Abstract
Healthy females outperform males on face recognition. Relative to healthy individuals, schizophrenia patients are impaired on face perception tasks. Yet, it is unclear whether the female advantage found in healthy controls is preserved in females with schizophrenia. In the present study, we compared male and female patients and healthy controls on two basic face perception tasks – detection and identity discrimination. In the detection task, subjects located an upright or inverted line-drawn face (or a tree) embedded within a larger line-drawing. In the identity discrimination task, subjects determined which of two side-by-side face images matched an earlier presented face image. Healthy females were significantly more accurate than healthy males on face detection, but not on identity discrimination. However, female patients were not more accurate than male patients on either task. On both upright face detection and face identity discrimination, healthy controls significantly outperformed patients. Patients’ performance on face detection was closely associated with tree detection and IQ scores, as well as level of psychosis. This pattern of results suggests that a female advantage in basic face perception is no longer available in schizophrenia, and that this absence may be related to a generalized deficit factor, which acts to level performance across sexes, and putative changes in sex-related neurobiological differences associated with schizophrenia.
Keywords: Sex Difference, Face Perception, Visual Processing, Generalized Deficit
1. Introduction
Face perception plays a foundational role in one’s ability to engage in meaningful social exchange (Haxby et al., 2002). Schizophrenia patients are deficient at face perception tasks, including face detection (Chen et al., 2008; Zivotofsky et al., 2008), facial identity discrimination (Shin et al., 2008; Chen et al., 2009), and facial emotion recognition (Walker et al., 1984; Heimberg et al., 1992). The way in which these face perceptual impairments impact the social lives of persons with schizophrenia is a current area of investigation (e.g. Green et al., 2007).
Healthy females are better than healthy males at affective (Miura, 1993; McClure, 2000) and non-affective (Lewin and Herlitz, 2002; Rehnman and Herlitz, 2007; McBain et al., 2009) face recognition.. While schizophrenia patients generally underperform on such tasks, it is not entirely clear whether the female advantage found in healthy controls persists in this patient population. The only study which has investigated along these lines (Scholten et al., 2005) found that female schizophrenia patients were better than male patients at recognizing negative facial emotions such as anger and disgust, but not at recognizing positive emotions such as happiness. However, it is unknown at present whether this female advantage in affective face recognition is underpinned by a general perceptual advantage in non-affective face recognition, or is instead specific to affective processing.
The relationship between affective and non-affective face recognition is complex. On the one hand, the two domains appear to be dissociable. For example, there have been several case studies which report individuals who can only process affective or non-affective face recognition (Humphreys et al., 1993; Duchaine et al., 2003); this suggests the two domains may operate independently. On the other hand, perception in one of these domains of face recognition has the ability to influence perception in the other domain (Schyns and Oliva, 1999; Calder and Young, 2005), as such indicating the existence of a bidirectional relationship between them.
A comparison of male and female schizophrenia patients’ performance on non-affective aspects of face recognition would provide indication as to whether the female patient advantage in affective face perception is related to basic processing of face information. In the present study, we compared performance of male and female schizophrenia patients and male and female healthy controls on two non-affective face processing tasks: detection and identity discrimination. Face detection is considered the first stage in face processing, during which a face is recognized as ‘a face,’ while facial identity discrimination is the process by which an individual picks out one particular facial identity from others (Bruce and Young, 1986). In order to avoid the influence of affective content on performance, both tasks utilized face images exhibiting only neutral facial expressions. Additionally, in order to experimentally dissociate visual processing of faces from other visual objects, we employed inverted face detection and tree detection tasks as comparison perceptual measures.
Based upon a recent study of ours which found a female advantage in healthy controls on face detection and facial identity discrimination tasks (McBain et al., 2009), we predicted an overall difference between genders on these tasks, although the female advantage may be diminished in the patient group due to the greater performance variability which often characterizes patient performance. Additionally, based upon the findings of previous studies of ours (Chen et al., 2008; Chen et al., 2009), we predicted a significant group difference between patients and controls.
2. Methods
2.1. Subjects
Forty-one patients (20 females) and 62 controls (35 females) participated in face detection, the main task. For the comparison tasks, a subsample of 41 patients and 39 controls completed inverted face detection, while 35 patients and 38 controls completed tree detection. A subsample of 33 patients (17 females) and 39 controls (19 females) also completed the facial identity discrimination task. The participants were recruited through advertisements posted in the McLean Hospital campus and local community. Inclusion criteria for both groups were: (1) age between 21–60 years-old, (2) verbal IQ greater than 70, (3) visual acuity of at least 20/30, (4) no recent history of drug or alcohol abuse (i.e., within the past six months), and (5) no history of organic brain disease. Intelligence quotient was assessed using the revised version of the Wechsler Adult Intelligence Scale (Wechsler, 1981). Visual acuity was measured using the Rosenbaum Pocket Vision Screener.
Patients were diagnosed with either schizophrenia (n=17) or schizoaffective disorder (n=24), based on the SCID-IV and a review of all available medical records (First et al., 2002). Within the patient population, there was a similar number of male and female subjects diagnosed with schizophrenia (8 vs. 9), and a similar number diagnosed with schizoaffective disorder (12 vs. 12). Average illness duration was 19.9 years (SD=9.5). Thirty-three patients were on atypical antipsychotic medications, 3 were on typical medication, and 5 were on both types. The average chlorpromazine equivalent for patients was 666 mg (577 mg). Average scores on the positive, negative and general scales of the Positive and Negative Syndrome Scale (PANSS) were 17.1 (8.1), 14.3 (6.9), and 31.7 (12.2), respectively (Kay et al., 1987).
Protocol was approved by the Institutional Review Board of McLean Hospital, and informed consent was attained from each participant. Table 1 provides an overview of demographic information of the sample.
Table 1.
Demographic information of the sample.
| 1. Participants | ||||
|---|---|---|---|---|
| Sex | Age | Verbal IQ | Education | |
| Healthy Controls | Female (n = 35) | 39.5 (14.2) | 110.3 (10.9) | 16.4 (2.3) |
| Male (n = 27) | 45.4 (13.7) | 110.5 (10.5) | 15.5 (2.2) | |
| Schizophrenia Patients | Female (n = 20) | 42.0 (8.3) | 100.3 (13.8) | 14.1 (2.3) |
| Male (n = 21) | 40.4 (9.5) | 100.1 (14.6) | 13.6 (1.4) | |
Means are reported above standard deviations. Verbal IQ was measured with the WAIS–R. Education and age are in years. There were significant differences between healthy controls and schizophrenia patients on verbal IQ and education (p<0.05). However, there were no significant differences between sexes on any of the demographic variables (p>0.05).
2.2. Stimuli
The target for face detection was a line-drawn face (or tree), which was located on either the left or right side of a larger, scrambled drawing (Fig 1). Orientation of faces could be upright or inverted, whereas trees were always presented upright. Drawings were displayed for 13, 26, 52, or 104 msec. For the facial identity discrimination task, the target was a face photograph image, with the presentation time set to 600 msec. Stimuli from both tasks were presented on a PowerMac G4 with a 17-inch Studio monitor. Both the drawings and the photograph images were 9 × 13.5cm. Subjects performed tasks in a darkened room, seated at a distance of 63cm from the monitor.
Figure 1.
Illustration of the upright face detection task.
2.3. Procedure
For the face detection task, a two-alternative forced choice method was employed: Subjects were asked to judge whether the face (or tree) was located on the left or right side of the line-drawing, and pressed one of two buttons on a keyboard to indicate their response. Stimulus presentation was blocked into sessions according to presentation time (13, 26, 52 and 104 msec), orientation (upright and inverted), and type (face and tree) of target. The order of sessions was randomized across subjects. Percent correct was measured and analyzed as the performance index.
For the facial identity discrimination task, participants were shown an initial face photograph, and then, after a 500 msec inter-stimulus interval (ISI) with a blank screen, were shown two more side-by-side face photographs (Fig 2). The use of this brief interval (500 msec) was used to reduce the likelihood that working memory constraints might facilitate a difference between groups. Degree of facial similarity between the side-by-side face photographs (5, 12.5, 25, 50, or 100%) was systematically varied via morphing. Morphed images were created using Gryphon Morph (Version1.0, Gryphon Software). Subjects were asked to determine which of the two face images matched the initial image by pressing one of two buttons on a keyboard. Individuals’ perceptual thresholds, defined as the minimum difference in facial similarity required to achieve 80% accuracy, was derived and analyzed as the performance index for this task.
Figure 2.
Illustration of the face identity discrimination task.
3. Results
3.1. Upright face detection
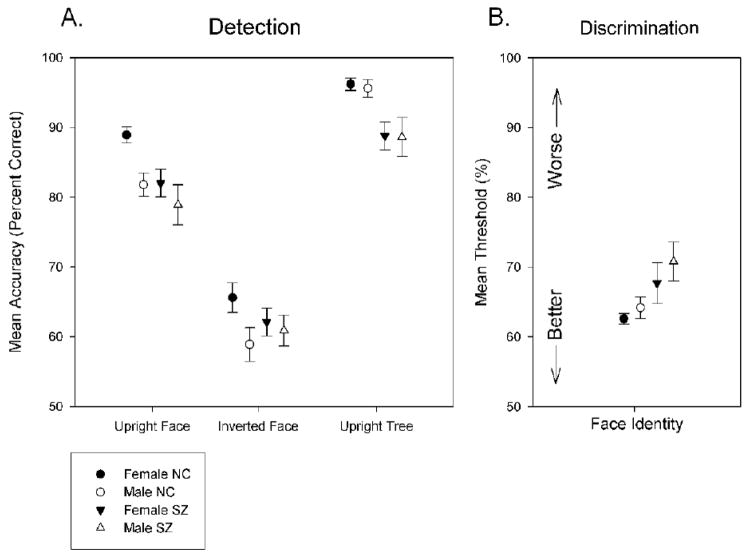
Using a three-way ANOVA (group × sex × presentation time), we found a significant main effect for group (F(1,101)=25.4, P<0.001, ES (effect size) = 0.54) on the upright face detection task, indicating that healthy controls outperformed patients. The same analysis also revealed a main effect for sex (F(1,101)= 27.7, P<0.001, ES=0.61), showing that women were more accurate than men regardless of group, and a main effect for presentation time (F(3, 408)=6.1, P<0.001), connoting higher accuracy when the stimulus was displayed for a longer durations. Lastly, there was a marginally significant interaction effect between sex and group (F(1,101)=3.8, P=0.050). Post-hoc ANOVAs revealed that healthy females performed significantly better than healthy males (F(1,60)=47.1, P<0.001, ES=0.91), but female patients did not perform significantly better than male patients (F(1,39)=3.1, P>0.05, ES=0.26). Figure 3A shows mean accuracy for male and female patients and controls on this task.
Figure 3.
A. Mean accuracy of male and female patients and controls on the three conditions of the detection task. The x-axis specifies the task condition. The y-axis represents mean accuracy, measured as percent of correct trials, of each group/sex. Error bars are representative of ±1 standard error. B. Mean thresholds of male and female patients and controls on face identity discrimination. The x-axis specifies the task condition; the y-axis is the minimum difference in facial similarity required for participants to achieve 80% accuracy, otherwise known as the ‘perceptual threshold.’ The lower a threshold is, the better the performance of the individual subject. Error bars are representative of ±1 standard error.
Patients diagnosed with schizophrenia were also compared with those diagnosed with schizoaffective disorder in order to see whether one group or the other accounted for a larger portion of the overall group difference between patients and controls. Using a three-way ANOVA (group (schizophrenia vs. schizoaffective) × sex × presentation time) we found no significant main or interaction effects (p>0.05).
3.2. Inverted face detection and tree detection
For inverted face detection, a three-way ANOVA (group × sex × presentation time) found a significant main effect for sex (F(1,78)=11.9, P<.001, ES=0.41) with women outperforming men; however, the effects for group (F(1,78)=0.40, P>0.05, ES=0.11) and presentation time (F(3,316)=0.85, P>0.05) were non-significant. The interaction between group and sex was significant (F(1,78)=5.8, P<0.05). Post-hoc ANOVAs revealed a significant performance difference between male and female healthy controls (F(1,37)=15.7, P<0.001, ES=0.69). This sex difference was not found in the patient group (F(1,39)=0.59, P>0.05, ES=0.13) (see Fig 3).
For tree detection, a three-way ANOVA (group × sex × presentation time) found significant main effects for group (F(1,71)=52.2, P<0.001, ES=0.92) and presentation time (F(3,288)=3.1, P<0.05), but not for sex (F(1,71)=0.31, P>0.05, ES=0.03). The interaction effect between group and sex was not significant (F(1,71)=0.13, P>0.05).
3.3. Facial identity discrimination
Performance on the facial identity discrimination task was assessed using a two-way ANOVA (group × sex) with perceptual thresholds as the dependent variable. This analysis yielded a main effect for group (F(1,70)=7.7, P<0.01, ES=−0.66), indicating that healthy controls had lower thresholds (better performance) than patients. There was no main effect found for sex (F(1,70)=1.2, P>0.05, ES=−0.22), nor was there a significant interaction effect between group and sex (F(1,70)=0.12, P>0.05). Figure 3B depicts mean perceptual thresholds for genders and groups on this task.
The performance of patients diagnosed with schizophrenia was again compared against the performance of those diagnosed with schizoaffective disorder. Using a two-way ANOVA (group × sex), there were no significant main or interaction effects (P>0.05). However, there was a trend towards a main effect for group (F(1, 31)=3.4, P=0.08), indicating that schizoaffective patients had lower thresholds than schizophrenia patients. This implies that those diagnosed with schizophrenia accounted for a larger proportion of the overall group difference between patients and controls.
3.4. Associations among perceptual and clinical variables
In both groups, performance scores on upright face detection were significantly correlated with tree detection scores (patients: r=0.67, P<0.001; controls: r=0.56, P<0.001). However, performance scores on face identity discrimination were not associated with tree detection scores (patients: r=−0.17, P>0.05; controls: r=−0.13, P>0.05). When tree detection performance was used as a covariate, the sex difference and the group difference in upright face detection were no longer significant (P>0.05), but the interaction between group and sex remained significant (F(1,71)=7.1, P<0.05). Post-hoc pairwise comparisons revealed the reason for this interaction: A significant sex difference between male and female controls (P<0.05) was countervailed by a non-significant difference between male and female patients (P>0.05).
In patients, IQ was also significantly correlated with upright face detection (r=0.50, P<0.01), but not face identity discrimination (r=−0.12, P>0.05), whereas, in normal controls, IQ was not significantly correlated with performance on either task (P>0.05). When IQ was used as a covariate, the sex difference and the group difference in upright face detection were no longer significant (P>0.05), but the interaction between sex and group remained significant (F(1,101)=7.0, P<0.05). Post-hoc pairwise comparisons revealed a significant sex difference in normal controls (P<0.05) but not in patients (P>0.05), which accounted for this interaction.
Female and male patients’ clinical statuses were compared using scores from the PANSS scale, which assesses positive (e.g., hallucinatory behavior, grandiosity), negative (e.g., blunted affect, poor rapport) and general (e.g., somatic concern, anxiety) symptomotology. Female and male patients did not differ significantly in terms of positive, negative, general, or overall scores (P>0.05). Additionally, genders did not differ in terms of antipsychotic mediation level, measured in chlorpromazine equivalent (P>0.05), suggesting that medication could not have played a significant role in affecting male versus female patient performance. On upright face detection, performance was moderately correlated with antipsychotic medication levels (r=0.34, P<0.05) and general PANSS scores (r=−0.37, P<0.05). Face identity discrimination performance was correlated with positive (r=0.59, P<0.01) and general (r=0.47, P<0.05) PANSS scores, but not medication levels (P>0.05).
4. Discussion
This study found that a perceptual advantage in face detection was present in healthy women, but not in women with schizophrenia.
4.1. Sex differences
A performance advantage in healthy females (as compared to healthy males) has been found on facial emotion recognition tasks (for a review, see McClure, 2000), and also on face perception memory tasks (Lewin and Herlitz, 2002; Rehnman and Herlitz, 2007). The data from the present study extend these findings by showing that healthy females also have an advantage at face detection, the putative first stage of face recognition.
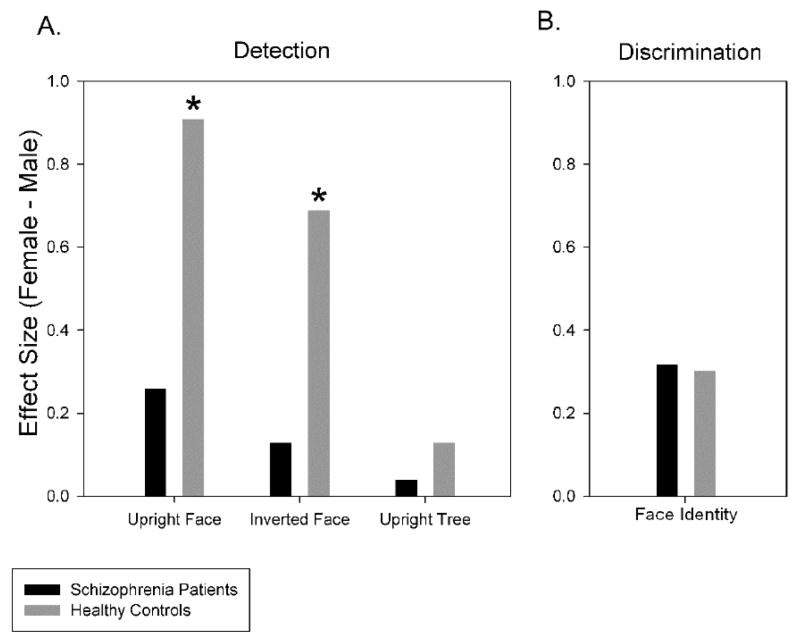
For three reasons, this female advantage appears to be specifically related to processing of face information rather than perceptual processing in general. First, while females outperformed males on upright and inverted face detection, no such sex differences were found on upright tree detection. Secondly, when tree detection and IQ scores were entered as covariates on upright face detection, the sex difference in healthy controls remained significant, suggesting that the performance difference in men and women was not determined by these factors. Lastly, pattern of effect sizes from upright face (ES=0.91), inverted face (ES=0.69) and upright tree detection (ES=0.13) indicate that the female advantage was greatest when face-processing mechanisms were most active (Kanwisher et al., 1998; Rossion and Gauthier, 2002).(see Fig. 4A).
Figure 4.
A. Effect size of the sex difference in patients and controls on the three conditions of the detection task. The x-axis specifies the task condition; the y-axis is effect size of the sex difference, defined as delta averaged across presentation times (13, 26, 52, 104 msec). B. Effect size of the sex difference in patients and controls on the identity discrimination task. The y-axis represents effect size of the sex difference, defined as delta.
Interestingly, this female advantage in face detection was not present in the patient group (see Fig. 4B). One prospective reason for this absence is that a generalized deficit acted to level performance across sexes. A generalized deficit in schizophrenia has been shown to affect patients’ performance across multiple cognitive domains (Chapman and Chapman, 1973; Dickinson et al., in press), and may similarly dampen performance on face recognition tasks. Two aspects of the results from the present study suggest that the generalized deficit influenced patients’ performance. First, patients not only performed worse than healthy controls on upright face detection and face identity discrimination, but also on tree detection, suggesting that patients’ performance was deficient, whether or not face recognition was involved. Second, both tree detection and IQ scores, which were used as assessments of general perceptual and cognitive functioning, were significantly correlated with upright face detection performance in patients.
Another possible reason for the absence of a sex difference in patients is that the neurobiological mechanisms mediating patients’ cognitive responses, including in face recognition, may be different and/or differentially affected in males and females (Rubin et al., 2008). There is some evidence suggesting that deficient estrogenic function in females with schizophrenia may contribute to impaired cognition in this group (Mortimer, 2007). For example, increased levels of estradiol in female patients are associated with better neuropsychological and cognitive performance (Hoff et al., 2001; Halari et al., 2004; Ko et al., 2006). While it remains to be seen how face detection in female patients is adversely affected by estrogenic dysfunction, it has been shown that estrogen functioning can modulate visual perception (Gupta et al., 2005), including face recognition (Gasbarri et al., 2008).
Recently, higher synaptic density in males was found in the temporal cortex, the area where face information is processed; this difference was not found in other cortical areas (Alonso-Nanclares et al., 2008). This sex difference at synaptic levels raises a question as to whether dendrite pruning is more intense in females, leading to the lower synapse density and in turn improving abilities in relevant cognitive functions such as face perception. Schizophrenia has been widely hypothesized as a disorder of synaptic dysfunction (Frankle et al., 2003; Stephan et al., 2006), and this synaptic dysfunction could compromise the sex difference in synapse density, and be a potential factor for the lack of a female advantage in face detection in patients.
While this study found no sex differences between male and female patients in the two non-affective face perception tasks, a recent study by Scholten and colleagues (2005) found a significant female advantage on affective face perception. This difference between these two types of face recognition tasks suggests that perceptual processing of faces is differentially affected in female patients, and that a female patient advantage in emotion recognition may be associated with relatively higher levels of social performance in female patients.
4.2. Group differences
Healthy controls significantly outperformed patients on all perceptual tasks except inverted face detection. On upright face detection, the difference between healthy females and female patients (ES=0.89) was much greater than that between healthy males and male patients (ES=0.24); the former thus accounted for a larger proportion of the overall group difference. This interaction between group and sex was not found on face identity discrimination or tree detection, indicating that sex played a considerably smaller role in influencing group differences on these tasks.
4.2.1. Face Detection
Two previous studies have shown that schizophrenia patients are less accurate than healthy controls at detecting faces (Chen et al., 2008; Zivotofsky et al., 2008). While this result was replicated in the present study, our findings also indicate that a generalized deficit may have played a role in poor patient performance on this task (refer to p. 10 for a review of reasons). Interestingly, upright face detection was significantly correlated with performance on general PANSS scores, as well as with antipsychotic medication levels. This correlation offers a tentative hypothesis that face detection performance is linked to the clinical status of patients.
4.2.2. Face Identity Discrimination
Patients underperformed on identity discrimination, which is consistent with a recent study by Shin and colleagues (2008) showing that patients are deficient at discriminating facial identities, particularly when discrimination requires the use of configural face information. Patients’ under-performance on this task was significantly correlated with the positive and general subscales of the PANSS, suggesting that facial identity discrimination is also associated with clinical status.
4.3. Limitations
There are a few notable limitations of this study. Firstly, while the gender difference in patients was non-significant for each aspect of basic face recognition, these null results should be confirmed in a larger patient population. Secondly, basic face detection relies on the processing of visual information, Deficient visual perception has been found in schizophrenia (Butler and Javitt, 2005; Chen et al., 2006). Whether some aspects of the visual processing deficit are gender-related and play a role in diminished performance on face detection have yet to be examined. Thirdly, the clinical statuses of male and female patients were similar in this study, whereas some studies have found that females’ clinical symptoms are less pronounced than males’ (e.g., Thorup et al., 2007). It is conceivable, therefore, that if male and female patients in this study had differed in terms of clinical status, the results of face recognition could also have differed.
Yet, it is also important to note that females with schizophrenia performed worse than females without schizophrenia to greater degree than males with schizophrenia versus males without schizophrenia. This result was found despite the fact that both males and females with schizophrenia exhibited similar levels of clinical symptoms. In other words, female patients were more adversely affected by the deficit in basic face detection, even though clinical status was similar for both genders.
4.4. Concluding Remarks
In summary, this study provides perceptual evidence that the normal female advantage in face detection is not present in schizophrenia. This lack of a sex difference in basic face perception is likely due to a generalized cognitive deficit which acted to level performance across males and females, and also putative changes in sex-related neurobiological differences associated with schizophrenia. Future studies should further investigate face processing deficits in schizophrenia by directly comparing male and female performance on affective and non-affective face processing tasks, and outlining the neurobiological mechanisms mediating responses across genders. Furthermore, perceptual training strategies for deficient face recognition in schizophrenia may take our pattern of perceptual results into account by targeting remediation of affect recognition impairments in male patients (Combs et al., 2008; Frommann et al., 2003), and focusing on remediation of more general face recognition impairments across both genders.
Acknowledgments
This study was funded in part by grants from the National Institutes of Health (Grant MH 61824) and Harvard University. We thank Ken Nakayama for contributions to the initial phase of this project, and Grace Masters and Charles Stromeyer IV for assistance in manuscript preparation.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Alonso-Nanclares L, Gonzalez-Soriano J, Rodriguez JR, DeFelipe J. Gender differences in human cortical synaptic density. Proceedings of the National Academy of the Sciences of the USA. 2008;105:14615–14619. doi: 10.1073/pnas.0803652105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruce V, Young A. Understanding face recognition. British Journal of Psychology. 1986;77:305–327. doi: 10.1111/j.2044-8295.1986.tb02199.x. [DOI] [PubMed] [Google Scholar]
- Butler PD, Javitt DC. Early-stage visual processing deficits in schizophrenia. Current Opinion in Psychiatry. 2005;18:151–7. doi: 10.1097/00001504-200503000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calder AJ, Young AW. Understanding the recognition of facial identity and facial expression. Nature Reviews: Neuroscience. 2005;6:641–651. doi: 10.1038/nrn1724. [DOI] [PubMed] [Google Scholar]
- Chapman L, Chapman J. Disordered Thought in Schizophrenia. Apple Century Crofts; New York: 1973. [Google Scholar]
- Chen Y, Bidwell LC, Norton D. Trait vs. state markers for schizophrenia: Identification and characterization through visual processes. Current Psychiatry Reviews. 2006;2:431–438. doi: 10.2174/157340006778699729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Norton D, McBain R, Ongur D, Heckers S. Visual and cognitive processing of face information in schizophrenia: Detection, discrimination and working memory. Schizophrenia Research. 2009;107:92–98. doi: 10.1016/j.schres.2008.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Norton D, Ongur D, Heckers S. Inefficient face detection in schizophrenia. Schizophrenia Bulletin. 2008;34:367–74. doi: 10.1093/schbul/sbm071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Combs DR, Tosheva A, Penn DL, Basso MR, Wanner JL, Laib K. Attentional-shaping as a means to improve emotion perception deficits in schizophrenia. Schizophrenia Research. 2008;105:68–77. doi: 10.1016/j.schres.2008.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickinson D, Ragland JD, Gold JM, Gur RC. General and Specific Cognitive Deficits in Schizophrenia: Goliath Defeats David? Biological Psychiatry. doi: 10.1016/j.biopsych.2008.04.005. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duchaine BC, Parker H, Nakayama K. Normal recognition of emotion in a prosopagnosic. Perception. 2003;32:827–838. doi: 10.1068/p5067. [DOI] [PubMed] [Google Scholar]
- First MB, Spitzer RL, Gibbon M, Williams JB. Structured clinical interview for DSM-IV-TR Axis I Disorders - Patient Edition (SCID - I/P, 11/2002 revision) Biometrics Research Department; New York: 2002. [Google Scholar]
- Frankle WG, Lerma J, Laruelle M. The synaptic hypothesis of schizophrenia. Neuron. 2003;17:205–216. doi: 10.1016/s0896-6273(03)00423-9. [DOI] [PubMed] [Google Scholar]
- Frommann N, Streit M, Wölwer W. Remediation of facial affect recognition impairments in patients with schizophrenia: a new training program. Psychiatry Research. 2003;117:281–4. doi: 10.1016/s0165-1781(03)00039-8. [DOI] [PubMed] [Google Scholar]
- Green MJ, Waldron JH, Coltheart M. Emotional context processing is impaired in schizophrenia. Cognitive Neuropsychiatry. 2007;12:259–80. doi: 10.1080/13546800601051847. [DOI] [PubMed] [Google Scholar]
- Güntekin B, Ba3ar E. Gender differences influence brain’s beta oscillatory responses in recognition of facial expressions. Neuroscience Letters. 2007;424:94–9. doi: 10.1016/j.neulet.2007.07.052. [DOI] [PubMed] [Google Scholar]
- Gupta PD, Nagpal K, Vasavada AR. Sex hormone receptors in the human eye. Survey of Opthalmology. 2005;50:274–284. doi: 10.1016/j.survophthal.2005.02.005. [DOI] [PubMed] [Google Scholar]
- Halari R, Kumari V, Mehrotra R, Wheeler M, Hines M, Sharma T. The relationship of sex hormones and cortisol with cognitive functioning in schizophrenia. Journal of Psychopharmacology. 2004;18:366–74. doi: 10.1177/026988110401800307. [DOI] [PubMed] [Google Scholar]
- Haxby JV, Hoffman EA, Gobbini MI. Human neural systems for face recognition and social communication. Biological Psychiatry. 2002;51:59–67. doi: 10.1016/s0006-3223(01)01330-0. [DOI] [PubMed] [Google Scholar]
- Heimberg C, Gur RE, Erwin RJ, Shtasel DL, Gur RC. Facial emotion discrimination: III. Behavioral findings in schizophrenia. Psychiatry Research. 1992;42:253–65. doi: 10.1016/0165-1781(92)90117-l. [DOI] [PubMed] [Google Scholar]
- Hoff AL, Kremen WS, Wieneke MH, Lauriello J, Blankfeld HM, Faustman WO, Csernansky JG, Nordahl TE. Association of estrogen levels with neuropsychological performance in women with schizophrenia. American Journal of Psychiatry. 2001;158:1134–1139. doi: 10.1176/appi.ajp.158.7.1134. [DOI] [PubMed] [Google Scholar]
- Humphreys GW, Donnelly N, Riddoch MJ. Expression is computed separately from facial identity, and it is computed separately for moving and static faces: Neuropsychological evidence. Neuropsychologia. 1993;31:173–181. doi: 10.1016/0028-3932(93)90045-2. [DOI] [PubMed] [Google Scholar]
- Johnson M. Subcortical Face Processing. Nature Reviews. Neuroscience. 2005;6:766–74. doi: 10.1038/nrn1766. [DOI] [PubMed] [Google Scholar]
- Kanwisher N, Tong F, Nakayama K. The effect of face inversion on the human fusiform face area. Cognition. 1998;68:B1–11. doi: 10.1016/s0010-0277(98)00035-3. [DOI] [PubMed] [Google Scholar]
- Kay S, Fiszbein A, Opler L. The Positive and Negative Syndrome Scale for schizophrenia. Schizophrenia Bulletin. 1987;13:261–276. doi: 10.1093/schbul/13.2.261. [DOI] [PubMed] [Google Scholar]
- Ko YH, Joe SH, Cho W, Park JH, Lee JJ, Jung IK, Kim L, Kim SH. Estrogen, cognitive function and negative symptoms in female schizophrenia. Neuropsychobiology. 2006;53:169–75. doi: 10.1159/000093780. [DOI] [PubMed] [Google Scholar]
- Køster A, Lajer M, Lindhardt A, Rosenbaum B. Gender differences in first episode psychosis. Social Psychiatry and Psychiatric Epidemiology. doi: 10.1007/s00127-008-0384-3. In Press. [DOI] [PubMed] [Google Scholar]
- Leung A, Chue P. Sex differences in schizophrenia, a review of the literature. Acta Psychiatrica Scandinavica. Supplementum. 2000;401:3–38. doi: 10.1111/j.0065-1591.2000.0ap25.x. [DOI] [PubMed] [Google Scholar]
- Lewin C, Herlitz A. Sex differences in face recognition-women’s faces make the difference. Brain and Cognition. 2002;50:121–8. doi: 10.1016/s0278-2626(02)00016-7. [DOI] [PubMed] [Google Scholar]
- McBain R, Norton D, Chen Y. Females excel at basic face perception. Acta Psychologica. 2009;130:168–173. doi: 10.1016/j.actpsy.2008.12.005. [DOI] [PubMed] [Google Scholar]
- McClure EB. A meta-analytic review of sex differences in facial expression processing and their development in infants, children, and adolescents. Psychological Bulletin. 2000;126:424– 453. doi: 10.1037/0033-2909.126.3.424. [DOI] [PubMed] [Google Scholar]
- Miura M. Individual differences in the perception of facial expression: the relation to sex difference and cognitive mode. Shinrigaku Kenkyu. 1993;63:409– 413. doi: 10.4992/jjpsy.63.409. [DOI] [PubMed] [Google Scholar]
- Mortimer A. Relationship between estrogen and schizophrenia. Expert Review of Neurotherapeutics. 2007;7:45–55. doi: 10.1586/14737175.7.1.45. [DOI] [PubMed] [Google Scholar]
- Ochoa S, Usall J, Haro JM, Araya S, Autonell J, Busquets E, Gost A, Grupo Nedes. Comparative study of the needs of patients with schizophrenia by gender. Actas Espanolas de Psiquiatria. 2001;29:165–71. [PubMed] [Google Scholar]
- Rehnman J, Herlitz A. Women remember more faces than men do. Acta Psychologica. 2007;124:344–55. doi: 10.1016/j.actpsy.2006.04.004. [DOI] [PubMed] [Google Scholar]
- Rossion B, Gauthier I. How does the brain process upright and inverted faces? Behavioral and Cognitive Neuroscience Reviews. 2002;1:63–75. doi: 10.1177/1534582302001001004. [DOI] [PubMed] [Google Scholar]
- Rubin LH, Hass GL, Keshavian MS, Sweeney JA, Maki PM. Sex difference in cognitive response to antipsychotic treatment in first-episode schizophrenia. Neuropsychopharmacology. 2008;34:290–297. doi: 10.1038/sj.npp.1301395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scholten M, Aleman A, Montagne B, Kahn R. Schizophrenia and processing of facial emotions: Sex matters. Schizophrenia Research. 2005;78:61–67. doi: 10.1016/j.schres.2005.06.019. [DOI] [PubMed] [Google Scholar]
- Schyns PG, Oliva A. Dr. Angry and Mr. Smile: When categorization flexibly modifies the perception of faces in rapid visual presentations. Cognition. 1999;1:243–265. doi: 10.1016/s0010-0277(98)00069-9. [DOI] [PubMed] [Google Scholar]
- Shin YW, Na MH, Ha TH, Kang DH, Yoo SY, Kwon JS. Dysfunction in configural face processing in patients with schizophrenia. Schizophrenia Bulletin. 2008;34:538–43. doi: 10.1093/schbul/sbm118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stephan KE, Baldeweg T, Friston KJ. Synaptic plasticity and dysconnection in schizophrenia. Biological Psychiatry. 2006;59:929–939. doi: 10.1016/j.biopsych.2005.10.005. [DOI] [PubMed] [Google Scholar]
- Thorup A, Petersen L, Jeppesen P, Ohlenschlaeger J, Christensen T, Krarup G, Jorgensen P, Nordentoft M. Gender differences in young adults with first-episode schizophrenia spectrum disorders at baseline in the Danish OPUS study. Journal of Nervous and Mental Disease. 2007;195:396–405. doi: 10.1097/01.nmd.0000253784.59708.dd. [DOI] [PubMed] [Google Scholar]
- Walker E, McGuire M, Bettes B. Recognition and identification of facial stimuli by schizophrenics and patients with affective disorders. British Journal of Clinical Psychology. 1984;23:37–44. doi: 10.1111/j.2044-8260.1984.tb00624.x. [DOI] [PubMed] [Google Scholar]
- Wechsler D. Manual for the Adult Intelligence Scale-Revised. Psychological Corporation; New York: 1981. [Google Scholar]
- Zivotofsky AZ, Oron L, Hibsher-Jacobson L, Weintraub Y, Strous RD. Finding the hidden faces: schizophrenic patients fare worse than healthy subjects. Neuropsychologia. 2008;46:2140–4. doi: 10.1016/j.neuropsychologia.2008.02.024. [DOI] [PubMed] [Google Scholar]