The authors describe their study on the role of CCR7-mediated, donor-derived antigen-presenting cells trafficking in high-risk corneal transplantation and demonstrate that not only is the CCR7-facilitated direct pathway important in the initiation of host T-cell priming, it is crucial for Treg-mediated tolerance.
Abstract
Purpose.
Chemokine receptor 7 (CCR7) is a key homing molecule for immune cell trafficking, including corneal antigen-presenting cell (APC) migration from the inflamed cornea to draining lymph nodes (LNs). Here, the authors investigated the effect of CCR7-facilitated donor APC trafficking on allosensitization, regulatory T-cell (Treg) function, and graft survival in corneal transplantation.
Methods.
CCR7−/− or wild-type (WT) allogeneic corneal grafts were transplanted onto the neovascularized high-risk recipient beds. Two weeks later, the frequency of directly alloprimed host T cells was measured by the IFN-γ ELISPOT assay. Treg function was tested by a coculture suppression assay and an IFN-γ ELISPOT assay. Kaplan-Meier analysis was performed to evaluate graft survival.
Results.
The recipients of CCR7−/− grafts had fewer migrated donor APCs and lower frequency of IFN-γ–producing T cells in the draining LNs. However, there was no statistically significant difference in transplant survival between recipients of CCR7−/− and those of WT grafts. Tregs from the CCR7−/− graft recipient group showed reduced regulatory potential for the suppression of proliferation of naive T cells and direct alloprimed T cells and expressed lower Foxp3 levels. In vitro studies confirmed that mature CCR7+ major histocompatibility complex class II+ CD86+ graft-derived dendritic cells were critical for Treg function.
Conclusions.
Not only is CCR7-mediated donor-derived APC trafficking to the draining LNs important in the initiation of host T-cell priming, it is crucial for Treg-mediated tolerance.
Alloantigens from grafted tissues are recognized by host T cells through their direct or indirect presentation by antigen-presenting cells (APCs) in the draining lymph nodes (LNs). The direct pathway involves the recognition of intact donor major histocompatibility complex (MHC) molecules on donor-derived APCs, whereas the indirect pathway involves recognition of donor major or minor MHC-derived peptides processed and presented by recipient APCs to host T cells.
Until recently, corneal grafts were considered entirely devoid of resident APCs, giving rise to the tenet that alloantigen recognition is mediated exclusively by the indirect pathway. Recently, however, resident MHC class II− APC populations were identified in the cornea.1,2 These cells are capable of expressing MHC class II antigen and accessory molecules (CD40, CD80) after transplantation and of migrating to draining LNs of allograft recipients, where they can prime host T cells through the direct pathway, especially in recipients of high-risk grafts (i.e., transplantations performed in inflamed host beds).3–5 This is important because although the survival rate of grafts performed onto avascular and uninflamed low-risk host beds approaches 90% under cover of topical steroids, well more than 50% of corneal grafts performed onto high-risk graft beds are rejected regardless of the immunosuppressive strategy used.6 Given that corneal transplants constitute the most frequent form of allograft performed and that allorejection remains a significant clinical problem, development of a strategy that could promote corneal graft survival without altering host immune competence is a priority.
Chemokine receptor 7 (CCR7) is an essential homing molecule that controls APC and T-cell migration to secondary lymphoid organs.7 We have previously demonstrated that CCR7 is expressed by CD11b+CD11c+MHC class II+ APCs in the inflamed cornea and that it mediates trafficking of these cells to the draining regional LNs.8 Thus, we hypothesized that CCR7 facilitates allorecognition in corneal transplantation and could be an important target for promoting graft survival. Indeed, the effects of targeting CCR7 on promoting graft survival have been demonstrated in several transplantation models, with results varying from limited to marked and even to permanent prolongation of graft survival.9–11 However, these studies used plt mice as recipients, CCL19-IgG administration to hosts, or CCR7−/− grafted tissue to CCR7−/− recipients. Although these studies have provided significant insight into CCR7-mediated processes, they could not be used to exclusively study the effect of CCR7 on direct allosensitization because their approaches affect the homing of both APCs and T cells.
In the present study using a high-risk corneal transplantation model in which CCR7−/− donor tissue was grafted onto wild-type (WT) hosts, we demonstrated that CCR7 has paradoxical effects on graft immunity. Although it mediates donor APC migration to host LN and T-cell priming, it also promotes the generation of Foxp3+ Tregs, suggesting dual roles that can promote or suppress graft longevity.
Materials and Methods
Animals
Six- to 8-week-old male BALB/c and C57BL/6 (Taconic Farms, Germantown, NY) mice were used in all experiments. CCR7−/− C57BL/6 mice were provided by Andrew Luster (Massachusetts General Hospital, Boston, MA) and Martin Lipp (Maz-Delbrueck-center of Molecular Medicine, Berlin, Germany). They were housed in a specific pathogen-free environment at the Schepens Eye Research Institute animal facility. All procedures were approved by the Institutional Animal Care and Use Committee, and all animals were treated according to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research.
High-Risk Orthotopic Corneal Transplantation
The well-established procedures for corneal high-risk transplantation have been described previously.3,12 Briefly, three interrupted 8-shaped sutures (11–0 nylon, 50-μm diameter needle; Sharpoint; Angiotech, Vancouver, BC, Canada) were placed in the central corneas of BALB/c mice to induce inflamed and neovascularized high-risk graft beds 14 days before orthotopic corneal transplantation. The central 2-mm diameter corneal grafts were excised from CCR7−/− or WT C57BL/6 mice with a trephine (Storz Instrument Co., St. Louis, MO) and microscissors. The recipient bed was prepared by excising 1.5-mm tissue from the central corneas of BALB/c host mice. The prepared donor corneal grafts were then transplanted onto the host bed with eight interrupted 11–0 nylon sutures. The sutures were removed 7 days after transplantation. All grafts were evaluated using slit-lamp biomicroscopy weekly over 8 weeks. Grafts were defined as rejected when they became opaque and the iris details could not be recognized clearly using a standardized opacity-grading (range, 0–5) scheme.13
Flow Cytometry
Ipsilateral draining submandibular and cervical LNs were harvested at 48 hours after transplantation. Based on our previous studies, 48 hours after HR transplantation is the peak time to identify donor-derived APCs in host draining LNs.3 Single-cell suspensions of these LNs or bone marrow–derived dendritic cells (BMDCs) were prepared. The isolated cells were stained with the following antibodies: anti–CCR7 PerCP-Cy5.5, anti–CD86 fluorescein isothiocyanate (FITC; BD PharMingen, San Jose, CA), anti–CD3 FITC, anti–Iab phycoerythrin (PE; BD PharMingen), anti–CD4 FITC, anti–CD25 PE, and anti–forkhead box P3 (FoxP3) PECy5. Except where noted, primary and isotype-matched antibodies were purchased from eBioscience (San Diego, CA). After staining, the cells were analyzed on a flow cytometer (Beckman Coulter).
Cell Sorting
T cells, CD4+CD25− effector T cells (Teff), and CD4+CD25+ regulatory T cells (Treg) from the LNs of different graft recipients or naive mice were isolated by magnetic separation using CD90+ cells, CD4+ T cells, and Treg isolation kits (Miltenyi Biotec, Auburn, CA). More than 97% sorted CD90+ cells were CD3+, and more than 95% CD4+CD25+ Treg were Foxp3+ in all the groups, as confirmed by flow cytometry.
Real-Time PCR
RNA was isolated (RNeasy Micro Kit; Qiagen, Valencia, CA) and reverse transcribed (Superscript III Kit; Invitrogen, Carlsbad, CA). Real-time PCR was performed using a PCR mix (TaqMan Universal PCR Mastermix; Invitrogen) and preformulated primers for FoxP3 (assay ID Mm00475156_ml) and GAPDH (assay ID Mm99999915_gl) (all Applied Biosystems, Austin, TX). Results were analyzed by the comparative threshold cycle method and normalized by GAPDH as an internal control.
ELISPOT Assay
The ELISPOT assay was performed to delineate the contribution of direct allosensitization to graft immunity, as described previously.14 Briefly, 96-well ELISPOT plates (Whatman Polyfiltronics, Rockland, MA) were coated with 4 g/mL primary anti–IFN-γ mAb (BD PharMingen) in sterile PBS overnight. The plates were then washed three times with PBS and blocked for 1.5 hours with PBS containing 1% BSA. Next, responder T cells were isolated from the draining LN ipsilateral to the grafted eyes (n = 8) and were added (5 × 105) to wells previously loaded with irradiated donor splenocytes as APCs (5 × 105) in a final volume of 200 μL AIM-V medium. T cells harvested from LNs of ungrafted animals served as controls. To test Treg suppression function, additional Treg (2.5 × 105) from each group were loaded. To isolate the CD4-mediated response, 25 μg/mL anti–CD8 (53–6.72) mAb (BD PharMingen) were used.4,5 Cells were incubated for 48 hours. The plates were washed three times with PBS, then four times with PBS containing 0.025% Tween 20. Biotinylated anti–IFN-γ detection mAbs were added at 2 μg/mL (BD PharMingen) and were incubated for 2 hours at room temperature. The washing steps were repeated; after 1 hour of incubation with avidin-HRP, the plates were washed again three times with PBS/0.025% Tween 20 and then three times with PBS alone. The spots were developed by the addition of the amino ethylcarbazole staining solution (Sigma-Aldrich, St. Louis, MO). The resultant spots were counted and analyzed on a computer-assisted ELISPOT image analyzer (Cellular Technology Ltd., Shaker Heights, OH).
Generation of Bone Marrow-Derived Dendritic Cells
BMDCs were generated as described by Lutz et al.15 Briefly, 6- to 8-week-old mice were humanely killed, and femurs were isolated. BM cells (2 × 106) collected from femurs were seeded in Petri dishes in 10 mL culture medium (RPMI-1640 supplemented with 10% fetal calf serum, 2 mM l-glutamine, 100 U/mL penicillin, 100 μg/mL streptomycin, and 50 mM 2-ME) containing 200 U/mL granulocyte macrophage colony-stimulating factor (GM-CSF; PeproTech, Rocky Hill, NJ). Medium was changed every 2 days. On day 5 or 6, nonadherent and loosely adherent cells were harvested as immature BMDCs. To generate mature BMDCs, 50 ng/mL TNF-α (R&D Systems, Minneapolis, MN) was added 24 hours before cell harvesting. The purity of the DCs, determined by flow analysis of surface CD11c staining, was greater than 97%. The maturity of the DCs was determined by flow analysis of surface CD86 staining.
Suppression Assay
As described previously,16 T cells (1 × 105) were cocultured with CD4+CD25+ Treg (5 × 104), T cell-depleted syngeneic splenocytes (1 × 105), and 1 μg/mL anti–CD3 antibody for 3 days. To measure the allospecificity of Treg, CD4+CD25− T cells (Teff, 1 × 105) and Treg (5 × 104) were cocultured with allogeneic BMDCs without anti–CD3 stimulation. We tested three different ratios of BMDC/Treg (1:1, 1:5, 1:10); accordingly, the number of BMDCs was 5 × 104, 1 × 104, and 5 × 103 in different groups. The proliferation of stimulated T cells without the addition of Treg was considered control proliferation with 0% suppression. Proliferation was measured using the BrdU incorporation assay (Millipore, Billerica, MA), and percentage suppression was calculated using the following formula: % suppression = [(T-cell proliferation without Treg − T cell proliferation with Treg)/T-cell proliferation without Treg] × 100.
Statistical Analysis
Data were presented as mean ± SEM and analyzed by the two-tailed Student's t-test (Prism, version 4.0; GraphPad, San Diego, CA) between groups. Values were considered statistically significant at P < 0.05. Kaplan-Meier analysis was adopted to construct survival curves, and the log-rank test was used to compare the rates of corneal graft survival in different settings.
Results
The Impairment by Deletion of CCR7 Expression on Donor-Derived APC
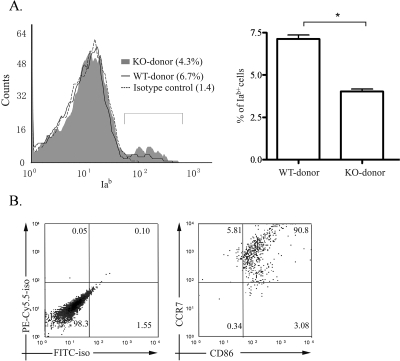
We analyzed the percentage of Iab+ (donor MHC class II) cells among the CD3− cell population in the draining LNs of BALB/c recipients of corneal transplants (Iad+) at 48 hours after corneal transplantation (Fig. 1). Compared with the group receiving WT grafts, the group with CCR7−/− grafts had a 43% lower frequency of donor MHC class II+ cells (Fig. 1A). Flow cytometric analysis of LN cells of recipients with WT grafts also showed that most donor MHC class II+ (Iab) cells expressed both CCR7 and CD86 (Fig. 1B), suggesting that CCR7 was involved in the migration of mature MHC class IIhigh CD86+ donor APCs.
Figure 1.
Comparison of donor MHC class II+ cells in the recipient draining LNs between recipient groups based on donor CCR7 expression. Draining LNs from the recipients were harvested 48 hours after high-risk transplantation. (A) Total LN cells from hosts were isolated and stained with anti–mouse CD3 FITC and anti–mouse Iab PE or isotype control 48 hours after high-risk corneal transplantation. During gating on the CD3− cell population, the percentages of donor-derived Iab+ cells were compared between the WT and the CCR7−/− donor groups. Left: one representative of three independent experiments is shown here. Right: values are expressed as the percentage of Iab+ cells in a CD3− cell population of the host LNs. Results represent the mean (± SEM) of three samples per group (each sample consisted of LNs from three mice; *P < 0.01, t-test). (B) From the recipients with WT donors, total LN cells were isolated and stained with anti–CD86 FITC, anti–Iab PE, and anti–CCR7 PE-Cy5.5. During gating on Iab+ cell population, most donor APCs were CCR7+CD86+. Left: isotype control. Results shown represent 1 of 3 independent experiments.
Suppression of Direct Allosensitization by Deletion of CCR7 Expression on Donor-Derived APC
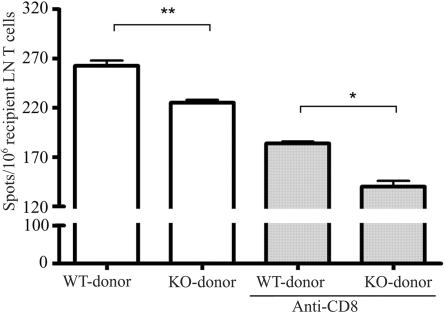
To determine the influence of CCR7−/− donor tissue on direct allosensitization, we investigated the frequency of IFN-γ–producing cells 14 days after surgery (a time point just preceding the manifestation of clinical rejection in the murine model). As shown in Figure 2, a significant reduction in the directly primed IFN-γ response was generated from the CCR7−/− donor group compared with the WT donor group. Anti–CD8 antibody was used to investigate the role of CCR7 in the direct priming of CD4+ versus CD8+ host T cells. When limiting the directly primed T cell response to the CD4+ compartment, we observed an even greater (20% vs. 13%) reduction in the direct response, suggesting that the depletion of CCR7-mediated donor APC trafficking led to an inhibition of recipient T cell priming, especially among host CD4+ T cells.
Figure 2.
The direct pathway of allorecognition in corneal graft recipients with CCR7−/− or WT allografts. Responder T cells were isolated from the draining LNs of graft recipients with WT or CCR7−/− allografts (n = 10) 14 days after transplantation and were stimulated with C57BL/6 splenic APCs. The frequency of directly primed T cells in grafted mice was measured by the IFN-γ ELISPOT assay. Anti–CD8 antibody was added to detect CD4+ T-cell reactivity. Results are depicted as the number (mean ± SEM) of spots per million responder T cells loaded and represent 1 of 3 independent experiments (*P < 0.05; **P < 0.01).
No Effect on Transplant Survival by Deletion of CCR7 Expression on Donor-Derived APC
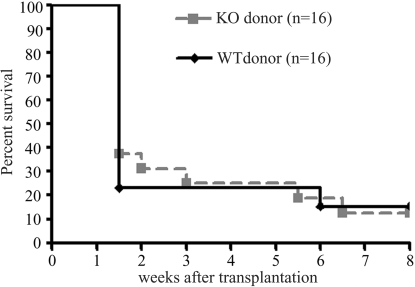
To test the effect of eliminating CCR7-facilitated direct allorecognition in high-risk graft survival, CCR7−/− C57BL/6 donor grafts were placed onto vascularized high-risk BALB/c beds. Corneas from WT C57BL/6 grafted onto high-risk BALB/c beds served as controls (Fig. 3). At the early time point (10–14 days), CCR7−/− grafts had a modestly higher survival rate (30%–40%) than WT grafts (20%). However, by 3 weeks, there was no difference in graft survival between the two groups, suggesting that the deletion of CCR7-facilitated direct allorecognition may only lead to a slight delay in corneal graft rejection.
Figure 3.
Fate of CCR7−/− or WT corneal allografts placed in high-risk vascularized host beds. BALB/c mice were grafted orthotopically with high-risk corneal allografts from either CCR7−/− (n = 16) or WT (n = 16) C57BL/6 mice and were scored clinically for 8 weeks. Data are reported as Kaplan-Meier survival curves.
Effects on Suppressor Function of Host Regulatory T Cells by Deletion of CCR7 Expression on Donor-Derived APC
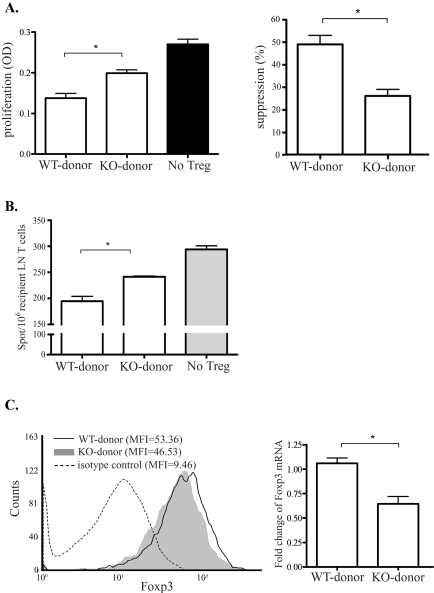
To analyze the regulatory potential of Tregs generated in the hosts of CCR7−/− compared with WT donors, we first tested the suppression of CD4+CD25+ Tregs on the proliferation of CD3-stimulated naive T cells by using a coculture suppression assay at 2 weeks after transplantation (Fig. 4). Tregs isolated from the CCR7−/− donor group showed significantly (46%) lower efficacy in suppressing the proliferation of naive T cells than those isolated from the WT donor group (26% vs. 49%; P = 0.01; Fig. 4A). We further studied the Treg suppressor function on directly primed T cells from WT graft recipients at high risk using the IFN-γ-ELISPOT assay. Similar to the suppressive effect on naive T-cell proliferation, the Tregs isolated from the CCR7−/− donor group showed a significantly (20%) lower potential to suppress the IFN-γ secretion of directly primed T cells compared with those isolated from the WT donor group (P = 0.03, Fig. 4B).
Figure 4.
Comparison of recipients' Treg function based on donor tissue CCR7 expression. Tregs (CD4+CD25+) were sorted from the draining LNs of recipients at 2 weeks after transplantation to evaluate their functionality. (A) Treg suppression assay showing the level of CD3-mediated proliferation of naive BALB/c CD4+ T cells with the Tregs from different recipient groups using BrdU assay (left). Accordingly, the suppression effect of these Treg was calculated as % suppression = [(T-cell proliferation without Treg − T-cell proliferation with Treg)/T-cell proliferation without Treg] × 100 (right). Results are depicted as the percentage of suppression (mean ± SEM) and represent the results of 1 of 3 independent experiments (*P < 0.01). (B) IFN-γ ELISPOT assay showing frequencies of directly primed T cells in different graft recipients. Responder T cells from the recipients at high risk with WT allografts were cocultured with the Tregs isolated from the recipients of CCR7−/− or WT allografts and were stimulated with C57BL/6 APCs. The results are depicted as the number (mean ± SEM) of spots per million responder T cells loaded and represent the results 1 of 3 independent experiments (*P < 0.05). (C) CD4+CD25+ Tregs isolated from the draining LNs of different graft recipient groups. Left: flow cytometric histogram showing expression levels (mean fluorescence intensity) of Foxp3 in these Tregs. Right: values are expressed as the fold change of Foxp3 mRNA expression levels of these Tregs in the CCR7−/− donor group over the WT donor group, normalized to GAPDH expression. Results represent the mean (± SEM) of four samples per group (each sample consisted of 1 LN), and the data are representative of two independent experiments (*P < 0.01).
Foxp3 expression levels are critical in defining regulatory activity16; therefore, we measured Foxp3 expression levels in the Tregs from each group to explain the difference in Treg function. Flow cytometric analysis of LN cell suspensions showed that CD4+CD25+ cells from the CCR7−/− donor group had a lower Foxp3 expression level (mean fluorescence [MFI], 46) than those from the WT donor group (MFI, 53). This result was confirmed by our real-time PCR result, which showed that CD4+CD25+ cells from the CCR7−/− donor group were expressing nearly half the levels of Foxp3 mRNA compared with cells from the WT donor group (Fig. 4C).
Promotion of Suppressor Function of Host Tregs by Donor Tissue–Derived Mature APC
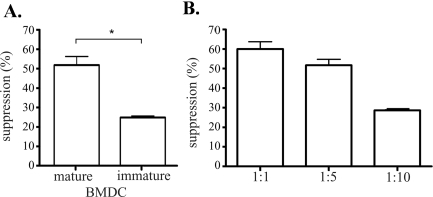
Because we demonstrated that CCR7 is important for the migration of mature donor APCs though we observed no enhancement of survival among CCR7−/− grafts, we hypothesized that not only are mature APCs relevant for allosensitization, they may play a critical role in promoting Treg function in high-risk corneal transplantation. To test this hypothesis, we compared the potential of mature BMDCs (MHC class IIhighCD86+) and immature BMDCs (MHC class IIlowCD86−) to promote Treg function in an allogeneic setting using an in vitro coculture assay (Fig. 5). C57BL/6 mature- or immature BMDCs and naive BALB/c CD4+CD25− T cells (Teff) were cocultured with CD4+CD25+ Tregs at indicated DC/Treg cell ratios. Tregs cocultured with mature BMDCs showed a higher suppressive effect on Teff than did those cocultured with immature BMDCs (Fig. 5A). In addition, Tregs cocultured with mature BMDCs at different cell ratios showed a direct relationship between their suppressor function and the numbers of mature BMDCs present in the culture (Fig. 5B). These results clearly demonstrate that mature BMDCs promote Treg suppressor function, supporting our hypothesis that CCR7-mediated trafficking of graft-derived mature APCs to draining LNs are essential for Treg function.
Figure 5.
Mature dendritic cells facilitate Treg suppressor function. (A) Mature or immature BMDCs were cocultured with allogeneic CD4+CD25− Teff and CD4+CD25+ Treg from naive mice. Total cell number of DC/Treg/Teff is 1:5:10. Treg suppression potency was compared between mature and immature BMDC groups. (B) Treg suppressor potential was also tested at different mature BMDC/Treg cell number ratios. Total cell number of Treg/Teff is 1:2. The x-axis shows the different ratios of mature BMDC/Treg. Results are depicted as the percentage of suppression (mean ± SEM) and represent the results of 1 of 3 independent experiments (*P < 0.05).
Discussion
Allorecognition is the result of host T cells detecting non–self-antigens by either the direct or the indirect pathway. Here, we report the relevance of CCR7-dependent donor-APC trafficking to the host lymph nodes on T-cell allosensitization and Treg function.
In this study, we first demonstrated a significant reduction in the frequency of donor MHC class II+ cells in the draining LN of CCR7−/− graft recipients, suggesting that CCR7 deletion impairs donor-derived APC migration to the host draining LNs. This impairment led to a blunted expansion of directly alloprimed IFN-γ–secreting Th1 cells in the draining LNs of recipients with CCR7−/− grafts compared with those in the WT donor control group. IFN-γ–secreting cells have been associated with effecting corneal transplant destruction at 2 weeks, and they correlate with the acquisition of donor-specific, delayed-type hypersensitivity.4 The Th1-type response represents the dominant effector T-cell population for acute rejection of corneal transplants.13,17,18 Indeed, the deletion of directly primed CD4+ Th1 cells by the use of MHC class II knockout donor grafts has shown the promotion of graft survival in high-risk corneal transplantation.3 These data led us to hypothesize that CCR7 deletion in the graft would lead to prolonged corneal transplant survival.
Interestingly, though we noted a slight delay in the rejection rate of CCR7 −/− grafts, the overall rate of rejection was nearly identical (∼80%) to that of WT grafts. We had observed only a 40% to 50% reduction in graft-derived APC trafficking from CCR7 −/− grafts compared with from WT grafts (Fig. 1); nevertheless, the fact that the overall graft rejection rate was no different from that observed with WT donor tissue led us to hypothesize that CCR7 function is potentially also relevant for the induction of tolerance.
Recent studies have shown that oral tolerance cannot be induced in CCR7-deficient mice that display impaired migration of DCs from the intestine to the mesenteric lymph nodes, and small intestinal lamina propria-DCs induce T cells to produce IL-4 and IL-10 by presenting intestinal epithelial cell-associated antigen in a CCR7-dependent manner.19,20 CD4+CD25+ Tregs, which constitute 5% to 10% of CD4+ T cells, have been shown to play a central role in inducing and maintaining tolerance in a number of clinical settings and animal models of transplantation.21–24 Our data demonstrate that Tregs isolated from the draining LNs of recipients receiving CCR7−/− grafts have a lower potential for suppressing the proliferation of naive T cells and alloprimed T cells. In addition, these Tregs express lower levels of Foxp3, an important observation; far more relevant than Treg frequency is their level of Foxp3 expression, which directly correlates with their potential for suppressing effector T cells.15 These observations suggest that CCR7-mediated pathways are relevant not only in the allopriming of CD4+ T cells but in establishing the regulatory effect of Tregs on these effector T cells.
CCR7 governs the migration of both semi-mature (MHC class II+ CD86low) and mature (MHC class II+ CD86high) DCs to the lymphoid tissues. These DC populations have also been shown to be crucially involved in the induction of peripheral tolerance.25 Under noninflammatory steady state conditions, semi-mature DCs induce alloantigen-specific T-cell anergy and derive de novo differentiation of naturally occurring Tregs from CD4+ T cells.26 Here, our results showed that most WT donor-derived APCs in the host draining LNs are mature and are of the MHC class II+ CD86highCCR7+ phenotype. In addition, our data from in vitro coculture experiments that mimic the interaction among DC, Tregs, and T cells in the lymphoid compartment after transplantation confirm that MHC class IIhigh CD86high DC are more efficient than their MHC class IIlowCD86− counterparts in promoting Treg function, as demonstrated previously.27–29 Together, these findings clearly explain why the deletion of CCR7 from donor-derived APCs leads to impaired Treg function in corneal graft recipients.
In conclusion, CCR7-mediated donor APC migration to the draining LNs after corneal transplantation promotes both allosensitization and Treg function in graft recipients. As such, CCR7 has a “paradoxical” and pleiotropic role in alloimmunity; though it clearly promotes direct priming by facilitating graft-derived APC migration to LNs, it also amplifies Treg function, underscoring the complexities of targeting APC trafficking for the promotion of transplant survival.
Footnotes
Supported by National Institutes of Health Grant R01-EY 12963 (RD).
Disclosure: Y. Jin, None; S.K. Chauhan, None; D.R. Saban, None; R. Dana, None
References
- 1.Hamrah P, Liu Y, Zhang Q, Dana MR. The corneal stroma is endowed with a significant number of resident dendritic cells. Invest Ophthalmol Vis Sci 2003; 44: 581–589 [DOI] [PubMed] [Google Scholar]
- 2.Hamrah P, Huq SO, Liu Y, Zhang Q, Dana MR. Corneal immunity is mediated by heterogeneous population of antigen-presenting cells. J Leukoc Biol 2003; 74: 172–178 [DOI] [PubMed] [Google Scholar]
- 3.Liu Y, Hamrah P, Zhang Q, Taylor AW, Dana MR. Draining lymph nodes of corneal transplant hosts exhibit evidence for donor major histocompatibility complex (MHC) class II-positive dendritic cells derived from MHC class II-negative grafts. J Exp Med 2002; 195: 259–268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huq S, Liu Y, Benichou G, Dana MR. Relevance of the direct pathway of sensitization in corneal transplantation is dictated by the graft bed microenvironment. J Immunol 2004; 173: 4464–4469 [DOI] [PubMed] [Google Scholar]
- 5.Boisgérault F, Liu Y, Anosova N, Dana R, Benichou G. Differential roles of direct and indirect allorecognition pathways in the rejection of skin and corneal transplants. Transplantation 2009; 87: 16–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dana MR, Qian Y, Hamrah P. Twenty-five-year panorama of corneal immunology: emerging concepts in the immunopathogenesis of microbial keratitis, peripheral ulcerative keratitis, and corneal transplant rejection. Cornea 2000; 19: 625–643 [DOI] [PubMed] [Google Scholar]
- 7.Förster R, Schubel A, Breitfeld D, et al. CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs. Cell 1999; 99: 23–33 [DOI] [PubMed] [Google Scholar]
- 8.Jin Y, Shen L, Chong EM, et al. The chemokine receptor CCR7 mediates corneal antigen-presenting cell trafficking. Mol Vis 2007; 13: 626–634 [PMC free article] [PubMed] [Google Scholar]
- 9.Beckmann JH, Yan S, Lührs H, et al. Prolongation of allograft survival in CCR7-deficient mice. Transplantation 2004; 12: 1809–1814 [DOI] [PubMed] [Google Scholar]
- 10.Ziegler E, Gueler F, Rong S, et al. CCL19-IgG prevents allograft rejection by impairment of immune cell trafficking. J Am Soc Nephrol 2006; 17: 2521–2532 [DOI] [PubMed] [Google Scholar]
- 11.Wang L, Han R, Lee I, et al. Permanent survival of fully MHC-mismatched islet allografts by targeting a single chemokine receptor pathway. J Immunol 2005; 175: 6311–6318 [DOI] [PubMed] [Google Scholar]
- 12.Sano Y, Ksander BR, Streilein JW. Murine orthotopic corneal transplantation in high-risk eyes: rejection is dictated primarily by weak rather than strong alloantigens. Invest Ophthalmol Vis Sci 1997; 38: 1130–1138 [PubMed] [Google Scholar]
- 13.Yamada J, Kurimoto I, Streilein JW. Role of CD4+ T cells in immunobiology of orthotopic corneal transplants in mice. Invest Ophthalmol Vis Sci 1999; 40: 2614–2621 [PubMed] [Google Scholar]
- 14.Benichou G, Valujskikh A, Heeger PS. Contributions of direct and indirect T cell alloreactivity during allograft rejection in mice. J Immunol 1999; 162: 352–358 [PubMed] [Google Scholar]
- 15.Lutz MB, Kukutsch N, Ogilvie AL, et al. An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. J Immunol Methods 1999; 223: 77–92 [DOI] [PubMed] [Google Scholar]
- 16.Chauhan SK, Saban DR, Lee HK, Dana R. Levels of Foxp3 in regulatory T cells reflect their functional status in transplantation. J Immunol 2009; 182: 148–153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yamada J, Ksander BR, Streilein JW. Cytotoxic T cells play no essential role in acute rejection of orthotopic corneal allografts in mice. Invest Ophthalmol Vis Sci 2001; 42: 386–392 [PubMed] [Google Scholar]
- 18.Haskova Z, Usiu N, Pepose JS, Ferguson TA, Stuart PM. CD4+ T cells are critical for corneal, but not skin, allograft rejection. Transplantation 2000; 69: 483–487 [DOI] [PubMed] [Google Scholar]
- 19.Jang MH, Sougawa N, Tanaka T, et al. CCR7 is critically important for migration of dendritic cells in intestinal lamina propria to mesenteric lymph nodes. J Immunol 2006; 176: 803–810 [DOI] [PubMed] [Google Scholar]
- 20.Worbs T, Bode U, Yan S, et al. Oral tolerance originates in the intestinal immune system and relies on antigen carriage by dendritic cells. J Exp Med 2006; 203: 519–527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hara M, Kingsley CI, Niimi M, et al. IL-10 is required for regulatory T cells to mediate tolerance to alloantigens in vivo. J Immunol 2001; 166: 3789–3796 [DOI] [PubMed] [Google Scholar]
- 22.van Maurik A, Herber M, Wood KJ, Jones ND. Cutting edge: CD4+CD25+ alloantigen-specific immunoregulatory cells that can prevent CD8+ T cell-mediated graft rejection: implications for anti–CD154 immunotherapy. J Immunol 2002; 169: 5401–5404 [DOI] [PubMed] [Google Scholar]
- 23.Sánchez-Fueyo A, Weber M, Domenig C, Strom TB, Zheng XX. Tracking the immunoregulatory mechanisms active during allograft tolerance. J Immunol 2002; 168: 2274–2281 [DOI] [PubMed] [Google Scholar]
- 24.Gregori S, Casorati M, Amuchastegui S, Smiroldo S, Davalli AM, Adorini L. Regulatory T cells induced by 1 alpha, 25-dihydroxyvitamin D3 and mycophenolate mofetil treatment mediate transplantation tolerance. J Immunol 2001; 167: 1945–1953 [DOI] [PubMed] [Google Scholar]
- 25.Ohl L, Mohaupt M, Czeloth N, et al. CCR7 governs skin dendritic cell migration under inflammatory and steady-state conditions. Immunity 2004; 21: 279–288 [DOI] [PubMed] [Google Scholar]
- 26.Morelli AE, Thomson AW. Tolerogenic dendritic cells and the quest for transplant tolerance. Nat Rev Immunol 2007; 7: 610–621 [DOI] [PubMed] [Google Scholar]
- 27.Yamazaki S, Iyoda T, Tarbell K, et al. Direct expansion of functional CD25+ CD4+ regulatory T cells by antigen-processing dendritic cells. J Exp Med 2003; 198: 235–247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brinster C, Shevach EM. Bone marrow-derived dendritic cells reverse the anergic state of CD4+CD25+ T cells without reversing their suppressive function. J Immunol 2005; 175: 7332–7340 [DOI] [PubMed] [Google Scholar]
- 29.Yamazaki S, Patel M, Harper A, et al. Effective expansion of alloantigen-specific Foxp3+ CD25+ CD4+ regulatory T cells by dendritic cells during the mixed leukocyte reaction. Proc Natl Acad Sci U S A 2006; 103: 2758–263 [DOI] [PMC free article] [PubMed] [Google Scholar]