Abstract
The endoderm germ layer contributes to the respiratory and gastrointestinal tracts, and all of their associated organs. Over the past decade, studies in vertebrate model organisms; including frog, fish, chick, and mouse; have greatly enhanced our understanding of the molecular basis of endoderm organ development. We review this progress with a focus on early stages of endoderm organogenesis including endoderm formation, gut tube morphogenesis and patterning, and organ specification. Lastly, we discuss how developmental mechanisms that regulate endoderm organogenesis are used to direct differentiation of embryonic stem cells into specific adult cell types, which function to alleviate disease symptoms in animal models.
Keywords: digestive system, foregut, liver, pancreas, intestine, nodal
INTRODUCTION
The definitive endoderm (DE) was first defined as the innermost tissue or germ layer found in all metazoan embryos. It gives rise to a vast array of highly specialized epithelial cell types that line the respiratory and digestive systems; and contributes to associated organs such as thyroid, thymus, lungs, liver, biliary system, and pancreas. In the adult, endodermally derived organs provide many essential functions including: gas exchange, digestion, nutrient absorption, glucose homeostasis, detoxification, and blood clotting. Perturbations in endodermal organ function are the underlying cause of thousands of human diseases that afflict millions of people every year. Basic studies of endoderm organ formation have proven invaluable for understanding the genetic basis of many human congenital diseases, and continued research will probably make it possible to grow endoderm organ tissue in vitro for future transplantation-based therapies, reviewed in Spence & Wells (2007).
Toward these goals, much has been learned about endoderm organogenesis over the past 20 years. The segregation of the three primary germ layers, the endoderm, mesoderm, and ectoderm, occurs during gastrulation; and it is one of the first cell fate decisions that is made in development. Increasing evidence suggests that the endoderm and mesoderm arise from a transient common precursor cell population referred to as mesendoderm. Mesendoderm induction and commitment to the endodermal lineage are controlled by an evolutionarily conserved gene regulatory network, which consists of Nodal growth factor signaling and a core group of downstream transcription factors.
After gastrulation, a series of morphogenetic movements transforms the naïve endoderm into a primitive gut tube that is surrounded by mesoderm. During this period, the gut tube becomes regionalized along the dorsal-ventral (D-V) and anterior-posterior (A-P) axes into broad foregut, midgut, and hindgut domains that can be observed at the molecular level by restricted gene expression patterns. Endoderm patterning is controlled by a series of reciprocal interactions with nearby mesoderm tissues. As development proceeds, broad gene expression patterns within the foregut, midgut, and hindgut become progressively refined into precise domains in which specific organs will form. The foregut gives rise to the esophagus, trachea, stomach, lungs, thyroid, liver, biliary system, and pancreas; whereas the midgut forms the small intestine and the hindgut forms the large intestine (Figure 1). Organ buds develop as outgrowths of endoderm epithelium that intermingle with the surrounding mesenchyme, and together these proliferate and ultimately differentiate during fetal development into functional organs. During organ formation, cell identity and the tissue morphogenesis must be tightly coordinated. These processes are controlled by many growth factor pathways including FGF, BMP, Wnt, retinoic acid (RA), Hedgehog, and Notch, which play multiple stage-specific roles during endoderm organogenesis.
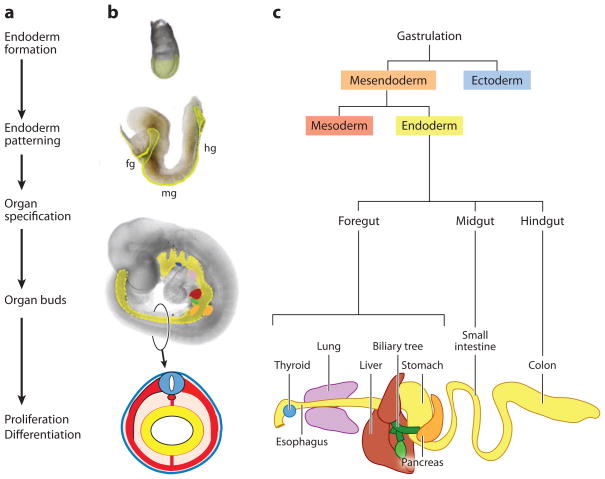
Figure 1.
Overview and timeline of endoderm organ formation. (a) The major events in endoderm organ formation are listed in chronological order and (b) illustrated with images of mouse embryos at e7.5 (top), e8.5, and e9.5 of development, with the endoderm shaded in (yellow). A schematic of a cross section through the e9.5 embryo illustrates the characteristic arrangement of the germ layers with the endoderm lining the gut tube (yellow), surrounded by mesoderm (red), and ectoderm (blue). (c) Endoderm cell lineages projected on to a schematic of the gastrointestinal tract. Fg; foregut, mg; midgut and hg; hindgut. The mouse embryo images are modified with permission from Zorn (2008).
This review summarizes our current understanding of endoderm organ development in vertebrates, from the establishment of the germ layers until organ bud formation. We highlight molecular mechanisms that are evolutionarily conserved between species and the emerging principles that can be drawn from these comparative studies. Finally, we discuss how this information has enabled researchers to direct the differentiation of embryonic stem cells into tissues such as hepatocytes or pancreatic β-cells, which represent a renewable source of therapeutic tissue for transplantation-based therapies.
DESCRIPTIVE EMBRYOLOGY OF THE ENDODERM
Fate mapping studies and embryological experiments in Xenopus, zebrafish, mouse, and chickens have defined which cells in the embryo give rise to the endoderm (Figure 2); and they have described the movements of those cells as they form a primitive gut tube (Figure 3). Although there are morphological differences in how these different species produce endoderm, they use a highly conserved molecular pathway to generate strikingly similar primitive gut tubes.
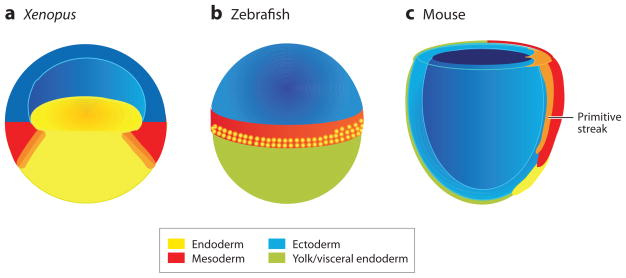
Figure 2.
Fate maps of (a) a Xenopus blastula, (b) a zebrafish blastula, and (c) an early streak-stage mouse gastrula, showing the presumptive endoderm (yellow), mesoderm (red) and ectoderm (blue). (a) The Xenopus blastula is depicted in section through the middle of the embryo to expose the bastocoel cavity, the putative mesendoderm (orange) at the border between the yolky vegetal endoderm cells, and the ring of equatorial mesoderm. The Xenopus blastula ectoderm is often referred to as the animal cap. (b) The external surface of a zebrafish blastula just prior to gastrulation is shown with the epiblast (also known as the blastoderm) sitting on top of the large yolk cell (green). The endoderm precursor cells are located in the 2–4 rows of marginal cells that are closest to the yolk (with proportionally more on the future dorsal—right side); and these are interspersed with mesoderm precursors that are found up to 8-cell rows from the margin. (c) The cut-away view of an early streak-stage mouse gastrula shows the definitive endoderm (yellow) emerging from the anterior end of the primitive streak (orange) and displacing the visceral endoderm (green). It is unclear if a transient mesendoderm progenitor resides within the primitive streak (orange).
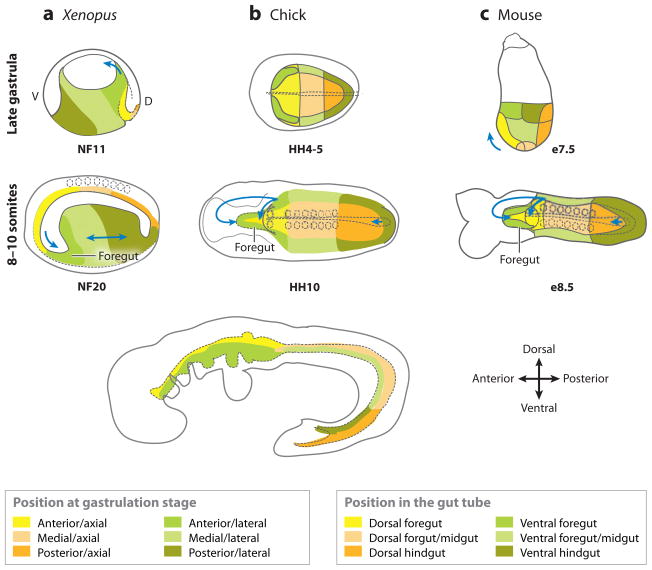
Figure 3.
Fate maps of (a) Xenopus, (b) chick, and (c) mouse embryos from the late gastrula to the early-somite stage. The (top panels) that show late-gastrula stage in the Xenopus (NF11) and mouse (e7.5) are a lateral view; and the chick (HH4) is a ventral view. The (dotted line) in the chick shows the developing notochord. The colored domains roughly correspond to the fate-map studies. The Xenopus embryos are depicted in mid-saggital section. The (middle panels) are a ventral view of 8–10 somite stage embryos. The (arrows) indicate the folding/migration direction of the foregut and hindgut; the (dotted circles) are somites; the (dotted line) along the midline is the notochord; and the (dotted line) in the anterior outlines the developing foregut. The (bottom panel) is a schematic showing a lateral view of an E9.5 mouse embryo. The colored domains shown at the bottom indicate the domains of the gastrula-stage embryos that roughly correspond with the equivalent domains of the gut tube. A, anterior; P, posterior; d, dorsal; v, ventral.
Xenopus
In the Xenopus blastula, prospective germ layers are arranged in a gradient along the animal-vegetal (top-bottom) axis (Figure 2a). The endoderm originates from the yolky vegetal cells, ectoderm is from the animal region on top of the bastocoel cavity, and the presumptive mesoderm is induced in a ring of equatorial tissue by signals from the vegetal cells (Dale & Slack 1987, Moody 1987, Nieuwkoop 1969). Initially, the presumptive endoderm and mesoderm territories overlap, and equatorial cells express both mesoderm and endoderm genes. However, by the late-gastrula stage, the domains are mutually exclusive (Lemaire et al. 1998, Wardle & Smith 2004). Single-cell transplantation experiments confirm that germ layer commitment is progressive; at the blastula stage, individual vegetal cells can contribute to all three germ layers, but in the gastrula they are committed to the endoderm lineage (Wylie et al. 1987).
In Xenopus, gastrulation initiates with the formation of the blastopore lip in the dorsal-anterior mesendoderm. The dorsal blastopore lip is the site of the Spemann organizer, which is analogous to the anterior primitive streak in amniotes and the shield in fish. The organizer has important roles in patterning all three germ layers along the dorsal-ventral and anterior-posterior axes. As gastrulation proceeds, the endoderm and mesoderm become internalized, and the leading edge of the dorsal-anterior endoderm migrates to the position of the future ventral foregut (Figure 3a) (Keller 1991). In the post-gastrula Xenopus embryo, the majority of the endoderm mass is located ventrally, and the presumptive organ domains are arranged along the A-P axis consistent with their final position in the gut (Figure 3a). During somite stages of development (stages 15–30), the mass of endodermal tissue elongates by radial cell intercalation and eventually cavitates to form a primitive gut tube (Chalmers & Slack 2000).
Zebrafish
In pregastrula zebrafish embryos, the epiblast is an inverted cup of cells that sits on top of a large yolk cell. The prospective germ layers are arranged along the animal-vegetal axis of the epiblast, with the endoderm progenitors restricted to the four rows of marginal cells closest to the yolk (Figure 2b). Mesoderm precursors are intermingled with the endoderm progenitors, but extend up to 8-cells from the yolk margin, and the ectoderm is derived from the animal portion of the epiblast (Kimmel et al. 1990, Warga & Nusslein-Volhard 1999). Single-cell labeling and transplantation experiments indicate that prior to gastrulation, single marginal cells can populate both endoderm and mesoderm, suggesting a common bi-potential mesendoderm progenitor (Ober et al. 2003, Schier & Talbot 2005).
During gastrulation, the marginal cells involute under the epiblast to form an internal layer between the epiblast and the yolk cell. Endoderm cells are the first to involute and migrate anteriorly to form a dispersed monolayer. The intervening cells between endoderm and the noninvoluting epiblast become mesoderm (Kimmel et al. 1990). Fate-mapping studies indicate that the presumptive organ domains are arranged in broad, overlapping regions that roughly correspond to their final anterior-posterior position in the GI tract (Warga & Nusslein-Volhard 1999). During early-somite stages of development, the single cell-layered endoderm sheet converges on the dorsal midline to form an endodermal rod, from which organ buds will emerge, and which will later cavitate to form a gut tube (Ober et al. 2003).
Mice and Chicks
In amniote embryos (mammals and birds), gastrulation initiates in an epithelial layer called the epiblast. Cells in the epiblast undergo an epithelial to mesenchymal transition (EMT), migrate through the primitive streak (PS), and incorporate in the middle (mesoderm) or outer (endoderm) layer. The presumptive definitive endoderm (DE) cells invade and displace an outer layer of extraembryonic tissue cells, the hypoblast in chick and the visceral endoderm (VE) in mouse, which form supporting structures such as the yolk sac. Lineage tracing and later studies with embryonic stem cells suggest that some endoderm derives from a transient mesendoderm progenitor in the streak. The first cells to exit the primitive streak migrate toward the anterior (Kimura et al. 2006, Lawson & Schoenwolf 2003, Lawson & Pedersen 1987), giving rise to medial and lateral anterior definitive endoderm (ADE) and axial mesoderm (Figure 2c). Endoderm that exits the PS later contributes to more posterior endoderm. At the end of gastrulation, the DE is a sheet of cells that surrounds the external surface of the mouse embryo. Early cell labeling studies, combined with molecular marker analysis indicated that DE from the primitive streak displaces the VE cells into the extraembryonic domain. However recent evidence suggests that some VE cells persist in the DE layer even during primitive gut tube formation at E8.5–9.5 (Kwon et al. 2008).
Lineage tracing of the late gastrula stage endoderm has provided insight on how a two-dimensional sheet of cells forms the primitive gut tube (E7.5 in mouse, HH4 in chick) (Kimura et al. 2006, Lawson & Schoenwolf 2003, Lawson et al. 1986, Rosenquist 1971, Tam et al. 2007, Tremblay & Zaret 2005). Morphogenesis begins when the epithelial sheet folds over at the anterior and posterior ends, forming foregut and hindgut pockets. Anterior axial and lateral endoderm fold toward the ventral midline forming the foregut, which then moves caudally like the cresting of a wave (Figure 3); until it hits the hindgut wave, which is expanding rostrally (Lawson et al. 1986). Remarkably, foregut organs such as the pancreas and liver are derived from different populations of medial and lateral endoderm that converge during foregut morphogenesis (Franklin et al. 2008, Tremblay & Zaret 2005). Similar bilateral organ domains have also been described in zebrafish (Ober et al. 2003).
ENDODERM FORMATION
Mesendoderm Induction by Nodal Signaling
In all vertebrates, the Nodal signaling pathway is necessary and sufficient to initiate endoderm and mesoderm development, and it is required for proper gastrulation and axial patterning. Nodal ligands are members of the TGFβ family of secreted growth factors. Whereas there is a single Nodal gene in mice, there are five nodal-related genes in Xenopus (xnr1,2,4,5,6); and two zebrafish genes, squint (sqt/ndr1) and cyclops (cyc/ndr2), that are all implicated in mesendoderm development. Nodal ligands signal via a complex of type I (Alk4 or Alk7) and type II (ActRIIA or ActRIIB) transmembrane serine-threonine kinase receptors; and an EGF-CFC family coreceptor (Cripto or Cryptic in mouse, Oep in zebrafish, and FRL/Xcr1–3 in Xenopus). The activated Alk4/7 phosphorylates the cytosolic proteins Smad2 or Smad3. Phosphorylated Smad2/3 then binds to Smad4, and translocates to the nucleus, where it associates with DNA-binding transcription factors; such as the Fork head box h1 (Foxh1/FAST1) or Mix-like homeodomain proteins; to stimulate the transcription of mesendoderm genes. In all species, examined loss-of-function in any core pathway component results in compromised mesendoderm development (Shen 2007).
Many studies support a model in which high levels of Nodal signaling promote endoderm development, whereas lower doses specify mesoderm identity. Although it was first demonstrated in Xenopus explant experiments (Clements et al. 1999, Green & Smith 1990), genetic and biochemical manipulations that progressively reduce endogenous Nodal activity demonstrate that this dose-dependent role is conserved in fish, mouse, and humans (D’Amour et al. 2005, Shen 2007, Zorn & Wells 2007). In all species, endoderm cells develop in close proximity to the Nodal source and require a sustained period to be specified (Aoki et al. 2002, Ben-Haim et al. 2006, Hagos & Dougan 2007). In Xenopus and zebrafish embryos, the endoderm arises from nodal-expressing cells, and the mesoderm is induced in the adjacent tissue. In mouse, DE comes from the anterior primitive streak, the source of Nodal (Figure 4).
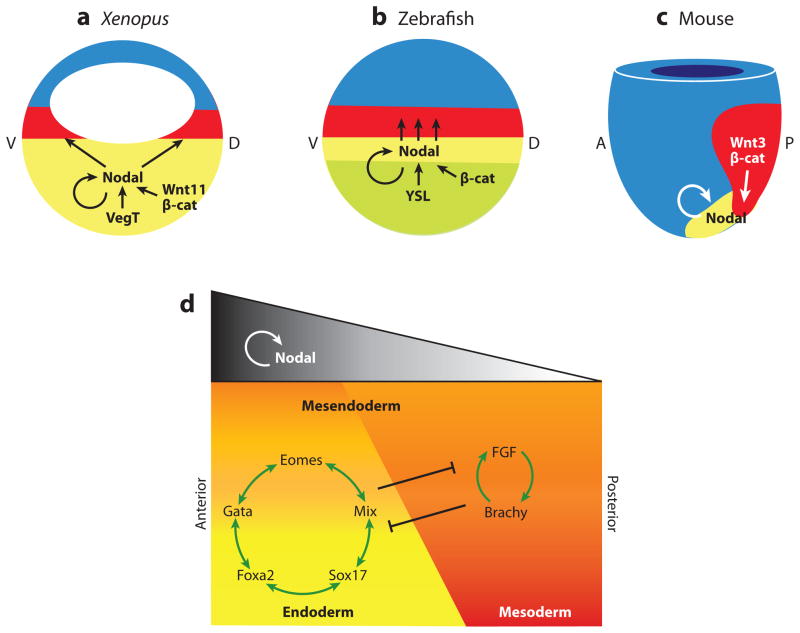
Figure 4.
Nodal signaling in mesendoderm induction. Fate maps of (a) Xenopus and (b) zebrafish blastula, and (c) an early mouse gastrula showing the endoderm (yellow), mesoderm (red), and ectoderm (blue), and the molecular pathways regulating Nodal gene expression and signaling in each species. (a) In Xenopus, Nodal transcription in the endoderm is activated by the maternal T-box transcription factor VegT, in cooperation with Wnt11/β-catenin on the future dorsal (d) side of the embryo. In zebrafish, an unknown signaling from the YSL induces Nodal transcription in the marginal mesendoderm in cooperation with maternal dorsal β-catenin. In both Xenopus and zebrafish, Nodal autoregulation maintains its own expression in the endoderm, and Nodal diffuses to induce mesoderm in the adjacent tissue. (c) In the mouse, high levels of Nodal expression in the node and primitive streak maintain Nodal autoregulation as well as Wnt3/β-catenin signaling. d, dorsal; v, ventral; a, anterior; p, posterior. (d) The schematic depicts a generalized model, incorporating data from frog, fish, and mouse; which shows endoderm and mesoderm induction by different levels of Nodal signaling; and how the downstream transcription factors might segregate these lineages from a common mesendoderm progenitor. High levels of Nodal are required to induce endoderm and anterior mesendoderm, whereas lower levels are sufficient for mesoderm and posterior tissue.
Nodal activity is positively controlled by an autoregulatory loop that maintains and amplifies its own transcription, via evolutionarily conserved Foxh1 DNA-binding sites in the first intron of Nodal, xnr1, and sqt genes (Fan et al. 2007, Norris et al. 2002, Osada et al. 2000). As a result, paracrine signaling between cells sustains the high nodal activity required to maintain endoderm gene expression. Negative feedback of Nodal activity occurs via its transcriptional target Lefty, which inhibits Nodal ligand-receptor interactions. Thus, in a classical reaction-diffusion model, Nodal autoregulation and Lefty negative feedback defines the mesendoderm territory and prevents its expansion into the ectoderm (Shen 2007).
There is considerable divergence in the way different species activate the nodal pathway (Figure 4). In Xenopus, the maternally inherited T-box transcription factor VegT is localized to the vegetal region of the oocyte and early blastula where it directly stimulates the transcription of many mesendoderm genes including xnr1 and xnr5 (Hyde & Old 2000, Xanthos et al. 2001). This role of VegT appears to be unique to frogs. In zebrafish, an unidentified maternal signal from the extraembryonic yolk syncytial layer (YSL) is thought to promote mesendoderm development, and the expression of zygotic cyc and sqt in the blastoderm margin (Fan et al. 2007, Schier & Talbot 2005). In the pregastrula mouse embryo, Nodal is expressed radially around the proximal epiblast; but then it becomes restricted to the presumptive posterior by the combined action of the antagonists Cerberus and Lefty in the anterior visceral endoderm (AVE), and the proteases that process Nodal ligands. Nodal signaling then initiates gastrulation in the posterior epiblast by regulating the Wnt, FGF, and BMP pathways (reviewed in Grapin-Botton & Constam 2007, Tam & Loebel 2007).
One similarity between frogs, fish, and mice is that canonical Wnt signaling via β-catenin and Tcf transcription factors stimulates high levels of Nodal gene transcription in the pregastrula embryo (Figure 4a). (see Wnt Signaling, sidebar below) In Xenopus and zebrafish, a maternal Wnt/β-catenin pathway that is active on the dorsal side of the embryo cooperates with VegT and the maternal YSL signal respectively, to promote high levels of nodal-related transcription in the dorsal-anterior mesendoderm (Fan et al. 2007, Hilton et al. 2003, Xanthos et al. 2002). Similarly in mouse, Wnt3 signaling appears to maintain Nodal expression in the streak, via evolutionary conserved Tcf DNA-binding sites in the Nodal promoter (Ben-Haim et al. 2006). Consistent with this, deletion of β-catenin in the early mouse gastrula impairs DE formation (Lickert et al. 2002).
Endoderm TranscriptionFactor Network
Nodal signaling promotes the expression of a conserved network of transcription factors within the endodermal lineage, which include Mix-like proteins: Foxa2, Sox17, Eomesodermin, and Gata4–6. Although the precise roles of these factors vary between species, together they activate a cascade of gene expression that functions to: (a) segregate the endoderm and mesoderm lineages, (b) commit cells to an endodermal fate, and (c) integrate signaling events that regionalize the nascent endoderm (Stainier 2002, Zorn & Wells 2007).
There are seven Mix-like homeobox genes in Xenopus (mix1, mix2, bix1, bix2, bix3, bix4, and mixer); four in zebrafish (bon, mezzo, mxt1, and mxt2); and only one in mice, Mixl1. Several Mix-like proteins physically interact with Smad2, and can mediate Nodal-dependent transcription (Germain et al. 2000). In mouse, Mixl1 is required for DE formation and for suppressing mesoderm (Hart et al. 2002, Tam et al. 2007). Of the three forkhead family members Foxa1–3 (Hnf3α, β, and γ), Foxa2 is particularly critical; and Foxa2−/− null embryos lack foregut endoderm (Dufort et al. 1998). The HMG box factor Sox17 is required for normal endoderm development in Xenopus and mice (Figure 5) (Hudson et al. 1997, Kanai-Azuma et al. 2002). In zebrafish, the Sox17-related gene casanova (cas/sox32) is essential for endoderm specification and cooperates with Pou51/Oct4 to activate sox17 and foxa2 transcription (Lunde et al. 2004, Reim et al. 2004, Stainier 2002).
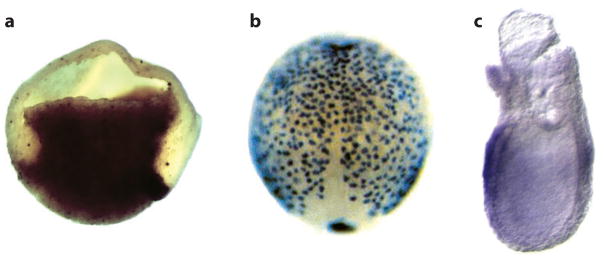
Figure 5.
In situ hybridization showing Sox17 expression in gastrula stage (a) Xenopus, (b) zebrafish, and (c) mouse embryos. (a) The Xenopus embryo is bisected with dorsal to the right. (b) The dorsal side of the zebrafish embryo is shown with anterior up, illustrating the dispersed layer of endoderm cells spread over the embryo. (c) A lateral view of the mouse embryo (posterior to the right) shows Sox17 expression in the endoderm layer surrounding the embryo. Reproduced with permission from Zorn and Wells (2007).
Whereas the functions of Mix, Sox17, and Foxa are highly conserved; other transcription factors appear to have species-specific functions. For example, in zebrafish and Xenopus; the zinc-finger factors Gata4, 5, and 6 are also involved in endoderm specification downstream of nodal signaling (Stainier 2002, Zorn & Wells 2007). In mice, Gata4–6 regulate extraembryonic endoderm lineages and do not appear to participate in DE specification, although they have essential, conserved roles in later endoderm organ development (Watt et al. 2007). The T-box transcription factor Eomesodermin (Eomes) is also essential for endoderm specification downstream of nodal signaling in zebrafish and mouse (Arnold et al. 2008, Bjornson et al. 2005), but its precise function in the frog endoderm remains to be determined.
A number of studies have begun to define the inter-relationships between the endoderm-specifying transcription factors. Epistatic analyses in zebrafish suggest a pathway in which Nodal activates the expression of Bon and Gata5; and they form a complex with Eomes to activate Cas, which in turn stimulates sox17 and foxa2 transcription (Alexander & Stainier 1999, Bjornson et al. 2005, Ober et al. 2003). Recent genome-wide expression analyses in various species indicate that these transcription factors probably do not act in a simple linear fashion, but rather form a complex gene regulatory network (GRN). The transcription factors influence each other expression through multiple feedback loops, and they regulate both distinct and overlapping transcriptional targets (Brown et al. 2008, Dickinson et al. 2006, Sinner et al. 2006, Tamplin et al. 2008) (Figure 4d). Several Web sites now provide interactive models of the increasingly complex GRN; integrating expression data, genomic information, and DNA-binding evidence along with functional data (see related resources section below) (Chan et al. 2008, Loose & Patient 2004, Oliveri & Davidson 2004). Interestingly, some components of the endoderm-GRN, such as Foxa and Gata factors, are evolutionarily ancient and known to regulate endoderm development in C. elegans, Drosophila, sea urchins, and ascidians (Zorn & Wells 2007).
A key function of the endoderm–GRN is to segregate the mesoderm and endoderm lineages. This is partly mediated by mutual antagonism with an analogous mesoderm-GRN, in which low doses of Nodal induce the expression of FGF ligands and the T-box transcription factor Brachyury. An autoregulatory loop between FGF and Brachyury constitutes the core of a mesoderm-GRN (Latinkic et al. 1997), which promotes mesoderm fate and represses endoderm gene expression (Mizoguchi et al. 2006, Poulain et al. 2006). This may act on several levels, because FGF stimulated MAP kinase can phosphorylate Smad2 and Sox32 in fish, which reduces their activity (Poulain et al. 2006, Zorn & Wells 2007). In addition, Mix-like proteins and Brachyury appear to repress each other’s expression (Hart et al. 2002, Kofron et al. 2004, Lemaire et al. 1998). Thus, although cells initially express both endoderm and mesoderm genes in response to Nodal signaling, these mutually repressive pathways resolve the lineages by mid-gastrula (Figure 4d).
ENDODERM PATTERNING
After gastrulation, naïve endoderm cells are plastic and not yet committed to specific organ fates. In the subsequent 48 hours, the endoderm germ layer forms a primitive gut tube from which organ buds emerge. During this time, the endoderm is patterned by a series of overlapping growth factor signals from the surrounding mesoderm that progressively subdivide the endoderm along the A-P axis; first into broad foregut and hindgut domains and then, ultimately, into committed organ primordia.
Initial Patterning Coincides with Endoderm Formation
The first regional differences can be detected in the early gastrula when the endoderm germ layer is being specified. In Xenopus and zebrafish, maternal Wnt/β-catenin signals that are active on the future dorsal side of the blastula (Figure 4a,b) cooperate with Nodal signaling to activate the expression of genes, such as the homeobox Hhex in anterior endoderm (Ho et al. 1999, Zorn et al. 1999). In mice, the high-nodal environment of the early primitive streak promotes anterior endoderm fate and expression of Hhex (Thomas et al. 1998). At later gastrula stages, cells in the posterior primitive streak are in a lower nodal environment and tend to contribute to posterior endoderm. The endoderm-GRN transcription factors also participate in endoderm patterning; Foxa2 and Mixl1 are preferentially required for anterior endoderm; and Sox17 is required for posterior endoderm (Dufort et al. 1998, Hart et al. 2002, Kanai-Azuma et al. 2002). Their different activities correlate with their timing of expression; with Foxa2 being is expressed first and Sox17 expressed slightly later, when the posterior DE emerges from the streak. Crosstalk with other signaling pathways may account for the different activities, because Sox17 physically interacts with β-catenin to promote the transcription of some target genes, whereas it represses others (Sinner et al. 2004).
A-P Patterning of the Primitive Gut Tube
By the end of gastrulation, the endoderm is partitioned into broad A-P domains that can be identified by the expression of Hhex, Sox2, and Foxa2 transcription factors in the anterior half of the embryo; and Caudal type homeobox genes Cdx1, 2, and 4 in the posterior half (Figure 6). These transcription factors are crucial for regional identity: Hhex, Foxa2, and Sox2 are required for foregut development, and the Cdx genes are required for the hindgut and positioning of the foregut-hindgut boundary (Chawengsaksophak et al. 2004, Dufort et al. 1998, Kinkel et al. 2008, Martinez Barbera et al. 2000.
Figure 6.
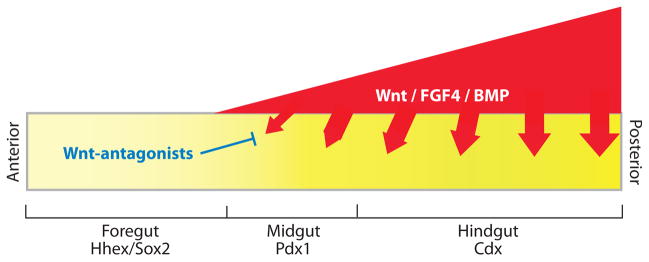
A model of early A-P patterning, incorporating recent data from frog, fish, mouse, and chick. During gastrulation and early somite stages of development, differential Wnt, FGF4, and BMP signaling along the A-P axis patterns the naïve endoderm into foregut, midgut, and hindgut progenitor domains; which express the transcription factors Hhex, Pdx1, and Cdx, respectively. Although this model remains to be validated genetically in the mouse, the available data suggests that Wnt, FGF, and BMP ligands; which are expressed in a graded fashion in the mesoderm (red); signal to the adjacent endoderm (yellow) to repress foregut identity and promote hindgut fate. Secreted Wnt-antagonists, such as Sfrp5 in the anterior endoderm, inhibit Wnt ligands to maintain foregut identity and proper foregut morphogenesis. Modified with permission from Zorn (2008).
Despite this differential gene expression, endoderm fate is still labile at this stage (Horb & Slack 2001, Kimura et al. 2007, Le Douarin 1968, Wells & Melton 2000). Dynamic tissue movements during gastrula and early somite stages bring the endoderm into proximity with different mesodermal tissues that secrete patterning factors (Chalmers & Slack 2000, Tam et al. 2007, Tremblay & Zaret 2005). These include FGF, Wnt, and BMP ligands; all of which appear to maintain hindgut identity and actively repress foregut fate in the posterior (Figure 6).
In mouse and chick, mesodermal FGF4 (but not FGF2, 5, or 8), signaling promotes Cdx expression in the presumptive hindgut endoderm and represses the expression of anterior genes Hhex and Foxa2 (Dessimoz et al. 2006, Haremaki et al. 2003, Wells & Melton 2000). In frog and zebrafish, Wnt/β-catenin signaling, similar to FGF4, is necessary and sufficient to promote hindgut development and inhibit foregut fate (Goessling et al. 2008, McLin et al. 2007). Moreover, in Xenopus, β-catenin activity must be repressed in the anterior endoderm by secreted Wnt-antagonists such as Sfrp5 to maintain the foregut progenitors, and for subsequent liver and pancreas development (Li et al. 2008). It is currently unclear if differential Wnt/β-catenin signaling also patterns the late gastrula amniote endoderm. Experiments in zebrafish, Xenopus, and chick indicate that, during these early stages of development, BMP signaling also promotes posterior endoderm development (Tiso et al. 2002, Wills et al. 2008) and that RA is important in establishing the foregut-hindgut boundary (Chen et al. 2004, Kumar et al. 2003, Stafford & Prince 2002).
Although foregut and hindgut progenitors are initially uncommitted, there is evidence that they have distinct developmental potentials and respond differently to subsequent inductive events. For example, in transplantation experiments, the foregut has a greater intrinsic hepatic potential than the hindgut (Fukuda-Taira 1981, Le Douarin 1968), which may be due to foregut enriched transcription factors such as Foxa2. In vivo DNA-binding studies suggest that Foxa2 binds to enhancer elements of the Albumin gene and opens chromatin, allowing it to be transcribed (Zaret 2002). Consistent with this, the conditional deletion of both Foxa1 and Foxa2 from the mouse foregut prevents liver induction (Lee et al. 2005). In another example, Cdx4-expressing hindgut endoderm is not competent to respond to subsequent pancreatic induction by RA signaling in zebrafish (Kinkel et al. 2008).
It is unclear how the effects of FGF, Wnt, BMP, and RA are integrated during endoderm patterning. However, there is evidence that these same signals coordinately regulate the transcription of Cdx and Hox genes in the mesoderm and neuro-ectoderm (Benahmed et al. 2008, Pilon et al. 2006, Shimizu et al. 2006); this suggests that these signaling factors synchronize A-P identity in all three germ layers. Indeed, a recent study found that RA and FGF4 cooperate to posteriorize the chick gastrula endoderm (Bayha et al. 2009).
MOLECULAR BASIS OF EARLY ENDODERM MORPHOGENESIS
During development, cell identity and tissue morphogenesis must be exquisitely orchestrated. Recent work has begun to reveal the molecular mechanisms that link endoderm formation and patterning with the cell-migration, cell-adhesion, and cytoskeletal dynamics that control endoderm morphogenesis.
Endoderm Migration in the Gastrula
In addition to their role in endoderm specification and patterning, Nodal, FGF, and Wnt signaling are all required for the coordinated series of cell movements that drive mesendoderm morphogenesis (reviewed in Keller 2005). During gastrulation, the dorsal mesendoderm elongates via polarized cell intercalations in a process known as convergent-extension, which is coordinately regulated by FGF and non-canonical Wnt/planar cell polarity (PCP) signaling (Keller 2005). The anterior endoderm cells, on the other hand, exhibit directional migration, which requires Nodal signaling and is mediated by dynamic cell-cell adhesion and integrin-fibronectin interactions with the extra cellular matrix (Davidson et al. 2002, Pezeron et al. 2008, Yamamoto et al. 2004).
In addition to specifying endodermal fate, Mix-like factors; Eomes, Lim1, Foxa2, and Gata4–6; also promote mesendoderm migration behavior (Arnold et al. 2008; Fletcher et al. 2006; Kofron et al. 2004; Luu et al. 2008; Tam et al. 2004, 2007); thus linking Nodal-induced endoderm identity with morphogenesis. Downstream targets of Nodal signaling and Mixer include the chemokine receptor Cxcr4 (Dickinson et al. 2006, Fukui et al. 2007, Sinner et al. 2006) and the ligand Sdf1, which acts as a chemoattractant for Cxcr4-expressing endoderm cells (Fukui et al. 2007, Mizoguchi et al. 2008, Nair & Schilling 2008). The disruption of Cxcr4/Sdf1 in zebrafish results in disrupted endoderm migration and gut-tube duplications (Mizoguchi et al. 2008, Nair & Schilling 2008). Cxcr4/Sdf1, as well as PDGF signaling, appear to act by regulating integrin fibronectin-mediated endoderm migration (Keller 2005). In Xenopus, the nodal target fibronectin-leucine rich transmembrane protein (FLRT3) also regulates cadherin-dependent cell adhesion and mesendoderm migration via the small GTPase Rand1 (Ogata et al. 2007). FLRT3−/− mouse embryos also have DE migration defects (Egea et al. 2008, Maretto et al. 2008).
In the mouse, gastrula FGF signaling, MAP kinase, and Eomes are required to downregulate E-cadherin and allow epiblast cells to undergo an EMT and ingress through the PS (Arnold et al. 2008, Ciruna & Rossant 2001, Zohn et al. 2006). However, less is known about what regulates the subsequent migration of DE cells and their incorporation into the visceral endoderm. Genetic analysis suggest that DE migration requires continued FGF signaling; the activity of Mixl1, Lim1, Foxa2; and the protein Nap1 (Burtscher & Lickert 2009, Garcia-Garcia & Anderson 2003, Rakeman & Anderson 2006, Sun et al. 1999, Tam et al. 2007). However, these mutants often have disruptions in the anterior visceral endoderm migration and the PS, which makes it difficult to study DE migration (Garcia-Garcia & Anderson 2003, Rakeman & Anderson 2006).
Early Gut-Tube Morphogenesis
Gut-tube morphogenesis occurs very differently across vertebrate species. In amniotes, it begins with the invagination, and rostral-to-caudal movement, of the foregut and hindgut pockets (Figure 3) (Franklin et al. 2008, Tremblay & Zaret 2005). Recent forward genetic screens in mice have revealed that this process requires a novel zinc finger protein Chato (Garcia-Garcia et al. 2008) and Lulu, a FERM domain cytoskeletal protein thought to regulate epithelial organization (Lee et al. 2007).
In zebrafish, vascular endothelial growth factor (VEGF) and Wnt/PCP pathways are required for convergent-extension of the dispersed endodermal sheet into a solid endodermal rod at the dorsal midline. Disruptions in this process result in foregut duplications with bilateral liver and pancreas (Matsui et al. 2005, Ober et al. 2004). Formation of a gut tube lumen within the solid endodermal rod is regulated by the transcription factor Hnf1β/Tcf2, through ion channel mediated fluid accumulation (Bagnat et al. 2007). Initial looping of the zebrafish gut appears to be controlled by asymmetric migration of the lateral plate mesoderm on one side of the embryo in response to global left-right asymmetry cues in the embryo (Horne-Badovinac et al. 2003).
The Wnt/PCP pathway also regulated gut elongation in Xenopus (Figure 3a) (Li et al. 2008). Moreover, the secreted Wnt-antagonist Sfrp5 appears to simultaneously restrict both Wnt11/PCP and Wnt11/β-catenin signaling in the anterior endoderm to coordinate foregut fate and morphogenesis (Li et al. 2008). Repression of Wnt11/PCP signaling is necessary to allow the foregut epithelium to form over the deep endoderm cells that are undergoing the convergent-extension movements that drive gut elongation. It is unclear if this mechanism is conserved in mammals, because Sfrp5−/− null mice do not exhibit foregut defects, although redundantly expressed Wnt-antagonists may be involved.
FOREGUT ORGAN DEVELOPMENT
During early somite stages of development, the broad foregut and hindgut territories become further subdivided into organ specific lineages by continued mesenchymal interactions. In mice this begins at ~e8.0 (2–4 somite stage), and it is largely complete by e9.5 when the anterior and posterior intestinal portals join at the midgut and organ primordia begin to bud from the gut tube.
An overview of endoderm organogenesis reveals some noteworthy features. First, it is clear that the same signaling pathways are used reiteratively throughout development and that the same cells can respond differently depending on the stage. For example, in cells that will give rise to the hepatic lineage, Wnt and FGF signaling are first required to initiate gastrulation and generate the early anterior endoderm. In late gastrula, Wnt and FGF4 repress anterior fate and must be excluded to maintain the foregut progenitors. Then, as we describe below, FGF and Wnt activity promote hepatic induction and liver bud growth. Amazingly, these changes in response are very rapid, often occurring within hours. Similarly, some of the transcription factors that control early regional identity such as Hhex, Cdx, and Foxa2 also have multiple roles in later organ formation.
On the other hand, some factors have similar roles in different organs. For example, mesenchymal FGF10 promotes epithelial proliferation and migration in several organs. Signaling from developing endothelial cells also promotes the growth of both liver and pancreatic buds, coordinating their development with the nascent vasculature. Finally, Notch signaling plays an analogous role in many organs to regulate the selection of specific cell subtypes, such as the segregation of hepatoblasts into hepatocytes or biliary epithelium, and the selection of the endocrine pancreas and intestinal epithelial lineages.
Subdividing the Foregut: Organ Induction
The foregut epithelium gives rise to the esophagus, trachea, lungs, thyroid, stomach, liver, pancreas, and hepatobiliary system (e.g., intrahepatic and extrahepatic bile ducts, common duct, gall bladder, cystic duct). Human congenital malformations often occur simultaneously in several of these organs, consistent with experimental evidence that they are derived from a common progenitor cell population in the ventral foregut (Zaret 2008).
Foregut explants and mouse genetics suggest a model in which varying concentrations of FGF2 act through MAP Kinase to induce different lineages from a common region of the ventral foregut (Calmont et al. 2006, Jung et al. 1999, Zaret 2008). High levels of FGF signaling induce expression of the homeobox gene Nkx2.1 in the lung and thyroid progenitors; moderate doses activate Albumin expression in liver progenitors; and low levels promote expression of the homeobox gene Pdx1 in the ventral pancreas and duodenum (Figure 7a) (Serls et al. 2005). It is unclear whether the proximity or duration of contact with the cardiac mesoderm controls the FGF dose in vivo. However, this may be regulated in part by Hhex-mediated cell proliferation, which displaces the presumptive ventral pancreatic epithelium away from the cardiac mesoderm (Bort et al. 2004).
Figure 7.
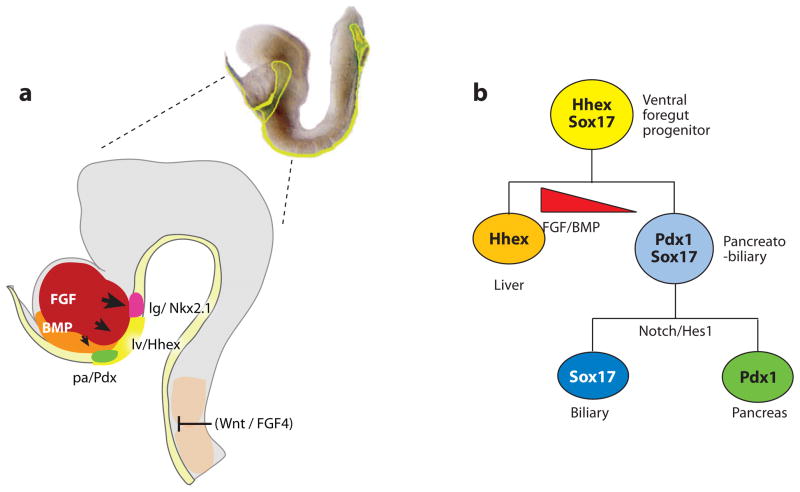
Foregut organ induction. (a) A lateral view of the anterior portion of an e8.25 (2–4 somite stage) mouse embryo (right). The developing head is shown in (gray). At this stage, the definitive endoderm (yellow) is forming a foregut pocket. The liver (lv) is marked by Hhex expression and is induced a portion of the ventral foregut by FGF signals from the cardiogenic mesoderm (red), and BMPs from the septum transversum mesenchyme (orange). The presumptive lung (lg) (red) and pancreas (pa) (green) are marked by Nkx2.1 and Pdx1 respectively. Signals from the axial mesoderm (candidates include FGF4 and Wnt ligands) repress hepatic fate in the dorsal endoderm. (b) Lineage model for the liver, pancreas, and biliary system. In this model, the ventral foregut progenitor expresses Hhex and Sox17. The high FGF/BMP environment adjacent to the cardiac mesoderm favors a liver fate (Hhex). Foregut cells caudal to the Hhex domain co-express Pdx1 and Sox17, and contribute to the ventral pancreas and biliary system. Segregation of biliary and pancreatic lineages depends on the cooperative activity of Sox17 and the Notch signaling effector Hes1 (Spence et al. 2009).
BMP signaling from the septum transversum mesenchyme also promotes hepatic fate, and at high levels, it appears to repress pancreas development (Figure 7) (Zaret 2008). BMP and FGF signaling similarly regulate liver and pancreas induction in zebrafish, chicken, and Xenopus (Shin et al. 2007, Zaret 2008). Although in zebrafish these factors (and hepatic promoting Wnt2b) originate from the lateral plate mesoderm and not cardiogenic and septum transversum mesenchyme (Ober et al. 2003, Zaret 2008). Recent single cell labeling experiments in zebrafish provided direct evidence that the liver, pancreas, and intestine indeed arise from a common pool of progenitors; and that proximity to the source of BMP2 signaling influences cell fate (Chung et al. 2008).
RA has multiple roles regulating cell identity in the pharyngeal arches, the anterior and posterior ends of the foregut, as well as a posteriorizing role in the hindgut (Duester 2008). Mouse embryos deficient for retinaldehyde dehydrogenase 2 (Raldh2) lack RA signaling in the foregut and fail to develop lungs, stomach, and dorsal pancreas; and have impaired liver growth (Molotkov et al. 2005, Wang et al. 2006). Experiments in chick, Xenopus, and zebrafish suggest that RA regulates the Pdx1-Cdx expression boundary and the position of the dorsal pancreas (Bayha et al. 2009, Chen et al. 2004, Kinkel et al. 2008, Stafford & Prince 2002, Kumar et al. 2003). RA signaling appears to act in part by directly activating the transcription of homeobox genes such as Hoxb1 and Hoxa5, which contain retinoic acid responsive enhancers (Grapin-Botton 2005). However, other effects of RA may be indirect owing to the activation of downstream factors including FGF10, Shh, and Tbx1; which regulate Raldh2 expression in a feedback loop (Ivins et al. 2005, Wang et al. 2006).
Dorso-ventral patterning of the gut tube is poorly understood, but critical for separating the dorsal esophagus from the ventral trachea, and it impacts the distinct ventral and dorsal pancreatic primordia. D-V patterning in the gut appears to be similar to neural tube patterning, in which mutual antagonism between Shh and BMP establishes D-V identity (Petryk et al. 2004, Que et al. 2006, Que et al. 2007, van den Brink 2007). Mouse knockouts of Shh, or its transcriptional effectors Gli2 and Gli3, cause a range of defects from lung agenesis to tracheo-esophageal fistulas, similar to a human congenital condition in which the trachea and esophagus fail to separate. Mutant mice that lack the BMP antagonists Noggin or Twisted-Gastrulation exhibit similar defects, with reduced Shh and Hhex expression in the ventral foregut. The separation of the trachea and esophagus also appears to involve the transcription factors Foxf1 in the mesenchyme (Mahlapuu et al. 2001) and mutual repression between dorsal Sox2 and ventral Nkx2.1 (Que et al. 2006, Que et al. 2007).
Translating Patterns into Organ Domains
Foregut organs are specified around the 6–7-somite stage of development in mouse and chick, but not until 20+ somite stages in Xenopus and zebrafish. Although we have begun to understand the cell signals that induce different foregut organ fates, it is unclear how the resulting gene expression patterns are actually translated into distinct organ primordia. Increasingly sophisticated maps of transcription factor expression domains suggest that there is not a one-to-one correspondence between any individual factor and a given cell lineage, but rather a complex combinatorial code of overlapping factors along the GI tract (Figure 8). A recent study of 15 transcription factors in the mouse foregut (Sherwood et al. 2009) identified over a dozen domains; with unique combination of factors that roughly correspond to the presumptive liver, biliary system, stomach, duodenum, and pancreas.
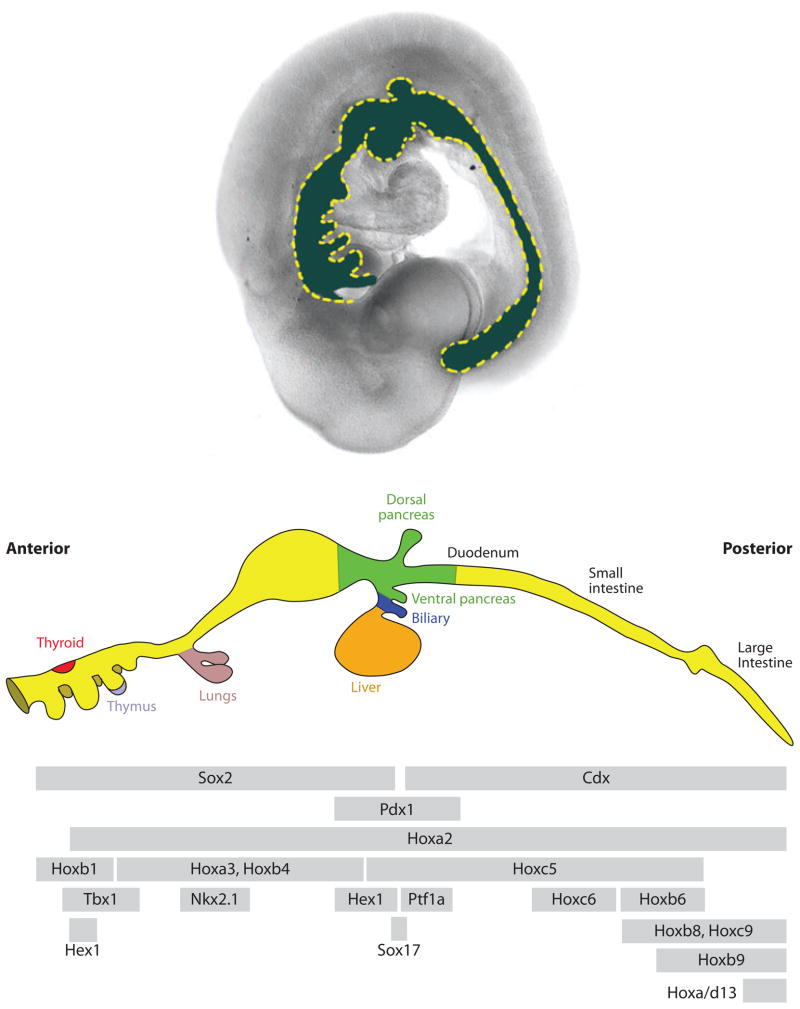
Figure 8.
Overlapping expression domains of transcription factors along the A-P axis of the gut tube. The (upper panel) shows a lateral view of an e9.5 mouse embryo with the gut tube false-colored (blue). The middle panel shows a schematic diagram of the bud-stage gut (~E10.5 in mouse). The (lower panel) indicates the relative A-P expression boundaries of several transcription factors. The expression of these factors is temporally dynamic, and these expression domains are not necessarily maintained throughout development. For example, Hhex is first expressed in one domain in the ventral foregut, then it is expressed in the presumptive thyroid and liver. The anterior and posterior expression limits on some of these factors are important in establishing organ domains. The lower panel was adapted from Grapin-Botton (2005).
Recent studies have begun to determine how combinations of transcription factors specify distinct lineages. For example, a domain in the ventral foregut that coexpresses Pdx1 and Sox17 was shown to give rise to the pancreas, gall-bladder, cystic duct, and common duct. Segregation of these extrahepatobiliary/pancreatic progenitors was Sox17-dependent. The deletion of Sox17 resulted in the loss of biliary structures and ectopic pancreas in the common duct, whereas maintained Sox17 expression suppressed pancreas development and induced ectopic biliary tissue (Spence et al. 2009). These and other studies support a model in which the common foregut progenitors first separate into Hhex+ liver cells and a multipotential Pdx1+/Sox17+ progenitor population (Figure 7b). Sox17 then acts to segregate the biliary from the pancreas lineage, possibly in concert with Hnf6 and Hnf1β, and the Notch effector Hes1 (Clotman et al. 2002, Coffinier et al. 2002, Hunter et al. 2007). Genetic analysis suggests that Foxf1 and FGF10 in the surrounding mesenchyme also regulate proliferation and correct positioning of the liver, pancreas, and various hepatobiliary ducts (Dong et al. 2007, Kalinichenko et al. 2002).
Below, we briefly summarize early liver, pancreas, and lung bud development (~e9–11 in the mouse), and for a review of pharyngeal organogenesis, including the thyroid and thymus, we refer readers to Graham (2008).
Liver
Once specified, the hepatic endoderm thickens into a columnar epithelium, the surrounding basement membrane breaks down, and the hepatic precursors (hepatoblasts) delaminate and migrate into the adjacent stroma and form the liver bud. This process requires the transcription factors Hhex, Gata4/6, Hnf6/Onecut1, Onecut2, and Prox1; and appears to be mediated by matrix remodeling metalloproteases. Continued signaling, which include FGF, BMP, Wnt, and HGF, from hepatic mesenchyme and endothelial cells, as well as hepatic transcription factors such as Hnf4α, Tbx3, and Hlx, promote liver bud growth and differentiation. The hepatoblasts are bi-potential, and those residing next to the portal vein mesenchyme are induced by a Notch-mediated process to form biliary epithelial cells, whereas those deeper in the parenchyma become hepatocytes. For a detailed review of liver differentiation, see Zaret (2008), Zhao & Duncan (2005), and Zorn (2008).
Pancreas
The pancreas arises from distinct dorsal and ventral buds, which are induced by different mesodermal structures. As described above, the ventral pancreas fate is induced in the portion of the ventral foregut that has low levels of cardiac FGF signaling. Dorsal pancreatic development requires RA signaling (Molotkov et al. 2005, Stafford & Prince 2002); as well as secreted factors from the notochord and dorsal aorta, including Activin and Fgf2, that repress Shh expression in the presumptive dorsal pancreatic epithelium (Figure 9). In both the dorsal and ventral buds, pancreatic progenitors express the essential transcription factor Pdx1 and Ptf1a/p48, but only the dorsal bud expresses Hoxb9 (Sherwood et al. 2009). Mesenchymal FGF10 then promotes the proliferation of pancreatic progenitors (Bhushan et al. 2001). Wnt signaling is required for the expansion of the exocrine pancreas (Murtaugh et al. 2005, Wells et al. 2007), whereas Notch-Delta signaling via the Ngn3 transcription factor is required for segregating the endocrine lineages. We refer readers to Gittes (2009) and Murtaugh (2007) for a detailed review on pancreas development and the differentiation of the five endocrine lineages (α-cells, β-cells, δ-cells, PP cells, and grelin cells).
Figure 9.
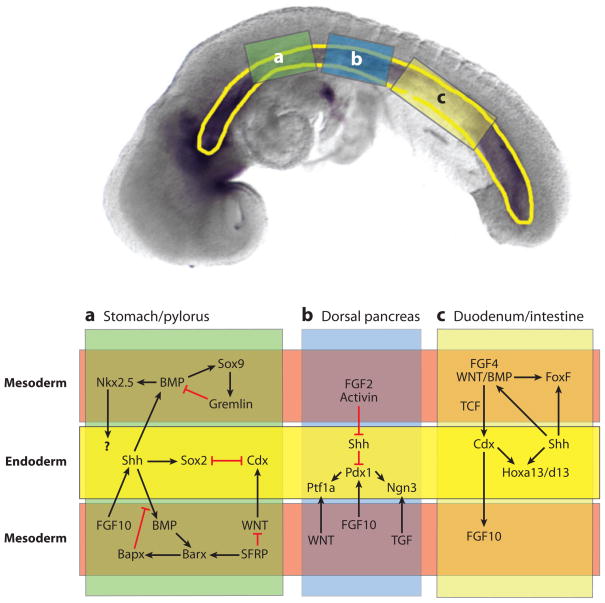
Reciprocal epithelial-mesenchymal signaling in the gut tube during development of the stomach (a), pancreas (b), and duodenum/small intestine (c). The (upper panel) shows a 10-somite stage mouse embryo with the gut tube outlined in (yellow). The three boxes indicate the regions of the gut that give rise to the stomach (a), pancreas (b), and duodenum (c). The (lower panel) schematically shows these regions with the endoderm-epitheium (yellow) flanked by adjacent mesoderm-mesenchyme (above and below) (light red). The mesoderm was divided to allow for a summary of the multiple signaling cascades that have been identified in different species and at different stages of development. This schematic broadly summarizes our current understanding. In some cases the molecular relationships depicted have been directly demonstrated, whereas in other cases, we have inferred a connection from studies in other species.
Lung
In response to high doses of cardiac FGF signaling, presumptive lung cells in ventral foregut upregulate the homeobox gene Nkx2.1. RA and Hedgehog signaling then represses TGFβ signaling and promotes the expression of Hoxa4/5 and Tbx4/5, as well as FGF10, in the mesenchyme (Chen et al. 2007). FGF10 in turn induces the proliferation and chemotaxis of the Fgfr2-expressing pulmonary epithelium, which is required for lung bud formation (Cardoso & Lu 2006). By e10.5 in the mouse, the growing lung bud undergoes a highly stereotyped branching morphogenesis to generate the tree-like structure of the lung. This is regulated by a series of negative feedback interactions between FGF10 and Tbx4/5 in the distal tip mesenchyme; with Shh, BMP4, Fgfr2, and the FGF-antagonist Sprouty in the growing distal tip epithelium (Cardoso & Lu 2006). The adult respiratory epithelium contains a diversity of cell types including ciliated, goblet, basal, and Clara cells. For a detailed review on the lung development and the specification of these mature cell types, see Cardoso & Lu (2006) and Maeda et al. (2007).
THE GASTROINTESTINAL TRACT
As the foregut organs are specified, reciprocal epithelial-mesenchymal interactions also establish overlapping gene expression domains along the GI tract (Figure 8), which are eventually translated into the functional segments of the adult GI tract. These include the esophagus, the stomach, and duodenum; the midgut giving rise to the jejunum, ileum, and the small intestine; and the hindgut becomes the large intestine, cloaca, and portions of the urogenital tract. The adult GI tract is a three-layered tube, with an endoderm epithelial mucosa lining the lumen; surrounded by connective mesenchyme and smooth muscle, which is innervated by neural crest-derived enteric neurons that control peristalsis (Rubin 2007, Sancho et al. 2004).
Establishing Domains in the Intestinal Tract
The available data support a model in which posteriorizing factors such as Wnt, FGF4, and Cdx progressively allocate more posterior regions of the gut; initially they are broadly active in the posterior and suppressed in the presumptive foregut but then become down-regulated in the midgut by early somite stages. In the hindgut, persistent FGF/Wnt signaling is required to maintain Cdx expression and define the anterior boundary of the intestine. Experimentally increasing FGF4 signaling in the gut shifts the Pdx-Cdx boundary and transforms the duodenum into intestine (Benahmed et al. 2008, Dessimoz et al. 2006), whereas deletion of FGF-Receptor 2b in mice results in duodenal atreasia (Rubin 2007). Gene targeting of various Wnt-pathway molecules, including Tcf1/Tcf4−/− or the noncanonical Wnt5a ligand in mice; results in a range of phenotypes including truncated intestinal development, downregulation of intestinal Cdx2, and a posterior expansion of the foregut marker Sox2 (Cervantes et al. 2009, Gregorieff et al. 2004). Conversely, activating Wnt or Cdx2 more anteriorly can cause intestinal metaplasia of the stomach, pancreas, or lung similar to several human conditions (Grapin-Botton 2005). Recent findings indicate that Cdx2 function in the endoderm is required for initiating the intestinal program and supressing anterior cell fate (Gao et al. 2009), suggesting that regulation of Cdx2 expression is a point of convergence by which signaling pathways regulate intestinal development.
Reciprocal signaling between Indian and Sonic hedgehog in the epithelium and the surrounding mesenchyme also regulates many aspects of GI tract development (Figure 9) (Kumar et al. 2003, van den Brink 2007). For example, hedgehog signaling promotes the expression of Foxf1 transcription factors in the intestinal mesenchyme, which in turn regulates Wnt signaling and BMP expression (Ormestad et al. 2006). In the stomach, mesenchymal FGF10 regulates epithelial Hedgehog expression, which in turn influences BMP and Nkx2.5 expression in the pyloric mesenchyme (De Santa Barbara et al. 2005, van den Brink 2007). Shh in the stomach/duodenal domain is also regulated by Activin, and mouse embryos with Activin receptor mutations have a posterior shift in Shh expression (Kim et al. 2000). Finally, epithelial Shh signaling regulates the cocentric architecture of the gut by restricting smooth muscle differentiation in the immediately adjacent mesoderm, thus allowing connective tissue to develop (van den Brink 2007).
One mechanism by which FGF, Wnt, RA; and the Parahox factors Pdx1 and Cdx; control GI segment identity is by directly regulating Hox gene expression in precise domains along the A-P axis (Figure 8) (Grapin-Botton 2005, Kawazoe et al. 2002, Shimizu et al. 2006). Hox gene mutations indicate that they are required for GI tract development, and anterior misexpression of posterior Hoxa13 resulted in a homeotic transformed of stomach into intestinal epithelium (Grapin-Botton 2005). The distribution of Hox genes regulates the regional expression of downstream factors such as Fgf10, which impacts regional proliferation in the intestine (Zacchetti et al. 2007).
Stomach and Duodenum
In the anterior part of the GI tract, Shh and Sox2 are required for proper esophagus development and to establish the boundary with the stomach (Que et al. 2007). Specification of the stomach requires the homeodomain transcription factor Barx1, which in mice is localized to the presumptive stomach mesenchyme (e15), in which it activates the expression of secreted Wnt-antagonists Sfrp1 and Sfrp2. These in turn locally inhibit Wnt signaling to repress Cdx in the epithelium, and enable stomach differentiation (Kim et al. 2005). Thus, mutual antagonism between an intestinal promoting Wnt/Cdx2 pathway and a Barx1/Sfrp/Sox2 pathway determines the stomach-intestine boundary (Figure 9) (Stringer et al. 2008). Barx1 also regulates mesenchymal expression of the homeodomain factor Bapx1 (Nkx3.2), which is required for (Verzi et al. 2009) pyloric sphincter formation. Bapx1 appears to function in a complex regulatory loop with FGF10, Shh, Nkx2.5, and Sox9 to locally repress mesenchymal BMP expression (Figure 9) (De Santa Barbara et al. 2005, Moniot et al. 2004). For details on the further differentiation of stomach epithelium into mature squamous, parietal, chief, and gastric endocrine cells see (Yasugi and Mizuno 2008).
Intestine
The transformation of the simple pseudostratified epithelium into mature intestinal mucosa with a cryptvillus axis begins at approximately e14 in the mouse, and proceeds in an A-P wave along the GI tract. Cyto-differentiation and villi formation require Wnt, BMP, and Cdx2 (Rubin 2007), resulting in the four main cell types: absorptive enterocytes (colonocytes in the colon), the three secretory lineages the goblet cells, enteroendocrine cells, and Paneth cells. Notch and its effector Hes1 control segregation of the absorptive and secretory lineages by repressing expression of the bHLH transcription factor Atoh1 (Math1), thus specifying absorptive lineages over secretory lineages. Finally, Ngn3, as in the pancreas, is required for differentiation of the endocrine lineage (Sancho et al. 2004, Shroyer et al. 2007).
PRODUCING ENDODERM FROM EMBRYONIC STEM CELLS
Diseases that involve endodermally derived organs include cystic fibrosis, chronic hepatitis, and diabetes; and they affect tens of millions of people in North America. Existing transplantation-based therapies are currently limited by the availability of donor-derived tissues. Human pluripotent/embryonic stem cells (HESCs) are a promising, renewable source of material for transplantation, because they can be expanded indefinitely in culture and can differentiate into all cell types of the body. Researchers are now taking advantage of our understanding of endoderm organogenesis to successfully direct the differentiation of ESCs into pancreas, liver, lung, and thyroid cells. Below, we summarize the efforts to derive liver and pancreatic cells from HESCs (Figure 10), and how these cells have been used alleviate symptoms in animal models of diabetes and liver disease.
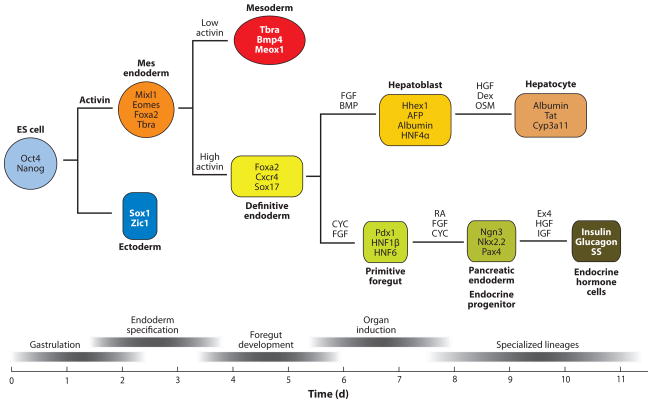
Figure 10.
Generating hepatocytes and β-cells from embryonic stem cells. The top diagram schematically shows the lineage relationship between ES cells, endoderm, liver, and pancreas. The lower panel shows the relative developmental stage and time in culture (days – d).
Culturing ES cells (ESCs) in activin mimics the activity of Nodal and induces specification of a mesendodermal progenitor. Persistent culture in high activin promotes definitive endoderm (DE) formation. DE cultures exposed to hepatic inducing factors (FGF and/or BMP) form hepatoblasts, whereas pancreatic inducing factors (CYC, cyclopamine; FGF10; and RA, Retinoic acid) promote posterior foregut and pancreatic fate. Further manipulations of the culture conditions promote formation of specialized organ lineages, such as hepatocytes and pancreatic endocrine cells. Molecular markers that define each cell type are shown within the colored box.
Abbreviations: HGF, hepatocyte growth factor; Dex, dexamethazone; OSM, oncostatin M; AFP, alphafetoprotein; Tat, tyrosine amino transferase; Cyp3a11, cytochrome superfamily p450 member 3a11.
A major advance in the field came with the observation that the TGFβ ligand Activin could mimic endogenous Nodal signaling, and promote a definitive endoderm fate in mouse and human embryonic stem cells (D’Amour et al. 2005, Kubo et al. 2004, Tada et al. 2005, Yasunaga et al. 2005). Remarkably, the effects of Activin on ES-cell differentiation were nearly identical to its effect on Xenopus blastula ectoderm; low levels of Activin promote a mesoderm fate (1–10 ng/ml), and high levels of Activin induced the formation of endoderm cells (10–100 ng/ml) (Green & Smith 1990, Hudson et al. 1997). Moreover, the timing of differentiation, and the molecular responses of ES cells to Activin, mimics mouse gastrulation: Day 1, initiation of gastrulation – Fgf8, Wnt3, and Eomes; Day 2 mesendoderm formation,– Mixl1 and Brachyury; Day 3–4, definitive endoderm formation – Sox17, Foxa2, and Cxcr4. These studies demonstrate that a thorough understanding of endoderm development has been essential for the directed differentiation of ES cells into definitive endoderm. Moreover, these ES cell studies have provided the best direct evidence for a transient mesendoderm progenitor (Tada et al. 2005), illustrating the utility of this system as a model for studying development. For example, recent studies have investigated the role of microRNAs in DE specification using human ES cells (Tzur et al. 2008).
Making Pancreatic β-cells and Hepatocytes for Therapy
Human ESC-derived endoderm has been further differentiated into the pancreatic endocrine lineage (Figure 10) (D’Amour et al. 2005, 2006; Kroon et al. 2008; reviewed in Spence and Wells 2007). Using a stepwise differentiation protocol designed to mimic pancreatic development, hESCs are first differentiated into DE with Activin; then into posterior foregut and pancreatic endoderm (PE) with a combination of retinoic acid, FGF10, and cyclopamine (a hedgehog inhibitor). PE cultures express foregut and pancreatic markers such as Hlxb9 and Pdx1. These PE cells were then cultured in the presence of Notch pathway inhibitors to promote endocrine fate and insulin, such as growth factor (IGF1) and hepatocyte growth factor (HGF), to promote maturation. The resulting cells had high levels of insulin, but were not responsive to glucose, suggesting that they were not fully mature β-cells. However, following engraftment into adult animals, PE/endocrine progenitor cells acquired features of mature pancreatic endocrine cells and restored normoglycemia in diabetic mice (Kroon et al. 2008).
In attempts to generate hepatocytes, researchers have treated Activin-induced HESC-derived endoderm with FGF2 and/or BMP4 to mimic hepatic induction. This has been followed by different combinations of HGF, EGF, FGF2, TGFα, VEGF, and Dexamothasone to mature the hepatic precursors and generate cells with hepatocyte-like properties (Gouon-Evans et al. 2006, Soto-Gutierrez et al. 2006, Zaret & Grompe 2008). Therapeutically, hepatocytes generated from both mouse and human ES cells have been shown to engraft in the liver (to a modest extent) and have restored some liver function in an animal model of liver failure (Zaret & Grompe 2008).
Although these reports are promising, in most cases the cells generated do not appear to be entirely mature and exhibit some fetal characteristics, suggesting that there are still gaps in our understanding. There is every reason to believe that further studies of liver and pancreatic development will continue to increase both the efficiency and functionality of HESC-derived cell types. It is hopeful that similar differentiation protocols can be used to direct induced pluripotent stem cells (iPSCs) into patient-specific therapeutic cell types.
RELATED RESOURCES
Jackson Labs mouse gene expression database: www.informatics.jax.org/
Xenbase, Xenopus gene expression database: www.xenbase.org
ZFIN, zebrafish gene expression database: www.zfin.org
Stembook, endoderm development: www.stembook.org/node/395
Xenopus mesendoderm GRN: www.nottingham.ac.uk/genetics/networks
Zebrafish mesendoderm GRN: www.zebrafishGRNs.org
Zorn lab: www.cincinnatichildrens.org/research/div/dev-biology/fac-labs/zorn-lab
Wells lab: www.cincinnatichildrens.org/research/div/dev-biology/fac-labs/wells-lab
WNT SIGNALING
Secreted Wnt ligands control many aspects of endoderm development. Wnts can signal via several distinct intracellular pathways (Clevers 2006, Semenov et al. 2007). In the canonical Wnt/β-catenin pathway, ligands interact with a Frizzled (Fz) and LRP5/6 receptor complex, causing the stabilization and nuclear accumulation of the key intracellular effector β-catenin, which interacts with Tcf/Lef transcription factors to activate target gene transcription. Non-canonical Wnt signaling refers to at least three intracellular pathways that are independent of β-catenin. These include the Wnt/PCP pathway, which is closely related to the Drosophila planar cell polarity pathway that signals via Fz to activate Rho and Rac GTPases. The Wnt/Ca2+ pathway signals via Fz, G-proteins, to mobilize intracellular calcium and activate Cam Kinase. Finally, a Wnt/Ror2 pathway signals via Ror/Ryk receptor tyrosine kinases to activate PI3 kinase, Cdc42, and JNK. Although the details of these noncanonical pathways are unresolved, they all regulate cytoskeleton dynamics and morphogenetic cell behaviors, including epithelial cell polarity, cell adhesion, and cell migration.
SUMMARY POINTS
The endoderm germ layer is specified during gastrulation by an evolutionarily conserved pathway that consists of Nodal signaling and a core group of downstream transcription factors.
There is emerging evidence that the endoderm and mesoderm are derived from a transient progenitor cell population known as the mesendoderm.
The primitive gut tube is progressively patterned, from broad foregut, midgut, and hindgut domains into organ primordia by a series of growth factor-mediated signaling between the endoderm epithelium and the surrounding mesenchyme.
Several signaling pathways, including those of FGF, hedgehog, BMP, RA, and Wnt; are used reiteratively during endoderm organ development.
Cells can exhibit dramatically different responses to the same growth factors at different times during the developmental continuum from endoderm formation, patterning, organ induction, and differentiation.
By the early somite stage, the developing gut tube is highly patterned and expresses genes in overlapping and distinct domains that predict where organ buds will form.
Disruption in embryonic patterning can lead to defects in the development of endoderm organs including the thymus, parathyroid, lungs, liver, pancreas, and intestine.
Information from endoderm organ development is being used to differentiate human embryonic stem cells into endoderm organ cell types, ultimately to treat degenerative diseases such as diabetes.
FUTURE ISSUES
Although recent results with directed differentiation of stem cells are promising, in most cases the resulting cells are not fully functional, suggesting that only part of the differentiation program has been activated. In vitro differentiation strategies would benefit from a better understanding of early endoderm patterning and factors that regulate maturation of fetal β-cells and hepatocytes.
Determine the cell-specific response to growth factor signaling in each stage of development, and determine how specificity arises and how the input from multiple pathways are integrated.
Determine, at the cell biological level, how lineage identity and tissue morphogenesis are coordinated during early organ formation at the cell biological level.
Identify the additional factors that regulate endoderm organ development and determine how these fit into the existing molecular pathways.
Use ES cells as a model to study mechanistic aspects of early human development that were previously inaccessible.
Acknowledgments
We apologize to colleagues whose work we could not properly cite owing to space constraints. We are grateful to the CHRF Endoderm Group for discussions, and to Scott Rankin and Jason Spence for comments on the manuscript. Endoderm research in the Zorn and Wells labs is supported by grants from the NIH.
- Definitive endoderm (DE)
the endoderm in an amniote embryo that gives rise to tissue in the embryo proper
- Gastrulation
the process of cell movement in the early embryo that generates the three primary germ layers: ectoderm, mesoderm and endoderm
- Mesendoderm
a transient cell population in the gastrula that gives rise to the both endoderm and mesoderm germ layers
- Foregut
anteriormost of three divisions of digestive tract (foregut, midgut, and hindgut)
- RA
retinoic acid
- Amniote
higher vertebrates, including reptiles, birds, and mammals that are capable of terrestrial reproduction and have an amnion, a developmental structure
- Epiblast
the embryonic cells in the pregastrula embryo
- PS
primitive streak
- Extraembryonic endoderm
an endoderm-like tissue in amniote embryos that d contributes to supporting structures, but does not contribute to the embryo proper
- VE
visceral endoderm
- Yolk syncytial layer (YSL)
an extra-embryonic tissue layer that lies between the epiblast and the yolk cell in the early zebrafish embryo
- GRN
gene regulatory network
- PCP
planar cell polarity
Footnotes
DISCLOSURE STATEMENT
The authors are not aware of any biases that might be perceived as affecting the objectivity of this review.
Contributor Information
Aaron M. Zorn, Email: aaron.zorn@cchmc.org.
James M. Wells, Email: james.wells@cchmc.org.
LITERATURE CITED
- Alexander J, Stainier DY. A molecular pathway leading to endoderm formation in zebrafish. Curr Biol. 1999;9:1147–57. doi: 10.1016/S0960-9822(00)80016-0. [DOI] [PubMed] [Google Scholar]
- Aoki TO, David NB, Minchiotti G, Saint-Etienne L, Dickmeis T, et al. Molecular integration of casanova in the Nodal signaling pathway controlling endoderm formation. Development. 2002;129:275–86. doi: 10.1242/dev.129.2.275. [DOI] [PubMed] [Google Scholar]
- Arnold SJ, Hofmann UK, Bikoff EK, Robertson EJ. Pivotal roles for eomesodermin during axis formation, epithelium-to-mesenchyme transition and endoderm specification in the mouse. Development. 2008;135:501–11. doi: 10.1242/dev.014357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bagnat M, Cheung ID, Mostov KE, Stainier DY. Genetic control of single lumen formation in the zebrafish gut. Nat Cell Biol. 2007;9:954–60. doi: 10.1038/ncb1621. [DOI] [PubMed] [Google Scholar]
- Bayha E, Jorgensen MC, Serup P, Grapin-Botton A. Retinoic acid signaling organizes endodermal organ specification along the entire antero-posterior axis. PLoS ONE. 2009;4:e5845. doi: 10.1371/journal.pone.0005845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben-Haim N, Lu C, Guzman-Ayala M, Pescatore L, Mesnard D, et al. The nodal precursor acting via activin receptors induces mesoderm by maintaining a source of its convertases and BMP4. Dev Cell. 2006;11:313–23. doi: 10.1016/j.devcel.2006.07.005. [DOI] [PubMed] [Google Scholar]
- Benahmed F, Gross I, Gaunt SJ, Beck F, Jehan F, et al. Multiple regulatory regions control the complex expression pattern of the mouse Cdx2 homeobox gene. Gastroenterology. 2008;135:1238–47. doi: 10.1053/j.gastro.2008.06.045. [DOI] [PubMed] [Google Scholar]
- Bhushan A, Itoh N, Kato S, Thiery JP, Czernichow P, et al. Fgf10 is essential for maintaining the proliferative capacity of epithelial progenitor cells during early pancreatic organogenesis. Development. 2001;128:5109–17. doi: 10.1242/dev.128.24.5109. [DOI] [PubMed] [Google Scholar]
- Bjornson CR, Griffin KJ, Farr GH, 3rd, Terashima A, Himeda C, et al. Eomesodermin is a localized maternal determinant required for endoderm induction in zebrafish. Dev Cell. 2005;9:523–33. doi: 10.1016/j.devcel.2005.08.010. [DOI] [PubMed] [Google Scholar]
- Bort R, Martinez-Barbera JP, Beddington RS, Zaret KS. Hex homeobox gene-dependent tissue positioning is required for organogenesis of the ventral pancreas. Development. 2004;131:797–806. doi: 10.1242/dev.00965. [DOI] [PubMed] [Google Scholar]
- Brown JL, Snir M, Noushmehr H, Kirby M, Hong SK, et al. Transcriptional profiling of endogenous germ layer precursor cells identifies dusp4 as an essential gene in zebrafish endoderm specification. Proc Natl Acad Sci USA. 2008;105:12337–42. doi: 10.1073/pnas.0805589105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burtscher I, Lickert H. Foxa2 regulates polarity and epithelialization in the endoderm germ layer of the mouse embryo. Development. 2009;136:1029–38. doi: 10.1242/dev.028415. [DOI] [PubMed] [Google Scholar]
- Calmont A, Wandzioch E, Tremblay KD, Minowada G, Kaestner KH, et al. An FGF response pathway that mediates hepatic gene induction in embryonic endoderm cells. Dev Cell. 2006;11:339–48. doi: 10.1016/j.devcel.2006.06.015. [DOI] [PubMed] [Google Scholar]
- Cardoso WV, Lu J. Regulation of early lung morphogenesis: questions, facts and controversies. Development. 2006;133:1611–24. doi: 10.1242/dev.02310. [DOI] [PubMed] [Google Scholar]
- Cervantes S, Yamaguchi TP, Hebrok M. Wnt5a is essential for intestinal elongation in mice. Dev Biol. 2009;326:285–94. doi: 10.1016/j.ydbio.2008.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chalmers AD, Slack JM. The Xenopus tadpole gut: fate maps and morphogenetic movements. Development. 2000;127:381–92. doi: 10.1242/dev.127.2.381. [DOI] [PubMed] [Google Scholar]
- Chan TM, Longabaugh W, Bolouri H, Chen HL, Tseng WF, et al. Developmental gene regulatory networks in the zebrafish embryo. Biochim Biophys Acta. 2008;1789:279–98. doi: 10.1016/j.bbagrm.2008.09.005. [DOI] [PubMed] [Google Scholar]
- Chawengsaksophak K, de Graaff W, Rossant J, Deschamps J, Beck F. Cdx2 is essential for axial elongation in mouse development. Proc Natl Acad Sci USA. 2004;101:7641–5. doi: 10.1073/pnas.0401654101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen F, Desai TJ, Qian J, Niederreither K, Lu J, Cardoso WV. Inhibition of Tgf beta signaling by endogenous retinoic acid is essential for primary lung bud induction. Development. 2007;134:2969–79. doi: 10.1242/dev.006221. [DOI] [PubMed] [Google Scholar]
- Chen Y, Pan FC, Brandes N, Afelik S, Solter M, Pieler T. Retinoic acid signaling is essential for pancreas development and promotes endocrine at the expense of exocrine cell differentiation in Xenopus. Dev Biol. 2004;271:144–60. doi: 10.1016/j.ydbio.2004.03.030. [DOI] [PubMed] [Google Scholar]
- Chung WS, Shin CH, Stainier DY. Bmp2 signaling regulates the hepatic versus pancreatic fate decision. Dev Cell. 2008;15:738–48. doi: 10.1016/j.devcel.2008.08.019. Using single-cell lineage-tracing, Chung et al. definitively demonstrated that pancreatic and hepatic lineages share a common progenitor that segregate in response to a BMP gradient. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciruna B, Rossant J. FGF signaling regulates mesoderm cell fate specification and morphogenetic movement at the primitive streak. Dev Cell. 2001;1:37–49. doi: 10.1016/s1534-5807(01)00017-x. [DOI] [PubMed] [Google Scholar]
- Clements D, Friday RV, Woodland HR. Mode of action of VegT in mesoderm and endoderm formation. Development. 1999;126:4903–11. doi: 10.1242/dev.126.21.4903. [DOI] [PubMed] [Google Scholar]
- Clevers H. Wnt/beta-Catenin Signaling in Development and Disease. Cell. 2006;127:469–80. doi: 10.1016/j.cell.2006.10.018. [DOI] [PubMed] [Google Scholar]
- Clotman F, Lannoy VJ, Reber M, Cereghini S, Cassiman D, et al. The onecut transcription factor HNF6 is required for normal development of the biliary tract. Development. 2002;129:1819–28. doi: 10.1242/dev.129.8.1819. [DOI] [PubMed] [Google Scholar]
- Coffinier C, Gresh L, Fiette L, Tronche F, Schutz G, et al. Bile system morphogenesis defects and liver dysfunction upon targeted deletion of HNF1beta. Development. 2002;129:1829–38. doi: 10.1242/dev.129.8.1829. [DOI] [PubMed] [Google Scholar]
- D’Amour KA, Agulnick AD, Eliazer S, Kelly OG, Kroon E, Baetge EE. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nat Biotechnol. 2005;23:1534–41. doi: 10.1038/nbt1163. [DOI] [PubMed] [Google Scholar]
- D’Amour KA, Bang AG, Eliazer S, Kelly OG, Agulnick AD, et al. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat Biotechnol. 2006;24:1392–401. doi: 10.1038/nbt1259. This work demonstrated that developmental signaling pathways can be used to direct the differentiation of human ES cells into hormone-expressing cells. [DOI] [PubMed] [Google Scholar]
- Dale L, Slack JM. Fate map for the 32-cell stage of Xenopus laevis. Development. 1987;99:527–51. doi: 10.1242/dev.99.4.527. [DOI] [PubMed] [Google Scholar]
- Davidson LA, Hoffstrom BG, Keller R, DeSimone DW. Mesendoderm extension and mantle closure in Xenopus laevis gastrulation: combined roles for integrin alpha(5)beta(1), fibronectin, and tissue geometry. Dev Biol. 2002;242:109–29. doi: 10.1006/dbio.2002.0537. [DOI] [PubMed] [Google Scholar]
- De Santa Barbara P, Williams J, Goldstein AM, Doyle AM, Nielsen C, et al. Bone morphogenetic protein signaling pathway plays multiple roles during gastrointestinal tract development. Dev Dyn. 2005;234:312–22. doi: 10.1002/dvdy.20554. [DOI] [PubMed] [Google Scholar]
- Dessimoz J, Opoka R, Kordich JJ, Grapin-Botton A, Wells JM. FGF signaling is necessary for establishing gut tube domains along the anterior-posterior axis in vivo. Mech Dev. 2006;123:42–55. doi: 10.1016/j.mod.2005.10.001. [DOI] [PubMed] [Google Scholar]
- Dickinson K, Leonard J, Baker JC. Genomic profiling of Mixer and Sox17beta targets during Xenopus endoderm development. Dev Dyn. 2006;235:368–81. doi: 10.1002/dvdy.20636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong PD, Munson CA, Norton W, Crosnier C, Pan X, et al. Fgf10 regulates hepatopancreatic ductal system patterning and differentiation. Nat Genet. 2007;39:397–402. doi: 10.1038/ng1961. [DOI] [PubMed] [Google Scholar]
- Duester G. Retinoic acid synthesis and signaling during early organogenesis. Cell. 2008;134:921–31. doi: 10.1016/j.cell.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dufort D, Schwartz L, Harpal K, Rossant J. The transcription factor HNF3beta is required in visceral endoderm for normal primitive streak morphogenesis. Development. 1998;125:3015–25. doi: 10.1242/dev.125.16.3015. [DOI] [PubMed] [Google Scholar]
- Egea J, Erlacher C, Montanez E, Burtscher I, Yamagishi S, et al. Genetic ablation of FLRT3 reveals a novel morphogenetic function for the anterior visceral endoderm in suppressing mesoderm differentiation. Genes Dev. 2008;22:3349–62. doi: 10.1101/gad.486708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan X, Hagos EG, Xu B, Sias C, Kawakami K, et al. Nodal signals mediate interactions between the extraembryonic and embryonic tissues in zebrafish. Dev Biol. 2007;310:363–78. doi: 10.1016/j.ydbio.2007.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fletcher G, Jones GE, Patient R, Snape A. A role for GATA factors in Xenopus gastrulation movements. Mech Dev. 2006;123:730–45. doi: 10.1016/j.mod.2006.07.007. [DOI] [PubMed] [Google Scholar]
- Franklin V, Khoo PL, Bildsoe H, Wong N, Lewis S, Tam PP. Regionalisation of the endoderm progenitors and morphogenesis of the gut portals of the mouse embryo. Mech Dev. 2008;125:587–600. doi: 10.1016/j.mod.2008.04.001. [DOI] [PubMed] [Google Scholar]
- Fukuda-Taira S. Hepatic induction in the avian embryo: Specificity of reactive endoderm and inductive mesoderm. J Embryol Exp Morph. 1981;63:111–25. [PubMed] [Google Scholar]
- Fukui A, Goto T, Kitamoto J, Homma M, Asashima M. SDF-1alpha regulates mesendodermal cell migration during frog gastrulation. Biochem Biophys Res Commun. 2007;354:472–77. doi: 10.1016/j.bbrc.2007.01.007. [DOI] [PubMed] [Google Scholar]
- Gao N, White P, Kaestner KH. Establishment of intestinal identity and epithelial-mesenchymal signaling by Cdx2. Dev Cell. 2009;16:588–99. doi: 10.1016/j.devcel.2009.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Garcia MJ, Anderson KV. Essential role of glycosaminoglycans in Fgf signaling during mouse gastrulation. Cell. 2003;114:727–37. doi: 10.1016/s0092-8674(03)00715-3. [DOI] [PubMed] [Google Scholar]
- Garcia-Garcia MJ, Shibata M, Anderson KV. Chato, a KRAB zinc-finger protein, regulates convergent extension in the mouse embryo. Development. 2008;135:3053–62. doi: 10.1242/dev.022897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Germain S, Howell M, Esslemont GM, Hill CS. Homeodomain and winged-helix transcription factors recruit activated Smads to distinct promoter elements via a common Smad interaction motif. Genes Dev. 2000;14:435–51. [PMC free article] [PubMed] [Google Scholar]
- Gittes GK. Developmental biology of the pancreas: a comprehensive review. Dev Biol. 2009;326:4–35. doi: 10.1016/j.ydbio.2008.10.024. [DOI] [PubMed] [Google Scholar]
- Goessling W, North TE, Lord AM, Ceol C, Lee S, et al. APC mutant zebrafish uncover a changing temporal requirement for wnt signaling in liver development. Dev Biol. 2008;320:161–74. doi: 10.1016/j.ydbio.2008.05.526. [DOI] [PubMed] [Google Scholar]
- Gouon-Evans V, Boussemart L, Gadue P, Nierhoff D, Koehler CI, et al. BMP-4 is required for hepatic specification of mouse embryonic stem cell-derived definitive endoderm. Nat Biotechnol. 2006;24:1402–11. doi: 10.1038/nbt1258. [DOI] [PubMed] [Google Scholar]
- Graham A. Deconstructing the pharyngeal metamere. J Exp Zoolog B Mol Dev Evol. 2008;310:336–44. doi: 10.1002/jez.b.21182. [DOI] [PubMed] [Google Scholar]
- Grapin-Botton A. Antero-posterior patterning of the vertebrate digestive tract: 40 years after Nicole Le Douarin’s PhD thesis. Int J Dev Biol. 2005;49:335–47. doi: 10.1387/ijdb.041946ag. [DOI] [PubMed] [Google Scholar]
- Grapin-Botton A, Constam D. Evolution of the mechanisms and molecular control of endoderm formation. Mech Dev. 2007;124:253–78. doi: 10.1016/j.mod.2007.01.001. [DOI] [PubMed] [Google Scholar]
- Green JB, Smith JC. Graded changes in dose of a Xenopus activin A homologue elicit stepwise transitions in embryonic cell fate. Nature. 1990;347:391–4. doi: 10.1038/347391a0. In a series of Xenopus studies, Smith and colleagues demonstrated that Activin signaling induces mesododerm fate in a dose-dependant manner, an observation that underpins endoderm induction in ES cells. [DOI] [PubMed] [Google Scholar]
- Gregorieff A, Grosschedl R, Clevers H. Hindgut defects and transformation of the gastro-intestinal tract in Tcf4(−/−)/Tcf1(−/−) embryos. EMBO J. 2004;23:1825–33. doi: 10.1038/sj.emboj.7600191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagos EG, Dougan ST. Time-dependent patterning of the mesoderm and endoderm by Nodal signals in zebrafish. BMC Dev Biol. 2007;7:22. doi: 10.1186/1471-213X-7-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haremaki T, Tanaka Y, Hongo I, Yuge M, Okamoto H. Integration of multiple signal transducing pathways on Fgf response elements of the Xenopus caudal homologue Xcad3. Development. 2003;130:4907–17. doi: 10.1242/dev.00718. [DOI] [PubMed] [Google Scholar]
- Hart AH, Hartley L, Sourris K, Stadler ES, Li R, et al. Mixl1 is required for axial mesendoderm morphogenesis and patterning in the murine embryo. Development. 2002;129:3597–608. doi: 10.1242/dev.129.15.3597. [DOI] [PubMed] [Google Scholar]
- Hilton E, Rex M, Old R. VegT activation of the early zygotic gene Xnr5 requires lifting of Tcf-mediated repression in the Xenopus blastula. Mech Dev. 2003;120:1127–38. doi: 10.1016/j.mod.2003.08.004. [DOI] [PubMed] [Google Scholar]
- Ho CY, Houart C, Wilson SW, Stainier DY. A role for the extraembryonic yolk syncytial layer in patterning the zebrafish embryo suggested by properties of the hex gene. Curr Biol. 1999;9:1131–34. doi: 10.1016/s0960-9822(99)80485-0. [DOI] [PubMed] [Google Scholar]
- Horb ME, Slack JM. Endoderm specification and differentiation in Xenopus embryos. Dev Biol. 2001;236:330–43. doi: 10.1006/dbio.2001.0347. [DOI] [PubMed] [Google Scholar]
- Horne-Badovinac S, Rebagliati M, Stainier DY. A cellular framework for gut-looping morphogenesis in zebrafish. Science. 2003;302:662–65. doi: 10.1126/science.1085397. [DOI] [PubMed] [Google Scholar]
- Hudson C, Clements D, Friday RV, Stott D, Woodland HR. Xsox17alpha and -beta mediate endoderm formation in Xenopus. Cell. 1997;91:397–405. doi: 10.1016/s0092-8674(00)80423-7. Identification of Sox17, the first unambiguous marker of endoderm progenitor cells. [DOI] [PubMed] [Google Scholar]
- Hunter MP, Wilson CM, Jiang X, Cong R, Vasavada H, et al. The homeobox gene Hhex is essential for proper hepatoblast differentiation and bile duct morphogenesis. Dev Biol. 2007;308:355–67. doi: 10.1016/j.ydbio.2007.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyde CE, Old RW. Regulation of the early expression of the Xenopus nodal-related 1 gene, Xnr1. Development. 2000;127:1221–29. doi: 10.1242/dev.127.6.1221. [DOI] [PubMed] [Google Scholar]
- Ivins S, Lammerts van Beuren K, Roberts C, James C, Lindsay E, et al. Microarray analysis detects differentially expressed genes in the pharyngeal region of mice lacking Tbx1. Dev Biol. 2005;285:554–69. doi: 10.1016/j.ydbio.2005.06.026. [DOI] [PubMed] [Google Scholar]
- Jung J, Zheng M, Goldfarb M, Zaret KS. Initiation of mammalian liver development from endoderm by fibroblast growth factors. Science. 1999;284:1998–2003. doi: 10.1126/science.284.5422.1998. Using embryonic foregut explants, Jung et al. determined that FGFs were the signal from the cardiogenic mesoderm that induce hepatic fate from the ventral foregut epithelium. [DOI] [PubMed] [Google Scholar]
- Kalinichenko VV, Zhou Y, Bhattacharyya D, Kim W, Shin B, et al. Haploinsufficiency of the mouse Forkhead Box f1 gene causes defects in gall bladder development. J Biol Chem. 2002;277:12369–74. doi: 10.1074/jbc.M112162200. [DOI] [PubMed] [Google Scholar]
- Kanai-Azuma M, Kanai Y, Gad JM, Tajima Y, Taya C, et al. Depletion of definitive gut endoderm in Sox17-null mutant mice. Development. 2002;129:2367–79. doi: 10.1242/dev.129.10.2367. [DOI] [PubMed] [Google Scholar]
- Kawazoe Y, Sekimoto T, Araki M, Takagi K, Araki K, Yamamura K. Region-specific gastrointestinal Hox code during murine embryonal gut development. Dev Growth Differ. 2002;44:77–84. doi: 10.1046/j.1440-169x.2002.00623.x. [DOI] [PubMed] [Google Scholar]
- Keller R. Early embryonic development of Xenopus laevis. Methods Cell Biol. 1991;36:61–113. doi: 10.1016/s0091-679x(08)60273-3. [DOI] [PubMed] [Google Scholar]
- Keller R. Cell migration during gastrulation. Curr Opin Cell Biol. 2005;17:533–41. doi: 10.1016/j.ceb.2005.08.006. [DOI] [PubMed] [Google Scholar]
- Kim BM, Buchner G, Miletich I, Sharpe PT, Shivdasani RA. The stomach mesenchymal transcription factor Barx1 specifies gastric epithelial identity through inhibition of transient Wnt signaling. Dev Cell. 2005;8:611–22. doi: 10.1016/j.devcel.2005.01.015. [DOI] [PubMed] [Google Scholar]
- Kim SK, Hebrok M, Li E, Oh SP, Schrewe H, et al. Activin receptor patterning of foregut organogenesis. Genes Dev. 2000;14:1866–71. [PMC free article] [PubMed] [Google Scholar]
- Kimmel CB, Warga RM, Schilling TF. Origin and organization of the zebrafish fate map. Development. 1990;108:581–94. doi: 10.1242/dev.108.4.581. [DOI] [PubMed] [Google Scholar]
- Kimura W, Yasugi S, Fukuda K. Regional specification of the endoderm in the early chick embryo. Dev Growth Differ. 2007;49:365–72. doi: 10.1111/j.1440-169X.2007.00933.x. [DOI] [PubMed] [Google Scholar]
- Kimura W, Yasugi S, Stern CD, Fukuda K. Fate and plasticity of the endoderm in the early chick embryo. Dev Biol. 2006;289:283–95. doi: 10.1016/j.ydbio.2005.09.009. [DOI] [PubMed] [Google Scholar]
- Kinkel MD, Eames SC, Alonzo MR, Prince VE. Cdx4 is required in the endoderm to localize the pancreas and limit beta-cell number. Development. 2008;135:919–29. doi: 10.1242/dev.010660. [DOI] [PubMed] [Google Scholar]
- Kofron M, Wylie C, Heasman J. The role of Mixer in patterning the early Xenopus embryo. Development. 2004;131:2431–41. doi: 10.1242/dev.01132. [DOI] [PubMed] [Google Scholar]
- Kroon E, Martinson LA, Kadoya K, Bang AG, Kelly OG, et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat Biotechnol. 2008;26:443–52. doi: 10.1038/nbt1393. [DOI] [PubMed] [Google Scholar]
- Kubo A, Shinozaki K, Shannon JM, Kouskoff V, Kennedy M, et al. Development of definitive endoderm from embryonic stem cells in culture. Development. 2004;131:1651–62. doi: 10.1242/dev.01044. [DOI] [PubMed] [Google Scholar]
- Kumar M, Jordan N, Melton D, Grapin-Botton A. Signals from lateral plate mesoderm instruct endoderm toward a pancreatic fate. Dev Biol. 2003;259:109–22. doi: 10.1016/s0012-1606(03)00183-0. [DOI] [PubMed] [Google Scholar]
- Kwon GS, Viotti M, Hadjantonakis AK. The endoderm of the mouse embryo arises by dynamic widespread intercalation of embryonic and extraembryonic lineages. Dev Cell. 2008;15:509–20. doi: 10.1016/j.devcel.2008.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Latinkic BV, Umbhauer M, Neal KA, Lerchner W, Smith JC, Cunliffe V. The Xenopus Brachyury promoter is activated by FGF and low concentrations of activin and suppressed by high concentrations of activin and by paired-type homeodomain proteins. Genes Dev. 1997;11:3265–76. doi: 10.1101/gad.11.23.3265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawson A, Schoenwolf GC. Epiblast and primitive-streak origins of the endoderm in the gastrulating chick embryo. Development. 2003;130:3491–501. doi: 10.1242/dev.00579. [DOI] [PubMed] [Google Scholar]
- Lawson KA, Meneses JJ, Pedersen RA. Cell fate and cell lineage in the endoderm of the presomite mouse embryo, studied with an intracellular tracer. Dev Biol. 1986;115:325–39. doi: 10.1016/0012-1606(86)90253-8. In a series of pioneering papers, Lawson et al. generated the first detailed mouse embryonic fate maps and description of early morphogenesis. [DOI] [PubMed] [Google Scholar]
- Lawson KA, Pedersen RA. Cell fate, morphogenetic movement and population kinetics of embryonic endoderm at the time of germ layer formation in the mouse. Development. 1987;101:627–52. doi: 10.1242/dev.101.3.627. [DOI] [PubMed] [Google Scholar]
- Le Douarin N. Synthesis of glycogen in hepatocytes undergoing differentiation: role of homologous and heterologous mesenchyma. Dev Biol. 1968;17:101–14. doi: 10.1016/0012-1606(68)90092-4. In a series of papers, Nichole Le Douarin used transplantation experiments in avian embryos to provide the first evidence of liver and pancreas induction from the mesenchyme. [DOI] [PubMed] [Google Scholar]
- Lee CS, Friedman JR, Fulmer JT, Kaestner KH. The initiation of liver development is dependent on Foxa transcription factors. Nature. 2005;435:944–47. doi: 10.1038/nature03649. [DOI] [PubMed] [Google Scholar]
- Lee JD, Silva-Gagliardi NF, Tepass U, McGlade CJ, Anderson KV. The FERM protein Epb4.1l5 is required for organization of the neural plate and for the epithelial-mesenchymal transition at the primitive streak of the mouse embryo. Development. 2007;134:2007–16. doi: 10.1242/dev.000885. [DOI] [PubMed] [Google Scholar]
- Lemaire P, Darras S, Caillol D, Kodjabachian L. A role for the vegetally expressed Xenopus gene Mix.1 in endoderm formation and in the restriction of mesoderm to the marginal zone. Development. 1998;125:2371–80. doi: 10.1242/dev.125.13.2371. [DOI] [PubMed] [Google Scholar]
- Li Y, Rankin SA, Sinner D, Kenny AP, Krieg PA, Zorn AM. Sfrp5 coordinates foregut specification and morphogenesis by antagonizing both canonical and noncanonical Wnt11 signaling. Genes Dev. 2008;22:3050–63. doi: 10.1101/gad.1687308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lickert H, Kutsch S, Kanzler B, Tamai Y, Taketo MM, Kemler R. Formation of multiple hearts in mice following deletion of beta-catenin in the embryonic endoderm. Dev Cell. 2002;3:171–81. doi: 10.1016/s1534-5807(02)00206-x. [DOI] [PubMed] [Google Scholar]
- Loose M, Patient R. A genetic regulatory network for Xenopus mesendoderm formation. Dev Biol. 2004;271:467–78. doi: 10.1016/j.ydbio.2004.04.014. [DOI] [PubMed] [Google Scholar]
- Lunde K, Belting HG, Driever W. Zebrafish pou5f1/pou2, homolog of mammalian Oct4, functions in the endoderm specification cascade. Curr Biol. 2004;14:48–55. doi: 10.1016/j.cub.2003.11.022. [DOI] [PubMed] [Google Scholar]
- Luu O, Nagel M, Wacker S, Lemaire P, Winklbauer R. Control of gastrula cell motility by the Goosecoid/Mix.1/Siamois network: basic patterns and paradoxical effects. Dev Dyn. 2008;237:1307–20. doi: 10.1002/dvdy.21522. [DOI] [PubMed] [Google Scholar]
- Maeda Y, Dave V, Whitsett JA. Transcriptional control of lung morphogenesis. Physiol Rev. 2007;87:219–44. doi: 10.1152/physrev.00028.2006. [DOI] [PubMed] [Google Scholar]
- Mahlapuu M, Enerback S, Carlsson P. Haploinsufficiency of the forkhead gene Foxf1, a target for sonic hedgehog signaling, causes lung and foregut malformations. Development. 2001;128:2397–406. doi: 10.1242/dev.128.12.2397. [DOI] [PubMed] [Google Scholar]
- Maretto S, Muller PS, Aricescu AR, Cho KW, Bikoff EK, Robertson EJ. Ventral closure, headfold fusion and definitive endoderm migration defects in mouse embryos lacking the fibronectin leucine-rich transmembrane protein FLRT3. Dev Biol. 2008;318:184–93. doi: 10.1016/j.ydbio.2008.03.021. [DOI] [PubMed] [Google Scholar]
- Martinez Barbera JP, Clements M, Thomas P, Rodriguez T, Meloy D, et al. The homeobox gene Hex is required in definitive endodermal tissues for normal forebrain, liver and thyroid formation. Development. 2000;127:2433–45. doi: 10.1242/dev.127.11.2433. [DOI] [PubMed] [Google Scholar]
- Matsui T, Raya A, Kawakami Y, Callol-Massot C, Capdevila J, et al. Noncanonical Wnt signaling regulates midline convergence of organ primordia during zebrafish development. Genes Dev. 2005;19:164–75. doi: 10.1101/gad.1253605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLin VA, Rankin SA, Zorn AM. Repression of Wnt/{beta}-catenin signaling in the anterior endoderm is essential for liver and pancreas development. Development. 2007;134:2207–17. doi: 10.1242/dev.001230. Directly demonstrated that Wnt signaling has distinct times at different times in hepatic development, first repressing foregut progenitors and then promoting liver. [DOI] [PubMed] [Google Scholar]
- Mizoguchi T, Izawa T, Kuroiwa A, Kikuchi Y. Fgf signaling negatively regulates Nodal-dependent endoderm induction in zebrafish. Dev Biol. 2006;300:612–22. doi: 10.1016/j.ydbio.2006.08.073. [DOI] [PubMed] [Google Scholar]
- Mizoguchi T, Verkade H, Heath JK, Kuroiwa A, Kikuchi Y. Sdf1/Cxcr4 signaling controls the dorsal migration of endodermal cells during zebrafish gastrulation. Development. 2008;135:2521–29. doi: 10.1242/dev.020107. [DOI] [PubMed] [Google Scholar]
- Molotkov A, Molotkova N, Duester G. Retinoic acid generated by Raldh2 in mesoderm is required for mouse dorsal endodermal pancreas development. Dev Dyn. 2005;232:950–57. doi: 10.1002/dvdy.20256. [DOI] [PubMed] [Google Scholar]
- Moniot B, Biau S, Faure S, Nielsen CM, Berta P, et al. SOX9 specifies the pyloric sphincter epithelium through mesenchymal-epithelial signals. Development. 2004;131:3795–804. doi: 10.1242/dev.01259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moody SA. Fates of the blastomeres of the 32-cell-stage Xenopus embryo. Dev Biol. 1987;122:300–19. doi: 10.1016/0012-1606(87)90296-x. [DOI] [PubMed] [Google Scholar]
- Murtaugh LC. Pancreas and beta-cell development: from the actual to the possible. Development. 2007;134:427–38. doi: 10.1242/dev.02770. [DOI] [PubMed] [Google Scholar]
- Murtaugh LC, Law AC, Dor Y, Melton DA. Beta-catenin is essential for pancreatic acinar but not islet development. Development. 2005;132:4663–74. doi: 10.1242/dev.02063. [DOI] [PubMed] [Google Scholar]
- Nair S, Schilling TF. Chemokine signaling controls endodermal migration during zebrafish gastrulation. Science. 2008;322:89–92. doi: 10.1126/science.1160038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieuwkoop PD. The formation of the mesoderm in the urodelean amphibians. I. Induction by the endoderm. Wilhelm Roux Arch EntwMech Org. 1969;162:341–73. doi: 10.1007/BF00578701. [DOI] [PubMed] [Google Scholar]
- Norris DP, Brennan J, Bikoff EK, Robertson EJ. The Foxh1-dependent autoregulatory enhancer controls the level of Nodal signals in the mouse embryo. Development. 2002;129:3455–68. doi: 10.1242/dev.129.14.3455. [DOI] [PubMed] [Google Scholar]
- Ober EA, Field HA, Stainier DY. From endoderm formation to liver and pancreas development in zebrafish. Mech Dev. 2003;120:5–18. doi: 10.1016/s0925-4773(02)00327-1. [DOI] [PubMed] [Google Scholar]
- Ober EA, Olofsson B, Makinen T, Jin SW, Shoji W, et al. Vegfc is required for vascular development and endoderm morphogenesis in zebrafish. EMBO Rep. 2004;5:78–84. doi: 10.1038/sj.embor.7400047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogata S, Morokuma J, Hayata T, Kolle G, Niehrs C, et al. TGF-beta signaling-mediated morphogenesis: modulation of cell adhesion via cadherin endocytosis. Genes Dev. 2007;21:1817–31. doi: 10.1101/gad.1541807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveri P, Davidson EH. Gene regulatory network controlling embryonic specification in the sea urchin. Curr Opin Genet Dev. 2004;14:351–60. doi: 10.1016/j.gde.2004.06.004. [DOI] [PubMed] [Google Scholar]
- Ormestad M, Astorga J, Landgren H, Wang T, Johansson BR, et al. Foxf1 and Foxf2 control murine gut development by limiting mesenchymal Wnt signaling and promoting extracellular matrix production. Development. 2006;133:833–43. doi: 10.1242/dev.02252. [DOI] [PubMed] [Google Scholar]
- Osada SI, Saijoh Y, Frisch A, Yeo CY, Adachi H, et al. Activin/nodal responsiveness and asymmetric expression of a Xenopus nodal-related gene converge on a FAST-regulated module in intron 1. Development. 2000;127:2503–14. doi: 10.1242/dev.127.11.2503. [DOI] [PubMed] [Google Scholar]
- Petryk A, Anderson RM, Jarcho MP, Leaf I, Carlson CS, et al. The mammalian twisted gastrulation gene functions in foregut and craniofacial development. Dev Biol. 2004;267:374–86. doi: 10.1016/j.ydbio.2003.11.015. [DOI] [PubMed] [Google Scholar]
- Pezeron G, Mourrain P, Courty S, Ghislain J, Becker TS, et al. Live analysis of endodermal layer formation identifies random walk as a novel gastrulation movement. Curr Biol. 2008;18:276–81. doi: 10.1016/j.cub.2008.01.028. [DOI] [PubMed] [Google Scholar]
- Pilon N, Oh K, Sylvestre JR, Bouchard N, Savory J, Lohnes D. Cdx4 is a direct target of the canonical Wnt pathway. Dev Biol. 2006;289:55–63. doi: 10.1016/j.ydbio.2005.10.005. [DOI] [PubMed] [Google Scholar]
- Poulain M, Furthauer M, Thisse B, Thisse C, Lepage T. Zebrafish endoderm formation is regulated by combinatorial Nodal, FGF and BMP signaling. Development. 2006;133:2189–200. doi: 10.1242/dev.02387. [DOI] [PubMed] [Google Scholar]
- Que J, Choi M, Ziel JW, Klingensmith J, Hogan BL. Morphogenesis of the trachea and esophagus: current players and new roles for noggin and Bmps. Differentiation. 2006;74:422–37. doi: 10.1111/j.1432-0436.2006.00096.x. [DOI] [PubMed] [Google Scholar]
- Que J, Okubo T, Goldenring JR, Nam KT, Kurotani R, et al. Multiple dose-dependent roles for Sox2 in the patterning and differentiation of anterior foregut endoderm. Development. 2007;134:2521–31. doi: 10.1242/dev.003855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rakeman AS, Anderson KV. Axis specification and morphogenesis in the mouse embryo require Nap1, a regulator of WAVE-mediated actin branching. Development. 2006;133:3075–83. doi: 10.1242/dev.02473. [DOI] [PubMed] [Google Scholar]
- Reim G, Mizoguchi T, Stainier DY, Kikuchi Y, Brand M. The POU domain protein spg (pou2/Oct4) is essential for endoderm formation in cooperation with the HMG domain protein casanova. Dev Cell. 2004;6:91–101. doi: 10.1016/s1534-5807(03)00396-4. [DOI] [PubMed] [Google Scholar]
- Rosenquist GC. The location of the pregut endoderm in the chick embryo at the primitive streak stage as determined by radioautographic mapping. Dev Biol. 1971;26:323–35. doi: 10.1016/0012-1606(71)90131-x. [DOI] [PubMed] [Google Scholar]
- Rubin DC. Intestinal morphogenesis. Curr Opin Gastroenterol. 2007;23:111–14. doi: 10.1097/MOG.0b013e3280145082. [DOI] [PubMed] [Google Scholar]
- Sancho E, Batlle E, Clevers H. Signaling pathways in intestinal development and cancer. Annu Rev Cell Dev Biol. 2004;20:695–723. doi: 10.1146/annurev.cellbio.20.010403.092805. [DOI] [PubMed] [Google Scholar]
- Schier AF, Talbot WS. Molecular genetics of axis formation in zebrafish. Annu Rev Genet. 2005;39:561–613. doi: 10.1146/annurev.genet.37.110801.143752. [DOI] [PubMed] [Google Scholar]
- Semenov MV, Habas R, Macdonald BT, He X. Snapshot: noncanonical Wnt signaling pathways. Cell. 2007;131:1378. doi: 10.1016/j.cell.2007.12.011. [DOI] [PubMed] [Google Scholar]
- Serls AE, Doherty S, Parvatiyar P, Wells JM, Deutsch GH. Different thresholds of fibroblast growth factors pattern the ventral foregut into liver and lung. Development. 2005;132:35–47. doi: 10.1242/dev.01570. [DOI] [PubMed] [Google Scholar]
- Shen MM. Nodal signaling: developmental roles and regulation. Development. 2007;134:1023–34. doi: 10.1242/dev.000166. [DOI] [PubMed] [Google Scholar]
- Sherwood RI, Chen TY, Melton DA. Transcriptional dynamics of endodermal organ formation. Dev Dyn. 2009;238:29–42. doi: 10.1002/dvdy.21810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu T, Bae YK, Hibi M. Cdx-Hox code controls competence for responding to Fgfs and retinoic acid in zebrafish neural tissue. Development. 2006;133:4709–19. doi: 10.1242/dev.02660. [DOI] [PubMed] [Google Scholar]
- Shin D, Shin CH, Tucker J, Ober EA, Rentzsch F, et al. Bmp and Fgf signaling are essential for liver specification in zebrafish. Development. 2007;134:2041–50. doi: 10.1242/dev.000281. [DOI] [PubMed] [Google Scholar]
- Shroyer NF, Helmrath MA, Wang VY, Antalffy B, Henning SJ, Zoghbi HY. Intestine-specific ablation of mouse atonal homolog 1 (Math1) reveals a role in cellular homeostasis. Gastroenterology. 2007;132:2478–88. doi: 10.1053/j.gastro.2007.03.047. [DOI] [PubMed] [Google Scholar]
- Sinner D, Kirilenko P, Rankin S, Wei E, Howard L, et al. Global analysis of the transcriptional network controlling Xenopus endoderm formation. Development. 2006;133:1955–66. doi: 10.1242/dev.02358. [DOI] [PubMed] [Google Scholar]
- Sinner D, Rankin S, Lee M, Zorn AM. Sox17 and beta-catenin cooperate to regulate the transcription of endodermal genes. Development. 2004;131:3069–80. doi: 10.1242/dev.01176. [DOI] [PubMed] [Google Scholar]
- Soto-Gutierrez A, Kobayashi N, Rivas-Carrillo JD, Navarro-Alvarez N, Zhao D, et al. Reversal of mouse hepatic failure using an implanted liver-assist device containing ES cell-derived hepatocytes. Nat Biotechnol. 2006;24:1412–19. doi: 10.1038/nbt1257. [DOI] [PubMed] [Google Scholar]
- Spence JR, Wells JM. Translational embryology: Using embryonic principles to generate pancreatic endocrine cells from embryonic stem cells. Dev Dyn. 2007;236:3218–27. doi: 10.1002/dvdy.21366. [DOI] [PubMed] [Google Scholar]
- Spence JR, Lange AW, Lin SC, Kaestner KH, Lowy AM, et al. Sox17 regulates organ lineage segregation of ventral foregut progenitor cells. Dev Cell. 2009 doi: 10.1016/j.devcel.2009.05.012. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stafford D, Prince VE. Retinoic acid signaling is required for a critical early step in zebrafish pancreatic development. Curr Biol. 2002;12:1215–20. doi: 10.1016/s0960-9822(02)00929-6. [DOI] [PubMed] [Google Scholar]
- Stainier DY. A glimpse into the molecular entrails of endoderm formation. Genes Dev. 2002;16:893–907. doi: 10.1101/gad.974902. [DOI] [PubMed] [Google Scholar]
- Stringer EJ, Pritchard CA, Beck F. Cdx2 initiates histodifferentiation of the midgut endoderm. FEBS Lett. 2008;582:2555–60. doi: 10.1016/j.febslet.2008.06.024. [DOI] [PubMed] [Google Scholar]
- Sun X, Meyers EN, Lewandoski M, Martin GR. Targeted disruption of Fgf8 causes failure of cell migration in the gastrulating mouse embryo. Genes Dev. 1999;13:1834–46. doi: 10.1101/gad.13.14.1834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tada S, Era T, Furusawa C, Sakurai H, Nishikawa S, et al. Characterization of mesendoderm: a diverging point of the definitive endoderm and mesoderm in embryonic stem cell differentiation culture. Development. 2005;132:4363–74. doi: 10.1242/dev.02005. First experimental evidence for a bipotential mesenoderm progenitor in mice, highlighting the power of ES cell cultures in the study of early mammalian development. [DOI] [PubMed] [Google Scholar]
- Tam PP, Khoo PL, Lewis SL, Bildsoe H, Wong N, et al. Sequential allocation and global pattern of movement of the definitive endoderm in the mouse embryo during gastrulation. Development. 2007;134:251–60. doi: 10.1242/dev.02724. [DOI] [PubMed] [Google Scholar]
- Tam PP, Khoo PL, Wong N, Tsang TE, Behringer RR. Regionalization of cell fates and cell movement in the endoderm of the mouse gastrula and the impact of loss of Lhx1(Lim1) function. Dev Biol. 2004;274:171–87. doi: 10.1016/j.ydbio.2004.07.005. [DOI] [PubMed] [Google Scholar]
- Tam PP, Loebel DA. Gene function in mouse embryogenesis: get set for gastrulation. Nat Rev Genet. 2007;8:368–81. doi: 10.1038/nrg2084. [DOI] [PubMed] [Google Scholar]
- Tamplin OJ, Kinzel D, Cox BJ, Bell CE, Rossant J, Lickert H. Microarray analysis of Foxa2 mutant mouse embryos reveals novel gene expression and inductive roles for the gastrula organizer and its derivatives. BMC Genomics. 2008;9:511. doi: 10.1186/1471-2164-9-511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas PQ, Brown A, Beddington RS. Hex: a homeobox gene revealing peri-implantation asymmetry in the mouse embryo and an early transient marker of endothelial cell precursors. Development. 1998;125:85–94. doi: 10.1242/dev.125.1.85. [DOI] [PubMed] [Google Scholar]
- Tiso N, Filippi A, Pauls S, Bortolussi M, Argenton F. BMP signaling regulates anteroposterior endoderm patterning in zebrafish. Mech Dev. 2002;118:29–37. doi: 10.1016/s0925-4773(02)00252-6. [DOI] [PubMed] [Google Scholar]
- Tremblay KD, Zaret KS. Distinct populations of endoderm cells converge to generate the embryonic liver bud and ventral foregut tissues. Dev Biol. 2005;280:87–99. doi: 10.1016/j.ydbio.2005.01.003. [DOI] [PubMed] [Google Scholar]
- Tzur G, Levy A, Meiri E, Barad O, Spector Y, et al. MicroRNA expression patterns and function in endodermal differentiation of human embryonic stem cells. PLoS ONE. 2008;3:e3726. doi: 10.1371/journal.pone.0003726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Den Brink GR. Hedgehog signaling in development and homeostasis of the gastrointestinal tract. Physiol Rev. 2007;87:1343–75. doi: 10.1152/physrev.00054.2006. [DOI] [PubMed] [Google Scholar]
- Verzi MP, Stanfel MN, Moses KA, Kim BM, Zhang Y, et al. Role of the homeodomain transcription factor Bapx1 in mouse distal stomach development. Gastroenterology. 2009;136:1701–10. doi: 10.1053/j.gastro.2009.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z, Dolle P, Cardoso WV, Niederreither K. Retinoic acid regulates morphogenesis and patterning of posterior foregut derivatives. Dev Biol. 2006;297:433–45. doi: 10.1016/j.ydbio.2006.05.019. [DOI] [PubMed] [Google Scholar]
- Wardle FC, Smith JC. Refinement of gene expression patterns in the early Xenopus embryo. Development. 2004;131:4687–96. doi: 10.1242/dev.01340. [DOI] [PubMed] [Google Scholar]
- Warga RM, Nusslein-Volhard C. Origin and development of the zebrafish endoderm. Development. 1999;126:827–38. doi: 10.1242/dev.126.4.827. [DOI] [PubMed] [Google Scholar]
- Watt AJ, Zhao R, Li J, Duncan SA. Development of the mammalian liver and ventral pancreas is dependent on GATA4. BMC Dev Biol. 2007;7:37. doi: 10.1186/1471-213X-7-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wells JM, Esni F, Boivin GP, Aronow BJ, Stuart W, et al. Wnt/beta-catenin signaling is required for development of the exocrine pancreas. BMC Dev Biol. 2007;7:4. doi: 10.1186/1471-213X-7-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wells JM, Melton DA. Early mouse endoderm is patterned by soluble factors from adjacent germ layers. Development. 2000;127:1563–72. doi: 10.1242/dev.127.8.1563. Demonstration that cell fate in the mouse gastrula endoderm is plastic and could be patterned by FGF4. [DOI] [PubMed] [Google Scholar]
- Wills A, Dickinson K, Khokha M, Baker JC. Bmp signaling is necessary and sufficient for ventrolateral endoderm specification in Xenopus. Dev Dyn. 2008;237:2177–86. doi: 10.1002/dvdy.21631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wylie CC, Snape A, Heasman J, Smith JC. Vegetal pole cells and commitment to form endoderm in Xenopus laevis. Dev Biol. 1987;119:496–502. doi: 10.1016/0012-1606(87)90052-2. [DOI] [PubMed] [Google Scholar]
- Xanthos JB, Kofron M, Tao Q, Schaible K, Wylie C, Heasman J. The roles of three signaling pathways in the formation and function of the Spemann Organizer. Development. 2002;129:4027–43. doi: 10.1242/dev.129.17.4027. [DOI] [PubMed] [Google Scholar]
- Xanthos JB, Kofron M, Wylie C, Heasman J. Maternal VegT is the initiator of a molecular network specifying endoderm in Xenopus laevis. Development. 2001;128:167–80. doi: 10.1242/dev.128.2.167. [DOI] [PubMed] [Google Scholar]
- Yamamoto M, Saijoh Y, Perea-Gomez A, Shawlot W, Behringer RR, et al. Nodal antagonists regulate formation of the anteroposterior axis of the mouse embryo. Nature. 2004;428:387–92. doi: 10.1038/nature02418. [DOI] [PubMed] [Google Scholar]
- Yasugi S, Mizuno T. Molecular analysis of endoderm regionalization. Dev Growth Differ. 2008;50(Suppl 1):S79–96. doi: 10.1111/j.1440-169X.2008.00984.x. [DOI] [PubMed] [Google Scholar]
- Yasunaga M, Tada S, Torikai-Nishikawa S, Nakano Y, Okada M, et al. Induction and monitoring of definitive and visceral endoderm differentiation of mouse ES cells. Nat Biotechnol. 2005;23:1542–50. doi: 10.1038/nbt1167. [DOI] [PubMed] [Google Scholar]
- Zacchetti G, Duboule D, Zakany J. Hox gene function in vertebrate gut morphogenesis: the case of the caecum. Development. 2007;134:3967–73. doi: 10.1242/dev.010991. [DOI] [PubMed] [Google Scholar]
- Zaret KS. Regulatory phases of early liver development: paradigms of organogenesis. Nat Rev Genet. 2002;3:499–512. doi: 10.1038/nrg837. [DOI] [PubMed] [Google Scholar]
- Zaret KS. Genetic programming of liver and pancreas progenitors: lessons for stem-cell differentiation. Nat Rev Genet. 2008;9:329–40. doi: 10.1038/nrg2318. [DOI] [PubMed] [Google Scholar]
- Zaret KS, Grompe M. Generation and regeneration of cells of the liver and pancreas. Science. 2008;322:1490–94. doi: 10.1126/science.1161431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao R, Duncan SA. Embryonic development of the liver. Hepatology. 2005;41:956–67. doi: 10.1002/hep.20691. [DOI] [PubMed] [Google Scholar]
- Zohn IE, Li Y, Skolnik EY, Anderson KV, Han J, Niswander L. p38 and a p38-interacting protein are critical for downregulation of E-cadherin during mouse gastrulation. Cell. 2006;125:957–69. doi: 10.1016/j.cell.2006.03.048. [DOI] [PubMed] [Google Scholar]
- Zorn A. StemBook. The Stem Cell Research Community. StemBook; 2008. Liver development. http://www.stembook.org. [DOI] [Google Scholar]
- Zorn AM, Butler K, Gurdon JB. Anterior endomesoderm specification in Xenopus by Wnt/beta-catenin and TGF-beta signaling pathways. Dev Biol. 1999;209:282–97. doi: 10.1006/dbio.1999.9257. [DOI] [PubMed] [Google Scholar]
- Zorn AM, Wells JM. Molecular basis of vertebrate endoderm development. Int Rev Cytol. 2007;259:49–111. doi: 10.1016/S0074-7696(06)59002-3. [DOI] [PubMed] [Google Scholar]