Abstract
Little is known about the architecture of cellular microenvironments that support stem and precursor cells during tissue development. Although adult stem cell niches are organized by specialized supporting cells, in the developing cerebral cortex, neural stem/precursor cells reside in a neurogenic niche lacking distinct supporting cells. Here we find that neural precursors themselves comprise the niche and regulate their own development. Precursor-precursor contact regulates β-catenin signaling and cell fate. In vivo knockdown of N-cadherin reduces β-catenin signaling, migration from the niche and neuronal differentiation in vivo. N-cadherin engagement activates β-catenin signaling via Akt, suggesting a mechanism through which cells in tissues can regulate their development. These results suggest that neural precursor cell interactions can generate a self-supportive niche to regulate their own number.
Keywords: Neural precursor, wnt, beta catenin, n-cadherin, niche, cerebral cortex, ventricular zone
Specialized microenvironments or niches coordinate the specification, self-renewal and differentiation of stem cells (Fuchs et al., 2004). Adult stem cell niches are distinct anatomical structures organized by specialized cells that create a supportive habitat for stem cells (Moore and Lemischka, 2006). In contrast, whether similar microenvironment structures regulate stem and precursor cells in developing tissues remains poorly understood (Alvarez-Buylla and Lim, 2004).
In the developing cerebral cortex, neural precursors reside in the ventricular zone (VZ), a neurogenic layer of epithelial cells that lines the neural tube. Residence in the VZ appears to sustain the proliferation of neural precursor cells, as when neural precursors are removed from the VZ, nearly all exit the cell cycle and differentiate (Temple, 2001). The observation that the VZ, unlike adult stem cell niches, consists almost exclusively of dividing precursors (Takahashi et al., 1993) suggested the possibility that the cortical precursors themselves could provide their own supportive environment in development.
Physical contact between stem cell and niche appears crucial to stem cell self-renewal. Recent studies indicate that adhesion mediated by adherens junctions anchor germline stem cells to the Drosophila ovary stem cell niche (Song et al., 2002), while similar adhesion complexes have been described between hematopoietic stem cells and their niche in the bone marrow (Zhang et al., 2003). In contrast, instead of adhering to specialized supporting cells, cerebral cortical neural precursors link to adjacent precursors through adherens junctions (Chenn et al., 1998). While adherens junctions appear to physically anchor stem cells to their niches and cortical neural precursors to each other, how adherens junctions might regulate signaling pathways that control stem cell renewal remains poorly understood. Here, we examine the role of the adherens junctions proteins N-cadherin and β-catenin in the neurogenic niche of the developing cerebral cortex.
Results
β-catenin Signaling Characterizes the Developing Cortical Neural Precursor Niche
During cortical development, β-catenin signaling can regulate neural precursor proliferation and differentiation (Chenn and Walsh, 2002; Hirabayashi et al., 2004; Woodhead et al., 2006). To examine the relationship of β-catenin signaling and the microenvironment of developing neural precursors, we first characterized endogenous β-catenin signaling in the developing cortex. In situ hybridization for β-gal in the BAT-Gal transgenic reporter mouse line (Maretto et al., 2003), and GFP expression in the Axin2-dGFP transgenic reporter mice (Jho et al., 2002) revealed β-catenin activity in the cortical ventricular zone (VZ) of the developing dorsal telencephalon in a medial high to lateral low gradient, while absent from ventral structures(Fig. 1A, B).
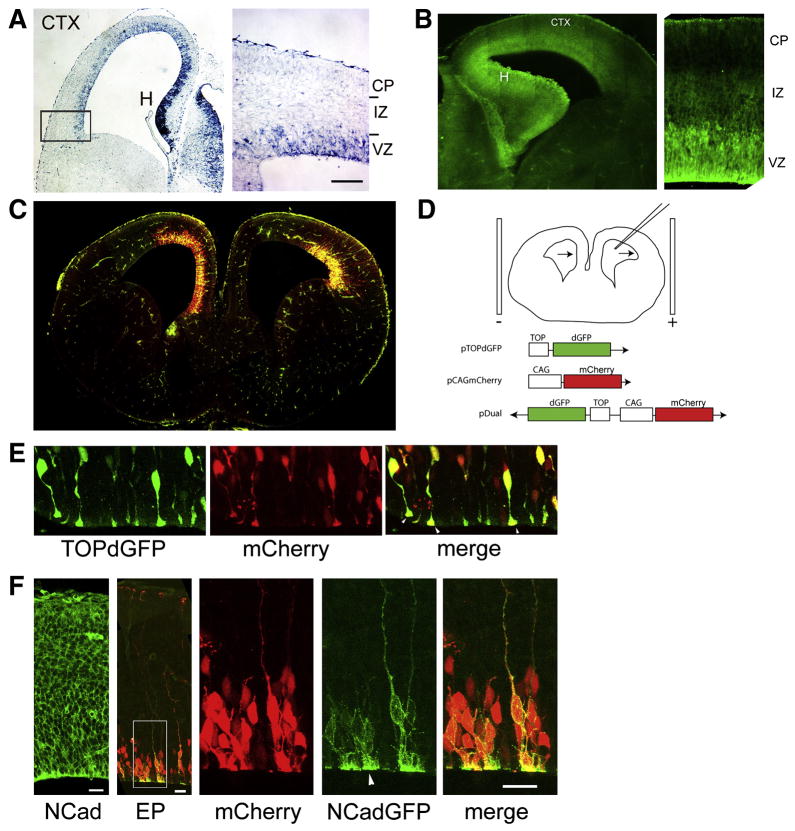
Figure 1. Relationship of N-cadherin expression and β-catenin signaling in cortical precursors.
(A)In situ hybridization for β-galactosidase transcript from transgenic BAT-Gal reporter mouse (E14.5 cortex). (B) Expression of GFP in cortex of Axin2-dGFP reporter mouse.. Higher power confocal images shows enrichment of β-gal and GFP signal in VZ, with reduced expression in IZ and CP. (C) β-catenin signaling revealed by co-electroporation of TOPdGFP reporter construct (green) and mCherry (red) of E14.5 brain. Low power image reveals that electroporated regions of lateral, dorsal, and medial cortical VZ (highlighted by co-electroporation of mCherry) are characterized by β-catenin signaling (green). (D) Schematic of electroporation protocol used in (C): DNA is injected into lateral ventricle and is introduced into the adjacent neuroepithelial cells directed towards the positive electrode, aligned outside the brain as shown. Cartoons of reporter constructs used in these experiments shown below. (E) Dual TOPdGFP/mCherry reporter (Figs. 1D, S1) highlights β-catenin signaling in precursor cell bodies and apical foot processes (arrowheads). Bar = 20 μm. (F) Antibody staining for N-cadherin in the E13.5 mouse cortical plate reveals staining throughout the developing cortical plate. Acute analysis of co-electroporation with GFP-tagged N-cadherin (right panels) and red fluorescent protein reveals N-cadherin labels VZ precursor cell membranes and is enriched at apical junctions (arrowhead) 14 hours after electroporation. Bars = 20 μm.
To examine at β-catenin signaling at a cellular level, we used a β-catenin reporter construct that expresses a destabilized green fluorescent protein (GFP) variant under the control of a well-characterized β-catenin responsive promoter, TOPdGFP (Dorsky et al., 2002), and confirmed that cells in the VZ throughout the lateral, dorsal, and medial cortical walls showed expression of destabilized GFP (Fig. 1C). Electroporation of a modified construct containing both mCherry and TOPdGFP (pDual, Fig. 1D) on a single plasmid revealed that signaling neural precursors had prominent apical endfeet connecting the cell body to the ventricular lumen (Fig. 1E).
This observation supported the possibility that components found at the apical endfeet such as N-cadherin might play important roles in β-catenin signaling in vivo. In utero electroporation of an expression construct driving an N-cadherin-GFP fusion revealed the subcellular distribution of newly-expressed (14 hours after electroporation) N-cadherin in individual precursors enriched at the apical endfeet, consistent with localization at adherens junctions (Fig. 1F, arrowhead). Together, these observations reveal 1) the presence of β-catenin signaling in the developing cortical VZ, and 2) co-expression of N-cadherin with β-catenin signaling in the VZ.
N-cadherin Regulates β-catenin Signaling in Cortical Precursors
Previous studies have shown that β-catenin signaling in VZ precursors regulates precursor number by influencing cell-cycle exit (Chenn and Walsh, 2002; Woodhead et al., 2006). What regulates β-catenin signaling in the ventricular zone niche? The observation that neural precursor contact with other precursors stimulates proliferation (Temple and Davis, 1994) suggested to us the possibility that cell contact in the niche might promote β-catenin signaling. To test whether neural precursor cell contact regulates β-catenin signaling, we examined the activity of the β-catenin-responsive reporter construct pSuper8TOPFLASH (DasGupta et al., 2005) in primary cortical cultures at varying cell densities and observed that high cortical cell density upregulates β-catenin signaling (Fig. 2A). Western blot analysis confirmed that increased β-catenin signaling was accompanied by increased active (unphosphorylated) β-catenin (van Noort et al., 2002) with higher cell densities (Fig. 2B). β-catenin signaling in low density cells was not restored by co-culture with a high density of cells separated by a Transwell compartment (Shen et al., 2004) despite an equivalent overall density of cells in the culture, suggesting that cell contact or close proximity is necessary to stimulate signaling (Fig. 2C).
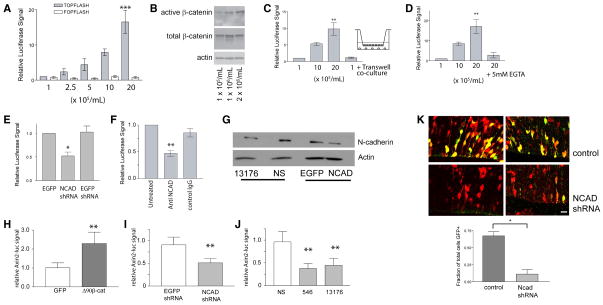
Figure 2. N-cadherin maintains β-catenin signaling in cortical precursors.
(A)β-catenin reporter assays in E14.5 rat primary cortical cultures transfected with pSUPER8TOPFLASH, with fold induction relative to signal from lowest cell density condition. Cell densities of 1, 2.5, 5, 10, and 20 × 105/ml represent 2.5, 6.25, 12.5, 25, and 50 X 104/cm2, respectively. (A) P< 0.001 by ANOVA; N = 11 experiments (1, 10, 20 × 105/ml), N = 2 experiments (2.5, 5 × 105/ml). ***P <0.001 for 20 × 105/ml vs. all other densities except P<0.01 vs. 5 × 105 condition. Neuman Keuls Post-hoc test. (B) High cell density increases the amount of dephosphorylated β-catenin. (C) Signaling is not stimulated in cultures of 1 × 105/ml cells with 2 × 106/ml co-cultured above in a Transwell chamber. P = 0.0016, N = 3 experiments; all pairwise comparisons P<0.05 except **P<0.01 for 2 × 106/ml vs. 1 × 105/ml or 1 × 105/ml +co-culture conditions, and P>0.05 1 × 105/ml vs. 1 × 105/ml + co-culture. (D) β-catenin signaling at high cell densities requires extracellular calcium. P=0.0041 N = 3 experiments. **P<0.01 2× 106/ml vs. 2× 10 6/ml + 5 mM EGTA; P >0.05 for 1 × 105/ml vs. 2× 106/ml + 5 mM EGTA. (E) shRNA to N-cadherin reduces β-catenin signaling. P= 0.0226. N = 3 experiments. * indicates P < 0.05 for NCAD shRNA vs. EGFP or shRNA against EGFP. (F) Function-blocking antibody to N-cadherin inhibits β-catenin signaling. P=0.0036. N = 3 experiments. ** indicates P < 0.01 for function blocking antibody vs. untreated or control IgG. (G) shRNA to N-cadherin reduces N-cadherin protein levels; shRNA constructs (NCAD, from (Maeda et. al, 2005) vs. shRNA against EGFP (middle); or clone 13176 from Open Biosystems vs. corresponding nonsilencing shRNA control) in primary neural precursors. Western blots performed on E13.5 mouse primary cortical precursors nucleofected with either control or N-cadherin knockdown shRNA constructs, and cultured for 24 hours before lysing cells. Western blots repeated three independent replicates with similar results. (H) Axin2 Luciferase Reporter is regulated identically as SUPER8TOPFLASH. E14.5 primary cortical cultures at 2×106/ml transfected with Axin2 Luciferase reporter show activation of reporter by stabilized β-catenin (Δ90β-catenin vs. GFP) (H), showing responsiveness of this reporter to β-catenin signaling. Three distinct shRNA constructs (NCAD, from (Maeda et. al, 2005) vs. shRNA against EGFP (I), or clones 546 and 13176 from Open Biosystems vs. corresponding nonsilencing shRNA control) show that knockdown of N-cadherin reduce Axin2 reporter signal (J). (H, I) ** indicates P < 0.01 by Students T-test. (J) P< 0.036 by repeated measures ANOVA; ** indicates P <0.01 vs. non-silencing shRNA. (K) Control E14.5 mouse forebrain co-electroporated with TOPdGFP (green), mCherry (red) and non-silencing shRNA construct. Green (yellow upon merge) represents activation of β-catenin signaling. Co-electroporation of TOPdGFP, mCherry, and shRNA to N-cadherin results in reduction of β-catenin signaling. * P = 0.0244 by Student’s T test; n = 4 brains. Repeated Measures ANOVA used for comparisons in C–F, J, with Neuman Keuls post hoc tests. Apical surface and lumen of lateral ventricle is down in all images; Bar = 200 μm (A, B), 20μm.
Because many factors might also regulate density dependent changes in signaling in neural precursors, we decided to examine the specific role of N-cadherin in β-catenin signaling in high-density cultures of primary neural precursors. We found that blocking N-cadherin function with 5mM EGTA (Fig. 2D), shRNA (Fig. 2E, G), or function-blocking N-cadherin antibody (Fig. 2F) reduced β-catenin signaling. Finally, we also examined the regulation of Axin2, an endogenous target of β-catenin signaling, using an Axin2 reporter construct driving luciferase (Jho et al., 2002). Co-transfection of this reporter construct along with three different shRNA’s against N-cadherin (vs. their respective controls) confirmed the findings that N-cadherin knockdown reduced β-catenin-mediated signaling (Fig. 2H–J).
To examine the requirement for N-cadherin in β-catenin signaling in vivo, we performed loss of function studies using in utero electroporation of short hairpin constructs targeting N-cadherin. Co-electroporation of the TOPdGFP reporter construct with the N-cadherin shRNA knockdown construct revealed that β-catenin signaling was reduced by N-cadherin knockdown (Fig. 2K, S2). Together, these studies (calcium dependence, function-blocking antibody, shRNA knockdown, and Axin2 responsiveness) suggest that the endogenous activation of β-catenin by neural precursor cell contact utilizes N-cadherin.
N-cadherin Knockdown In Vivo Causes Premature Migration, and Neuronal Differentiation
To examine the effects of reducing N-cadherin on neural precursor cell fate in vivo, we examined the migration and differentiation of cells following in utero electroporation of shRNA against N-cadherin. We observed that reduction of N-cadherin resulted in increased cell migration from the VZ (Fig. 3A–D), premature neuronal differentiation (Fig. 3A–C, E), and increased cell cycle exit (Fig. 3F, G). We found that restoring β-catenin signaling by introducing a stabilized, active form of the protein (Δ90β-catenin) along with N-cadherin shRNA could rescue the proliferative phenotype (Fig 3F, G). We found no evidence that N-cadherin reduction led to non-cell-autonomous effects on cell proliferation when non-electroporated cells were analyzed (Fig. S3). Together, the findings that N-cadherin reduction leads to reduction of β-catenin signaling, increased neuronal differentiation, and increased cell-cycle exit, suggest that N-cadherin interactions between precursors serve to maintain precursor self-renewal by maintaining β-catenin signaling.
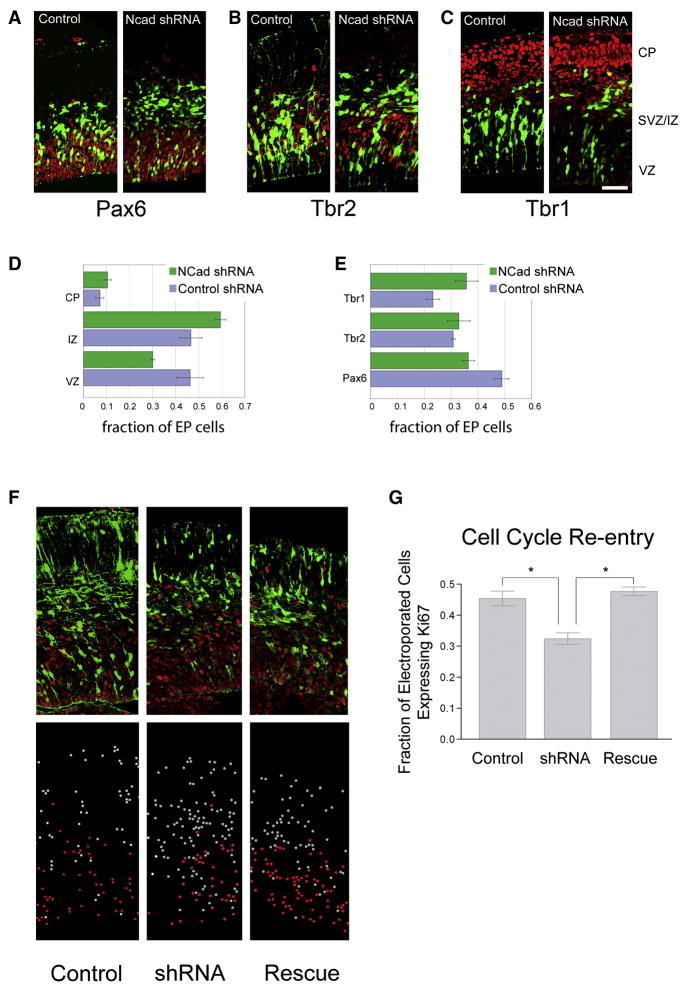
Figure 3. N-cadherin knockdown increases neuronal differentiation and cell cycle exit in vivo.
(A-C) E13.5 mouse forebrain co-electroporated in utero with pCAG-GFP and 4-fold (by mass) excess of non-silencing shRNA construct (control), or shRNA targeting N-cadherin (Ncad shRNA). Sections of electroporated brains stained for radial glial marker Pax6 (A), intermediate progenitor marker Tbr2 (B), and early postmitotic neuronal marker Tbr1 (C). Electroporated cells are green, and the respective antigens, red. Bar = 50 μm. Cell histograms represent mean fraction of total electroporated cells found in each brain region, showing increased exit from the VZ (D) or the fraction of cells that express each marker after electroporation, showing premature neuronal differentiation (E). For distribution, shRNA vs. control χ2[2]= 58.4, p < 0.0001 (control n = 4 brains, 1060 cells; shRNA n = 3 brains, 527 cells). For cell identity, shRNA vs. control χ2[2]= 172, p < 0.001 (control n = 2 brains, 1887 cells; shRNA n = 4 brains, 2226 cells). Error bars = 1 SEM. (F, G) N-cadherin knockdown reduces cell cycle re-entry and is rescued by stabilized β-catenin. E13.5 embryos electroporated with control non-silencing shRNA, N-cadherin shRNA, or N-cadherin shRNA and stabilized β-catenin (Δ90-βcatenin-GFP). Electroporated cells are labeled green and Ki67 staining red. Bottom images in (F) show the visual representation of the individual cells where all cells targeted by electroporation that also express Ki67 are labeled red, while electroporated cells that do not express Ki67 are white. (G) Histogram displaying the proportion of electroporated cells expressing Ki67 +/− SEM. (n = 3 brains each, 2490 cells total). P = 0.0028 by Repeated Measures ANOVA; **P < 0.01 by Neuman Keuls post-hoc test.
N-cadherin Regulates β-catenin Signaling Through Akt Activation
How does N-cadherin engagement mediate β-catenin signaling? We observed that N-cadherin shRNA, function-blocking antibodies (Fig. 2), and inhibitors of canonical Wnt signaling (Fig. S4A) could all reduce endogenous β-catenin signaling. However, blocking N-cadherin did not inhibit Wnt-stimulated activation of β-catenin (Fig. S4B), suggesting that N-cadherin function in β-catenin signaling might utilize an alternative non-Wnt-mediated mechanism. Increasing evidence suggests that specific post-translational modifications (such as specific phosphorylation) of β-catenin can contribute to further regulation of its activity. In intestinal stem cells, β-catenin phosphorylation at Ser 552 (via AKT) leads to its stabilization and nuclear localization (He et al., 2004). As N-cadherin adhesion can lead to activation of the phosphatidylinositol 3-kinase/AKT pathway (Tran et al., 2002), we reasoned that this pathway might contribute to the N-cadherin-dependent activation of β-catenin we observed in neural precursors.
Using an antibody that recognizes β-catenin phosphorylated at Ser 552, we observed that β-catenin Ser552-P was found in dividing neural precursors in the VZ (Fig. 4A). Co-staining with the mitotic marker phosphorylated histone H3 (pHH3) revealed that the β-catenin Ser552-P expressing cells were mitotic cells (Fig. 4A). These findings suggested that N-cadherin could activate β-catenin via AKT-mediated phosphorylation at Ser 552.
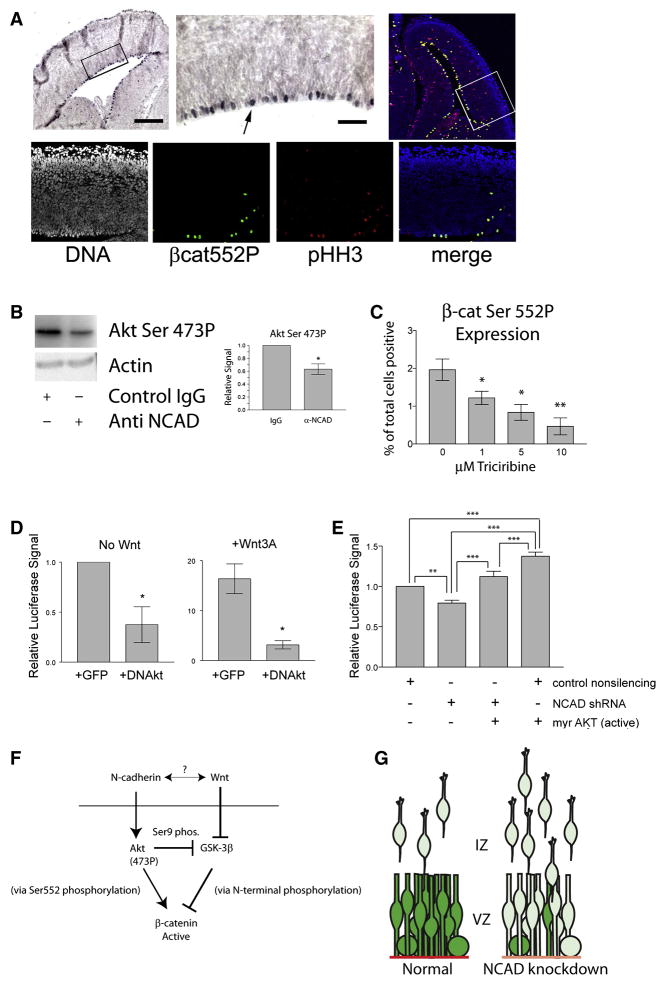
Figure 4. β-catenin Ser 552 P in the developing ventricular zone and interactions of N-cadherin, Akt, and β-catenin signaling.
(A) Staining (purple) for β-catenin Ser552P shows nuclear staining in mitotic cells along the ventricle (higher magnification of inset shown). Immunofluorescence reveals β-catenin Ser552P-expressing cells (green) co-stain for the mitotic marker phosphorylated Histone H3 (pHH3, red; co-stained cells, yellow). Higher magnification confocal optical sections of inset area (bottom) confirm co-localization. Bar = 200 μm and 20 μm. (B) Inhibition of N-cadherin engagement with function-blocking antibody on primary neural precursors leads to reduction of Ser473P (active) Akt by western blot; quantitation, right. N = 3, P = 0.0467 by Paired T test. (C) Inhibition of Akt activity with Triciribine reduces number of β-catenin Ser552P in primary cortical precursor culture at 24 hr; N = 3; P= 0.0094 by Repeated Measures ANOVA; * P < 0.05; ** P < 0.01 vs. untreated, Neuman Keuls post-hoc. (D) Dominant negative (kinase dead, HA-Akt-K179M) Akt inhibits endogenous (N = 4; P = 0.0402) and Wnt-stimulated β-catenin signaling (N = 4; P = 0.0284) in TOPFlash reporter assay (Paired T test). (E) Inhibition of β-catenin signaling by N-cadherin shRNA is rescued by myristoylated (active) Akt. N = 3; P<0.0001 by Repeated Measures ANOVA; ** P < 0.01; *** P < 0.001 by Neuman-Keuls post hoc. In D and E, luciferase signal from pSUPER8TOPFLASH is normalized to cotransfected internal control pSUPER8FOPrenilla, containing mutated LEF-1/TCF-1 binding sites and is unresponsive to β-catenin signaling (as in Figure 2; see also Figure S1). (F) Illustration of the interaction of the N-cadherin and Wnt regulation of β-catenin signaling. N-cadherin engagement activates Akt via phosphorylation at Ser473. Active Akt can lead to increased β-catenin stability indirectly by Ser 9 phosphorylation and inhibition of GSK3β or directly by phosphorylation of β-catenin at Ser 552. Wnt signaling leads to inactivation of GSK3β leading to the accumulation of N-terminally unphosphorylated β-catenin. Wnts may also regulate N-cadherin levels (Toyofuku et al., 2000; Tufan and Tuan, 2001). N-cadherin engagement may also facilitate canonical Wnt signaling by increasing the physical association of neural precursor cells. (G) Model of the VZ niche in development. VZ precursors exhibit β-catenin signaling (green) and are joined to each other via N-cadherin adherens junctions (red). Following N-cadherin reduction, β-catenin signaling is also reduced, causing increased neuronal differentiation and migration into the intermediate zone (IZ) towards the cortical plate.
We investigated whether Akt might link N-cadherin to β-catenin activation in cortical precursors. We found that function-blocking antibodies to N-cadherin (Fig. 4B) or shRNA to N-cadherin (Fig. S4C, D) led to a significant reduction in phosphorylated (active) Akt in primary cortical precursors. To test the link between Akt activation and phosphorylation of β-catenin at Ser552, we inhibited Akt in neural precursors using triciribine (API-2), a small molecule Akt pathway inhibitor (Yang et al., 2004). Triciribine treatment of primary cortical precursors reduced the fraction of cells expressing β-catenin Ser552 in a dose-dependent fashion (Fig. 4C). Finally, expression of a dominant negative (kinase-dead) Akt also reduced both baseline β-catenin signaling in high-density primary cortical precursor cultures as well as Wnt-stimulated β-catenin signaling (Fig. 4D). To confirm whether Akt functions downstream of N-cadherin to mediate β-catenin signaling, we co-expressed myristoylated (active) Akt along with shRNA to N-cadherin and measured β-catenin signaling by TOP-flash reporters. We found that that myrAkt rescued β-catenin signaling following N-cadherin knockdown (Fig. 4E). We also found that myrAkt alone could increase β-catenin signaling, a finding consistent with the idea that this pathway may exist in parallel with the canonical Wnt signaling pathway. Together, these observations suggest that N-cadherin engagement leads to phosphorylation of Akt and subsequent Akt-mediated phosphoryation and activation of β-catenin.
Discussion
Here, we demonstrate 1) a type of niche regulation where neural precursor cells generate their own self-supportive niche, and 2) a mechanism where N-cadherin engagement activates beta catenin signaling through Akt in neural precursors. Our observations suggest that neural precursors comprise a niche in which the precursor cells themselves function as supporting cells to sustain their own normal development. Furthermore, our findings provide evidence that neural stem/precursor cell self-renewal is promoted by signals produced by the cells themselves and suggest that N-cadherin is a crucial mediator of precursor-precursor signaling. Inhibition of N-cadherin leads to reduction of β-catenin signaling, premature neuronal differentiation, cell cycle exit, and increased migration towards the developing cortical plate.
Although it has been suggested that cell contact and cadherin stabilization leads to reduction of β-catenin signaling by titration of cytoplasmic β-catenin (Nelson and Nusse, 2004), we found that high cortical cell density surprisingly upregulates β-catenin signaling, in a fashion requiring N-cadherin. Our studies suggest a model in which N-cadherin engagement leads to Akt activation. Akt-dependent phosphorylation of β-catenin at Ser552 results in stabilization of β-catenin and increased transcriptional activation (Fig. 4F).
Our observations that N-cadherin regulates maintenance of precursors in the niche support studies of other stem cell types (Zhang et al., 2003) suggesting N-cadherin mediates attachment of stem cells to their niches. However, the developing cortical VZ is unusual amongst most well-characterized stem/progenitor niches as it consists not of specialized supporting cells, but of other precursor cells. Instead of facilitating interactions between stem cells and supporting cells, N-cadherin in the VZ mediates interactions between proliferating precursors with each other. While our studies support findings that cell-cell contact in tissues regulates key signaling pathways during differentiation by mediating a “community effect” (Gurdon, 1988), adult stem cell niches are characterized by distinct supporting cells and microenvironmental structures (Fuchs et al., 2004; Moore and Lemischka, 2006). Although whether adult stem cell self-interactions also have self-supportive function or share molecular mechanisms is not known, high local cell density of human embryonic stem cells promotes self-renewal and inhibits differentiation (Peerani et al., 2007).
Other factors that regulate neural precursors may function similarly; endothelial factors that promote neural precursor self-renewal lead to increased precursor cell contact and increased β-catenin levels (Shen et al., 2004), and Numb and Numblike proteins can regulate N-cadherin function (Rasin et al., 2007). Although disruption of epithelial integrity and cell polarity can cause hyperproliferation (Bilder et al., 2000), leading to suggestions that cell contact negatively regulates proliferation (Lien et al., 2006), our findings that N-cadherin can regulate cell fate and signaling even in areas without tissue disorganization suggest a more complex regulation of proliferation and cell fate by cell adhesion molecules. Our studies support recent findings suggesting that disruptions of adherens junctions and cell polarity in VZ progenitors lead to premature differentiation (Cappello et al., 2006; Yokota et al., 2009). Alterations in N-cadherin signaling may mediate the alterations in cell fate observed when adherens junctions are disrupted and cell polarity is lost. In addition to cell-cell adhesion, cell to extracellular matrix adhesion also appears critical in maintaining VZ architecture (Loulier et al., 2009). Further understanding of how precursor cells integrate signals from cell density and their microenvironment will lend insight into the mechanisms that underlie the growth of cells during development as well as the regulation of tumor growth.
Experimental Procedures
Statistical Analysis
Statistical analysis was performed with Graphpad Prism, with error bars on all graphs representing 1 SEM.
In Utero Electroporation
In utero electroporation and cell analysis performed as in (Woodhead et al., 2006); (plasmid and antibody information listed in supplemental information). For in vivo TOP-d-GFP signaling studies, co-electroporation with DN-TCF4 and ICAT confirmed specificity (Woodhead et al., 2006).
Cell Culture and Luciferase Assays
Primary cortical cultures and luciferase assays performed as described in (Noles and Chenn, 2007). For co-culture assays, 1 X 106 cells were placed onto 6.5 mm transwell membrane inserts with 0.4 μm pore size (Costar) above freshly plated and transfected cells at 1 X 105/ml (2.5 X 104/cm2) as in (Shen et al., 2004). For function blocking antibodies, cultures at high density [2 × 106/ml (0.5 X 106/cm2)] were incubated with 50 μg/ml N-cadherin function blocking antibody (De Wever et al., 2004; Makrigiannakis et al., 1999; Wallerand et al., 2008) (ACAM, GC-4, Sigma) or control IgG1κ isotype control (BD Pharmingen cat. 554721) 5 hours after transfection of reporter constructs. For protein analysis, 5 million primary cortical precursors were transfected by AMAXA Nucleofection (Amaxa Biosystems, Gaithersburg, MD) following manufacturer protocols. For triciribine treatment primary cortical cells were plated at a density of 2 million/ml, immediately treated with 0, 1, 5, or 10 μM triciribine (EMD), for 24 hr.
Cell-cycle re-entry studies
E13.5 embryonic cortices were electroporated with Ncad-shRNA or control shRNA, together with pCAG-EGFP as control. For the rescue experiment, Ncad-shRNA, pCAG-Δ90β-catenin-GFP or pCAG-EGFP was electroporated. After 48 hours, the E15.5 embryonic brains were dissected, fixed, cryosectioned, and stained for GFP and ki67 as in (Stocker and Chenn, 2008).
Supplementary Material
Acknowledgments
The first two authors contributed equally to this study. Supported by the NINDS RO1 NS047191, Searle Scholars, Sontag Foundation Distinguished Scientist Award, and March of Dimes Research Grant 6-FY07-401 (A. C.), F30NS053303 (G. W.) F30NS051864 (C. M), NIH T32 GM08061 (A. S.) and the Katten Muchin Rosenman Travel Scholarship (G. W. and S. N.). We thank R. Dorsky (Utah) for pTOP-dGFP, X. He (Harvard) for DN-LRP6, A. Kenney (Memorial Sloan Kettering) for DN-AKT, L. Li (Stowers) for β-catenin Ser552 antibody, R.T. Moon (UW, Seattle) for Super8xTOPflash and Super8xFOPflash constructs, N. Perrimon (Harvard) for pRL –polIII construct, R. Tsien (University of California, San Diego) for mCherry, M. Wheelock (Nebraska) for pSuper N-cadherin shRNA and control shRNA constructs, Ed Monuki (Irvine) for in situ hybridization expertise and C. J. Gottardi (Northwestern) for advice.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Alvarez-Buylla A, Lim DA. For the long run: maintaining germinal niches in the adult brain. Neuron. 2004;41:683–686. doi: 10.1016/s0896-6273(04)00111-4. [DOI] [PubMed] [Google Scholar]
- Bilder D, Li M, Perrimon N. Cooperative Regulation of Cell Polarity and Growth by Drosophila Tumor Suppressors. Science. 2000;289:113–116. doi: 10.1126/science.289.5476.113. [DOI] [PubMed] [Google Scholar]
- Cappello S, Attardo A, Wu X, Iwasato T, Itohara S, Wilsch-Brauninger M, Eilken HM, Rieger MA, Schroeder TT, Huttner WB, et al. The Rho-GTPase cdc42 regulates neural progenitor fate at the apical surface. Nat Neurosci. 2006;9:1099–1107. doi: 10.1038/nn1744. [DOI] [PubMed] [Google Scholar]
- Chenn A, Walsh CA. Regulation of cerebral cortical size by control of cell cycle exit in neural precursors. Science. 2002;297:365–369. doi: 10.1126/science.1074192. [DOI] [PubMed] [Google Scholar]
- Chenn A, Zhang YA, Chang BT, McConnell SK. Intrinsic polarity of mammalian neuroepithelial cells. Molecular and cellular neurosciences. 1998;11:183–193. doi: 10.1006/mcne.1998.0680. [DOI] [PubMed] [Google Scholar]
- DasGupta R, Kaykas A, Moon RT, Perrimon N. Functional genomic analysis of the Wnt-wingless signaling pathway. Science. 2005;308:826–833. doi: 10.1126/science.1109374. [DOI] [PubMed] [Google Scholar]
- De Wever O, Westbroek W, Verloes A, Bloemen N, Bracke M, Gespach C, Bruyneel E, Mareel M. Critical role of N-cadherin in myofibroblast invasion and migration in vitro stimulated by colon-cancer-cell-derived TGF-beta or wounding. Journal of cell science. 2004;117:4691–4703. doi: 10.1242/jcs.01322. [DOI] [PubMed] [Google Scholar]
- Dorsky RI, Sheldahl LC, Moon RT. A transgenic Lef1/beta-catenin-dependent reporter is expressed in spatially restricted domains throughout zebrafish development. Dev Biol. 2002;241:229–237. doi: 10.1006/dbio.2001.0515. [DOI] [PubMed] [Google Scholar]
- Fuchs E, Tumbar T, Guasch G. Socializing with the neighbors: stem cells and their niche. Cell. 2004;116:769–778. doi: 10.1016/s0092-8674(04)00255-7. [DOI] [PubMed] [Google Scholar]
- Gurdon JB. A community effect in animal development. Nature. 1988;336:772–774. doi: 10.1038/336772a0. [DOI] [PubMed] [Google Scholar]
- He XC, Zhang J, Tong WG, Tawfik O, Ross J, Scoville DH, Tian Q, Zeng X, He X, Wiedemann LM, et al. BMP signaling inhibits intestinal stem cell self-renewal through suppression of Wnt-beta-catenin signaling. Nat Genet. 2004;36:1117–1121. doi: 10.1038/ng1430. [DOI] [PubMed] [Google Scholar]
- Hirabayashi Y, Itoh Y, Tabata H, Nakajima K, Akiyama T, Masuyama N, Gotoh Y. The Wnt/beta-catenin pathway directs neuronal differentiation of cortical neural precursor cells. Development. 2004;131:2791–2801. doi: 10.1242/dev.01165. [DOI] [PubMed] [Google Scholar]
- Jho EH, Zhang T, Domon C, Joo CK, Freund JN, Costantini F. Wnt/beta-catenin/Tcf signaling induces the transcription of Axin2, a negative regulator of the signaling pathway. Mol Cell Biol. 2002;22:1172–1183. doi: 10.1128/MCB.22.4.1172-1183.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lien WH, Klezovitch O, Fernandez TE, Delrow J, Vasioukhin V. alphaE-catenin controls cerebral cortical size by regulating the hedgehog signaling pathway. Science. 2006;311:1609–1612. doi: 10.1126/science.1121449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loulier K, Lathia JD, Marthiens V, Relucio J, Mughal MR, Tang SC, Coksaygan T, Hall PE, Chigurupati S, Patton B, et al. beta1 integrin maintains integrity of the embryonic neocortical stem cell niche. PLoS biology. 2009;7:e1000176. doi: 10.1371/journal.pbio.1000176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makrigiannakis A, Coukos G, Christofidou-Solomidou M, Gour BJ, Radice GL, Blaschuk O, Coutifaris C. N-cadherin-mediated human granulosa cell adhesion prevents apoptosis: a role in follicular atresia and luteolysis? Am J Pathol. 1999;154:1391–1406. doi: 10.1016/S0002-9440(10)65393-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maretto S, Cordenonsi M, Dupont S, Braghetta P, Broccoli V, Hassan AB, Volpin D, Bressan GM, Piccolo S. Mapping Wnt/beta-catenin signaling during mouse development and in colorectal tumors. Proc Natl Acad Sci U S A. 2003;100:3299–3304. doi: 10.1073/pnas.0434590100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore KA, Lemischka IR. Stem cells and their niches. Science. 2006;311:1880–1885. doi: 10.1126/science.1110542. [DOI] [PubMed] [Google Scholar]
- Nelson WJ, Nusse R. Convergence of Wnt, beta-catenin, and cadherin pathways. Science. 2004;303:1483–1487. doi: 10.1126/science.1094291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noles SR, Chenn A. Cadherin inhibition of beta-catenin signaling regulates the proliferation and differentiation of neural precursor cells. Molecular and cellular neurosciences. 2007 doi: 10.1016/j.mcn.2007.04.012. in press. [DOI] [PubMed] [Google Scholar]
- Peerani R, Rao BM, Bauwens C, Yin T, Wood GA, Nagy A, Kumacheva E, Zandstra PW. Niche-mediated control of human embryonic stem cell self-renewal and differentiation. The EMBO journal. 2007;26:4744–4755. doi: 10.1038/sj.emboj.7601896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasin MR, Gazula VR, Breunig JJ, Kwan KY, Johnson MB, Liu-Chen S, Li HS, Jan LY, Jan YN, Rakic P, et al. Numb and Numbl are required for maintenance of cadherin-based adhesion and polarity of neural progenitors. Nat Neurosci. 2007 doi: 10.1038/nn1924. [DOI] [PubMed] [Google Scholar]
- Shen Q, Goderie SK, Jin L, Karanth N, Sun Y, Abramova N, Vincent P, Pumiglia K, Temple S. Endothelial cells stimulate self-renewal and expand neurogenesis of neural stem cells. Science. 2004;304:1338–1340. doi: 10.1126/science.1095505. [DOI] [PubMed] [Google Scholar]
- Song X, Zhu CH, Doan C, Xie T. Germline stem cells anchored by adherens junctions in the Drosophila ovary niches. Science. 2002;296:1855–1857. doi: 10.1126/science.1069871. [DOI] [PubMed] [Google Scholar]
- Stocker A, Chenn A. Focal Reduction of alphaE-catenin Causes Premature Differentiation and Reduction of beta-catenin Signaling During Cortical Development. 2008. In revision. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi T, Nowakowski RS, Caviness VS., Jr Cell cycle parameters and patterns of nuclear movement in the neocortical proliferative zone of the fetal mouse. J Neurosci. 1993;13:820–833. doi: 10.1523/JNEUROSCI.13-02-00820.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Temple S. The development of neural stem cells. Nature. 2001;414:112–117. doi: 10.1038/35102174. [DOI] [PubMed] [Google Scholar]
- Temple S, Davis AA. Isolated rat cortical progenitor cells are maintained in division in vitro by membrane-associated factors. Devel. 1994;120:999–1008. doi: 10.1242/dev.120.4.999. [DOI] [PubMed] [Google Scholar]
- Toyofuku T, Hong Z, Kuzuya T, Tada M, Hori M. Wnt/frizzled-2 signaling induces aggregation and adhesion among cardiac myocytes by increased cadherin-beta-catenin complex. J Cell Biol. 2000;150:225–241. doi: 10.1083/jcb.150.1.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran NL, Adams DG, Vaillancourt RR, Heimark RL. Signal transduction from N-cadherin increases Bcl-2. Regulation of the phosphatidylinositol 3-kinase/Akt pathway by homophilic adhesion and actin cytoskeletal organization. J Biol Chem. 2002;277:32905–32914. doi: 10.1074/jbc.M200300200. [DOI] [PubMed] [Google Scholar]
- Tufan AC, Tuan RS. Wnt regulation of limb mesenchymal chondrogenesis is accompanied by altered N-cadherin-related functions. Faseb J. 2001;15:1436–1438. doi: 10.1096/fj.00-0784fje. [DOI] [PubMed] [Google Scholar]
- van Noort M, Meeldijk J, van der Zee R, Destree O, Clevers H. Wnt signaling controls the phosphorylation status of beta-catenin. J Biol Chem. 2002;277:17901–17905. doi: 10.1074/jbc.M111635200. [DOI] [PubMed] [Google Scholar]
- Wallerand H, Cai Y, Wainberg ZA, Garraway I, Lascombe I, Nicolle G, Thiery JP, Bittard H, Radvanyi F, Reiter RR. Phospho-Akt pathway activation and inhibition depends on N-cadherin or phospho-EGFR expression in invasive human bladder cancer cell lines() Urol Oncol. 2008 doi: 10.1016/j.urolonc.2008.09.041. [DOI] [PubMed] [Google Scholar]
- Woodhead GJ, Mutch CA, Olson EC, Chenn A. Cell-Autonomous beta-Catenin Signaling Regulates Cortical Precursor Proliferation. J Neurosci. 2006;26:12620–12630. doi: 10.1523/JNEUROSCI.3180-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L, Dan HC, Sun M, Liu Q, Sun XM, Feldman RI, Hamilton AD, Polokoff M, Nicosia SV, Herlyn M, et al. Akt/protein kinase B signaling inhibitor-2, a selective small molecule inhibitor of Akt signaling with antitumor activity in cancer cells overexpressing Akt. Cancer research. 2004;64:4394–4399. doi: 10.1158/0008-5472.CAN-04-0343. [DOI] [PubMed] [Google Scholar]
- Yokota Y, Kim WY, Chen Y, Wang X, Stanco A, Komuro Y, Snider W, Anton ES. The adenomatous polyposis coli protein is an essential regulator of radial glial polarity and construction of the cerebral cortex. Neuron. 2009;61:42–56. doi: 10.1016/j.neuron.2008.10.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Niu C, Ye L, Huang H, He X, Tong WG, Ross J, Haug J, Johnson T, Feng JQ, et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature. 2003;425:836–841. doi: 10.1038/nature02041. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.