Abstract
Cell-attached patches from isolated epithelial cells from larval bullfrog skin revealed a cation channel that was activated by applying suction (−1 kPa to −4.5 kPa) to the pipette. Activation was characterized by an initial large current spike that rapidly attenuated to a stable value and showed a variable pattern of opening and closing with continuing suction. Current–voltage plots demonstrated linear or inward rectification and single channel conductances of 44–56 pS with NaCl or KCl Ringer's solution as the pipette solution, and a reversal potential (−Vp) of 20–40 mV. The conductance was markedly reduced with N-methyl-D-glucamide (NMDG)-Cl Ringer's solution in the pipette. Neither amiloride nor ATP, which are known to stimulate an apical cation channel in Ussing chamber preparations of larval frog skin, produced channel activation nor did these compounds affect the response to suction. Stretch activation was not affected by varying the pipette concentrations of Ca2+ between 0 mmol l−1 and 4 mmol l−1 or by varying pH between 6.8 and 8.0. However, conductance was reduced with 4 mmol l−1 Ca2+. Western blot analysis of membrane homogenates from larval bullfrog and larval toad skin identified proteins that were immunoreactive with mammalian TRPC1 and TRPC5 (TRPC, canonical transient receptor potential channel) antibodies while homogenates of skin from newly metamorphosed bullfrogs were positive for TRPC1 and TRPC3/6/7 antibodies. The electrophysiological response of larval bullfrog skin resembles that of a stretch-activated cation channel characterized in Xenopus oocytes and proposed to be TRPC1. These results indicate this channel persists in all life stages of anurans and that TRP isoforms may be important for sensory functions of their skin.
Keywords: amphibian, larva, skin
INTRODUCTION
The skin of larval bullfrogs, Rana (Lithobates) catesbeiana, contributes little to the exchange of solutes with the environment (Alvarado and Moody, 1970). This is in contrast to the skin of adult frogs in which passive loss and regulated active uptake of NaCl take place. Structurally, the larval skin is simpler than that of the adult with an outer layer of apical cells supported on a layer of basal cells with intermediate filament-rich skein cells interspersed between the apical and basal cells (Weed, 1933; Robinson and Heintzelman, 1987). Cox and Alvarado showed that the larval skin mounted in an Ussing-type chamber that minimized edge damage to the apical cell layer had very high resistance and generated a small short-circuit current (Isc) with NaCl Ringer's solution bathing either side of tissue (Cox and Alvarado, 1979). A similar or even greater Isc could be obtained with KCl or other alkali metal cation Ringer's solutions as the outer solution (Cox and Alvarado, 1979), and this current was shown to be carried by a non-selective cation channel that could be activated by amiloride, which is an inhibitor of epithelial sodium channels in the adult skin (Hillyard et al., 1982). Subsequent experiments with the larval skin have shown similar stimulation of non-selective cation conductance by acetylcholine and ATP (Cox, 1992; Cox, 1993; Cox, 1997).
The nature of the larval cation channel or channels remains unclear. Jensik et al. (Jensik et al., 2001) used reverse transcription polymerase chain reaction (RT-PCR) to demonstrate the existence of a purinergic (P2X) receptor while Takada et al. (Takada et al., 2006) have shown that adult-like epithelial Na+ channel (ENaC) subunits exist in the larval skin even though they appear not to be functional. Other evidence suggests the activators are acting on a common channel: (1) stimulation of Isc by amiloride, acetylcholine or ATP exhibits a similar pattern of transient activation with a large current spike that rapidly declines to a level slightly above the control value. (2) The stimulation of Isc becomes refractory following treatment with any of the above compounds, i.e. treatment with a low dose of any of these activators reduces the stimulation by a higher secondary dose of any of the others (Hillyard and Van Driessche, 1989). (3) Stimulation of Isc by any of the activators is much greater in the absence of Ca2+, and calcium channel blockers inhibit the transient activation (Cox, 1992).
The present study was initially designed to obtain cell-attached patch recordings from isolated apical cells and to test the hypothesis that amiloride and ATP activate a common channel. We found that neither amiloride nor ATP in the pipette solutions resulted in channel activity but channel activity could be obtained by applying suction to the pipette, suggesting a mechanosensitive or stretch-activated channel (SAC). These observations were of interest because there is historical evidence for a mechanosensory role for epithelial cells in the larval amphibian skin (Wintrebert, 1905; Wintrebert, 1920; Muntz, 1964). In these experiments early stage larvae, prior to the development of peripheral sensory innervation, reacted with body movements when the skin was stimulated with a thin bristle. By carefully cutting away sections of the skin it was possible to show a path of conduction of sensory information by the epidermis. Roberts recorded from the epidermis of early stage Xenopus larvae with microelectrodes and observed transient all-or-none impulses generated by gently prodding the skin propagated in all directions from the point of stimulation (Roberts, 1969; Roberts, 1971). The mechanosensing element was not identified.
The electrophysiological properties of the SAC recorded in our experiments resembled those of the SAC that Hamill and co-workers have characterized in Xenopus oocytes (reviewed by Hamill and McBride, 1994). Maroto et al. (Maroto et al., 2005) recently suggested the oocyte channel to be a member of the canonical transient receptor potential channel family (TRPC1) and serve a mechanosensory function. This prompted us to examine the hypothesis that members of the TRPC family might exist in larval as well as adult frog skin and whether there might be a broader taxonomic occurrence of these channels. Western blots of homogenates of isolated cells from larval frog and toad skin demonstrated proteins that are immunoreactive with mammalian TRPC1 and also TRPC5 antibodies. TRPC1 continues to be expressed in epidermal cells of frogs following metamorphosis, and TRPC3/6/7 immunoreactivity is detectable in the adult stage while TRPC5 is not.
MATERIALS AND METHODS
All experiments were conducted in compliance with animal welfare regulations pertaining to biological experiments at the University of Copenhagen, Denmark, and the University of Nevada, Las Vegas, NV, USA. Larval bullfrogs (Rana catesbeiana Shaw) were obtained from commercial suppliers, maintained in tap water and fed with plant chips (Vitakraft, Bremen, Germany) ad libitum. Larvae were anesthetized with 0.1% tricaine methane sulfonate and killed by severing the conus arteriosus. The ventral skin was dissected free with the abdominal muscles attached and placed for 5 min in a calcium-free normal Ringer's (NR) solution (113 mmol l−1 NaCl, 3.7 mmol l−1 KCl, 3 mmol l−1 Na acetate, 11 mmol l−1 glucose, 10 mmol l−1 Hepes adjusted to pH 7.4 with NaOH) that also contained 5 mmol l−1 EGTA, which caused the apical cells to separate from the basal cells (Takada, et al., 1995). The cells were gently scraped free and suspended in calcium-free NR solution. The suspended cells were then subjected to centrifugation at 3000 g for 3 min and resuspended in NR solution with 1 mmol l−1 CaCl2. The resuspended cells were placed in small Petri dishes for patch-clamp recording.
The resuspended cells lost their columnar form and appeared spherical so pipette placement was not identified as to being apical or basolateral. Pipettes were filled with NR solution that contained variable Ca2+ concentrations (0–4 mmol l−1), variable pH (6.8–8.0), with KCl Ringer's solution (113 mmol l−1 KCl substituted for NaCl) or with N-methyl-D-glucamide (NMDG) Ringer's solution (113 mmol l−1 NMDG-Cl substituted for NaCl). Seals were made by gentle suction. Once a stable current record had been obtained suction was applied either by mouth or by a syringe and monitored by a pressure gauge (Eirelec Ltd, Dundalk, Ireland). Recordings were made with an Axopatch 200B voltage clamp (Molecular Devices, Foster City, CA, USA). Because reverse potential (Vp) in cell-attached configuration refers to an extracellular potential, it is conventional to depict the current as a function of −Vp and the inward-directed current as negative. Recordings were made with −Vp values between −100 mV and +80 mV, and single channel currents were analyzed with Axon Instuments Clampfit software (Axon Instruments, Inc., Union City, CA, USA). In accordance with common practice currents are displayed against the negative pipette potential (−Vp) to maintain physiological polarity. With this polarity convention inward currents are depicted negative. The potential across the patch membrane (Vm) relates to the general cell membrane potential (Vc) by the equation Vm=−Vp+Vc, i.e. it is displaced by −Vp from the general Vc.
Western blots were made with homogenates of epithelial cells from larval skin and skin from newly metamorphosed bullfrogs. For phylogenetic comparison epithelial cells were obtained from two cohorts of larval toads Bufo woodhouseii. Cells were isolated as described above and homogenized in a buffer that contained 25 mmol l−1 Tris, pH 7.4, 1% Triton X-100, 1% deoxycholate, 0.1% SDS, 137 mmol l−1 NaCl, 1 mmol l−1 sodium vanadate and 1 mmol l−1 phenylmethylsulfonyl fluoride. Western blot analysis of the solubilized proteins was performed using the procedure described by Banes-Bercelli et al. (Banes-Bercelli et al., 2006). Briefly, homogenates were resolved by SDS-PAGE (10%), transferred to nitrocellulose membranes and blocked by 60 min incubation at room temperature (22°C) in TTBS (TBS with 0.05% Tween 20, pH 7.4) plus 5% skimmed milk powder. Nitrocellulose membranes were incubated overnight at 4°C with primary antibodies raised against mammalian TRPC1, TRPC5 and TRPC3/6/7 (SantaCruz Biotechnology, Santa Cruz, CA, USA; anti goat). Subsequently, nitrocellulose membranes were washed twice for 10 min each with TTBS and incubated with rabbit anti-goat IgG horseradish peroxidase conjugate. After extensive washing, bound antibody was visualized on Kodak Biomax film (Sigma-Aldrich, St Louis, MO, USA) using a Pierce Supersignal substrate chemiluminescence detection kit (Rockford, IL, USA).
RESULTS
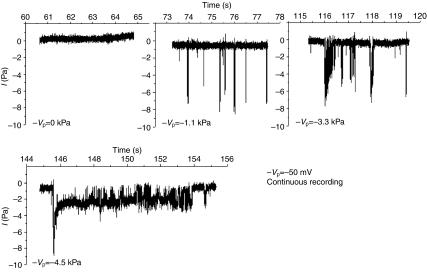
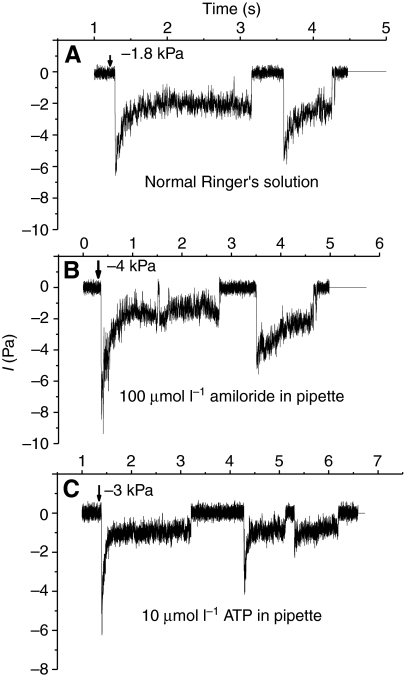
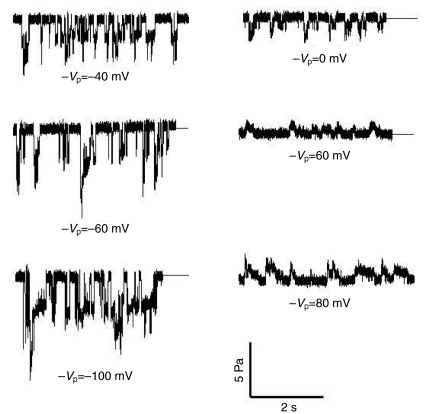
Stable patches with gigaohm resistances typically showed no spontaneous channel activity with NR solution alone or with NR solution containing amiloride (10 μmol l−1 and 100 μmol l−1) or ATP (10–100 μmol l−1) as the pipette solution. The application of suction, typically −1 kPa to −4.5 kPa (1 kPa=7.5 mmHg=10.2 cm H2O), resulted in progressive channel activation that frequently began with a large current that rapidly declined to a fairly stable value (Fig. 1). Our method of applying suction to the pipette did not allow fine resolution relative to the current traces as reported by McBride and Hamill (McBride and Hamill, 1992). Generally, initial activation occurred as soon as the applied suction was noted on the meter. However, there was sometimes a delay of several seconds, and even when pressure was continuously applied there were multiple channel openings and closings. Fig. 2 shows that no difference in the pattern of SAC activation was observed with pipettes filled with NR solution or with NR solution containing amiloride or ATP. Fig. 3 shows the single channel currents measured during continuous stretch activation as −Vp was varied between −100 mV and +80 mV. Note that the channels fluctuated between open and closed states when suction was continuously applied and that not all openings showed the characteristic large and rapidly declining currents.
Fig. 1.
The progressive application of suction to the pipette activated a cation channel with a large initial current that quickly relaxed. Note at lower suction values the initial activation is of a larger current while the relaxation occurs at higher suction pressures where longer openings are observed. −Vp, negative membrane potential.
Fig. 2.
(A) The application of suction to the pipette of cell-attached patches of larval skin epithelial cells stimulated cation channel activity that rapidly adapted after initial activation. Bath and pipette solutions were normal Ringer's solutions with 1 mmol l−1 Ca2+. (B) The presence of amiloride (100 μmol l−1) or (C) ATP (10 μmol l−1) in the pipette solution did not produce channel activity or affect activation by pipette suction.
Fig. 3.
A stepwise progression of pipette potential (−Vp) between −100 mV and +80 mV in a single patch under continuous activation with suction levels between −1.7 kPa and −2.8 kPa.
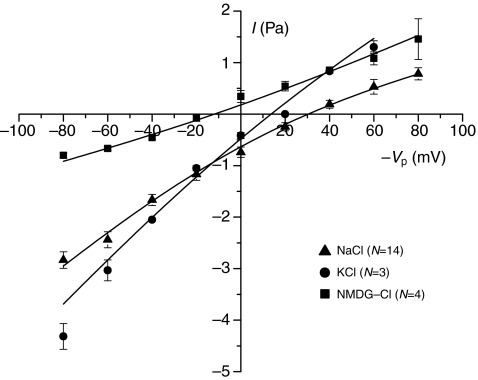
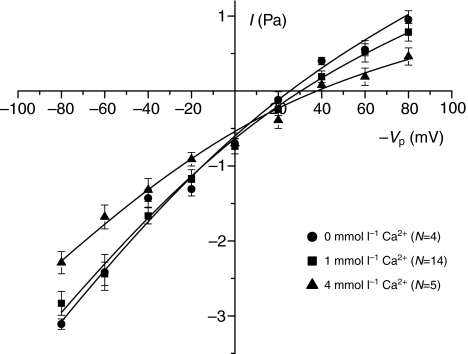
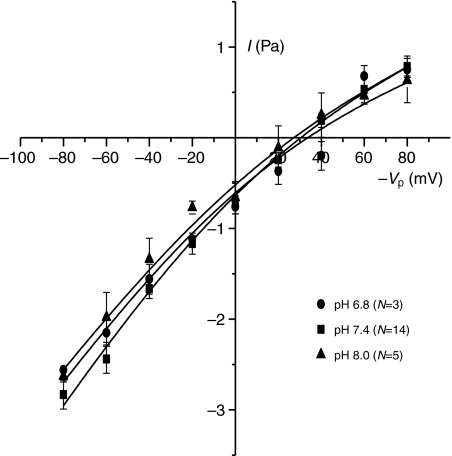
As noted earlier, Vp in cell-attached configuration refers to an extracellular potential, and it is conventional to depict the current as a function of −Vp in order to keep the physiological polarity (Penner, 1995). In the following figures this sign notation is adopted. The current–voltage relationship with either Na+ and K+ as the primary cation showed a similar pattern of slightly inward rectification with conductances of 44–56 pS and 14–24 pS at large negative and positive −Vp values, respectively. The data were fitted with the Goldman–Hodgkin–Katz equation assuming voltage-independent permeabilities. The permeabilities obtained from the best fits were 7.9×10−14 cm3 s−1 and 1.0×10−13 cm3 s−1 for NaCl and KCl, respectively, indicating similar permeabilities for Na+ and K+ (i.e. properties as a non-selective cation channel). The reversal potential was ~30 mV and ~15 mV for NaCl and KCl, respectively (Fig. 4). By contrast, conductance with NMDG as the cation was greatly reduced (permeability obtained from best fit: 2.6×10−14 cm3 s−1) and the reversal potential left-shifted to −20 mV. Specifically, the inward currents at negative −Vp values (where cations will enter the cell) were strongly reduced. Current–voltage plots obtained with variable pipette Ca2+ concentrations showed that with 0 mmol l−1 and 1 mmol l−1 Ca2+ the conductances were practically identical whereas there was a modest reduction in conductance when the Ca2+ in the pipette solution was increased to 4 mmol l−1 (Fig. 5). In terms of permeabilities obtained from the best fits, exposure to 4 mmol l−1 Ca2+ caused a reduction from 7.9–8.3×10−14 cm3 s−1 to 6.1×10−14 cm3 s−1. Variation of extracellular pH in the range 6.8–8.0 (Fig. 6) had no effect on either reversal potential (27–33 mV), conductance or fitted permeability value (6.9–7.2×10−14 cm3 s−1).
Fig. 4.
A current–voltage plot with NaCl or KCl Ringer's solutions as the pipette solutions shows the channel to be relatively non-selective for K+ over Na+. The conductance is much lower with NMDG-Cl as the pipette solution, and the reversal potential shifts to a more negative value on the −Vp (membrane potential) axis. Each point is the mean ± 1 s.e.m. for the number of trials indicated. The data have been fitted with the Goldman–Hodgkin–Katz equation, and calculated permeability values are given in the text. NMDG, N-methyl-D-glucamide.
Fig. 5.
Varying the concentration of Ca2+ in the pipette Ringer's solution from 1 mmol l−1 to 4 mmol l−1 resulted in a modest reduction in conductance at the higher concentration. Permeability values calculated from Goldman–Hodgkin–Katz equation fits are presented in the text. Each point is the mean ± 1 s.e.m. for the number of trials indicated.
Fig. 6.
Varying the pH of the pipette Ringer's solution between 6.8 and 8.0 had little effect on the current–voltage relationship. Each point is the mean ±1 s.e.m. for the number of trials indicated, and data are fitted with the Goldman–Hodgkin–Katz equation.
Western blots identified proteins from the epidermis of larval and adult frogs and also from larval toads that were immunoreactive with mammalian TRPC1 antibody (Fig. 7). Larval frog and toad epidermis also had proteins that were detected by mammalian TRPC5 antibody while proteins from newly metamorphosed frogs were immunoreactive with the TRPC3/6/7 antibody.
Fig. 7.
Western blot analysis of proteins obtained from larval bullfrogs and toads showed similar bands indicating the presence of TRPC1 and TRPC5. TRPC1 was similarly identified in isolated epithelial cells from adult bullfrogs. However, an immunoreactive band was observed with the TRPC3/6/7 antibody rather than with TRPC5. TRPC, canonical transient receptor potential channel.
DISCUSSION
Previous patch-clamp studies with larval skin (Hillyard et al., 2002) characterized an outward rectifying Cl− channel that was spontaneously active and resembled a basolateral Cl− channel identified by noise analysis in Ussing chamber preparations. Cell-attached patches for these experiments were obtained from cells isolated using Ringer's solutions that contained collagenase and trypsin. These cells were fragile and frequently burst when additional pressure was applied to the pipette. By contrast, we found that cells could be isolated with EGTA treatment alone and were more rigid, which allowed the application of suction pressures above those required for obtaining a successful patch. The larger outward rectifying channel rarely appeared in these cells. Attempts to obtain whole-cell patches resulted in loss of the gigaseal so membrane potential could not be measured.
The pattern of stretch activation that we observed in the larval SAC resembles the SAC from Xenopus oocytes (Hamill and McBride, 1992; Hamill and McBride, 1994; McBride and Hamill, 1992) in that: (1) increasing suction produced a progressive increase in channel openings (Fig. 1); (2) the single channel current rapidly adapts (Fig. 2); (3) the single channel conductance is similar (ca. 25–40 pS) with NR solution in the pipette (Fig. 3); (4) the channel is poorly selective for Na+ over K+ (Fig. 4); and (5) the suction required for channel activation is in the same range (ca. from −1 kPa to −4 kPa). Unlike the oocyte channel, amiloride had no inhibitory effect on stretch activation or channel conductance. The 10–100 μmol l−1 amiloride concentrations were selected because activation of the larval cation channel in Ussing chamber preparations occurs in this range and our original goal was to study this property of the channel. The IC50 for amiloride inhibition of the oocyte SAC is approximately 500 μmol l−1 (Lane et al., 1991), which might explain our failure to see an effect. Single channel conductance was modestly reduced by 22–26% at more negative −Vp values with 4 mmol l−1 Ca2+ in the pipette solution. Taglitellii and Toselli found a larger reduction of conductance in cell-attached patches of a SAC in frog (Rana esculenta) oocytes with 2 mmol l−1 Ca2+ vs a Ca2+-free solution in the pipette; however, the Ca2+-free solution also contained 5 mmol l−1 EGTA, and the current–voltage plots were extended to −200 mV (Taglitellii and Toselli, 1988). These authors also were able to demonstrate the oocyte channel conducts Ca2+ with higher (75 mmol l−1) Ca2+ concentration in the pipette. Because our goal was to characterize a cation channel that was not selective for Na+ vs K+, the Ca 2+ conductance was not measured for the larval SAC.
Maroto et al. (Maroto et al., 2005) isolated a protein in a membrane fraction from Xenopus oocytes that showed similar SAC properties when studied in liposomes and identified it immunologically as the canonical transient receptor protein, TRPC1, initially characterized from an oocyte cDNA library by Bobanovic et al. (Bobanovic et al., 1999). The oocyte TRPC1 had approximately 80% homology with mammalian TRPC1s. The identity of TRPC1 in larval and adult amphibian epidermis with mammalian antibodies is thus reasonable evidence for its existence in amphibian epidermal cells and for the possibility that TRPC1 mediates the SAC activity we have recorded. Future studies will be required to see if TRPC channel blockers such as gadolinium and ruthenium red affect the larval SAC.
The role of TRPC1 as a SAC and the physiological role of SACs in general remain unresolved. Maroto et al. (Maroto et al., 2005) reported an increase in mechanosensitive cation channel expression in mammalian (CHO-K1) cultured cells transfected with human (h)TRPC1 and suggested it to be an obligatory component of vertebrate SACs. More recently, Gottleib et al. noted that overexpression of TRPC1 and TRPC6 into Chinese hamster ovary (CHO) and green monkey kidney (COS) cells did not increase the mechanosensitive current, and that expression of TRPC subunits in heterologous systems ‘can be problematic’ (Gottleib et al., 2008). Furthermore, a TRPC1−/− knockout mouse line shows no phenotypic abnormalities (Dietrich et al., 2007).
SACs in oocytes have been suggested to be involved in cell volume regulation (Bryan-Sisneros et al., 2003) and may be important in development (Hamill and McBride, 1995). However, Wilkinson et al. found that concentrations of SAC blockers (gadolinium, amiloride) that inhibit channel activity in membrane patch recordings did not adversely affect Xenopus development from oogenesis to fertilization to the free-swimming larval stage (Wilkinson et al., 1998). Such a channel could provide sensory information to early stage larvae regarding their interaction with objects in their natural environment or perhaps act as a sensor for the oocyte relative to its placement relative to substrates or other oocytes. This latter function would not necessarily affect survival in a controlled laboratory environment but could conceivably be important in natural situations.
The selectivity properties of the larval SAC are similar to that of the non-selective cation channel that Hillyard et al. (Hillyard et al., 1982) characterized in the apical membrane of isolated larval skin in Ussing chamber preparations, and it is tempting to draw parallels between the two. However, the SAC in membrane patches did not respond to either ATP or amiloride and, because the cells lost their polarity when separated, the SAC could not be localized to the apical membrane. It is possible that the loss of polarity could dissociate the SAC from putative interactions with activators associated with ATP or amiloride receptors. Maroto observed TRPC1 that was isolated from oocytes remained stretch-activated in liposomes, indicating that SAC properties of the oocyte channel are independent of cytosolic or associated membrane proteins (Maroto, 2005). This could give a SAC with no effect of ATP or amiloride. Also, the frequency of patches with SAC activity was low, suggesting the channels are widely dispersed in the isolated cells.
It is of interest that TRPC5 is co-expressed with TRPC1 in the skin of larval frogs and toads. Gomis et al. (Gomis et al., 2008) found that whole-cell currents obtained from human embryonic kidney (HEK) cells transfected with mammalian TRPC5 showed stretch activation associated with increased cytosolic Ca2+, in contrast to the inhibition of single channel currents by Ca2+ in the pipette that was observed in frog oocytes. TRPC5 was also demonstrated by RT-PCR and immunohistochemistry to occur in mechanosensory neurons, indicating a mechanosensory function for the channel. Stretch activation of TRPC5 required delays of seconds after the application of pressure, and the authors suggested possible coupling between the channel and force-sensing mechanism (Gomis et al., 2008). In this regard, TRPC1 and TRPC5 are known to form hetero-oligomeric channels in the mammalian brain (Strubing et al., 2001), and when expressed in HEK cells, TRPC5 could be stimulated by carbachol while the TRPC1–C5 heteromer could be stimulated by ATP and was poorly selective for Na+ over Cs+, both of which are properties of the larval cation channel. It should be noted, however, that the single channel conductance of the TRPC1–C5 heteromer was reduced to 5 pS, which is much smaller that we recorded for the SAC in larval epidermal cells. We cannot at this time determine if the two form hetero-oligomeric channels, co-immunoprecipitation would be necessary but is beyond the scope of the present study. TRPC1 and TRPC3/6/7 are both detectable in the adult skin. Liu et al. have demonstrated that TRPC1 and TRPC3 co-assemble to form a heteromer that functions as a store-operated cation channel in human parotid gland (HSY) epithelial cells (Liu et al., 2005). As with TRPC1 and TRPC5 it is beyond the scope of the present study to determine the heteromeric structure of TRPC1 and TRP3/6/7 isoforms.
Van Driessche and Zeiske characterized a non-selective cation channel in isolated skin of adult frogs (Van Driessche and Zeiske, 1985), and Nilius has suggested that calcium impermeable, monovalent, cation channels might be TRP channel isoforms (Nilius, 2003). The identification of TRP channels in mammalian keratinocytes has generated hypotheses that they may serve sensory functions, which include mechanoreception and thermoreception (Lumpkin and Caterina, 2007). Amphibian skin serves as a respiratory, ionic and osmoregulatory surface in addition to its thermal and mechanosensory function. Characterization of TRP channel isoforms in amphibian skin will improve our understanding of how these animals perceive their environment.
ACKNOWLEDGEMENTS
The investigators were supported in part by the following research grants: NSF IOB 9986008 (S.D.H.), Danish Natural Research Foundation (N.J.W.), NIH HL-58135 and DK-5026A (M.B.M.). Deposited in PMC for release after 12 months.
Footnotes
- EnaC
- epithelial Na+ channel
- Isc
- short-circuit current
- NMDG
- N-methyl-D-glucamide
- NR solution
- normal Ringer's solution
- SAC
- stretch-activated channel
- TRPC
- canonical transient receptor potential channel
- Vc
- cell membrane potential
- Vm
- patch membrane potential
- Vp
- pipette potential
REFERENCES
- Alvarado R. A., Moody A. (1970). Sodium and chloride transport in tadpoles of the bullfrog, Rana catesbieana. Am. J. Physiol. 218, 1510-1516 [DOI] [PubMed] [Google Scholar]
- Banes-Berceli A. K., Shaw S., Ma G., Brands M., Eaton D. C., Stern D. M., Fulton D., Caldwell R. W., Marrero M. B. (2006). Effect of simvastatin on high glucose- and angiotensin II-induced activation of the JAK/STAT pathway in mesangial cells. Am J. Physiol. Renal Physiol. 291, F116-F121 [DOI] [PubMed] [Google Scholar]
- Bobanovic L. K., Laine M., Petersen C. C., Bennett D. L., Berridge M. J., Lipp P., Ripley S. J., Bootman M. D. (1999). Molecular cloning and immunolocalization of a novel vertebrate trp homologue from Xenopus. Biochem. J. 340, 593-599 [PMC free article] [PubMed] [Google Scholar]
- Bryan-Sisneros A. A., Fraser S. P., Djamgoz M. B. A. (2003). Electrophysiological, mechanosensitive responses of Xenopus laevis oocytes to direct, isotonic increase in intracellular volume. J. Neurosci. Methods 125, 103-111 [DOI] [PubMed] [Google Scholar]
- Cox T. C. (1992). Calcium channel blockers inhibit amiloride-stimulated short-circuit current in frog tadpole skin. Am. J. Physiol. 263, R827-R833 [DOI] [PubMed] [Google Scholar]
- Cox T. C. (1993). Low affinity mixed acetylcholine-reponsive receptors at the apical membrane of frog tadpole skin. Am. J. Physiol. 265, R552-R558 [DOI] [PubMed] [Google Scholar]
- Cox T. C. (1997). Apical regulation of nonselective cation channel by ATP in larval bullfrog skin. J. Exp. Zool. 279, 220-227 [PubMed] [Google Scholar]
- Cox T. C., Alvarado R. A. (1979). Electrical and transport characteristics of skin of larval Rana catesbeiana. Am. J. Physiol. 237, R74-R79 [DOI] [PubMed] [Google Scholar]
- Dietrich A., Kalwa H., Storch U., Mederos Y., Schnitzer M., Salinova B., Pinkenburg O., Dubrovska G., Essin K., Gollasch M., et al. (2007). Pressure-induced and store-operated cation influx in vascular smooth muscle cells is independent of TRPC1. Pflugers Arch. 455, 465-477 [DOI] [PubMed] [Google Scholar]
- Gomis A., Soriano S., Belmonte C., Viana F. (2008). Hypoosmotic and pressure-induced membrane stretch activate TRPC5 channels. J. Physiol. 586, 5633-5649 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gottleib P., Folgering J., Maroto R., Raso A., Wood T. G., Kurosky A., Bowman C., Bichet D., Patel A., Sachs F., et al. (2008). Revisiting TRPC1 and TRPC6 mechanosensitivity. Pflugers Arch. 455, 1097-1103 [DOI] [PubMed] [Google Scholar]
- Hamill O. P., McBride D. W., Jr (1992). Rapid adaptation of the mechanosensitive channel in Xenopus oocytes. Proc. Natl. Acad. Sci. USA 89, 7462-7466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamill O. P., McBride D. W., Jr (1994). Molecular mechanisms of mechanoreceptor adaptation. New Physiol. Sci. 9, 53-69 [Google Scholar]
- Hamill O. P., McBride D. W., Jr (1995). Mechanoreceptive membrane ion channels. Am. Sci. 83, 30-37 [Google Scholar]
- Hillyard S. D., Van Driessche W. (1989). Effect of amiloride on the poorly selective cation channel of larval bullfrog skin. Am. J. Physiol. 256, C168-C174 [DOI] [PubMed] [Google Scholar]
- Hillyard S. D., Zeiske W., Van Driessche W. (1982). A fluctuation analysis study of the development of amiloride-sensitive Na+ transport in the skin of larval bullfrogs (Rana catesbeiana). Biochim. Biophys. Acta 692, 445-461 [DOI] [PubMed] [Google Scholar]
- Hillyard S. D., Rios K., Larsen E. H. (2002). Basolateral Cl− channels in the larval bullfrog skin epithelium. J. Comp. Physiol. B. 172, 577-586 [DOI] [PubMed] [Google Scholar]
- Jensik P. J., Holbird D., Collard M. W., Cox T. C. (2001). Cloning and characterization of a functional P2X receptor from larval bullfrog skin. Am. J. Physiol. 281, C954-C962 [DOI] [PubMed] [Google Scholar]
- Lane J. W., McBride D. W., Jr, Hamill O. P. (1991). Amiloride block of the mechanosensitive cation channel in Xenopus oocytes. J. Physiol. 44, 347-366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X., Bandyopadhyay B. C., Singh B. B., Groschener K., Ambudkar I. S. (2005). Molecular analysis of a store operated and 2-acetyl-sn-glycerol-sensitive non selective cation channel. J. Biol. Chem. 280, 21600-21606 [DOI] [PubMed] [Google Scholar]
- Lumpkin E. A., Caterina M. J. (2007). Mechanisms of sensory transduction in the skin. Nature 445, 858-865 [DOI] [PubMed] [Google Scholar]
- Maroto R., Raso A., Wood T. G., Kurosky A., Martinac B., Hamill O. P. (2005). TRPC1 forms the stretch-activated cation channel in vertebrate cells. Nature Cell Biol. 7, 1443-1446 [DOI] [PubMed] [Google Scholar]
- McBride D. W., Jr, Hamill O. P. (1992). Pressure clamp: a method for rapid step perturbation of mechanosensitive channels. Pflugers Arch. 421, 606-612 [DOI] [PubMed] [Google Scholar]
- Muntz L. (1964). Neuromuscular foundations of behavior in embryonic and larval stages of the auran, Xenopus laevis PhD Dissertation University of Bristol, UK: [Google Scholar]
- Nilius B. (2003). Calcium-impermeable monovalent cation channels: a TRP connection? British. J. Pharmacol. 138, 5-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penner R. (1995). A practical guide to patch clamping. In Single-Channel Recording, 2nd edition (ed. Sakmann B., Neher E.), pp. 3-30 New York: Plenum Press; [Google Scholar]
- Roberts A. (1969). Conducted impulses in the skin of young tadpoles. Nature 222, 1265-1266 [DOI] [PubMed] [Google Scholar]
- Roberts A. (1971). The role of propagated impulses in the sensory system of young tadpoles. Z. vergl. Physiol. 75, 388-401 [Google Scholar]
- Robinson D. H., Heintzelman M. B. (1987). Morphology of ventral epidermis of Rana catesbeiana during metamorphosis. Anat. Rec. 217, 305-317 [DOI] [PubMed] [Google Scholar]
- Strubing C., Krapivinsky G., Clapham D. C. (2001). TRPC1 and TRPC5 form a novel cation channel in mammalian brain. Neuron 29, 645-655 [DOI] [PubMed] [Google Scholar]
- Taglietti V., Toselli M. (1988). A study of stretch activated channels in the membrane of frog oocytes: interactions with Ca2+ ions. J. Physiol. 407, 311-328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takada M., Yai H., Takayama-Arita K. (1995). Corticoid-induced differentiation of amiloride-blockable active Na+ transport across larval bullfrog skin in vitro. Am. J. Physiol. 268, C218-C226 [DOI] [PubMed] [Google Scholar]
- Takada M., Shimomura T., Hokari S., Jensik P. A., Cox T. C. (2006). Larval bullfrog skin expresses ENaC despite having no amiloride – blockable epithelial Na+ transport. J. Comp. Physiol. B 176, 287-293 [DOI] [PubMed] [Google Scholar]
- Van Driessche W., Zeiske W. (1985). Ca2+-sensitive, spontaneously fluctuating cation channels in the apical membrane of the adult frog skin. Pflugers Arch. 405, 250-259 [DOI] [PubMed] [Google Scholar]
- Weed I. G. (1933). Cytological studies of the epidermis of Rana pipiens and Rana climitans tadpoles with special references to the figures of Eberth. J. Morphol. 56, 213-229 [Google Scholar]
- Wilkinson N. S., Gao F., Hamill O. P. (1998). The effects of blockers of mechanically-gated channels on Xenopus oocyte growth and development. J. Membr. Biol. 165, 161-174 [DOI] [PubMed] [Google Scholar]
- Wintrebert P. (1905). Sur l'existence d'us irritablite excite-motrice primitive independente de voies nerveuses chez les embryons cilieas des batrachians. C. R. Seances Soc. Biol. Fil. 57, 645-647 [Google Scholar]
- Wintrebert P. (1920). La conduction aneurale de l'ectoderme chez les embryons d'amphibiens. C. R. Acad. Sci. III, Sci. Vie 171, 680-682 [Google Scholar]