Abstract
Cisplatin is an important chemotherapeutic agent available; however the nephrotoxicity is limiting its clinical use. Enhanced inflammatory response and oxidative/nitrosative stress appear to play a key role in the development of cisplatin-induced nephropathy. Activation of cannabinoid-2 (CB2) receptors with selective agonists exerts anti-inflammatory and tissue protective effects in various disease models. We have investigated the role of CB2 receptors in cisplatin-induced nephrotoxicity using selective CB2 receptor agonist HU-308 and CB2 knockout mice. Cisplatin significantly increased inflammation (leukocyte infiltration, CXCL1/2, MCP-1, TNF-α and IL1β levels), expressions of adhesion molecule ICAM-1 and superoxide generating enzymes NOX2, NOX4 and NOX1, enhanced ROS generation, iNOS expression, nitrotyrosine formation, apoptotic and poly(ADP-ribose) polymerase-dependent cell death in the kidneys of mice, associated with marked histopathological damage and impaired renal function (elevated serum BUN and creatinine levels) 3 days following the administration of the drug. CB2 agonist attenuated the cisplatin-induced inflammatory response, oxidative/nitrosative stress and cell death in the kidney and improved renal function, while CB2 knockouts developed enhanced inflammation and tissue injury. Thus, the endocannabinoid system through CB2 receptors protects against cisplatin-induced kidney damage by attenuating inflammation and oxidative/nitrosative stress, and selective CB2 agonists may represent a promising novel approach to prevent this devastating complication of chemotherapy.
Introduction
The platinum compound cisplatin is a widely used and potent chemotherapy agent available to treat a variety of solid tumors and other malignancies [1, 2], however its clinical use is limited by development of dose-dependent nephrotoxicity in about 30% of patients preventing the use of high doses to maximize the therapeutic efficacy [1, 2].
Numerous interrelated processes may be involved in the development of cisplatin-induced nephrotoxicity including inflammation [3, 4], formation of reactive oxygen [5, 6] and nitrogen species [7, 8], DNA damage [1], caspase [1] and poly (ADP-ribose) polymerase (PARP) activation [9]. Increasing number of recent studies highlight the importance of inflammatory mechanisms in the pathogenesis and progression of cisplatin-induced nephropathy through the recruitment of inflammatory cells, such as leukocytes and macrophages, which contribute to the cisplatin-induced tissue injury [3, 4, 10, 11]. Unfortunately, efficient pharmacotherapies to decrease this devastating complication of cisplatin chemotherapy are not available.
Activation of the cannabinoid-2 (CB2) receptors (expressed predominantly in immune cells and to a much less extent in various other cell types (e.g. endothelial and certain parenchymal cells) by recently recognized endogenous lipid mediators (termed endocannabinoids; produced and present in virtually all tissues/organ systems [12-14]), or selective synthetic agonists, has been shown to protect against tissue damage in various experimental models of ischemic-reperfusion injury [15-18], atherosclerosis/cardiovascular inflammation [19-21], neurodegenerative [22, 23], gastrointestinal [24-26] and other disorders by limit inflammatory cell chemotaxis/infiltration, activation and interrelated oxidative/nitrosative stress [14, 27-29]. Furthermore, CB2 receptors are over-expressed in a variety of cancers, and CB2 activation may decrease the proliferation/growth of various cancer cells and tumors [30].
In this study we aimed to explore the role of the CB2 receptors in an important and widely used chemotherapeutic drug, the cisplatin-induced inflammation, oxidative/nitrosative stress, and tissue injury in the kidney, utilizing a well-established mouse model of cisplatin-induced nephropathy. These results may have important implications for the prevention of the cisplatin-induced nephrotoxicity, as well as for the therapy of other inflammatory kidney diseases.
Material and methods
Animals and drug treatment
All animal experiments conformed to National Institutes of Health (NIH) guidelines and were approved by the Institutional Animal Care and Use Committee of the National Institute on Alcohol Abuse and Alcoholism (NIAAA; Bethesda, MD, USA). Six to 8-week-old male C57Bl/6J mice were obtained from The Jackson Laboratory (Bar Harbor, ME, USA). CB2 knockout mice (CB2−/−) and their wild-type littermates (CB2+/+) were developed as described previously and had been backcrossed to a C57Bl/6J background [15]. All animals were kept in a temperature-controlled environment with a 12-h light–dark cycle and were allowed free access to food and water at all times, and were cared for in accordance with National Institutes of Health (NIH) guidelines. Mice were sacrificed 72 hrs following a single injection of cisplatin (cis-Diammineplatinum(II) dichloride 20 mg/kg i.p.; Sigma) administration. The selective CB2 receptor agonist HU-308 [31] was obtained and dissolved as described [16], and used at 10 mg/kg, i.p. every day, starting 1.5 hours before the cisplatin exposure. In another set of experiment, HU-308 treatment started 6, 48 and 60 hours following the cisplatin injection, and continued every day until mice were sacrificed and tissues collected for the biochemical and histological measurements at 72 hours.
Renal function monitoring
On the day of the sacrifice, blood was collected immediately and serum levels of creatinine and Blood Urea Nitrogen (BUN) were measured using Prochem-V chemical analyzer (Texas, USA) or Idexx VetTest 8008 (Idexx Laboratories, Westbrook, Maine, ME).
Western blot analysis
Anti-iNOS and anti-beta-actin antibodies were obtained from Cell Signaling Technology (Danvers, MA) and Chemicon (MA). The kidney protein samples were mixed in Laemmli loading buffer, boiled for 10 min, and then subjected to SDS-PAGE. After electrophoresis, proteins were transferred onto nitrocellulose membranes and blotted against primary antibody (1:1000 dilution) for 16 hrs. Membranes were washed with PBS-T and incubated with a secondary antibody (1:1000 dilution) for 2 hrs. Protein bands were visualized by chemiluminescence reaction using SuperSignal West Pico Substrate (Thermo-Fisher).
Histological examination for tubular damage
Following fixation of the kidneys with 10% formalin, renal tissues were sectioned and stained with periodic acid-Schiff (PAS) reagents for histological examination. Tubular damage in PAS-stained sections was examined under the microscope (200× magnification) and scored based on the percentage of cortical tubules showing epithelial necrosis: 0=normal; =<10%; 2=10–25%; 3=26–75%; 4=>75%. Tubular necrosis was defined as the loss of the proximal tubular brush border, blebbing of apical membranes, tubular epithelial cell detachment from the basement membrane or intraluminal aggregation of cells and proteins as described [32, 33]. The morphometric examination was performed in a blinded manner by two independent investigators.
Myeloperoxidase (MPO) and nitrotyrosine staining
Paraffin-embedded sections were cut, deparaffinized, and hydrated by in descending gradations of ethanol, followed by microwave treatment. Next, sections were incubated in 0.3% H2O2 in PBS to block endogenous peroxidase activity. The sections were then incubated with anti-MPO (Invitrogen, Carlsbad, CA) or anti-nitrotyrosine (1:200 dilution, Cayman Chemical, Ann Arbor, MI) overnight at 4°C in a moist chamber. Biotinylated secondary antibodies and ABC Reagent were added as per kit's instructions (Vector Laboratories, Burlingame, CA). Color development was induced by incubation with a DAB kit (Vector Laboratories, Burlingame, CA) for 3–5 min, and specific staining was visualized by light microscopy as described[15, 32, 34, 35].
Lipid peroxidation/ROS production
Malondialdehyde (MDA) is one of the end products of lipid peroxidation and an indicator of ROS production. MDA was quantified in tissues as previously described, with some minor modifications [34, 36]. Briefly, tissues were homogenized (100 mg/mL) in 1.15% KCl buffer, homogenates (200 μL) were then added to a reaction mixture consisting of 1.5 mL of 0.8% thiobarbituric acid, 200 μL of 8.1% sodium dodecyl sulfate, 1.5 mL of 20% acetic acid (pH 3.5), and 600 μL of distilled H2O, and heated for 45 min at 90°C. After cooling to room temperature, the samples were centrifuged at 10,000 g for 10 min, and the absorbance of the supernatant at 532 nm was measured with 1,1,3,3-tetramethoxypropane as an external standard. The level of lipid peroxides was expressed as nmol MDA/mg protein.
In addition to MDA, 4-hydroxynonenal (4-HNE) in the renal tissues was also determined using a kit from Cell Biolabs (San Diego, CA). In brief, renal tissue extracts (10μg/mL) are adsorbed on to a 96-well plate for 12hrs at 4°C. 4-HNE adducts present in the sample or standard are probed with anti-HNE antibody, followed by an HRP conjugated secondary antibody. The HNE-protein adducts content in an unknown sample is determined by comparing with a standard curve as described in the manufacturer's protocol.
Detection of apoptosis by TUNEL, renal DNA fragmentation and caspase 3/7 activity assays
Paraffin sections were dewaxed and in situ detection of apoptosis in the renal tissues was performed by terminal deoxynucleotodyltransferase mediated nick-end labeling (TUNEL) assay according to the instruction of the manufacturer kit (Roche Diagnostics, Indianapolis). After TUNEL labeling, nucleus was labeled with Hoechst 33258 (Molecular probes, Invitrogen, CA) and the TUNNEL positive labeled kidney cells were observed using Olympus IX81 fluorescence microscope using 20× objective at 2048 × 2048 resolution with 1.6 times zoom. The morphometric examination was performed by two independent, blinded investigators. The average number of apoptotic cells in each group was calculated by taking the average of TUNEL-positive apoptotic cells in 10 fields from each kidney sample (n=5-6/group) with 320× magnification.
Caspase-3/7 activity in tissue lysate was measured using Apo-One Homogenous caspase-3/7 Assay Kit (Promega Corp., Madison, WI). An aliquot of caspase reagent was added to each well, mixed on a plate shaker for 1 h at room temperature shielded from light, and the fluorescence was measured.
The DNA fragmentation assay is based on measuring the amount of mono- and oligonucleosomes in the cytoplasmic fraction of tissue extracts using the commercially available kit (Roche, GmbH) according to manufacturer's instructions as described [16, 37].
Renal myeloperoxidase activity assay
Myeloperoxidase [MPO, (EC1.11.1.7)] was measured by InnoZyme™ Myeloperoxidase Activity Kit (EMD Gibbstown, NJ) according to manufacturer's instruction [15]. Myeloperoxidase activities were expressed as fold changes compared to the vehicle-treated control sample.
Determination renal KC (CXCL1) content
Mouse KC, also known as CXCL1(chemokine (C-X-C motif) ligand 1) or N51 was measured by Quantikine Mouse CXCL1/KC Immunoassay (R&D Systems, Minneapolis, MN) according to manufacturer's protocol. Levels were presented as fold changes compared to vehicle-treated control sample.
Renal poly (ADP-ribose) polymerase (PARP) activity and nitrotyrosine (NT) content
PARP activity was determined by assay kit according to manufacturer's instructions (Trevigen, Gaithersburg, MD) [32, 33]. NT was measured by the NT ELISA kit from Hycult Biotechnology (Cell Sciences, Canton, MA) from tissue homogenites as described [35]. Levels were presented as fold changes compared to vehicle-treated control sample.
Real-time PCR analyses
Total RNA was isolated from kidney homogenate using Trizol LS reagents (Invitrogen, Carlsbad, CA) according to manufacturer's instruction. The isolated RNA was treated with RNase-free DNase (Ambion, Austin, TX) to remove traces of genomic DNA contamination. One μg of total RNA of was reverse-transcribed to cDNA using the Super-Script II (Invitrogen, Carlsbad, CA). The target gene expression was quantified with Power Syber Green PCR Master Mix using ABI 7500 Realtime PCR Instrument. Each amplified sample in all wells was analyzed for homogeneity using dissociation curve analysis. After denaturation at 95 °C for 2 min, 40 cycles were performed at 95 °C for 10 s, 60 °C for 30 s. Relative quantification was calculated using the comparative CT method (2-ΔΔCt method : ΔΔCt = ΔCt sample - ΔCt reference). Lower ΔCT values and lower ΔΔCT reflect a relatively higher amount of gene transcript. Statistical analyses were carried out for at least 6 to 15 replicate experimental samples in each set.
Primers used:
TNF-α 5′AAGCCTGTAGCCCACGTCGTA3′ and 5′AGGTACAACCCATCGGCTGG3′
IL-1β 5′AAAAAAGCCTCGTGCTGTCG3′ and 5′GTCGTTGCTTGGTTCTCCTTG3′
MCP-1 5′ TCAGCCAGATGCAGTTAACGC3′ and 5′ TCTGGACCCATTCCTTCTTGG3′
MIP-2 5′ GGATGGCTTTCATGGAAGGAG3′ and 5′ TTGCTAAGCAAGGCACTGTGC3′
ICAM-1 5′AACTTTTCAGCTCCGGTCCTG 3′ and 5′AAATGAAGTGGACTCCACGCG 3′
iNOS 5′ATTCACAGCTCATCCGGTACG3′ and 5′GGATCTTGACCATCAGCTTGC3′
NOX1 5′TCGAACGCTACAGAAGAAGCC3′ and 5′TGGCAATCACTCCAGTAAGGC3′
gp91phox 5′ GACCATTGCAAGTGAACACCC3′ and 5′AAATGAAGTGGACTCCACGCG 3′
NOX4 5′ TCATTTGGCTGTCCCTAAACG3′ and 5′ AAGGATGAGGCTGCAGTTGAG3′
Actin 5′TGCACCACCAACTGCTTAG3′ and 5′GGATGCAGGGATGATGTTC3′.
Statistical analysis
Results are reported as mean±SEM. Statistical significance between 2 measurements was determined by the 2-tailed unpaired Student's t test (and among groups it was determined by ANOVA followed by post-hoc Student-Newman-Keuls) by using GraphPad Prism 4.3 software (San Diego, CA). Probability values of P<0.05 were considered significant.
Results
CB2 activation attenuates the cisplatin-induced renal dysfunction in mice; increased cisplation-induced damage in CB2 knockout mice
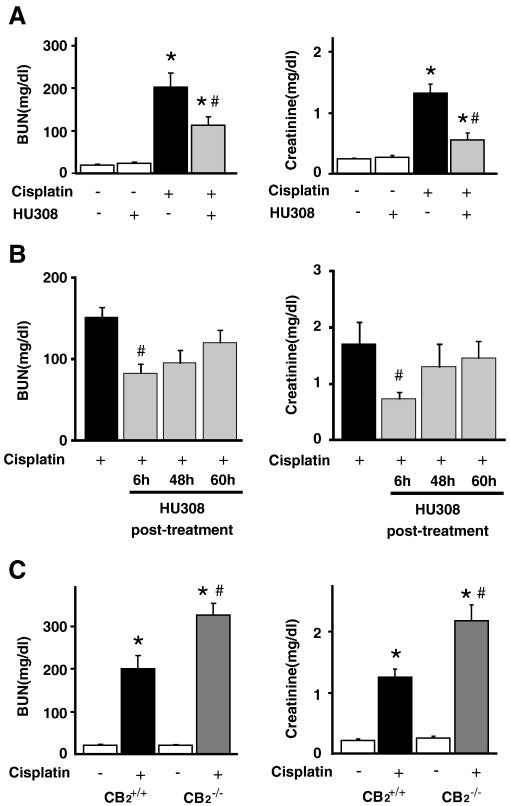
Levels of BUN and creatinine were measured at 72 h after cisplatin or vehicle administration in the serum of both HU-308-treated and untreated mice or in CB2-/- or CB2+/+ mice treated with cisplatin or vehicle. As shown in Figure 1, cisplatin administration induced severe renal dysfunction, which was attenuated by CB2 agonist HU-308 (administered starting from 1.5 hrs before the cisplatin injection and every 24 hours thereafter until the measurements were taken at 72 hours; Figure 1A; n=8-10/group, P<0.01). Starting from 6 hrs after the cisplatin administration HU-308 was still effective (Figure 1B), however it lost efficacy when it was administered after the full inflammatory response already developed at 48 or 60 hours (Figure 1B). The renal dysfunction was aggravated in CB2-/- mice compared to CB2+/+ littermates (Figure 1C; n=6-7/group, P<0.01). HU-308 alone had no effects on BUN and creatinine levels as compared to the vehicle-treated group (n=5).
Figure 1. CB2 activation attenuates the cisplatin-induced renal dysfunction; enhanced cisplatin-induced dysfunction in CB2 knockout mice.
Cisplatin induced profound renal dysfunction 72 h after the administration to mice evidenced by increased serum levels of blood urea nitrogen (BUN) and creatinine (Panel A), which were attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection and every 24 hours thereafter until the measurements were taken at 72 hours). Starting from 6 hrs after the cisplatin administration HU-308 was still effective, however it lost efficacy when it was administered after the full inflammatory response already developed at 48 or 60 hours (panel B). Results are mean±S.E.M. of n=8-10/group, *P<0.01 vs. vehicle; #P<0.05 vs. cisplatin). The cisplatin-induced renal dysfunction was enhanced in CB2-/- mice compared to CB2+/+ littermates (Panel C). Results are mean±S.E.M. of 6-7 experiments/group *P<0.01 vs. vehicle in CB2+/+ or CB2-/- mice; #P<0.05 vs. cisplatin in CB2+/+ mice.
CB2 activation attenuates the cisplatin-induced tubular necrosis and cell death; increased cisplatin-induced damage in CB2 knockout mice
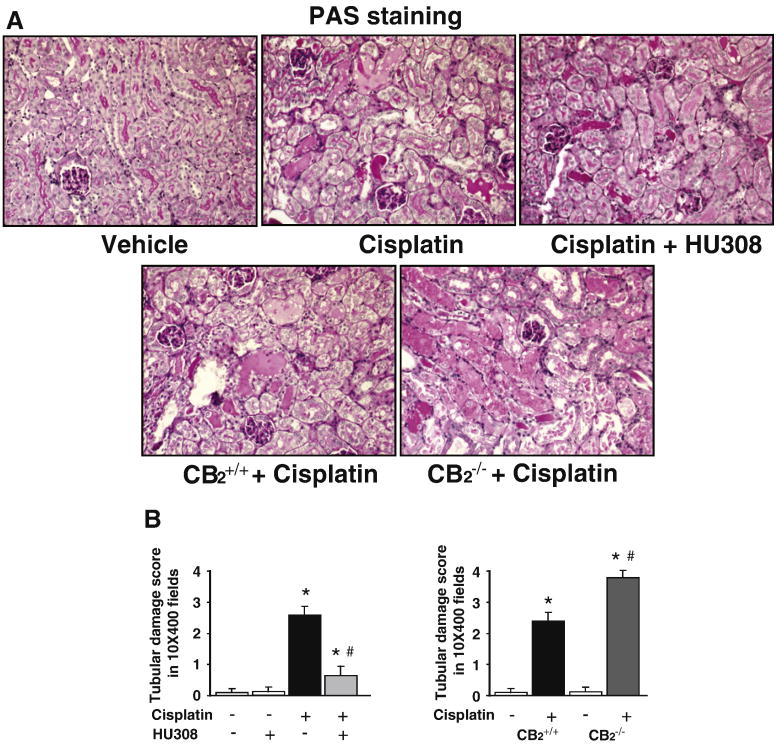
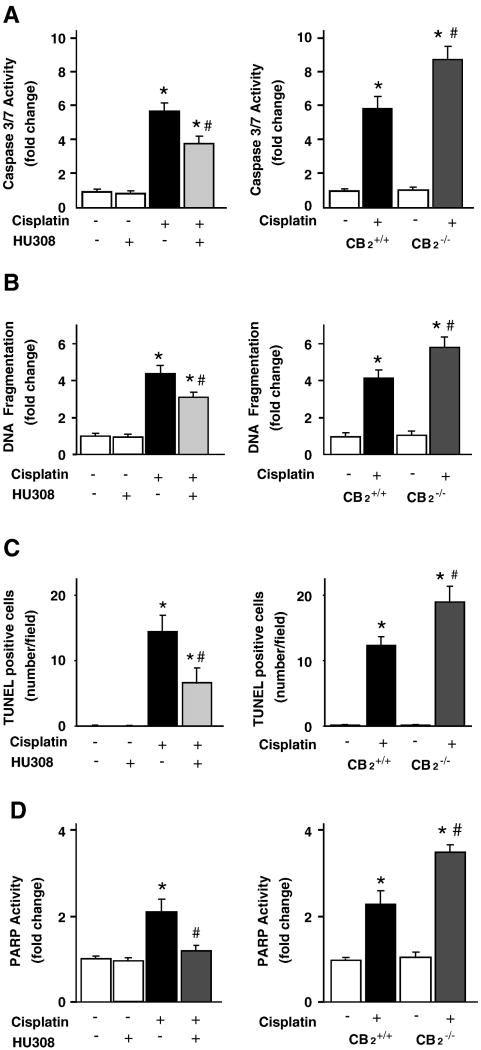
Histological examination revealed necrosis, protein cast, vacuolation and desquamation of epithelial cells in the renal tubules of the cisplatin-treated control group. Treatment with HU-308 (starting from 1.5 hrs before the cisplatin injection) significantly improved the cisplatin-induced renal tubular damage (Figure 2AB, n=6-7/group, P<0.01). The cisplatin-induced renal tubular damage was enhanced in CB2-/- mice compared to CB2+/+ littermates (n=6-7, P<0.05). Cell death was evaluated by caspase 3/7 activity (Figure 3A), DNA fragmentation (Figure 3B), PARP activity (Figure 3D) assays, and TUNEL staining (Figures 3C and 4). As shown in Figures 3 and 4 all markers of cisplatin-induced cell death were attenuated by HU-308 treatment (starting from 1.5 hrs before the cisplatin injection; (n=6-7/group, P<0.01) and were enhanced in CB2-/- mice compared to CB2+/+ littermates (n=6-7, P<0.05). HU-308 alone had no effects on the above mentioned variables (n=5) in control mice.
Figure 2. CB2 activation attenuates the cisplatin-induced renal histopathological damage; enhanced cisplatin-induced damage in CB2 knockout mice.
As shown in the representative images (panel A) cisplatin induced profound histopathological renal injury 72 h after the administration to mice, evidenced by protein cast, vacuolation and desquamation of epithelial cells in the renal tubules using PAS staining, which were attenuated by HU-308 treatment starting from 1.5 hrs before the cisplatin injection (Panels A upper row, panel B left). The cisplatin-induced renal damage was enhanced in CB2-/- mice compared to CB2+/+ littermates (Panels A bottom row, panel B right). Results are mean±S.E.M. of 6-7 experiments/group *P<0.01 vs. vehicle in CB2+/+ or CB2-/- mice; #P<0.01 vs. cisplatin in CB2+/+ mice.
Figure 3. CB2 activation attenuates the cisplatin-induced renal cell death; enhanced cisplatin-induced cell death in CB2 knockout mice.
Cell death in the kidneys was evaluated by caspase 3/7 activity, DNA fragmentation and PARP activity assays (Panels A-D). As shown in panels 3A-D left columns, all markers of cisplatin-induced cell death in the kidneys were attenuated by HU-308 treatment starting from 1.5 hrs before the cisplatin injection. Results are mean±S.E.M. of 5-7 experiments in each group *P<0.01 vs. vehicle; #P<0.05 vs. The cisplatin-induced cell death was enhanced in kidneys of CB2-/- mice compared to CB2+/+ littermates (Panels A-D right column). Results are mean±S.E.M. of 6-7 experiments/group *P<0.01 vs. vehicle in CB2+/+ or CB2-/- mice; #P<0.01 vs. cisplatin in CB2+/+ mice.
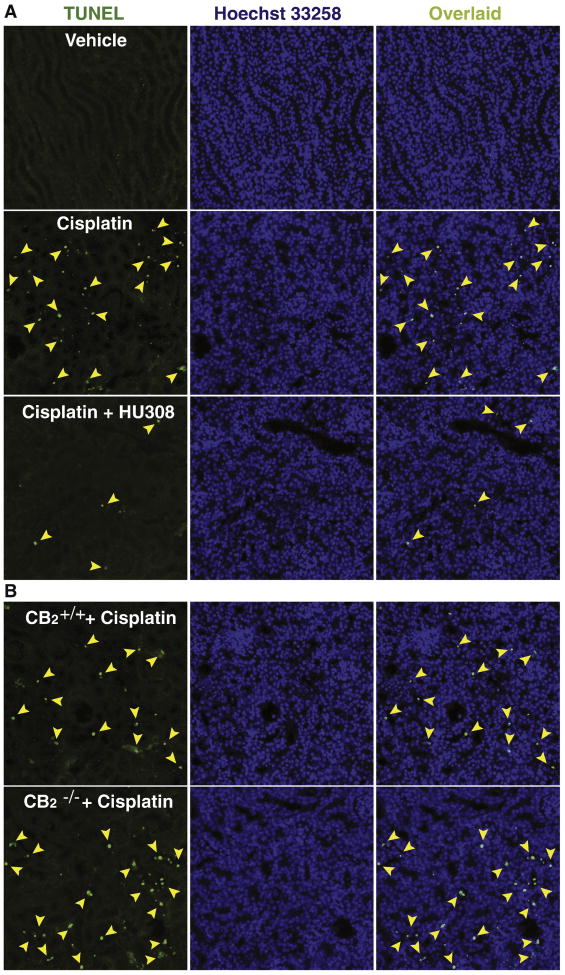
Figure 4. CB2 activation attenuates the cisplatin-induced renal TUNEL staining; enhanced cisplatin-induced TUNEL staining in kidneys of CB2 knockout mice.
Late apoptotic cell death in kidneys was evaluated by fluorescent TUNEL staining (Panels A and B, left column). As shown in representative images (320×magnification), cisplatin markedly increased the number of TUNEL positive cells (left column, shown in green color) in the kidneys. Middle column depicts nuclear staining with Hoechst 33258 (blue color), and the collocalization is shown with TUNEL at right. The number of TUNEL positive cells was attenuated by HU-308 treatment starting from 1.5 hrs before the cisplatin injection (Panel A). The cisplatin-induced cell death (number of TUNEL positive cells) was increased in kidneys of CB2-/- mice compared to CB2+/+ littermates (Panel B). See also Figure 3C for quantification.
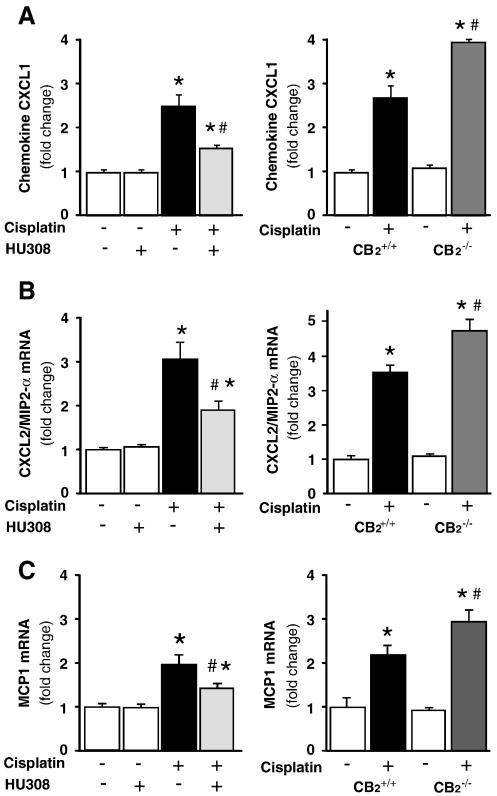
CB2 activation attenuates the cisplatin-induced enhanced renal chemokine mRNA expressions, increased cisplatin-induced chemokine expression in CB2 knockout mice
Cisplatin significantly increased chemokine (C-X-C motif) ligand 1 (CXCL1 or KC), chemokine (C-X-C motif) ligand 2 (CXCL2 or macrophage-inflammatory protein-2 (MIP-2)), monocyte chemoattractant protein-1 (MCP-1) mRNA expressions in the kidneys (Figure 5A-C; n=8-10/group, P<0.01), which were attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection; n=8-10/group, P<0.01; Figure 5A-C, left panels), and were enhanced in CB2-/- mice compared to CB2+/+ littermates (Figures 5A-C, right panels; n=6-7/group, P<0.05). HU-308 alone had no effects on the above mentioned variables (n=5) in control mice.
Figure 5. CB2 activation attenuates the cisplatin-induced enhanced renal chemokine mRNA expressions, increased cisplatin-induced chemokine expression in CB2 knockout mice.
Cisplatin significantly increased chemokine (C-X-C motif) ligand 1 (CXCL1 or KC; Panel A), chemokine (C-X-C motif) ligand 2 (CXCL2 or macrophage-inflammatory protein-2 (MIP-2); Panel B), monocyte chemoattractant protein-1 (MCP-1, Panel C) mRNA expressions in the kidneys (Panels A-C), which were attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection; Panels 5A-C, left panels). Results are mean±S.E.M. of 5-10 experiments in each group *P<0.01 vs. vehicle; #P<0.05 vs. cisplatin. The cisplatin-induced increased chemokine expressions were enhanced in kidneys of CB2-/- mice compared to CB2+/+ littermates (Panels A-C right panels). Results are mean±S.E.M. of 6-7 experiments/group *P<0.01 vs. vehicle in CB2+/+ or CB2-/- mice; #P<0.05 vs. cisplatin in CB2+/+ mice.
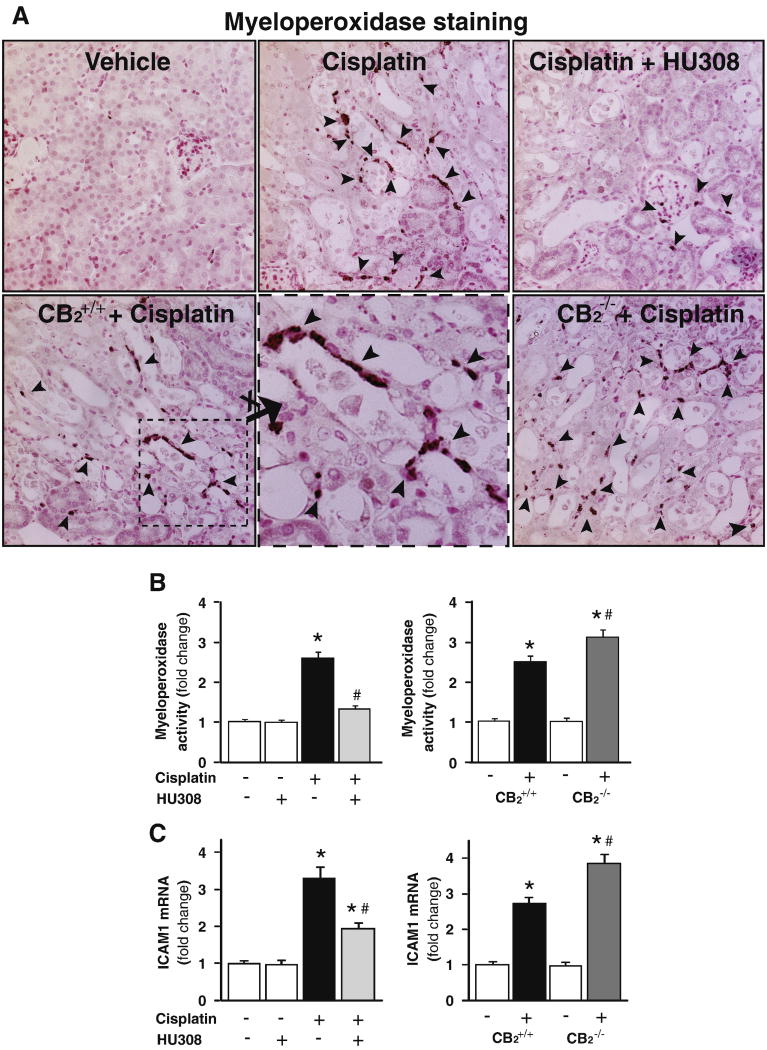
CB2 activation attenuates the cisplatin-induced enhanced renal inflammatory cell infiltration and adhesion molecule expression, increased cisplatin-induced inflammatory cell infiltration and adhesion molecule expression in CB2 knockout mice
Cisplatin significantly increased neurtophil infiltration (evidenced by increased number of myeloperoxidase positive inflammatory cells around the damaged tubular structures (Figure 6A) or increased kidney myeloperoxidase activity (Figure 6B)) and expression of intercellular adhesion molecule 1 (ICAM-1; Figure 6C; n=8-10/group, P<0.01), which were attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection; n=8-10/group, P<0.01; Figure 6B and C, left panels), and were enhanced in CB2-/- mice compared to CB2+/+ littermates (Figures 6B and C, right panels; n=6-7/group, P<0.05). HU-308 alone had no effects on the above mentioned variables (n=5) in control mice.
Figure 6. CB2 activation attenuates the cisplatin-induced increased renal leukocyte infiltration and adhesion molecule ICAM-1 expression; enhanced cisplatin-induced inflammation in CB2 knockout mice.
Cisplatin significantly increased the number of renal myeloperoxidase positive cells around the damaged tubular structures (Panel A; 400× magnification) and/or tissue myeloperoxidase activity (indicator of leukocyte infiltration, Panel B) and ICAM-1 expression (Panel C), which were attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection). Results are mean±S.E.M. of 5-10 experiments in each group *P<0.01 vs. vehicle; #P<0.05 vs. cisplatin. The cisplatin-induced increased renal leukocyte infiltration and ICAM-1 expression were enhanced in kidneys of CB2-/- mice compared to CB2+/+ littermates (Panel A lower row of images; Panels B and C, right panels). Results are mean±S.E.M. of 6-7 experiments/group *P<0.01 vs. vehicle in CB2+/+ or CB2-/- mice; #P<0.05 vs. cisplatin in CB2+/+ mice.
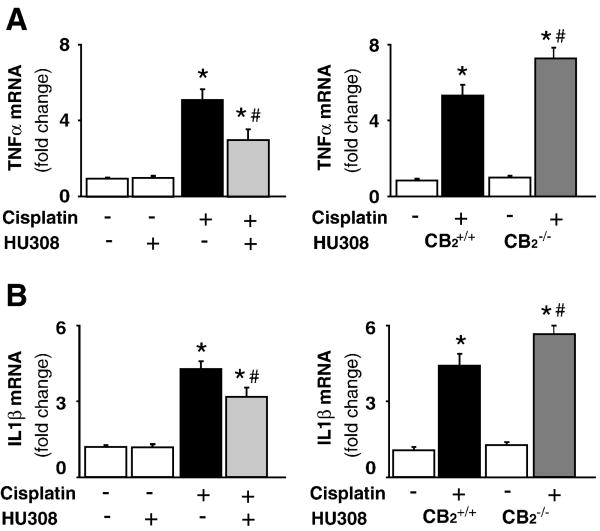
CB2 activation attenuates the cisplatin-induced enhanced renal inflammatory cytokine mRNA expression, increased cisplatin-induced inflammatory cytokine mRNA expression in CB2 knockout mice
Cisplatin significantly increased mRNA expressions of pro-inflammatory cytokines tumor necrosis factor alpha (TNFα) and interleukin 1 beta (IL-1β; Figure 7A and B; n=8-10/group, P<0.01), which were attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection; n=8-10/group, P<0.05; Figure 7A and B, left panels), and were enhanced in CB2-/- mice compared to CB2+/+ littermates (Figures 7A and B, right panels; n=6-7/group, P<0.05). HU-308 alone had no effects on the above mentioned variables (n=5).
Figure 7. CB2 activation attenuates the cisplatin-induced increased expression of mRNA of TNF-α and IL1β; enhanced cisplatin-induced inflammatory response in CB2 knockout mice.
Cisplatin significantly increased mRNA expression of TNF-α and IL1β (panels A and B) in the kidneys 72 h following the administration to mice, which were attenuated by HU-308 treatment (starting from 1.5 hrs before the cisplatin injection). Results are mean±S.E.M. of 5-10 experiments in each group *P<0.01 vs. vehicle; #P<0.05 vs. cisplatin. The cisplatin-induced increased renal TNF-α and IL1β mRNA expression was enhanced in kidneys of CB2-/- mice compared to CB2+/+ littermates (Panel A and B, right panels). Results are mean±S.E.M. of 6-7 experiments/group *P<0.01 vs. vehicle in CB2+/+ or CB2-/- mice; #P<0.05 vs. cisplatin in CB2+/+ mice.
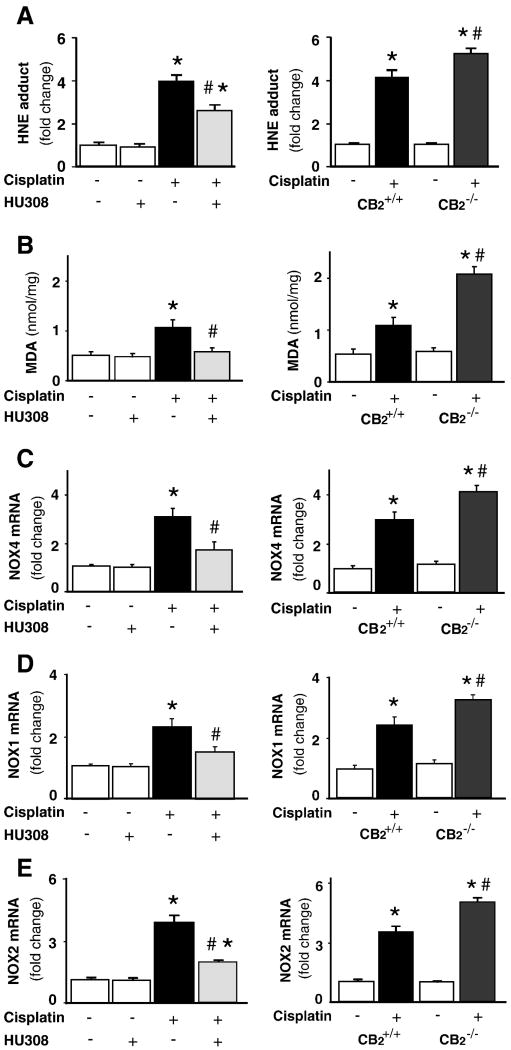
CB2 activation attenuates the cisplatin-induced increased renal lipid peroxidation/ROS generation and enhanced expression of superoxide generating enzymes RENOX (NOX4), NOX1 and NOX2; enhanced cisplatin-induced ROS generation in CB2 knockout mice
Cisplatin induced significant elevation of renal HNE adduct (Figure 8A) and malondialdehyde (MDA) levels (Figure8B; n=8-10/group, P<0.01), end products of lipid peroxidation and/or ROS generation, which were attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection; n=8-10/group, P<0.05; Figure 8A and B, left panels), and were elevated in CB2-/- mice compared to CB2+/+ littermates (Figure 8A and B, n=6-7/group, P<0.05). HU-308 alone had no effects on MDA (n=5).
Figure 8. CB2 activation attenuates the cisplatin-induced increased lipid peroxidation/ROS generation, overexpression of superoxide generating enzymes NOX4(RENOX), NOX2(gp91phox) and NOX1; increased cisplatin-induced ROS generation in CB2 knockout mice.
Cisplatin induced marked increases in lipid peroxidation/ROS generation (HNE and MDA; panela A and B); mRNA expression of NOX4, NOX2 and NOX1 (Panels C-E) in the kidneys 72 h following the administration to mice, which were attenuated by HU-308 treatment (starting from 1.5 hrs before the cisplatin injection, Panels A and B, left). Results are mean±S.E.M. of 5-10 experiments in each group *P<0.01 vs. vehicle; #P<0.05 vs. cisplatin. The cisplatin-induced increased renal NOX1, NOX2 and NOX4 mRNA expression was enhanced in kidneys of CB2-/- mice compared to CB2+/+ littermates (Panels C-E, right). Results are mean±S.E.M. of 6-7 experiments/group *P<0.01 vs. vehicle in CB2+/+ or CB2-/- mice; #P<0.05 vs. cisplatin in CB2+/+ mice.
The mRNAs for RENOX (NOX4), NOX1 and phagocyte NADPH oxidase (gp91phox/NOX2) were significantly increased in the kidneys of cisplatin-treated mice (Figure 8 C-E; n=8-10/group, P<0.01). Such increases were significantly attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection; n=8-10/group, P<0.05; Figure 8C-E, left panels), and were enhanced in CB2-/- mice compared to CB2+/+ littermates (Figure 8C-E, n=6-7/group, P<0.05). HU-308 alone had no effects on the mRNA expression of NOX4, NOX1, and NOX2 (n=5).
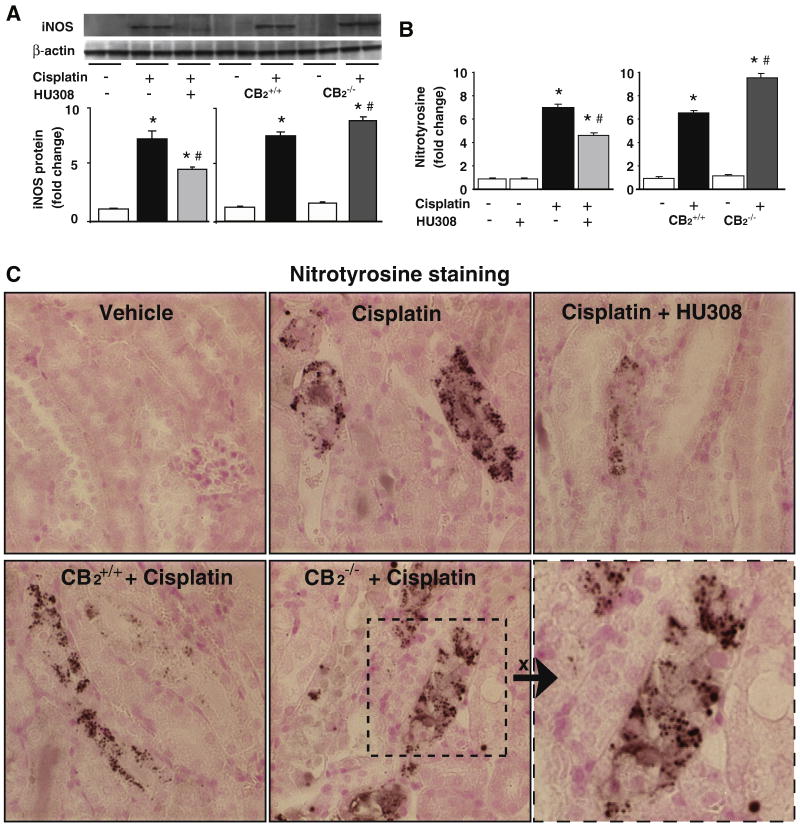
CB2 activation attenuates the cisplatin-induced increased renal iNOS protein expression and nitrotyrosine formation, enhanced cisplatin-induced nitrosative stress in CB2 knockout mice
Cisplatin significantly increased renal iNOS protein expression (Figure 9A) and nitrotyrosine (NT) formation (Figure 9B and C). Interestingly, the cisplatin-induced increased nitrotyrosine formation was mostly localized in damaged tubular cells as well as in some surrounding inflammatory cells (Figure 9C). These increases were significantly attenuated by CB2 agonist HU-308 treatment (starting from 1.5 hrs before the cisplatin injection; n=6-7/group, P<0.01; Figure 9A and B, left panels; Figure 9C upper row images), and were enhanced in CB2-/- mice compared to CB2+/+ littermates (Figure 9A and B, right panels; Figure 9C lower row images; n=6-7/group, P<0.05). HU-308 alone had no effects on the iNOS expression and NT formation (n=5).
Figure 9. CB2 activation attenuates the cisplatin-induced enhanced renal expression of iNOS and 3-nitrotyrosine (NT) formation; increased cisplatin-induced iNOS expression and NT formation in CB2 knockout mice.
Cisplatin increased iNOS protein expression (A) and NT formation (B) in the kidneys 72 h following the administration to mice, which were attenuated by HU-308 treatment (starting from 1.5 hrs before the cisplatin injection). Results are mean±S.E.M. of 5-7 experiments in each group *P<0.01 vs. vehicle; #P<0.05 vs. cisplatin. The cisplatin-induced increased renal iNOS expression and NT formation was enhanced in kidneys of CB2-/- mice compared to CB2+/+ littermates (Panels A and B, right). Results are mean±S.E.M. of 6-7 experiments/group *P<0.01 vs. vehicle in CB2+/+ or CB2-/- mice; #P<0.05 vs. cisplatin in CB2+/+ mice. Representative kidney sections demonstrate that the NT staining (dark staining) is predominantly localized in damaged tubular cells and to a lesser extent to inflammatory cells at close proximity. 400× magnification
Discussion
In the current study, we used an agonist of the cannabinoid CB2 receptor as well as CB2 receptor knockout mice to outline the role of the endocannabinoid system in an in vivo model of cisplatin-induced nephropathy. We show that treatment of mice with a CB2 receptor agonist HU-308 attenuates cisplatin-induced increased chemokine production and inflammatory cell infiltration in the kidney, and the consequent release of reactive oxidants and inflammatory mediators, leading to decreased cell death in tubular cells associated with marked improvement of the cisplatin-induced compromised renal function. These findings suggest that targeting CB2 cannabinoid receptors may represent a novel protective strategy against cisplatin-induced nephropathy. These findings are consistent with numerous recent reports demonstrating that CB2 receptor activation by synthetic agonists decreases inflammatory cell chemotaxis, adhesion to endothelial cells/activation, transendothelial migration followed by the attachment to parenchimal cell and consequent activation and release of pro-inflammatory mediators and oxidants, in experimental models of ischemic-reperfusion injury (e.g. hepatic, myocardial and cerebral)[15-18, 38], colitis [24], pancreatitis [26], cardiovascular inflammation and/or atherosclerosis [19-21], neurodegenerative-inflammatory disorders [22, 23], and other pathologies (reviewed in [14, 27-29, 39, 40]). In most of these studies CB2 agonists not only attenuated the inflammatory response, but also reduced the interrelated oxidative/nitrosative stress.
It is well known that inflammatory cells upon activation produce plethora of various reactive oxygen and nitrogen species (e.g. superoxide, inducible nitric oxide synthase-derived nitric oxide (NO) and consequently peroxynitrite via diffusion-limited reaction of superoxide with NO, just to mention a few [41, 42]), which contribute to tissue injury via numerous complex interrelated mechanisms comprising of increased lipid peroxidation [43], changes in pro-inflammatory gene expressions in both inflammatory and parenchimal cells, secretion of pro-inflammatory mediators (e.g. cytokines, chemokines), oxidation/nitration of key regulatory proteins involved in cell metabolism, signaling processes implicated in proliferation, survival and/or death, eventually leading to the activation of various mitochondrial-dependent or -independent cell death pathways culminating into organ dysfunction and failure [44, 45].
Increased inflammation [3, 4], oxidative [5, 6] and nitrosative [7, 8] stress and the activation of downstream effector cell death pathways (e.g. PARP [9]) also play a critical role in the pathogenesis of cisplatin-induced cell death and renal dysfunction.
The cellular sources of increased superoxide generation may be multiple including NADH/NADPH and xanthine oxidases, the mitochondrial respiratory chain, just to mention a few. We found significant overexpression of NOX4 ((RENOX), NAD(P)H oxidase isoform considered to be an important source of ROS production in the kindey [46]) and phagocyte NAD(P)H oxidase gp91phox/NOX2, and NOX1, accompanied by enhanced lipid peroxidation/ROS generation in kidneys of cisplatin-treated mice. We also found increased inflammatory cell infiltration around the damaged tubular structures (Figure 6A) accompanied by marked upregulation of mRNA of pro-inflammatory cytokines (TNF-α and IL1β) and chemokines (CXCL1/KC, CXCL2/MIP-2, and MCP1) in the kidneys of cisplatin-treated animals 72 hours following the drug administration, supporting an important role of the inflammatory component. Indeed inflammation, particularly cytokine TNF-α, appears to play a central role in the drug-induced nephrotoxicity [10, 11], and the cisplatin-induced kidney injury largely depends on TNF-α, since TNF-α-deficient mice and TNF-α antibody-treated wild-type mice display resistance to cisplatin-induced kidney toxicity [10, 11].
The cisplatin-induced ROS generation might also favor augmented expression of iNOS and adhesion molecules (e.g. intercellular adhesion molecule 1 (ICAM-1)) through the activation of NF-κB, which increases the generation of nitric oxide (NO). We found increased expression of adhesion molecule ICAM-1 in kidneys of cisplatin-treated mice, which may promote adhesion of inflammatory cells to the activated endothelium and/or certain parenchymal cells facilitating the increased inflammatory cell recruitment and consequent activation and interrelated tissue injury. Consistently with previous reports demonstrating that selective iNOS inhibition reduces the cisplatin-induced renal injury [8], we found significantly increased iNOS expression in the kidneys of cisplatin-treated mice. The simultaneously increases superoxide and NO production may facilitate the formation of the potent cytotoxin peroxynitrite via a diffusion-limited reaction, which attacks various biomolecules, leading to organ dysfunction via multiple mechanisms [44, 45]. In fact, nitrosative stress and/or peroxynitrite, and the activation of interrelated downstream effector pathways such as nuclear enzyme poly(ADP)ribose polymerase (PARP), have importantly been implicated in the development of cisplatin-induced cell death and subsequent nephropathy [7-9]. Underlying the importance of the nitrosative/nitrative stress-PARP pathway, we also found markedly enhanced nitrotyrosine formation (marker of peroxynitrite generation or more broadly nitrosative/nitrative stress) [45] and PARP activation in the kidneys of cisplatin-treated mice. Interestingly, nitrotyrosine was predominantly localized in damaged tubular cells and in some inflammatory and endothelial cells at close proximity of the tubular damage (Figure 9C), supporting the idea that increased peroxynitrite formation and/or protein nitration may play a important role in triggering cell death [45, 47-49].
Treatment of mice with the CB2 agonist HU-308 not only attenuated the cisplatin-induced increased inflammatory response (chemokine secretion, inflammatory cell infiltration, TNF-α and IL1β levels), but also reduced the expression of ROS generating enzymes NOX4, NOX2 and NOX1, and the consequent renal oxidative stress. Furthermore, HU-308 also decreased the cisplatin-induced increased iNOS expression and nitrotyrosine formation in the kidneys, and consequent cell death (both apoptotic and necrotic) and renal dysfunction. This is consistent with the anti-inflammatory and cytoprotective effects of CB2 activation observed in numerous above mentioned preclinical studies. On the basis of our results, the most likely mechanisms of the protective effects of the CB2 receptor activation in cisplatin-induced nephropathy model are the reduction of the endothelial cell activation (e.g. adhesion molecule expression, chemokines such as MCP-1 secretion, etc.) and attenuation of the inflammatory cell migration to the site of tubular injury and adhesion to the activated endothelium and/or damaged parenchymal cells. Interestingly, a recent study has demonstrated that CB2 receptors directly regulate ROS generation in macrophages [50] raising a possibility that the attenuation of the inflammatory cell activation may also be involved in the beneficial effect of CB2 receptor agonist in our nephropathy model. These results are also in good agreement with the protective effects of CB2 agonists observed in models of ischemic-reperfusion injury and vascular inflammation [27]. We also found increased cisplatin-induced kidney inflammation, oxidative/nitrosative stress, cell death and dysfunction in CB2-/- mice compared to their wild type CB2+/+ littermates, suggesting that the endocannabinoid system may exert protective effects via tonic activation of CB2 receptors, similarly to the effects reported in models of ischemic-reperfusion injury and neuroinflammatory disorders [14, 27].
Collectively, our results suggest that the endocannabinoid system through CB2 receptors limits the cisplatin-induced nephropathy by attenuating inflammation, oxidative/nitrosative stress and cell death, and that selective CB2 agonists may represent a promising novel approach to prevent this devastating complication of chemotherapy. This is particularly exciting, because selective CB2 receptor agonists are devoid of psychoactive side effects characteristic of the CNS cannabinoid receptor-1 activation[14]. Furthermore, CB2 receptors are over-expressed in a variety of cancers in which CB2 activation appears to decrease the proliferation/growth of cancer cells[30, 51]. These results may also have important implications for the treatment of other kidney diseases associated with inflammation and interrelated oxidative/nitrosative stress.
Acknowledgments
This study was supported by the Intramural Research Program of NIH/NIAAA (to P.P.). P.P. dedicates this study to his beloved mother Iren Bolfert, who died from the complications of chemotherapy. Authors are indebted to Prof. Raphael Mechoulam for providing continuous support/tools and valuable comments on the manuscript.
List of abbreviations
- CB2 receptor
cannabinoid-2 receptor
- ROS
reactive oxygen species
- HU-308
a potent, selective CB2 agonist
- CB2-/- mice
CB2 knockout mice
- PARP
poly (ADP-ribose) polymerase
- iNOS
inducible nitric oxide synthase
- NO
nitric oxide
- MCP-1
monocyte chemoattractant protein 1
- ICAM-1/CD54
intercellular adhesion molecule 1
- CXCL1/KC/N51
chemokine (C-X-C motif) ligand 1
- CXCL2/MIP-2
chemokine (C-X-C motif) ligand 2/macrophage inflammatory protein
- MPO
myeloperoxidase
Footnotes
Disclosures: No conflict of interest to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Ries F, Klastersky J. Nephrotoxicity induced by cancer chemotherapy with special emphasis on cisplatin toxicity. Am J Kidney Dis. 1986;8:368–379. doi: 10.1016/s0272-6386(86)80112-3. [DOI] [PubMed] [Google Scholar]
- 2.Schrier RW. Cancer therapy and renal injury. J Clin Invest. 2002;110:743–745. doi: 10.1172/JCI16568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yamate J, Sato K, Ide M, Nakanishi M, Kuwamura M, Sakuma S, Nakatsuji S. Participation of different macrophage populations and myofibroblastic cells in chronically developed renal interstitial fibrosis after cisplatin-induced renal injury in rats. Vet Pathol. 2002;39:322–333. doi: 10.1354/vp.39-3-322. [DOI] [PubMed] [Google Scholar]
- 4.Faubel S, Lewis EC, Reznikov L, Ljubanovic D, Hoke TS, Somerset H, Oh DJ, Lu L, Klein CL, Dinarello CA, Edelstein CL. Cisplatin-induced acute renal failure is associated with an increase in the cytokines interleukin (IL)-1beta, IL-18, IL-6, and neutrophil infiltration in the kidney. J Pharmacol Exp Ther. 2007;322:8–15. doi: 10.1124/jpet.107.119792. [DOI] [PubMed] [Google Scholar]
- 5.Matsushima H, Yonemura K, Ohishi K, Hishida A. The role of oxygen free radicals in cisplatin-induced acute renal failure in rats. J Lab Clin Med. 1998;131:518–526. doi: 10.1016/s0022-2143(98)90060-9. [DOI] [PubMed] [Google Scholar]
- 6.Davis CA, Nick HS, Agarwal A. Manganese superoxide dismutase attenuates Cisplatin-induced renal injury: importance of superoxide. J Am Soc Nephrol. 2001;12:2683–2690. doi: 10.1681/ASN.V12122683. [DOI] [PubMed] [Google Scholar]
- 7.Chirino YI, Hernandez-Pando R, Pedraza-Chaverri J. Peroxynitrite decomposition catalyst ameliorates renal damage and protein nitration in cisplatin-induced nephrotoxicity in rats. BMC Pharmacol. 2004;4:20. doi: 10.1186/1471-2210-4-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chirino YI, Trujillo J, Sanchez-Gonzalez DJ, Martinez-Martinez CM, Cruz C, Bobadilla NA, Pedraza-Chaverri J. Selective iNOS inhibition reduces renal damage induced by cisplatin. Toxicol Lett. 2008;176:48–57. doi: 10.1016/j.toxlet.2007.10.006. [DOI] [PubMed] [Google Scholar]
- 9.Racz I, Tory K, Gallyas F, Jr, Berente Z, Osz E, Jaszlits L, Bernath S, Sumegi B, Rabloczky G, Literati-Nagy P. BGP-15 - a novel poly(ADP-ribose) polymerase inhibitor - protects against nephrotoxicity of cisplatin without compromising its antitumor activity. Biochem Pharmacol. 2002;63:1099–1111. doi: 10.1016/s0006-2952(01)00935-2. [DOI] [PubMed] [Google Scholar]
- 10.Ramesh G, Reeves WB. TNF-alpha mediates chemokine and cytokine expression and renal injury in cisplatin nephrotoxicity. J Clin Invest. 2002;110:835–842. doi: 10.1172/JCI15606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang B, Ramesh G, Norbury CC, Reeves WB. Cisplatin-induced nephrotoxicity is mediated by tumor necrosis factor-alpha produced by renal parenchymal cells. Kidney Int. 2007;72:37–44. doi: 10.1038/sj.ki.5002242. [DOI] [PubMed] [Google Scholar]
- 12.Mechoulam R, Fride E, Di Marzo V. Endocannabinoids. Eur J Pharmacol. 1998;359:1–18. doi: 10.1016/s0014-2999(98)00649-9. [DOI] [PubMed] [Google Scholar]
- 13.Howlett AC, Barth F, Bonner TI, Cabral G, Casellas P, Devane WA, Felder CC, Herkenham M, Mackie K, Martin BR, Mechoulam R, Pertwee RG. International Union of Pharmacology. XXVII. Classification of cannabinoid receptors. Pharmacol Rev. 2002;54:161–202. doi: 10.1124/pr.54.2.161. [DOI] [PubMed] [Google Scholar]
- 14.Pacher P, Batkai S, Kunos G. The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev. 2006;58:389–462. doi: 10.1124/pr.58.3.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Batkai S, Osei-Hyiaman D, Pan H, El-Assal O, Rajesh M, Mukhopadhyay P, Hong F, Harvey-White J, Jafri A, Hasko G, Huffman JW, Gao B, Kunos G, Pacher P. Cannabinoid-2 receptor mediates protection against hepatic ischemia/reperfusion injury. FASEB J. 2007;21:1788–1800. doi: 10.1096/fj.06-7451com. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rajesh M, Pan H, Mukhopadhyay P, Batkai S, Osei-Hyiaman D, Hasko G, Liaudet L, Gao B, Pacher P. Cannabinoid-2 receptor agonist HU-308 protects against hepatic ischemia/reperfusion injury by attenuating oxidative stress, inflammatory response, and apoptosis. J Leukoc Biol. 2007;82:1382–1389. doi: 10.1189/jlb.0307180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Montecucco F, Lenglet S, Braunersreuther V, Burger F, Pelli G, Bertolotto M, Mach F, Steffens S. CB(2) cannabinoid receptor activation is cardioprotective in a mouse model of ischemia/reperfusion. J Mol Cell Cardiol. 2009;46:612–620. doi: 10.1016/j.yjmcc.2008.12.014. [DOI] [PubMed] [Google Scholar]
- 18.Zhang M, Adler MW, Abood ME, Ganea D, Jallo J, Tuma RF. CB2 receptor activation attenuates microcirculatory dysfunction during cerebral ischemic/reperfusion injury. Microvasc Res. 2009;78:86–94. doi: 10.1016/j.mvr.2009.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Steffens S, Veillard NR, Arnaud C, Pelli G, Burger F, Staub C, Karsak M, Zimmer A, Frossard JL, Mach F. Low dose oral cannabinoid therapy reduces progression of atherosclerosis in mice. Nature. 2005;434:782–786. doi: 10.1038/nature03389. [DOI] [PubMed] [Google Scholar]
- 20.Rajesh M, Mukhopadhyay P, Batkai S, Hasko G, Liaudet L, Huffman JW, Csiszar A, Ungvari Z, Mackie K, Chatterjee S, Pacher P. CB2-receptor stimulation attenuates TNF-alpha-induced human endothelial cell activation, transendothelial migration of monocytes, and monocyte-endothelial adhesion. Am J Physiol Heart Circ Physiol. 2007;293:H2210–2218. doi: 10.1152/ajpheart.00688.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Batkai S, Rajesh M, Mukhopadhyay P, Hasko G, Liaudet L, Cravatt BF, Csiszar A, Ungvari Z, Pacher P. Decreased age-related cardiac dysfunction, myocardial nitrative stress, inflammatory gene expression, and apoptosis in mice lacking fatty acid amide hydrolase. Am J Physiol Heart Circ Physiol. 2007;293:H909–918. doi: 10.1152/ajpheart.00373.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Maresz K, Pryce G, Ponomarev ED, Marsicano G, Croxford JL, Shriver LP, Ledent C, Cheng X, Carrier EJ, Mann MK, Giovannoni G, Pertwee RG, Yamamura T, Buckley NE, Hillard CJ, Lutz B, Baker D, Dittel BN. Direct suppression of CNS autoimmune inflammation via the cannabinoid receptor CB1 on neurons and CB2 on autoreactive T cells. Nat Med. 2007;13:492–497. doi: 10.1038/nm1561. [DOI] [PubMed] [Google Scholar]
- 23.Sagredo O, Gonzalez S, Aroyo I, Pazos MR, Benito C, Lastres-Becker I, Romero JP, Tolon RM, Mechoulam R, Brouillet E, Romero J, Fernandez-Ruiz J. Cannabinoid CB2 receptor agonists protect the striatum against malonate toxicity: relevance for Huntington's disease. Glia. 2009;57:1154–1167. doi: 10.1002/glia.20838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Storr MA, Keenan CM, Zhang H, Patel KD, Makriyannis A, Sharkey KA. Activation of the cannabinoid 2 receptor (CB(2)) protects against experimental colitis. Inflamm Bowel Dis. 2009 doi: 10.1002/ibd.20960. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pacher P, Gao B. Endocannabinoids and liver disease. III. Endocannabinoid effects on immune cells: implications for inflammatory liver diseases. Am J Physiol Gastrointest Liver Physiol. 2008;294:G850–854. doi: 10.1152/ajpgi.00523.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Michalski CW, Laukert T, Sauliunaite D, Pacher P, Bergmann F, Agarwal N, Su Y, Giese T, Giese NA, Batkai S, Friess H, Kuner R. Cannabinoids ameliorate pain and reduce disease pathology in cerulein-induced acute pancreatitis. Gastroenterology. 2007;132:1968–1978. doi: 10.1053/j.gastro.2007.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pacher P, Hasko G. Endocannabinoids and cannabinoid receptors in ischaemia-reperfusion injury and preconditioning. Br J Pharmacol. 2008;153:252–262. doi: 10.1038/sj.bjp.0707582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Di Marzo V. Targeting the endocannabinoid system: to enhance or reduce? Nat Rev Drug Discov. 2008;7:438–455. doi: 10.1038/nrd2553. [DOI] [PubMed] [Google Scholar]
- 29.Pertwee RG. Emerging strategies for exploiting cannabinoid receptor agonists as medicines. Br J Pharmacol. 2009;156:397–411. doi: 10.1111/j.1476-5381.2008.00048.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pisanti S, Bifulco M. Endocannabinoid system modulation in cancer biology and therapy. Pharmacol Res. 2009;60:107–116. doi: 10.1016/j.phrs.2009.03.011. in press. [DOI] [PubMed] [Google Scholar]
- 31.Hanus L, Breuer A, Tchilibon S, Shiloah S, Goldenberg D, Horowitz M, Pertwee RG, Ross RA, Mechoulam R, Fride E. HU-308: a specific agonist for CB(2), a peripheral cannabinoid receptor. Proceedings of the National Academy of Sciences of the United States of America. 1999;96:14228–14233. doi: 10.1073/pnas.96.25.14228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pan H, Mukhopadhyay P, Rajesh M, Patel V, Mukhopadhyay B, Gao B, Hasko G, Pacher P. Cannabidiol attenuates cisplatin-induced nephrotoxicity by decreasing oxidative/nitrosative stress, inflammation, and cell death. J Pharmacol Exp Ther. 2009;328:708–714. doi: 10.1124/jpet.108.147181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pan H, Shen Z, Mukhopadhyay P, Wang H, Pacher P, Qin X, Gao B. Anaphylatoxin C5a contributes to the pathogenesis of cisplatin-induced nephrotoxicity. Am J Physiol Renal Physiol. 2009;296:F496–504. doi: 10.1152/ajprenal.90443.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Moon KH, Hood BL, Mukhopadhyay P, Rajesh M, Abdelmegeed MA, Kwon YI, Conrads TP, Veenstra TD, Song BJ, Pacher P. Oxidative inactivation of key mitochondrial proteins leads to dysfunction and injury in hepatic ischemia reperfusion. Gastroenterology. 2008;135:1344–1357. doi: 10.1053/j.gastro.2008.06.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mukhopadhyay P, Rajesh M, Batkai S, Kashiwaya Y, Hasko G, Liaudet L, Szabo C, Pacher P. Role of superoxide, nitric oxide, and peroxynitrite in doxorubicin-induced cell death in vivo and in vitro. Am J Physiol Heart Circ Physiol. 2009;296:H1466–1483. doi: 10.1152/ajpheart.00795.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pacher P, Liaudet L, Bai P, Mabley JG, Kaminski PM, Virag L, Deb A, Szabo E, Ungvari Z, Wolin MS, Groves JT, Szabo C. Potent metalloporphyrin peroxynitrite decomposition catalyst protects against the development of doxorubicin-induced cardiac dysfunction. Circulation. 2003;107:896–904. doi: 10.1161/01.cir.0000048192.52098.dd. [DOI] [PubMed] [Google Scholar]
- 37.Mukhopadhyay P, Batkai S, Rajesh M, Czifra N, Harvey-White J, Hasko G, Zsengeller Z, Gerard NP, Liaudet L, Kunos G, Pacher P. Pharmacological inhibition of CB1 cannabinoid receptor protects against doxorubicin-induced cardiotoxicity. J Am Coll Cardiol. 2007;50:528–536. doi: 10.1016/j.jacc.2007.03.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Murikinati S, Juttler E, Keinert T, Ridder DA, Muhammad S, Waibler Z, Ledent C, Zimmer A, Kalinke U, Schwaninger M. Activation of cannabinoid 2 receptors protects against cerebral ischemia by inhibiting neutrophil recruitment. FASEB J. 2009 doi: 10.1096/fj.09-141275. [DOI] [PubMed] [Google Scholar]
- 39.Benito C, Tolon RM, Pazos MR, Nunez E, Castillo AI, Romero J. Cannabinoid CB2 receptors in human brain inflammation. Br J Pharmacol. 2008;153:277–285. doi: 10.1038/sj.bjp.0707505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Izzo AA, Camilleri M. Cannabinoids in intestinal inflammation and cancer. Pharmacol Res. 2009;60:117–125. doi: 10.1016/j.phrs.2009.03.008. in press. [DOI] [PubMed] [Google Scholar]
- 41.Ischiropoulos H, Zhu L, Beckman JS. Peroxynitrite formation from macrophage-derived nitric oxide. Arch Biochem Biophys. 1992;298:446–451. doi: 10.1016/0003-9861(92)90433-w. [DOI] [PubMed] [Google Scholar]
- 42.McCord JM, Fridovich I. The biology and pathology of oxygen radicals. Ann Intern Med. 1978;89:122–127. doi: 10.7326/0003-4819-89-1-122. [DOI] [PubMed] [Google Scholar]
- 43.Radi R, Beckman JS, Bush KM, Freeman BA. Peroxynitrite-induced membrane lipid peroxidation: the cytotoxic potential of superoxide and nitric oxide. Arch Biochem Biophys. 1991;288:481–487. doi: 10.1016/0003-9861(91)90224-7. [DOI] [PubMed] [Google Scholar]
- 44.Szabo C, Ischiropoulos H, Radi R. Peroxynitrite: biochemistry, pathophysiology and development of therapeutics. Nat Rev Drug Discov. 2007;6:662–680. doi: 10.1038/nrd2222. [DOI] [PubMed] [Google Scholar]
- 45.Pacher P, Beckman JS, Liaudet L. Nitric oxide and peroxynitrite in health and disease. Physiol Rev. 2007;87:315–424. doi: 10.1152/physrev.00029.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Geiszt M, Kopp JB, Varnai P, Leto TL. Identification of renox, an NAD(P)H oxidase in kidney. Proceedings of the National Academy of Sciences of the United States of America. 2000;97:8010–8014. doi: 10.1073/pnas.130135897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Levrand S, Vannay-Bouchiche C, Pesse B, Pacher P, Feihl F, Waeber B, Liaudet L. Peroxynitrite is a major trigger of cardiomyocyte apoptosis in vitro and in vivo. Free Radic Biol Med. 2006;41:886–895. doi: 10.1016/j.freeradbiomed.2006.04.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Beckman JS. Understanding peroxynitrite biochemistry and its potential for treating human diseases. Arch Biochem Biophys. 2009;484:114–116. doi: 10.1016/j.abb.2009.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Liaudet L, Vassalli G, Pacher P. Role of peroxynitrite in the redox regulation of cell signal transduction pathways. Front Biosci. 2009;14:4809–4814. doi: 10.2741/3569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Han KH, Lim S, Ryu J, Lee CW, Kim Y, Kang JH, Kang SS, Ahn YK, Park CS, Kim JJ. CB1 and CB2 cannabinoid receptors differentially regulate the production of reactive oxygen species by macrophages. Cardiovasc Res. 2009 doi: 10.1093/cvr/cvp240. in press. [DOI] [PubMed] [Google Scholar]
- 51.Guzman M. Cannabinoids: potential anticancer agents. Nat Rev Cancer. 2003;3:745–755. doi: 10.1038/nrc1188. [DOI] [PubMed] [Google Scholar]