Abstract
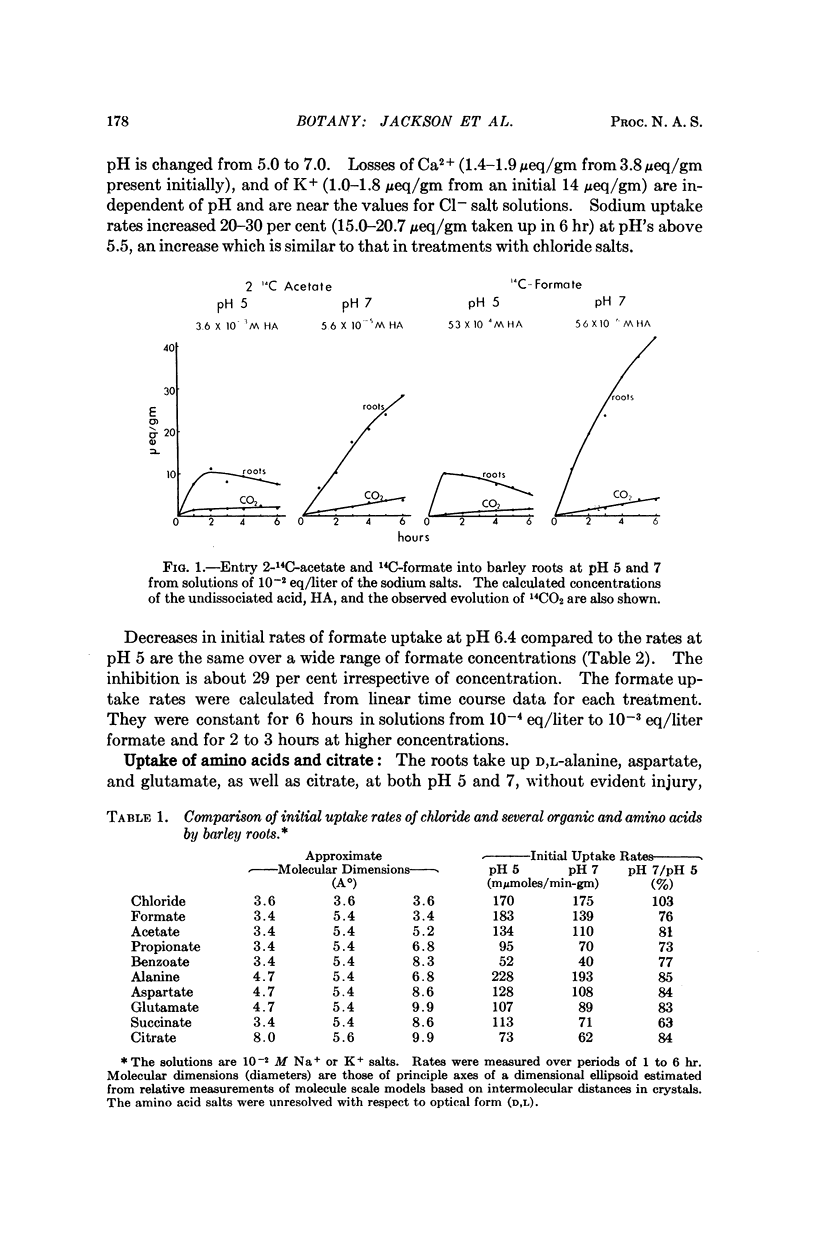
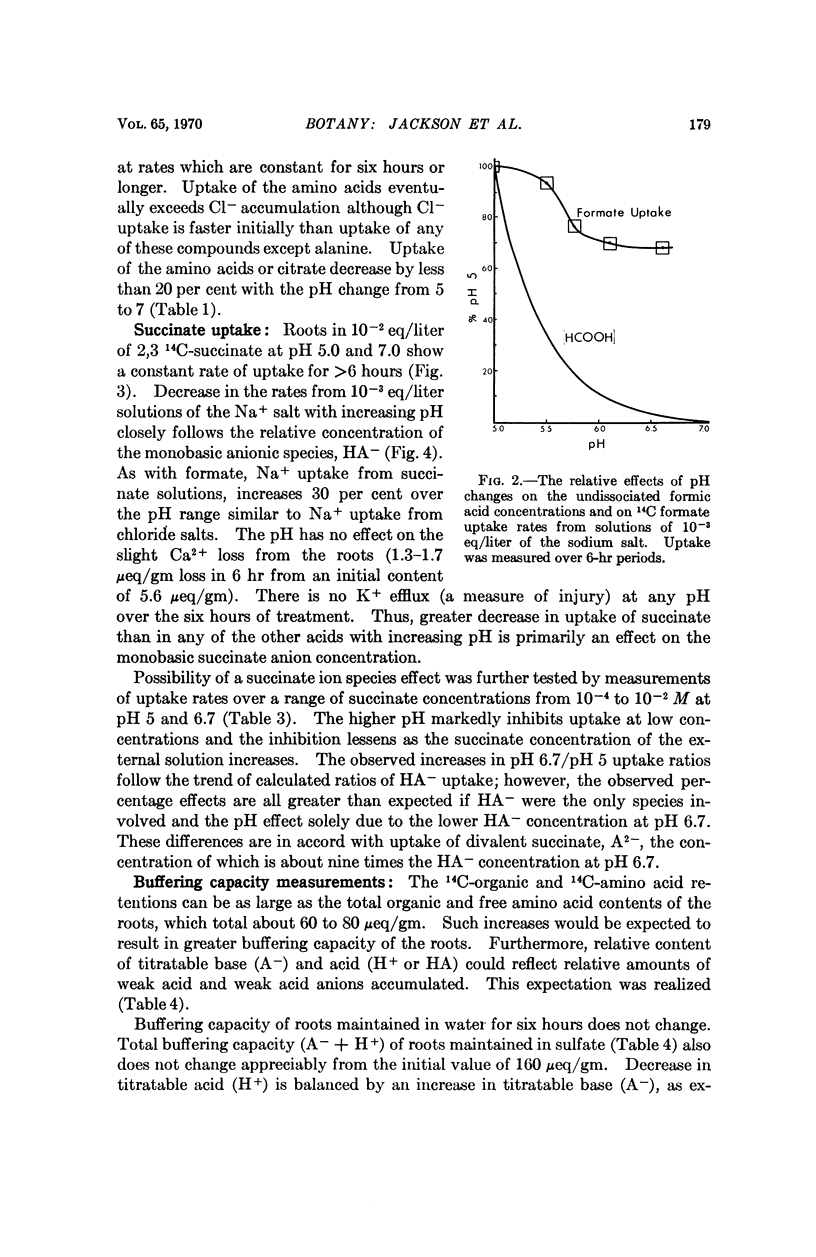
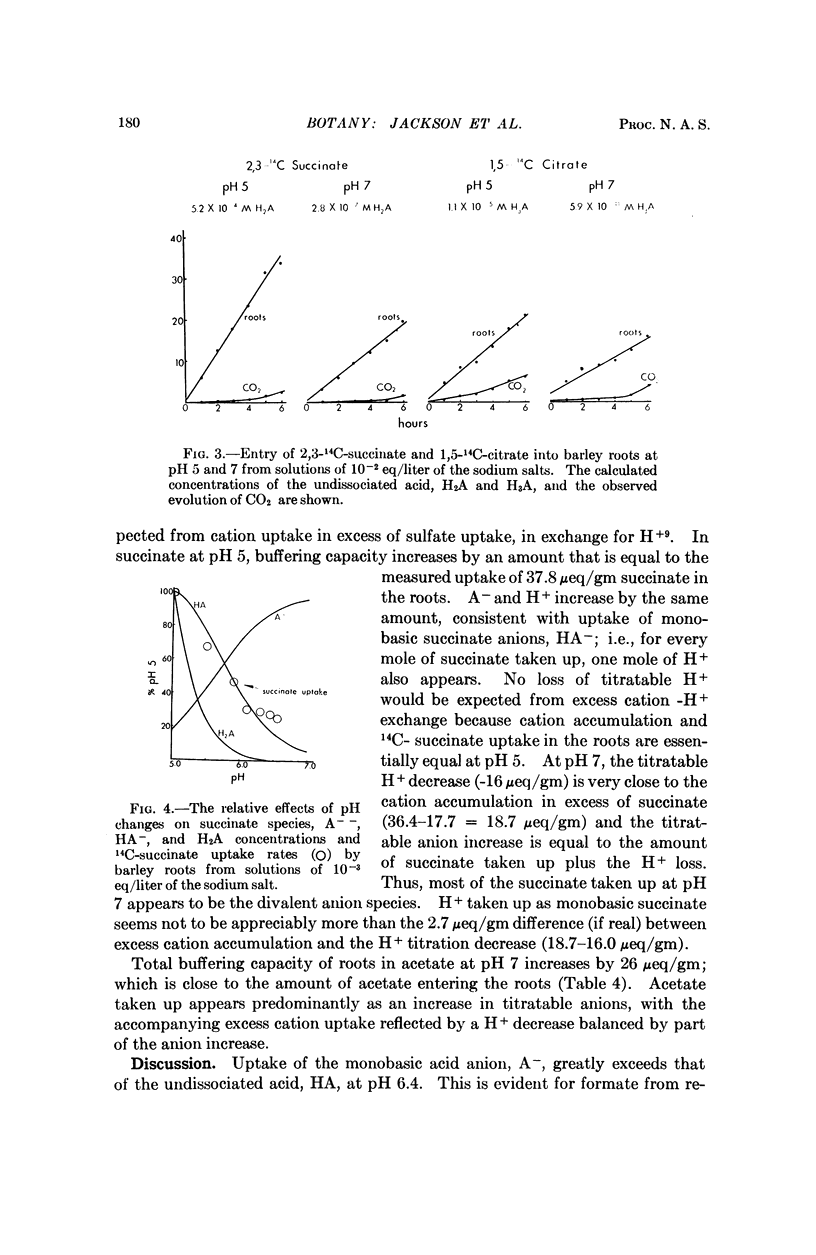
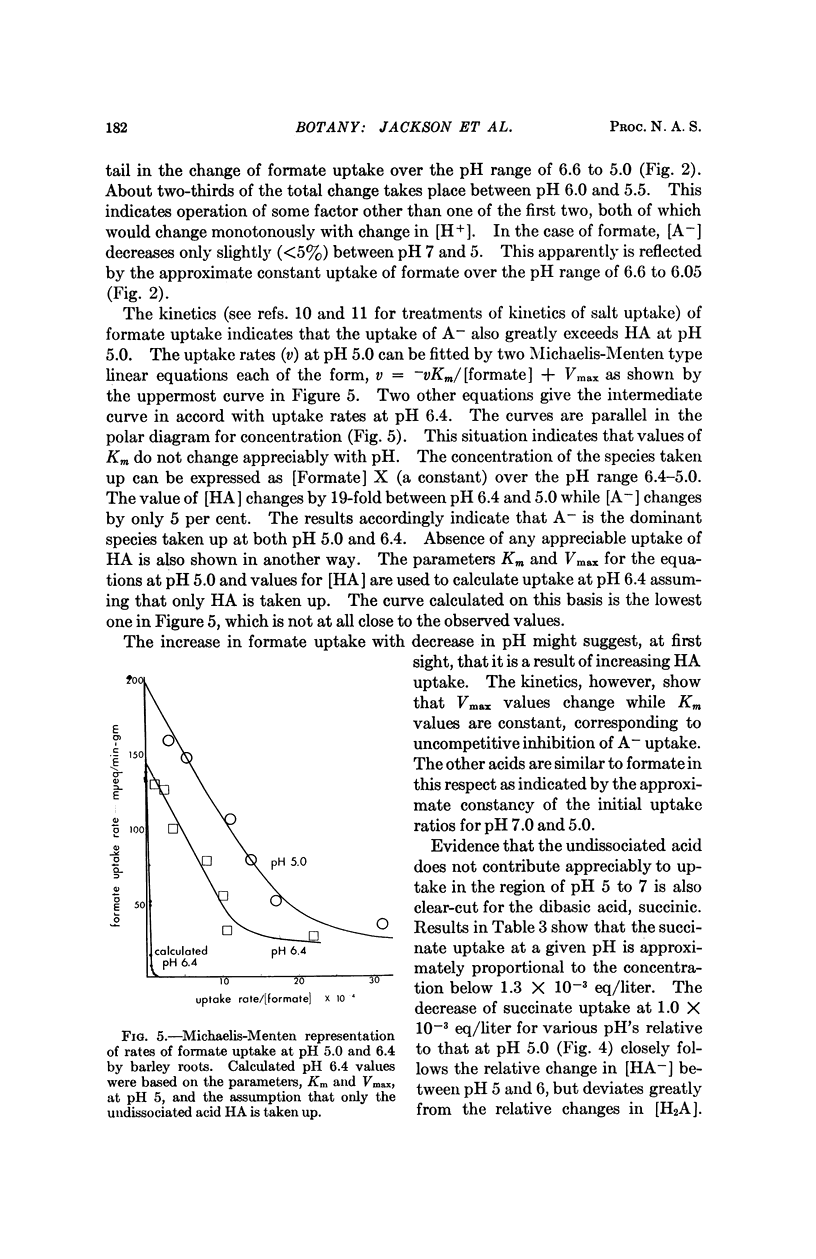
Entry of formate, acetate, succinate, and salts of several other organic acids into barley roots takes place chiefly as ions from solutions at pH 5 to 7. The results are in accord with entry of the organic acid predominantly at pH 5 as ions through hydrophylic regions of the limiting membranes rather than by distribution of the undissociated acid to lipid-rich regions, as has previously been held. These results with a plant tissue parallel observations of others indicative of similar hydrophylic properties of membranes from animals relative to transfer of ions and small nonelectrolytes.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- GIEBEL O., PASSOW H. [The permeability of erythrocyte membranes for organic anions. On the problem of diffusion through the pores]. Pflugers Arch Gesamte Physiol Menschen Tiere. 1960;271:378–388. [PubMed] [Google Scholar]
- Hagen C. E., Leggett J. E., Jackson P. C. THE SITES OF ORTHOPHOSPHATE UPTAKE BY BARLEY ROOTS. Proc Natl Acad Sci U S A. 1957 Jun 15;43(6):496–506. doi: 10.1073/pnas.43.6.496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JACKSON P. C., ADAMS H. R. Cation-anion balance during potassium and sodium absorption by barley roots. J Gen Physiol. 1963 Jan;46:369–386. doi: 10.1085/jgp.46.3.369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KOEFOED-JOHNSEN V., USSING H. H. The contributions of diffusion and flow to the passage of D2O through living membranes; effect of neurohypophyseal hormone on isolated anuran skin. Acta Physiol Scand. 1953 Mar 31;28(1):60–76. doi: 10.1111/j.1748-1716.1953.tb00959.x. [DOI] [PubMed] [Google Scholar]
- Lieb W. R., Stein W. D. Biological membranes behave as non-porous polymeric sheets with respect to the diffusion of non-electrolytes. Nature. 1969 Oct 18;224(5216):240–243. doi: 10.1038/224240a0. [DOI] [PubMed] [Google Scholar]
- Lips S. H., Beevers H. Compartmentation of Organic Acids in Corn Roots II. The Cytoplasmic Pool of Malic Acid. Plant Physiol. 1966 Apr;41(4):713–717. doi: 10.1104/pp.41.4.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MULLINS L. J. An analysis of pore size in excitable membranes. J Gen Physiol. 1960 May;43:105–117. doi: 10.1085/jgp.43.5.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solomon A. K. Characterization of biological membranes by equivalent pores. J Gen Physiol. 1968 May;51(5 Suppl):335S+–335S+. [PubMed] [Google Scholar]
- Villegas R., Bruzual I. B., Villegas G. M. Equivalent pore radius of the axolemma of resting and stimulated squid axons. J Gen Physiol. 1968 May;51(5 Suppl):81S+–81S+. [PubMed] [Google Scholar]



