Abstract
Background and objectives: Sparse longitudinal data exist on how diet influences microalbuminuria and estimated GFR (eGFR) decline in people with well-preserved kidney function.
Design, settings, participants, & measurements: Of the 3348 women participating in the Nurses' Health Study who had data on urinary albumin to creatinine ratio in 2000, 3296 also had data on eGFR change between 1989 and 2000. Cumulative average intake of nutrients over 14 years was derived from semiquantitative food frequency questionnaires answered in 1984, 1986, 1990, 1994, and 1998. Microalbuminuria presence and eGFR decline ≥30% were the outcomes of interest.
Results: Compared with the lowest quartile, the highest quartile of animal fat (odds ratio (OR): 1.72; 95% confidence interval (CI): 1.12 to 2.64) and two or more servings of red meat per week (OR: 1.51; 95% CI: 1.01 to 2.26) were directly associated with microalbuminuria. After adjustment for other nutrients individually associated with eGFR decline ≥30%, only the highest quartile of sodium intake remained directly associated (OR: 1.52; 95% CI: 1.10 to 2.09), whereas β-carotene appeared protective (OR: 0.62, 95% CI: 0.43 to 0.89). Results did not vary by diabetes status for microalbuminuria and eGFR outcomes or in those without hypertension at baseline for eGFR decline. No significant associations were seen for other types of protein, fat, vitamins, folate, fructose, or potassium.
Conclusions: Higher dietary intake of animal fat and two or more servings per week of red meat may increase risk for microalbuminuria. Lower sodium and higher β-carotene intake may reduce risk for eGFR decline.
Dietary modification represents an attractive intervention to prevent kidney function decline. Higher-protein diets have been hypothesized to result in progressive chronic kidney disease (CKD) (1,2), possibly through glomerular hyperfiltration (3), but scarce data are available on other nutrients or in people with preserved kidney function.
Diet studies of macrovascular cardiovascular disease (CVD) may be useful in constructing hypotheses for kidney microvascular disease manifesting as microalbuminuria (MA) and estimated GFR (eGFR) decline. Higher saturated and transunsaturated fat intakes have been associated with an increased risk of CVD (4,5), whereas monounsaturated and polyunsaturated fats (4), high fiber, and antioxidant vitamin supplementation (6,7) may be protective against vascular endothelial injury.
Therefore, we investigated how individual dietary nutrients, including protein (total, animal, vegetable, dairy, and nondairy), fat (total, saturated, trans, monosaturated, polyunsaturated, animal, and vegetable), cholesterol, fiber (total, soluble, and insoluble), antioxidant vitamins (vitamins A, C, E, and β-carotene), vitamin D, folate, fructose, sodium, and potassium, are associated with MA and eGFR decline. We hypothesized that higher intake of total and animal protein, saturated fat, transunsaturated fats, cholesterol, fructose, and sodium would increase risk for MA and eGFR decline, whereas higher intake of vegetable protein, monounsaturated and polyunsaturated fat, fiber, antioxidant vitamins, vitamin D, and potassium would be associated with a decreased risk.
Materials and Methods
Participants
The Nurses' Health Study (NHS) was initiated in 1976 with the enrollment of 121,700 U.S. nurses ages 30 to 55 years. This cohort is followed through biennial mailed questionnaires related to lifestyle factors and health outcomes. Between 1989 and 1990, 32,826 participants provided blood samples that were shipped overnight and stored at −130°C (8). In the year 2000, 18,720 of these participants submitted a second blood and spot urine specimen under the same handling and storage conditions. Participants who did and did not return blood samples were similar in terms of demographics and lifestyle characteristics.
Participants were from a substudy of analgesic use and renal function (9) or a substudy of type 2 diabetes and kidney function who had plasma creatinine measured in both 1989 and 2000 collections and/or urinary albumin to creatinine ratio (ACR) measured from the 2000 collection. The women in the analgesic study (n = 3876) had returned supplemental questionnaires regarding lifetime analgesic use. Among those who returned the analgesic questionnaire, 2712 women had plasma creatinine and urinary ACR data; we also included 730 women from the diabetes substudy. From this combined group (n = 3442), we included those with cumulative average dietary and urine ACR data in the year 2000 (n = 3348). The majority (n = 3296) also had plasma creatinine measured in samples collected in 1989 and 2000.
Assessment of Nutrient Intake
The semiquantitative food frequency questionnaires (FFQs) were designed to assess average food intake of more than 130 foods and beverages over the preceding year (10). A standard portion size and nine possible frequency of consumption responses, ranging from “never or less than once per month” to “6 or more times per day,” were given for each food item. Total energy and nutrient intakes were calculated from the reported frequency of consumption of each specified unit of food or beverage and from published data on the nutrient content of the specified portions (10). Previous validation studies among members of the NHS revealed good correlations between nutrients assessed by the FFQ and multiple weeks of food records completed over the preceding year (11). For example, correlation coefficients between 1986 FFQ and diet records obtained in 1986 were 0.68 for saturated fat, 0.48 for polyunsaturated fat, and 0.78 for crude fiber (12).
For analyses of MA, cumulative average energy adjusted nutrients for each participant were calculated from FFQs returned in 1984, 1986, 1990, 1994, and 1998, as described previously (4). Because direct associations were consistently seen between MA and nutrients derived from animal foods, we also examined the association between cumulative average red meat consumption (including beef, regular and lean hamburger, bacon, hot dogs, and processed meats) and MA.
For analyses of eGFR decline, cumulative averages of nutrients in 1984, 1986, and 1990 were used. We also examined associations between only the most recent FFQ and MA (1998) or eGFR decline (1990) to assess whether the cumulative effect of diet over several years or the immediate preceding diet were associated with the renal outcomes of interest differently.
Measurement of Urinary ACR
Urinary assays were performed on spot collections submitted in the year 2000; urinary creatinine concentration was measured by a modified Jaffe method (coefficient of variation (CV) 1.6%). Urinary albumin was measured using solid-phase fluorescence immunoassay using the Hitachi 911 analyzer and Roche diagnostics reagents (Indianapolis, IN) with a lower limit of detection of 0.1 mg/L (CV 8.0%). A urinary ACR of 25 to 355 μg/mg was used to define MA; this sex-specific cutpoint for women has been reported to approximate a urinary albumin excretion rate of 30 to 300 mg per 24 hours (13), which is traditionally considered clinically relevant MA. In this study, 205 women (6.1%) met the criterion for MA. There were 36 women with macroalbuminuria (ACR >355 μg/mg) who were excluded from the albuminuria analyses.
Measurement of Kidney Function Decline
Plasma creatinine was analyzed using a modified kinetic Jaffe reaction (CV 10%). In 2007, repeat measurement of 20 NHS plasma samples collected in 1989 (with a range of 0.6 to 1.4 mg/dl) and initially measured in 2000 revealed a mean recalibration coefficient (new value/original value) of 0.97 and confirmed that plasma creatinine is stable for many years under our storage conditions.
Glomerular filtration was estimated by the four-variable Modification in Diet in Renal Disease equation, where estimated GFR (ml/min per 1.73 m2) = 186 × [PCr (mg/dl)]−1.154 × [Age]−0.203 × [0.472 if female] × [1.21 if black] (14). An eGFR decline of ≥30% between 1989 and 2000 was considered to be a clinically significant change and has been used in previous analyses of renal function decline in NHS participants (15). We also examined “rapid” eGFR decline, defined as ≥3 ml/min per 1.73 m2 per year, which has been previously used as a cutoff that reflects three times more rapid decline than expected by normal aging (16).
Assessment of Covariates
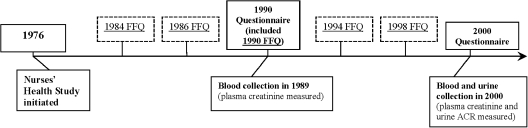
Race and height were initially reported on the 1992 questionnaire. Other self-reported clinical and lifestyle variables, including weight, hypertension (HTN), smoking status, physical activity, CVD (angina, myocardial infarction, coronary artery bypass surgery, percutaneous coronary revascularization, or stroke), and BP medication use, were reported on the biennial questionnaires. Questionnaire data collected closest to the year when kidney function was measured (the 1988 questionnaire for eGFR decline and the 2000 questionnaire for urinary ACR) were used (Figure 1). Many of these variables have been previously validated through direct medical records review (17,18).
Figure 1.
Timeline of questionnaire and biological data collection in NHS for these analyses. Questionnaires are administered every 2 years beginning in 1976, but only questionnaire data that included diet assessments are shown, except for the year 2000 questionnaire. FFQs asking about diet over the previous 12 months were administered in 1984, 1986, 1990, 1994, and 1998.
Participants reported new physician-diagnosed diseases, including diabetes, by physicians on the biennial questionnaires. We mailed a diabetes supplementary questionnaire to all women reporting diabetes to obtain further information about the date of diagnosis, symptoms, diagnostic tests, and treatment. We used the National Diabetes Data Group criteria to define diabetes self-reported up to the 1996 biennial questionnaire (19); the American Diabetes Association diagnostic criteria for diabetes released in 1997 were used for incident cases of diabetes reported in 1998 and after (20). Self-reported diagnosis of type 2 diabetes using the diabetes supplementary questionnaire has been established as 98% accurate in a separate validation study through medical records review (21). Because the onset of abnormalities in glucose handling can precede the diagnosis of type 2 diabetes by 10 years or more (22,23), we considered a participant who was diagnosed with diabetes up through the year 2000 as having diabetes.
Statistical Analysis
Wilcoxon rank sum and χ2 tests were used to compare differences between groups. Independent variables were quartiles of cumulative average nutrients, with the lowest quartile as the referent category. A cumulative average approach was chosen because it generally reflects long-term diet and also likely reduces measurement error from intraindividual variation over time (4). Logistic regression was used to examine associations between quartiles of nutrients and presence of MA in 2000 or eGFR decline ≥30% decline or “rapid” eGFR of ≥3 ml/min per 1.73 m2 per year between 1989 and 2000.
Multivariable models were adjusted for age, hypertension, body mass index (BMI), ever smoked, physical activity, CVD, and diabetes. For the outcome of MA, angiotensin-converting enzyme inhibitor or angiotensin-type 2 receptor blocker medication use and eGFR in 2000 were also included.
Change in dietary intake over time was also assessed using an ANOVA (24). All analyses were performed with SAS software, version 9.1 (SAS Institute Inc., Cary, NC). This study was approved by the Partners' Healthcare Brigham and Women's Hospital Human Research Committee Institutional Review Board.
Results
Study Participants and Dietary Factors
Median age was 67 years, 97% were Caucasian, 54% had HTN, and 24% were diabetic (Table 1). Cumulative average medians and ranges of relevant energy-adjusted nutrients are given in Table 2. There was <15% mean difference in energy-adjusted dietary intake for each individual nutrient between each questionnaire year and baseline FFQ data, suggesting that dietary intake remained relatively similar over time.
Table 1.
Demographic and clinical characteristics of participants in the NHS in 2000
| All NHS (n = 3348) | ACR <25 μg/mg (n = 3107) | ACR 25 to 355 μg/mg (n = 205) | GFR Decline <30% (n = 2916) | eGFR Decline ≥30% (n = 380) | |
|---|---|---|---|---|---|
| Age, yr | 67 | 67 | 69a | 67 | 69a |
| Caucasian, % | 97.3 | 97.3 | 98.0 | 97.3 | 97.9 |
| Hypertension, % | 54.4 | 52.7 | 73.7a | 52.0 | 69.2a |
| Diabetes, % | 23.7 | 20.8 | 57.6a | 21.2 | 37.6a |
| Cardiovascular disease, % | 6.2 | 5.1 | 16.1a | 4.9 | 15.3a |
| Current smoker, % | 6.0 | 5.7 | 6.8 | 6.0 | 4.2 |
| Ever smoker, % | 53.0 | 52.9 | 58 | 52.6 | 57.4 |
| Alcohol intake, g/d | 1.7 | 1.8 | 0.5a | 1.8 | 1.1 |
| Calorie intake, kcal/d | 1726 | 1728 | 1704 | 1728 | 1702 |
| Activity level, mets/wk | 11.4 | 11.8 | 7.7a | 12.1 | 7.7a |
| BMI, kg/m2 | 26.4 | 26.1 | 29.8a | 26.1 | 27.4a |
| ACE inhibitor or ARB medication use, % | 15.6 | 14.7 | 25.9a | 14.3 | 23.7a |
| Plasma creatinine in 1989, mg/dl | 0.74 | 0.75 | 0.76 | 0.76 | 0.69a |
| Plasma creatinine in 2000, mg/dl | 0.80 | 0.80 | 0.82b | 0.80 | 1.04a |
| eGFR in 2000, ml/min per 1.73 m2 | 76 | 76 | 72b | 78 | 56a |
| Urinary ACR, μg/mg | 3.4 | 3.2 | 45 | 3.3 | 4.3a |
Results expressed as median or %. Note: There were 3348 with ACR data, but 36 women with ACR >355 were excluded from ACR analyses; 3296 with eGFR decline data. ACE, angiotensin-converting enzyme inhibitor; ARB, angiotensin-type 2 receptor blocker.
P < 0.001 when compared to ACR <25 μg/mg or eGFR decline ≤30% referent groups.
P < 0.01 when compared to ACR <25 μg/mg or eGFR decline ≤30% referent groups.
Table 2.
Medians and ranges for quartiles of cumulative averaged energy adjusted individual nutrients as estimated by FFQs
| Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | |
|---|---|---|---|---|
| Animal protein, g/d | 41.9 (0.69 to 46.2) | 49.7 (46.3 to 52.7) | 55.5 (52.8 to 59.0) | 61.2 (59.1 to 90.6) |
| Vegetable protein, g/d | 17.5 (10.6 to 18.8) | 20.0 (18.9 to 21.0) | 22.0 (21.1 to 23.1) | 24.9 (23.2 to 50.2) |
| Low-fat dairy protein, g/d | 0.82 (0 to 1.2) | 1.7 (1.2 to 2.1) | 2.8 (2.2 to 3.5) | 4.6 (3.6 to 8.9) |
| Saturated fat, g/d | 14.9 (4.8 to 16.4) | 17.7 (16.5 to 18.8) | 20.0 (18.9 to 21.0) | 22.9 (21.1 to 38.1) |
| Monounsaturated fat, g/d | 16.9 (8.7 to 18.6) | 20.0 (18.7 to 21.0) | 22.1 (21.1 to 23.4) | 25.1 (23.4 to 42.0) |
| Animal fat, g/d | 22.3 (0.2 to 25.3) | 27.6 (25.4 to 29.8) | 31.9 (29.9 to 34.4) | 37.9 (34.5 to 72.2) |
| Cholesterol, mg/d | 184 (9 to 208) | 230 (208 to 246) | 264 (247 to 284) | 316 (284 to 722) |
| Sodium, g/d | 1.7 (1.1 to 1.7) | 1.9 (1.8 to 1.9) | 2.1 (2.0 to 2.2) | 2.4 (2.3 to 4.9) |
| Total β-carotene, mg/d | 2.2 (0.2 to 2.8) | 3.3 (2.8 to 3.8) | 4.5 (3.9 to 5.3) | 5.3 (5.4 to 26.0) |
| Total vitamin E, mg/d | 6.0 (2.6 to 8.1) | 13.6 (8.2 to 23.0) | 73.0 (23.1 to 140) | 292.9 (140 to 1187) |
| Vitamin E from foods, mg/d | 4.9 (2.1 to 5.4) | 5.8 (5.4 to 6.1) | 6.6 (6.1 to 7.1) | 8.3 (7.1 to 38.1) |
| Supplemental vitamin E, mg/d | 0.0 (0.0 to 0.0) | 7.5 (0.03 to 16.1) | 66.7 (16.1 to 133.3) | 300 (133 to 900) |
| Total folate, μg/d | 244 (111 to 270) | 292 (270 to 312) | 334 (312 to 359) | 398 (359 to 726) |
Only nutrients with significant multivariable ORs are included.
Cumulative average dietary animal protein was significantly inversely correlated with vegetable protein (r = −0.25, P < 0.001). Saturated fat was highly correlated with animal fat (Spearman r = 0.84, P < 0.001) and moderately correlated with cholesterol (Spearman r = 0.43, P < 0.001). Animal protein and animal fat were significantly directly correlated (r = 0.41, P < 0.001), as were animal fat and cholesterol (r = 0.63, P < 0.001). Total dietary vitamin E and folate were also correlated (r = 0.54, P < 0.001). Sodium intake was moderately associated with dietary fats (Spearman correlations 0.11 to 0.18) and minimally associated with vitamin intake (Spearman correlations −0.01 to −0.04).
We present only those results that were significant in multivariable adjusted models for either MA or eGFR decline ≥30% when comparing quartile 4 to quartile 1 of dietary intake.
Microalbuminuria
In this cohort, 205 women (6.1%) had MA. After adjusting for covariates, animal protein, saturated fat, animal fat, and cholesterol remained independently associated with presence of MA (Table 3). Higher intake of vegetable protein had a borderline inverse association with MA. BMI and diabetes status substantially attenuated the associations between these nutrients and MA. When animal protein, vegetable protein, and animal fat were included in a multivariable model, the magnitudes of the associations were attenuated and lost statistical significance. We did not create multivariable models that include animal fat and saturated fat or cholesterol because of the high correlations between these nutrients. Intake of two or more servings a week of red and processed meat was associated with MA (multivariable odds ratio (OR): 1.51; 95% confidence interval (CI): 1.01 to 2.26) when compared with less than one serving per week.
Table 3.
Dietary factors with significant adjusted ORs [95% CI] for quartile 4 versus quartile 1 of cumulative averaged energy adjusted individual protein and fat nutrients and presence of MA (ACR 25 to 355 μg/mg) or eGFR decline ≥30%
| MA (ACR 25 to 355 μg/mg) | eGFR Decline ≥30% | |
|---|---|---|
| Animal protein | ||
| age-adjusted | 2.54 [1.67, 3.88] | 1.34 [0.99, 1.31] |
| MV modela | 1.74 [1.11, 2.75] | 1.07 [0.79, 1.45] |
| MV + animal fat | 1.43 [0.88, 2.31] | 0.91 [0.65, 1.27] |
| Vegetable protein | ||
| age-adjusted | 0.66 [0.59, 0.99] | 0.84 [0.62, 1.17] |
| MVa + animal protein | 0.65 [0.42, 1.02] | 0.93 [0.66, 1.30] |
| Low-fat dairy protein | ||
| age-adjusted | 0.86 [0.57, 1.30] | 0.74 [0.44, 1.00] |
| MV modela | 0.91 [0.59, 1.39] | 0.71 [0.52, 0.97] |
| Saturated fat | ||
| age-adjusted | 2.37 [1.58, 3.56] | 1.61 [1.16, 2.22] |
| MVa + monounsaturated and polyunsaturated fat | 1.60 [0.86, 2.99] | 1.62 [1.02, 2.59] |
| Monounsaturated fat | ||
| age-adjusted | 1.95 [1.31, 2.90] | 1.65 [1.21, 2.26] |
| MV modela | 1.41 [0.93, 2.15] | 1.48 [1.08, 2.04] |
| MVa + saturated and polyunsaturated fat | 1.40 [0.71, 2.74] | 1.06 [0.64, 1.76] |
| Animal fat | ||
| age-adjusted | 2.80 [1.87, 4.18] | 1.75 [1.29, 2.38] |
| MVa + vegetable fat | 1.66 [1.08, 2.57] | 1.49 [1.08, 2.07] |
| Vegetable fat | ||
| age-adjusted | 0.63 [0.41, 0.95] | 0.93 [0.69, 1.24] |
| MVa + animal fat | 0.76 [0.49, 1.17] | 1.06 [0.78, 1.45] |
| Cholesterol | ||
| age-adjusted | 2.58 [1.71, 3.89] | 1.37 [1.00, 1.88] |
| MV modela | 1.64 [1.06, 2.55] | 1.12 [0.81, 1.56] |
| Sodium | ||
| age-adjusted | 1.56 [1.06, 2.29] | 1.75 [1.29, 2.38] |
| MV modela | 0.94 [0.63, 1.41] | 1.53 [1.11, 2.09] |
MV, multivariable.
Adjusted for age, hypertension, BMI, diabetes, cigarette smoking, activity (mets/wk), and cardiovascular disease. MA models also adjusted for eGFR and angiotensin-converting enzyme inhibitor/angiotensin-type 2 receptor blocker medication use.
When the nutrient data from the most recent questionnaire year (1998 only) were used instead of cumulative average values, results were consistent but attenuated (data not shown). Using eGFR decline instead of eGFR in the 2000 in adjusted models of MA also did not meaningfully change the results.
eGFR Decline
Of the 3296 women with data for eGFR change, 380 (11.5%) experienced an eGFR decline ≥30% between 1989 and 2000; this reflected a median increase in plasma creatinine of 0.33 mg/dl. Median eGFR in 1989 was 84.5 ml/min per 1.73 m2, and only 147 women (4.5%) had eGFR <60 ml/min per 1.73 m2 at that time. Those who had eGFR decline ≥30% were more likely to have HTN, diabetes, CVD, higher BMI, and be on an angiotensin-converting enzyme inhibitor or angiotensin-type 2 receptor blocker medication (Table 1).
Higher intakes of total fat, saturated fat, animal fat, and sodium were significantly directly associated with eGFR decline in multivariable models, whereas low-fat dairy was inversely associated (Table 3). After adjusting for nondietary covariates, higher intakes of β-carotene (OR: 0.56; 95% CI: 0.40 to 0.78), vitamin E (OR: 0.68; 95% CI: 0.50 to 0.93), and folate (OR: 0.74; 95% CI: 0.55 to 1.00) were inversely associated with eGFR decline, but only total β-carotene intake remained significant after adjusting for vitamin E and folate (Table 4). Limiting the nutrient analyses to those with baseline eGFR <80 ml/min per 1.73 m2 at baseline (n = 1278) (9) did not meaningfully change any of the observed associations (data not shown). In a model that included all nutrients that remained significantly associated with eGFR decline after multivariable adjustment, only higher dietary sodium intake remained directly associated with eGFR decline ≥30%, whereas higher β-carotene intake was inversely associated (Table 5). In those without baseline HTN (n = 2329), the highest quartile of sodium intake was still associated with eGFR decline ≥30% (multivariable OR: 1.69; 95% CI: 1.13 to 2.54, P for trend = 0.008).
Table 4.
ORs [95% CI] for quartile 4 versus quartile 1 of β-carotene, vitamin E, and folate intake and presence of MA (ACR 25 to 355 μg/mg) or eGFR decline ≥30%
| MA (ACR 25 to 355 μg/mg) | eGFR Decline ≥30% | |
|---|---|---|
| Total β-carotene | ||
| age-adjusted | 0.99 [0.65, 1.49] | 0.56 [0.41, 0.78] |
| MVa + vitamin E + folate | 1.07 [0.66, 1.72] | 0.60 [0.42, 0.85] |
| Total vitamin E | ||
| age-adjusted | 1.19 [0.91, 1.76] | 0.64 [0.47, 0.87] |
| MVa + folate + β-carotene | 1.57 [0.97, 2.57] | 0.68 [0.47, 1.00] |
| Total folate | ||
| age-adjusted | 0.72 [0.48, 1.10] | 0.70 [0.52, 0.95] |
| MVa + vitamin E + β-carotene | 0.78 [0.46, 1.34] | 1.22 [0.80, 1.88] |
MV, multivariable.
Adjusted for age, hypertension, BMI, diabetes, cigarette smoking, activity (mets/wk), and cardiovascular disease. MA models also adjusted for eGFR and angiotensin-converting enzyme inhibitor/angiotensin-type 2 receptor blocker medication use.
Table 5.
Multivariable-adjusted model including multiple dietary nutrients for quartile 4 versus quartile 1 of each nutrient (ORs [95% CI])
| Multivariable Modela Including | eGFR Decline ≥30% |
|---|---|
| Low-fat dairy | 0.76 [0.55, 1.05] |
| Animal fat | 1.20 [0.82, 1.76] |
| Monounsaturated fat | 1.01 [0.67, 1.52] |
| Sodium | 1.52 [1.10, 2.09] |
| β-carotene | 0.62 [0.43, 0.89] |
| Vitamin E | 0.78 [0.57, 1.08] |
Adjusted for age, hypertension, BMI, diabetes, cigarette smoking, activity (mets/wk), and cardiovascular disease. MA models also adjusted for eGFR and angiotensin-converting enzyme inhibitor/angiotensin-type 2 receptor blocker medication use (note: very similar results if saturated fat used instead of animal fat).
Red and processed meat intake was not associated with eGFR decline ≥30% nor with eGFR decline ≥3 ml/min per 1.73 m2 per year. In multivariable models, only the highest quartile of sodium remained significantly associated with eGFR decline ≥3 ml/min per 1.73 m2 per year (OR: 1.68; 95% CI: 1.14 to 2.46).
Stratification by Diabetes Status
Because those with diabetes comprised a large percentage of those with MA or eGFR decline, stratified analysis by diabetes status was performed. Associations between dietary nutrients and MA or renal function decline did not vary by presence of diabetes (all interaction terms P > 0.25).
Discussion
MA and modest decrements in eGFR are powerfully associated with subsequent overt kidney disease (25), cardiovascular risk (26–28), and all-cause mortality (28,29). We report that dietary factors may be important modifiable risk factors in decreasing risk for MA and kidney function decline in people with well-preserved eGFR at baseline. Specifically, diets higher in animal protein, animal fat, and cholesterol may be associated with MA. Diets lower in sodium but higher in β-carotene may be protective against eGFR decline. Results using only the most recent FFQ data were quantitatively very similar, but associations were attenuated for most nutrients, suggesting that long-term intake may be more important.
Our findings are consistent with an analysis of diet patterns and albuminuria in over 5000 participants of the Multi-Ethnic Study of Atherosclerosis, which reported a direct association between each additional serving per day of nondairy animal food intake with ACR (30). No results for eGFR decline were reported in the Multi-Ethnic Study of Atherosclerosis, however.
Consumption of animal fat significantly alters kidney structure and function. Wistar rats fed a high-animal fat diet of lard and egg yolk had decreased glomerular number and size (and greatest increase in BP) when compared with rats fed a high-fat diet of canola oil, which is high in monounsaturated fat (31). In aging rats fed canola oil, fish oil, or butter, eGFR decline was greatest in those animals who received butter, which is high in saturated fat (32). Published literature on dietary fat and MA in humans is currently very limited, however. One observational dietary study of 48 Australian type 1 diabetics with MA compared with 130 diabetics without MA found an OR of 4.9 (1.2 to 20.0) when comparing the highest to lowest quintile of saturated fat intake for risk of MA (33). We are not aware of other published data on the relation of dietary fat intake and kidney function decline in adults either general population studies or in type 2 diabetes.
Because high intake of animal protein promotes renal damage in experimental animal models (3), the effect of low-protein diets on eGFR decline in humans has been of interest. The Modification in Diet in Renal Disease study was the largest randomized trial examining the effect of a low-protein diet (0.6 g/kg per day versus 1.3 g/kg per day) on progression of renal insufficiency in 585 subjects with moderate chronic kidney disease (GFR 25 to 55 cc/min); of note, only 3% had type 2 diabetes. With a mean follow-up of only 2.2 years, the investigators did not find a significant difference between the diet groups or BP groups (34). The utility of protein restriction in GFR decline has remained controversial in light of two subsequent meta-analyses. Pedrini et al. (2) found that a low-protein diet significantly reduced risk for renal failure or death (relative risk: 0.67; CI: 0.50 to 0.89). By incorporating additional subsequent trials, the second meta-analysis detected a very small benefit of only 0.53 ml/min per year in reduction of decline of GFR; however, the beneficial effect of a lower-protein diet was more evident in diabetics (5.4 ml/min per year benefit) (35). We did not find associations between either total or animal protein intake and eGFR decline after adjusting for clinical covariates in this study. Compared with the previous investigations, our observational study has the advantage of an 11-year follow-up for eGFR change and inclusion of over 700 diabetics in a group with well-preserved kidney function at baseline.
Of the 3348 women in this study, 1624 women were included in a previously published analysis reporting an association between animal protein and eGFR decline over 11 years (9). The association was seen only in those with baseline creatinine clearance of <80 ml/min (−1.69 ml/min per 1.73 m2 over 11 years per 10-g increase in nondairy animal protein) (9), but we did not observe a statistically significant association in this expanded study population. One difference is that in the first study, eGFR change as a continuous variable was the primary outcome, whereas a dichotomous categorization for eGFR decline was used here. Also, cumulative average nutrients were used here, whereas the previous analysis used protein intake from the 1990 FFQ only.
The association of higher salt intake with eGFR decline was consistently seen in our analyses. A previous observational retrospective study also reported that higher salt intake (4.6 g versus 2.3 g per day measured by urinary sodium excretion) was associated with faster creatinine clearance decline (−0.41 versus −0.25 ml/min per month, P < 0.05) over 3 years in 57 Italians with baseline creatinine clearance between 10 and 40 ml/min (36). Modulation of TGF-β has been proposed as a key link between high dietary sodium and progressive kidney function decline on the basis of multiple animal studies (37). Elevated BP from higher sodium intake may also play an important role in eGFR decline. This study provides additional evidence that higher sodium is associated with eGFR decline in people with well-preserved kidney function.
Antioxidant vitamin supplementation decreases intrarenal free-radical damage in streptozocin-induced diabetic rats (38). We found no published data on β-carotene (abundant in green leafy and root vegetables) and kidney function in animals or humans, however, and only sparse data on vitamin E and long-term kidney outcomes in humans. An 8-month, double-blinded, randomized crossover trial of 36 people with type 1 diabetes found that vitamin E supplementation normalized supranormal creatinine clearance (39). We previously noted that higher intake of vitamin E was significantly inversely associated with presence of eGFR <60 ml/min per 1.73 m2 in type 2 diabetics in NHS women as well as in the Health Professionals Follow-Up Study in a cross-sectional analysis (40). One randomized, double-blind study of 93 patients with mild to moderate CKD reported that intervention with pravastatin, vitamin E supplementation, and homocysteine-lowering therapy improved vascular parameters but showed no difference in renal function after 2 years (41); the small number of participants and relatively short follow-up make it difficult to draw strong conclusions.
Some important limitations of this investigation deserve mention. The study population was composed of mostly older Caucasian women, so the results may not necessarily be generalizable to other races or ethnicities, age groups, or men; however, there is no pathophysiologic evidence to suggest associations between diet and kidney function would be different. Specific values for systolic and diastolic BP for participants were also not available. In addition, ACR was assessed only once in 2000, and therefore the ACR analyses are cross-sectional. The presence of residual confounding is also possible. The possibility of survival bias is present because those women who died before 2000 would not have been included in this study; however, we would expect this to bias the results toward the null in that participants with faster eGFR decline would be more likely to die earlier.
In summary, diets lower in animal protein, animal fat, and cholesterol may be protective against MA. Notably, consumption of two or more servings of red or processed meat per day was associated with an approximately 50% increased odds for MA. Lower sodium and higher β-carotene intake may be protective against GFR decline. These associations appear to be stronger when considering cumulative average intake over many years and are similar in nondiabetics and diabetics. These findings suggest that dietary modification may be an important strategy in maintaining kidney health.
Disclosures
None.
Acknowledgments
We would like to thank Elaine Coughlan-Gifford, Tricia Li, and Peter Gaccione for statistical programming support and Manyee To for assistance in manuscript preparation. This work was supported by National Institutes of Health grants K08 DK066246 and R03 DK078551 (to J.L.), R01DK066574 (to G.C.C.), R01HL065582 (to F.B.H.), and P01CA055075. A portion of this work was published as an abstract and presented as an oral free communication at the American Society of Nephrology meeting in San Diego, CA, in October 2009.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.The Modification of Diet in Renal Disease Study: Design, methods, and results from the feasibility study. Am J Kidney Dis 20: 18– 33, 1992 [DOI] [PubMed] [Google Scholar]
- 2.Pedrini MT, Levey AS, Lau J, Chalmers TC, Wang PH: The effect of dietary protein restriction on the progression of diabetic and nondiabetic renal diseases: A meta-analysis. Ann Intern Med 124: 627– 632, 1996 [DOI] [PubMed] [Google Scholar]
- 3.Brenner BM, Meyer TW, Hostetter TH: Dietary protein intake and the progressive nature of kidney disease: The role of hemodynamically mediated glomerular injury in the pathogenesis of progressive glomerular sclerosis in aging, renal ablation, and intrinsic renal disease. N Engl J Med 307: 652– 659, 1982 [DOI] [PubMed] [Google Scholar]
- 4.Hu FB, Stampfer MJ, Rimm E, Ascherio A, Rosner BA, Spiegelman D, Willett WC: Dietary fat and coronary heart disease: A comparison of approaches for adjusting for total energy intake and modeling repeated dietary measurements. Am J Epidemiol 149: 531– 540, 1999 [DOI] [PubMed] [Google Scholar]
- 5.Lopez-Garcia E, Hu FB: Nutrition and the endothelium. Curr Diab Rep 4: 253– 259, 2004 [DOI] [PubMed] [Google Scholar]
- 6.Hu FB, Willett WC: Optimal diets for prevention of coronary heart disease. JAMA 288: 2569– 2578, 2002 [DOI] [PubMed] [Google Scholar]
- 7.Brown AA, Hu FB: Dietary modulation of endothelial function: implications for cardiovascular disease. Am J Clin Nutr 73: 673– 686, 2001 [DOI] [PubMed] [Google Scholar]
- 8.Schulze MB, Hoffmann K, Manson JE, Willett WC, Meigs JB, Weikert C, Heidemann C, Colditz GA, Hu FB: Dietary pattern, inflammation, and incidence of type 2 diabetes in women. Am J Clin Nutr 82: 675– 684, 2005; quiz 714–715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Knight EL, Stampfer MJ, Hankinson SE, Spiegelman D, Curhan GC: The impact of protein intake on renal function decline in women with normal renal function or mild renal insufficiency. Ann Intern Med 138: 460– 467, 2003 [DOI] [PubMed] [Google Scholar]
- 10.Willett WC, Sampson L, Stampfer MJ, Rosner B, Bain C, Witschi J, Hennekens CH, Speizer FE: Reproducibility and validity of a semiquantitative food frequency questionnaire. Am J Epidemiol 122: 51– 65, 1985 [DOI] [PubMed] [Google Scholar]
- 11.Willett W: Nutritional Epidemiology, New York, Oxford University Press, 1998 [Google Scholar]
- 12.Salvini S, Hunter DJ, Sampson L, Stampfer MJ, Colditz GA, Rosner B, Willett WC: Food-based validation of a dietary questionnaire: The effects of week-to-week variation in food consumption. Int J Epidemiol 18: 858– 867, 1989 [DOI] [PubMed] [Google Scholar]
- 13.Warram JH, Gearin G, Laffel L, Krolewski AS: Effect of duration of type I diabetes on the prevalence of stages of diabetic nephropathy defined by urinary albumin/creatinine ratio. J Am Soc Nephrol 7: 930– 937, 1996 [DOI] [PubMed] [Google Scholar]
- 14.Stevens LA, Coresh J, Greene T, Levey AS: Assessing kidney function–measured and estimated glomerular filtration rate. N Engl J Med 354: 2473– 2483, 2006 [DOI] [PubMed] [Google Scholar]
- 15.Curhan GC, Knight EL, Rosner B, Hankinson SE, Stampfer MJ: Lifetime nonnarcotic analgesic use and decline in renal function in women. Arch Intern Med 164: 1519– 1524, 2004 [DOI] [PubMed] [Google Scholar]
- 16.Rifkin DE, Shlipak MG, Katz R, Fried LF, Siscovick D, Chonchol M, Newman AB, Sarnak MJ: Rapid kidney function decline and mortality risk in older adults. Arch Intern Med 168: 2212– 2218, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hu FB, Willett WC, Colditz GA, Ascherio A, Speizer FE, Rosner B, Hennekens CH, Stampfer MJ: Prospective study of snoring and risk of hypertension in women. Am J Epidemiol 150: 806– 816, 1999 [DOI] [PubMed] [Google Scholar]
- 18.Fung TT, Rexrode KM, Mantzoros CS, Manson JE, Willett WC, Hu FB: Mediterranean diet and incidence of and mortality from coronary heart disease and stroke in women. Circulation 119: 1093– 1100, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. National Diabetes Data Group. Diabetes 28: 1039– 1057, 1979 [DOI] [PubMed] [Google Scholar]
- 20.Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 20: 1183– 1197, 1997 [DOI] [PubMed] [Google Scholar]
- 21.Shai I, Schulze MB, Manson JE, Rexrode KM, Stampfer MJ, Mantzoros C, Hu FB: A prospective study of soluble tumor necrosis factor-alpha receptor II (sTNF-RII) and risk of coronary heart disease among women with type 2 diabetes. Diabetes Care 28: 1376– 1382, 2005 [DOI] [PubMed] [Google Scholar]
- 22.Pradhan AD, Rifai N, Buring JE, Ridker PM: Hemoglobin A1c predicts diabetes but not cardiovascular disease in nondiabetic women. Am J Med 120: 720– 727, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tirosh A, Shai I, Tekes-Manova D, Israeli E, Pereg D, Shochat T, Kochba I, Rudich A: Normal fasting plasma glucose levels and type 2 diabetes in young men. N Engl J Med 353: 1454– 1462, 2005 [DOI] [PubMed] [Google Scholar]
- 24.Littell RC, Pendergast J, Natarajan R: Modelling covariance structure in the analysis of repeated measures data. Stat Med 19: 1793– 1819, 2000 [DOI] [PubMed] [Google Scholar]
- 25.Verhave JC, Gansevoort RT, Hillege HL, Bakker SJ, De Zeeuw D, de Jong PE: An elevated urinary albumin excretion predicts de novo development of renal function impairment in the general population. Kidney Int Suppl: S18– S21, 2004 [DOI] [PubMed] [Google Scholar]
- 26.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY: Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 351: 1296– 1305, 2004 [DOI] [PubMed] [Google Scholar]
- 27.Anavekar NS, McMurray JJ, Velazquez EJ, Solomon SD, Kober L, Rouleau JL, White HD, Nordlander R, Maggioni A, Dickstein K, Zelenkofske S, Leimberger JD, Califf RM, Pfeffer MA: Relation between renal dysfunction and cardiovascular outcomes after myocardial infarction. N Engl J Med 351: 1285– 1295, 2004 [DOI] [PubMed] [Google Scholar]
- 28.Wachtell K, Ibsen H, Olsen MH, Borch-Johnsen K, Lindholm LH, Mogensen CE, Dahlof B, Devereux RB, Beevers G, de Faire U, Fyhrquist F, Julius S, Kjeldsen SE, Kristianson K, Lederballe-Pedersen O, Nieminen MS, Okin PM, Omvik P, Oparil S, Wedel H, Snapinn SM, Aurup P: Albuminuria and cardiovascular risk in hypertensive patients with left ventricular hypertrophy: The LIFE study. Ann Intern Med 139: 901– 906, 2003 [DOI] [PubMed] [Google Scholar]
- 29.Solomon SD, Lin J, Solomon CG, Jablonski KA, Rice MM, Steffes M, Domanski M, Hsia J, Gersh BJ, Arnold JM, Rouleau J, Braunwald E, Pfeffer MA: Influence of albuminuria on cardiovascular risk in patients with stable coronary artery disease. Circulation 116: 2687– 2693, 2007 [DOI] [PubMed] [Google Scholar]
- 30.Nettleton JA, Steffen LM, Palmas W, Burke GL, Jacobs DR, Jr.: Associations between microalbuminuria and animal foods, plant foods, and dietary patterns in the Multiethnic Study of Atherosclerosis. Am J Clin Nutr 87: 1825– 1836, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Aguila MB, Mandarim-De-Lacerda CA: Effects of chronic high fat diets on renal function and cortical structure in rats. Exp Toxicol Pathol 55: 187– 195, 2003 [DOI] [PubMed] [Google Scholar]
- 32.Valente Gamba C, Zeraib Caraviello A, Matsushita A, Alves GM, Nunes Da Silva L, Nascimento Gomes G, Zaladek Gil F: Effects of dietary lipids on renal function of aged rats. Braz J Med Biol Res 34: 265– 269, 2001 [DOI] [PubMed] [Google Scholar]
- 33.Riley MD, Dwyer T: Microalbuminuria is positively associated with usual dietary saturated fat intake and negatively associated with usual dietary protein intake in people with insulin-dependent diabetes mellitus. Am J Clin Nutr 67: 50– 57, 1998 [DOI] [PubMed] [Google Scholar]
- 34.Klahr S, Levey AS, Beck GJ, Caggiula AW, Hunsicker L, Kusek JW, Striker G: The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. Modification of Diet in Renal Disease Study Group. N Engl J Med 330: 877– 884, 1994 [DOI] [PubMed] [Google Scholar]
- 35.Kasiske BL, Lakatua JD, Ma JZ, Louis TA: A meta-analysis of the effects of dietary protein restriction on the rate of decline in renal function. Am J Kidney Dis 31: 954– 961, 1998 [DOI] [PubMed] [Google Scholar]
- 36.Cianciaruso B, Bellizzi V, Minutolo R, Tavera A, Capuano A, Conte G, De Nicola L: Salt intake and renal outcome in patients with progressive renal disease. Miner Electrolyte Metab 24: 296– 301, 1998 [DOI] [PubMed] [Google Scholar]
- 37.Sanders PW: Effect of salt intake on progression of chronic kidney disease. Curr Opin Nephrol Hypertens 15: 54– 60, 2006 [DOI] [PubMed] [Google Scholar]
- 38.Mekinova D, Chorvathova V, Volkovova K, Staruchova M, Grancicova E, Klvanova J, Ondreicka R: Effect of intake of exogenous vitamins C, E and beta-carotene on the antioxidative status in kidneys of rats with streptozotocin-induced diabetes. Nahrung 39: 257– 261, 1995 [DOI] [PubMed] [Google Scholar]
- 39.Bursell SE, Clermont AC, Aiello LP, Aiello LM, Schlossman DK, Feener EP, Laffel L, King GL: High-dose vitamin E supplementation normalizes retinal blood flow and creatinine clearance in patients with type 1 diabetes. Diabetes Care 22: 1245– 1251, 1999 [DOI] [PubMed] [Google Scholar]
- 40.Lin J, Hu FB, Curhan G: Antioxidant vitamins and renal dysfunction in type 2 diabetes mellitus. J Am Soc Nephrol 16: 338A, 2006 [Google Scholar]
- 41.Nanayakkara PW, van Guldener C, ter Wee PM, Scheffer PG, van Ittersum FJ, Twisk JW, Teerlink T, van Dorp W, Stehouwer CD: Effect of a treatment strategy consisting of pravastatin, vitamin E, and homocysteine lowering on carotid intima-media thickness, endothelial function, and renal function in patients with mild to moderate chronic kidney disease: Results from the Anti-Oxidant Therapy in Chronic Renal Insufficiency (ATIC) Study. Arch Intern Med 167: 1262– 1270, 2007 [DOI] [PubMed] [Google Scholar]