Abstract
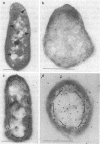
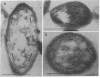
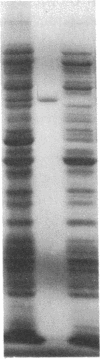
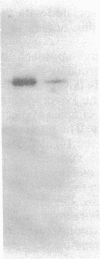
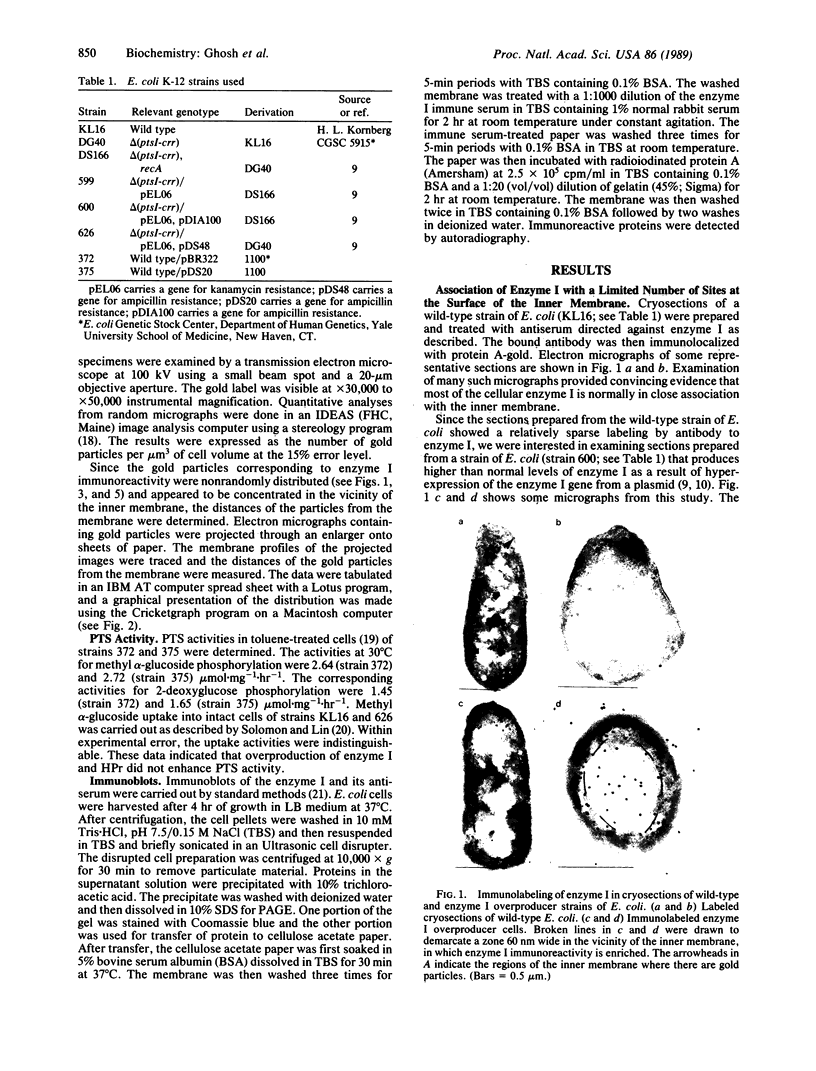
The phosphoenolpyruvate:sugar phosphotransferase system of Escherichia coli constitutes a major pathway for sugar translocation. It is composed of integral membrane proteins (enzyme II components) that recognize specific extracellular sugars as well as phosphocarrier proteins, one of which is called enzyme I. While enzyme I plays a role in energizing the enzyme II for sugar transfer, its precise cellular distribution had not previously been defined. This study was designed to elucidate the cellular location of this protein by immunoelectron microscopy. Enzyme I antibody bound to E. coli cryosections was visualized with protein A-gold. The gold particles in sections of wild-type E. coli were found primarily associated with the surface of the inner membrane. A strain of E. coli harboring a plasmid encoding the gene for enzyme I was also tested for its distribution of enzyme I. Consistent with the biochemically established overproduction of enzyme I, this strain showed an approximately 80-fold higher density of gold particles per unit cell volume than the wild-type cells. The substantial overproduction of immunoreactive enzyme I was associated with a significant (approximately 20-fold) increase in the amount of that protein bound to the inner membrane. In addition, a substantial fraction of the total enzyme I accumulated within a 60-nm-wide zone in the vicinity of the inner membrane. A model to explain the zonal distribution of enzyme I under conditions of overexpression of the protein is presented.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baty D., Knibiehler M., Verheij H., Pattus F., Shire D., Bernadac A., Lazdunski C. Site-directed mutagenesis of the COOH-terminal region of colicin A: effect on secretion and voltage-dependent channel activity. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1152–1156. doi: 10.1073/pnas.84.5.1152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danchin A. Membrane integration of carbohydrate transport in bacteria. Microbiol Sci. 1987 Sep;4(9):267–269. [PubMed] [Google Scholar]
- Gachelin G. A new assay of the phosphotransferase system in Escherichia coli. Biochem Biophys Res Commun. 1969 Feb 21;34(4):382–387. doi: 10.1016/0006-291x(69)90392-1. [DOI] [PubMed] [Google Scholar]
- Geli V., Baty D., Lazdunski C. Use of a foreign epitope as a "tag" for the localization of minor proteins within a cell: the case of the immunity protein to colicin A. Proc Natl Acad Sci U S A. 1988 Feb;85(3):689–693. doi: 10.1073/pnas.85.3.689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths G., McDowall A., Back R., Dubochet J. On the preparation of cryosections for immunocytochemistry. J Ultrastruct Res. 1984 Oct;89(1):65–78. doi: 10.1016/s0022-5320(84)80024-6. [DOI] [PubMed] [Google Scholar]
- Guan T., Ghosh A., Ghosh B. K. Immunoelectron microscopic double labeling of alkaline phosphatase and penicillinase with colloidal gold in frozen thin sections of Bacillus licheniformis 749/C. J Bacteriol. 1985 Oct;164(1):107–113. doi: 10.1128/jb.164.1.107-113.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harwood J. P., Gazdar C., Prasad C., Peterkofsky A., Curtis S. J., Epstein W. Involvement of the glucose enzymes II of the sugar phosphotransferase system in the regulation of adenylate cyclase by glucose in Escherichia coli. J Biol Chem. 1976 Apr 25;251(8):2462–2468. [PubMed] [Google Scholar]
- Kaback H. R. The role of the phosphoenolpyruvate-phosphotransferase system in the transport of sugars by isolated membrane preparations of Escherichia coli. J Biol Chem. 1968 Jul 10;243(13):3711–3724. [PubMed] [Google Scholar]
- Liberman E., Reddy P., Gazdar C., Peterkofsky A. The Escherichia coli adenylate cyclase complex. Stimulation by potassium and phosphate. J Biol Chem. 1985 Apr 10;260(7):4075–4081. [PubMed] [Google Scholar]
- Liberman E., Saffen D., Roseman S., Peterkofsky A. Inhibition of E. coli adenylate cyclase activity by inorganic orthophosphate is dependent on IIIglc of the phosphoenolpyruvate:glycose phosphotransferase system. Biochem Biophys Res Commun. 1986 Dec 30;141(3):1138–1144. doi: 10.1016/s0006-291x(86)80162-0. [DOI] [PubMed] [Google Scholar]
- Misset O., Robillard G. T. Escherichia coli phosphoenolpyruvate-dependent phosphotransferase system: mechanism of phosphoryl-group transfer from phosphoenolpyruvate to HPr. Biochemistry. 1982 Jun 22;21(13):3136–3142. doi: 10.1021/bi00256a016. [DOI] [PubMed] [Google Scholar]
- Postma P. W., Lengeler J. W. Phosphoenolpyruvate:carbohydrate phosphotransferase system of bacteria. Microbiol Rev. 1985 Sep;49(3):232–269. doi: 10.1128/mr.49.3.232-269.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy P., Meadow N., Roseman S., Peterkofsky A. Reconstitution of regulatory properties of adenylate cyclase in Escherichia coli extracts. Proc Natl Acad Sci U S A. 1985 Dec;82(24):8300–8304. doi: 10.1073/pnas.82.24.8300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy A., Danchin A. The cya locus of Escherichia coli K12: organization and gene products. Mol Gen Genet. 1982;188(3):465–471. doi: 10.1007/BF00330050. [DOI] [PubMed] [Google Scholar]
- Saffen D. W., Presper K. A., Doering T. L., Roseman S. Sugar transport by the bacterial phosphotransferase system. Molecular cloning and structural analysis of the Escherichia coli ptsH, ptsI, and crr genes. J Biol Chem. 1987 Nov 25;262(33):16241–16253. [PubMed] [Google Scholar]
- Saier M. H., Jr, Cox D. F., Feucht B. U., Novotny M. J. Evidence for the functional association of enzyme I and HPr of the phosphoenolpyruvate-sugar phosphotransferase system with the membrane in sealed vesicles of Escherichia coli. J Cell Biochem. 1982;18(2):231–238. doi: 10.1002/jcb.1982.240180210. [DOI] [PubMed] [Google Scholar]
- Solomon E., Lin E. C. Mutations affecting the dissimilation of mannitol by Escherichia coli K-12. J Bacteriol. 1972 Aug;111(2):566–574. doi: 10.1128/jb.111.2.566-574.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tinglu G., Ghosh A., Ghosh B. K. Subcellular localization of alkaline phosphatase in Bacillus licheniformis 749/C by immunoelectron microscopy with colloidal gold. J Bacteriol. 1984 Aug;159(2):668–677. doi: 10.1128/jb.159.2.668-677.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsang V. C., Peralta J. M., Simons A. R. Enzyme-linked immunoelectrotransfer blot techniques (EITB) for studying the specificities of antigens and antibodies separated by gel electrophoresis. Methods Enzymol. 1983;92:377–391. doi: 10.1016/0076-6879(83)92032-3. [DOI] [PubMed] [Google Scholar]
- Weigel N., Waygood E. B., Kukuruzinska M. A., Nakazawa A., Roseman S. Sugar transport by the bacterial phosphotransferase system. Isolation and characterization of enzyme I from Salmonella typhimurium. J Biol Chem. 1982 Dec 10;257(23):14461–14469. [PubMed] [Google Scholar]