Abstract
Erythroderma is an unusual cutaneous finding associated with dermatomyositis. There are only five cases of erythrodermic dermatomyositis reported in the English literature. We treated a case of erythrodermic dermatomyositis associated with a Bormann type 1 gastric cancer. The patient had a generalized, erythematous scaly eruption consistent with erythroderma and Gottron's papules as well as a heliotrope rash; these are the hallmark skin manifestations of dermatomyositis.
Keywords: Dermatomyositis, Erythroderma, Gastric cancer, Malignancy
INTRODUCTION
Dermatomyositis is a rare systemic autoimmune disorder characterized by specific cutaneous manifestations and symmetric proximal muscle weakness. In addition to the skin and skeletal muscles, a variety of internal organs, such as the heart, lung, gastrointestinal tract, and eyes, can also be affected1,2. Life threatening complications can develop including internal malignancies, calcinosis, and vasculopathy1,2. Erythroderma is defined as a diffuse scaly erythema of the skin involving more than 90 percent of the total body skin surface area3. It can be caused by a variety of cutaneous and systemic diseases. It has been reported that pre-existing skin diseases including psoriasis, atopic dermatitis, drug reactions, seborrheic dermatitis, and congenital dermatoses play a role in the development of erythroderma in approximately 52% of the cases3,4. In addition, a variety of drugs and malignancies, including solid tumors and lymphoproliferative diseases, can cause erythroderma. Among the malignancies, cutaneous T-cell lymphoma is the most common cause of erythroderma. In cases of paraneoplastic erythroderma, the cutaneous finding of erythroderma may occur as the only symptom of a malignancy. Therefore, patients with a history of recurrent erythroderma without a known cause should be evaluated periodically for an occult malignancy3,5.
Erythroderma can present as a cutaneous finding associated with dermatomyositis3; however, this is unusual. We could find only five cases of erythrodermic dermatomyositis in the English literature (Table 1). Among the documented cases, two were associated with internal malignancies of the stomach4 and liver6. Here we report a case of erythrodermic dermatomyositis associated with gastric cancer.
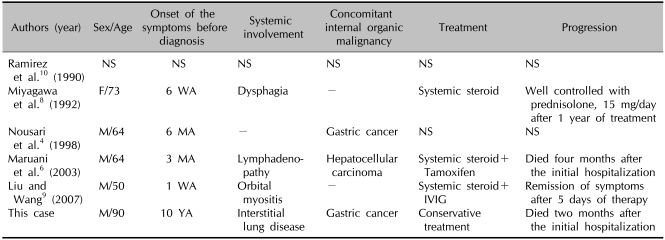
Table 1.
Reported cases of erythrodermic dermatomyositis
IVIG: intravenous immunoglobulin, MA: months ago, NS: not stated, WA: weeks ago, YA: years ago.
CASE REPORT
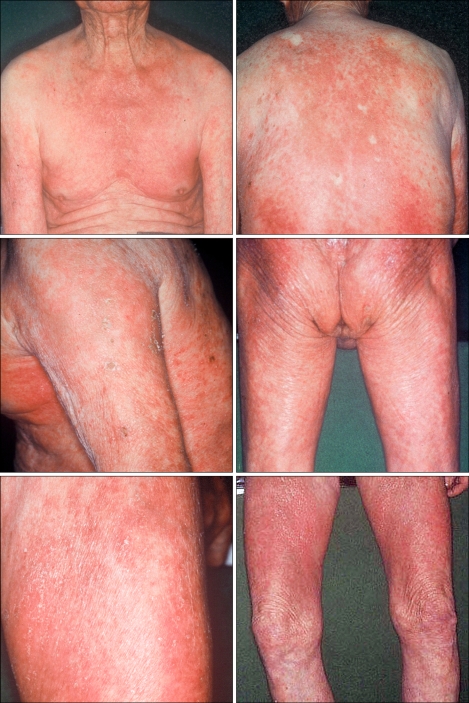
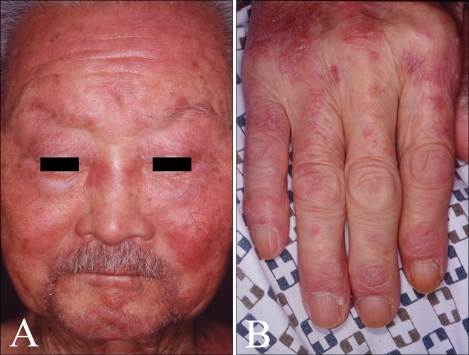
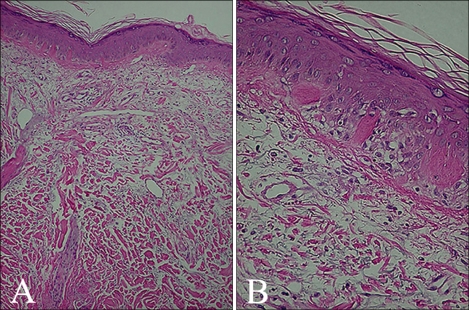
A 90-year-old male with a 10-year history of generalized pruritus presented with aggravation of his skin lesions and weakness of the proximal extremities. The skin lesions were observed as violaceous to erythematous, confluent, scaly patches involving more than 90 percent of the total body area (Fig. 1). Periorbital, edematous, violaceous erythema and scaly papules affecting the extensor aspects of the interphalangeal joints of the hands were noted (Fig. 2). The patient complained of worsening of the skin lesions and pruritus after exposure to sunlight. In addition, he complained of difficulty in raising his hands over the level of his shoulders, sitting down on a chair and standing up, which started six years ago and has gradually worsened. Histopathology of the erythematous scaly patch showed epidermal atrophy, hydropic degeneration of the basal cell layer, papillary dermal edema, and a mild dermal perivascular lymphocytic infiltration (Fig. 3). Laboratory investigations demonstrated elevated serum creatine kinase (452 U/L, reference: 25~90 U/L), lactate dehydrogenase (587 U/L, reference: 100~190 U/L), and aspartate transaminase levels (68 U/L, reference: 0~35 U/L). The electromyography revealed short duration muscle unit action potentials in the deltoid and iliopsoas muscles. A muscle biopsy of the deltoid muscle showed small atrophic myofibers and focal inflammatory infiltration suggestive of an inflammatory myopathy. Additional history revealed a medical history of untreated gastric cancer that was diagnosed five years previously. The gastroendoscopy showed a Bormann type 1 gastric cancer. The patient was transferred to the neurology department and conservative management was provided for the diagnosis of dermatomyositis. Two months later, the patient died of complications associated with interstitial lung disease and a malignant pleural effusion.
Fig. 1.
Erythematous scaly patches involving more than 90 percent of the total body area.
Fig. 2.
Hallmark cutaneous skin manifestations of dermatomyositis including (A) heliotrope rash and (B) Gottron's papules were noted.
Fig. 3.
Histopathology of the skin lesion showed a hyperkeratotic stratum corneum, epidermal atrophy, hydropic degeneration of the basal cell layer, papillary dermal edema, dermal mild perivascular lymphocytic infiltration and increased mucin deposition (H&E, A: ×40, B: ×200).
DISCUSSION
The idiopathic inflammatory myopathies are a heterogeneous group of autoimmune disorders that predominately target the skin and skeletal musculature. This term previously referred to cases with polymyositis/dermatomyositis1,2. The idiopathic inflammatory myopathy classification system does not include cutaneous-only (amyopathic) or cutaneous predominant (hypomyopathic) subsets of dermatomyositis. Clinically amyopathic dermatomyositis is a designation that refers both to amyopathic and hypomyopathic dermatomyositis1,2. As the hallmark cutaneous skin lesions are the only clue to the diagnosis of dermatomyositis until the myopathic symptoms become evident; dermatologists are especially interested this type of dermatomyositis. From this point of view, Sontheimer1 proposed a more inclusive categorization of disease referred to as 'idiopathic inflammatory dermatomyopathies' encompassing the subset of clinically amyopathic dermatomyositis.
In the case presented here, the diagnosis of dermatomyositis was made using the criteria of Bohan et al.7 including the typical skin rash, symmetrical proximal muscle weakness with or without dysphagia or respiratory muscle involvement, abnormal findings on the muscle biopsy specimen, elevation of the skeletal muscle-derived enzymes, and abnormal electromyogram. If more than four criteria, as described above, are fulfilled, a definite diagnosis of dermatomyositis can be made. The patient presented here met all of the above criteria. The hallmark skin manifestations include heliotrope rash and Gottron's papules; these are characteristic and pathognomonic cutaneous findings associated with dermatomyositis2. Other cutaneous features include poikiloderma, malar erythema, periungual telangiectasia, and photosensitivity8.
There are five reported cases of dermatomyositis presenting as erythroderma in the English literature (Table 1)4,6,8-10. Even though only a few cases have been reported in the literature, erythroderma is not likely to be a rare form of dermatomyositis. Maruani et al.6 suggested that the terms used in previous reports, 'widespread erythema'11, 'diffuse rash'7, and 'generalized eczema'10 were describing cases with erythroderma. In addition, it is noteworthy that two of the five documented cases were associated with internal malignancies, gastric cancer4 and hepatocellular carcinoma6. It is well known that dermatomyositis is highly associated with malignancies1,2. Since the concomitant malignancy indicates a poor prognosis with dermatomyositis1, many studies have attempted to identify the clinical features associated with a greater risk of malignancy in patients with dermatomyositis. Advanced age12, normal serum creatine kinase level13, increased erythrocyte sedimentation rate14, presence of cutaneous vasculitis15, and cutaneous necrosis14 are risk factors. However, these risk factors have not been confirmed by all studies. For example, Fudman and Schnitzer13 reported that a normal creatine kinase level was associated with a poor prognosis. However, Sparsa et al.16 reported that patients with a higher level of creatine kinase were more likely to have cancer.
Maruani et al.6 proposed that erythroderma was a predictive factor for a malignancy in patients with dermatomyositis. In this context, our case is a good example that supports the report by Maruani et al.6. The frequency of malignancy in the non-erythrodermic dermatomyositis has been reported to be between 15% and 45%6, which is not different from cases of erythrodermic dermatomyositis. Thus, additional evidence is needed to confirm that erythroderma is a predictive factor for a malignancy in patients with dermatomyositis.
Yamashita et al.17 reported dermatomyositis as a paraneoplastic manifestation secondary to gastric cancer. In this case, the symptoms of dermatomyositis completely resolved after removal of the tumor without further treatment for dermatomyositis itself. Similar to this report, we can speculate that erythroderma developed as a paraneoplastic symptom in the case reported here. However, more data is needed to clarify the relationship of dermatomyositis, erythroderma and internal malignancies. In our case, the long history of systemic skin lesions implied that the erythroderma preceded the gastric cancer and was secondary to the dermatomyositis.
Herein we report an additional case of erythrodermic dermatomyositis associated with internal malignancy. Although no precise association has been revealed between erythroderma and internal malignancy in dermatomyositis, additional case reports will be helpful to clarify the relationship.
In conclusion, erythroderma can be a cutaneous manifestation of dermatomyositis; in patients with these findings an internal malignancy should be ruled out.
References
- 1.Sontheimer RD. Dermatomyositis: an overview of recent progress with emphasis on dermatologic aspects. Dermatol Clin. 2002;20:387–408. doi: 10.1016/s0733-8635(02)00021-9. [DOI] [PubMed] [Google Scholar]
- 2.Sontheimer RD, Costner MI. Dermatomyositis. In: Wolff K, Goldsmith LA, Katz SI, Gilchrest BA, Paller AS, Leffell DJ, editors. Fitzpatrick's dermatology in general medicine. 7th ed. New York: McGraw-Hill; 2008. pp. 1536–1553. [Google Scholar]
- 3.Grant-Kels JM, Bernstein ML, Rothe MJ. Exfoliative dermatitis. In: Wolff K, Goldsmith LA, Katz SI, Gilchrest BA, Paller AS, Leffell DJ, editors. Fitzpatrick's dermatology in general medicine. 7th ed. New York: McGraw-Hill; 2008. pp. 225–232. [Google Scholar]
- 4.Nousari HC, Kimyai-Asadi A, Spegman DJ. Paraneoplastic dermatomyositis presenting as erythroderma. J Am Acad Dermatol. 1998;39:653–654. doi: 10.1016/s0190-9622(98)70022-2. [DOI] [PubMed] [Google Scholar]
- 5.Turchin I, Barankin B. Dermacase. Erythroderma secondary to cutanous T-cell lymphoma. Can Fam Physician. 2005;51:963, 971–973. [PMC free article] [PubMed] [Google Scholar]
- 6.Maruani A, Armingaud P, Nseir A, Luthier F, Esteve E. Erythroderma and multiple cutaneous necrosis revealing a dermatomyositis. Ann Dermatol Venereol. 2003;130:353–356. [PubMed] [Google Scholar]
- 7.Bohan A, Peter JB, Bowman RL, Pearson CM. Computer-assisted analysis of 153 patients with polymyositis and dermatomyositis. Medicine (Baltimore) 1977;56:255–286. doi: 10.1097/00005792-197707000-00001. [DOI] [PubMed] [Google Scholar]
- 8.Miyagawa S, Okazaki A, Minowa R, Shirai T. Dermatomyositis presenting as erythroderma. J Am Acad Dermatol. 1992;26:489–490. doi: 10.1016/s0190-9622(08)80580-4. [DOI] [PubMed] [Google Scholar]
- 9.Liu ZH, Wang XD. Acute-onset adult dermatomyositis presenting with erythroderma and diplopia. Clin Exp Dermatol. 2007;32:751–752. doi: 10.1111/j.1365-2230.2007.02447.x. [DOI] [PubMed] [Google Scholar]
- 10.Ramirez G, Asherson RA, Khamashta MA, Cervera R, D'Cruz D, Hughes GR. Adult-onset polymyositis-dermatomyositis: description of 25 patients with emphasis on treatment. Semin Arthritis Rheum. 1990;20:114–120. doi: 10.1016/0049-0172(90)90024-a. [DOI] [PubMed] [Google Scholar]
- 11.Pierson JC, Taylor JS. Erythrodermic dermatomyositis. J Am Acad Dermatol. 1993;28:136. doi: 10.1016/s0190-9622(08)80870-5. [DOI] [PubMed] [Google Scholar]
- 12.Chen YJ, Wu CY, Shen JL. Predicting factors of malignancy in dermatomyositis and polymyositis: a case-control study. Br J Dermatol. 2001;144:825–831. doi: 10.1046/j.1365-2133.2001.04140.x. [DOI] [PubMed] [Google Scholar]
- 13.Fudman EJ, Schnitzer TJ. Dermatomyositis without creatine kinase elevation. A poor prognostic sign. Am J Med. 1986;80:329–332. doi: 10.1016/0002-9343(86)90036-7. [DOI] [PubMed] [Google Scholar]
- 14.Basset-Seguin N, Roujeau JC, Gherardi R, Guillaume JC, Revuz J, Touraine R. Prognostic factors and predictive signs of malignancy in adult dermatomyositis. A study of 32 cases. Arch Dermatol. 1990;126:633–637. [PubMed] [Google Scholar]
- 15.Feldman D, Hochberg MC, Zizic TM, Stevens MB. Cutaneous vasculitis in adult polymyositis/dermatomyositis. J Rheumatol. 1983;10:85–89. [PubMed] [Google Scholar]
- 16.Sparsa A, Liozon E, Herrmann F, Ly K, Lebrun V, Soria P, et al. Routine vs extensive malignancy search for adult dermatomyositis and polymyositis: a study of 40 patients. Arch Dermatol. 2002;138:885–890. doi: 10.1001/archderm.138.7.885. [DOI] [PubMed] [Google Scholar]
- 17.Yamashita K, Hosokawa M, Hirohashi S, Arimura Y, Endo T, Denno R, et al. Epstein-Barr virus-associated gastric cancer in a patient with dermatomyositis. Intern Med. 2001;40:96–99. doi: 10.2169/internalmedicine.40.96. [DOI] [PubMed] [Google Scholar]