Abstract
To explore the similarities and differences of regulatory circuits among budding yeasts, we characterized the role of the unscheduled meiotic gene expression 6 (UME6) gene in Kluyveromyces lactis. We found that Ume6 was required for transcriptional silencing of the cryptic mating-type loci HMLα and HMRa. Chromatin immunoprecipitation (ChIP) suggested that Ume6 acted directly by binding the cis-regulatory silencers of these loci. Unexpectedly, a MATa ume6 strain was mating proficient, whereas a MATα ume6 strain was sterile. This observation was explained by the fact that ume6 derepressed HMLα2 only weakly, but derepressed HMRa1 strongly. Consistently, two a/α-repressed genes (MTS1 and STE4) were repressed in the MATα ume6 strain, but were expressed in the MATa ume6 strain. Surprisingly, ume6 partially suppressed the mating defect of a MATa sir2 strain. MTS1 and STE4 were repressed in the MATa sir2 ume6 double-mutant strain, indicating that the suppression acted downstream of the a1/α2-repressor. We show that both STE12 and the MATa2/HMRa2 genes were overexpressed in the MATa sir2 ume6 strain. Consistent with the idea that this deregulation suppressed the mating defect, ectopic overexpression of Ste12 and a2 in a MATa sir2 strain resulted in efficient mating. In addition, Ume6 served as a block to polyploidy, since ume6/ume6 diploids mated as pseudo a-strains. Finally, Ume6 was required for repression of three meiotic genes, independently of the Rpd3 and Sin3 corepressors.
EXPLORING the regulation of gene expression of orthologous genes in related organisms can tease out themes and variations in the evolution of regulatory mechanisms. Several studies suggest that the evolution of regulatory mechanisms is a major driving force in the generation of biological diversity (for example, Levine and Tjian 2003 and Tsong et al. 2006). Among closely related organisms, regulatory mechanisms are often conserved and regulatory regions share high degrees of identity. DNA sequences specifying binding sites for regulators in fact can be identified by aligning regulatory regions of orthologous genes between closely related organisms (Cliften et al. 2003; Kellis et al. 2003). The usefulness of such approaches diminishes, however, as the evolutionary distance increases. As an example, Saccharomyces cerevisiae and Kluyveromyces lactis separated by 50 × 106 to 150 × 106 years of evolution rarely produce meaningful promoter alignments. While the two genomes show high degrees of synteny, the regulatory mechanisms or protein-binding sites have diverged to the point where in silico-based methods for predicting regulatory mechanisms are rendered unreliable. It is nevertheless interesting to compare such organisms since a greater variation is expected. Hence, such comparisons should pinpoint regulatory mechanisms that are strictly conserved and identify mechanisms that have evolved quickly.
Unscheduled meiotic gene expression 6 (Ume6) is thoroughly characterized in S. cerevisiae. Ume6 binds to the URS1 sequence present in the regulatory regions of several early meiotic genes (Anderson et al. 1995). Ume6 regulates these genes both by repressing them during vegetative growth and by being required for their full induction during meiosis (Buckingham et al. 1990; Strich et al. 1994; Steber and Esposito 1995). This molecular switch is achieved by the interaction of Ume6 with different factors during mitosis and meiosis (Rubin-Bejerano et al. 1996; Kadosh and Struhl 1997). During mitosis, Ume6 interacts with the Sin3-Rpd3 corepressor complex. Rpd3 deacetylates histone H3 and H4 in proximity to the Ume6-binding site facilitating transcriptional repression (Kadosh and Struhl 1998; Rundlett et al. 1998). In addition, Ume6 recruits the Isw2 chromatin-remodeling complex, which also contributes to transcriptional repression in a parallel pathway to Rpd3-Sin3 (Goldmark et al. 2000). During meiosis, Ume6 becomes phosphorylated by the yeast GSK3β homologs Rim11 and Mck1, leading to its association with Ime1 (Malathi et al. 1997; Xiao and Mitchell 2000), This Ume6-Ime1 complex is important for the induction of meiotic genes. Recently, the view that Ume6 is converted to an activator during meiosis was challenged. Ume6 was shown to be degraded during early meiosis by ubiquitin-mediated proteolysis, and this degradation was important for meiotic gene expression and progression (Mallory et al. 2007). Microarray analysis of ume6 mutant strains showed that the Ume6 regulon involves carbon/nitrogen metabolism genes in addition to meiotic genes. Hence, Esposito and co-workers suggested that Ume6 couples metabolic responses to nutritional cues with the initiation of meiosis in diploid cells (Williams et al. 2002).
Silencing of the cryptic mating-type loci in S. cerevisiae requires a combination of regulatory sequences and dedicated proteins (Rusche et al. 2003). The HMR-E silencer contains binding sites for Rap1, Abf1, and ORC (Brand et al. 1987; Shore and Nasmyth 1987). These proteins recruit Sir proteins (Rine and Herskowitz 1987) to the silencer. Sir2 deacetylates the N-terminal tails of histones H3 and H4 (Imai et al. 2000; Landry et al. 2000; Smith et al. 2000). Sir3 and Sir4 bind strongly to the deacetylated histone tails (Hecht et al. 1995), to each other, and to Sir2 (Hecht et al. 1996; Triolo and Sternglanz 1996; Moazed et al. 1997; Ghidelli et al. 2001). Other notable molecular contacts include Rap1 interacting with Sir3/Sir4 (Moretti et al. 1994; Cockell et al. 1995) and Orc1 interacting with Sir1 (Triolo and Sternglanz 1996; Gardner and Fox 2001). In combination, these molecular interactions, coupled to a deacetylation of histones, lead to the spread of a stable silencing complex encompassing the entire HMRa locus (Rusche et al. 2002). Silencing of the cryptic mating-type loci is important for mating proficiency in S. cerevisiae because simultaneous expression of genes specifying both mating types leads to sterility. In MATa/MATα diploids or sir mutant haploid strains, the a1 and α2 homeodomain transcription factors form a heterodimer that represses transcription of haploid-specific genes (hsgs) (Herskowitz 1988). Hsgs include those genes encoding the subunits of the heterotrimeric G-protein (STE4, STE18, and GPA1) (Galgoczy et al. 2004). The heterotrimeric G-protein is essential for mating pheromone signaling, which explains the mating defect of silencing-deficient strains.
K. lactis also contains cryptic mating-type loci that are transcriptionally silenced (Åström and Rine 1998). During an earlier characterization of the K. lactis HMLα silencer, we discovered a minimal DNA fragment (102 bp long), which was capable of mediating silencing (Åström et al. 2000). In this minimal silencer, three short DNA sequences essential for silencing were identified (Sjöstrand et al. 2002). These sites were named the A-, B-, and C-boxes, respectively. The B-box is a binding site for the Myb-domain protein Reb1, and strains carrying temperature-sensitive reb1 alleles could not silence HMLα at the restrictive temperature. The proteins binding to the A- and C-boxes are unknown, but the A-box overlapped with a potential binding site for Ume6. Additionally, the HMRa silencer contained potential binding sites for both Ume6 and Reb1.
In this study, we investigated the role of Ume6 in the silencing and regulation of meiotic genes in K. lactis. Ume6 was required for silencing of both HMRa and HMLα and also for repressing a subset of meiotic genes during vegetative growth. We also found an unanticipated role for Ume6 in promoting the MATa/MATα cellular identity.
MATERIALS AND METHODS
Yeast strains:
The strains used in this study are listed in Table 1. Unless noted otherwise, all gene deletions were generated using a one-step gene disruption procedure (Rothstein 1983) with a NAT or kanMX PCR fragment amplified from pAG25 (Goldstein and McCusker 1999) or pFA6a-kanMX (Bahler et al. 1998) and confirmed by DNA blots or locus-specific PCR. SAY536 (MATα) was generated by crossing SAY119 with SAY186 (Åström et al. 2000), followed by tetrad analysis. SAY537 and SAY538 (MATα nej1∷LEU2) were generated by crossing SAY536 with SAY572 (MATa nej1∷LEU2) (Kegel et al. 2006). The strains SAY549 (ume6∷kanMX), SAY839 (sin3∷NAT), and SAY1084 (ste4∷NAT) were generated in SAY572. SAY541 (ume6∷kanMX) and SAY544 (sir2∷kanMX) were generated in SAY538. SAY726 (hmra∷NAT) and SAY838 (sin3∷NAT) were generated in SAY509 (MATα nej1∷LEU2) (Kegel et al. 2006). SAY571 (MATα sir2∷kanMX) was generated by crossing SAY544 with CK213. SAY276 (rpd3∷LEU2) was generated by a two-step procedure deleting RPD3 in CK213. The double mutants SAY586 (sir2∷kanMX ume6∷kanMX), SAY945 (sir2:kanMX rpd3∷LEU2), and SAY947 (sir2∷kanMX sin3∷NAT) were generated by crossing the respective single mutant with SAY571. SAY732 (hmra∷NAT ume6∷kanMX) was generated by crossing SAY726 with SAY549, followed by tetrad analysis. Selecting LEU2+ segregants from the cross between SAY276 with SAY119 generated SAY789 (MATα rpd3∷LEU2). SAY721 (UME6-13XMYC) was generated by amplifying a 13XMYC-containing PCR fragment with 50 bp of flanking DNA corresponding to the UME6 3′ end and introducing it in SAY537. Diploid strains used in the a/α-cell identity mating assay and fluorescence activated cell sorter (FACS) analyses were made by crossing SAY538 (WT) with SAY572 (WT), SAY538 with SAY549 (ume6∷kanMX), and SAY541 (ume6∷kanMX) with SAY549, thus generating the WT, heterozygous (UME6/ume6), and homozygous (ume6/ume6) mutant diploids, respectively.
TABLE 1.
Yeast strains used in this study
| Name | Genotype | Reference |
|---|---|---|
| WM52 | MATα ade1 adeX his7 uraA1 | Chen and Clark-Walker (1994) |
| CK213 | MATaleu2 lysA1 metA1 trp1 uraA1 | Chen and Clark-Walker (1994) |
| SAY102 | MATaleu2 lysA1 metA1 sir2∷URA3 trp1 uraA1 | Åström and Rine (1998) |
| SAY119 | MATα ade1 leu2 trp1 metA1 uraA1 | Åström et al. (2000) |
| SAY186 | MATaleu2 lysA1 metA1 trp1 uraA1 hmlαΔp | Åström et al. (2000) |
| SAY189 | MATahmlαΔp leu2 sir2∷URA3 trp1 uraAI | Åström et al. (2000) |
| SAY276 | MATaleu2 lysA1 metA1 rpd3∷LEU2 trp1 uraA1 | This study |
| SAY509 | MATα ade1 leu2 metA1 nej1∷LEU2 trp1 uraA1 | Kegel et al. (2006) |
| SAY536 | MATα ade1 hmlαΔp leu2 metA1 trp1 uraA1 | This study |
| SAY537 | MATα ade1 hmlαΔp leu2 metA1 nej1∷LEU2 trp1 uraA1 | This study |
| SAY538 | MATα ade1 hmlαΔp leu2 metA1 nej1∷LEU2 trp1 uraA1 | This study |
| SAY541 | MATα ade1 hmlαΔp leu2 metA1 nej1∷LEU2 trp1 ume6∷kanMX uraA1 | This study |
| SAY544 | MATα ade1 hmlαΔp leu2 metA1nej1∷LEU2 trp1 sir2∷kanMX uraA1 | This study |
| SAY549 | MATaleu2 lysA1 metA1 nej1∷LEU2 trp1 ume6∷kanMX uraA1 | This study |
| SAY569 | MATaleu2 uraA1 sir2∷kanMX trp1 metA1 | This study |
| SAY571 | MATα hmlαΔp leu2 uraA1 sir2∷kanMX trp1 metA1 | This study |
| SAY572 | MATaleu2 lysA1 metA1 nej1∷LEU2 trp1 uraA1 | Kegel et al. (2006) |
| SAY586 | MATaleu2 lysA1 metA1 sir2∷kanMX trp1 ume6∷kanMX uraA1 | This study |
| SAY721 | MATα ade1 hmlαΔp leu2 nej1∷LEU2 metA1 trp1 UME6-13xMYC∷kanMX uraA1 | This study |
| SAY726 | MATα ade1 hmra∷NAT leu2 metA1 nej1∷LEU2 trp1 uraA1 | This study |
| SAY732 | MATα ade1 hmra∷NAT leu2 metA1 nej1∷LEU2 trp1 ume6∷kanMX uraA1 | This study |
| SAY789 | MATα ade1 leu2 metA1 rpd3∷LEU2 trp1 uraA1 | This study |
| SAY838 | MATα ade1 leu2 metA1 nej1∷LEU2 sin3∷NAT trp1 uraA1 | This study |
| SAY839 | MATaleu2 lysA1 metA1 nej1∷LEU2 sin3∷NAT trp1 uraA1 | This study |
| SAY945 | MATalysA1 trp1 metA1 leu2 uraA1 rpd3∷LEU2 sir2∷KanMX | This study |
| SAY947 | MATaleu2 lysA1 metA1 sin3∷NAT sir2∷kanMX trp1 uraA1 | This study |
| SAY1081 | MATaleu2 lysA1 metA1 nej1∷LEU2 ste4∷NAT trp1 ume6∷kanMX uraA1 | This study |
| SAY1084 | MATaleu2 lysA1 metA1 nej1∷LEU2 ste4∷NAT trp1 uraA1 | This study |
Media and standard methods:
Growth media for yeast and bacteria; preparation of DNA, RNA, and protocols for DNA and RNA blots; and transformation of yeast and bacteria are described elsewhere (Schiestl and Gietz 1989; Ausubel 1999). Mating assays were performed as described (Åström and Rine 1998) by mixing 5 μl (A600 ∼0.5) of each mating partner on a 2 × 2-cm area on a YEPD or selection plate. The mating plate was incubated for 18–24 hr at 30°, replica printed on selective media, and incubated for a further 72–96 hr before the result was scored. For RNA blots, the probes used corresponded to the following regions of the genes investigated: STE4 (+788–1271 bp), MTS1 (ORF), ACT1 (+382–1408 bp), DIG1 (+446–1015 bp), a2 (+40–456 bp), STE12 (+1531–1982 bp), HOP1 (+669–1092 bp), SPO69 (+782–1262 bp), and CST9 (+119–638 bp). Detection and quantification of signals from RNA blots were done using a FLA-3000 phosphorimager (Fuji, Tokyo) and the Multi Gauge software (Fujifilm).
Plasmids:
Plasmids p291 and p414 are described elsewhere (Åström et al. 2000). The pEB119 (pCXJ20-pADH1-a2) plasmid was generated by a three-factor cloning using a BamHI–PstI PCR fragment containing the S. cerevisiae ADH1 promoter (−492 to −13 bp, relative to the start codon), a PstI–HindIII PCR fragment containing the K. lactis a2 ORF and BamHI–HindIII-digested pCXJ20 (LEU2). The pEB18 (pCXJ18-pGAL1-STE12) vector was generated in two steps. pPMB35 (pCXJ18-pGAL1-MTS1) (Barsoum et al. 2010) linearized with SmaI was cotransformed with a PCR fragment containing K. lactis STE12 and 50 bp of homology on each side to the Gal1 promoter and cloning cassette, respectively. Recombination in vivo generated pEB18.
FACS analysis:
A 1-ml sample of exponentially growing cells (A600 ∼0.5) was centrifuged and fixed by dissolving the pellet in cold 70% ethanol while vortexing. The cells were centrifuged and resuspended in 0.8 ml 50 mm Tris–HCl (pH 7.8) and then 20 μl RNase A (10 mg/ml) was added, followed by an incubation at 37° for 12 hr. Cells were resuspended in 0.5 ml 200 mm Tris–HCl (pH 7.5), 211 mm NaCl, and 78 mm MgCl2. Then, 55 μl of 1 mg/ml propidium iodide was added; the cells were sonicated and analyzed by FACS.
Chromatin immunoprecipitation and quantitative PCR:
ChIP was performed as described (Ausubel 1999). Briefly, cells grown in 50 ml of YEPD (OD600 ∼1) were fixed in 1% formaldehyde for 15 min and then quenched with 125 mm glycine for 5 min at room temperature. Cells were resuspended in 250 μl lysis buffer [0.1% deoxycholic acid, 1 mm EDTA, 50 mm HEPES (pH 7.5), 140 mm NaCl, 1% Triton X-100] containing Complete Mini protease inhibitors (Roche). Then the cells were disrupted with glass beads in a multi-vortexer for 30 min at 4°. The chromatin was sheared, and immunoprecipitation was performed with 20 μg/ml of anti c-Myc antibody 9E10 (Santa Cruz). Input DNA was recovered before the immunoprecipitation. The precipitates were washed 5 min with several buffers according to the protocol. The crosslinking was reversed, and the DNA was purified and RNase A treated. Quantitative PCR (qPCR) reactions were performed in triplicates using SYBR Green PCR Master Mix (Applied Biosystems) on an ABI-Prism 7000 instrument. As reference, standard curves were determined on dilutions of input DNA. For quantitative RT–PCR, total RNA from logarithmically growing cells was converted into cDNA using a cDNA synthesis kit and the conditions recommended by the manufacturer (Invitrogen). Briefly, 5 μg of RNA was treated with DNase I followed by first-strand cDNA synthesis using Superscript II RT and an oligo(dT)20 primer. As reference, the ACT1 gene was used, and quantification was performed by the comparative CT method. The sequences of primers used are available in the supporting information, Table S1. Analysis of the data was performed using the 7000 System sequence detection software.
Random spore analysis:
Cells were scraped off a sporulation plate and resuspended in 1 ml TE (10 mm Tris and 1 mm EDTA), and the total cell count was determined using a Bürkner chamber. The suspension was divided in two aliquots where one was put aside as a control. To the other aliquot, 50 μl of 2 mg/ml Zymolyase was added followed by incubation at 37° for 10 min. All vegetative cells were killed by an additional incubation at 55° for 10 min. Both suspensions were serially diluted in TE and plated on YEPD plates.
RESULTS
Comparison of S. cerevisiae and K. lactis Ume6:
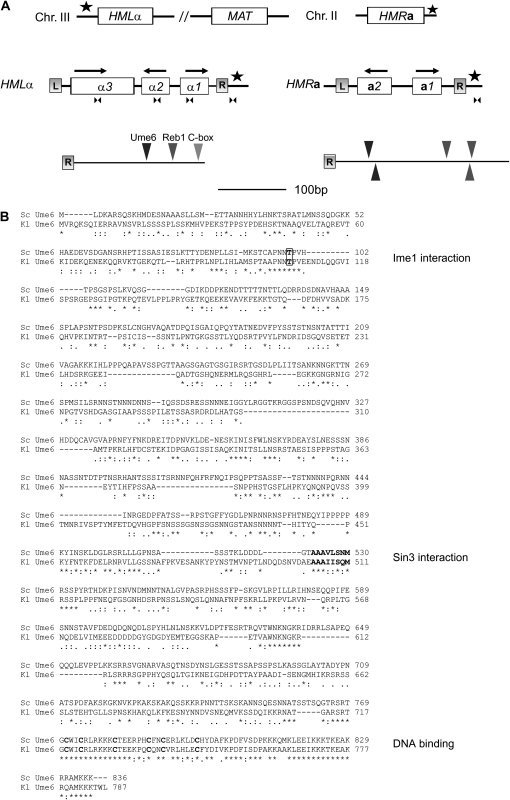
A BLAST search using the Génolevures database (http://cbi.labri.fr/Genolevures) revealed the presence of a single UME6 ortholog in the K. lactis genome. KlUme6 (787 amino acids) was 26% identical and 41% similar compared with ScUme6 (838 amino acids). The alignment between the proteins (Figure 1) revealed that the C-terminal DNA-binding domain was highly conserved between yeasts (∼90% identity), indicating that the DNA sequences that the two proteins recognize were similar. This domain forms a C6 zinc cluster and exhibits a zinc-dependent binding to the URS1 DNA sequence (Anderson et al. 1995) in S. cerevisiae. An earlier study (Washburn and Esposito 2001) of the interaction between ScUme6 and the Sin3 corepressor identified a number of mutations in Ume6 that abolished this interaction. These mutations were clustered to amino acids 523–530 of ScUme6 and were predicted to be part of an amphipathic α-helix. This stretch of amino acids was highly conserved in KlUme6 (Figure 1), indicating that a similar interaction also occurs in milk yeast. In addition, the NH2-terminal domain (1–158) of ScUme6 interacts with the Ime1 transcriptional activator (Rubin-Bejerano et al. 1996). Threonine 99 in ScUme6 is probably phosphorylated by Rim1 (Malathi et al. 1997), which in turn is important for this interaction. Threonine 99 as well as two other threonine residues in the vicinity were also conserved between species.
Figure 1.—
Similarity between S. cerevisiae and K. lactis Ume6. (A) Schematic of the mating-type loci in K. lactis, including their chromosomal position (top) and the ORFs in the HMLα and HMRa loci with the transcriptional orientation depicted by arrows (middle). The boxes with L (left) and R (right) show the repetitive sequences that flank the mating-type loci, and the stars represent the position of the silencers. The arrowheads below the boxes represent the positions of the primers used in the qPCR experiments in Figure 2. (Bottom) Ume6- and Reb1-binding sites and the C-box in the HMLα and HMRa silencers. (B) Pairwise alignment of ScUme6p and KlUme6p displaying 41% similarity and 26% identity. Threonine 99/106 is boxed; the cysteines of the C-terminal Zn2+-finger domain and the residues of the amphiphatic helix are in boldface type.
Ume6 is required for silencing of HMRa and HMLα:
To investigate the role of Ume6 in K. lactis, we generated strains of the opposite mating type in which the UME6 ORF was replaced by a kanMX marker gene. Both the MATα ume6∷kanMX and MATa ume6∷kanMX strains displayed normal growth. Next, these strains were tested for silencing defects. We investigated the expression of the MATα1/HMLα1, MATα2/HMLα2, MATa1/HMRa1, and MATa2/HMRa2 genes and normalized to the expression of ACT1. Total RNA isolated from MATa and MATα wild-type strains and isogenic ume6 mutants was subjected to quantitative RT–PCR. We began by investigating the expression of the HMRa1 and HMRa2 genes (Figure 2A). The HMRa1 gene was efficiently silenced in the MATα wild-type strain, and the ume6 mutation resulted in derepression of a1 expression, consistent with Ume6-mediated silencing of HMRa1. Surprisingly, the HMRa2 gene was expressed in the wild-type MATα strain. The levels of a2 mRNA in the wild-type MATα strain were ∼50% of the levels in the wild-type MATa strain, indicating a gene dosage effect. In the MATα ume6 strain, a2 expression was only moderately increased. Since this result was unexpected, we repeated the assay using an independent primer pair from which similar results were obtained (data not shown; see discussion).
Figure 2.—
Ume6 is required for silencing of the cryptic mating-type loci. (A) Quantitative RT–PCR analysis using primers specific for the a1 and a2 genes and RNA from MATα (SAY538), MATα ume6 (SAY541), MATa (SAY572), and MATa ume6 (SAY549) strains. (B) Same as in A, but using primers specific for the α1 and α2 genes. The amount of cDNA in each sample was first normalized to the ACT1 gene. The expression levels of a1 and a2 in SAY572 was set to 1.0 in A, and the expression levels of α1 and α2 in SAY538 was set to 1.0 in B. Values shown are the average of two independent experiments with the maximum and minimum values indicated. (C) ChIP was performed on strains SAY537 (UME6) and SAY721 (UME6-13XMYC) using an anti-Myc antiserum (9E10). Following ChIP, DNA associated with the HMRa and HMLα silencers and the MATα1/HMLα1, MATα2/HMLα2, MATα3/HMLα3, and ACT1 ORFs was analyzed by qPCR using locus-specific primers (as indicated by the colors). Mock immunoprecipitations, in which the 9E10 antiserum was omitted, are included. The y-axis represents the amount of precipitated DNA relative to a standard curve obtained from dilutions of the input DNA. Values shown are the standard error of the mean resulting from three independent experiments.
Next we investigated the expression of the HMLα1 and HMLα2 genes (Figure 2B). Both the HMLα1 and the HMLα2 genes were efficiently silenced in the MATa wild-type strain. Absence of Ume6 derepressed their expression, but the effect was weaker compared to the derepression of HMRa. In the MATa ume6 strain, the HMLα1 and HMLα2 genes were derepressed to only 37% and 6.5%, respectively, of their expression levels in the wild-type MATα strain. In contrast, in the MATα ume6 strain, the HMRa1 and HMRa2 genes were efficiently derepressed (68% and 78%, respectively, of their expression levels in a wild-type MATa strain). We concluded that Ume6 had an essential role in the silencing of HMRa, but also an important role in the silencing of HMLα.
Ume6 interacts with both the HMRa and the HMLα loci:
We showed previously that if the potential Ume6-binding site in the HMLα silencer was mutated, then silencer function was compromised (Sjöstrand et al. 2002). In addition, the HMRa silencer contained two potential Ume6-binding sites (Figure 1A), but their contribution to silencing remains unknown. These observations, together with the finding that ume6 mutant strains derepressed both HMRa and HMLα, strongly suggested that binding of Ume6 to the silencers was important for silencing. To investigate if Ume6 could bind the HMRa and HMLα silencers in vivo, we performed a ChIP experiment followed by qPCR using a Ume6-13XMyc fusion protein and an antiserum (9E10) that specifically recognized the Myc epitope. We amplified DNA from several regions (Figure 1A), two of which were in close proximity to the predicted Ume6-binding sites in the HMRa and HMLα silencers. In addition, we analyzed regions located in the MATα1/HMLα1, MATα2/HMLα2, and MATα3/HMLα3 ORFs. Finally, we used the ACT1 ORF as a negative control because it contains no discernible Ume6-binding site. The Ume6-13XMyc fusion protein appeared to function normally because the MATα UME6-13XMyc strain mated with normal efficiency (data not shown; see below). As a control, we compared the UME6-13XMyc strain with an isogenic untagged (UME6) strain. Relative to input DNA, in the UME6-13XMyc strain we recovered 1.4% of the HMRa locus and 1.1% of the region close to the Ume6-binding site in HMLα. In contrast, in the untagged strain using the same primer pairs, we recovered only 0.10% of the HMRa silencer and 0.09% of the HMLα silencer (Figure 2C). In addition, ChIP samples in which the 9E10 antiserum was omitted recovered only background levels of HMRa and HMLα. When we used primer pairs that amplified in the MATα1/HMLα1, MATα2/HMLα2, and MATα3/HMLα3 ORFs, lower levels of DNA were precipitated from the UME6-13XMyc strain (0.44–0.43% of input) comparable to the ACT1 ORF (0.34% of input). Hence, Ume6 did not spread efficiently over the HMLα locus. We concluded that Ume6 could bind the silencers in vivo and thus most likely had a direct effect on silencing.
A MATα ume6 strain is sterile but a MATa ume6 strain is fertile:
An alternative measure of silencing of the cryptic mating-type loci is mating assays. The logic for this is that derepression of the cryptic mating-type loci results in sterility due to the simultaneous expression of the a1 and α2 proteins. The MATα ume6 strain mated very inefficiently with a MATa tester strain compared to the wild-type parent. However, a MATa ume6 strain showed no mating defect when mixed with a MATα tester strain (Figure 3A). Lack of Ume6 thus resulted in a MATα-specific mating defect. These results were consistent with the gene expression analyses because the absence of Ume6 efficiently derepressed HMRa1, but only weakly derepressed HMLα2.
Figure 3.—
Mating phenotypes of ume6 strains. (A) Mating tests of MATa (CK213) and MATa ume6 (SAY549) (left) and of MATα (SAY538) and MATα ume6 (SAY541) (right) strains to MATα (WM52) and MATa (CK213) tester strains. Diploids were selected on synthetic dextrose plates containing uracil (SD + Ura; left) or on SD + Ura, Trp, and Met; right. (B) Mating tests of MATα hmra (SAY726), MATα ume6 (SAY541), and MATα hmra ume6 (SAY732) strains to a MATa tester strain (CK213). Diploids were selected on SD + Ura, Trp, and Met. (C) Mating tests of MATa strains; WT (SAY572), sir2 (SAY102), sir2 hmlαΔp (SAY189), and sir2 ume6 (SAY586) to MATα tester mater (WM52). Diploids were selected on SD + Ura. Below each panel are the results of quantitative matings in which the mating efficiency of the wild-type MATa or MATα strain was set to 1.0.
If the α-specific mating defect was solely due to the derepression of HMRa, then the deletion of this locus should suppress the mating defect. We generated a MATα ume6 hmra strain and tested the mating proficiency. Deletion of HMRa substantially improved mating in the MATα ume6 strain, but did not completely suppress the mating defect (Figure 3B). Hence, the mating defect of a MATα ume6 strain was largely explained by the derepression of HMRa, but loss of Ume6 also compromised some other aspect of α-mating.
Haploid-specific genes are repressed in the MATα ume6 strain but expressed in the MATa ume6 strain:
To further explore the MATα-specific mating defect of ume6 strains, we investigated the transcription of hsgs. We wanted to compare the transcription of hsgs in MATα ume6 and MATa ume6 strains. Both the gene expression analyses and the mating phenotypes suggested that hsgs were repressed in MATα ume6 but expressed in MATa ume6. The only gene previously reported to be an hsg in K. lactis is MTS1 (Barsoum et al. 2010), the ortholog of S. cerevisiae RME1. K. lactis NEJ1 is not repressed in diploids (Kegel et al. 2006), and K. lactis does not contain a functional HO gene (Fabre et al. 2005). To uncover additional K. lactis hsgs, we identified the orthologs of the S. cerevisiae FUS3, GPA1, STE4, STE18, and STE5 genes. These genes are directly repressed by the a1/α2 heterodimer in S. cerevisiae (Galgoczy et al. 2004). We tested the cell-type-specific transcription of these genes on RNA blots (data not shown). The STE4 transcript was reduced severalfold in the presence of the a1/α2 repressor and is thus a bona fide hsg.
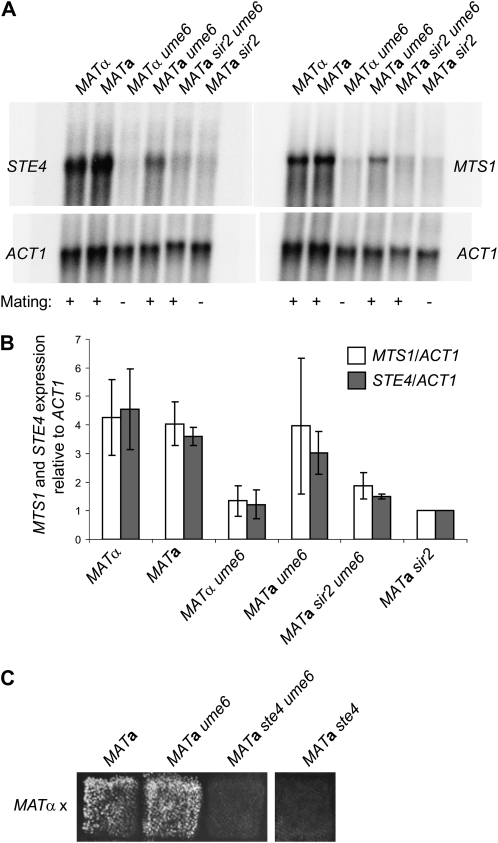
Next we tested the expression of STE4 and MTS1 in MATα ume6, MATa ume6, and their corresponding wild-type parents (Figure 4A). Consistent with the mating phenotypes, the MATa ume6 strain expressed close to normal levels of the transcripts, but the MATα ume6 strain showed reduced expression of both STE4 and MTS1. Hence, while partial derepression of HMLα2 in ume6 strains was not sufficient to induce a/α repression, a robust derepression of HMRa1 was.
Figure 4.—
The ume6 mutation influences expression of haploid-specific genes. (A) RNA-blot analysis using strains MATα (SAY538), MATa (SAY572), MATα ume6 (SAY541), MATa ume6 (SAY549), MATa sir2 ume6 (SAY586), and MATa sir2 (SAY102). The 32P-labeled probes used to detect transcripts were MTS1, STE4, or ACT1 specific, as indicated. (B) Quantification of the blot shown in A plus an independent experiment, indicating the minimum and maximum values as error bars. Shown is the ratio between the MTS1/ACT1 and STE4/ACT1 transcripts, with the ratio in SAY102 defined as 1.0. (C) Mating tests of MATa (CK213), MATa ume6 (SAY549), MATa ste4 ume6 (SAY1081), and MATa ste4 (SAY1084) strains to the MATα tester strain (WM52). Diploids were selected on SD + Ura.
The ume6 mutation suppressed the mating defect of a MATa sir2 strain:
We generated a MATa ume6 sir2 double-mutant strain in which we expected a strong derepression of HMLα. As shown previously, sir2 is required for the silencing of the HMLα locus, and consequently MATa strains that harbor a sir2 mutation display a severe mating defect (Chen and Clark-Walker 1994; Åström et al. 2000) (Figure 3C). Importantly, the MATa sir2 mating defect was efficiently suppressed by a mutation (hmlαΔp), which abolished HMLα1/2 transcription. Surprisingly, the MATa sir2 ume6 double-mutant strain mated more efficiently than the sir2 single-mutant strain (Figure 3C). Hence, the absence of Ume6 partially suppressed the mating defect that normally results from derepression of HMLα in a MATa background. Thus, the role of Ume6 in mating in K. lactis was complex and needed further examination.
We investigated the transcription of hsgs in the MATa sir2 ume6 double-mutant strain. Inconsistent with the mating phenotype, the MATa sir2 ume6 mating-proficient strain showed clear signs of a/α repression, almost to the same extent as the repression observed in the mating-deficient MATa sir2 strain (Figure 4A). We concluded that the absence of Ume6 must affect processes distinct from the a/α repression of hsgs to suppress the mating defect.
ume6 is not a bypass suppressor:
STE4 encodes the β-subunit of the heterotrimeric G-protein necessary for mating pheromone signaling and is essential for mating in S. cerevisiae (Whiteway et al. 1989) and K. lactis (Kawasaki et al. 2005). We investigated the possibility that the lack of Ume6 activated an unusual MATa mating pathway that was independent of mating pheromone signaling. We thus generated a ste4 null mutant strain and combined this mutation with the ume6 mutation. Both the ste4 single-mutant and the ste4 ume6 double-mutant strains were completely sterile in both MATa and MATα backgrounds (Figure 4C; data not shown). Hence ume6 was not a bypass suppressor, and ume6 strains required the normal mating pheromone pathway for mating proficiency.
Ume6 maintains the a/α cell type:
A previous study showed that dig1/dig1 dig2/dig2 diploid S. cerevisiae strains expressed a-specific genes and mated as pseudo a-cells (Gelli 2002). In addition, a dig1/dig1 dig2/dig2 diploid S. cerevisiae strain repressed the hsg RME1, indicating that the a1/α2 repressor complex is still active in the absence of Dig1/2. This phenotype was analogous to our observations in the MATa sir2 ume6 background, and we thus tested if Ume6 was important to maintaining sterility of MATa/MATα diploids in K. lactis. Homozygous ume6 mutant diploids mated with a MATα tester strain, while a heterozygous UME6/ume6 diploid strain could not (Figure 5A). Neither diploid strains mated with a MATa tester strain (data not shown). To directly demonstrate the presence of triploid cells, we scraped cells from the mating plates and measured the DNA content in a FACS. A peak corresponding to a higher DNA content than a control MATa/MATα diploid was uniquely present in the cells resulting from mating the MATa/MATα ume6/ume6 strain with the MATα tester strain (Figure 5B). The triploid state appeared unstable, however, because we also observed peaks corresponding to a diploid DNA content in this sample. We concluded that Ume6 was necessary for diploid sterility in K. lactis.
Figure 5.—
Ume6 is required for maintaining the a/α cell identity. (A) Mating tests of the MATa/MATα diploid strains UME6/ume6 (SAY538X549) and ume6/ume6 (SAY541X549) to the MATα tester strain (WM52). Triploids were selected on SD + Ura. (B) FACS analysis of a haploid MATα strain (WM52), a wild-type diploid strain (SAY538XSAY572), and mating mixtures obtained from mixing WM52 with the UME6/ume6 heterozygous diploid (SAY538X549) or the ume6/ume6 homozygous diploid (SAY541X549). (C) RNA-blot analysis of total RNA prepared from the strains indicated above the lanes (same as in Figure 4A). The probes used in each panel are shown on the left side of each blot. Below the blots are quantifications of the relative expression levels expressed as the DIG1, a2, and STE12/ACT1 ratio. The average of two independent experiments is shown with the minimum and maximum values indicated as error bars. (D) Mating tests of a MATa sir2 strain (SAY569) containing empty plasmids (pCXJ18 + pCXJ20), a plasmid overexpressing the a2 gene from the ADH1 promoter (pCXJ18 + pADH-a2), a plasmid overexpressing the STE12 gene from the GAL1-10 promoter (pGAL-STE12 + pCXJ20), or plasmids overexpressing both STE12 and a2 (pGAL-STE12 + pADH-a2). The MATα tester strain was WM52, and diploids were selected on SD.
Strains lacking Ume6 and Sir2 overexpressed a2 and Ste12:
We next investigated the transcription of the single DIG ortholog in the K. lactis genome. In addition, we examined the transcript levels of K. lactis STE12 and HMRa2/MATa2 (Figure 5C) because these genes encode regulators of the mating response. Ume6 single-mutant strains did not show large differences (<1.5-fold) compared to the isogenic wild-type strains for any of the transcripts investigated. It should be noted that the expression of HMRa2/MATa2 was low in wild-type strains and hence difficult to detect, but was observed in the quantitative RT–PCR analysis (Figure 2A). The mating-proficient MATa sir2 ume6 double-mutant strain overexpressed both STE12 (2.4-fold) and a2 (5-fold), both averages of two independent experiments, compared to a MATa wild-type strain. The a2 gene was also overexpressed in the sir2 single-mutant strain, but not to the same extent as in the sir2 ume6 double mutant. Hence, a plausible explanation for the mating proficiency of the MATa sir2 ume6 strain, despite the a/α repression, was the derepression of STE12 and/or MATa2/HMRa2 transcription. We set out to test this idea by introducing plasmid-borne copies of these genes into a MATa sir2 strain. The STE12 gene was controlled by the GAL1 promoter and the MATa2 gene was expressed from the ADH1 promoter. The plasmid-encoded STE12 and a2 genes were functional because they complemented the mating defect of the corresponding mutant strain (data not shown). While the increased expression of a2 or STE12 was unable to suppress the mating defect of the MATa sir2 strain, the overexpression of both proteins simultaneously could (Figure 5D). This remarkable result suggests that increased expression of Ste12 and a2 provides a molecular explanation for ume6 suppression of the MATa sir2 mating defect.
Ume6 is required for repression of a subset of meiotic genes in K. lactis:
It is possible that the set of genes repressed by Ume6 in S. cerevisiae and K. lactis completely changed, i.e., from meiotic genes to the cryptic mating-type loci. To learn if Ume6 repressed meiotic genes in K. lactis also, we searched the genome using the consensus Ume6-binding sites from S. cerevisiae as query (TCGGCGGCTAA). To limit the number of positive hits, we considered only perfect matches to the 11-bp Ume6 consensus site. An exact match to an 11-bp sequence should occur stochastically only once in every 4 × 106 bp and approximately three times in the K. lactis genome. Fourteen hits were recovered and 11 resided in intergenic, potential regulatory regions. One of the perfect matches was in the HMRa silencer, but the potential Ume6-binding site in the HMLα silencer was not identified since this sequence had two mismatches compared to the 11-bp consensus. Strikingly, 6 of the consensus Ume6-binding sites resided in the potential regulatory regions of the NDT80, MER1, SPO22, SPO69, HOP1, and CST9 genes. The S. cerevisiae orthologs of these genes were previously implicated as direct Ume6 targets and/or as having meiosis-specific functions (Engebrecht and Roeder 1990; Xu et al. 1995; Williams et al. 2002). Using RNA blots from wild-type and ume6 mutant strains, we investigated if the expression of these genes was affected by the absence of Ume6. The CST9, HOP1, and SPO69 transcripts accumulated in both MATa ume6 and MATα ume6 strains, but were undetectable in the wild-type MATα strain (Figure 6A). The NDT80, MER1, and SPO22 transcripts were not detectable in either the absence or the presence of Ume6 (data not shown). Since the CST9, HOP1, and SPO69 genes contained Ume6-binding sites in their regulatory regions and Ume6 was required for their transcriptional repression, they were likely direct Ume6 target genes. To test this idea for SPO69, ChIP/qPCR was performed using primers specific for the SPO69 regulatory region (amplifying close to the potential Ume6-binding site). Relative to input DNA, we recovered 0.63% DNA from the UME6-13XMyc-tagged strain, but only 0.08% was recovered from the untagged strain (Figure 6C). Hence, Ume6-13XMyc associated with the SPO69 regulatory region. We concluded that, similar to S. cerevisiae Ume6, K. lactis Ume6 repressed meiosis-specific genes during vegetative growth.
Figure 6.—
Ume6 represses meiotic genes during vegetative growth independently of Rpd3/Sin3. (A) RNA-blot analysis of total RNA prepared from MATα ume6 (SAY541), MATα (SAY119), and MATa ume6 (SAY549) strains as indicated above the lanes. The gene-specific probes used are indicated on the right, and the control hybridizations with an ACT1 probe are shown on the left. (B) RNA-blot analysis using RNA from strains MATa (SAY572), MATα (SAY119), MATa ume6 (SAY549), MATα ume6 (SAY541), MATa rpd3 (SAY276), MATα rpd3 (SAY789), MATa sin3 (SAY839), and MATα sin3 (SAY838). (C) ChIP was performed as described in Figure 2C by using locus-specific primers for the SPO69 regulatory region. Values on the y-axis are the average of two independent experiments displaying the maximum and minimum values as error bars. (D) Mating test of the MATa strains WT (CK213), rpd3 (SAY276), rpd3 sir2 (SAY945), sin3 (SAY839), and sin3 sir2 (SAY947) to the MATα tester strain (WM52) (left, top and bottom). Diploids were selected on SD + Ura. Mating tests of MATα strains WT (SAY119), rpd3 (SAY789), and sin3 (SAY838) to the MATa tester strain (CK213) (right, top and bottom). Diploids were selected on SD + Ura, Leu, Trp, and Met.
Ume6 is required for efficient sporulation:
Next we determined if Ume6 was required for sporulation in K. lactis. We generated a homozygous ume6/ume6 diploid and examined the formation of tetrads using light microscopy at various time points after transfer to solid sporulation media. A wild-type diploid has visible tetrads already after 24 hr while after 72 hr ∼10% of the diploids have sporulated. In contrast, the ume6/ume6 diploid did not form any tetrads even after prolonged incubation (∼1 week). Visible inspection of cells did not rule out that a low frequency of sporulation was completed in the ume6/ume6 diploid. To further characterize this phenotype, we performed random spore analysis in which vegetative cells were killed by heat treatment. The number of cells in the original culture was determined by counting cells in a Bürkner chamber. This analysis revealed that sporulation occurred with a >100-fold reduced efficiency in the ume6/ume6 diploid compared to the wild-type diploid (Table 2). Interestingly, the viable cell count was almost 30-fold lower compared to the total cell count in the ume6/ume6 diploid strain before heat treatment, indicating that ume6/ume6 diploids were largely inviable after exposure to sporulation conditions. We concluded that Ume6 was required for sporulation in K. lactis.
TABLE 2.
Sporulation defect of ume6/ume6 diploids
| Genotype | Total cell count (cells/ml × 106) | Viable cell count (cells/ml × 106) | Viable cells after heat treatment (cells/ml × 106) |
|---|---|---|---|
| ume6/ume6 | 28 | 1.1 | 0.053 |
| UME6/UME6 | 26 | 16 | 7.6 |
Ume6 acts independently of Sin3 and Rpd3 in silencing of the cryptic mating-type loci and repression of meiotic genes:
S. cerevisiae Ume6 directly interacts with Sin3/Rpd3 in a large protein assembly known as the Rpd3L complex (Carrozza et al. 2005). We generated sin3 and rpd3 null mutant strains assuming that, if K. lactis Ume6 acted as part of an Rpd3L complex, then these null alleles would phenocopy one or several of the phenotypes displayed by ume6 strains. First, we explored the role of Sin3 and Rpd3 in silencing and repression of meiotic genes. Neither gene was required for silencing of HMLα (data not shown) or for repression of SPO69 transcription during vegetative growth (Figure 6B). Moreover, deletion of these genes did not suppress the mating defect of a MATa sir2 strain or generate mating defects in either a MATa or a MATα background (Figure 6D). Homozygous rpd3/rpd3 and sin3/sin3 diploid strains had a defect in successful tetrad production, however, indicating that these genes were required for some aspect of meiosis. Hence, Ume6 appeared to act independently of Sin3/Rpd3 in silencing of the cryptic mating-type loci and repression of SPO69 transcription during vegetative growth.
DISCUSSION
We explored the role of Ume6 in K. lactis with respect to transcriptional silencing of the HM-loci, repression of meiotic genes, and mating. Ume6 apparently had important roles during both mitosis and meiosis and, unexpectedly, a role in diploid cells to maintain the a/α-cell identity.
Our results strongly suggest a direct role for Ume6 in silencing of the cryptic mating-type loci. We have three arguments for this notion: (1) deletion of the UME6 gene led to derepression of both HM-loci; (2) both the HMLα and HMRa silencers contain Ume6-binding sites and the site present in the HMLα silencer was important for silencing (Sjöstrand et al. 2002); and (3) ChIP experiments showed that Ume6-13XMyc associated specifically with the silencers.
The silencers in K. lactis are bound by the Reb1 and Ume6 proteins as well as by an unknown protein that presumably binds to the C-box in the HMLα silencer (Sjöstrand et al. 2002). A major role for these proteins should be to recruit Sir proteins to the HM loci. However, we have been unable to demonstrate any direct interactions between Reb1/Ume6 and Sir2. We speculate that either the as-yet-unknown C-box-binding protein is responsible for Sir2 recruitment or the potential interaction between Reb1/Ume6 and Sir2 requires an unknown adaptor protein. K. lactis contains a functional Sir4 homolog, which also is required for efficient silencing (Åström and Rine 1998; Hickman and Rusche 2009). In two-hybrid experiments, we have failed to observe an interaction between Sir4 and Reb1/Ume6. The silencer-binding proteins in S. cerevisiae are ORC, Rap1, and Abf1. The differences between Kluyveromyces and Saccharomyces are apparent, and a reasonable hypothesis is that the nature of silencer-binding proteins evolves quickly. Consistent with this notion, it was recently observed that among Saccharomyces sensu stricto species the base-pair substitution rate at silenced chromatin, including HMRa and HMLα, was higher than in nonsilenced chromatin (Teytelman et al. 2008). It was suggested that this increased base-pair substitution rate was due to the effects of the silencing machinery on DNA replication or repair. How the differences in silencer-binding sites between Kluyveromyces and Saccharomyces developed is an interesting subject since silencing presumably was maintained at all stages. We speculate that other yeasts may utilize transition-state silencers compared to modern K. lactis and S. cerevisiae, as shown elegantly for cell-type-specific promoters by others (Tsong et al. 2006). Hence, it would be interesting to study HM silencers in other budding yeasts whose phylogeny suggest an intermediate position compared to S. cerevisiae and K. lactis.
Deletion of the UME6 gene resulted in a MATα-specific mating defect. This was largely due to the fact that MATα ume6 strains repressed hsgs. Deletion of HMRa did not completely suppress the mating defect of MATα ume6 strains, indicating that Ume6 had additional roles in MATα cells. We speculate that Ume6 was required for optimal levels of a factor promoting α-mating. In support of this idea, earlier studies in S. cerevisiae (Strich et al. 1994) showed that levels of α-factor were reduced in ume6 mutants through derepression of the a-specific gene BAR1, encoding an α-factor degrading enzyme.
The role of Ume6 in MATa-specific mating was complex. In MATa ume6 mutant strains, hsgs were not repressed even though HMLα was derepressed. This was most likely due to the relatively weak derepression of HMLα2. We showed that the level of the a1/α2 repressor was not high enough in the MATa ume6 mutant strain to induce repression of the hsgs MTS1 and STE4. Previously, we showed that base-pair substitutions in the Ume6-binding site of the HMLα silencer resulted in a strong mating defect of a MATa strain (Sjöstrand et al. 2002). However, in this experiment, we used a plasmid-based assay containing a minimal silencer fragment of only 102 bp. We speculate that, in the genomic context, removal of Ume6 results in only partial derepression of HMLα due to compensatory protein-binding sites outside of the 102-bp minimal silencer.
The suppression of the MATa sir2 mating defect by ume6 was unexpected and paralleled only by the SUM1-1 mutation in S. cerevisiae (Livi et al. 1990). Whereas SUM1-1 suppresses the mating defect in S. cerevisiae sir mutants by reinstating silencing (Laurenson and Rine 1991), ume6 suppressed the mating defect in K. lactis by acting on the downstream components of the mating pathway. In the mating-competent sir2 ume6 double-mutant strain, expression of hsgs was low and similar to the expression levels in the sterile sir2 mutant strain. Importantly, a ste4 ume6 double mutant was completely sterile, showing that the absence of Ume6 did not bypass the requirement for mating pheromone signaling, but rather facilitated mating in the presence of low levels of gene products from hsgs. Instead, the results indicate that the MATa sir2 ume6 double-mutant strain mates because the a2 and STE12 transcripts were overexpressed. Consistent with this idea, ectopic overexpression of Ste12 and a2 substantially improved mating of a MATa sir2 strain. There is an analogous observation in S. cerevisiae, where the diploid dig1 dig2 double-mutant strains override the a/α repression of hsgs and mate as a pseudo a-strain. Dig1 and Dig2, also known as Rst1 and Rst2, form complexes with Ste12, thus acting negatively on Ste12-dependent transcription of mating-specific genes (Olson et al. 2000). It was hypothesized that the elevated Ste12 activity in dig1 dig2 strains resulted in increased expression of a-specific genes. In addition, the Mcm1/α2 complex, which normally represses a-specific genes, is defective in the dig1 dig2 mutant strain, possibly as α2 is rendered unstable (Gelli 2002).
The MATa2 gene is essential for a-mating in K. lactis (data not shown) and is an activator of a-specific genes in Candida albicans (Tsong et al. 2003). We hypothesize that a2 activates a-specific genes also in K. lactis. Moreover, we suggest that increased transcription of both a2 and Ste12 leads to a pseudo a-mating phenotype even when the a1/α2 repressor is present. The reason for the synergistic increase in a2 and STE12 transcription in the double-mutant strain compared to the single-mutant strains remains unknown. A recent study (Hickman and Rusche 2009) showed that KlSir2 acts in both a promoter-specific and a long-range manner to regulate transcription. In addition, KlSir2 interacts with KlSum1 to repress transcription of both a subset of sporulation genes and cell-type-specific genes. It is hence possible that the synergistic effect observed by us is due to indirect regulatory effects.
Ume6 was required for the MATa/MATα cell identity and thus has an important function of acting as a barrier to polyploidy. We speculate that this function of Ume6 may be related to the deregulation of a2 and STE12. The ascomycetes can broadly be divided into two groups: one group that has gone through a whole-genome duplication followed by massive gene loss and another group that has not duplicated their genome (Kellis et al. 2004). K. lactis belongs to the latter group, and it is possible that the role of Ume6 in blocking polyploidization has contributed to this.
KlUme6 mediated repression of meiotic genes during vegetative growth, suggesting that this is a conserved role for Ume6 in many fungi. More unexpected was the observation that Ume6 acts independently of a potential Rpd3L complex (Carrozza et al. 2005), at least independently of the Rpd3 and Sin3 subunits, in mediating repression of meiotic genes and in regulating mating. It is possible that the recruitment of an Isw2-containing complex has a more important role in Ume6-mediated repression in K. lactis than it has in S. cerevisiae.
A remarkable finding was that HMRa2 was expressed despite the silenced HMRa1 gene sharing the regulatory region with HMRa2. It is hence possible that there are DNA sequences in this 850-bp intergenic region that insulate HMRa2 from the silencing machinery. Consistent with this idea, the silenced HMRa1 gene is proximal to the silencer sequence that presumably nucleates silent chromatin formation. In this context, it should be noted that the a2 transcript was very scarce in a wild-type MATa strain. The much higher levels of the a2 transcript observed in MATa sir2 and MATa sir2 ume6 strains indicate that the a2 gene is inducible by conditions not yet defined. Hence, the expression of HMRa2 that we observed is probably at a very low basal level that may result from incomplete silencing. In summary, we obtained evidence for a conserved function of Ume6 in yeasts in repressing meiotic gene expression, but found a unique role for K. lactis Ume6 in preserving the a/α-cell identity and in silencing of the cryptic mating-type loci.
Acknowledgments
We thank past and present members of the laboratory for scientific input. This study was supported by grants from the Swedish Research Council (2007-5516) and the Swedish Cancer Society (2007/808).
Supporting information is available online at http://www.genetics.org/cgi/content/full/genetics.110.114678/DC1.
References
- Anderson, S. F., C. M. Steber, R. E. Esposito and J. E. Coleman, 1995. UME6, a negative regulator of meiosis in Saccharomyces cerevisiae, contains a C-terminal Zn2Cys6 binuclear cluster that binds the URS1 DNA sequence in a zinc-dependent manner. Protein Sci. 4 1832–1843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Åström, S. U., and J. Rine, 1998. Theme and variation among silencing proteins in Saccharomyces cerevisiae and Kluyveromyces lactis. Genetics 148 1021–1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Åström, S. U., A. Kegel, J. O. Sjostrand and J. Rine, 2000. Kluyveromyces lactis Sir2p regulates cation sensitivity and maintains a specialized chromatin structure at the cryptic α-locus. Genetics 156 81–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ausubel, F. M., 1999. Short Protocols in Molecular Biology: A Compendium of Methods From Current Protocols in Molecular Biology. John Wiley & Sons, New York.
- Bahler, J., J. Q. Wu, M. S. Longtine, N. G. Shah, A. McKenzie, III et al., 1998. Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast 14 943–951. [DOI] [PubMed] [Google Scholar]
- Barsoum, E., P. Martinez and S. U. Åström, 2010. Alpha3, a transposable element that promotes host sexual reproduction. Genes Dev. 24 33–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brand, A. H., G. Micklem and K. Nasmyth, 1987. A yeast silencer contains sequences that can promote autonomous plasmid replication and transcriptional activation. Cell 51 709–719. [DOI] [PubMed] [Google Scholar]
- Buckingham, L. E., H. T. Wang, R. T. Elder, R. M. McCarroll, M. R. Slater et al., 1990. Nucleotide sequence and promoter analysis of SPO13, a meiosis-specific gene of Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 87 9406–9410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrozza, M. J., L. Florens, S. K. Swanson, W. J. Shia, S. Anderson et al., 2005. Stable incorporation of sequence specific repressors Ash1 and Ume6 into the Rpd3L complex. Biochim. Biophys. Acta 1731 77–87; discussion 75–76. [DOI] [PubMed] [Google Scholar]
- Chen, X. J., and G. D. Clark-Walker, 1994. sir2 mutants of Kluyveromyces lactis are hypersensitive to DNA-targeting drugs. Mol. Cell. Biol. 14 4501–4508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cliften, P., P. Sudarsanam, A. Desikan, L. Fulton, B. Fulton et al., 2003. Finding functional features in Saccharomyces genomes by phylogenetic footprinting. Science 301 71–76. [DOI] [PubMed] [Google Scholar]
- Cockell, M., F. Palladino, T. Laroche, G. Kyrion, C. Liu et al., 1995. The carboxy termini of Sir4 and Rap1 affect Sir3 localization: evidence for a multicomponent complex required for yeast telomeric silencing. J. Cell Biol. 129 909–924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engebrecht, J., and G. S. Roeder, 1990. MER1, a yeast gene required for chromosome pairing and genetic recombination, is induced in meiosis. Mol. Cell. Biol. 10 2379–2389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fabre, E., H. Muller, P. Therizols, I. Lafontaine, B. Dujon et al., 2005. Comparative genomics in hemiascomycete yeasts: evolution of sex, silencing, and subtelomeres. Mol. Biol. Evol. 22 856–873. [DOI] [PubMed] [Google Scholar]
- Galgoczy, D. J., A. Cassidy-Stone, M. Llinas, S. M. O'Rourke, I. Herskowitz et al., 2004. Genomic dissection of the cell-type-specification circuit in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 101 18069–18074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner, K. A., and C. A. Fox, 2001. The Sir1 protein's association with a silenced chromosome domain. Genes Dev. 15 147–157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelli, A., 2002. Rst1 and Rst2 are required for the a/alpha diploid cell type in yeast. Mol. Microbiol. 46 845–854. [DOI] [PubMed] [Google Scholar]
- Ghidelli, S., D. Donze, N. Dhillon and R. T. Kamakaka, 2001. Sir2p exists in two nucleosome-binding complexes with distinct deacetylase activities. EMBO J. 20 4522–4535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldmark, J. P., T. G. Fazzio, P. W. Estep, G. M. Church and T. Tsukiyama, 2000. The Isw2 chromatin remodeling complex represses early meiotic genes upon recruitment by Ume6p. Cell 103 423–433. [DOI] [PubMed] [Google Scholar]
- Goldstein, A. L., and J. H. McCusker, 1999. Three new dominant drug resistance cassettes for gene disruption in Saccharomyces cerevisiae. Yeast 15 1541–1553. [DOI] [PubMed] [Google Scholar]
- Hecht, A., T. Laroche, S. Strahl-Bolsinger, S. M. Gasser and M. Grunstein, 1995. Histone H3 and H4 N-termini interact with SIR3 and SIR4 proteins: a molecular model for the formation of heterochromatin in yeast. Cell 80 583–592. [DOI] [PubMed] [Google Scholar]
- Hecht, A., S. Strahl-Bolsinger and M. Grunstein, 1996. Spreading of transcriptional repressor SIR3 from telomeric heterochromatin. Nature 383 92–96. [DOI] [PubMed] [Google Scholar]
- Herskowitz, I., 1988. Life cycle of the budding yeast Saccharomyces cerevisiae. Microbiol. Rev. 52 536–553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hickman, M. A., and L. N. Rusche, 2009. The Sir2-Sum1 complex represses transcription using both promoter-specific and long-range mechanisms to regulate cell identity and sexual cycle in the yeast Kluyveromyces lactis. PLoS Genet. 5 e1000710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imai, S., C. M. Armstrong, M. Kaeberlein and L. Guarente, 2000. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature 403 795–800. [DOI] [PubMed] [Google Scholar]
- Kadosh, D., and K. Struhl, 1997. Repression by Ume6 involves recruitment of a complex containing Sin3 corepressor and Rpd3 histone deacetylase to target promoters. Cell 89 365–371. [DOI] [PubMed] [Google Scholar]
- Kadosh, D., and K. Struhl, 1998. Targeted recruitment of the Sin3-Rpd3 histone deacetylase complex generates a highly localized domain of repressed chromatin in vivo. Mol. Cell. Biol. 18 5121–5127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawasaki, L., A. L. Savinon-Tejeda, L. Ongay-Larios, J. Ramirez and R. Coria, 2005. The Gbeta(KlSte4p) subunit of the heterotrimeric G protein has a positive and essential role in the induction of mating in the yeast Kluyveromyces lactis. Yeast 22 947–956. [DOI] [PubMed] [Google Scholar]
- Kegel, A., P. Martinez, S. D. Carter and S. U. Åström, 2006. Genome wide distribution of illegitimate recombination events in Kluyveromyces lactis. Nucleic Acids Res. 34 1633–1645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kellis, M., N. Patterson, M. Endrizzi, B. Birren and E. S. Lander, 2003. Sequencing and comparison of yeast species to identify genes and regulatory elements. Nature 423 241–254. [DOI] [PubMed] [Google Scholar]
- Kellis, M., B. W. Birren and E. S. Lander, 2004. Proof and evolutionary analysis of ancient genome duplication in the yeast Saccharomyces cerevisiae. Nature 428 617–624. [DOI] [PubMed] [Google Scholar]
- Landry, J., A. Sutton, S. T. Tafrov, R. C. Heller, J. Stebbins et al., 2000. The silencing protein SIR2 and its homologs are NAD-dependent protein deacetylases. Proc. Natl. Acad. Sci. USA 97 5807–5811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laurenson, P., and J. Rine, 1991. SUM1–1: a suppressor of silencing defects in Saccharomyces cerevisiae. Genetics 129 685–696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine, M., and R. Tjian, 2003. Transcription regulation and animal diversity. Nature 424 147–151. [DOI] [PubMed] [Google Scholar]
- Livi, G. P., J. B. Hicks and A. J. Klar, 1990. The sum1–1 mutation affects silent mating-type gene transcription in Saccharomyces cerevisiae. Mol. Cell. Biol. 10 409–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malathi, K., Y. Xiao and A. P. Mitchell, 1997. Interaction of yeast repressor-activator protein Ume6p with glycogen synthase kinase 3 homolog Rim11p. Mol. Cell. Biol. 17 7230–7236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mallory, M. J., K. F. Cooper and R. Strich, 2007. Meiosis-specific destruction of the Ume6p repressor by the Cdc20-directed APC/C. Mol. Cell 27 951–961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moazed, D., A. Kistler, A. Axelrod, J. Rine and A. D. Johnson, 1997. Silent information regulator protein complexes in Saccharomyces cerevisiae: a SIR2/SIR4 complex and evidence for a regulatory domain in SIR4 that inhibits its interaction with SIR3. Proc. Natl. Acad. Sci. USA 94 2186–2191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moretti, P., K. Freeman, L. Coodly and D. Shore, 1994. Evidence that a complex of SIR proteins interacts with the silencer and telomere-binding protein RAP1. Genes Dev. 8 2257–2269. [DOI] [PubMed] [Google Scholar]
- Olson, K. A., C. Nelson, G. Tai, W. Hung, C. Yong et al., 2000. Two regulators of Ste12p inhibit pheromone-responsive transcription by separate mechanisms. Mol. Cell. Biol. 20 4199–4209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rine, J., and I. Herskowitz, 1987. Four genes responsible for a position effect on expression from HML and HMR in Saccharomyces cerevisiae. Genetics 116 9–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothstein, R. J., 1983. One-step gene disruption in yeast. Methods Enzymol. 101 202–211. [DOI] [PubMed] [Google Scholar]
- Rubin-Bejerano, I., S. Mandel, K. Robzyk and Y. Kassir, 1996. Induction of meiosis in Saccharomyces cerevisiae depends on conversion of the transcriptional represssor Ume6 to a positive regulator by its regulated association with the transcriptional activator Ime1. Mol. Cell. Biol. 16 2518–2526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rundlett, S. E., A. A. Carmen, N. Suka, B. M. Turner and M. Grunstein, 1998. Transcriptional repression by UME6 involves deacetylation of lysine 5 of histone H4 by RPD3. Nature 392 831–835. [DOI] [PubMed] [Google Scholar]
- Rusche, L. N., A. L. Kirchmaier and J. Rine, 2002. Ordered nucleation and spreading of silenced chromatin in Saccharomyces cerevisiae. Mol. Biol. Cell 13 2207–2222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rusche, L. N., A. L. Kirchmaier and J. Rine, 2003. The establishment, inheritance, and function of silenced chromatin in Saccharomyces cerevisiae. Annu. Rev. Biochem. 72 481–516. [DOI] [PubMed] [Google Scholar]
- Schiestl, R. H., and R. D. Gietz, 1989. High efficiency transformation of intact yeast cells using single stranded nucleic acids as a carrier. Curr. Genet. 16 339–346. [DOI] [PubMed] [Google Scholar]
- Shore, D., and K. Nasmyth, 1987. Purification and cloning of a DNA binding protein from yeast that binds to both silencer and activator elements. Cell 51 721–732. [DOI] [PubMed] [Google Scholar]
- Sjöstrand, J. O., A. Kegel and S. U. Åström, 2002. Functional diversity of silencers in budding yeasts. Eukaryot. Cell 1 548–557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith, J. S., C. B. Brachmann, I. Celic, M. A. Kenna, S. Muhammad et al., 2000. A phylogenetically conserved NAD+-dependent protein deacetylase activity in the Sir2 protein family. Proc. Natl. Acad. Sci. USA 97 6658–6663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steber, C. M., and R. E. Esposito, 1995. UME6 is a central component of a developmental regulatory switch controlling meiosis-specific gene expression. Proc. Natl. Acad. Sci. USA 92 12490–12494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strich, R., R. T. Surosky, C. Steber, E. Dubois, F. Messenguy et al., 1994. UME6 is a key regulator of nitrogen repression and meiotic development. Genes Dev. 8 796–810. [DOI] [PubMed] [Google Scholar]
- Teytelman, L., M. B. Eisen and J. Rine, 2008. Silent but not static: accelerated base-pair substitution in silenced chromatin of budding yeasts. PLoS Genet. 4 e1000247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Triolo, T., and R. Sternglanz, 1996. Role of interactions between the origin recognition complex and SIR1 in transcriptional silencing. Nature 381 251–253. [DOI] [PubMed] [Google Scholar]
- Tsong, A. E., M. G. Miller, R. M. Raisner and A. D. Johnson, 2003. Evolution of a combinatorial transcriptional circuit: a case study in yeasts. Cell 115 389–399. [DOI] [PubMed] [Google Scholar]
- Tsong, A. E., B. B. Tuch, H. Li and A. D. Johnson, 2006. Evolution of alternative transcriptional circuits with identical logic. Nature 443 415–420. [DOI] [PubMed] [Google Scholar]
- Washburn, B. K., and R. E. Esposito, 2001. Identification of the Sin3-binding site in Ume6 defines a two-step process for conversion of Ume6 from a transcriptional repressor to an activator in yeast. Mol. Cell. Biol. 21 2057–2069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whiteway, M., L. Hougan, D. Dignard, D. Y. Thomas, L. Bell et al., 1989. The STE4 and STE18 genes of yeast encode potential beta and gamma subunits of the mating factor receptor-coupled G protein. Cell 56 467–477. [DOI] [PubMed] [Google Scholar]
- Williams, R. M., M. Primig, B. K. Washburn, E. A. Winzeler, M. Bellis et al., 2002. The Ume6 regulon coordinates metabolic and meiotic gene expression in yeast. Proc. Natl. Acad. Sci. USA 99 13431–13436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao, Y., and A. P. Mitchell, 2000. Shared roles of yeast glycogen synthase kinase 3 family members in nitrogen-responsive phosphorylation of meiotic regulator Ume6p. Mol. Cell. Biol. 20 5447–5453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu, L., M. Ajimura, R. Padmore, C. Klein and N. Kleckner, 1995. NDT80, a meiosis-specific gene required for exit from pachytene in Saccharomyces cerevisiae. Mol. Cell. Biol. 15 6572–6581. [DOI] [PMC free article] [PubMed] [Google Scholar]