Abstract
Male mice deficient in ESR1 (ERalpha) (Esr1KO mice) are infertile, and sperm recovered from the cauda epididymis exhibit reduced motility and fail to fertilize eggs in vitro. These effects on sperm appear to result from defective epididymal function and not a direct effect on spermatogenesis, as Esr1KO germ cells transplanted into wild-type testes yield normal offspring. We hypothesized that the previously described defect in efferent duct fluid reabsorption would lead to alterations in the epididymal fluid milieu, which would negatively impact sperm function. Analysis of the epididymal fluid revealed that the Esr1KO maintains a higher luminal pH throughout the epididymis, confirming an inability of the efferent ducts and/or epididymis to properly acidify the luminal contents. Subsequent studies showed that these abnormalities were not the result of global defects in epididymal function since protein secretion by the Esr1KO epididymis appeared normal as judged by SDS-PAGE of total secreted proteins and by immunoblotting of candidate secreted proteins. To gain insight into the basis of the aberrant fluid homeostasis in the Esr1KO epididymis, the expression of several enzymes and transporters known to be involved in acid/base regulation were analyzed. The levels of SLC9A3 (NHE3) as well as carbonic anhydrase XIV and SLC4A4 (NBC1) were all reduced in the proximal portion of the Esr1KO epididymis, while other components appeared unaffected, including other ion transporters and ATP6V0A1 (V-ATPase). The altered luminal milieu of the Esr1KO epididymis was shown to lead to a corresponding increase in the intracellular pH of Esr1KO sperm, relative to sperm from control animals. Since pH and bicarbonate ions are critical regulators of sperm cAMP levels and motility, we attempted to bypass the abnormal luminal and intracellular environment by supplementing sperm with exogenous cAMP. This treatment rescued all defective motility parameters, as assayed by CASA, further showing that motility defects are not intrinsic to the sperm but, rather, result from the abnormal epididymal milieu.
Keywords: epididymis, estradiol receptor, male reproductive tract, sperm maturation, sperm motility and transport
Estrogen receptor alpha is essential for the regulation of the epididymal luminal microenvironment.
INTRODUCTION
The creation of transgenic mouse models lacking a functional Esr1 gene, collectively known as the ERα knockout (Esr1KO or Esr1−/−), has provided insight into the importance of ESR1 in the regulation of male fertility and reproductive tract function [1–3]. Animals that lack a functional Esr1 gene are sterile, and sperm recovered from the cauda epididymis exhibit a low percent motility, beat less vigorously, and are ineffective at in vitro fertilization. However, this infertility is not due to a primary defect in spermatozoa, as transplantation of Esr1KO germ cells into a wild-type (WT) reproductive tract yields normal offspring [4, 5]. Recent studies, however, suggest that Sertoli cells may express ESR1 and respond to estrogen [6], which could help explain age-dependent testicular degeneration in the aromatase knockout mouse [7].
As sperm leave the testis, they are funneled through a series of tubes called the efferent ducts, which function to reabsorb testicular fluid [8, 9]. The absorptive capacity of the efferent ducts concentrates the sperm as they enter the initial segment of the epididymis. Together, these two regions help to create and maintain a luminal microenvironment that is optimal for sperm viability and function. It is now known that in the absence of Esr1, the efferent ducts fail to execute their primary function of fluid reabsorption, causing an accumulation of fluid in the lumen and subsequent backpressure atrophy of the testis [3, 10, 11].
Previous studies have reported that the efferent ducts of the Esr1KO animals have reduced expression of several proteins that are important for controlling fluid/ion dynamics, including the sodium/hydrogen exchanger 3 (NHE3, official symbol SLC9A3), carbonic anhydrase II (CAR2), and two water channels known as aquaporin 1 and 9 (AQP1 and AQP9) [12, 13]. Analysis of mRNA levels concluded that ESR1 has a primary effect on SLC9A3 transcription, while lowered expression of CAR2, AQP1, and AQP9 are likely secondary to morphological defects of the efferent duct epithelium. ESR1 is also expressed in the proximal regions of the epididymis [14]; however, a detailed examination of transport channels in the Esr1KO epididymis has yet to be determined.
The epididymis plays an important role in sperm maturation, transport, protection, and storage [15–17] and consists of a single convoluted duct that can be subdivided into four regions known as the initial segment, caput, corpus, and cauda [18, 19]. Each of these regions has distinct and overlapping functional properties that stem from differences in the constituent epithelial cell types. There are two main cell types in the epididymis that contribute to providing a specialized luminal environment for sperm. The most abundant is the principal cell, which comprises approximately 65%–80% of the total epithelial cell population and synthesizes all proteins secreted into the epididymal lumen [20, 21]. The second major cell type are the clear cells, also known as narrow cells in the initial segment. These cells are large endocytic cells that are responsible for the uptake of a number of different proteins and osmolytes excreted by the epididymal epithelium as well as for the secretion of protons to facilitate luminal acidification [22–24].
Acidification of the epididymal lumen, coupled with low  concentrations, is required to keep spermatozoa quiescent before ejaculation [25–27]. Bicarbonate concentration must be maintained low as spermatozoa express an adenylyl cyclase (ADCY10, previous symbol SACY) that is directly activated by bicarbonate [28–30]. ADCY10 activation by bicarbonate leads to a rise in intracellular cAMP and subsequent activation of protein kinase A. This leads to an increase in the tyrosine phosphorylation of several sperm proteins that are important for the process of capacitation and in initiating motility [31–33].
concentrations, is required to keep spermatozoa quiescent before ejaculation [25–27]. Bicarbonate concentration must be maintained low as spermatozoa express an adenylyl cyclase (ADCY10, previous symbol SACY) that is directly activated by bicarbonate [28–30]. ADCY10 activation by bicarbonate leads to a rise in intracellular cAMP and subsequent activation of protein kinase A. This leads to an increase in the tyrosine phosphorylation of several sperm proteins that are important for the process of capacitation and in initiating motility [31–33].
There are several transepithelial acid/base transport systems expressed in the epididymis that contribute to maintaining low luminal pH and bicarbonate concentrations, many of which are shared with the efferent ducts and kidney [34]. Sperm leave the testis in a solution that is high in bicarbonate. The proximal region of the epididymis expresses an apically localized sodium hydrogen exchanger, SLC9A3 [35, 36], which functions to pump out protons into the lumen, acidifying the fluid as well as providing a hydrogen ion for the conjugation of  by membrane-localized carbonic anhydrases, forming H2O and CO2. In the proximal region, the primary carbonic anhydrase present on the luminal surface is CAR14 (CA-XIV). The newly formed CO2 can readily diffuse into the epithelial cell, where it is hydrated by CAR2 to form H+ and
by membrane-localized carbonic anhydrases, forming H2O and CO2. In the proximal region, the primary carbonic anhydrase present on the luminal surface is CAR14 (CA-XIV). The newly formed CO2 can readily diffuse into the epithelial cell, where it is hydrated by CAR2 to form H+ and  . Intracellular
. Intracellular  diffuses passively across the basolateral membrane via the Na+-
diffuses passively across the basolateral membrane via the Na+- cotranspoter SLC4A4 (NBC1 or NBCe1) or the anion exchanger SLC4A2 (AE2). In addition to proton secretion occurring via SLC9A3 in principal cells, vacuolar-H+ATPase (ATP6V0A1), an enzyme that couples ATP hydrolysis to proton pumping across membranes, is active in the clear and narrow cells of the epididymis [37–39]. Thus, bicarbonate reabsorption and net luminal acidification is achieved by the coordinated functions of ATP6V0A1, SLC9As, carbonic anhydrases, and bicarbonate transporters [40].
cotranspoter SLC4A4 (NBC1 or NBCe1) or the anion exchanger SLC4A2 (AE2). In addition to proton secretion occurring via SLC9A3 in principal cells, vacuolar-H+ATPase (ATP6V0A1), an enzyme that couples ATP hydrolysis to proton pumping across membranes, is active in the clear and narrow cells of the epididymis [37–39]. Thus, bicarbonate reabsorption and net luminal acidification is achieved by the coordinated functions of ATP6V0A1, SLC9As, carbonic anhydrases, and bicarbonate transporters [40].
We hypothesized that the previously described defect in efferent duct fluid reabsorption would lead to alterations in the epididymal fluid milieu, negatively impacting sperm function. Results here show that the Esr1KO epididymis fails to properly acidify the luminal milieu because of defects in the expression of specific acid/base regulators. This leads to defects in sperm intracellular pH and motility that can be partially rescued by bypassing the consequences of the abnormal epididymal milieu.
MATERIALS AND METHODS
Animals and Tissue Preparation
The present study used homozygous Esr1KO males produced by ZP3-Cre-mediated excision of exon 3, which generates a fully disrupted Esr1 mutant that lacks all transcripts downstream of exon 2 [41, 42]. The phenotype of these males has been reported to be indistinguishable from that of the original Esr1KO null mutation [1, 3, 41, 43]. Esr1KO male mice are infertile, whereas heterozygous mice show normal fertility and sperm motility. Comparisons were made between Esr1KO and heterozygous (HET) or WT littermates, as indicated. Only sexually mature males were used in this study, ranging from 7 to 13 wk of age. For each analysis, at least three age-matched mice of each genotype were used. The mice were euthanized by cervical dislocation. For CASA analysis, mice were lightly anesthetized via carbon dioxide inhalation prior to cervical dislocation. All experiments involving animals were conducted according to Emory University, University of Illinois, and University of Kentucky institutional standards for the care and use of experimental animals. Epididymides were dissected clean of connective tissue and subdissected into four regions: the initial segment and the remaining caput, corpus, and cauda.
Sperm Collection
All reagents were purchased from Sigma Aldrich unless noted otherwise. Sperm were collected and incubated in either a modified Krebs-Ringer buffer (dmKBRT) [44] or Medium B (127 mM NaCl, 5.3 mM KCl, 18.2 mM HEPES, pH 7.4). Initial segment and caput sperm were collected by puncturing tissue with a 27½-gauge needle and gently expressing fluid out of the perforations into the collection media. Cauda sperm were collected by shredding tissue in the appropriate media, as indicated, and filtering through a nylon mesh (03–35/16 Nitex; Sefar Filtration Inc.). In some instances, the highly motile cauda sperm (“swim out”) were collected by making three to five small incisions into cauda tissue and collecting the sperm after 5 min of incubation at 37°C. The remaining cauda sperm were collected by shredding the cauda tissue, triturating with a transfer pipette, and filtering through nylon mesh.
Single-Dimension Electrophoresis, Western Blot Analysis, and Quantification
Epididymal fluid was collected by pooling the indicated anatomical regions from one animal and expressing the fluid into a small volume of Medium B. Sperm and cell debris were pelleted, and the supernatant was saved. Epididymal tissue lysates were prepared from tissue that was pooled and snap frozen in liquid nitrogen and stored at −80°C. Tissue was homogenized in 0.5 ml of lysis buffer previously described [45]. The membrane-bound vesicle fraction was isolated by collecting fluid as described previously in 200 μl of Medium B with 1 mM EDTA. Sperm and cell debris were removed by centrifugation. To separate the whole membrane fraction from total luminal fluid, the sample was subjected to ultracentrifugation at 200 000 × g for 1 h, as previously described [46]. The supernatant was collected as the soluble fraction. The remaining pellet was sonicated in a buffer containing 50 μl of Medium B supplemented with 1 mM EDTA and 0.1% Triton X-100.
Protein concentration was determined using the RC DC Protein Assay (Bio-Rad Laboratories), and equal protein loads were resolved by SDS-PAGE using Tris-HCl precast gels (Criterion System; Bio-Rad Laboratories) after heating at 90°C for 8 min in the presence of DTT. Gels were silver-stained for protein visualization. Resolved proteins were transferred onto Immobilon-P membranes, PVDF (Millipore) and blocked with 5% (w/v) nonfat dry milk powder for 1 h at room temperature. The membranes were probed using the following commercially available antibodies: rabbit anti-CA-II (CAR2) raised against human erythrocyte CA-II (Chemicon); mouse anti-CA-XIV (CAR14) raised against amino acids 194–301 (BD Biosciences); rabbit anti-NHE3 (SLC9A3) raised against amino acids 665–834 (Santa Cruz Biotechnology); rabbit anti-V-ATPase (ATP6V0A1) raised against amino acids 334–513 (Santa Cruz Biotechnology); hamster monoclonal anti-SED1/MFG-E8, which reacts with all SED1/MFG-E8 isoforms (MBL International); and mouse anti-α-tubulin (Sigma). Rabbit antibodies against CRISP1/CAP-A [47], ClC-5 (CLCN5) (C2) [48], and NBC1/NBCe1 (SLC4A4) [49] were kindly provided by the cited investigators. All secondary HRP-conjugated antibodies were used at a 1:25 000 dilution (Santa Cruz Biotechnology). Peroxidase-bound protein bands were visualized using the ECL or ECL-Plus method (GE Healthcare Life Sciences). To check for equal protein loads, blots were incubated in stripping buffer (0.25 M glycine, 0.5% SDS, pH 2.5) and reprobed using an antibody against α-tubulin. Intensities of bands on films were quantified using spot densitometer software (FluorChem SP; Alpha Innotech Corp.).
Determination of Luminal pH
Luminal fluid pH was determined as previously described with minor modifications [45]. Single pairs of age-matched control and Esr1KO mice were sacrificed by cervical dislocation, and epididymides were removed and subdissected by anatomical region (initial segment, caput, and cauda). Tissue was gently manipulated to exude luminal contents through the cut side of tissue onto Hydrion pH paper (Microfine; Micro Essential Laboratory). The pH was determined by two independent investigators by comparing the expressed luminal fluid to standard phosphate-buffered solutions with pH 6.4–7.4 as well as to the standards provided with the litmus kit. The recorded measurements from each investigator were within 0.1 units of each other, and at least five independent samples of luminal fluid from each genotype and each region were assayed. Means were compared using an unpaired Welch-corrected Student t-test; differences were considered significant when P ≤ 0.05.
Determination of Intracellular Sperm pH
Intracellular sperm pH (pHi) was measured using 2,7-bicarboxyethyl-5,6-carboxyfluorescein-acetoxymethyl ester (BCECF-AM; Invitrogen Molecular Probes), a fluorophore that diffuses freely through the plasma membrane. In the cell, BCECF-AM is hydrolyzed by nonspecific esterases, releasing BCECF, which is charged and therefore retained within the cytoplasm. Once inside the cell, BCECF has a pH-dependent fluorescent excitation profile. The utility of BCECF to assess the intracellular pH of mouse sperm has been previously reported [50, 51] and verified in this study by incubating control sperm in buffers of varying pH in the presence of the K+/H+ ionophore, nigericin (Invitrogen Molecular Probes), which causes rapid equilibration of intracellular and extracellular pH in the presence of a depolarizing concentration of extracellular K+. Intracellular sperm fluorescence intensity was confirmed to be proportional to pH within a range of 6.6–8.0.
The previously published protocol for the use of BCECF-AM in mouse sperm was modified slightly [50, 51]. Briefly, BCECF-AM was dissolved in dimethylsulfoxide (DMSO; 1 mM stock solution) and stored in aliquots of 20 μl at −20°C. Spermatozoa recovered from the initial segment, caput, and cauda in Medium B and were incubated with BCECF-AM (final concentration: 4 μM) in the dark (37°C) for 30 min and then centrifuged (203 × g) for 5 min. The supernatant was discarded, and the sperm were resuspended in appropriate media. Immediately prior to imaging, the sperm suspension was spread on a ProbeOn Plus microscope slide (Fisher Scientific) and mounted with a cover slip. The spermatozoa solution was excited at 488 nm, and the fluorescence emission was recorded with band pass of 505–550 nm. All scans were taken within 30 sec of each other. Using the LSM 510 software (Zeiss), the images were processed by selecting the region of interest (sperm head) and subtracting the background fluorescence. The analysis software provided the mean pixel intensity value for each individual sperm.
BCECF statistical analysis.
For each assay, sperm were collected from the initial segment, caput, and cauda epididymis of age-matched genotype pairs (WT/HET vs. Esr1KO), and their pixel intensities were recorded. Mean intensities of sperm from each region were compared between genotypes (Esr1KO vs. WT/HET). Although within each pair of mice assayed on a given day the fluorescence intensity was consistently elevated in the Esr1KO sperm populations, we pooled data from multiple assays to acquire a statistically significant sample set. Fluorescent intensity was standardized between assays from different days by subtracting the experimental mean for all sperm from each observation and dividing this value by the experimental standard deviation for all sperm. All analyses utilized Systat 12 (Systat, Inc.). Data were modeled using a linear mixed-model analysis to account for observations that are correlated. The data were modeled by declaring each experiment and animal a random effect, therefore taking into account the multiple measurements of sperm taken from one animal. To assess the effect of genotype in each region of the epididymis, we used the computed type 3 analysis of variance. The Kruskal-Wallis test and the Kolomogorov-Smirnov two-sample test were used to examine differences between distributions. Significance was determined as P ≤ 0.05.
Motility Induction with cAMP
Caput and cauda epididymal sperm were collected in dmKBRT as described previously. Aliquots of the recovered sperm suspension were placed into a treatment or control droplet (v:v 1:1). The treatment droplet consisted of db-cAMP (dibutyryl-cyclic adenosine monophosphate sodium salt) dissolved in DMSO (final concentration: 1 mM) and the phosphodiesterase inhibitor caffeine (final concentration: 15 mM). The control droplet consisted of an equivalent volume of DMSO in dmKBRT. Treated and control samples were placed in a humidified incubator (37°C, 5% CO2) for 25 min. After incubation, a 15-μl aliquot of the treated and control samples was transferred into each of two compartments on a glass cannula slide for computer-assisted sperm analysis (CASA) using the integrated visual optical system (IVOS) motility analyzer (Hamilton-Thorne Research, Inc.). Thirty frames were acquired at a frame rate of 60 Hz. The operational settings of the IVOS were as follows: minimum contrast (40) and size (four pixels), gate thresholds 0.38/1.65 for intensity and 0.42/2.34 for size, static elongation 0/75, progressive minimum path velocities of sperm (VAP) 50 μm/sec, straightness threshold 50%, and magnification 0.82.
CASA statistical analysis.
Parameters of motility were analyzed using a mixed model wherein litters and males nested within a litter were random factors. To assess the assumption of equal variances, the Levene test was performed. The resulting P-value of the Levene test was greater than 0.05 for all but two of the motility parameters (BCF and LIN). For all analyses, residuals were normally distributed based on visual inspection of probability plots. The Tukey honestly significant difference test was utilized to perform pairwise comparisons for all combinations of genotype and treatment for each motility parameter. Means were considered significantly different if P ≤ 0.05.
RESULTS
The Loss of Functional Esr1 Gene Results in Elevated Epididymal Fluid pH
Previous studies using the Esr1KO mouse model have demonstrated a decrease in Slc9a3 mRNA expression as well as reduced immunostaining of AQP1, AQP9, and CAR2 within the efferent duct epithelium [12, 13]. Another infertile mouse model, the Ros1 knockout male, also exhibits altered expression of several epithelial transporters and enzymes as well as a consequent increase in the luminal pH of the cauda epididymis [45]. Given these reports and the role that SLC9A3 plays in acid/base regulation and fluid homeostasis, we measured luminal pH from the initial segment, caput, and cauda for comparison between Esr1KO and control animals.
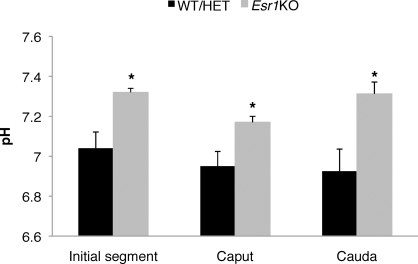
Fluid harvested from control animals displayed a slow acidification as it progressed through the lumen, from the proximal to the distal regions of the epididymis (Fig. 1; initial segment: 7.04 ± 0.08; caput: 6.95 ± 0.07; cauda: 6.93 ± 0.11). In contrast, the luminal contents of the Esr1KO initial segment was ∼0.30 units higher and remained more alkaline than control fluid in all epididymal segments measured (initial segment: 7.32 ± 0.02; caput: 7.17 ± 0.03; cauda: 7.31 ± 0.05).
FIG. 1.
The Esr1KO epididymis fails to acidify the luminal fluid. Epididymides were isolated and subdissected, and the pH of the luminal contents was measured using an ex vivo assay. In control animals, the luminal pH decreases during epididymal transit, whereas it remains alkaline in the Esr1KO epididymis. In this regard, fluid isolated from the initial segment of Esr1KO animals was significantly more alkaline than controls (control: 7.04 ± 0.08, Esr1KO: 7.32 ± 0.02). This difference persists in the caput (control: 6.95 ± 0.07, Esr1KO: 7.17 ± 0.03) as well as in the cauda (control: 6.93 ± 0.11, Esr1KO: 7.31 ± 0.05). At least five independent samples of luminal fluid from each genotype and each region were assayed. Bars = SEM. WT, wild type; HET, heterozygote; *P ≤ 0.04.
The Esr1KO Epididymis Does Not Exhibit Global Defects in Protein Composition or Secretion
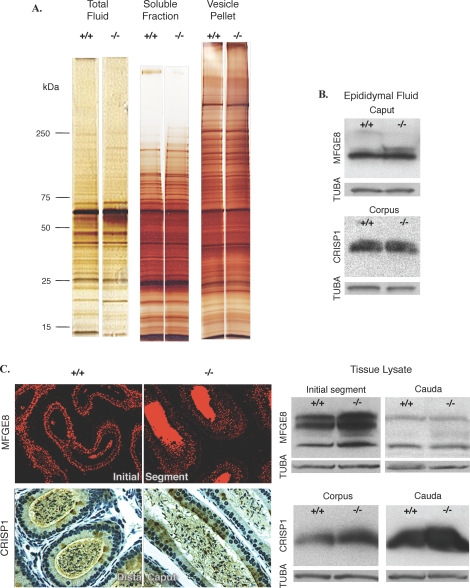
To determine if the failure of the epididymal fluid to acidify was due to global defects in epididymal function, we compared protein expression and secretion between Esr1KO and control epididymides. The total protein composition of luminal fluid collected from the proximal epididymis (caput and initial segment) was examined via single dimension electrophoresis and silver stain (Fig. 2A). Although not intended to detect small, minor differences in protein composition, the overall protein abundance, and profile, as judged by 1D SDS-PAGE, were similar between genotypes. For a closer examination of the epididymal secretions that occur in membrane-bound vesicles, known as epididymosomes, we further fractionated the luminal contents into membrane-bound and soluble secretions. Analysis of these fractions by 1D SDS-PAGE revealed that the soluble and membrane-bound secretions were fairly distinct, with the exception of polypeptides at ∼70, 40, 25, and 15 kDa that were common to both fractions (Fig. 2A). Similar to that seen with total luminal proteins, the relative abundance and profile of proteins in both the membrane-bound and the soluble fractions appeared to be indistinguishable between Esr1KO and control mice at this resolution.
FIG. 2.
Many epididymal proteins are expressed and localized normally in the Esr1KO epididymis. As a simple readout of overall epididymal function, total epididymal protein expression was compared between control and Esr1KO mice, as was the immunolocalization of MFGE8 and CRISP1, two epididymal secreted proteins important in sperm function. A) Total luminal fluid protein was separated by single-dimension electrophoresis and visualized by silver stain. At this resolution, the relative protein abundance and separation profiles appeared to be indistinguishable between genotypes. Furthermore, the protein composition of soluble and membrane-bound fractions were analyzed by SDS-PAGE and silver staining and shown to be similar between Esr1KO and control animals. B) Western blot analysis of MFGE8 and CRISP1 in epididymal fluid revealed similar levels in the Esr1KO and control caput and corpus, respectively. C) Immunolocalization of MFGE8 and CRISP1 in the Esr1KO epididymis was indistinguishable from that observed in control animals and similar to that previously reported [47, 52, 53]. Original magnification ×400. Furthermore, the relative abundance of CRISP and the known MFGE8 isoforms, as judged by Western blot analysis, was similar between genotypes. (A large bolus of unlabeled protein present in the epididymal fluid distorts the migration of the higher-molecular-weight MFGE8 isoform, which is easily seen in tissue lysates.) For all blots, α-tubulin, TUBA, serves as a loading control.
Even though the profile of total secreted proteins appeared similar in control and Esr1KO epididymides, we examined the expression of two specific proteins known to be essential for sperm maturation and fertilization: MFGE8 (SED1) and CRISP1 (Fig. 2, B and C). As previously reported, MFGE8 exists as multiple isoforms that are highly expressed in the principal cells of the initial segment, where it is concentrated in the Golgi region, apically localized punctate foci, and the epididymal lumen, a distribution that is characteristic of the vesicular secretory pathway [52]. The distribution for MFGE8 within the epididymal duct was found to be similar between control and Esr1KO genotypes (Fig. 2C).
CRISP1 expression has been documented in the mouse epididymal lumen from the corpus to the cauda regions, with prominent expression within the perinuclear area of corpus cells and the apical region of cauda cells [47, 53]. In addition to these regions, we found strong CRISP1 immunoreactivity within a subset of principal cells and within the luminal compartment of the distal caput (Fig. 2C). The immunocytochemical localization of CRISP1 in all segments of the epididymis was comparable between Esr1KO and WT tissues. Furthermore, the level of MFGE8 isoforms and CRISP1 expression within the epididymal tissue and luminal fluid, as judged by Western blot analysis, was also similar between genotypes (Fig. 2, B and C).
The Esr1KO Epididymis Has Reduced Expression of Several Proteins That Are Involved in Acid/Base Regulation
The increased luminal fluid pH in the Esr1KO epididymis is consistent with an inappropriate regulation of fluid homeostasis. We therefore considered possible mechanisms whereby loss of ESR1 may affect this aspect of epididymal physiology. Given its essential role in modifying the epididymal environment, our analysis focused on the initial segment of the epididymis.
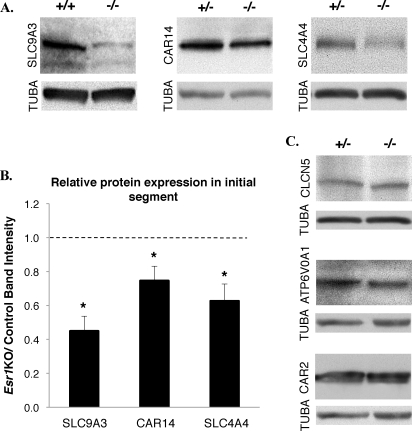
The composition of the epididymal luminal fluid is regulated by a number of ion transporters, membrane pumps/channels, and enzymes. We investigated a subset of these proteins with known expression in the mouse or rat epididymis and characterized the level of protein expression by Western blot analysis. Components of the regulatory pathway that are believed to be highly active within the principal cells, such as the sodium/hydrogen exchanger SLC9A3 [35, 36], sodium/bicarbonate cotransporter SLC4A4 [54], and CAR14 [55], all have significantly reduced expression in the Esr1KO initial segment when compared to control tissue (Fig. 3A). Quantification of all experiments showed that in comparison to control tissue, SLC9A3 was reduced by 50%, SLC4A4 by 35%, and CAR14 by 25% in the Esr1KO initial segment (Fig. 3B).
FIG. 3.
Reduced expression of cell-specific components involved in acid/base homeostasis. A) Western blot analysis of principle cell-specific regulatory proteins SLC9A3 (83 kDa), CAR14 (50 kDa), and SLC4A4 (130 kDa) showed reduced expression within the Esr1KO initial segment. α-tubulin, TUBA, serves as a loading control. B) Densitometric quantification of Western blots expressed as ratios of the control value revealed statistically significant reduction of SLC9A3, CAR14, and SLC4A4 in Esr1KO tissue. Histograms represent the mean ± SEM; *P ≤ 0.03. The dotted line represents equal levels of protein expression between Esr1KO and control animals. C) Other components involved in fluid regulation that are specifically expressed in narrow cells, such as ATP6V01 and CLCN5, or that are expressed by both principle and narrow cells, that is, CAR2, have similar levels of expression between genotypes. α-tubulin, TUBA, serves as a loading control.
In contrast to that seen in principle cells, enzymes and transporters specific to narrow cells, such as the chloride channel CLCN5 [48] and the ATP6V0A1 proton pump [38], showed similar expression in Esr1KO and control tissue (Fig. 3C). Furthermore, some components that participate in acid/base regulation are found in both principle and narrow cells, such as cytosolic CAR2. Although we occasionally observed decreased levels of CAR2 in the Esr1KO initial segment, its ubiquitous expression resulted in inconsistent findings, making interpretations difficult. Collectively, these findings suggest that the reduced expression of principal cell components SLC9A3, SLC4A4, and CAR14, individually or in combination, are responsible for the failure of the Esr1KO epididymis to appropriately acidify the luminal compartment.
Esr1KO Sperm Display Aberrant Intracellular pH During Epididymal Transit
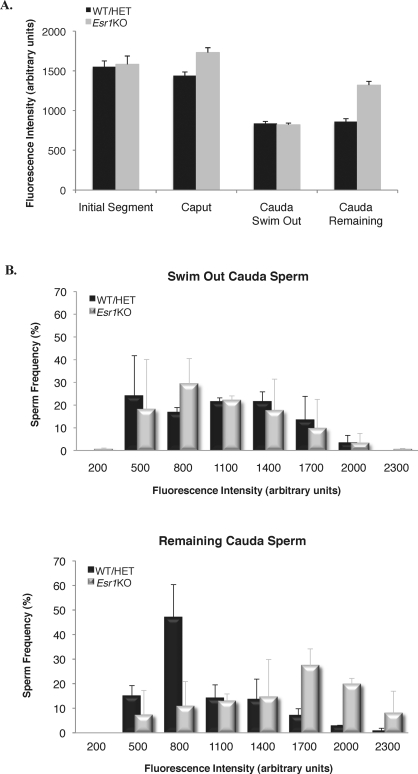
To investigate the effect of luminal pH on intracellular sperm pH (pHi), we utilized the pH-sensitive fluorophore BCECF, which has been used by others to assess mouse sperm pHi [50, 51]. Sperm from the initial segment, caput, and cauda epididymis were isolated and treated with BCECF, and their fluorescence intensity was measured (Fig. 4A). Our findings indicate that control sperm enter the epididymis with an alkaline pHi, which gradually acidifies as sperm transit to the distal regions of the epididymal duct. This decrease in intracellular pH mimics the slow acidification of the epididymal lumen. Similarly, Esr1KO sperm also possess an alkaline pHi as they enter the epididymis in the initial segment, but unlike that seen with control sperm, Esr1KO sperm fail to acidify their intracellular environment in the caput and remain alkaline.
FIG. 4.
Intracellular pH of Esr1KO sperm fails to acidify during epididymal transit. Sperm from the initial segment, caput, and cauda epididymis were loaded with BCECF, a pH-dependent fluorophore, and their fluorescence intensity was measured. A) Esr1KO and control sperm recovered from the initial segment have a similar fluorescent intensity. However, caput Esr1KO sperm show increased fluorescent intensity and pHi, relative to controls (P = 0.04). Cauda sperm were separated on the basis of motility, revealing that the overall mean fluorescent intensity of Esr1KO sperm that are sufficiently motile to swim out of the cauda is similar to that of control sperm, while the fluorescent intensity of the remaining nonmotile Esr1KO sperm is significantly increased. Presented are mean values ± SEM; *p ≤ 0.05. B) Frequency distributions of cauda sperm fluorescence intensity reveals that the relative pHi is similar between both control sperm populations and the Esr1KO swim-out sperm but is significantly more alkaline in the nonmotile Esr1KO cells.
Because of the previously described motility defect characteristic of Esr1KO cauda sperm [12], we evaluated the pHi of the most motile sperm, which are able to swim out of the cauda epididymal tissue, relative to those with compromised motility and that fail to swim out (Fig. 4B). Esr1KO sperm that were sufficiently motile to swim out of the cauda epididymides had a pHi that was statistically similar to that of WT cells. However, those sperm that failed to swim out of the cauda epididymis had an intracellular pH that was distinctly more alkaline, indicating that the difference in pHi seen in unfractionated Esr1KO cauda sperm is most prominent in the nonmotile sperm population.
Esr1KO Sperm Motility Defects Are Rescued by Treatment with Exogenous cAMP
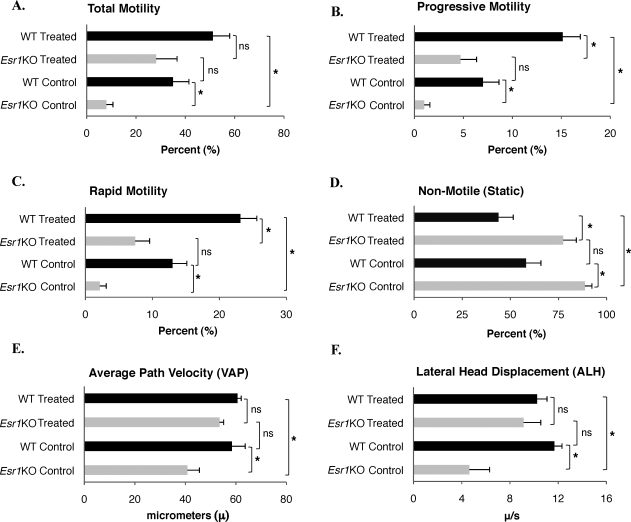
Since pH and bicarbonate ions are critical regulators of sperm adenylyl cyclase and consequent motility, we sought to bypass the abnormal Esr1KO luminal and intracellular pH by supplying Esr1KO sperm with a membrane-permeable cAMP analog in an attempt to recover motility deficits. We chose caput sperm for this analysis since other studies have shown that the severity of Esr1KO-associated sperm anomalies increases as sperm progress through the epididymal duct [56]. We reasoned, therefore, that cauda sperm may be irreversibly damaged and that caput sperm may be more amenable to pharmacological rescue of their motility defects. Utilizing CASA, we were able to obtain objective and quantitative descriptions of changes in sperm kinematic parameters in response to exogenous cAMP (Fig. 5). Caput Esr1KO sperm possess a significantly lower percent motility than WT cells (control motility: 35 ± 6.5%; Esr1KO motility: 8 ± 2.7%; Fig. 5A). CASA measurements indicate that both the raw numbers and the relative percentages of sperm classified as having progressive and rapid motility were significantly lower in the Esr1KO (Fig. 5, B and C), while those sperm classified as static were drastically increased (Fig. 5D). The data further indicate that the VAP and amplitudes of their lateral head displacement were all greatly reduced in Esr1KO sperm (Fig. 5, E and F).
FIG. 5.
Exogenous treatment with cAMP rescues Esr1KO sperm motility deficits. CASA measurements of caput sperm indicate that the raw numbers and relative percentages of overall sperm motility, as well as progressive and rapid motility, were significantly lower in the Esr1KO, whereas those sperm classified as static were drastically increased. The data also indicate that the path velocities of sperm (VAP) and amplitudes of their lateral head displacement (ALH) are both greatly reduced in the Esr1KO sperm. Treatment of Esr1KO sperm with a dp-cAMP cocktail significantly increased their total motility (A), progressive motility (B), and rapid motility (C) and decreased their static motility (D) to levels comparable to control cells. Similarly, the path velocities of sperm (VAP; E) and amplitudes of their lateral head displacement (ALH; F) were also increased to levels statistically similar to control cells following cAMP treatment. Although WT sperm generally showed stronger responses to exogenous cAMP treatment, the increase in Esr1KO total motility, ALH, and VAP induced by cAMP treatment were statistically similar to that seen in treated wild-type cells. *P ≤ 0.03; ns, not significant.
When Esr1KO sperm were treated with a cocktail of db-cAMP and caffeine, their motility and velocity parameters increased to levels statistically similar to that of WT control cells (Fig. 5, A–F). All six of the motility parameters that were analyzed as being statistically different under basal conditions recovered to normal control values after treatment (% motility, % progressive, % rapid, % static, average path velocity, and lateral head displacement). These results further argue that the defective motility seen in Esr1KO sperm is not intrinsic to the sperm but rather is a physiological manifestation of the abnormal luminal environment that is acquired during epididymal transit.
DISCUSSION
Results presented here indicate that the epididymal duct of the Esr1KO male is unable to properly regulate the luminal fluid milieu, consistent with previously reported findings of aberrant fluid regulation in Esr1KO efferent ducts. The efferent ducts and the initial segment of the epididymis play an integral role in reabsorbing testicular fluid and acidification of the luminal milieu. Although previous studies of the Esr1KO males have reported aberrant expression of critical regulators of fluid transport within the efferent duct epithelium [13], the downstream effects on epididymal function, luminal composition, and consequent sperm maturation had yet to be explored.
Spermatozoa acquire their ability to become motile and to fertilize an egg in the lumen of the epididymis. Given this important role, failure of the epididymis to provide an adequate environment for sperm maturation has profound effects on male fertility. One of the most critical features of the epididymal luminal fluid is that it is acidified to keep spermatozoa in a quiescent, immotile state while they undergo maturation and are stored in the epididymis. The principal and clear (or narrow) cells of the epididymal epithelium are able to regulate the luminal pH by utilizing various membrane pumps, channels, and transporter systems [40, 55, 57].
We show here that in contrast to control animals, the luminal pH of the Esr1KO fluid remains alkaline in all regions, indicating that these animals fail to properly acidify the epididymal lumen. Interestingly, the absolute difference in pH between the Esr1KO and control animals is similar to that reported using the same assay to assess luminal pH in the infertile Ros1 knockout males, a model that is also deficient in pH regulation [45].
To assess the mechanisms underlying the Esr1KO phenotype, we evaluated the expression of several transmembrane proteins that drive fluid reabsorption and acidification in the initial segment of the epididymis. Western blot analysis indicates that the Esr1KO initial segment has significantly reduced levels of SLC9A3, CAR14, and SLC4A4, all of which are thought to be highly active within the principal cells of this region. Other components of the pH regulatory pathway that are localized to the narrow cells are unaffected in Esr1KO. Furthermore, the global expression of proteins within the epididymal tissue, as well as the expression and localization of the principal cell-secreted proteins MFGE8 and CRISP1, appear to be indistinguishable between genotypes. Although the analysis of total protein composition is limited by the resolving capacity of single-dimension polyacrylamide gels, together these results suggest that the Esr1KO epididymal epithelium shows a relatively specific defect in its ability to properly regulate the composition of the luminal fluid.
A model for acidification within the initial segment has been previously proposed for the rat [40]. It is believed that the main sodium/hydrogen exchanger present in the initial segment, SLC9A3, is critical for bicarbonate reabsorption as well as for driving protons out of the cell. These luminal protons are then combined with bicarbonate ions under the activity of CAR14 present on the apical surface of principal cells in the initial segment. The activity of CAR14 produces CO2 that is hydrated by the cytosolic CAR2 to form H+ and bicarbonate. The protons recycle back into the lumen via SLC9A34, whereas  diffuses across the basolateral membrane via Na+/
diffuses across the basolateral membrane via Na+/ cotransporters. Given these proposed functions, it is reasonable to propose that the reduced abundance of SLC9A3 alone or in conjugation with SLC9A3 and CAR14 is likely responsible for the alkaline pH of Esr1KO epididymal fluid. Still unknown, however, is the mechanistic regulation of these components. The decreased transcription of SLC9A3 previously reported in the Esr1KO efferent ducts implies that this component is also under transcriptional control within the initial segment, where ESR1 is abundantly expressed [14]. Given that only a subset of fluid transport components was evaluated, we cannot rule out the involvement and alteration of other critical transporters that may contribute to this phenotype. Nevertheless, the finding that acid/base regulatory components expressed by the narrow cells of the initial segment are expressed normally in the Esr1KO epididymis indicates at least some degree of biochemical specificity underlying this phenotype.
cotransporters. Given these proposed functions, it is reasonable to propose that the reduced abundance of SLC9A3 alone or in conjugation with SLC9A3 and CAR14 is likely responsible for the alkaline pH of Esr1KO epididymal fluid. Still unknown, however, is the mechanistic regulation of these components. The decreased transcription of SLC9A3 previously reported in the Esr1KO efferent ducts implies that this component is also under transcriptional control within the initial segment, where ESR1 is abundantly expressed [14]. Given that only a subset of fluid transport components was evaluated, we cannot rule out the involvement and alteration of other critical transporters that may contribute to this phenotype. Nevertheless, the finding that acid/base regulatory components expressed by the narrow cells of the initial segment are expressed normally in the Esr1KO epididymis indicates at least some degree of biochemical specificity underlying this phenotype.
Despite the clear role of luminal acidification in regulating sperm maturation and respiration, it is less clear what role sperm intracellular pH plays in this process. It is assumed that sperm are able to actively regulate their pHi because they maintain their initial pH when exposed to an acidic environment [58]. However, the mechanisms that sperm utilize to regulate  concentration and pHi remain unknown. To our knowledge, this is the first report of relative sperm pHi as a function of transit through the epididymal duct. This analysis revealed that control sperm undergo gradual acidification as they proceed from the proximal regions of the epididymis to the distal cauda, much like the luminal fluid itself. In contrast, Esr1KO sperm recovered from the caput are significantly more alkaline than control cells, a finding that is reflective of the alkaline luminal environment. Interestingly, the luminal pH of all segments of the Esr1KO epididymis is more alkaline than controls, although the intracellular sperm pH is indistinguishable from WT in the initial segment and becomes more alkaline than controls only in the caput. This suggests that the relative pH changes that occur in the Esr1KO luminal fluid and sperm compartments are out of phase. The lack of synchrony between the luminal and intracellular pH could simply reflect the time required for normal sperm to begin intracellular acidification, which fails to occur in the Esr1KO, or, rather, reflect differences in assay methods.
concentration and pHi remain unknown. To our knowledge, this is the first report of relative sperm pHi as a function of transit through the epididymal duct. This analysis revealed that control sperm undergo gradual acidification as they proceed from the proximal regions of the epididymis to the distal cauda, much like the luminal fluid itself. In contrast, Esr1KO sperm recovered from the caput are significantly more alkaline than control cells, a finding that is reflective of the alkaline luminal environment. Interestingly, the luminal pH of all segments of the Esr1KO epididymis is more alkaline than controls, although the intracellular sperm pH is indistinguishable from WT in the initial segment and becomes more alkaline than controls only in the caput. This suggests that the relative pH changes that occur in the Esr1KO luminal fluid and sperm compartments are out of phase. The lack of synchrony between the luminal and intracellular pH could simply reflect the time required for normal sperm to begin intracellular acidification, which fails to occur in the Esr1KO, or, rather, reflect differences in assay methods.
The frequency distribution of sperm pHi in cells that swam out from the cauda epididymis was similar in both genotypes, whereas the relatively immotile Esr1KO sperm forcibly collected from the cauda were skewed to the alkaline range. Importantly, Esr1KO sperm with higher pHi are viable because only viable sperm retain BCECF in the cytoplasm. In control sperm, cytoplasmic alkalinization is known to play important regulatory roles at multiple stages of sperm function. On insemination, there is a rise in sperm pHi as sperm move from a quiescent to a motile and active state [25, 59, 60]; there is a second increase in pHi during acrosomal exocytosis [61–63]. It is likely that the first event is a consequence of increased  concentration in the female reproductive tract, a known activator of the cAMP pathway. In any event, a mechanistic relationship between the increased pHi and the motility defects of Esr1KO sperm cannot be made at this time since the relationship between sperm
concentration in the female reproductive tract, a known activator of the cAMP pathway. In any event, a mechanistic relationship between the increased pHi and the motility defects of Esr1KO sperm cannot be made at this time since the relationship between sperm  concentration and pHi is unknown. However, it is plausible that the more alkaline Esr1KO sperm represent a subpopulation that are in the process of undergoing spontaneous acrosome reactions since it has been reported that increased pH contributes to spontaneous acrosomal exocytosis [64], which is more prevalent in Esr1KO sperm than in control cells [56].
concentration and pHi is unknown. However, it is plausible that the more alkaline Esr1KO sperm represent a subpopulation that are in the process of undergoing spontaneous acrosome reactions since it has been reported that increased pH contributes to spontaneous acrosomal exocytosis [64], which is more prevalent in Esr1KO sperm than in control cells [56].
Despite these defects in luminal and intracellular pH, Esr1KO sperm retain the ability to respond to cAMP as revealed by CASA. Evaluation of motility parameters showed that Esr1KO caput sperm had significantly reduced overall motility, progressive motility, and other related parameters. In all cases, treatment with exogenous cAMP restored motility to levels observed in WT cells although not always to levels observed in WT cells treated with cAMP. This finding illustrates that Esr1KO sperm have the machinery to respond to cAMP despite their abnormal external and internal environment.
Nevertheless, compared to fertile mice, there is a significant change in the luminal and intracellular sperm pH in Esr1KO animals, which undoubtedly negatively impacts sperm maturation and function and is likely correlated with their infertility. For example, the transfer of proteins from epididymosomes, which are released by the initial segment epithelium, to sperm is favored at an acidic pH [65], and therefore the Esr1KO sperm would be at a disadvantage despite the appropriate expression and secretion of these proteins by the epididymis. Similarly, the activities of several enzymes that modify sperm surface proteins during epididymal transit are pH dependent, with highest specific activity at the acidic pH of the normal epididymis [66, 67]. The Esr1KO animal therefore serves as model to better understand the role of luminal and intercellular sperm pH in fertility.
Acknowledgments
We would like to thank Criss Hartzell for technical input; Paul Cooke, Indrani Bagchi, Mary Laws, and Daryl Meling for animal use; and Inyeong Choi, Kenneth Roberts, and Alan Yu for generously providing antibodies. This manuscript represents a portion of a thesis submitted by A.J. to the University of Illinois at Urbana-Champaign Graduate College as partial fulfillment of the requirements for a Ph.D.
Footnotes
Supported by National Institutes of Health grants F31 HD 54330 (A.J.), RO1 HD 23479 (B.D.S.), and P20 RR 15592 (C.K.).
REFERENCES
- Eddy EM, Washburn TF, Bunch DO, Goulding EH, Gladen BC, Lubahn DB, Korach KS.Targeted disruption of the estrogen receptor gene in male mice causes alteration of spermatogenesis and infertility. Endocrinology 1996; 137: 4796–4805. [DOI] [PubMed] [Google Scholar]
- Lubahn DB, Moyer JS, Golding TS, Couse JF, Korach KS, Smithies O.Alteration of reproductive function but not prenatal sexual development after insertional disruption of the mouse estrogen receptor gene. Proc Natl Acad Sci U S A 1993; 90: 11162–11166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hess RA, Bunick D, Lee KH, Bahr J, Taylor JA, Korach KS, Lubahn DB.A role for oestrogens in the male reproductive system. Nature 1997; 390: 509–512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahato D, Goulding EH, Korach KS, Eddy EM.Estrogen receptor-alpha is required by the supporting somatic cells for spermatogenesis. Mol Cell Endocrinol 2001; 178: 57–63. [DOI] [PubMed] [Google Scholar]
- Mahato D, Goulding EH, Korach KS, Eddy EM.Spermatogenic cells do not require estrogen receptor-alpha for development or function. Endocrinology 2000; 141: 1273–1276. [DOI] [PubMed] [Google Scholar]
- Lucas TF, Siu ER, Esteves CA, Monteiro HP, Oliveira CA, Porto CS, Lazari MF.17beta-estradiol induces the translocation of the estrogen receptors ESR1 and ESR2 to the cell membrane, MAPK3/1 phosphorylation and proliferation of cultured immature rat Sertoli cells. Biol Reprod 2008; 78: 101–114. [DOI] [PubMed] [Google Scholar]
- Robertson KM, O'Donnell L, Jones ME, Meachem SJ, Boon WC, Fisher CR, Graves KH, McLachlan RI, Simpson ER.Impairment of spermatogenesis in mice lacking a functional aromatase (cyp 19) gene. Proc Natl Acad Sci U S A 1999; 96: 7986–7991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ilio KY, Hess RA.Structure and function of the ductuli efferentes: a review. Microsc Res Tech 1994; 29: 432–467. [DOI] [PubMed] [Google Scholar]
- Hess RA.The Efferent Ductules: Structure and Functions. Robaire B, Hinton BT.The Epididymis: From Molecules to Clinical Practice New York:Kluwer Academic/Plenum Publishers;2002: 49–80. [Google Scholar]
- Hess RA.Estrogen in the adult male reproductive tract: a review. Reprod Biol Endocrinol 2003; 1: 52 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hess RA, Bunick D, Lubahn DB, Zhou Q, Bouma J.Morphologic changes in efferent ductules and epididymis in estrogen receptor-alpha knockout mice. J Androl 2000; 21: 107–121. [PubMed] [Google Scholar]
- Ruz R, Gregory M, Smith CE, Cyr DG, Lubahn DB, Hess RA, Hermo L.Expression of aquaporins in the efferent ductules, sperm counts, and sperm motility in estrogen receptor-alpha deficient mice fed lab chow versus casein. Mol Reprod Dev 2006; 73: 226–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Q, Clarke L, Nie R, Carnes K, Lai LW, Lien YH, Verkman A, Lubahn D, Fisher JS, Katzenellenbogen BS, Hess RA.Estrogen action and male fertility: roles of the sodium/hydrogen exchanger-3 and fluid reabsorption in reproductive tract function. Proc Natl Acad Sci U S A 2001; 98: 14132–14137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Q, Nie R, Prins GS, Saunders PT, Katzenellenbogen BS, Hess RA.Localization of androgen and estrogen receptors in adult male mouse reproductive tract. J Androl 2002; 23: 870–881. [PubMed] [Google Scholar]
- Hermo L, Oko R, Morales CR.Secretion and endocytosis in the male reproductive tract: a role in sperm maturation. Int Rev Cytol 1994; 154: 106–189. [PubMed] [Google Scholar]
- Hinton BT, Palladino MA, Rudolph D, Labus JC.The epididymis as protector of maturing spermatozoa. Reprod Fertil Dev 1995; 7: 731–745. [DOI] [PubMed] [Google Scholar]
- Setchell BP, Sanchez-Partida LG, Chairussyuhur A.Epididymal constituents and related substances in the storage of spermatozoa: a review. Reprod Fertil Dev 1993; 5: 601–612. [DOI] [PubMed] [Google Scholar]
- Abou-Haila A, Fain-Maurel MA.Regional differences of the proximal part of mouse epididymis: morphological and histochemical characterization. Anat Rec 1984; 209: 197–208. [DOI] [PubMed] [Google Scholar]
- Turner TT, Bomgardner D, Jacobs JP, Nguyen QA.Association of segmentation of the epididymal interstitium with segmented tubule function in rats and mice. Reproduction 2003; 125: 871–878. [DOI] [PubMed] [Google Scholar]
- Hermo L.Structural features and functions of principal cells of the intermediate zone of the epididymis of adult rats. Anat Rec 1995; 242: 515–530. [DOI] [PubMed] [Google Scholar]
- Trasler JM, Hermo L, Robaire B.Morphological changes in the testis and epididymis of rats treated with cyclophosphamide: a quantitative approach. Biol Reprod 1988; 38: 463–479. [DOI] [PubMed] [Google Scholar]
- Flickinger CJ, Herr JC, Klotz KL.Immunocytochemical localization of the major glycoprotein of epididymal fluid from the cauda in the epithelium of the mouse epididymis. Cell Tissue Res 1988; 251: 603–610. [DOI] [PubMed] [Google Scholar]
- Hermo L, Dworkin J, Oko R.Role of epithelial clear cells of the rat epididymis in the disposal of the contents of cytoplasmic droplets detached from spermatozoa. Am J Anat 1988; 183: 107–124. [DOI] [PubMed] [Google Scholar]
- Vierula ME, Rankin TL, Orgebin-Crist MC.Electron microscopic immunolocalization of the 18 and 29 kilodalton secretory proteins in the mouse epididymis: evidence for differential uptake by clear cells. Microsc Res Tech 1995; 30: 24–36. [DOI] [PubMed] [Google Scholar]
- Acott TS, Carr DW.Inhibition of bovine spermatozoa by caudal epididymal fluid: II. Interaction of pH and a quiescence factor. Biol Reprod 1984; 30: 926–935. [DOI] [PubMed] [Google Scholar]
- Carr DW, Usselman MC, Acott TS.Effects of pH, lactate, and viscoelastic drag on sperm motility: a species comparison. Biol Reprod 1985; 33: 588–595. [DOI] [PubMed] [Google Scholar]
- Hinton BT, Palladino MA.Epididymal epithelium: its contribution to the formation of a luminal fluid microenvironment. Microsc Res Tech 1995; 30: 67–81. [DOI] [PubMed] [Google Scholar]
- Chen Y, Cann MJ, Litvin TN, Iourgenko V, Sinclair ML, Levin LR, Buck J.Soluble adenylyl cyclase as an evolutionarily conserved bicarbonate sensor. Science 2000; 289: 625–628. [DOI] [PubMed] [Google Scholar]
- Pastor-Soler N, Beaulieu V, Litvin TN, Da Silva N, Chen Y, Brown D, Buck J, Levin LR, Breton S.Bicarbonate-regulated adenylyl cyclase (sAC) is a sensor that regulates pH-dependent V-ATPase recycling. J Biol Chem 2003; 278: 49523–49529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinclair ML, Wang XY, Mattia M, Conti M, Buck J, Wolgemuth DJ, Levin LR.Specific expression of soluble adenylyl cyclase in male germ cells. Mol Reprod Dev 2000; 56: 6–11. [DOI] [PubMed] [Google Scholar]
- Visconti PE, Bailey JL, Moore GD, Pan D, Olds-Clarke P, Kopf GS.Capacitation of mouse spermatozoa. I. Correlation between the capacitation state and protein tyrosine phosphorylation. Development 1995; 121: 1129–1137. [DOI] [PubMed] [Google Scholar]
- Visconti PE, Moore GD, Bailey JL, Leclerc P, Connors SA, Pan D, Olds-Clarke P, Kopf GS.Capacitation of mouse spermatozoa. II. Protein tyrosine phosphorylation and capacitation are regulated by a cAMP-dependent pathway. Development 1995; 121: 1139–1150. [DOI] [PubMed] [Google Scholar]
- Lin M, Lee YH, Xu W, Baker MA, Aitken RJ.Ontogeny of tyrosine phosphorylation-signaling pathways during spermatogenesis and epididymal maturation in the mouse. Biol Reprod 2006; 75: 588–597. [DOI] [PubMed] [Google Scholar]
- Hamm LL, Alpern RJ.Cellular Mechanisms of renal tubular acidification. Seldin DW, Giebisch G.The Kidney, Physiology and Pathophysiology Philadelphia:Lippincott Williams & Wilkins;2000: 1935–1979. [Google Scholar]
- Bagnis C, Marsolais M, Biemesderfer D, Laprade R, Breton S.Na+/H+-exchange activity and immunolocalization of NHE3 in rat epididymis. Am J Physiol Renal Physiol 2001; 280: F426–F436. [DOI] [PubMed] [Google Scholar]
- Pushkin A, Clark I, Kwon TH, Nielsen S, Kurtz I.Immunolocalization of NBC3 and NHE3 in the rat epididymis: colocalization of NBC3 and the vacuolar H+-ATPase. J Androl 2000; 21: 708–720. [PubMed] [Google Scholar]
- Da Silva N, Shum WW, Breton S.Regulation of vacuolar proton pumping ATPase-dependent luminal acidification in the epididymis. Asian J Androl 2007; 9: 476–482. [DOI] [PubMed] [Google Scholar]
- Hermo L, Adamali HI, Andonian S.Immunolocalization of CA II and H+ V-ATPase in epithelial cells of the mouse and rat epididymis. J Androl 2000; 21: 376–391. [PubMed] [Google Scholar]
- Pietrement C, Sun-Wada GH, Silva ND, McKee M, Marshansky V, Brown D, Futai M, Breton S.Distinct expression patterns of different subunit isoforms of the V-ATPase in the rat epididymis. Biol Reprod 2006; 74: 185–194. [DOI] [PubMed] [Google Scholar]
- Pastor-Soler N, Pietrement C, Breton S.Role of acid/base transporters in the male reproductive tract and potential consequences of their malfunction. Physiology (Bethesda) 2005; 20: 417–428. [DOI] [PubMed] [Google Scholar]
- Dupont S, Krust A, Gansmuller A, Dierich A, Chambon P, Mark M.Effect of single and compound knockouts of estrogen receptors alpha (ERalpha) and beta (ERbeta) on mouse reproductive phenotypes. Development 2000; 127: 4277–4291. [DOI] [PubMed] [Google Scholar]
- Gieske MC, Kim HJ, Legan SJ, Koo Y, Krust A, Chambon P, Ko C.Pituitary gonadotroph estrogen receptor-alpha is necessary for fertility in females. Endocrinology 2008; 149: 20–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen M, Hsu I, Wolfe A, Radovick S, Huang K, Yu S, Chang C, Messing EM, Yeh S.Defects of prostate development and reproductive system in the estrogen receptor-alpha null male mice. Endocrinology 2009; 150: 251–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodeheffer C, Shur BD.Sperm from beta1,4-galactosyltransferase I-null mice exhibit precocious capacitation. Development 2004; 131: 491–501. [DOI] [PubMed] [Google Scholar]
- Yeung CH, Breton S, Setiawan I, Xu Y, Lang F, Cooper TG.Increased luminal pH in the epididymis of infertile c-ros knockout mice and the expression of sodium-hydrogen exchangers and vacuolar proton pump H+-ATPase. Mol Reprod Dev 2004; 68: 159–168. [DOI] [PubMed] [Google Scholar]
- Gatti JL, Metayer S, Belghazi M, Dacheux F, Dacheux JL.Identification, proteomic profiling, and origin of ram epididymal fluid exosome-like vesicles. Biol Reprod 2005; 72: 1452–1465. [DOI] [PubMed] [Google Scholar]
- Roberts KP, Ensrud KM, Hamilton DW.A comparative analysis of expression and processing of the rat epididymal fluid and sperm-bound forms of proteins D and E. Biol Reprod 2002; 67: 525–533. [DOI] [PubMed] [Google Scholar]
- Isnard-Bagnis C, Da Silva N, Beaulieu V, Yu AS, Brown D, Breton S.Detection of ClC-3 and ClC-5 in epididymal epithelium: immunofluorescence and RT-PCR after LCM. Am J Physiol Cell Physiol 2003; 284: C220–C232. [DOI] [PubMed] [Google Scholar]
- Bevensee MO, Schmitt BM, Choi I, Romero MF, Boron WF.An electrogenic Na(+)-HCO(−)(3) cotransporter (NBC) with a novel COOH-terminus, cloned from rat brain. Am J Physiol Cell Physiol 2000; 278: C1200–C1211. [DOI] [PubMed] [Google Scholar]
- Hernandez-Gonzalez EO, Sosnik J, Edwards J, Acevedo JJ, Mendoza-Lujambio I, Lopez-Gonzalez I, Demarco I, Wertheimer E, Darszon A, Visconti PE.Sodium and epithelial sodium channels participate in the regulation of the capacitation-associated hyperpolarization in mouse sperm. J Biol Chem 2006; 281: 5623–5633. [DOI] [PubMed] [Google Scholar]
- Zeng Y, Oberdorf JA, Florman HM.pH regulation in mouse sperm: identification of Na(+)-, Cl(−)-, and HCO3(−)-dependent and arylaminobenzoate-dependent regulatory mechanisms and characterization of their roles in sperm capacitation. Dev Biol 1996; 173: 510–520. [DOI] [PubMed] [Google Scholar]
- Raymond AS, Shur BD.A novel role for SED1 (MFG-E8) in maintaining the integrity of the epididymal epithelium. J Cell Sci 2009; 122: 849–858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eberspaecher U, Roosterman D, Kratzschmar J, Haendler B, Habenicht UF, Becker A, Quensel C, Petri T, Schleuning WD, Donner P.Mouse androgen-dependent epididymal glycoprotein CRISP-1 (DE/AEG): isolation, biochemical characterization, and expression in recombinant form. Mol Reprod Dev 1995; 42: 157–172. [DOI] [PubMed] [Google Scholar]
- Jensen LJ, Schmitt BM, Berger UV, Nsumu NN, Boron WF, Hediger MA, Brown D, Breton S.Localization of sodium bicarbonate cotransporter (NBC) protein and messenger ribonucleic acid in rat epididymis. Biol Reprod 1999; 60: 573–579. [DOI] [PubMed] [Google Scholar]
- Hermo L, Chong DL, Moffatt P, Sly WS, Waheed A, Smith CE.Region- and cell-specific differences in the distribution of carbonic anhydrases II, III, XII, and XIV in the adult rat epididymis. J Histochem Cytochem 2005; 53: 699–713. [DOI] [PubMed] [Google Scholar]
- Joseph A, Shur BD, Ko C, Chambon P, Hess RA.Epididymal hypo-osmolality induces abnormal sperm morphology and function in the estrogen receptor alpha knockout mouse. Biol Reprod 2010; 82: 958–967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shum WW, Da Silva N, Brown D, Breton S.Regulation of luminal acidification in the male reproductive tract via cell-cell crosstalk. J Exp Biol 2009; 212: 1753–1761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babcock DF, Pfeiffer DR.Independent elevation of cytosolic [Ca2+] and pH of mammalian sperm by voltage-dependent and pH-sensitive mechanisms. J Biol Chem 1987; 262: 15041–15047. [PubMed] [Google Scholar]
- Wong PY, Lee WM, Tsang AY.The effects of extracellular sodium on acid release and motility initiation in rat caudal epididymal spermatozoa in vitro. Exp Cell Res 1981; 131: 97–104. [DOI] [PubMed] [Google Scholar]
- Hamamah S, Gatti JL.Role of the ionic environment and internal pH on sperm activity. Hum Reprod 1998; 13(suppl 4):20–30. [DOI] [PubMed] [Google Scholar]
- Lee MA, Storey BT.Evidence for plasma membrane impermeability to small ions in acrosome-intact mouse spermatozoa bound to mouse zonae pellucidae, using an aminoacridine fluorescent pH probe: time course of the zona-induced acrosome reaction monitored by both chlortetracycline and pH probe fluorescence. Biol Reprod 1985; 33: 235–246. [DOI] [PubMed] [Google Scholar]
- Florman HM, Tombes RM, First NL, Babcock DF.An adhesion-associated agonist from the zona pellucida activates G protein-promoted elevations of internal Ca2+ and pH that mediate mammalian sperm acrosomal exocytosis. Dev Biol 1989; 135: 133–146. [DOI] [PubMed] [Google Scholar]
- Working PK, Meizel S.Correlation of increased intraacrosomal pH with the hamster sperm acrosome reaction. J Exp Zool 1983; 227: 97–107. [DOI] [PubMed] [Google Scholar]
- Hyne RV, Garbers DL.Requirement of serum factors for capacitation and the acrosome reaction of guinea pig spermatozoa in buffered medium below pH 7.8. Biol Reprod 1981; 24: 257–266. [DOI] [PubMed] [Google Scholar]
- Sullivan R, Frenette G, Girouard J.Epididymosomes are involved in the acquisition of new sperm proteins during epididymal transit. Asian J Androl 2007; 9: 483–491. [DOI] [PubMed] [Google Scholar]
- Marengo SR.Maturing the sperm: unique mechanisms for modifying integral proteins in the sperm plasma membrane. Anim Reprod Sci 2008; 105: 52–63. [DOI] [PubMed] [Google Scholar]
- Tulsiani DR.Glycan modifying enzymes in luminal fluid of rat epididymis: are they involved in altering sperm surface glycoproteins during maturation? Microsc Res Tech 2003; 61: 18–27. [DOI] [PubMed] [Google Scholar]