Abstract
Purpose
19 To evaluate the presence of retinal nerve fiber layer (RNFL) defects in patients with XLRS using high speed, high resolution, Fourier domain OCT (FD-OCT).
Methods
Twenty four patients with XLRS seen by the authors were enrolled in the study. All patients underwent a complete eye examination. FD-OCT was performed using Optovue technology. A quadrant of the RNFL was considered to be thinned if at least 2 of the 4 segments in the quadrant were reduced in thickness.
Results
The average age of the 24 patients in the study was 28.8 years ± 14.7 years. Thinning of the RNFL in 1 quadrant was seen in 10 patients (41.7%), and thinning in 2 or more quadrants was seen in 8 patients (33.3%). Thinning in the inferior quadrant was most commonly seen and was observed in 12 patients (50%), followed by the temporal quadrant in 8 patients (33.3%), nasal quadrant in 4 patients (16.7%), and the superior quadrant in 4 patients (16.7%).
Conclusions
Among our 24 patients with XLRS, 15 patients (62.5%) showed a thinning of the RNFL in one or more quadrants in at least one eye and 9 patients (37.5%) in both eyes. The use of high speed, high resolution FD-OCT may be useful to determine the presence of possible changes in RNFL thickness in patients with XLRS. Reductions in RNFL thickness in such patients could be relevant in their selection for future therapeutic trials.
Keywords: X-linked retinoschisis, nerve fiber layer thickness, Fourier-domain OCT
Introduction
Juvenile X-linked retinoschisis (XLRS) was described as early as 1898 by Haas1 and several years later, it was identified by Mann and MacRae2 in 1938 to have an X-linked recessive inheritance. It occurs almost exclusively in males and is characterized by a progressive decrease of central visual acuity in the first years of life and the presence of foveal cystic, spoke wheel-like lesions at the macula apparent on fundus examination. A typical feature of this disease is a selective or predominant reduction of the scotopic b-wave amplitude (negative ERG) upon stimulation of the retina with a white-light single flash.3 However, Renner et al, reported that 40% of XLRS patients did not show a negative ERG.3
Mutations in the RS1 gene have been described in patients affected with this disease.4 The gene has six exons encoding for the retinoschisin polypeptide of 224 amino acids, which is synthesized in photoreceptor cell inner segments and provides cell adhesion and proper interaction between cells within the inner nuclear layer as well as synaptic connections between bipolar cells and photoreceptors.5-9 The mutant RS1 gene causes primarily the inner retinal layers to split. Histopathologic examinations suggest that the likely primary lesion in X-linked retinoschisis occurs initially in the innermost portion of Müller cells or at the border of the retinal nerve fiber layer.10-12
In patients with XLRS, mutations in the RS1 gene lead to the accumulation of retinoschisin within the inner retina13 and the development of cystic-like spaces primarily in the inner nuclear and outer plexiform layers of the retina observed by optical coherence tomography (OCT).14-16
A recent study by Apushkin et al suggested that reduction of visual acuity does not correspond to the size of cystic areas or retinal thickness of the macula as evaluated by time-domain optical coherence tomography (OCT).17 Gerth et al, using high-resolution OCT, concluded that there is disturbed foveal photoreceptor cell layer integrity in XLRS patients. Retinal layer abnormalities correlated with age but did not appear to correlate with visual acuity loss or genotypic variation.18
The purpose of the current study was to use high speed, high resolution Fourier domain OCT (FD-OCT) to evaluate the presence of any retinal nerve fiber layer (RNFL) defects in patients with XLRS.
Materials and methods
Participants
The study was conducted in the Department of Ophthalmology at the University of Illinois at Chicago. It followed the tenets of the Declaration of Helsinki and was approved by an institutional review board at the University of Illinois. Twenty four patients (43 eyes) with XLRS were included, and informed consent was obtained on all participants.
The diagnosis of XLRS was based on the history of a decrease in central vision within the first decade of life, selective or predominant reduction of the ERG b-wave in light and dark adapted responses and presence of characteristic fundus cystic lesions of the macula.
Patients with a diagnosis of XLRS who were seen by the authors (M.G. & G.A.F.) during their routine clinical follow up examinations were included in the present study if they were willing to undergo OCT testing. Additional patients previously seen by one of the authors (G.A.F.) were contacted by telephone and asked to participate in the study based on their prior diagnosis of XLRS.
Exclusion criteria included an inability to maintain steady fixation, past history of glaucoma and or increased intraocular pressure of ≥ 21 mmHg, pseudophakic and aphakic patients, posterior uveitis, diabetic retinopathy, optic neuropathies, spherical refraction of more than ± 6 diopters (D) or cylinder refraction of ± 2 D , or any central media opacity sufficient to hinder an OCT examination. No patients with a history of any systemic diseases such as hypertension or diabetes mellitus were included.
Ocular examination
All patients underwent a complete eye examination, including best-corrected visual acuity (BCVA) using an Early Treatment Diabetic Retinopathy Study (ETDRS) chart (The Lighthouse, Long Island City, NY), slit-lamp biomicroscopic examination of the anterior segment, and intraocular pressure (IOP) measurement with Goldmann applanation tonometry. A pachymetry measurement (Nidek Inc., Fremont, CA) was made if the IOP pressure was between 18-20 mmHg on two readings or the optic disc showed any degree of pallor. Both eyes were dilated with 2.5% phenylephrine and 1% tropicamide. Fundus examination was performed using both direct and indirect ophthalmoscopy as well as biomicroscopy with a noncontact 78 D lens and any optic disc pallor was recorded.
Optical coherence tomography (OCT) techniques
OCT was performed using Optovue technology (RTvue version 3.5; Optovue Inc., Fremont, CA). The NMH4 protocol was used for scan acquisition which consisted of 12 radial line scans, 3.4 mm in length and 6 concentric rings (2.5-4.0mm diameter), all centered on the optic disc. Internal fixation was used for all patients. The total time for a single scan acquisition was 0.39 seconds. Scans were accepted only if they had a signal strength index greater than 35 and were free of artifacts.
RNFL thickness was measured automatically by the existing software at a diameter of 3.45 mm around the center of the optic disc. The total number of A-scans at that circumference was 9510. RNFL thickness was measured in the superior (46°-135°), nasal (316°-45° for the right and 136°- 225° for the left), inferior (226°-315°), and temporal (136°- 225° for the right and 316°-45°for the left) quadrants. OCT software automatically measured 4 smaller segments within each quadrant (Figs 1-2). Data thus obtained were compared with the normative database provided with the software, taking the patient age and size of the optic disc into account.
Figure 1.
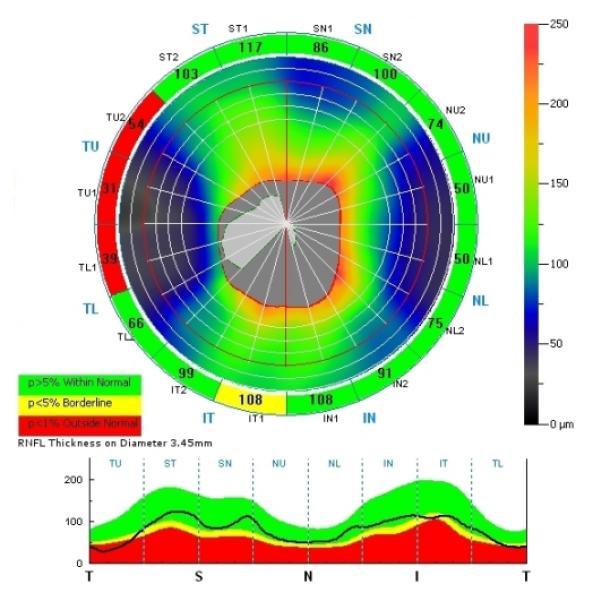
Abnormal thinning in 3 of 4 segments in the temporal quadrant of the right eye in a patient with XLRS. A single segment is abnormal in the inferior quadrant.
Figure 2.
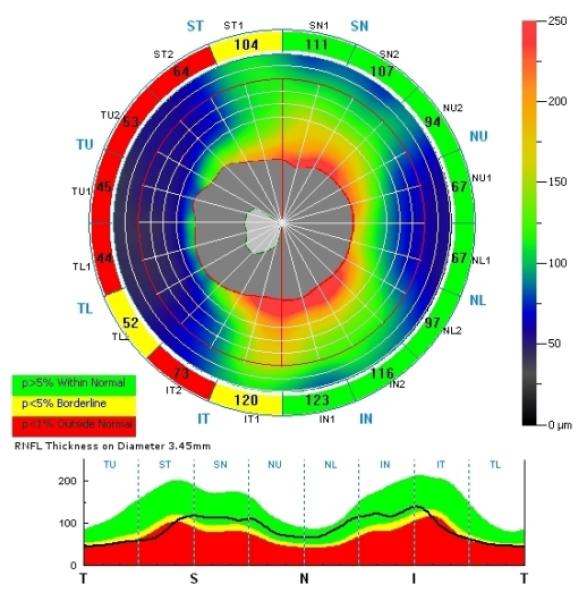
Thinning of the superior, inferior, and temporal quadrants of the RNFL in the right eye of a patient with XLRS. All 4 segments in the temporal quadrant are thin while 2 of 4 in the superior and inferior quadrants are thin.
RNFL thickness was measured at least 3 times in each eye and data were used only if the scans were reproducible, within either a normal or abnormal range, in at least 2/3 of the scan acquisitions. Also, eyes with scans inconsistent on the first set of 3 OCT RNFL scans were rescanned with another set of 3 scans. We then used the criteria that 2/3 of these scans should be consistent as either normal or abnormal in order to be included in the analysis. In 29 eyes (67.4%) only one set of scans was required, while in 14 eyes (32.6%) two sets of scans were required.
One hundred forty three OCT scans were obtained on 43 eyes (1 eye each was tested in 5 patients and both eyes were tested in 19 patients). Inner and outer automated machine drawn contour lines which mark the boundary of the nerve fiber layer were examined in each scan and scans with boundary line misplacements leading to distortion of the contour lines were excluded. The contour of the optic nerve head was manually traced using a fundus image generated by the OCT.
For detecting changes within the macula in the XLRS patients, we also scanned the macula using both the MM5 and the Radial Lines protocols. The radial scan consisted of twelve 6-mm radial scans at 15° intervals passing through the center of the fovea. All 12 scans were acquired simultaneously and the total time taken for their acquisition was 0.27 seconds. Each radial line consisted of 1024 A-scans.
The MM5 scan protocol consisted of 11 horizontal and 11 vertical line scans with a 5mm scan length and 0.5mm interval and 6 horizontal and 6 vertical line scans with a 3mm scan length and 0.5mm interval, all centered at the fovea, with a total acquisition time of 0.78 seconds.
Data analyses
- Segmental analysis with reference to color-coded normative data
The RNFL was considered abnormally thin for an individual if its value was less than the 5th percentile of the age- and optic disc size-adjusted normal values and thick if it was more than the 95th percentile. Abnormal thinning or thickness in at least 2 of the 4 segments within a quadrant, if reproducible, was considered significant for that quadrant.
- Quantitative analysis for individual quadrants
Additionally, we measured the thickness of the RNFL ± 2 SD quantitatively in each of four quadrants and compared these values with normative data provided to us by the company that was not corrected for age and optic disc size and which was retrieved from 268 eyes of 134 normative control subjects (mean age of 44.1 ± 15.5 years).
Statistical analysis
Differences in the mean of peripapillary RNFL thickness in our study population and that from the normative data were analyzed by the two-tailed Student t-test. A probability value of less than 0.05 was considered statistically significant. Since most of participants had a symmetrically equivalent RNFL thickness between eyes, the mean of RNFL thickness of each patient whose scans were included for both eyes was calculated as a single number, so that an analysis was performed in a by-person basis. Differences were studied for the thickness in overall circumference, hemispheres (superior and inferior), and quadrants (superior, nasal, inferior and temporal). The association between the presence of RNFL thinning (by segmental analysis) and age was also analyzed using Fisher’s Exact test in the by-eye analysis.
Results
Twenty four patients with the diagnosis of juvenile XLRS were included in our study. We included 43 eyes; 23 right eyes (53.5%) and 20 left eyes (46.5%). In 5 patients (20.8%), only one eye was included due to the presence of a traumatic cataract, rotatory nystagmus, old retinal detachment, or poor patient fixation which made the acquisition of a reliable RNFL scan difficult.
The average age of the 24 patients in the study was 28.8 years ± 14.7 years with a range from 10–54 years. There were 22 Caucasian (91.7%), and 2 Hispanic (8.3%) patients enrolled in the study. In 14 patients (58.3%), the abnormal disease causing gene was previously identified (Table 1).
Table 1.
Genetic mutation sequence in the studied patients
| Patient No./Age, y | XLRS1 genetic mutation |
OD/OS VA logMAR |
OD/OS Color vision on Ishihara test plates |
Macular schisis* |
Peripheral schisis** |
|---|---|---|---|---|---|
| 1/50 | His626Arg exon 6 residue 209 |
0.34 | 19/21 OD | + | − |
| 2/39 | Trp96Arg exon 4 | 0.40 / 0.20 | 21/21 OU | − | − |
| 3/32 | Trp96Arg exon 4 | 0.80 / 0.88 | 21/21 OU | + | + |
| 4/24 | Arg141His exon 5 | 0.58 / 0.42 | 21/21 OU | + | + (OD only) |
| 5/20 | Deletion of splicing acceptor site IVS4del ttCtcgg |
1.30 / 0.90 | 9/21-19/21 | + | + (OD only) |
| 6/15 | Large deletion in exon 1 |
0.38 / 0.28 | 21/21 OU | + | − |
| 7/38 | Gly70 Ser exon 4 | 0.52 / 0.48 | 20/21-21/21 | + | − |
| 8/14 | G to T at nucleotide 88 in exon 3 lead to premature stop codon |
0.36 / 0.40 | 21/21 OU | + | − |
| 9/32 | Gly70Ser exon 4 | 0.70 / 0.64 | 21/21 OU | + | + |
| 10/12 | Arg141His exon 5 | 0.30 / 0.88 | 20/21-7/21 | + | − |
| 11/10 | Arg141His exon 5 | 0.44 / 0.26 | 21/21 OU | + | − |
| 12/15 | Arg3ins1cG exon 1 | 0.10 / 0.76 | 4/21 OU | + | − |
| 13/12 | Arg3ins1cG exon 1 | 0.40 / 0.40 | 21/21 OU | + | + (OD only) |
| 14/27 | 20bp insertion exon 4 codon 74 |
0.30 / 0.30 | 18/21 OU | − | − |
Diagnosed by OCT and fundus exam
Diagnosed by Indirect Ophthalmoscopy
‘+’, presence; ‘-’, absence
The average best-corrected visual acuity in the right eyes was 0.50 logMAR units with a SD of 0.24 (range, 0.10-1.00), equivalent to 20/25 - 20/200. The average best-corrected visual acuity in the left eyes was 0.48 logMAR units with a SD of 0.24 (range, 0.04-1.00), equivalent to 20/22 – 20/200.
The range of IOP for all patients was from 8 to 17 mmHg in the right eyes (mean, 13.0 mmHg, and SD of 2.4), while the range of IOP in left eyes was from 10 to 17 mmHg (mean 12.8 mmHg, and SD of 2.2). None of the patients had a glaucomatous appearance to the optic disc. Three patients (14.3%) had mild temporal disc pallor clinically in both eyes. Two out of the three patients showed thinning in the RNFL thickness in the temporal quadrants, while the other patient showed thinning of the RNFL in the superior, nasal and inferior quadrants with normal RNFL thickness in the temporal quadrant.
Among our 24 patients with XLRS, 15 patients (62.5%) showed a thinning of the RNFL in one or more quadrants in at least one eye and 9 patients (37.5%) in both eyes. Thinning of the RNFL in 1 quadrant was seen in 12 eyes (27.9%) of 10 patients (41.7%), and thinning in 2 or more quadrants was seen in 12 eyes (27.9%) of 8 patients (33.3%).
Of note, the presence of RNFL thinning was present in 7 (87.5%) of 8 patients who were older than 35 years of age. All of the seven patients had thinning of RNFL in at least one quadrant in all studied eyes (5 patients whose both eyes were included, and 2 patients where only one eye was studied). For those who were younger than 35 years old, thinning of RNFL in at least one quadrant was observed in 9 (56.3%) of 16 patients. In those 9 patients, 7 patients were studied bilaterally, and 4 of the 7 patients showed thinning in only one eye. There was a statistically significant difference for the presence of RNFL thinning in at least one quadrant in the older (≥ 35 years old) compared to the younger age group (< 35 years old) (p = 0.0028). The significant difference was still demonstrated at the cut-off age of 30 years old (p = 0.0477).
Regarding the distribution for abnormal thinning of the RNFL among the 4 quadrants, thinning in the inferior quadrant was most commonly seen in 18 eyes (41.9%) of 12 patients (50%), followed by the temporal quadrant in 12 eyes (27.9%) of 8 patients (33.3%), nasal quadrant in 5 eyes (11.6%) of 4 patients (16.7%), and the superior quadrant in 5 eyes (11.6%) of 4 patients (16.7%) (Figs 1-2).
We also found that the overall average RNFL thickness at a diameter of 3.45mm in our study population to be 100.95 μm with a SD ± 16.02, compared to normative data, where the average RNFL thickness was 106.72 μm with a SD ±11.34. The patients with retinoschisis had a statistically significant difference of mean RNFL thickness compared to the normative data (p = 0.034).
By assessing the RNFL thickness quantatively in each quadrant, compared to the values of the mean RNFL thickness ± 2SD provided by the manufacturer, using the criteria of calculating the average RNFL thickness ± 2SD for each quadrant, we found that 14 eyes (32.6%) had statistically significant thinning of the RNFL in the inferior quadrant, while only 1 eye (2.3%) was found to have a thicker than normal RNFL in the inferior quadrant. We additionally found that 5 eyes (11.6%) had thinning of the RNFL in the temporal quadrant while 4 eyes (9.3%) were found to have a thicker than normal RNFL in the temporal quadrant. We were unable to find any RNFL thickness defects in either the superior or nasal quadrants by this quantitative method of analysis not accounting for age or optic disc size.
Further, we calculated the average RNFL thickness on all patients from all quadrants and compared these values to the normative values provided by the manufacturer uncorrected for age or optic disc size. As shown in Table 2, statistically significant differences in mean RNFL thickness were seen only in the inferior and temporal quadrants. There were no statistically significant differences in mean RNFL thickness in any quadrants, when comparing between the right and left eyes (p values ranged between 0.516-0.998)
Table 2.
Mean peripapillary RNFL thickness in each quadrant, compared to normative data
| Quadrant | RNFL thickness (Mean ± SD) in our patients |
RNFL thickness (Mean ± SD) from normative data |
P-value |
|---|---|---|---|
| Superior | 130.67 ± 19.54 | 129.03 ± 17.83 | 0.6831 |
| Nasal | 78.94 ± 13.51 | 78.43 ± 13.54 | 0.8652 |
| Inferior | 119.64 ± 26.20 | 139.78 ± 17.08 | <0.0001 |
| Temporal | 72.67 ± 16.30 | 79.63 ± 12.03 | 0.0149 |
The overall average for the RNFL thickness in the superior hemisphere was 105.69 μm (SD ± 15.82), while in the control group the average thickness was 106.59μm (SD± 12.68). In the inferior hemisphere the overall average was 95.43μm (SD ± 17.48), while the control group was 106.84μm (SD ± 11.53). There was no statistically significant difference shown in the superior hemisphere (p = 0.7586), but a statistical difference was demonstrated in the inferior hemisphere (p < 0.0001).
All patients included in the study were also examined by FD-OCT for the presence of cystic changes within the macula using the MM5 and Radial Lines examination protocols. Macular cysts were observed in 29 eyes (67.4%) from 18 patients (75.0%).
Discussion
In our cohort of 24 patients with XLRS, 15 patients (62.5%) showed a thinning of the RNFL in one or more quadrants in at least one eye by criteria referenced to color-coded normative data for segments within each quadrant. Abnormal thinning was more commonly found in the inferior and temporal quadrants. The nasal and the superior quadrants were equally affected and both showed thinning in fewer eyes than were seen in the inferior and temporal quadrants. A direct quantitative comparison between quadrants using data computed directly for each quadrant also confirmed a significant reduction of mean RNFL thickness in the inferior and temporal quadrants in our patients compared to normative data.
The segmental analysis for determining quadrant defects was based on normative data corrected for age and disc size provided by the manufacturer. Due to a proprietary limitation, the normal values from specific age groups were unavailable for our directly computed quantitative analysis of each quadrant. The normative data used herein were retrieved from visually normal subjects at a wide range of age. Therefore, the mean age of our study subjects was lower than that of the normative data. However, this limitation would not likely have affected our conclusions since we would more likely have anticipated a thicker RNFL in the younger patients had their RNFL not been affected by their disease.22 It is possible that we may have found even greater statistically significant results in our analysis if we could have used data from age-similar controls in this portion of our directly computed quadrant analysis.
The finding of changes in the RNFL of our XLRS patients might have been anticipated in reference to a report by Yanoff et al.,10 which stated that the primary pathology in XLRS was located in the inner retinal layers. Other previous studies based on histopathologic analysis suggested that the likely primary lesion in XLRS occurs initially in the innermost portion of Müller cells or at the border of the RNFL.10-12
Also, our finding of RNFL thinning is in agreement with previous studies by Manschot and Condon et al.,11-12 which described optic nerve atrophy in XLRS patients. Using a time-domain scanning procedure of the macula, Apushkin et al.,17 did not observe any thinning of the papillo-macular bundle RNFL thickness on OCT imaging in patients with juvenile XLRS and no clinical findings of optic disc pallor.
One of the current study’s potential limitations was that the drawing of the margins of the optic discs was done manually for all patients; hence, the size of the optic disc might have been subjectively influenced by the operator. Further, another parameter which might have influenced the RNFL thickness measured by OCT was the axial length of the eye,19-21 since we did not correct for this factor in the directly computed quadrant quantitative analysis portion of our study when using values for various quadrants compared to the manufacturer’s provided normative data that also did not correct for axial length. However, we only included patients with a lower degree of refractive error in whom a normal or close to normal axial length might be assumed.
Our study showed that older XLRS patients were more likely to show RNFL thinning which suggests that the younger patients might have a higher potential to benefit from treatment. This has potential significance for future treatment trials in patients with XLRS where patient selection might be influenced by the degree of thinning in their RNFL. However, this hypothesis needs to be confirmed in future treatment trials. Nonetheless, use of high-speed, high-resolution tools such as FD-OCT may be useful to measure RNFL thickness during patient selection in future therapeutic trials for patients with XLRS.
Acknowledgements
Supported by funds from the Foundation Fighting Blindness, Owings Mills, Maryland; Grant Healthcare Foundation, Lake Forest, Illinois; NIH core grant EYO1792; and an unrestricted departmental grant from Research to Prevent Blindness.
Footnotes
The authors have no proprietary interests in this work.
References
- 1.Haas J. Ueber das Zusammenvorkommen von Veränderungen der Retina und Chorioidea. Arch Augenheikd. 1898;37:343–348. [Google Scholar]
- 2.Mann I, MacRae A. Congenital vascular veils in the vitreous. Br J Ophthalmol. 1938;22:1–10. doi: 10.1136/bjo.22.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Renner AB, Kellner U, Fiebig B, Cropp E, Foerster MH, Weber BH. ERG variabilityin X-linked congenital retinoschisis patients with mutations in the RS1 gene and the diagnostic importance of fundus autofluorescence and OCT. Doc Ophthalmol. 2008;116:97–109. doi: 10.1007/s10633-007-9094-5. [DOI] [PubMed] [Google Scholar]
- 4.Sauer CG, Gehrig A, Warneke-Wittstock R, Marquardt A, Ewing CC, Gibson A, et al. Positional cloning of the gene associated with X-linked juvenile retinoschisis. Nat Genet. 1997;17:164–170. doi: 10.1038/ng1097-164. [DOI] [PubMed] [Google Scholar]
- 5.Grayson C, Reid SN, Ellis JA, Rutherford A, Sowden JC, Yates JR, et al. Retinoschisin, the X-linked retinoschisis protein,is a secreted photoreceptor protein, and is expressed and released by Weri-Rbs1 cells. Hum Mol Genet. 2000;9:1873–1879. doi: 10.1093/hmg/9.12.1873. [DOI] [PubMed] [Google Scholar]
- 6.Reid SN, Yamashita C, Farber DB. Retinoschisin, a photoreceptor-secreted protein, and its interaction with bipolar and Müller cells. J Neurosci. 2003;23:6030–6040. doi: 10.1523/JNEUROSCI.23-14-06030.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Molday LL, Hicks D, Sauer CG, Weber BH, Molday RS. Expression of the X-linked retinoschisis protein RS1 in photoreceptor and bipolar cells. Invest Ophthalmol Vis Sci. 2001;42:816–825. [PubMed] [Google Scholar]
- 8.Reid SN, Akhmedov NB, Piriev NI, Kozak CA, Danciger M, Farber DB. The mouse X-linked juvenile retinoschisis cDNA: expression in photoreceptors. Gene. 1999;227:257–266. doi: 10.1016/s0378-1119(98)00578-2. [DOI] [PubMed] [Google Scholar]
- 9.Weber BH, Schrewe H, Molday LL, Gehrig A, White KL, Seeliger MW, et al. Inactivation of the murine X-linked juvenile retinoschisis gene, Rs1h, suggests a role of retinoschisin in retinal cell layer organization and synaptic structure. Proc Natl Acad Sci USA. 2002;99:6222–6227. doi: 10.1073/pnas.092528599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yanoff M, Rahn EK, Zimmermann LE. Histopathology of juvenile retinoschisis. Arch Ophthalmol. 1968;79:49–53. doi: 10.1001/archopht.1968.03850040051014. [DOI] [PubMed] [Google Scholar]
- 11.Manschot WA. Pathologyof hereditaryjuvenile retinoschisis. Arch Ophthalmol. 1972;88:131–138. doi: 10.1001/archopht.1972.01000030133002. [DOI] [PubMed] [Google Scholar]
- 12.Condon GP, Brownstein S, Wang NS, Kearns AF, Ewing CC. Congenital hereditary (juvenile X-linked) retinoschisis. Histopathologic and ultrastructural findings in three eyes. Arch Ophthalmol. 1986;104:576–583. doi: 10.1001/archopht.1986.01050160132029. [DOI] [PubMed] [Google Scholar]
- 13.Kirsch LS, Brownstein S, de Wolff-Rouendaal D. A histopathological, ultrastructural and immunohistochemical study of congenital hereditary retinoschisis. Can J Ophthalmol. 1996;31:301–310. [PubMed] [Google Scholar]
- 14.Ozdemir H, Karacorlu S, Karacorlu M. Optical coherence tomography findings in familial foveal retinoschisis. Am J Ophthalmol. 2004;137:179–181. doi: 10.1016/s0002-9394(03)00736-0. [DOI] [PubMed] [Google Scholar]
- 15.Brucker AJ, Spaide RF, Gross N, Klancnik J, Noble K. Optical coherence tomography of X-linked retinoschisis. Retina. 2004;24:151–152. doi: 10.1097/00006982-200402000-00021. [DOI] [PubMed] [Google Scholar]
- 16.Azzolini C, Pierro L, Codenotti M, Brancato R. OCT images and surgeryof juvenile macular retinoschisis. Eur J Ophthalmol. 1997;7:196–200. doi: 10.1177/112067219700700214. [DOI] [PubMed] [Google Scholar]
- 17.Apushkin MA, Fishman GA, Janowicz MJ. Correlation of optical coherence tomography findings with visual acuity andmacular lesions in patients with X-linked retinoschisis. Ophthalmology. 2005;112:495–501. doi: 10.1016/j.ophtha.2004.08.027. [DOI] [PubMed] [Google Scholar]
- 18.Gerth C, Zawadzki RJ, Werner JS, Héon E. Retinal morphological changes of patients with X-linked retinoschisis evaluated by Fourier-domain optical coherence tomography. Arch Ophthalmol. 2008;126:807–811. doi: 10.1001/archopht.126.6.807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Budenz DL, Anderson DR, Varma R, Schuman J, Cantor L, Savell J, Greenfield DS, et al. Determinants of normal retinal nerve fiber layer thickness measured by Stratus OCT. Ophthalmology. 2007;114:1046–1052. doi: 10.1016/j.ophtha.2006.08.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bayraktar S, Bayraktar Z, Yilmaz OF. Influence of scan radius correction for ocular magnification and relationship between scan radius with retinal nerve fiber layer thickness measured byoptical coherence tomography. J Glaucoma. 2001;10:163–169. doi: 10.1097/00061198-200106000-00004. [DOI] [PubMed] [Google Scholar]
- 21.Sony P, Sihota R, Tewari HK, Venkatesh P, Singh R. Quantification of the retinal nerve fibre layer thickness in normal Indian eyes with optical coherence tomography. Indian J Ophthalmol. 2004;52:303–309. [PubMed] [Google Scholar]
- 22.Parikh RS, Parikh SR, Sekhar GC, Prabakaran S, Babu JG, Thomas R. Normal age-related decayofretinal nerve fiber layer thickness. Ophthalmology. 2007;114:921–926. doi: 10.1016/j.ophtha.2007.01.023. [DOI] [PubMed] [Google Scholar]


