Small "priming" quantities of type I interferon enhance cellular responses to type II interferon by maintaining basal levels of STAT1, explaining the observed crosstalk between these two cytokines.
Abstract
Autocrine priming of cells by small quantities of constitutively produced type I interferon (IFN) is a well-known phenomenon. In the absence of type I IFN priming, cells display attenuated responses to other cytokines, such as anti-viral protection in response to IFNγ. This phenomenon was proposed to be because IFNα/β receptor1 (IFNAR1) is a component of the IFNγ receptor (IFNGR), but our new data are more consistent with a previously proposed model indicating that regulated expression of STAT1 may also play a critical role in the priming process. Initially, we noticed that DNA binding activity of STAT1 was attenuated in c-Jun −/− fibroblasts because they expressed lower levels of STAT1 than wild-type cells. However, expression of STAT1 was rescued by culturing c-Jun −/− fibroblasts in media conditioned by wild-type fibroblasts suggesting they secreted a STAT1-inducing factor. The STAT1-inducing factor in fibroblast-conditioned media was IFNβ, as it was inhibited by antibodies to IFNAR1, or when IFNβ expression was knocked down in wild-type cells. IFNAR1−/− fibroblasts, which cannot respond to this priming, also expressed reduced levels of STAT1, which correlated with their poor responses to IFNγ. The lack of priming in IFNAR1−/− fibroblasts was compensated by over-expression of STAT1, which rescued molecular responses to IFNγ and restored the ability of IFNγ to induce protective anti-viral immunity. This study provides a comprehensive description of the molecular events involved in priming by type I IFN. Adding to the previous working model that proposed an interaction between type I and II IFN receptors, our work and that of others demonstrates that type I IFN primes IFNγ-mediated immune responses by regulating expression of STAT1. This may also explain how type I IFN can additionally prime cells to respond to a range of other cytokines that use STAT1 (e.g., IL-6, M-CSF, IL-10) and suggests a potential mechanism for the changing levels of STAT1 expression observed during viral infection.
Author Summary
Cells of the immune system release interferons (IFNs) in response to pathogens or tumor cells; these proteins signal to other immune cells to initiate the body's defense mechanisms. The two classes of IFNs—types I and II—have different receptors and distinct effects on the cells; however, there is “crosstalk” between them. In particular, small quantities of type I IFN can “prime” cells to produce a robust response to type II IFN. In this paper, we provide evidence to explain the molecular basis of this crosstalk. We show that continuous expression of the transcriptional activator c-Jun is responsible for producing basal, priming levels of a type I IFN; this signals to immune cells with the type I IFN receptor (IFNAR1) to maintain expression of STAT1 inside these cells. STAT1 is a key factor for immune cell responses to type II IFN. Thus, signaling by low levels of type I IFN primes the cells with sufficient STAT1 to respond robustly to a subsequent type II IFN signal. This work provides an alternative explanation of the priming phenomenon to a previous proposal that the ligand-bound type I receptor, IFNAR1, acts as a component of the type II IFN receptor.
Introduction
Although type I and type II interferons (IFNs) have distinct roles in immune responses, there is substantial overlap between the genes and cellular responses they regulate. It has been known for some time that many cells secrete small priming quantities of type I IFNs that facilitate more potent responses to subsequent stimuli [1]–[3]. Moreover, cellular responses to CSF-1 or IFNγ can be affected by neutralizing type I IFN antibodies or knockout of type I IFN-Receptors (IFNAR) [2],[4],[5]. Notably, the protective anti-viral effects of IFNγ were much less potent in IFNAR1 −/− than wild-type fibroblasts which appeared to be caused by a lack of type I IFN priming [4],[5]. The molecular events that underpin these priming events have not been fully characterized, although it has been proposed that type I and II IFNs shared receptor components [5]. However, as the majority of responses to type I and II IFNs require the expression of the STAT1 transcription factor [6], this is also a possible point of crosstalk between them.
STAT1 is a key mediator of cytokine-induced gene expression as it is activated either as homo- or heterodimer with other STATs by many cytokines including type I and type II IFNs, interleukin (IL)-6 and IL-10. STAT1 activity is of particular importance to the IFN system as STAT1−/− mice display many similar phenotypes to mice lacking IFNAR1 or the IFN Receptor (IFNGR)1. In particular, anti-viral, anti-mycobacterial, and anti-tumor responses are compromised [6]–[9]. Induction of STAT1 expression is a potential explanation for the priming activity of type I IFN because it is an IFN-stimulated gene (ISG) itself [10]–[12] and its 5′ promoter region contains an IRF/gamma activated sequence (GAS) element bound by IFN-stimulated transcription factors [13]. Inducing the expression of STAT1 would increase the pool of this factor available for activation by IFNγ. Consistent with such a hypothesis, low expression of STAT1 correlated with IFN-resistance in melanoma samples when compared to surrounding normal tissue [14].
In unstimulated cells, STAT1 resides in the cytoplasm as a latent factor that is activated by a series of post-translational modifications initiated when it is recruited to cytokine receptors following receptor ligation [15]. At the receptor, STAT1 is phosphorylated on tyrosine 701, by Janus family kinase (JAK)s, which facilitates its dimerization either with other STAT1 molecules or other STAT proteins depending on the cytokine receptor. In addition, STAT1 proteins are phosphorylated on serine 727 prior to nuclear translocation which is essential for their full transcriptional activity [16]. Conversely, STAT1 activity is negatively regulated by phosphatases, SOCS proteins, and the SUMO ligase Protein Inhibitor of Activated STAT (PIAS)1 [15].
Recently, in the course of our studies on IFNγ-activated AP-1 DNA binding, we noticed that IFNγ-induced GAS DNA binding was suppressed in c-Jun −/− cells compared to wild-type cells [17] and this correlated reduced levels of STAT1 in c-Jun −/− cells. The level of STAT1 expression in c-Jun −/− murine embryonic fibroblasts (MEFs) were restored to wild-type levels following culture in media conditioned by wild-type fibroblasts suggesting that c-Jun deficiency caused the disruption of an autocrine/paracrine loop that regulated STAT1 expression. The STAT1-inducing component of media conditioned by wild-type fibroblasts was IFNβ, because the activity could be blocked by neutralizing antibodies directed against type I IFN and antibodies used were raised against IFNAR and attenuated by targeted knockdown of IFNβ by RNA interference (RNAi). While c-Jun has been demonstrated to co-operate with ATF-2, IRF-3, and NFκB for virus-induced production of IFNβ [18], to our knowledge our studies are the first to demonstrate that c-Jun is necessary for basal expression of low-level IFNβ. Fibroblasts in which this autocrine/paracrine loop was disrupted by the loss of components of type I IFN receptors also express lower levels of STAT1. As many biological functions of IFN require STAT1 [6],[7], this suggested that previous observations of attenuated responses to IFN in IFNAR1−/− cells may be related to the reduced STAT1 expression that has been observed [19]. Consistent with this hypothesis, restoring STAT1 expression in IFNAR1 −/− fibroblasts rescued IFNγ-induced gene transcription and anti-viral properties.
In summary, this study provides evidence of an autocrine/paracrine stimulatory loop that requires the expression of c-Jun, IFNβ, and IFNAR to regulate the expression of STAT1. Importantly, this basal IFNβ production occurs via a mechanism distinct from the pathogen-stimulated IFNβ production mediated by IRF and NFκB pathways [18]. One model to explain crosstalk between type I and II IFNs states that type I and II IFN-R physically interact in a ligand-dependant manner, such that the presence of type I IFNs is essential for a fully competent IFNγ response [5]. Herein, we demonstrated that attenuated IFNγ-mediated gene induction and an associated defective anti-viral response to IFNγ that is observed in IFNAR1-deficient cells can be rescued by re-expressing STAT1 and is therefore independent of IFNAR1. We propose that an alternative model to explain the functional synergy between type I and II IFNs is based on the regulated expression of STAT1 via c-Jun-mediated production of basal levels of IFNβ.
Results
STAT1 Expression Is Attenuated in c-Jun−/− MEFs
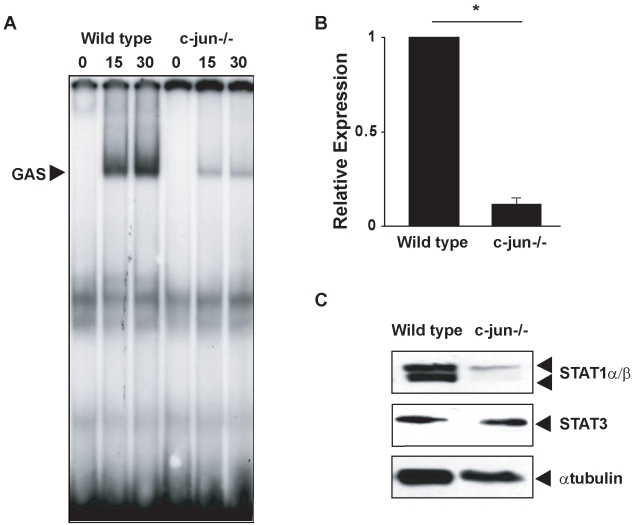
In the course of our studies of IFN-induced signaling and gene expression, we performed elecrophoretic mobility shift assays (EMSAs) assessing GAS binding species in nuclear extracts from IFNγ-stimulated wild-type and c-Jun −/− MEFs. A GAS binding complex was detected in both wild-type and matched c-Jun −/− MEFs following 15–30 min of exposure to IFNγ, however in the absence of c-Jun, IFNγ-induced GAS binding activity was markedly attenuated (Figure 1A). The decrease in GAS binding activity in c-Jun −/− MEFs was a consequence of reduced expression of STAT1. Both STAT1 mRNA and protein were ∼10-fold lower in c-Jun −/− MEFs compared to wild-type cells (Figure 1B and C). However, expression of STATs was not globally affected, as expression of STAT3, another GAS-binding transcription factor, remained unchanged (Figure 1C). Reduced STAT1 expression was not a clone-specific phenomenon as similar results were obtained using an independently derived matched pair of wild-type and c-Jun −/− MEFs (Figure S1).
Figure 1. STAT1 DNA binding and expression is attenuated in c-Jun-deficient MEFs.
(A) EMSAs were performed using radiolabeled oligonucleotides containing a GAS consensus sequence, and nuclear extracts from wild-type or c-Jun −/− MEFs treated with 100 IU/mL IFNγ for indicated times. (B) RNA was extracted from wild-type or c-Jun −/− MEFs, cDNA synthesized, and qRT-PCR performed with primers complementary to murine STAT1. Histograms represent mean and error bars the SEM of four independent experiments and are expressed relative to the levels detected in wild-type cells (* p<0.05). (C) SDS-PAGE and Western blotting with antibodies against STAT1 and STAT3 were performed using whole cell extracts from wild-type or c-Jun −/− MEFs. As a control, the expression of α-tubulin was also tested by Western blot.
c-Jun Maintains Levels of STAT1 Expression by Stimulating Autocrine Production of a Soluble Factor
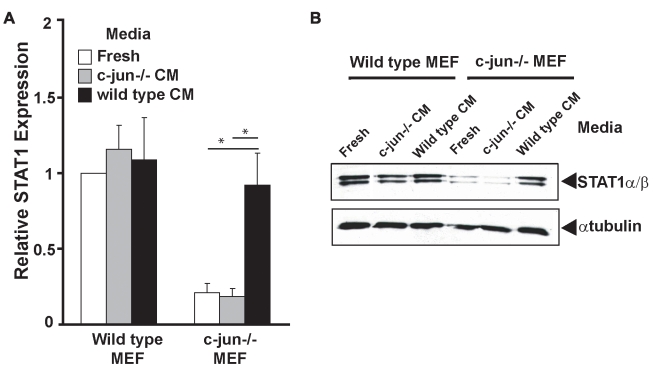
To determine if c-Jun could regulate STAT1 levels by inducing the secretion of a soluble factor that acted in autocrine/paracrine fashion to induce STAT1 expression, conditioned media from wild-type or c-Jun −/− MEFs were cultured in (i) fresh media, (ii) media conditioned by c-Jun −/− MEFs, or (iii) media conditioned by wild-type MEFs. Cells were harvested after 16 h of culture in conditioned media and STAT1 mRNA and protein expression was assessed. Expression of STAT1 mRNA and protein was unaltered in wild-type MEFs cultured in fresh media or conditioned media from wild-type or c-Jun −/− MEFs (Figure 2A and B). In c-Jun −/− MEFs, basal expression of STAT1 was much lower than in wild-type cells and was not increased when the cells were cultured in either fresh media or conditioned media from c-Jun −/− MEFs (Figure 2A and B). In contrast, when c-Jun −/− MEFs were cultured in media conditioned by wild-type MEFs, STAT1 mRNA and protein expression was induced almost to the levels observed in wild-type cells (Figure 2A and B). These data confirmed that fibroblasts secrete a c-Jun-dependent soluble factor that induces STAT1 expression through an autocrine/paracrine feedback loop.
Figure 2. c-Jun is necessary for constitutive IFNβ secretion and subsequent maintenance of STAT1 expression.
Wild-type or c-Jun −/− MEFs were cultured in fresh media (Fresh) or conditioned media from either c-Jun−/− (c-Jun−/− CM) or wild-type (wild-type CM) MEFs for 16 h. Cells were harvested, and (A) RNA was extracted and STAT1 mRNA expression assessed by qRT-PCR. Histograms represent mean and error bars the SEM of three independent experiments and are expressed relative to the levels detected in wild-type cells cultured in fresh media (* p<0.05 between indicated samples). (B) Whole cell lysates from treated cells were analyzed by SDS-PAGE and Western blot probed with an antibody specific for STAT1 and α-tubulin to confirm equivalent protein loading.
Constitutive Secretion of IFNβ Maintains Basal Expression of STAT1
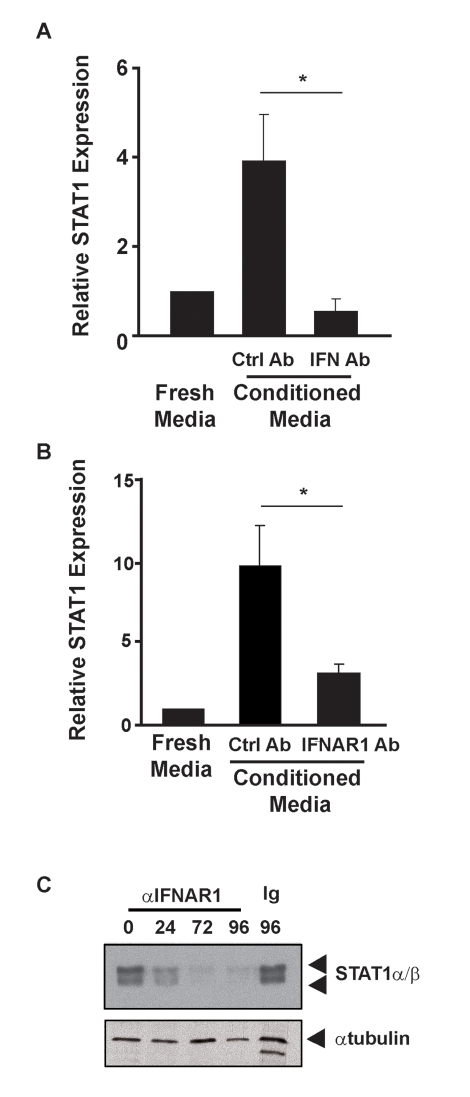
Type I IFN is constitutively secreted from unstimulated fibroblasts and can induce STAT1 expression [10]. To determine if type I IFN was the STAT1-inducing active component of fibroblast conditioned media, c-Jun −/− MEFs were cultured in either fresh or conditioned media from wild-type cells in the presence of a type I IFN blocking antibody [20]. STAT1 expression was increased in c-Jun −/− MEFs cultured in conditioned media from wild-type cells in the presence of control antibodies (Figure 3A) and this enhanced expression was entirely blocked by the presence of type I IFN neutralizing antibodies used at concentrations capable of neutralizing ∼5 IU/mL IFNβ. Additional studies (Figure 3B and C) revealed that the STAT1-inducing activity of wild-type-conditioned media was almost ablated by a blocking mAb raised against IFNAR1 [21]. Together, these data demonstrate that type I IFN is a component of conditioned media from wild-type cells that is necessary for the rescue of STAT1 expression in c-Jun −/− cells.
Figure 3. Antibodies that block type I IFN function modulate STAT1 expression.
c-Jun −/− MEFs were cultured in fresh media or media conditioned by wild-type MEFs for 16 h. RNA was extracted and STAT1 mRNA expression assessed by qRT-PCR. (A) Conditioned media were supplemented with control antibody (Ctrl Ab) or type I IFN neutralizing antibody (IFN Ab). Histograms represent the mean and error bars the SEM of four independent experiments. (B) Conditioned media were supplemented with control antibody (Ctrl) or blocking antibody to IFNAR1 (IFNAR1 Ab). Histograms represent the mean and error bars the SEM of five independent experiments. In each case STAT1 levels are expressed relative to the levels detected in c-Jun −/− cells cultured in fresh media. (* p<0.05 between indicated samples). (C) Wild-type MEFs were cultured for various times in media containing antibodies to IFNAR1 or a control antibody for 96 h. At each time point, cells were harvested, whole cell extracts prepared, and STAT1 expression assessed by Western blot. Tubulin expression was assessed as a loading control.
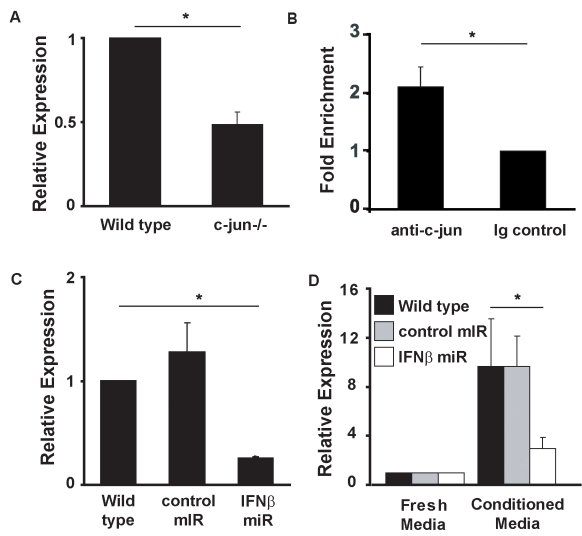
It has been reported that STAT1 levels are diminished in IFNβ −/− cells [22] indicating that IFNβ could be the key component of the conditioned media from wild-type cells shown to induce expression of STAT1 in c-Jun −/− MEFs. Treatment of c-Jun −/− MEFs with doses as low as 1 IU/mL IFNβ induced STAT1 mRNA and doses between 5 and 10 IU/mL were sufficient to restore STAT1 mRNA and protein expression to levels seen in wild-type cells (Figure S2A and B). STAT1 mRNA levels were slightly increased in wild-type MEFs treated with IFNβ (Figure S2C), which is consistent with studies demonstrating that STAT1 expression is induced in fibosarcoma cell lines treated with IFNα or β [12] and in splenic leukocytes where STAT1 levels were increased following virus infection in a type I IFN-dependent manner [11]. Comparison of the levels of expression of IFNβ mRNA in wild-type and c-Jun −/− cells revealed that c-Jun −/− MEFs expressed ∼50% of the wild-type levels of IFNβ mRNA (Figure 4A). AP-1 sites are known to be important for inducible expression of IFNβ [23], but little is known of what regulates constitutive production of type I IFN in unstimulated cultured fibroblasts. Chromatin immunoprecipitation (ChIP) assays on unstimulated wild-type and c-Jun −/− MEFs demonstrated a >2-fold increase in c-Jun bound to the murine IFNβ promoter when compared to Ig control samples (Figure 4B). Together, these data imply that expression of c-Jun and subsequent occupation of the IFNβ promoter by c-Jun is required for basal secretion of IFNβ.
Figure 4. c-Jun binds to the IFNβ promoter and is required to maintain its expression in unstimulated cells.
(A) RNA from wild-type and c-Jun −/− MEFs were assayed for IFNβ expression by qRT-PCR. Histograms represent mean and error bars the SEM of three independent experiments and are expressed relative to the levels detected in wild-type cells (arbitrarily set as one). (B) ChIP assays were performed using chromatin from wild-type MEFs using an anti c-Jun antibody or an isotype control antibody. Immuno-precipitated chromatin was assessed for enrichment of the IFNβ promoter by qRT-PCR using primers flanking PRD IV. Loading of c-Jun on PRD IV was calculated relative to isotype control. Histograms represent the mean and error bars the SEM of three independent experiments (* p<0.05 between indicated samples). (C) RNA was prepared from wild-type MEFs infected with recombinant lentivirus encoding microRNAs (miRs) targeting the IFNβ gene or a control miR. Expression of IFNβ was assessed by qRT-PCR. Histograms represent the mean and SEM of three independent experiments (* p<0.05 between indicated samples). (D) c-Jun −/− MEFs were treated with conditioned media from untransduced wild-type MEFs and MEFs transduced with lentivirus encoding either miRs targeting the IFNβ gene or a non-silencing miR or with fresh media. After 24 h, cells were harvested, RNA prepared, and STAT1 expression assessed by qRT-PCR. Histograms represent the mean and error bars the SEM of five independent experiments (* p<0.05 between indicated samples).
To determine if IFNβ was the type I IFN necessary to maintain STAT1 expression, we used RNAi to knock down IFNβ in wild-type MEFs (Figure 4C) and assessed the ability of conditioned media from these cells to induce the expression of STAT1 mRNA in c-Jun −/− MEFs. As expected, STAT1 mRNA levels were greater when c-Jun −/− MEFs were cultured in conditioned media from wild-type MEFs or from MEFs expressing a control knockdown vector than if these cells were cultured in fresh media (Figure 4D). In contrast, the ability of conditioned media from wild-type cells with RNAi-mediated knockdown of IFNβ to induce STAT1 expression in c-Jun −/− MEFs was significantly reduced (Figure 4D). These data confirm that IFNβ is expressed by unstimulated wild-type fibroblasts and is necessary for the maintenance of STAT1 expression.
IFNAR-Deficient Cells Express Reduced Levels of STAT1
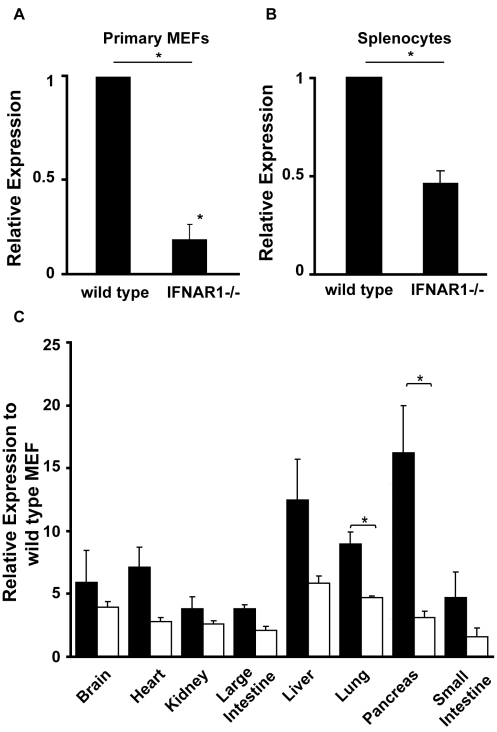
As disruption of autocrine/paracrine stimulation by IFNβ affected the level of STAT1 expression in c-Jun −/− MEFs, we predicted that cells lacking either chain of the type I IFN receptor would also express less STAT1 than wild-type cells. Primary MEFs (Figure 5A) and splenocytes (Figure 5B) from either IFNAR1 −/− or IFNAR2 −/− (unpublished data) mice expressed significantly lower levels of STAT1 than wild-type cells. We extended these studies to compare the expression of STAT1 across multiple tissues in wild-type versus IFNAR1 −/− mice. As shown in Figure 5C, the levels of STAT1 were consistently reduced in all tissues from IFNAR1 −/− mice compared to their wild-type counterparts, suggesting this defect may have broad physiological importance. Interestingly expression of STAT2 was also reduced in IFNAR11 −/− MEFs while the levels of STAT3 were unaffected by knockout of the type I IFN receptor (Figure S3A).
Figure 5. STAT expression in primary IFNAR-deficient cells.
Wild-type and strain matched IFNAR1−/− mice were sacrificed and RNA from (A) primary MEFs, (B) splenocytes, or (C) various indicated tissues was extracted, cDNA synthesized, and STAT1 mRNA expression assessed by qRT-PCR. STAT1 expression was normalized to untreated wild-type cells. Histograms show the mean of three independent experiments and error bars SEM (* p<0.05 between indicated samples).
Our model predicted that, unlike c-Jun deficiency that affected production of an autocrine stimulus, IFNAR1 deficiency affects responses to the autocrine stimulus. In support of this model, wild-type-conditioned media was able to rescue the expression of STAT1 in c-Jun −/− MEFs, but in IFNAR1 −/− MEFs STAT1 expression was unaffected by culture in wild-type-conditioned media (Figure S3B). These data support the existence of an autocrine loop involving IFNβ that regulates basal STAT1 expression levels and suggest that defects in any part of this loop are likely to affect the expression of STAT1.
Re-Expression of STAT1 in IFNAR-Deficient Cells Restores IFNγ Signaling and Gene Expression
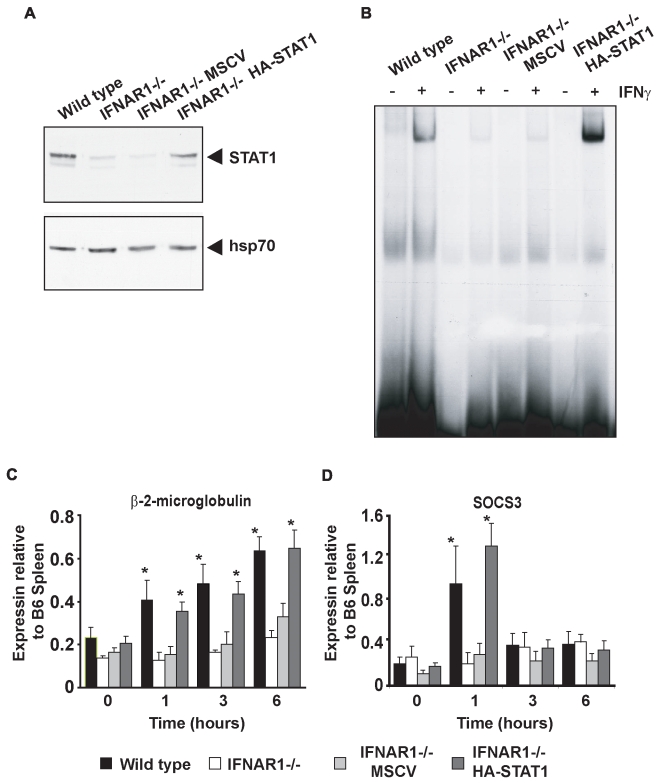
STAT1 is important for not only IFNα/β signaling but also the signaling of several other cytokines, including IFNγ [15]. The expression of approximately two-thirds of IFNγ-induced genes is dependent upon STAT1 expression, however not all IFNγ-mediated biological responses are entirely dependent on STAT1 expression [17],[24]. It has previously been reported that IFNAR1 −/−cells are refractory to IFNγ treatment due to the proposed interaction between IFNAR1 and IFNGR [5]. To determine if decreased expression of STAT1 may confer the observed decrease of IFNγ-mediated responses in IFNAR1−/− cells, STAT1 levels were restored in these cells by retroviral transduction (Figure 6A). GAS binding activity was assessed by EMSA using nuclear extracts from IFNγ-treated wild-type MEFs, IFNAR1 −/− MEFs, and IFNAR1 −/− MEFs reconstituted with empty vector (IFNAR1 −/− MSCV) or STAT1 (IFNAR1 −/− HA-STAT1). Consistent with previous studies [5],[25], IFNγ induced less GAS binding in IFNAR1 −/− cells than wild-type cells (Figure 6B). This low level of GAS binding was also observed in cells transduced with empty vector but was rescued in cells reconstituted with HA-STAT1α. These data demonstrated that the reduced GAS binding observed in IFNAR1 −/− cells was caused by reduced STAT1 expression rather than being a direct consequence of IFNAR1 deficiency.
Figure 6. Restoration of STAT1α expression in IFNAR1−/− MEFs rescues their IFNγ responsiveness.
(A) Protein extracts from wild-type MEFs, IFNAR1−/− MEFs, and IFNAR1−/− MEFs transduced with empty vector (IFNAR1−/− MSCV) or IFNAR1−/− MEFs transduced with HA tagged STAT1α (IFNAR1−/− STAT1) were subjected to SDS-PAGE and probed with an antibody specific to STAT1,and membranes were stripped and re-probed with antibodies specific to hsp70 as a loading control. (B) EMSAs were performed using radiolabeled oligonucleotides containing a GAS consensus sequence, and nuclear extracts from wild-type MEFs, IFNAR1−/− MEFs, IFNAR1−/− MEFs transduced with empty vector (IFNAR1−/− MSCV), or IFNAR1−/− MEFs transduced with HA tagged STAT1α (IFNAR1−/− STAT1) treated in the presence or absence of 100 IU/mL IFNγ. (C, D) Wild-type MEFs, IFNAR1−/− MEFs, and IFNAR1−/− MEFs transduced with empty vector (IFNAR1−/− MSCV) or IFNAR1−/− MEFs transduced with HA-tagged STAT1α (IFNAR1−/− STAT1) were treated with 100 IU/mL IFNγ for 0, 1, or 6 h. RNA was extracted, cDNA synthesized, and qRT-PCR performed with primers specific for β-2-microglobulin and SOCS3. mRNA levels are expressed relative to those of wild-type C57/BL6 (B6) splenocytes. Histograms represent the mean and error bars the standard error of four independent experiments. (* p<0.05 for samples that were significantly induced).
Previous studies demonstrated that IFNγ-induced gene expression was attenuated in IFNAR1 −/− cells [5]. We therefore assessed the impact of re-expression of STAT1α in IFNAR1−/− cells upon the IFNγ-induced expression of genes such as β-2-microglobulin and SOCS3 that require STAT1 expression [26]. Both genes were induced in response to IFNγ in wild-type cells, although with differing kinetic profiles, but induction was weak or absent in IFNAR1 −/− cells. IFNγ-induced expression of both β-2-microglobulin and SOCS3 was restored in cells that re-expressed HA-STAT1α, but not in cells transduced with an empty vector (Figure 6C, D). Similar results were observed when other IFNγ-responsive genes were tested (Figure S4).
Expression of STAT1 in IFNAR-Deficient Cells Restores Their Protective Anti-Viral Response Following Treatment with IFNγ
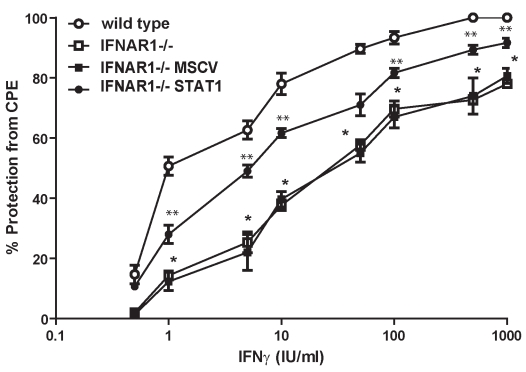
In order to determine whether the reduced levels of STAT1 in IFNAR1 −/− cells could affect biological responses to IFNγ, we investigated whether re-expression of STAT1 in IFNAR1 −/− cells impacted upon the ability of IFNγ to protect them against infection by the cytopathic virus murine encephalomyocarditis virus (EMCV). Wild-type, IFNAR1 −/−, IFNAR1 −/− MSCV, and IFNAR1 −/− HA-STAT1 MEFs were infected with a dose of virus sufficient to induce 100% lysis of wild-type MEFs in the presence or absence of various doses of IFNγ, and the cytopathic effects were determined by assessing cell viability after 24 h. As was shown previously [5], the ability of IFNγ to protect cells from EMCV-mediated lysis was significantly reduced in IFNAR1 −/− MEFs when compared to wild-type MEFs at most doses of IFNγ and the concentration of IFNγ (500 IU/ml) required to provide 80% protection from the virus for IFNAR1 −/− cells was much greater than that required to provide a similar level of protection for wild-type cells (10 IU/ml). The response of IFNAR1 −/− MEFs transduced with empty vector to IFNγ was not significantly different from the untransduced IFNAR1 −/− MEFs at any dose of IFNγ and the concentration of IFNγ required to provide 80% protection (450 IU/ml) was of a similar order of magnitude (Figure 7). In contrast, protection from virus-induced lysis was significantly enhanced in IFNAR1 −/− HA-STAT1 MEFs at most doses of IFNγ. These data provide direct evidence that the attenuated protective anti-viral responses to IFNγ observed in IFNAR1 −/− cells is a consequence of reduced STAT1 expression.
Figure 7. Restoration of STAT1α expression in IFNAR1−/− MEFs rescues the ability of IFNγ to protect cells from cytopathic virus.
Wild-type MEFs, IFNAR1 −/− MEFs, and IFNAR1 −/− MEFs transduced with vector (IFNAR1 −/− MSCV) or IFNAR1 −/− MEFs transduced with HA-tagged STAT1α (IFNAR1 −/− STAT1) were infected with EMCV (0.1 moi) in the presence or absence of various doses of IFNγ. After 24 h, cells were stained with crystal violet and viability assessed by measuring OD550 of the solubilized stain. The effect of IFNγ was determined by comparison of OD550 with samples of known viability. Data points are the mean and error bars represent standard error from three independent experiments. (* p<0.05 between wild-type and IFNAR1 −/− ** p<0.05 between wild-type and IFNAR1 −/−).
Discussion
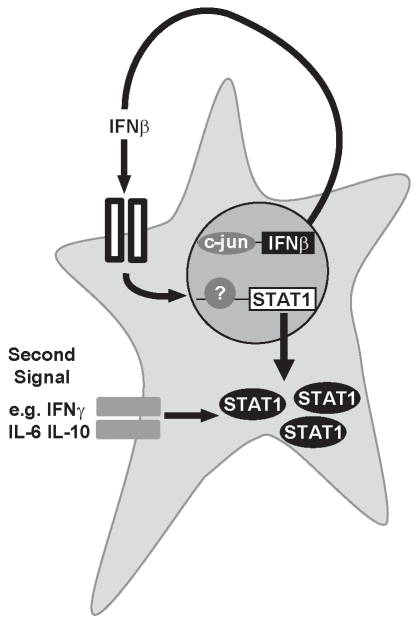
Herein we demonstrate that c-Jun is essential for the constitutive production of small quantities of IFNβ that initiates autocrine or paracrine feedback loops required to maintain the expression of STAT1 (Figure 8). This system was disrupted either by c-Jun deficiency, which prevents production of IFNβ, or by IFNAR deficiency, which affects the ability of cells to respond to the autocrine stimulus. Consistent with our data, others found that cells lacking IFNβ also express much lower levels of STAT1 [22] and virus-mediated induction of STAT1 is dependent on type I IFN signaling [11]. As IFNγ signaling is attenuated when the autocrine stimulus is blocked (Figure 8) but restored by adding back STAT1, it appears the level of STAT1 expressed by the cell determines the response of the cell to other cytokines. These results suggested the ability of IFNγ to induce a protective anti-viral state was due to the type I IFN-mediated maintenance of STAT1 expression rather than the recruitment of IFNAR1 into the IFNγR complex as has been previously proposed [5]. These findings define a novel mechanism through which STAT1-mediated signals can be regulated and highlight the importance of crosstalk between type I and II IFNs for anti-viral immunity.
Figure 8. STAT1 levels are regulated by autocrine/paracrine stimulation by type I IFN.
c-Jun regulates secretion of small amounts of type I IFN, which then stimulate adjacent cells and induce the expression of STAT1. The level of STAT1 that is expressed by a cell dictates how it responds to other stimuli, such as IFNγ, and other cytokines.
It has been known for some time that, as well as being produced in large quantities following viral infections, cells can secrete low levels of type I IFN constitutively [1],[2],[27]. Virus-induced activation of the IFNβ enhanceasome is one of the best-characterized transcriptional modules [18],[23]. Viral activation of the IFNβ promoter involves the binding of NFκB, IRF3, and ATF2/c-Jun complexes to a series of DNA elements termed PRD I-IV [23]. In this setting, c-Jun binds to PRD IV of the promoter and facilitates co-operative binding of the other factors. Removing PRD IV from the promoter, or even reversing its orientation, has a major impact on the transcriptional activity of the promoter [23], suggesting the role of c-Jun is critical in the context of viral infection. In contrast, little is known of the molecular mechanisms of constitutive type I IFN production. Our study indicates that PRD IV of the IFNβ promoter is occupied by c-Jun even in “resting” cultured cells (Figure 4B). This requirement for c-Jun explains why we found that constitutive IFNβ production and hence the expression of STAT1 was attenuated in c-Jun −/− cells. In addition to regulating basal expression of IFNβ, we have recently demonstrated that c-Jun is activated following IFNγ treatment and may also play a direct role in regulating the expression of a subset of IFNγ-responsive genes (ISGs) [17]. Indeed we identified ISGs that were dependent on c-Jun for induction by IFNγ, others that required STAT1, and others that required both c-Jun and STAT1 for increased expression following treatment with IFNγ [17]. These results, coupled with the functional data provided herein, highlight the complex molecular interplay between c-Jun and canonical mediators of type I and II IFN signaling such as STAT1 in regulating a comprehensive response to IFN treatment.
Takaoka and colleagues previously demonstrated the importance of IFNβ in the production of an IFNγ-mediated anti-viral response [5]. In that paper the authors showed that IFNβ −/− MEFs were defective in mounting an IFNγ-induced antiviral response. These data mirror what we have demonstrated herein where we show that IFNAR1 −/− MEFs show a similar defect in mounting an IFNγ-induced antiviral response. However, we showed that restoring STAT1 expression in IFNAR1 −/− cells significantly rescued the ability of IFNγ to protect cells against EMCV, suggesting that regulating the levels of STAT1 expression through the autocrine loop may play an important role in responses to this challenge. The ability of type I and II IFNs to co-operate, for example, in treatment of melanoma tissue [28] or priming of macrophage cytotoxicity [29] has long been recognized. Interestingly, at a cellular level, IFNAR1 −/− cells were known to have an anomalously poor response to IFNγ with respect to induction of GAS DNA binding, induction of gene expression, and protection against the cytopathic effects of EMCV [4],[5],[25]. IFNγ function is not entirely compromised in IFNAR1 −/− animals because IFNGR1 −/− mice have distinct phenotypic differences from IFNAR1 −/− mice [30]. Inhibiting autocrine priming by type I IFN does not only affect signaling by IFNγ. Therefore its is not surprising that IL-6 signaling [31] and CSF-1 signaling are affected by inhibiting priming by type I IFN [2] and that signals induced by IL-10 can be affected by priming with IFNs [32].
It was proposed that the ligand-bound IFNAR1 chain acts as a component of the IFNGR and promotes recruitment of STAT1 to the IFNGR because IFN receptors are clustered within caveolar membrane fractions to facilitate their association [5]. Such a hypothesis is inconsistent with mapping of the docking site of STAT1 to the IFNAR2 chain of the type I IFN-R rather than the IFNAR1 as specified by the shared receptor model [33]. We demonstrated herein that IFNAR1 −/− cells express lower basal levels of STAT1 relative to wild-type controls (Figure 5), and as STAT1 is a critical mediator of IFN signaling, this is an alternative reason why these cells may lack sensitivity to IFNγ. Our model not only explains the inability of IFNγ to prime IFNAR1 −/− cells for an anti-viral response and the rescue of IFNγ function in IFNAR1 −/− cells by STAT1α expression but also the attenuated responses to other cytokines, such as IL-6 and CSF-1, observed in IFNAR1 −/− cells [2],[31] and predicts they may also be rescued by expression of STAT1. As IFNγ function was not entirely recovered following re-expression of STAT1 in IFNAR −/− cells, we cannot exclude that the shared receptor mechanism makes a contribution, but there are other reasons why reconstitution of STAT1 may not have fully rescued IFNγ function. These include the absence of other as yet unidentified signal transducing proteins from cells of this genotype.
The level of STAT1 expression in cells can have functional consequences with respect to immune responses. In response to viral infection, Ag-specific CD8+ T cells express peak levels of STAT1 for a shorter period of time than CD4+ cells [34]. This decreased sensitivity to IFN-induced growth inhibition allows expansion of Ag-specific CD8+ cells while the proliferation of cells with higher STAT1 is inhibited [34]. The relative amounts of different STATs can also affect the biological responses to cytokines. For example STAT1:3 and STAT1:4 ratios have been shown to alter cellular responses, and thus regulating the levels of these transcription factors will affect the outcome of immune responses [11],[35].
Our previous studies revealed that loss of IFN signaling abrogated the immune-mediated neo-natal lethality of SOCS1−/− deficiency [36], and more recently we discovered that deleting IFNAR1 also rescued this pathology [37] to a level equivalent to SOCS1 −/− IFN+/ −. Although SOCS1 directly regulated type I IFN signaling, another reason why IFNAR1 deficiency can protect SOCS1−/− animals may be the similarities and crosstalk between type I and II IFN signaling pathways. These data highlight the patho-physiological importance and mechanism of crosstalk between type I and II IFN that are important considerations in understanding the contributions of individual cytokines to host defense and in their therapeutic targeting.
Materials and Methods
Cells and Reagents
c-Jun −/−, IFNAR1 −/− [38], IFNAR2 −/− [39], and wild-type matched MEFs were derived from embryos and either used as early passage primary MEFs or immortalized by the “3T3” method. IFNAR1 −/− MEFs were transduced with Murine stem cell leukemia virus supernatants encoding GFP alone or cDNA encoding HA-tagged STAT1 (generous gift from Thomas Decker). Supernatants were produced by transient transfection of PhoenixE cells with MSCV vector by calcium phosphate precipitation using standard methods.
c-Jun −/− MEFs were transduced with lentiviral supernatants encoding miR sequences targeting IFNβ (Open Biosystems, Huntsville, AL, USA; product numbers: RMM4431-98755134, RHS4346). Supernatants were produced by transient transfection of 293T cells with pGIPZ vector using Lentiphos HT kit (Clontech, Mountain View, CA, USA) according to the manufacturer's instructions.
Cells were cultured in DMEM supplemented with 5% foetal bovine serum (JRH Biosciences, Lenexa, KS, USA) and 2 mM L-Glutamine (JRH Biosciences, Lenexa, KS, USA). All tissue culture reagents were certified sterile and free of Mycoplasma and pyrogens. Antibodies for the following targets were used: STAT1 (BD Biosciences Franklin Lakes, NJ, USA), STAT3 (Santa Cruz Biotech, Santa Cruz, CA, USA), HA (Cell Signaling Technology, Beverly, MA, USA), c-Jun (Santa Cruz Biotech, Santa Cruz, CA, USA), α-tubulin (Sigma Chemical Co., St. Louis, MO, USA), and hsp70 (Clone N6 was a kind gift of Dr. Robin Anderson; Peter MacCallum Cancer Centre, Melbourne, Australia). Neutralizing anti type I IFN [20] and anti-IFNAR1 were described previously [40]. HRP-conjugated secondary antibodies were purchased from Dako (Glostrup, Denmark).
Production of Conditioned Media
3×106 MEFs were cultured in 175 cm2 tissue culture flasks in 20 mL media for 3 d. Supernatant was collected, cell debris removed by centrifugation (670 g, 4 min), sterilized using a 0.22 µM filter, and stored at 4°C.
SDS-PAGE and Western Blotting
Western blotting was performed as previously described [17]. Briefly, cells were washed, resuspended in whole cell lysis buffer (50 mM Tris-HCl pH 8, 0.1% Triton X-100, 150 mM NaCl, 0.1 mM EDTA, 0.1 mM EGTA, 10% glycerol, 1 µg/mL aprotinin, 0.5 µg/mL leupeptin, and 0.2 mM PMSF), and after (4°C 10 min) lysates were cleared by centrifugation. Proteins were separated by SDS-PAGE, transferred to immobilon P membranes (Millipore), and probed with specific antibodies. Secondary antibodies were conjugated to horseradish peroxidase and images were visualized by chemiluminescence (ECL, GE Healthcare, Bucks, UK).
Nuclear Extraction and EMSA
Nuclear extractions and EMSAs were performed as previously described [17]. Briefly, cells were resuspended in hypotonic lysis buffer (10 mM HEPES, 1.5 mM MgCl2, 10 mM KCl, and protease inhibitors) (4°C, 5 min), NP-40 was added to a final concentration of 0.25%, and the nuclei isolated by centrifugation (2,000 g, 10 min). Nuclei were resuspended in hypertonic lysis buffer (5 mM HEPES pH 8, 1.5 mM MgCl2, 0.2 mM EDTA, 0.5 M NaCl, 25% glycerol, and protease inhibitors) (4°C, 1 h). For binding reactions 5–10 µg of nuclear lysate was incubated (4°C, 30 min) with 5×104 cpm T4 PNK-32P-labeled oligonucleotides in binding buffer (20 mM Tris/HCl pH 8, 6 mM KCl, 2 mM MgCl2, 12% Glycerol, 5 µM DTT, 2.5 µg polydI.dC.polydI.dC, and 0.05% NP-40). Complexes were separated by 5% native PAGE, and gels were dried and visualized by autoradiography on X-ray film (Kodak). The sequence of the GAS oligonucleotide 5′-TAGGGATTTACGGGAAATTGATGAAGCTGATC-3′ was derived from the FcγRI promoter; the AP-1 oligonucleotides were described previously [17].
Quantitative Real Time PCR
RNA was extracted using Trizol (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's instructions. cDNA was synthesized from 2 µg RNA using superscript III (Invitrogen, Carlsbad, CA, USA) as per the manufacturer's instructions. The abundance of specific genes in the samples was quantitated using the SYBR Green dye detection method (Applied Biosystems, Foster City, CA, USA). Primers to murine GBP-1 (5′-TGTGGTTGCTGGATGAGCAGAGTA-3′; 5′-AAGGAAACACAGTAGGCTGGAGCA-3′), SOCS3 (5′-CCTTCAGCTCCAAAAGCGAG-3′; 5′-GCTCTCCTGCAGCTTGCG-3′), and IFNβ (5′-AGCTCCAAGAAAGGACGAACAT-3′; 5′-GCCCTGTAGGTGAGGTTGATCT-3′) were designed using Primer Express 2 software (Applied Biosystems, Foster City, CA, USA). Primers to murine STAT1 gene (5′-CGCGCATGCAAGTGGCATATAACT-3′; 5′-AAGCTCGAACCACTGTGACATCCT-3′) were designed using PrimerQuest software (Integrated DNA Technologies). Primers to ribosomal L32 (5′-TTCCTGGTCCACAACGTCAAG-3′; 5′-TGTGAGCGATCTCGGCAC-3′) were as previously described [17]. Threshold cycle numbers (Ct) were measured in the exponential phase for all samples. Relative abundance of sample genes was calculated using the ΔΔCt method relative to the L32 control gene [17]. mRNA abundance was normalized to the untreated samples of each genotype.
ChIP
ChIP assays were performed as described previously [41] using 5 µg of anti c-Jun or rabbit IgG control antibodies. The abundance of specific sequences in ChIP samples was quantitated using the SYBR Green dye detection method (Applied Biosystems, Warrington, UK). Primers used for PCR reactions were mIFN PRDIV (5′-ATTCCTCTGAGGCAGAAAGGACCA; 5′-GCAAGATGAGGCAAAGGCTGTCAA) and were designed using Primer Express 2 software. Threshold cycle values (Ct) were measured in the exponential phase, and promoter occupancy was calculated using the formula 2(Ct Ig − Ct c-Jun).
Statistical Analysis
Statistical significance was tested using one-way ANOVA testing with OriginLab 7.5 software (Northampton, MA, USA) or Prism Software Graphpad (La Jolla, CA, USA).
Viral Protection Assays
103 cells of each genotype were plated in duplicate wells in a 96 well plate and allowed to adhere. Media was replaced with fresh media containing murine EMCV (M.O.I of 0.1) and various concentrations of IFNγ (0–1,000 IU/mL) and cultured for 16 h. As controls, cells were cultured in fresh media alone (100% survival) or with EMCV alone (0% survival). Cells were washed in PBS, formalin fixed (10 min at RT), washed (twice with PBS), and stained in 0.5% Crystal Violet/20% methanol. Stained cells were extensively washed, crystal violet was solubilized in 10% acetic acid, and OD550 nm was recorded. Viability was calculated by comparison against a standard curve.
Supporting Information
STAT1 expression is decreased in c-Jun knockout cells. SDS-PAGE and Western blotting with antibodies against STAT1 was performed using whole cell extracts from an independently derived set of wild-type or c-Jun −/− MEFs. As a control, the expression of α-tubulin was also tested by Western blot.
(0.35 MB TIF)
IFNβ regulates the expression of STAT1. (A) c-Jun −/− MEFs were treated with various doses of IFNβ for 24 h, RNA was isolated, and expression of STAT1 was assessed by qRT-PCR. STAT1 expression in untreated wild-type MEFs was also assessed as a control. Histograms represent mean and error bars the SEM of three independent experiments and are expressed relative to the levels detected in c-Jun −/− cells (arbitrarily set as one). (B) c-Jun −/− MEFs were treated in the presence or absence of 10 IU IFNβ for 24 h, cell lysates were prepared, and expression of STAT1 was assessed by Western blot. (C) Wild-type MEFs were treated in the presence or absence of various doses of IFNβ for 24 h, RNA was isolated, and expression of STAT1 was assessed by qRT-PCR. Histograms represent mean and error bars the SEM of three independent experiments (arbitrarily set as one).
(0.81 MB TIF)
Expression of STAT1, 2, and 3 in IFNAR1 knockout cells. (A) The expression of STAT2 mRNA (top panel) and STAT3 protein (bottom panel) was determined in wild-type and IFNAR1 −/− MEFs by QRT-PCR and Western blotting, respectively. (B) Wild-type, IFNAR1 −/−, or c-Jun −/− MEFs were cultured in fresh media (white bars) or conditioned media from wild-type MEFs (black bars) for 16 h. RNA was extracted, cDNA synthesized, and STAT1 mRNA expression assessed by qRT-PCR. STAT1 mRNA expression was normalized to that of untreated IFNAR1−/− MEFs cultured in fresh media. Data are representative of three similar experiments.
(0.65 MB TIF)
Reconstitution of STAT1 in IFNAR1 knockout cells restores IFNγ-mediated upregulation of IFN response genes. Wild-type MEFs, IFNAR1−/− MEFs, and IFNAR1 −/− MEFs transduced with empty vector (IFNAR1−/− MSCV) or IFNAR1−/− MEFs transduced with HA-tagged STAT1α (IFNAR1−/− STAT1) were treated with 100 IU/mL IFNγ for 0, 1, or 6 h. RNA was extracted, cDNA synthesized, and qRT-PCR performed with primers specific for caspase 4 (CASP 4), CISH, CXCL11, and MYD88. mRNA levels are expressed relative to those of wild-type C57/BL6 (B6) splenocytes. Histograms represent the mean and error bars the standard error of four independent experiments (* p<0.05 for samples that were significantly induced).
(1.56 MB TIF)
Acknowledgments
The authors would like to acknowledge Dr. J. Oliaro and Drs. N. Waterhouse, I. Voskoboinik, and M. Bots for helpful discussions.
Abbreviations
- ChIP
chromatin immunoprecipitation
- EMCV
encephalomyocarditis virus
- EMSA
elecrophoretic mobility shift assay
- GAS
gamma activated sequence
- IFN
interferon
- IFNAR1
IFNα/β receptor1
- IFNGR
IFNγ receptor
- IL
interleukin
- ISG
IFN-stimulated gene
- JAK
Janus family kinase
- MEFs
murine embryonic fibroblasts
- PIAS
Protein Inhibitor of Activated STAT
- RNAi
RNA interference
Footnotes
The authors have declared that no competing interests exist.
This work was funded by National Health and Medical Research Council (NHMRC) program (454569) and project grants (506970). RWJ is a Pfizer Australia Senior Research Fellow and a Senior Research Fellow of the NHMRC of Australia. PJH is a Principal Research Fellow of NHMRC of Australia. JAT is a Senior Principal Research Fellow of NHMRC Australia. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Vogel S. N, Fertsch D. Endogenous interferon production by endotoxin-responsive macrophages provides an autostimulatory differentiation signal. Infect Immun. 1984;45:417–423. doi: 10.1128/iai.45.2.417-423.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hamilton J. A, Whitty G. A, Kola I, Hertzog P. J. Endogenous IFN-alpha beta suppresses colony-stimulating factor (CSF)-1-stimulated macrophage DNA synthesis and mediates inhibitory effects of lipopolysaccharide and TNF-alpha. J Immunol. 1996;156:2553–2557. [PubMed] [Google Scholar]
- 3.Marie I, Durbin J. E, Levy D. E. Differential viral induction of distinct interferon-a genes by positive feedback through interferon regulatory factor-7. EMBO J. 1998;17:6660–6669. doi: 10.1093/emboj/17.22.6660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Muller U, Steinhoff U, Reis L. F, Hemmi S, Pavlovic J, et al. Functional role of type I and type II interferons in antiviral defense. Science. 1994;264:1918–1921. doi: 10.1126/science.8009221. [DOI] [PubMed] [Google Scholar]
- 5.Takaoka A, Mitani Y, Suemori H, Sato M, Yokochi T, et al. Cross talk between interferon-gamma and -alpha/beta signaling components in caveolar membrane domains. Science. 2000;288:2357–2360. doi: 10.1126/science.288.5475.2357. [DOI] [PubMed] [Google Scholar]
- 6.Durbin J. E, Hackenmiller R, Simon M. C, Levy D. E. Targeted disruption of the mouse Stat1 gene results in compromised innate immunity to viral disease. Cell. 1996;84:443–450. doi: 10.1016/s0092-8674(00)81289-1. [DOI] [PubMed] [Google Scholar]
- 7.Meraz M. A, White J. M, Sheehan K. C, Bach E. A, Rodig S. J, et al. Targeted disruption of the Stat1 gene in mice reveals unexpected physiologic specificity in the JAK-STAT signaling pathway. Cell. 1996;84:431–442. doi: 10.1016/s0092-8674(00)81288-x. [DOI] [PubMed] [Google Scholar]
- 8.Kaplan D. H, Shankaran V, Dighe A. S, Stockert E, Aguet M, et al. Demonstration of an interferon gamma-dependent tumor surveillance system in immunocompetent mice. Proc Natl Acad Sci U S A. 1998;95:7556–7561. doi: 10.1073/pnas.95.13.7556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shankaran V, Ikeda H, Bruce A. T, White J. M, Swanson P. E, et al. IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature. 2001;410:1107–1111. doi: 10.1038/35074122. [DOI] [PubMed] [Google Scholar]
- 10.Pine R, Canova A, Schindler C. Tyrosine phosphorylated p91 binds to a single element in the ISGF2/IRF-1 promoter to mediate induction by IFN alpha and IFN gamma, and is likely to autoregulate the p91 gene. EMBO J. 1994;13:158–167. doi: 10.1002/j.1460-2075.1994.tb06245.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Miyagi T, Gil M. P, Wang X, Louten J, Chu W. M, et al. High basal STAT4 balanced by STAT1 induction to control type 1 interferon effects in natural killer cells. J Exp Med. 2007;204:2383–2396. doi: 10.1084/jem.20070401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Der S. D, Zhou A, Williams B. R. G, Silverman S. H. Identification of genes differentially regulated by interferon α, β and γ using oligonucleotide arrays. Proc Natl Acad Sci U S A. 1998;95:15623–15628. doi: 10.1073/pnas.95.26.15623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wong L. H, Sim H, Chatterjee-Kishore M, Hatzinisiriou I, Devenish R. J, et al. Isolation and characterization of a human STAT1 gene regulatory element. Inducibility by interferon (IFN) types I and II and role of IFN regulatory factor-1. J Biol Chem. 2002;277:19408–19417. doi: 10.1074/jbc.M111302200. [DOI] [PubMed] [Google Scholar]
- 14.Wong L. H, Krauer K. G, Hatzinisiriou I, Estcourt M. J, Hersey P, et al. Interferon-resistant human melanoma cells are deficient in ISGF3 components, STAT1, STAT2, and p48-ISGF3gamma. J Biol Chem. 1997;272:28779–28785. doi: 10.1074/jbc.272.45.28779. [DOI] [PubMed] [Google Scholar]
- 15.Levy D. E, Darnell J. E., Jr Stats: transcriptional control and biological impact. Nat Rev. 2002;3:651–662. doi: 10.1038/nrm909. [DOI] [PubMed] [Google Scholar]
- 16.Sadzak I, Schiff M, Gattermeier I, Glinitzer R, Sauer I, et al. Proc Natl Acad Sci U S A. 2008 Jul 1 105:8944–8949. doi: 10.1073/pnas.0801794105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gough D. J, Sabapathy K, Ko E. Y, Arthur H. A, Schreiber R. D, et al. A novel c-Jun-dependent signal transduction pathway necessary for the transcriptional activation of interferon gamma response genes. J Biol Chem. 2007;282:938–946. doi: 10.1074/jbc.M607674200. [DOI] [PubMed] [Google Scholar]
- 18.Maniatis T, Falvo J. V, Kim T. H, Kim T. K, Lin C. H, et al. Structure and function of the interferon-beta enhanceosome. Cold Spring Harb Symp Quant Biol. 1998;63:609–620. doi: 10.1101/sqb.1998.63.609. [DOI] [PubMed] [Google Scholar]
- 19.Fleetwood A. J, Dinh H, Cook A. D, Hertzog P. J, Hamilton J. A. GM-CSF- and M-CSF-dependent macrophage phenotypes display differential dependence on Type I interferon signaling. J Leuk Biol. 2009;86:411–421. doi: 10.1189/jlb.1108702. [DOI] [PubMed] [Google Scholar]
- 20.Gresser I, Maury C, Kress C, Blangy D, Maunoury M. T. Role of interferon in the pathogenesis of virus diseases in mice as demonstrated by the use of anti-interferon serum. VI. Polyoma virus infection. Int J Cancer. 1979;24:178–183. doi: 10.1002/ijc.2910240209. [DOI] [PubMed] [Google Scholar]
- 21.Sheehan K. C, Lai K. S, Dunn G. P, Bruce A. T, Diamond M. S, et al. Blocking monoclonal antibodies specific for mouse IFN-alpha/beta receptor subunit 1 (IFNAR-1) from mice immunized by in vivo hydrodynamic transfection. J Interferon Cytokine Res. 2006;26:804–819. doi: 10.1089/jir.2006.26.804. [DOI] [PubMed] [Google Scholar]
- 22.Thomas K. E, Galligan C. L, Newman R. D, Fish E. N, Vogel S. N. Contribution of interferon-beta to the murine macrophage response to the toll-like receptor 4 agonist, lipopolysaccharide. J Biol Chem. 2006;281:31119–31130. doi: 10.1074/jbc.M604958200. [DOI] [PubMed] [Google Scholar]
- 23.Falvo J. V, Parekh B. S, Lin C. H, Fraenkel E, Maniatis T. Assembly of a functional beta interferon enhanceosome is dependent on ATF-2-c-jun heterodimer orientation. Mol Cell Biol. 2000;20:4814–4825. doi: 10.1128/mcb.20.13.4814-4825.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ramana C. V, Gil M. P, Han Y, Ransohoff R. M, Schreiber R. D, et al. Stat1-independent regulation of gene expression in response to IFN-gamma. Proc Natl Acad Sci U S A. 2001;98:6674–6679. doi: 10.1073/pnas.111164198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Owczarek C. M, Hwang S. Y, Holland K. A, Gulluyan L. M, Tavaria M, et al. Cloning and characterization of soluble and transmembrane isoforms of a novel component of the murine type I interferon receptor, IFNAR 2. J Biol Chem. 1997;272:23865–23870. doi: 10.1074/jbc.272.38.23865. [DOI] [PubMed] [Google Scholar]
- 26.Varinou L, Ramsauer K, Karaghiosoff M, Kolbe T, Pfeffer K, et al. Phosphorylation of the Stat1 transactivation domain is required for full-fledged IFN-gamma-dependent innate immunity. Immunity. 2003;19:793–802. doi: 10.1016/s1074-7613(03)00322-4. [DOI] [PubMed] [Google Scholar]
- 27.Hata N, Sato M, Takaoka A, Asagiri M, Tanaka N, et al. Constitutive IFN-alpha/beta signal for efficient IFN-alpha/beta gene induction by virus. Biochem Biophys Res Comm. 2001;285:518–525. doi: 10.1006/bbrc.2001.5159. [DOI] [PubMed] [Google Scholar]
- 28.Dezfouli S, Hatzinisiriou I, Ralph S. J. Enhancing CTL responses to melanoma cell vaccines in vivo: synergistic increases obtained using IFNgamma primed and IFNbeta treated B7-1+ B16-F10 melanoma cells. Immunol Cell Biol. 2003;81:459–471. doi: 10.1046/j.0818-9641.2003.01189.x. [DOI] [PubMed] [Google Scholar]
- 29.Pace J. L. Synergistic interactions between IFN-gamma and IFN-beta in priming murine macrophages for tumor cell killing. J Leukoc Biol. 1988;44:514–520. doi: 10.1002/jlb.44.6.514. [DOI] [PubMed] [Google Scholar]
- 30.van den Broek M. F, Muller U, Huang S, Zinkernagel R. M, Aguet M. Immune defence in mice lacking type I and/or type II interferon receptors. Immunol Rev. 1995;148:5–18. doi: 10.1111/j.1600-065x.1995.tb00090.x. [DOI] [PubMed] [Google Scholar]
- 31.Mitani Y, Takaoka A, Kim S. H, Kato Y, Yokochi T, et al. Cross talk of the interferon-alpha/beta signalling complex with gp130 for effective interleukin-6 signalling. Genes Cells. 2001;6:631–640. doi: 10.1046/j.1365-2443.2001.00448.x. [DOI] [PubMed] [Google Scholar]
- 32.Sharif M. N, Tassiulas I, Hu Y, Mecklenbrauker I, Tarakhovsky A, et al. IFN-alpha priming results in a gain of proinflammatory function by IL-10: implications for systemic lupus erythematosus pathogenesis. J Immunol. 2004;172:6476–6481. doi: 10.4049/jimmunol.172.10.6476. [DOI] [PubMed] [Google Scholar]
- 33.Zhao W, Lee C, Piganis R, Plumlee C, de Weerd N, et al. A conserved IFN-alpha receptor tyrosine motif directs the biological response to type I IFNs. J Immunol. 2008;180:5483–5489. doi: 10.4049/jimmunol.180.8.5483. [DOI] [PubMed] [Google Scholar]
- 34.Gil M. P, Salomon R, Louten J, Biron C. A. Modulation of STAT1 protein levels: a mechanism shaping CD8 T-cell responses in vivo. Blood. 2006;107:987–993. doi: 10.1182/blood-2005-07-2834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Qing Y, Stark G. R. Alternative activation of STAT1 and STAT3 in response to interferon-gamma. J Biol Chem. 2004;279:41679–41685. doi: 10.1074/jbc.M406413200. [DOI] [PubMed] [Google Scholar]
- 36.Alexander W. S, Starr R, Fenner J. E, Scott C. L, Handman E, et al. SOCS1 is a critical inhibitor of interferon gamma signaling and prevents the potentially fatal neonatal actions of this cytokine. Cell. 1999;98:597–608. doi: 10.1016/s0092-8674(00)80047-1. [DOI] [PubMed] [Google Scholar]
- 37.Fenner J. E, Starr R, Cornish A. L, Zhang J. G, Metcalf D, et al. Suppressor of cytokine signaling 1 regulates the immune response to infection by a unique inhibition of type I interferon activity. Nat Immunol. 2006;7:33–39. doi: 10.1038/ni1287. [DOI] [PubMed] [Google Scholar]
- 38.Hwang S. Y, Hertzog P. J, Holland K. A, Sumarsono S. H, Tymms M. J, et al. A null mutation in the gene encoding a type I interferon receptor component eliminates antiproliferative and antiviral responses to interferons alpha and beta and alters macrophage responses. Proc Natl Acad Sci U S A. 1995;92:11284–11288. doi: 10.1073/pnas.92.24.11284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hardy M. P, Owczarek C. M, Trajanovska S, Liu X, Kola I, et al. The soluble murine type I interferon receptor Ifnar-2 is present in serum, is independently regulated, and has both agonistic and antagonistic properties. Blood. 2001;97:473–482. doi: 10.1182/blood.v97.2.473. [DOI] [PubMed] [Google Scholar]
- 40.Sheehan K. C, Calderon J, Schreiber R. D. Generation and characterization of monoclonal antibodies specific for the human IFN-gamma receptor. J Immunol. 1988;140:4231–4237. [PubMed] [Google Scholar]
- 41.Baker E. K, Johnstone R. W, Zalcberg J. R, El-Osta A. Epigenetic changes to the MDR1 locus in response to chemotherapeutic drugs. Oncogene. 2005;24:8061–8075. doi: 10.1038/sj.onc.1208955. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
STAT1 expression is decreased in c-Jun knockout cells. SDS-PAGE and Western blotting with antibodies against STAT1 was performed using whole cell extracts from an independently derived set of wild-type or c-Jun −/− MEFs. As a control, the expression of α-tubulin was also tested by Western blot.
(0.35 MB TIF)
IFNβ regulates the expression of STAT1. (A) c-Jun −/− MEFs were treated with various doses of IFNβ for 24 h, RNA was isolated, and expression of STAT1 was assessed by qRT-PCR. STAT1 expression in untreated wild-type MEFs was also assessed as a control. Histograms represent mean and error bars the SEM of three independent experiments and are expressed relative to the levels detected in c-Jun −/− cells (arbitrarily set as one). (B) c-Jun −/− MEFs were treated in the presence or absence of 10 IU IFNβ for 24 h, cell lysates were prepared, and expression of STAT1 was assessed by Western blot. (C) Wild-type MEFs were treated in the presence or absence of various doses of IFNβ for 24 h, RNA was isolated, and expression of STAT1 was assessed by qRT-PCR. Histograms represent mean and error bars the SEM of three independent experiments (arbitrarily set as one).
(0.81 MB TIF)
Expression of STAT1, 2, and 3 in IFNAR1 knockout cells. (A) The expression of STAT2 mRNA (top panel) and STAT3 protein (bottom panel) was determined in wild-type and IFNAR1 −/− MEFs by QRT-PCR and Western blotting, respectively. (B) Wild-type, IFNAR1 −/−, or c-Jun −/− MEFs were cultured in fresh media (white bars) or conditioned media from wild-type MEFs (black bars) for 16 h. RNA was extracted, cDNA synthesized, and STAT1 mRNA expression assessed by qRT-PCR. STAT1 mRNA expression was normalized to that of untreated IFNAR1−/− MEFs cultured in fresh media. Data are representative of three similar experiments.
(0.65 MB TIF)
Reconstitution of STAT1 in IFNAR1 knockout cells restores IFNγ-mediated upregulation of IFN response genes. Wild-type MEFs, IFNAR1−/− MEFs, and IFNAR1 −/− MEFs transduced with empty vector (IFNAR1−/− MSCV) or IFNAR1−/− MEFs transduced with HA-tagged STAT1α (IFNAR1−/− STAT1) were treated with 100 IU/mL IFNγ for 0, 1, or 6 h. RNA was extracted, cDNA synthesized, and qRT-PCR performed with primers specific for caspase 4 (CASP 4), CISH, CXCL11, and MYD88. mRNA levels are expressed relative to those of wild-type C57/BL6 (B6) splenocytes. Histograms represent the mean and error bars the standard error of four independent experiments (* p<0.05 for samples that were significantly induced).
(1.56 MB TIF)