Abstract
Metabolites of arachidonic acid influence sodium chloride (NaCl) transport in the thick ascending limb. Because a 10 pS Cl channel is the major type of chloride channel in the basolateral membrane of this nephron segment, we explored the effect of arachidonic acid on this channel in cell-attached patches. Addition of 5μmol arachidonic acid significantly decreased channel activity (a product of channel number and open probability) while linoleic acid had no effect. To determine if this was mediated by acachidonic acid per se or by its metabolites, we measured channel activity in the presence of the cyclooxygenase inhibitor indomethacin, the selective lipoxygenase inhibitor nordihydroguaiaretic acid, and the cytochrome P-450 (CYP)-ω-hydroxylation inhibitor 17-octadecynoic acid. Neither cyclooxygenase nor lipoxygenase inhibition had an effect on basal chloride channel activity; further they failed to abolish the inhibitory effect of arachidonate on the 10 pS channel. However, inhibition of CYP-ω-hydroxylation completely abolished the effect of arachidonic acid. The similarity of the effects of 20-hydroxyeicosatetraenoic acid (20-HETE) and arachidonic acid suggests that the effect of arachidonic acid was mediated by CYP-ω-hydroxylation-dependent metabolites. We conclude that arachidonic acid inhibits the 10 pS chloride channel in the basolateral membrane of the medullary thick ascending limb, an effect mediated by the CYP-ω-hydroxylation-dependent metabolite 20-HETE.
Keywords: Cl transport, cyclooxygenase, CYP-ω-hydroxylation, 20-HETE
The thick ascending limb (TAL) is responsible for reabsorption of 25% filtered NaCl load and has a key role in urinary concentrating mechanism.1–3 For salt reabsorption, Na and Cl enter the cell across the apical membrane through type 2 Na/K/2Cl cotransporter,4 whereas Na leaves cell through basolateral Na-K-ATPase and Cl exits across the basolateral membrane through Cl channels.5 Although type 1 KCl cotransporter has been also shown to be expressed in the basolateral membrane of the TAL,6,7 the role of KCl cotransporter in mediating Cl exit is not explored. The driving force for Cl exit across the basolateral membrane is the cell membrane potential such that a hyperpolarization enhances while a depolarization decreases Cl exit.3 Patch-clamp studies have shown that two types of Cl channels, a 8–9 pS and a 20–40 pS, are expressed in the basolateral membrane of the TAL and that the 8–9 pS Cl channel is a main type of Cl channels expressed in the basolateral membrane.8 Molecular cloning has also identified two types of Cl channels, ClC-K1 and ClC-K2, in the Henle's loop.9,10 Moreover, immunostaining shows that ClC-K1 is mainly expressed in the thin ascending limb of Henle's loop,11 whereas ClC-K2 which has two isoforms, CLC-K2L and CLC-K2S,10 is predominantly expressed in the basolateral membrane of the TAL in the rat kidney,12 suggesting that ClC-K2 is responsible for the basolateral Cl conductance. Furthermore, cystic fibrosis transmembrane conductance regulator (CFTR), a Cl channel expressed in the lung and pancreatic tissue, is also expressed in the TAL.13 However, CFTR seems not to have an important function in mediating Cl absorption in the TAL because Cl absorption in the TAL is normal in CFTR-null mice.14
Although the basolateral Cl channels have such an important function, their regulatory mechanism is not completely understood. It has been shown that the 8–9 pS Cl channels are pH sensitive and stimulated by cAMP.15 A large body of evidence indicates that cytochrome P450 (CYP) monooxygenase-dependent metabolites of arachidonic acid (AA) have an important function in regulating NaCl absorption in the TAL.16–18 It has been shown that 20-hydroxyeicosatetraenoic acid (20-HETE), a metabolite of CYP monooxygenase, inhibits Na/K/2Cl cotransporter,16,19 the apical 70-pS K channels which is essential for K recycling and maintaining the Na/K/2Cl cotransporter activity20 and the basolateral 50-pS K channels in the medullary TAL (mTAL).21 Because an integrated mechanism of NaCl transport in the mTAL requires the involvement of basolateral Cl channels, it is conceivable that basolateral Cl channel activity must work in concert with Na/K/2Cl cotransporter. Therefore, the aim of this study is to examine the role of CYP-ω-hydroxylation-dependent AA metabolism in regulating the basolateral Cl channels.
Results
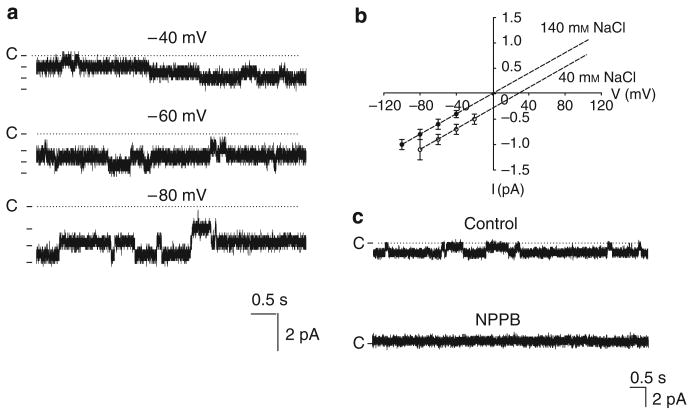
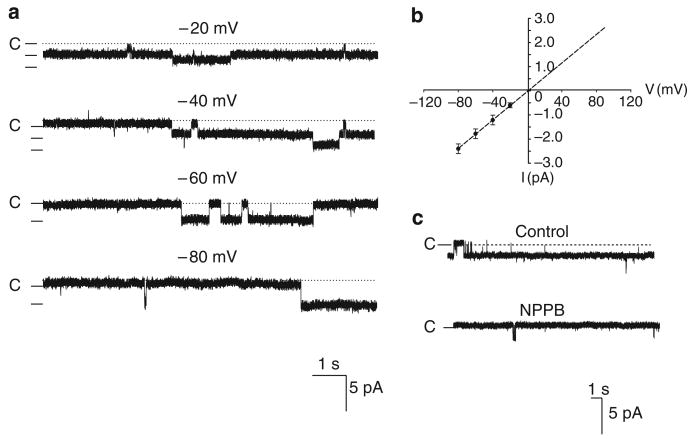
We confirmed the report by Guinamard et al.8 that two types of Cl channels, a small conductance and an intermediated conductance, are expressed in the basolateral membrane of the TAL. Figure 1a is a channel recording showing the small-conductance Cl channel activity at −40, −60, and −80 mV in a cell-attached patch with 140 mmol NaCl in both pipette and bath solutions. Figure 1b is an I/V curve of the Cl channel in inside-out patches measured with a symmetrical 140 mmol NaCl in both bath and pipette (filled circle) at −100 (the trace not included), −80, −60, and −40 mV or with 140 mmol NaCl in the bath and 40 mmol NaCl/100 mmol sodium gluconate in the pipette (circles) at −80, −60, −40, and −20 mV, respectively. Both I/V curves yield a channel conductance of 10.4±1.6 pS (n=10). Moreover, reducing pipette Cl concentration from 140 to 40 mmol shifts a reverse potential by 30±5mV (n=3), indicating that the 10 pS channel is a Cl-sensitive channel. This notion is also supported by the finding that application of 0.1 mmol NPPB in the bath facing the cytoplasmic side of an inside-out patch inhibited the small-conductance Cl channels (Figure 1c). In addition to 10 pS Cl channel, we have also identified an intermediate-conductance Cl channel. Figure 2a is a channel recording made in a cell-attached patch showing the channel activity with symmetrical 140 mmol NaCl in both bath and pipette at −20, −40, −60, and −80 mV, respectively. Figure 2b is an I/V curve measured with symmetrical 140 mmol NaCl solution in the pipette and bath and it yields a slope conductance of 30±2pS (n=5). We were not able to measure the Cl current with 40 mmol NaCl/100 mmol sodium gluconate in the pipette for the 30 pS Cl channel because the probability of finding the 30 pS Cl channel was low. However, the finding that this channel was also inhibited by 0.1 mmol NPPB applied in the bath facing the cytoplasmic side of an inside-out patch suggests that the 30 pS channel is a Cl channel (Figure 2c). We observed 10 pS Cl channel activity in 418 patches, the 30 pS Cl channels in 46 patches, and both Cl channels in the same patch in 19 patches from the total 1468 patches. Thus, the 10 pS Cl channel is the major type of Cl channels expressed in the TAL.
Figure 1. A 10 pS Cl channel in the thick ascending limb.
(a) A channel recording showing the activity of the basolateral 10 pS Cl channel in a cell-attached patch with symmetrical 140 mmol NaCl in both the pipette and the bath solutions. The holding potential is indicated on the top of each trace and ‘C’ indicates the channel-closed level. (b) The I/V curve of the 10 pS Cl channel in inside-out patches measured with symmetrical 140 mmol NaCl in both the bath and the pipette (filled circles) or with 140 mmol NaCl in the bath and 40 mmol NaCl/100 mmol sodium gluconate in the pipette (circles), respectively. (c) Effects of 0.1 mmol NPPB on the 10 pS Cl channel in an inside-out patch (holding potential was −80 mV).
Figure 2. A 30 pS Cl channel in the thick ascending limb.
(a) A channel recording showing the activity of the basolateral 30 pS Cl channel in a cell-attached patch with symmetrical 140mmol NaCl in both the pipette and the bath solutions. The holding potential is indicated on the top of each trace and ‘C’ indicates the channel-closed level. (b) The I/V curve of the Cl channel in inside-out patches measured with symmetrical 140mmol NaCl in both bath and pipette. (c) Effects of 0.1 mmol NPPB on Cl channel in an inside-out patch (holding potential was −80mV).
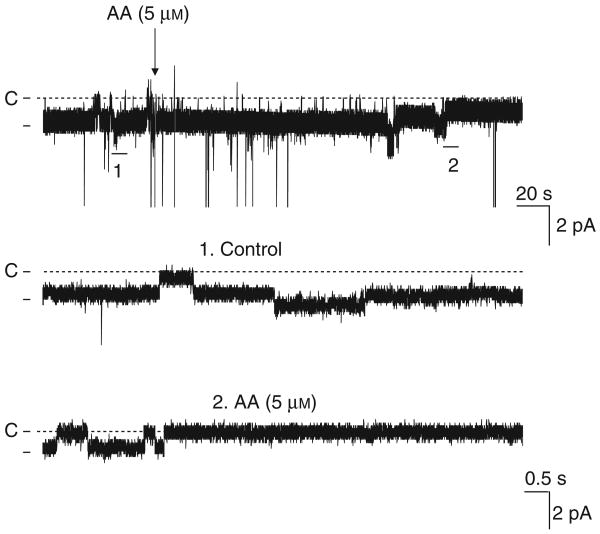
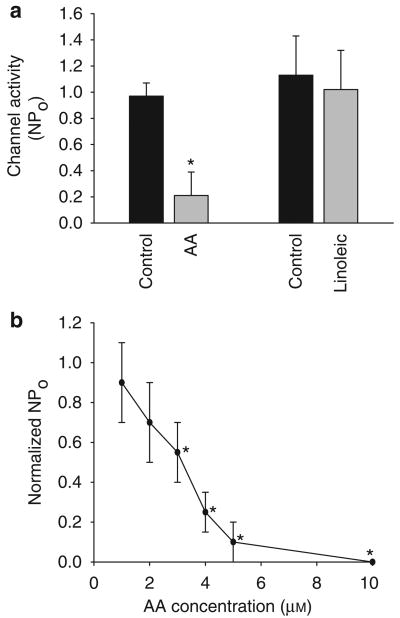
Because the 10 pS Cl channel is the major type of Cl channels in the basolateral membrane of the mTAL, we focused on investigating the regulation of the 10 pS Cl channels by AA. Figure 3 is a representative recording showing that AA inhibits the basolateral 10 pS Cl channel in a cell-attached patch. Addition of 5 μmol AA decreased channel activity, defined by NPo, from 0.97±0.08 to 0.21±0.18 (n=8, P<0.01) (Figure 4a). Figure 4b is a dose–response curve of AA effect on channel activity in cell-attached patches, showing that 3 μmol AA could significantly inhibit Cl channels by 50%. Because free AA concentrations inside of cells are in micromole range (Personal communication from Dr J Capdevila, University of Vanderbilt), the finding that 3 μm AA inhibited the 10 pS Cl channels by 50% suggests that AA is a physiological player for regulating transepithelial Cl transport in the TAL. To test the specificity of the AA effect, we also examined the effect of linoleic acid, an 18-carbon fatty acid with two double bonds, on the 10 pS Cl channel in a cell-attached patch. Figure 4a summarizes results showing that addition of 5 μmol linoleic acid did not inhibit the 10 pS Cl channels (control NPo, 1.13±0.32; linoleic acid 1.01±0.3) (n=5, P>0.05). This suggests that the effect of AA is not due to changing membrane lipid fluidity or other nonspecific fatty acid-induced effect.
Figure 3. Effect of arachidonic acid (AA) (5μmol) on the 10 pS Cl channel activity in the basolateral membrane of the medullary thick ascending limb.
The experiment was performed in a cell-attached patch with 140 mmol NaCl in pipette and 5 mmol KCl/140 mmol NaCl in the bath. The top trace shows the time course of the experiment. Two parts of the trace, indicated by numbers, are extended to show the fast time resolution. Holding potential was −80 mV. The large downward-deflection is an electrical interference.
Figure 4. AA specifically inhibits Cl channels.
(a) Effect of arachidonic acid (AA) and linoleic acid (5 μmol) on the 10 pS Cl channel activity in the basolateral membrane of the medullary thick ascending limb. The experiment is performed in a cell-attached patch. (b) A dose–response curve of the AA effect on the basolateral 10 pS Cl channels in cell-attached patches. Asterisk indicates a significant difference in comparison with control (no AA) (P<0.05).
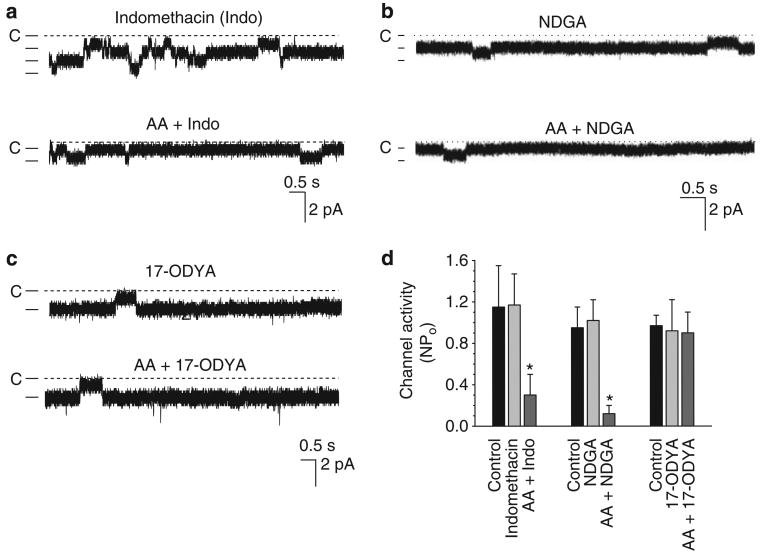
After establishing that AA specifically inhibits the 10 pS Cl channels, we explored whether this effect depends on metabolic pathways of AA, which are metabolized by three types of enzymes: COX, lipoxygenase, and CYP monooxygenase. Although both COX and CYP monooxygenase are expressed and involved in regulating epithelial transport in the mTAL,22–25 two types lipoxygenases, 5 and 12-lipoxygenase, are also shown to be expressed in the renal tissue including distal convoluted tubule cells.26,27 Therefore, we explored whether the effect of AA was mediated by COX-dependent, lipoxygenase-dependent, or CYP-monooxygenase-dependent metabolites of AA. Figure 5a is a representative channel recording in a cell-attached patch showing the effect of AA on 10 pS Cl channels in the presence of indomethacin (5 μmol), a non-selective COX1 and COX2 inhibitor. It is apparent that indomethacin did not affect channel activity significantly and failed to block the AA-mediated inhibition. Results summarized in Figure 5d show that addition of 5 μm AA still inhibited the Cl channel, and decreased NPo from 1.17±0.3 to 0.30±0.20 (n=5, P<0.05). This suggests that it is unlikely that the AA-induced acute inhibition of the 10 pS Cl channel was mediated by a COX-dependent metabolite of AA.
Figure 5. CYP-ω-hydroxylation of AA inhibits Cl channels.
(a) Effect of arachidonic acid (AA) on the basolateral 10 pS Cl channel activity in a cell-attached patch in the presence of indomethacin. The channel closed level is indicated by a dotted line and ‘C’. (b) Effect of AA (5 μmol) on the 10 pS Cl channels in a cell-attached patch in the presence of NDGA (2.5 μmol). (c) Effect of AA (5μmol) on the 10 pS Cl channels in a cell-attached patch in the presence of 17-ODYA (5 μmol). (d) Bar graphs summarize the effect of AA (5 μmol) on 10 pS Cl channels in the presence of indomethacin (5 μmol), NDGA (2.5 μmol), and 17-ODYA (5 μmol), respectively. The holding potentials in experiments were always at −80mV and asterisk indicates a significant difference in comparison with control value (P<0.05). NDGA, nordihydroguaiaretic acid.
We also examined the effect of AA on the 10 pS Cl channel in the presence of nordihydroguaiaretic acid (NDGA), a non-selective lipoxygenase inhibitor.28 Although chronic use of NDGA-containing substances has been shown to affect the CYP enzyme system thereby causing liver and renal damage,29 NDGA at concentrations of micromole ranges inhibits mainly lipoxygenases. Figure 5b is a channel recording showing that AA inhibited the 10 pS Cl channel in the presence of NDGA (2.5 μm). Results from five experiments are summarized in Figure 5d, showing that NDGA had no significant effect on the Cl channel activity (NPo, 1.02±0.2), a value not significantly different from the control (0.95±0.2). Moreover, 5 μm AA decreased NPo to 0.12±0.08 in the presence of NDGA, suggesting that the effect of AA was not mediated by lipoxygenase-dependent pathway. We next examined the effect of AA on Cl channel activity in the presence of 17-ODYA, an inhibitor of CYP-ω-hydroxylation of AA. Figure 5c is a channel recording illustrating the effect of AA on channel activity in a cell-attached patch in the presence of 5 μm 17-ODYA. Although inhibiting the CYP-ω-hydroxylation of AA had no significant effect on the basal level of channel activity (0.92±0.30) (Figure 5d), it completely abolished the inhibitory effect of AA, because 5 μm AA did not decrease NPo (0.90±0.20, n=5).
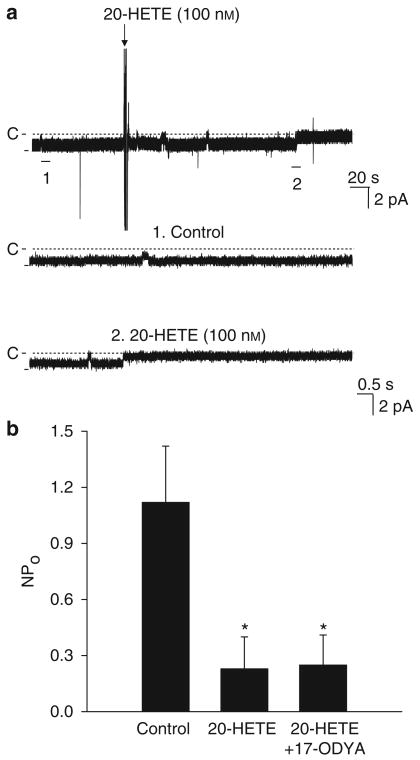
After showing that the AA-induced inhibition of Cl channels was mediated by CYP-ω-hydroxylase-dependent metabolic pathway of AA, we examined the effect of 20-HETE on the 10 pS Cl channels, a main product of CYP-ω-hydroxylation of AA in the mTAL.30 Figure 6a is a typical channel recording in a cell-attached patch showing that the application of 100 nm 20-HETE inhibited the activity of the 10 pS Cl channels and decreased NPo from 1.12±0.30 to 0.23±0.17 (n=7, P<0.05). Moreover, the effect of 20-HETE was also observed in the presence of 5 μm 17-ODYA and Figure 6b shows that 100 nm 20-HETE decreased NPo to 0.25±0.16. 17-ODYA completely blocked the effect of AA on the 10 pS Cl channel but failed to abolish the 20-HETE-mediated inhibition, suggesting that the inhibitory effect of AA is mediated by the CYP-ω-hydroxylation-dependent pathway and that 20-HETE is a mediator of AA effect.
Figure 6. 20-HETE inhibits Cl channels.
(a) Effect of 100 nm 20-HETE on the 10 pS Cl channels in the basolateral membrane of the medullary thick ascending limb. The top trace shows the time course of experiments and two parts of trace indicated by numbers are extended to show the fast time resolution. The holding potential is −60 mV and the channel closed level is indicated by a dotted line and ‘C’. The experiments were performed in a cell-attached patch. (b) The bar graph summarizes the effect of 20-HETE (100 nm) on the10 pS Cl channel in the absence of or in the presence of 17-ODYA (5 μm), respectively. Asterisk indicates a significant difference in comparison with control value (P<0.05). 20-HETE, 20-hydroxyeicosatraenoic acid.
Discussion
Molecular cloning has identified three types of Cl channels, CFTR, ClC-K1, and ClC-K2, which are expressed in the renal tubules including TAL.5,6 However, the observation that Cl transport in the TAL is not altered in cystic fibrosis mice14 suggests that CFTR may not have an important function in maintaining Cl transport in the TAL. The notion that Cl channels other than CFTR are responsible for the basolateral Cl conductance is also supported by the patch-clamp experiments in which two types of Cl channels, the small conductance (8–10 pS) and the intermediate conductance (20–40 pS), are detected in the basolateral membrane of the TAL.8 However, a large-conductance (80–90 pS) Cl channel was also detected when vesicles isolated from the rabbit mTAL were incorporated into lipid bilayer.31 But ours and other studies failed to detect the large-conductance Cl channels in both rat and mouse TAL.8 Although two types of Cl channels are expressed in the TAL, the previous studies and the present investigation have suggested that the small-conductance Cl channels should be mainly responsible for Cl exit across the basolateral membrane. Guinamard et al.8 have reported that the small-conductance Cl channels were found in 43–57% patches in non-forskolin-treated TAL and 82–87% in forskolin-treated tubules. This study conformed their finding and we observed the small-conductance Cl channels in approximately 30% patches of non-forskolin-treated TAL. Moreover, similar to the observation by Guinamard et al., the possibility of finding the intermediate-conductance Cl channel is lower (4%) than that of the small-conductance Cl channels. Thus, it is safe to conclude that the 10 pS Cl channels should be mainly responsible for the Cl exit across the basolateral membrane. Because 10 pS Cl channel shares some biophysical properties of ClC-K2, it may be a pore-forming component of the 10 pS Cl channel and mainly responsible for the basolateral Cl conductance.32 This notion is also supported by in situ hybridization and immunostaining showing that ClC-K1 is mainly expressed in the thin ascending limb,11 whereas ClC-K2 which has two isoforms, CLC-K2L and CLC-K2S,10 is predominantly expressed in the basolateral membrane of TAL in the rat kidney.12
The basolateral Cl channels have an important function in the regulation of transepithelial Cl absorption in the TAL because they provide the major pathway for Cl exit. Inhibition of Cl channel activity is expected to increase intracellular Cl concentrations, and a high Cl concentration leads to blocking the Na/K/2Cl cotransporter activity hereby inhibiting the transepithelial Cl absorption.33 The role of basolateral Cl channels in maintaining NaCl absorption in the TAL is best demonstrated by the observation that defective gene products encoding human basolateral Cl channel, ClC-Kb (equal to rat ClC-K2), and Barttin, a subunit of ClC-Kb, cause type III and IV Bartter's syndrome, respectively.5 Thus, the regulation of basolateral Cl channels is an important component for modulating epithelial transport in the TAL.
A large body of evidence suggests that COX-dependent and CYP-ω-hydroxylation-dependent AA metabolism have an important function in regulating Na and Cl transport in the TAL.24,34–37 Both COX and CYP monooxygenase are present in the mTAL.22,38 Previous studies showed that the COX-dependent AA metabolites, such as prostaglandin E2 (PGE2), inhibited the stimulatory effect of vasopressin on Cl reabsorption39 or Na/K/2Cl cotransporter.40 We have previously demonstrated that PGE2 at low concentrations (<1 μm) abolished the vasopressin-induced increase in the apical 70-pS K channel activity, whereas PGE2 at high concentrations inhibited the 70-pS K channels by a protein kinase C-dependent mechanism.41 Although COX-dependent AA metabolites have been reported to inhibit Cl reabsorption in the mTAL,36,37 it is unlikely that the inhibitory effect of AA on the Cl channels was induced by a COX-dependent metabolite such as PGE2. This notion is supported by the observation that indomethacin failed to abolish the AA-induced inhibition of channel activity. We speculate that PGE2 inhibiting Cl transport in the mTAL is generated by renal tissue other than mTAL or that AA applied to the mTAL is preferably metabolized by CYP monooxygenase. COX inhibitors such as indomethacin have been used to treat patients with Bartter's disease in clinical practice. However, the beneficiary effect of indomethacin on Bartter's disease may be partially due to changing renal hemodynamics, glomerular filtration rate, and renin release.42
In addition to COX, CYP-ω-hydroxylation-dependent AA metabolites have an important function in the regulation of membrane transport in the TAL: 20-HETE has been shown to inhibit Na/K/2Cl cotransporter,16 apical 70 pS K channels, and basolateral K channels.20 Two lines of evidence have indicated that CYP-ω-hydroxylation of AA inhibits the 10 pS Cl channel in the basolateral membrane of the mTAL: first, inhibition of CYP-ω-hydroxylation of AA metabolism abolished the effect of AA on channel activity. Second, addition of 20-HETE mimics the effect of AA and inhibits the 10 pS Cl channel even in the presence of 17-ODYA. In addition to CYP-ω-hydroxylation, AA may also be metabolized by CYP epoxygenase which is expressed in the mTAL.43 However, the role of CYP epoxygenase-dependent AA metabolism in regulating the epithelial transport in the TAL is not explored. Considering that CYP epoxygenase activity is upregulated by high Na intake and high K diet,44,45 it is possible that CYP epoxygenase-dependent AA metabolites should also inhibit the basolateral Cl channels. However, it is possible that CYP epoxygenase and CYP-ω-hydroxylase may function independently under different physiological conditions.
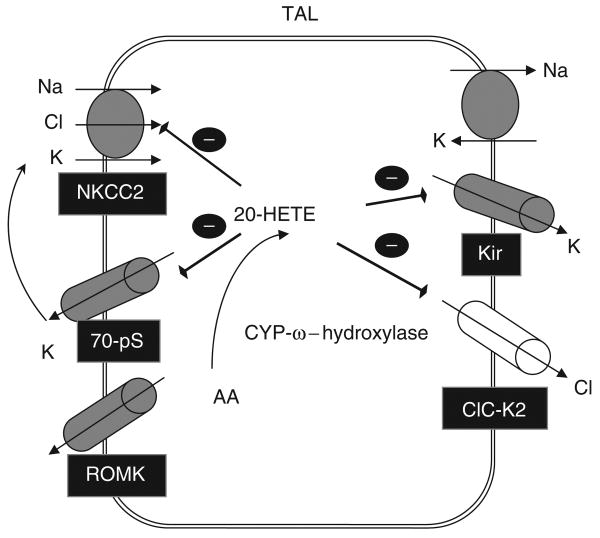
Figure 7 is a cell model illustrating the cellular mechanism by which 20-HETE inhibits NaCl transport in the TAL. Increasing 20-HETE production in the apical membrane inhibits the apical K channels and K recycling,18 which has an important function for maintaining Na/K/2Cl cotransporter function.46 In contrast, high 20-HETE level inhibits both the Cl channels and K channels in the basolateral membrane,21 leading to suppressing Cl exit across the basolateral membrane and indirectly inhibiting the apical Na/K/2Cl cotransporter function through elevated intracellular Cl concentrations.33 In addition, CYP-ω-hydroxylation-dependent metabolites of AA such as 20-HETE have been shown to inhibit the basolateral Na-K-ATPase.17 Thus, the coordinated effect of 20-HETE on apical K channels, Na/K/2Cl cotransporter, basolateral Cl, K channels, and Na-K-ATPase can effectively block NaCl transport in the TAL in response to high salt intake. The notion that 20-HETE may have a function in regulating salt-sensitive hypertension has been suggested by the report that high salt intake stimulates 20-HETE generation and that defective regulation of 20-HETE generation results in salt-sensitive hypertension.47 Therefore, the 20-HETE-mediated inhibition of the Cl channels in the mTAL is an integrated component of the antihypertensive effect of 20-HETE in response to a high salt intake.
Figure 7. A cell scheme illustrates the effect of 20-HETE on the apical Na/K/2Cl cotransporter, apical K channels, and basolateral K and Cl channels.
NKCC2 (Na/K/2Cl cotransporter II), Kir (inwardly-rectifying K channel), ClC-K2 (Cl channel-K2), ROMK (Kir1.1). ROMK, renal outer medullary potassium channel.
The physiological upstream signaling by which 20-HETE serves as a mediator for regulating the basolateral Cl channels is not clear. Stimulation of the Ca2+-sensing receptor has been shown to increase 20-HETE production.48 Because the Ca2+-sensing receptor is located in the basolateral membrane of the mTAL, it is conceivable that the stimulation of the Ca2+-sensing receptor may also inhibit the basolateral 10 pS Cl channel. Further experiments are required to test this possibility. We conclude that two types of Cl channels are expressed in the basolateral membrane of the TAL and that 20-HETE mediates the inhibitory effect of AA on Cl channels.
Materials and Methods
Preparation of the TAL
Pathogen-free Sprague-Dawley rats of either sex (<90 g) were purchased from the animal facility of the Second Affiliated Hospital of Harbin Medical University (Harbin, China). The animals were fed a normal rat chow and had free access to water before experiments. Rats were killed by cervical dislocation and the kidneys were removed immediately. We followed the methods described by Guinamard et al.8 to prepare TAL for the patch-clamp studies. Briefly, the kidney was cut into 1mm thick slices with a razor blade and the kidney slices were incubated in an HEPES buffer solution containing collagenase type 1A (1 mg/ml; Sigma, St Louis, MO, USA) at 37°C for 45 min. After the collagenase treatment, the kidney slices were rinsed with a HEPES-buffered solution containing (in mmol): 140 NaCl, 5 KCl, 1.8 MgCl2, 1.8 CaCl2, and 10 HEPES (pH=7.4) at 4°C. Three types of nephron segments: thin descending limb, TAL, and medullar collecting duct, can be dissected in the renal medulla. However, mTAL is very easy to recognize because the size of the mTAL is larger than that of thin descending limb but smaller than that of collecting duct. Moreover, we have carried out a large amount of experiments studying the apical ROMK and 70 pS K channels, which is expressed uniquely in the TAL.49 Thus, we are very familiar with the morphological characteristic of the mTAL. The single TAL was dissected and the isolated tubule was transferred onto a 5×5-mm cover glass-coated with polylysine (Sigma) to immobilize the tubule. The cover glass was placed in a chamber mounted on an inverted microscope (Nikon, Melville, NY, USA). The TAL was superfused with HEPES-buffered NaCl solution plus 5 mmol glucose. We selected cells with a clear border, an indication of no cell swelling, for patch-clamp experiments. The animal protocol was approved by the animal care and use committee of Harbin Medical University.
Patch-clamp technique
The patch pipette was pulled with Narishige PP-830 (Tokyo, Japan) electrode-puller and the pipette solution contained (in mmol): 140 NaCl, 1.8 MgCl2, and 10 HEPES (pH=7.4). We used an Axon 200B patch-clamp amplifier (Burlingame, CA, USA) to record channel current. The current was low-pass filtered at 0.2–0.5 kHz and digitized by an Axon interface (Digidata 1320, Burlingame, CA, USA). Data were collected by an IBM-compatible Pentium computer at a rate of 2 kHz and analyzed by using pClamp software system 9.2 (Axon Instruments, Burlingame, CA, USA). Channel activities were defined as NPo, a product of channel open probability (Po) and channel number (n). The NPo was calculated from data samples of 30–60 s duration in the steady state as follows:
in which ti is the fractional open time spent at each of the observed current levels. NPo was calculated from data samples of 30–60 s duration. Specifically, we used an all-point histogram (pClamp 9.2) to obtain the channel activity. The slope conductance of the channel was determined by the measurement of the Cl current at several holding potentials.
Chemicals and experimental solution
The bath solution was composed of (in mmol) 140 NaCl, 5 KCl, 1.8 CaCl2, 1.8 MgCl2, and 10 HEPES (pH=7.4). AA and lenoleic acid were purchased from Nu-Chek Prep (Elysian, MN, USA). Whereas NDGA, indomethacin and 17-ODYA were obtained from Sigma. The above chemicals were dissolved in 100% ethanol and the final concentrations of ethanol in the bath were less than 0.1%, which had no significant effect on channel activity.
Statistics
Data are shown as means±s.e.m. We used paired Student's t-tests to determine the significance of the difference between the control and experimental periods. Statistical significance was taken as P<0.05.
Acknowledgments
This work was supported by National Nature Science Foundation of China no. 30570673 (R-MG), no. 30770800 (R-MG), the Research Fund of High Education for Doctoral Program no. 20040226 (R-MG), Youth Science Foundation of Medical Basic Subjects of Harbin Medical University, no. 060035 (LY) and NIH HL34300 (W-HW).
Footnotes
DISCLOSURE: All the authors declared no competing interests.
References
- 1.Giebisch G. Renal potassium transport: mechanisms and regulation. Am J Physiol Renal Physiol. 1998;274:F817–F833. doi: 10.1152/ajprenal.1998.274.5.F817. [DOI] [PubMed] [Google Scholar]
- 2.Greger R. Ion transport mechanisms in thick ascending limb of Henle's loop of mammalian nephron. Physiol Rev. 1985;65:760–797. doi: 10.1152/physrev.1985.65.3.760. [DOI] [PubMed] [Google Scholar]
- 3.Hebert SC, Andreoli TE. Control of NaCl transport in the thick ascending limb. Am J Physiol (Renal Fluid Electrolyte Physiol) 1984;246:F745–F756. doi: 10.1152/ajprenal.1984.246.6.F745. [DOI] [PubMed] [Google Scholar]
- 4.Gamba G, Miyanoshita A, Lombardi M, et al. Molecular cloning, primary structure and characterization of two members of the mammalian electroneutral sodium-(potassium)-chloride cotransporter family expressed in kidney. J Biol Chem. 1994;269:17713–17722. [PubMed] [Google Scholar]
- 5.Kramer BK, Bergler T, Stoelcker B, et al. Mechanisms of Disease: the kidney-specific chloride channels ClCKA and ClCKB, the Barttin subunit, and their clinical relevance. Nat Clin Pract Neph. 2008;4:38–46. doi: 10.1038/ncpneph0689. [DOI] [PubMed] [Google Scholar]
- 6.van Kuijck MA, van Aubel RA, Busch AE, et al. Molecular cloning and expression of a cyclic AMP-activated chloride conductance regulator: a novel ATP-binding cassette transporter. Proc Natl Acad Sci USA. 1996;93:5401–5406. doi: 10.1073/pnas.93.11.5401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Di Stefano A, Jounier S, Wittner M. Evidence supporting a role for KCl cotransporter in the thick ascending limb of Henle's loop. Kidney Int. 2001;60:1809–1823. doi: 10.1046/j.1523-1755.2001.00994.x. [DOI] [PubMed] [Google Scholar]
- 8.Guinamard R, Chraibi A, Teulon J. A small-conductance Cl− channel in the mouse thick ascending limb that is activated by ATP and protein kinase A. J Physiol. 1995;485:97–112. doi: 10.1113/jphysiol.1995.sp020715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kieferle S, Fong P, Bens M, et al. Two highly homologous members of the ClC chloride channel family in both rat and human kidney. Proc Natl Acad Sci USA. 1994;91:6943–6947. doi: 10.1073/pnas.91.15.6943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Adachi S, Uchida S, Ito H, et al. Two isoforms of a chloride channel predominantly expressed in thick ascending limb of Henle's loop and collecting ducts of rat kidney. J Biol Chem. 1994;269:17677–17683. [PubMed] [Google Scholar]
- 11.Uchida S, Sasaki S, Nitta K, et al. Localization and functional characterization of rat kidney-specific chloride channel, ClC-K1. J Clin Invest. 1995;95:104–113. doi: 10.1172/JCI117626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Winters CJ, Zimniak L, Reeves WB, et al. Cl- channels in basolateral renal medullary membranes XII Anti-rbClC-Ka antibody blocks MTAL Cl− channels. AJP -Renal Physiol. 1997;273:F1030–F1038. doi: 10.1152/ajprenal.1997.273.6.F1030. [DOI] [PubMed] [Google Scholar]
- 13.Sheppard DN, Welsh MJ. Structure and function of the CFTR chloride channel. Physiol Rev. 1999;79:S23–S45. doi: 10.1152/physrev.1999.79.1.S23. [DOI] [PubMed] [Google Scholar]
- 14.Marvao P, Jesus Ferreira MC, Bailly C, et al. Cl− absorption across the thick ascending limb is not altered in cystic fibrosis mice A role for a pseudo-CFTR Cl− channel. J Clin Invest. 1998;102:1986–1993. doi: 10.1172/JCI4074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guinamard R, Paulais M, Teulon J. Inhibition of a small-conductance cAMP-dependent Cl– channel in the mouse thick ascending limb at low internal pH. J Physiol. 1996;490:759–765. doi: 10.1113/jphysiol.1996.sp021183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Escalante B, Erlij D, Falck JR, et al. Effect of cytochrome P450 arachidonate metabolites on ion transport in rabbit kidney loop of Henle. Science. 1991;251:799–802. doi: 10.1126/science.1846705. [DOI] [PubMed] [Google Scholar]
- 17.Schwartzman M, Ferreri NR, Carroll MA, et al. Renal cytochrome P450-related arachidonate metabolite inhibits (Na++K+)ATPase. Nature. 1985;314:620–622. doi: 10.1038/314620a0. [DOI] [PubMed] [Google Scholar]
- 18.Wang WH, Lu M, Hebert SC. Cytochrome P-450 metabolites mediate extracellular Ca2+-induced inhibition of apical K+ channels in the TAL. Am J Physiol. 1996;271:C103–C111. doi: 10.1152/ajpcell.1996.271.1.C103. [DOI] [PubMed] [Google Scholar]
- 19.Escalante BA, Ferreri NR, Dunn CE, et al. Cytokines affect ion transport in primary cultured thick ascending limb of Henle's loop cells. Am J Physiol. 1994;266:C1568–C1576. doi: 10.1152/ajpcell.1994.266.6.C1568. [DOI] [PubMed] [Google Scholar]
- 20.Wang W, Lu M. Effect of arachidonic acid on activity of the apical K+ channel in the thick ascending limb of the rat kidney. J Gen Physiol. 1995;106:727–743. doi: 10.1085/jgp.106.4.727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gu RM, Wang WH. Arachidonic acid inhibits K channels in basolateral membrane of the thick ascending limb. Am J Physiol Renal Physiol. 2002;283:F407–F414. doi: 10.1152/ajprenal.00002.2002. [DOI] [PubMed] [Google Scholar]
- 22.McGiff JC. Cytochrome P-450 metabolism of arachidonic acid. Annu Rev Pharmacol Toxicol. 1991;31:339–369. doi: 10.1146/annurev.pa.31.040191.002011. [DOI] [PubMed] [Google Scholar]
- 23.Jim K, Hassid A, Sun F, et al. Lipoxygenase activity in rat kidney glomeruli, glomerular epithelial cells, and cortical tubules. J Biol Chem. 1982;257:10294–10299. [PubMed] [Google Scholar]
- 24.Ferreri NR, McGiff JC, Vio CP, et al. TNFα regulates renal COX-2 in the rat thick ascending limb (TAL) Thromb Res. 2003;110:277–280. doi: 10.1016/s0049-3848(03)00381-5. [DOI] [PubMed] [Google Scholar]
- 25.Ito O, Alonso-Galicia M, Hopp KA, et al. Localization of cytochrome P-450 4A isoforms along the rat nephron. Am J Physiol (Renal Physiol) 1998;274:F395–F404. doi: 10.1152/ajprenal.1998.274.2.F395. [DOI] [PubMed] [Google Scholar]
- 26.Perico N, Cornejo RP, Benigni A, et al. Endothelin induces diuresis and natriuresis in the rat by acting on proximal tubular cells through a mechanism mediated by lipoxygenase products. J Am Soc Nephrol. 1991;2:57–69. doi: 10.1681/ASN.V2157. [DOI] [PubMed] [Google Scholar]
- 27.Gonzalez-Nunez D, Sole M, Natarajan R, et al. 12-Lipoxygenase metabolism in mouse distal convoluted tubule cells. Kidney Int. 2005;67:178–186. doi: 10.1111/j.1523-1755.2005.00068.x. [DOI] [PubMed] [Google Scholar]
- 28.Salari H, Braquet P, Borgeat P. Comparative effects of indomethacin, acetylenic acids, 15-HETE, nordihydroguaiaretic acid and BW755C on the metabolism of arachidonic acid in human leukocytes and platelets. Prostaglandins Leukot Med. 1984;13:53–60. doi: 10.1016/0262-1746(84)90102-1. [DOI] [PubMed] [Google Scholar]
- 29.Arteaga S, Andrade-Cetto A, Cárdenas R. Larrea tridentata (Creosote bush), an abundant plant of Mexican and US-American deserts and its metabolite nordihydroguaiaretic acid. J Ethnopharmacol. 2005;98:231–239. doi: 10.1016/j.jep.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 30.Roman RJ. P450 metabolites of arachidonic acid in the control of cardiovascular function. Physiol Rev. 2002;82:131–185. doi: 10.1152/physrev.00021.2001. [DOI] [PubMed] [Google Scholar]
- 31.Reeves WB, Andreoli TE. Cl− transport in basolateral renal medullary vesicles: II. Cl− channels in planar lipid bilayers. J Membr Biol. 1990;113:57–65. doi: 10.1007/BF01869606. [DOI] [PubMed] [Google Scholar]
- 32.Nissant A, Paulais M, Lachheb S, et al. Similar chloride channels in the connecting tubule and cortical collecting duct of the mouse kidney. AJP - Renal Physiol. 2006;290:F1421–F1429. doi: 10.1152/ajprenal.00274.2005. [DOI] [PubMed] [Google Scholar]
- 33.Russell JM. Sodium-potassium-chloride cotransport. Physiol Rev. 2000;80:211–276. doi: 10.1152/physrev.2000.80.1.211. [DOI] [PubMed] [Google Scholar]
- 34.McGiff JC, Quilley J. 20-HETE and the kidney: resolution of old problems and new beginnings. Am J Physiol. 1999;277:R607–R623. doi: 10.1152/ajpregu.1999.277.3.R607. [DOI] [PubMed] [Google Scholar]
- 35.Ye WL, Zhang H, Hillas E, et al. Expression and function of COX isoforms in renal medulla: evidence for regulation of salt sensitivity and blood pressure. A J Physiol Renal Physiol. 2006;290:F542–F549. doi: 10.1152/ajprenal.00232.2005. [DOI] [PubMed] [Google Scholar]
- 36.Stokes JB. Effect of prostaglandine on chloride transport across the rabbit thick ascending limb of Henle. J Clin Invest. 1979;64:495–502. doi: 10.1172/JCI109487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Peterson LN, McKay AJ, Borzecki JS. Endogenous prostaglandin E2 mediates inhibition of rat thick ascending limb Cl reabsorption in chronic hypercalcemia. J Clin Invest. 1993;91:2399–2407. doi: 10.1172/JCI116473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ferreri NR, An SJ, McGiff JC. Cyclooxygenase-2 expression and function in the medullary thick ascending limb. Am J Physiol. 1999;277:F360–F368. doi: 10.1152/ajprenal.1999.277.3.F360. [DOI] [PubMed] [Google Scholar]
- 39.Culpepper RM, Andreoli TE. Interactions among prostaglandin E2, antidiuretic hormone, and cyclic adenosine monophosphate in modulating Cl− absorption in single mouse medullary thick ascending limbs of Henle. J Clin Invest. 1983;71:1588–1601. doi: 10.1172/JCI110915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kaji DM, Chase HS, Jr, Eng JP, et al. Prostaglandin E2 inhibits Na-K-2Cl cotransport in medullary thick ascending limb cells. Am J Physiol. 1996;271:F354–F361. doi: 10.1152/ajpcell.1996.271.1.C354. [DOI] [PubMed] [Google Scholar]
- 41.Liu HJ, Wei Y, Ferreri N, et al. Vasopressin and PGE2 regulate the apical 70 pS K channel in the thick ascending limb of rat kidney. Am J Phyiol (Cell Physiol) 2000;278:C905–C913. doi: 10.1152/ajpcell.2000.278.5.C905. [DOI] [PubMed] [Google Scholar]
- 42.Harris RC, Breyer MD. Update on cyclooxygenase-2 inhibitors. Clin J Am Soc Nephrol. 2006;1:236–245. doi: 10.2215/CJN.00890805. [DOI] [PubMed] [Google Scholar]
- 43.Sun P, Lin DH, Wang T, et al. Low Na intake suppresses the expression of CYP2C23 and the arachidonic acid-induced inhibition of ENaC. Am J Physiol Renal Physiol. 2006;291:1192–1200. doi: 10.1152/ajprenal.00112.2006. [DOI] [PubMed] [Google Scholar]
- 44.Sun P, Liu W, Lin DH, et al. Epoxyeicosatrienoic acid (EET) activates the Ca2+-dependent bid conductance K channel in the cortical collecting duct. J Am Soc Nephrol. 2008;20:523. doi: 10.1681/ASN.2008040427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nakagawa K, Holla VR, Wei Y, et al. Salt sensitive hypertension is associated with a dysfunctional Cyp4a10 gene and kidney epithelial sodium channel. J Clin Invest. 2006;116:1696–1702. doi: 10.1172/JCI27546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Simon DB, Karet FE, Rodriguez-Soriano J, et al. Genetic heterogeneity of Bartter's syndrome revealed by mutations in the K+ channel, ROMK. Nat Genet. 1996;14:152–156. doi: 10.1038/ng1096-152. [DOI] [PubMed] [Google Scholar]
- 47.Zhou AP, Drummond HA, Roman RJ. Role of 20-HETE in elevating loop chloride reabsorption in Dahl SS/Jr rats. Hypertension. 1996;27:631–635. doi: 10.1161/01.hyp.27.3.631. [DOI] [PubMed] [Google Scholar]
- 48.Wang WH, Lu M, Hebert SC. Phospholipase A2 is involved in mediating the effect of Ca2+ on the apical K+ channels in rat TAL. Am J Physiol (Renal Physiol) 1997;273:F421–F429. doi: 10.1152/ajprenal.1997.273.3.F421. [DOI] [PubMed] [Google Scholar]
- 49.Gu RM, Wei Y, Jiang HL, et al. K depletion enhances the extracellular Ca2+-induced inhibition of the apical K channels in the mTAL of rat kidney. J Gen Physiol. 2002;119:33–44. doi: 10.1085/jgp.119.1.33. [DOI] [PMC free article] [PubMed] [Google Scholar]