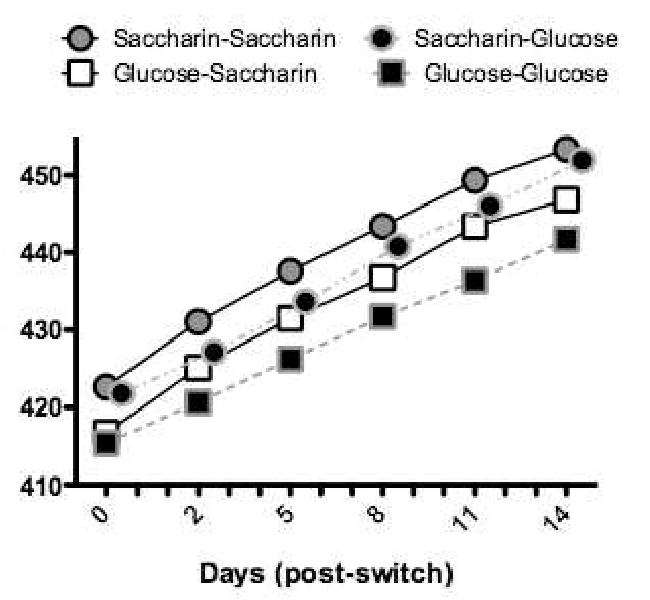
Body weight gain in all rats given saccharin-sweetened yogurt or refried beans was significantly greater compared to animals given access to glucose-sweetened diets (n's =12- 14 per group). Adapted from Adapted from 19. Used with permission.
Thermogenesis: a potential physiological mechanism
It is well-established that food intake elicits a reflexive cephalic phase thermogenic response in both human and non human animals. This form of heat production appears to be mediated, in part, by orosensory stimuli that precede and signal the absorption of nutrients in the gut. In humans when food is tasted but not swallowed, as well as in dogs allowed to taste food but not digest it, the thermogenic response to the oral stimulation produced by food has been reported to exceed that produced by a meal that is ingested normally28. Alternatively, when nutrients bypass the oropharyngeal cavity (e.g., via feeding tube) cephalic phase thermogenic responses for both humans and dogs are either not observed or much weaker than those produced by normal feeding 29,30. These responses, along with other cephalic phase reflexes (e.g., hormonal, gastric, exocrine), are generally thought to perform anticipatory functions that prepare the GI tract for the arrival of nutrients, increase the efficiency of nutrient utilization, and minimize the degree to which those nutrients perturb homeostasis by producing positive energy balance 17,31,32.
One component of this thermogenic response is an increment in core body temperature, the magnitude of which is determined primarily by the energy content of the food 33,34. If sweet tastes evoke thermic responses based, in part, on the degree to which they predict the arrival of calories in the gut, one might expect that tasting a sweet, high-calorie food would evoke a greater increase in core body temperature for rats that have been exposed to a highly reliable predictive relationship between sweet tastes and calories compared to rats that for which sweet taste is a less reliable signal for calories. One implication of this hypothesis is that if consuming non-nutritive sweeteners reduces the validity of sweet taste as a signal for calories, then one effect of consuming these sweeteners would be a reduced ability for sweet tasting foods to evoke increments in core body temperature.
We tested this prediction by implanting miniature radio-frequency transmitters in the abdominal cavity of 16 adult male rats to allow us to measure remotely changes in core body temperature immediately before, during, and after the rats consumed premeals and test meals that contained nutritive or non-nutritive sweeteners 21. During training, rats received 30 g of a low-fat, plain yogurt daily for 14 days in addition to ad-lib lab chow and water. As in our earlier studies, the yogurt was sweetened on a quasi-random half of the training days, and was unsweetened on the remaining days. For half of the rats, the sweetener used was always saccharin, whereas glucose was the sweetener for the other rats. When training was complete, all the rats were tested following overnight food deprivation with a premeal of 5-g thickened Chocolate Ensure Plus. During training and testing temperature was recorded at 1-min intervals.
Figure 8A shows core temperature data during the first 60 min after presentation of sweetened and unsweetened yogurt, averaged across all training sessions, for rats that received yogurt sweetened with glucose. Comparable data for rats that received yogurt sweetened with saccharin are shown in Figure 8B. The results indicate that while intake of yogurt sweetened with glucose produced a larger and more sustained increment in core temperature relative to unsweetened yogurt, consuming yogurt sweetened with saccharin did not have this effect. This is not surprising given that the energy content of glucose sweetened yogurt was substantially greater than for unsweetened yogurt, whereas the energy content of saccharin-sweetened and unsweetened yogurt were the same. However, this pattern of results indicated that our measures of core temperature were consistent with previous reports showing that energy density is an important determinant of the thermic effect of food35.
Figure 8.
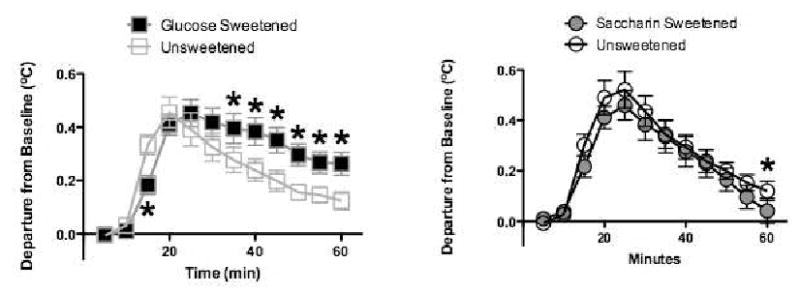
Increases in core body temperature across two weeks of exposure to plain, unsweetened yogurt and yogurt sweetened with glucose (left) or saccharin (right) (n's =6 – 8 per group). Adapted from 21. Used with permission.
*p < 0.05 compared to unsweetened yogurt
Of greater interest are the results from when the rats in the glucose-trained and saccharin-trained groups were tested with a common novel, sweet, and high-calorie supplement, Chocolate Ensure Plus. We reasoned that compared to the rats trained with glucose, rats trained with saccharin should exhibit a weaker thermic response to the sweet taste of the Ensure. That is because, for those rats, prior consumption of sweet, non-nutritive saccharin should have reduced the ability of sweet taste to predict calories and thus, should have reduced the ability of sweet taste to evoke increments in core temperature. This prediction was confirmed (see Figure 9). That is, the increment in core temperature that occurred for the saccharin group was significantly smaller than that for the glucose group when both groups were tested with same novel sweet-tasting diet. These results suggest that experience with non-nutritive sweeteners may perturb at least one physiological mechanism involved with energy regulation, in a manner that was anticipated with a theoretical framework based on Pavlovian conditioning 21.
Figure 9.
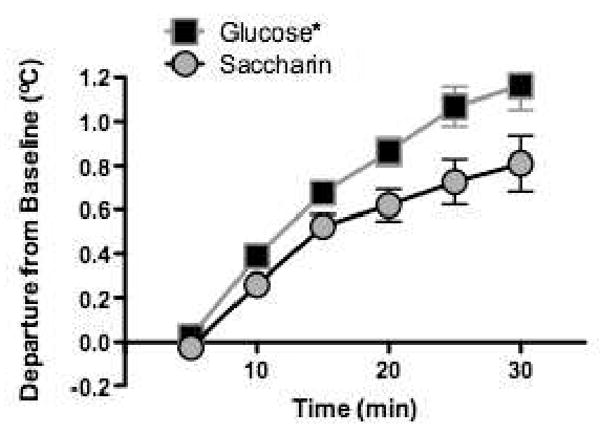
Increases in core body temperature following a premeal of a novel, sweet diet were significantly higher in animals with previous experience consuming glucose-sweetened yogurt diets compared to animals with previous experience consuming saccharin-sweetened yogurt diets (n's =6 – 8 per group). Adapted from 21. Used with permission.
*p < 0.05 compared to saccharin group
Sweet taste-caloric relations: fluids
Caloric compensation
As noted in the Introduction, much of the concern about the effectiveness using non-nutritive sweeteners as a method of weight control stems from epidemiological studies that indicate a link between body weight gain and the consumption of diet beverages. We have also used our animal model to investigate this link.
As an initial test of this idea, Swithers 18assigned juvenile male and female rats (∼35 days old) to one of two groups. The first group (Consistent) received experience with two sweet diets that contained calories - 10% sucrose and 10% glucose (w/v). The second group of rats (Inconsistent) received experience with one caloric sweet diet, 10% glucose, and one non-caloric sweet diet, 0.3% saccharin. All diets were flavored with 0.1% grape or cherry Kool-Aid. The order of presentation of the diets and the Kool-Aid flavor associated with each diet were counterbalanced across litters. The rats were given 5 days of training where a fluid supplement was offered for 24 hr, then removed and replaced with the alternative supplement. Following this training, the rats were food deprived overnight, then given a small (10 g) preload of a novel, sweet-tasting, and high-caloric premeal of Rich Chocolate Ensure Plus® to consume for 1 hr. One hour after the end of this premeal period, intake of their regular rat chow diet was assessed.
Analogous to our account for the data obtained with semi-solid foods, our hypothesis was that rats in the Inconsistent group would compensate less well for the sweet-tasting preload compared to rats animals for which sweet taste was a consistent predictor of calories (i.e., the Consistent group). The results showed (see Figure 10) that although intake of the premeal did not differ between the groups, subsequent chow intake for the Inconsistent group during was significantly greater compared to the Consistent Group, for which sweet taste was a highly reliable signal for calories. These data indicate that inconsistent experiences with sweet tastes and calories may have impaired compensation for the calories in the sweet premeal.
Figure 10.
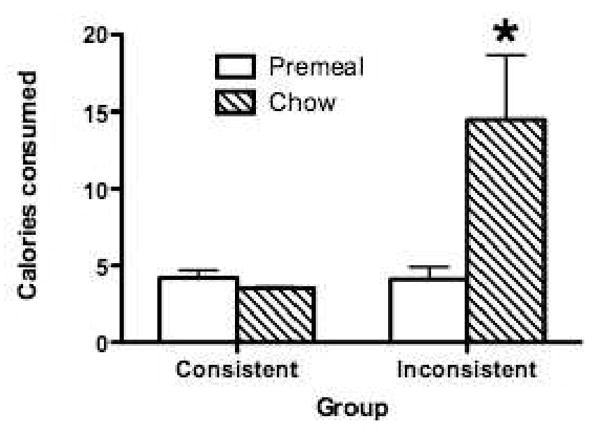
In juvenile rats given experience with Inconsistent relations between sweet taste and calories (glucose and saccharin solutions), chow intake following a novel, sweet premeal was significantly greater compared to juvenile rats given previous experience with Consistent relations between sweet taste and calories (glucose and sucrose solutions; n = 10 per group). Adapted from 18. Used with permission.
* p < 0.05 compared to Consistent group
Food intake and body weight gain
In addition to data showing that exposure to sweet, but non-nutritive fluids may increase short-term intake by impairing caloric compensation, very recent studies in our laboratories have assessed the effects of such exposure on food intake and body weight gain. In one study, adult male rats were assigned to one of two groups, matched on ad libitum body weight. One group was given water sweetened with 10% glucose (n = 16) and the other was given water sweetened with 0.3% saccharin (n = 13). The solutions were presented overnight for ten consecutive days. Each rat received 10 ml of its sweetened solution for the first two days, after which they were given 20 ml of the solution on each of the next eight days. The rats were also given free access to lab chow and water throughout the experiment. Body weight and amount of lab chow consumed were recorded for all rats on Days 1, 5, and 10 of this period.
Figure 11 shows mean body weight for the groups given saccharin or glucose solutions on the day immediately before, and 1, 5, and 10 days after the solutions were presented. Mean daily intake of lab chow is shown for each group over the same period in Figure 12. The results indicate that the rats given the saccharin solutions both gained weight significantly more rapidly and ate significantly more chow overall than the rats given the glucose solution.
Figure 11.
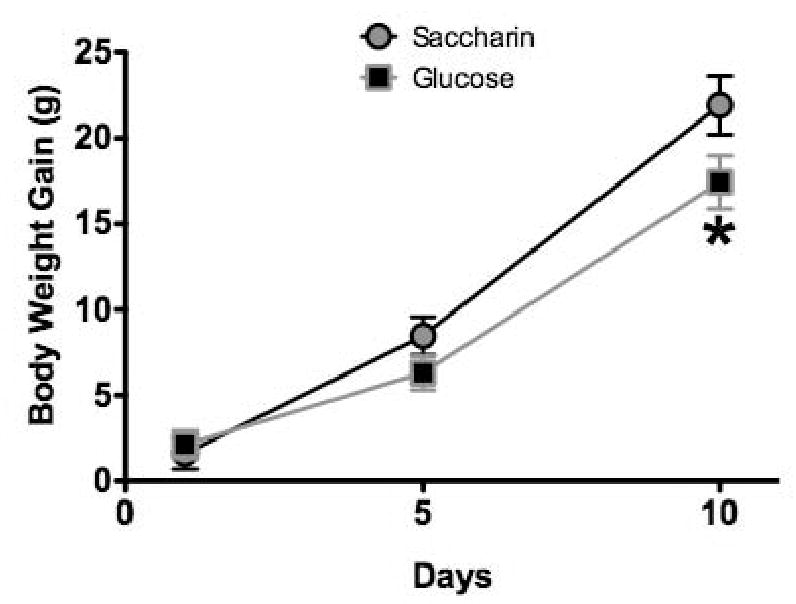
Body weight gain was significantly greater in rats (n = 13- 16 per group) following 10 days exposure to saccharin solutions compared to glucose solutions.
* p < 0.05 compared to saccharin
Figure 12.
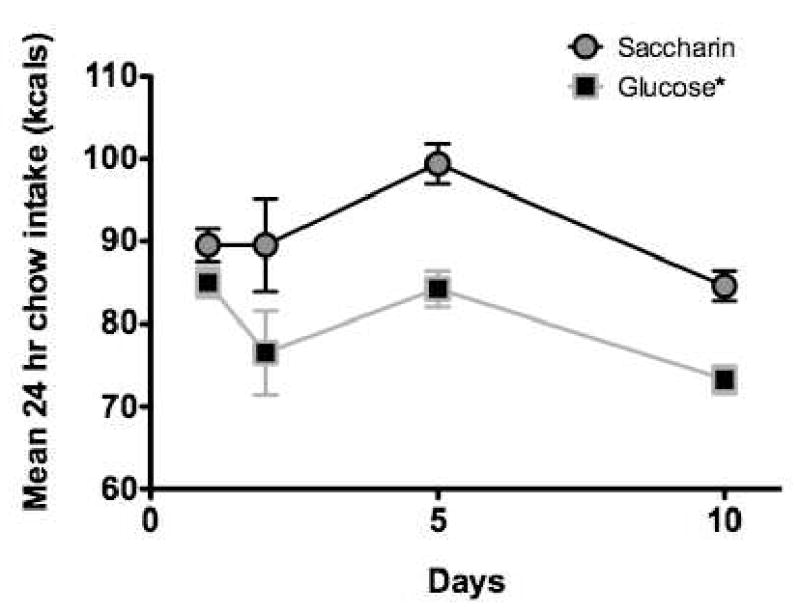
Chow intake was significantly greater during 10 days exposure to in saccharin solutions compared to 10 days exposure to glucose solutions (n = 13- 16 per group).
* p <0.05 compared to saccharin
Stevia solutions have effects on body weight gain similar to saccharin solutions
Recently, sweeteners extracted from the leaves of the Stevia rebaudiana plant have been introduced as “natural” non-nutritive sweeteners in the U.S. market. Like the “artificial” sweeteners AceK and saccharin, stevioside is reported to be significantly sweeter than sugar (250 to 300 times). To assess the generality of the effects of consuming saccharin solutions on body weight gain that we found in the preceding study, we gave one group of adult male rats access to a 30 – 50 ml of a solution of Stevia extract (Steviva) mixed in water (0.1% w/w) overnight for 15 days and compared body weight gain for this group with that of rats given the same type of exposure to 0.3% saccharin or 10% glucose solutions (n = 18 for each group). Lab chow and tap water were available throughout the experiment. Weight gain for rats given Stevia and those given saccharin did not differ significantly any point during this training (Figure 13). In contrast, by the end of 15 days of exposure weight gain for each of these groups was significantly greater than that for the group given the glucose solution. Thus, just as the effects on body weight gain of consuming semi-solid yogurt mixed with non-nutritive sweeteners are not specific to saccharin, the results of this study support the same conclusion when sweet, non-nutritive solutions are consumed.
Figure 13.
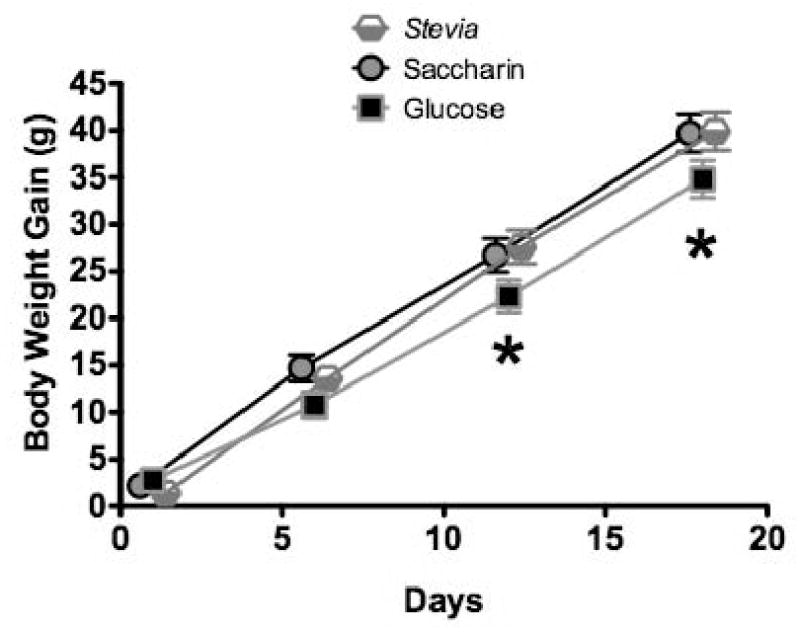
Body weight gain was significantly greater in animals consuming either saccharin solutions or Stevia solutions compared to animals consuming glucose solutions (n=18 per group).
* p< 0.05 compared to saccharin or Stevia
Summary and Conclusions
The research reported here provides an answer to the question “Can consuming non-nutritively-sweetened foods or fluids promote increased energy intake and body weight gain compared to sugar-sweetened foods and fluids?” In principle, the answer clearly seems to be yes. Using a rodent model, we found that intake of foods or fluids containing non-nutritive sweeteners was accompanied by increased food intake, body weight gain, accumulation of body fat, and weaker caloric compensation, compared to consumption of foods and fluids containing glucose. Our research also provided evidence consistent with the hypothesis that these effects of consuming saccharin may be associated with a decrement in the ability of sweet taste to evoke thermic responses, and perhaps other physiological, cephalic phase, reflexes that are thought to help maintain energy balance. Furthermore, our studies were generated within a theoretical framework provided by Pavlovian conditioning, and the results we obtained seem readily interpretable within a mechanistic approach that integrates fundamental concepts from learning and physiology. Thus, the claim that there is no viable mechanistic explanation of how intake of non-nutritive sweeteners could promote weight gain can be repudiated.
However, it is always possible to question whether any “in principle” demonstration, obtained with an animal model, under well-controlled laboratory conditions, can shed light on factors that are currently promoting excess energy intake and body weight gain in the much more complex and uncontrolled human food environment. Yet, despite this complexity, it is plausible, if not probable, that (a) both human and nonhuman animals rely on learning about the relationship between the sensory properties of foods and their nutritive postingestive consequences to help maintain energy balance; and (b) consuming diets in which the orosensory properties of foods and beverage are not strongly predictive of their caloric consequences can disrupt energy regulation. Thus, it is conceivable that just as exposure to non-predictive sweet taste-calorie relationships in the laboratory promotes increased intake, body weight and body adiposity in rats, the widespread use of non-caloric sweeteners by humans outside of the laboratory may have similar effects on the predictive validity of sweet tastes and ultimately on the ability of humans to control their intake and body weight.
Acknowledgments
We thank Melissa McCurley, Chelsea Baker, Jennie Mak, Lindsey Schier, Anna Barajas, Alycia LaBoy and Matt Gifford for technical assistance. Supported by NIH grants R01DK076078 and P01HD052112.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Chen L, Appel LJ, Loria C, Lin PH, Champagne CM, Elmer PJ, Ard JD, Mitchell D, Batch BC, Svetkey LP, et al. Reduction in consumption of sugar-sweetened beverages is associated with weight loss: the PREMIER trial. Am J Clin Nutr. 2009;89(5):1299–306. doi: 10.3945/ajcn.2008.27240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dhingra R, Sullivan L, Jacques PF, Wang TJ, Fox CS, Meigs JB, D'Agostino RB, Gaziano JM, Vasan RS. Soft drink consumption and risk of developing cardiometabolic risk factors and the metabolic syndrome in middle-aged adults in the community. Circulation. 2007;116(5):480–8. doi: 10.1161/CIRCULATIONAHA.107.689935. [DOI] [PubMed] [Google Scholar]
- 3.Dubois L, Farmer A, Girard M, Peterson K. Regular sugar-sweetened beverage consumption between meals increases risk of overweight among preschool-aged children. J Am Diet Assoc. 2007;107(6):924–34. doi: 10.1016/j.jada.2007.03.004. discussion 934-5. [DOI] [PubMed] [Google Scholar]
- 4.Fung TT, Malik V, Rexrode KM, Manson JE, Willett WC, Hu FB. Sweetened beverage consumption and risk of coronary heart disease in women. Am J Clin Nutr. 2009;89(4):1037–42. doi: 10.3945/ajcn.2008.27140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Malik VS, Schulze MB, Hu FB. Intake of sugar-sweetened beverages and weight gain: a systematic review. Am J Clin Nutr. 2006;84(2):274–88. doi: 10.1093/ajcn/84.1.274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lutsey PL, Steffen LM, Stevens J. Dietary intake and the development of the metabolic syndrome: the Atherosclerosis Risk in Communities study. Circulation. 2008;117(6):754–61. doi: 10.1161/CIRCULATIONAHA.107.716159. [DOI] [PubMed] [Google Scholar]
- 7.Nettleton JA, Polak JF, Tracy R, Burke GL, Jacobs DR., Jr Dietary patterns and incident cardiovascular disease in the Multi-Ethnic Study of Atherosclerosis. Am J Clin Nutr. 2009;90(3):647–54. doi: 10.3945/ajcn.2009.27597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fowler SP, Williams K, Resendez RG, Hunt KJ, Hazuda HP, Stern MP. Fueling the obesity epidemic? Artificially sweetened beverage use and long-term weight gain. Obesity (Silver Spring) 2008;16(8):1894–900. doi: 10.1038/oby.2008.284. [DOI] [PubMed] [Google Scholar]
- 9.Mattes RD, Popkin BM. Nonnutritive sweetener consumption in humans: effects on appetite and food intake and their putative mechanisms. Am J Clin Nutr. 2009;89(1):1–14. doi: 10.3945/ajcn.2008.26792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bellisle F, Drewnowski A. Intense sweeteners, energy intake and the control of body weight. Eur J Clin Nutr. 2007;61(6):691–700. doi: 10.1038/sj.ejcn.1602649. [DOI] [PubMed] [Google Scholar]
- 11.Blundell JE, Green SM. Effect of sucrose and sweeteners on appetite and energy intake. Int J Obes Relat Metab Disord. 1996;20 2:S12–7. [PubMed] [Google Scholar]
- 12.Pavlov IP. In: Conditioned Reflexes. Anrep GV, translator. London: Oxford University Press; 1927. [Google Scholar]
- 13.Baxter DA, Byrne JH. Feeding behavioral of Aplysia: A model system for comparing cellular mechanisms of classical and operant conditioning. Learning & Memory. 2006;13:669–680. doi: 10.1101/lm.339206. [DOI] [PubMed] [Google Scholar]
- 14.Rescorla RA. Behavioral studies of Pavlovian conditioning. Annu Rev Neurosci. 1988;11:329–52. doi: 10.1146/annurev.ne.11.030188.001553. [DOI] [PubMed] [Google Scholar]
- 15.Sclafani A. Learned controls of ingestive behaviour. Appetite. 1997;29(2):153–8. doi: 10.1006/appe.1997.0120. [DOI] [PubMed] [Google Scholar]
- 16.Welzl H, D'Adamo P, Lipp HP. Conditioned taste aversion as a learning and memory paradigm. Behav Brain Res. 2001;125(1-2):205–13. doi: 10.1016/s0166-4328(01)00302-3. [DOI] [PubMed] [Google Scholar]
- 17.Woods SC, Ramsay DS. Pavlovian influences over food and drug intake. Behav Brain Res. 2000;110(1-2):175–82. doi: 10.1016/s0166-4328(99)00194-1. [DOI] [PubMed] [Google Scholar]
- 18.Davidson TL, Swithers SE. A Pavlovian approach to the problem of obesity. Int J Obes Relat Metab Disord. 2004;28(7):933–5. doi: 10.1038/sj.ijo.0802660. [DOI] [PubMed] [Google Scholar]
- 19.Swithers SE, Baker CR, Davidson TL. General and persistent effects of high-intensity sweeteners on body weight gain and caloric compensation in rats. Behav Neurosci. 2009;123(4):772–80. doi: 10.1037/a0016139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Swithers SE, Davidson TL. Obesity: outwitting the wisdom of the body? Curr Neurol Neurosci Rep. 2005;5(3):159–62. doi: 10.1007/s11910-005-0041-0. [DOI] [PubMed] [Google Scholar]
- 21.Swithers SE, Davidson TL. A role for sweet taste: calorie predictive relations in energy regulation by rats. Behav Neurosci. 2008;122(1):161–73. doi: 10.1037/0735-7044.122.1.161. [DOI] [PubMed] [Google Scholar]
- 22.Mattes RD. Physiologic responses to sensory stimulation by food: nutritional implications. J Am Diet Assoc. 1997;97(4):406–13. doi: 10.1016/S0002-8223(97)00101-6. [DOI] [PubMed] [Google Scholar]
- 23.Rescorla RA, Wagner AR. A theory of Pavlovian conditioning: Variations in the effectiveness of reinforcement and nonreinforcement. In: Black AH, Prokasy WF, editors. Classical Conditioning II: Current Research and Theory. New York: Appleton Century Crofts; 1972. pp. 64–99. [Google Scholar]
- 24.Booth DA. Conditioned satiety in the rat. J Comp Physiol Psychol. 1972;81(3):457–71. doi: 10.1037/h0033692. [DOI] [PubMed] [Google Scholar]
- 25.Foltin RW, Fischman MW, Moran TH, Rolls BJ, Kelly TH. Caloric compensation for lunches varying in fat and carbohydrate content by humans in a residential laboratory. Am J Clin Nutr. 1990;52(6):969–80. doi: 10.1093/ajcn/52.6.969. [DOI] [PubMed] [Google Scholar]
- 26.Mazlan N, Horgan G, Stubbs RJ. Energy density and weight of food effect short-term caloric compensation in men. Physiol Behav. 2006;87(4):679–86. doi: 10.1016/j.physbeh.2006.01.032. [DOI] [PubMed] [Google Scholar]
- 27.Rowland NE, Nasrallah N, Robertson KL. Accurate caloric compensation in rats for electively consumed ethanol-beer or ethanol-polycose mixtures. Pharmacol Biochem Behav. 2005;80(1):109–14. doi: 10.1016/j.pbb.2004.10.010. [DOI] [PubMed] [Google Scholar]
- 28.LeBlanc J. Nutritional implications of cephalic phase responses. Appetite. 2000;34:214–216. doi: 10.1006/appe.1999.0283. [DOI] [PubMed] [Google Scholar]
- 29.Diamond P, Brondel L, LeBlanc J. Palatability and postprandial thermogenesis in dogs. Am J Physiol. 1985;248(1 Pt 1):E75–9. doi: 10.1152/ajpendo.1985.248.1.E75. [DOI] [PubMed] [Google Scholar]
- 30.LeBlanc J, Cabanac M, Samson P. Reduced postprandial heat production with gavage as compared with meal feeding in human subjects. Am J Physiol. 1984;246(1 Pt 1):E95–101. doi: 10.1152/ajpendo.1984.246.1.E95. [DOI] [PubMed] [Google Scholar]
- 31.Powley TL, Berthoud HR. Diet and cephalic phase insulin responses. Am J Clin Nutr. 1985;42(5 Suppl):991–1002. doi: 10.1093/ajcn/42.5.991. [DOI] [PubMed] [Google Scholar]
- 32.Teff K. Nutritional implications of the cephalic-phase reflexes: endocrine responses. Appetite. 2000;34(2):206–13. doi: 10.1006/appe.1999.0282. [DOI] [PubMed] [Google Scholar]
- 33.de Graaf C, Blom WA, Smeets PA, Stafleu A, Hendriks HF. Biomarkers of satiation and satiety. Am J Clin Nutr. 2004;79(6):946–61. doi: 10.1093/ajcn/79.6.946. [DOI] [PubMed] [Google Scholar]
- 34.Westerterp KR. Diet induced thermogenesis. Nutr Metab (Lond) 2004;1(1):5. doi: 10.1186/1743-7075-1-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jequier E. Thermogenic responses induced by nutrients in man: their importance in energy balance regulation. Experientia Suppl. 1983;44:26–44. doi: 10.1007/978-3-0348-6540-1_3. [DOI] [PubMed] [Google Scholar]


