Abstract
This study aimed to improve, using the zebrafish model, our understanding of the distinct roles of pituitary gonadotropins FSH and LH in regulating testis functions in teleost fish. We report, for the first time in a vertebrate species, that zebrafish Leydig cells as well as Sertoli cells express the mRNAs for both gonadotropin receptors (fshr and lhcgr). Although Leydig cell fshr expression has been reported in other piscine species and may be a common feature of teleost fish, Sertoli cell lhcgr expression has not been reported previously and might be related to the undifferentiated gonochoristic mode of gonadal sex differentiation in zebrafish. Both recombinant zebrafish (rzf) gonadotropins (i.e. rzfLH and rzfFSH) stimulated androgen release in vitro and in vivo, with rzfFSH being significantly more potent than rzfLH. Forskolin-induced adenylate cyclase activation mimicked, whereas the protein kinase A inhibitor H-89 significantly reduced, the gonadotropin-stimulated androgen release. Therefore, we conclude that both FSH receptor and LH/choriogonadotropin receptor signaling are predominantly mediated through the cAMP/protein kinase A pathway to promote steroid production. Despite this similarity, other downstream mechanisms seem to differ. For example, rzfFSH up-regulated the testicular mRNA levels of a number of steroidogenesis-related genes both in vitro and in vivo, whereas rzfLH or human chorionic gonadotropin did not. Although not fully understood at present, these differences could explain the capacity of FSH to support both steroidogenesis and spermatogenesis on a long-term basis, whereas LH-stimulated steroidogenesis might be a more acute process, possibly restricted to periods during which peak steroid levels are required.
Both zebrafish gonadotropin receptors show overlapping cellular expression in the testis and share downstream signaling steps, but the biological activities of each gonadotropin still differ substantially, in particular with regard to modifying testicular gene expression levels.
The pituitary gonadotropins LH and FSH play critical roles in regulating male reproduction across vertebrates (1,2,3). In mammals, the specific activities of both gonadotropins are clearly defined, given the highly specific interactions between each hormone and its respective receptor, LH/choriogonadotropin receptor (Lhcgr) and FSH receptor (Fshr). In addition, Lhcgr and Fshr expression in mammalian testis is restricted to Leydig and Sertoli cells, respectively (1,2,4). Hence, LH predominantly controls Leydig cell steroidogenesis, whereas FSH regulates Sertoli cell functions (2,4,5,6).
In teleost fish, conversely, the biological activities of FSH and LH seem to be broader because both are strong steroidogenic hormones (7,8,9,10,11,12,13,14,15,16). Our present concept on how gonadotropins develop their bioactivity in fish testis is mainly based on a groundbreaking study in coho salmon, Oncorhynchus kisutch, showing LH binding to Leydig cells (indicating the presence of Lhcgr) and FSH binding to Sertoli cells (indicating the presence of Fshr), although FSH binding to Leydig cells could not be unequivocally demonstrated or clearly excluded (17). Recently, Fshr protein (Japanese eel, Anguilla japonica) (18) and mRNA (African catfish, Clarias gariepinus) (7) have been demonstrated in teleost Leydig cells, findings compatible with a direct trophic effect of FSH on Leydig cell functioning, including the regulation of steroid release (7). Thus, a revision of the concept of gonadotropin mode of action in fish testis is required. For instance, in the presence of Fshr in both Leydig and Sertoli cells, FSH alone may regulate the activities of both cell types during early-mid spermatogenesis, when plasma LH levels are very low or undetectable in seasonally reproducing species (19,20,21).
Another distinctive feature of teleost gonadotropin bioactivity is the limited hormone-binding selectivity that the gonadotropin receptors exhibit (7,14,16,17,22,23,24,25,26). This may lead to receptor cross-activation, most probably LH interaction with Fshr (see references above), especially during the spawning season when LH reaches peak plasma levels (19,20,21,27). The physiological relevance of this phenomenon (if any) is unknown at present.
To understand how gonadotropins modulate gonadal functions, knowledge about the identity and the relevant characteristics of the gonadotropin target cells is imperative. This information is still missing in zebrafish, Danio rerio, an important vertebrate model species. Thus, the first objective of the present report was to identify the cell types expressing either the fshr or the lhcgr mRNA in zebrafish testis. We then set out to produce recombinant zebrafish (rzf) gonadotropins for studies on their biological activities regarding testicular androgen release and expression of selected testicular genes.
Materials and Methods
Animals
Sexually mature zebrafish from the Tübingen AB strain, either wild type or transgenic [expressing enhanced green fluorescent protein (EGFP) under the control of the germ cell-specific vas promoter; vas::EGFP (28)], and outbred fish were used. Animal housing (29) and experimentation were consistent with Dutch national regulations and were approved by the Utrecht University Animal Use and Care Committee.
Cellular localization of gonadotropin receptor gene expression in zebrafish testis
The localization of lhcgr and fshr mRNA expression in zebrafish testis was investigated by in situ hybridization, laser microdissection of testis sections, and fluorescence-activated sorting of testicular cell suspensions.
In situ hybridization for lhcgr, fshr, and insulin-like peptide 3 (insl3), a Leydig cell-specific transcript (30), was performed on 10-μm-thick cryosections from wild-type Tübingen AB zebrafish testis using digoxigenin-labeled cRNA probes (7). Gene-specific primers used to generate DNA templates for probe synthesis are shown in Supplemental Table 1 (published on The Endocrine Society’s Journals Online web site at http://endo.endojournals.org).
Laser microdissection of zebrafish testis sections was carried out using a PALM MicroBeam Instrument (PALM Microlaser Technologies, Bernried, Germany). Two microdissected fractions were analyzed from two independent biological samples for lhcgr and fshr mRNA abundance: interstitial tissue, identified by 3β-hydroxysteroid dehydrogenase (3β-Hsd) staining of Leydig cells, and intratubular tissue, containing spermatogenic cysts (germ/Sertoli cells units). See Supplemental Materials and Methods and Supplemental Fig. 1 for further details.
Fluorescence-activated cell sorting (FACS) was used to isolate a germ-cell-enriched population from vas::EGFP zebrafish testis. Both EGFP intensity and cell size decrease as spermatogenesis progresses (31), whereas somatic cells are EGFP negative and have variable sizes. This allowed obtaining cell populations enriched in spermatogonia and primary spermatocytes by selecting for cells showing strong EGFP intensity and large size (Supplemental Fig. 1, D and E). Dissociated testicular cells were prepared from two independent batches of 10–12 fish each (32), resuspended in 1 ml D-PBS+ (Invitrogen, Carlsbad, CA), and then immediately subjected to FACS using an inFlux cell sorter (Becton Dickinson Biosciences, Franklin Lakes, NJ). The obtained cell suspension was centrifuged at 50 × g for 10 min followed by total RNA extraction using the RNAqueous-Micro kit (Ambion, Austin, TX). Synthesis of cDNA from total RNA samples was performed as described (26).
Primers to detect zebrafish fshr mRNA, lhcgr mRNA, piwi-like 1 (piwil1) mRNA (predominantly expressed in spermatogonia) (33), synaptonemal complex protein 3 like (sycp3l) mRNA (expressed by primary spermatocytes) (34,35), outer dense fiber 3 like (odf3l) mRNA (expressed by spermatids) (35), gonadal somatic cell-derived factor (gsdf) mRNA (expressed by Sertoli cells) (36), insl3 mRNA (expressed by Leydig cells) (30), and the reference endogenous control gene β-actin1 (Supplemental Table 2) were designed and validated for specificity and amplification efficiency on serial dilutions of testis cDNA (26). All real-time quantitative PCRs (qPCRs) and calculations were performed as described previously (7,26,37).
Gonadotropins
The rzfFSH and rzfLH proteins used for these experiments were produced as detailed in the Supplemental Materials and Methods and Supplemental Fig. 2. Human chorionic gonadotropin (hCG) was obtained from Organon (Oss, The Netherlands).
In vitro androgen release response to increasing gonadotropins and forskolin concentrations
Testicular tissue was challenged in concentration-response bioassays with either rzfFSH (from 12.5–1000 ng protein/ml), rzfLH (from 100-2000 ng protein/ml), or the adenylate cyclase activator forskolin (from 0.1–25 μm; Sigma-Aldrich, St. Louis, MO). Testis tissue was collected from 12 outbred zebrafish per condition tested, and the two testes from each fish were incubated in parallel, one of them (randomly chosen left or right) serving as control for the contralateral one. Incubations lasted 18 h in a humidified air atmosphere at 25 C in 96-well flat-bottom plates (Corning Inc., Corning, NY) using a final volume of 200 μl culture medium (38). After incubation, tissue explants were weighed and discarded, while the medium was processed for the quantification of 11-ketotestosterone (11-KT) and 11β-hydroxyandrostenedione (OHA) levels by RIA (39).
Because of the experimental design used (one testis assigned to basal condition and the contralateral one to experimental condition), we obtained data for basal steroid release for all concentrations of the compounds assayed. Homogeneity of basal steroid release among the different replicates was tested by one-way ANOVA. Because no statistically significant differences (P > 0.05) were identified, basal steroid release data were compiled into one single basal steroid release condition for each compound tested. Thereafter, significant differences among the different concentrations of each substance were identified by one-way ANOVA followed by the Student-Newman-Keuls test (P < 0.05).
Role of the cAMP/protein kinase A (PKA) pathway on the gonadotropin-mediated stimulation of androgen release in vitro
Testis tissue explants were incubated with rzfFSH (250 ng/ml) or rzfLH (1000 ng/ml) in the absence or presence of 100 μm of the PKA inhibitor H-89 (Sigma-Aldrich) (7). Fish origin, batch and age, number of replicates per condition tested, tissue preparation, culture conditions, and analyses performed were the same as described above. For each fish, one testis was incubated with recombinant gonadotropin, whereas the contralateral one was incubated with gonadotropin plus H-89. Significant differences were identified by the Student’s paired t test (P < 0.05).
In vitro short-term actions of gonadotropins on testis functions
The capacities of rzfFSH (100 ng/ml) and rzfLH (500 ng/ml) to modulate the mRNA levels of a number of testicular genes were investigated over a 2-h incubation period. Origin of the fish (n = 8 per condition), tissue preparation, culture conditions, and analyses performed were the same as described above, except that testis explants were saved for gene expression studies.
Total RNA was extracted from testis explants using the RNAqueous-Micro kit (Ambion). Further processing to determine the threshold cycle (Cq) values of the reference endogenous control gene β-actin1 as well as of fshr; lhcgr; the Leydig cell genes insl3, steroidogenic acute regulatory protein (star), and cytochrome P450, family 17, subfamily A, polypeptide 1 (cyp17a1); and the Sertoli cell genes androgen receptor (ar; expression not detectable in zebrafish Leydig cells by in situ hybridization) (37), anti-Müllerian hormone (amh), and gsdf (primer sequences are listed in Supplemental Table 2) by qPCR analysis was performed as reported (26,37). No significant differences (P > 0.05) were found among the mean β-actin1 Cq values in the different treatment groups (Supplemental Fig. 3A), thus validating β-actin1 as a suitable reference for the current experiments.
Because the two testes from each fish were incubated in parallel, the amounts of androgens released into the incubation media and the relative mRNA expression levels were compared between treated and respective control groups by the Student’s paired t test (P < 0.05). Thereafter, values were expressed as percentage of respective basal levels, and differences between each gonadotropin treatment were identified by the Student’s unpaired t test (P < 0.05).
In vitro medium-term actions of gonadotropins on testis functions
The capacities of rzfFSH (100 ng/ml) and rzfLH (500 ng/ml) to modulate the mRNA levels of a number of testicular genes were investigated in a medium-term organ culture system (38). In addition, the relative contribution of steroid production to gonadotropin-induced changes in gene expression was assessed by including 25 μg/ml of the 3β-Hsd inhibitor trilostane (Chemos, Regenstauf, Germany) in the media. After 2 d incubation in a humidified air atmosphere at 25 C, explants (n = 8 per condition) were processed as above for gene expression analysis. No significant differences (P > 0.05) were found among the mean β-actin1 Cq values in the different treatment groups (Supplemental Fig. 3B).
Incubation media were recovered after culture and stored at −25 C until 11-KT and/or OHA quantification (39). Pilot studies determined that the 11-KT antibody cross-reacted with trilostane at the concentration used, and therefore, trilostane-containing incubation media were assayed for OHA only. Recovery studies using tritiated androgens showed that 47 ± 1% of total steroids added to culture wells was present in the incubation medium after an overnight equilibration period (n = 8), whereas the remaining steroid was trapped in the agar cylinder. The results obtained were corrected accordingly.
Data were compared between treated and respective control explants by the Student’s paired t test (P < 0.05). Thereafter, values were expressed as percentage of respective basal levels, and differences between treatments (i.e. gonadotropin vs. gonadotropin plus trilostane) were identified by the Student’s unpaired t test (P < 0.05).
In vivo short-term actions of gonadotropins on testis functions
In this experiment, outbred zebrafish received an ip injection of 100 ng/g body weight rzfFSH or rzfLH or 10 IU/g body weight hCG in a total volume of approximately 5 μl (n = 8 fish per condition). Control fish received a 5-μl PBS injection. Two hours after the injections, fish were euthanized in ice water, the caudal peduncle cut, and a sample of blood collected using heparinized syringes. Samples were then transferred to heparinized tubes and the 11-KT plasma levels quantified (39). Testes were used for gene expression analysis as reported above. No significant differences (P > 0.05) were found among the mean β-actin1 Cq values in the different treatment groups (Supplemental Fig. 3C).
Significant differences among the different treatments were identified by one-way ANOVA followed by the Student- Newman-Keuls test (P < 0.05).
Results
Zebrafish Leydig and Sertoli cells express both fshr and lhcgr mRNA
A clear in situ hybridization signal on interstitial Leydig cells was obtained with fshr antisense probe (Fig. 1A). The arrangement of cells stained was characteristic of Leydig cells as observed on histological sections (Fig. 1B) and resembled those obtained with insl3 antisense probe (Fig. 1D) and with the 3β-Hsd enzyme-histochemical reaction (Fig. 1E). Sertoli and germ cells were negative for fshr mRNA by in situ hybridization. No signal was obtained for lhcgr mRNA. The fshr mRNA-positive Leydig cells present were stained with an approximately similar intensity, and no apparent spatial distribution pattern of Leydig cells (e.g. rostrocaudal, ventrodorsal, or central vs. peripheral in the testis) was observed. Hybridization with sense cRNA probes for fshr (Fig. 1C), lhcgr, or insl3 (not shown) did not yield any staining.
Figure 1.
Cellular localization of fshr and lhcgr mRNA expression in zebrafish testis. A and C, In situ hybridization with either an antisense (A) or a sense (C) riboprobe for zebrafish fshr; note the positive staining in Leydig cells (Lc) in the interstitial tissue in A and the absence of signal in C. In both cases, germ cells were devoid of staining. B, Toluidine blue-stained 2-μm-thick plastic section showing groups of Leydig cells (Lc) within the interstitial compartment (enclosed by broken lines). D and E, Leydig cells are also identified by their positive staining with an insl3 antisense cRNA probe (D) and with the 3β-Hsd enzymatic reaction (E). Scale bars, 20 μm. F, Relative fshr and lhcgr mRNA expression levels in two microdissected testis tissue fractions (interstitial and intratubular) and a germ-cell-enriched population obtained from vas::EGFP transgenic zebrafish by FACS (germ cells only). Data correspond to values from two experiments, each with duplicate measurements (mean ± sem), normalized to β-actin1 mRNA levels, and expressed as percentage of fshr transcript amounts in the interstitial fraction. Note the logarithmic scale.
Confirmation of fshr mRNA expression by interstitial Leydig cells was obtained by qPCR analysis of laser microdissected testis fractions (Fig. 1F). This analysis also revealed expression of fshr mRNA in the intratubular compartment [∼3-fold lower than in the interstitial (Leydig cell) fraction]. Moreover, lhcgr mRNA expression was found in the interstitial and, remarkably, also in the intratubular samples (∼24- and ∼18-fold lower than fshr mRNA levels measured in the interstitial compartment, respectively). The purity of the intratubular fraction was confirmed by measuring mRNA levels of the Leydig cell-specific gene insl3. Its expression level was approximately 265-fold lower in the intratubular than in the interstitial fraction (Supplemental Fig. 1C). Contamination of the interstitial testis tissue fraction with intratubular Sertoli cells was assessed by measuring the mRNA levels of the Sertoli cell-specific gene gsdf. Its expression level in the interstitial fraction was approximately 11-fold lower than in the intratubular fraction (Supplemental Fig. 1C). These data indicated a small degree of Sertoli cell contamination in the interstitial fraction but negligible contamination of the intratubular fraction with interstitial elements. Still, lhcgr mRNA levels were even 1.3-fold higher in the intratubular than in the interstitial fraction.
Identification of the intratubular cell type expressing the gonadotropin receptor mRNAs was accomplished by qPCR analysis of a germ-cell-enriched population obtained by FACS of testis cell suspensions prepared from vas::EGFP transgenic zebrafish (Fig. 1F). This cell population exhibited abundant piwi1l and sycp3l mRNA expression in addition to intermediate odf3l mRNA levels (i.e. the cell population contained mainly spermatogonia and spermatocytes but also spermatids), whereas the levels of the somatic transcripts gsdf and insl3 were at least 60-fold lower than piwi1l (Supplemental Fig. 1F), indicating the high purity of the samples obtained. Because both fshr and lhcgr mRNA levels were very low in the germ cells compared with the whole intratubular compartment (Fig. 1F), we concluded that the intratubular expression of both fshr and lhcgr genes resided in the only non-germ-cell type in the intratubular compartment, the Sertoli cell.
Altogether, our results indicate that in zebrafish testis, both Leydig and Sertoli cells express both fshr and lhcgr mRNA, whereas germ cells are devoid of such transcripts.
Recombinant, single-chain zebrafish gonadotropins and forskolin stimulate androgen production in testicular explants
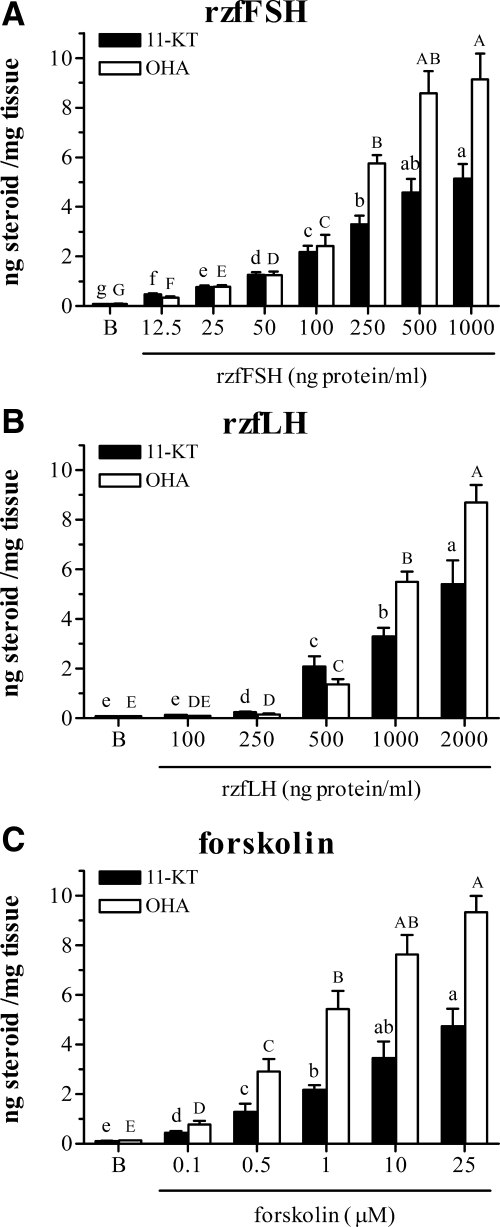
When zebrafish testes were incubated with rzfFSH, androgen secretion increased gradually with the gonadotropin concentrations (Fig. 2A). The lowest rzfFSH concentration tested (12.5 ng/ml) elicited significant elevations of both 11-KT and OHA release (∼4.7- and ∼3.5-fold above basal levels, respectively). Maximal androgen release (∼47-fold for 11-KT and ∼84-fold for OHA) was reached at 500 ng/ml rzfFSH. No further increase was observed by doubling the concentration.
Figure 2.
Stimulation of androgen release by zebrafish testicular explants. Amounts of 11-KT and OHA (mean ± sem) measured in incubation media after overnight (18 h) exposure to increasing concentrations of zfFSH (panel A), rzfLH (panel B), or the adenylate cyclase activator forskolin (panel C). B, Basal release. Values represent compiled data from two experiments, each with six replicates per ligand concentration. Different letters denote significant differences among groups (P < 0.05).
For rzfLH (Fig. 2B), 250 ng/ml induced the first significant increase in both 11-KT and OHA secretion (∼2.5-and ∼1.8-fold above basal levels, respectively). From 250-2000 ng/ml, androgen secretion kept increasing significantly. Higher rzfLH concentrations could not be tested. However, the androgen release induced by 2000 ng/ml rzfLH was similar to those observed in response to maximally effective rzfFSH or forskolin concentrations, suggesting that maximal androgen release had been reached.
Incubation with 0.1 μm forskolin, the lowest concentration tested, already induced significant increases in both 11-KT and OHA production by zebrafish testis (∼4.0- and ∼5.9-fold above basal levels, respectively) (Fig. 2C). The maximal steroidogenic response was obtained with forskolin concentrations between 10 and 25 μm.
The cAMP/PKA pathway is involved in both the FSH- and LH-stimulated androgen production
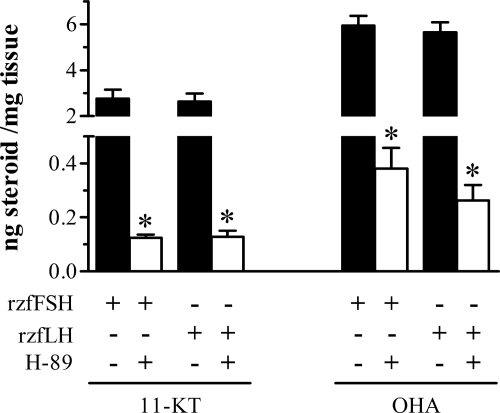
When testis tissue was incubated with 250 ng/ml rzfFSH or 1000 ng/ml rzfLH in the presence of 100 μm of the PKA inhibitor H-89, androgen production was strongly reduced (22- to 16-fold) compared with the levels measured in the absence of the inhibitor (Fig. 3). Therefore, for both rzf gonadotropins, the cAMP-PKA pathway is the major mediator of the steroidogenic response. This is further supported by comparing the residual 11-KT release observed in the presence of gonadotropin and H-89 with basal 11-KT release from 49–82 individual testis incubations of fish of the same origin and age (basal release from the dose-response experiments described above; see Fig. 2): all 11-KT release levels were within the basal release range, whereas the values of the precursor OHA showed a somewhat higher dispersion with part of the data being above the basal range (Supplemental Fig. 4).
Figure 3.
Effects of the PKA inhibitor H-89 on the gonadotropin-stimulated androgen release by zebrafish testicular explants. Amounts of 11-KT and OHA (mean ± sem) measured in incubation media after overnight (18 h) exposure to 250 ng/ml rzfFSH or 1000 ng/ml rzfLH alone and in combination with 100 μm H-89. Values represent compiled data from two experiments, each with six to seven replicates per condition. *, Values are significantly different (P < 0.05) from the respective gonadotropin-only condition.
The rzf gonadotropins show differential effects on steroidogenesis and testicular gene expression in vitro and in vivo
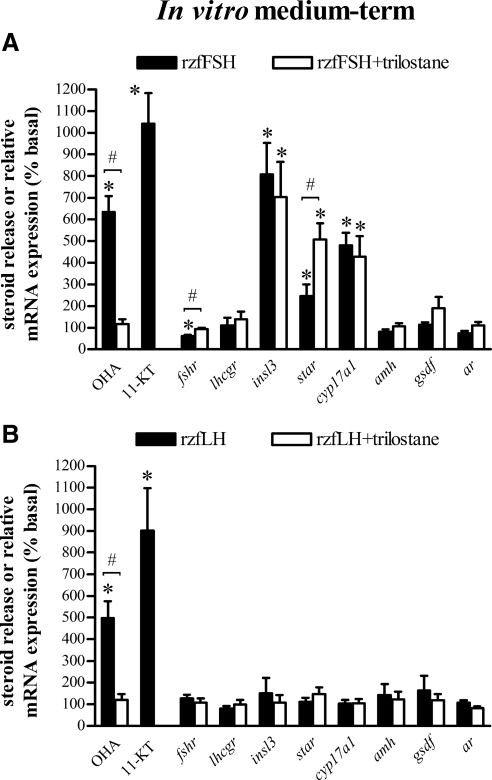
Zebrafish testis explants incubated for 2 d with either 100 ng/ml rzfFSH or 500 ng/ml rzfLH displayed a significant up-regulation (5- to 10-fold) of androgen release (Fig. 4). However, we observed significant changes in gene expression levels only in response to rzfFSH. The steady-state mRNA levels of a number of Leydig cell genes (i.e. insl3, star, and cyp17a1) increased (2.5- to 8-fold), whereas fshr mRNA levels were reduced to 60% of control values (Fig. 4A). No significant changes were observed for the Sertoli cell genes amh, gsdf, or ar mRNA levels (Fig. 4A). In the presence of the 3β-Hsd inhibitor trilostane, both rzfFSH- and rzfLH-stimulated androgen release was completely abolished (Fig. 4). Although trilostane did not modify the rzfFSH-stimulated insl3 and cyp17a1 mRNA overexpression, it prevented rzfFSH-induced fshr mRNA down-regulation and further increased star mRNA levels (Fig. 4A). The presence of trilostane in combination with rzfLH did not modify the mRNA levels of any of the transcripts analyzed (Fig. 4B).
Figure 4.
In vitro medium-term (2 d) modulation of androgen release and testicular gene expression by rzf gonadotropins. Amounts of the androgens OHA and 11-KT measured in incubation media and relative mRNA expression levels of several testicular genes (fshr, lhcgr, insl3, star, cyp17a1, amh, gsdf, and ar) after 2 d exposure to 100 ng/ml rzfFSH (A) or 500 ng/ml rzfLH (B) alone (black bars) and in combination with 25 μg/ml of the 3β-Hsd inhibitor trilostane (white bars). Data (mean ± sem) come from an experiment with eight replicates per condition and are expressed as percentage of basal levels, which were set to 100% for each parameter analyzed. Basal androgen release was 237 ± 18 pg OHA/mg tissue and 147 ± 17 pg 11-KT/mg tissue. Gene expression levels were normalized to β-actin1 mRNA levels. *, Values are significantly different (P < 0.05) from the respective basal condition in the absence of recombinant gonadotropin; #, significant difference (P < 0.05) between the absence and the presence of trilostane.
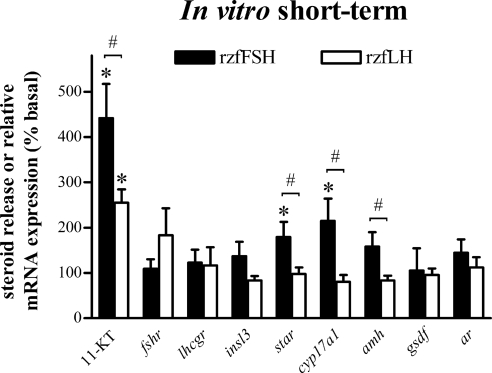
The short-term culture approach (Fig. 5) provided a picture similar to that obtained in the medium-term study; both rzfFSH and rzfLH stimulated testicular 11-KT release (P < 0.05), whereas significant changes in steroidogenesis-related transcripts (star and cyp17a1) were detected only after rzfFSH treatment. Incubation with rzfLH elicited a 1.8-fold increase in fshr mRNA levels, but statistical significance was not reached. Moreover, no significant changes were observed for any of the Sertoli cell genes assayed as compared with basal samples, although amh mRNA expression differed statistically between rzfFSH- and rzfLH-treated explants.
Figure 5.
In vitro short-term (2 h) modulation of androgen release and testicular gene expression by rzf gonadotropins. Amounts of androgen (11-KT) measured in incubation media and relative mRNA expression levels of several testicular genes (fshr, lhcgr, insl3, star, cyp17a1, amh, gsdf, and ar) after 2 h exposure to 100 ng/ml rzfFSH (black bars) or 500 ng/ml rzfLH (white bars). Data (mean ± sem) come from an experiment with eight replicates per condition and are expressed as percentage of basal levels, which were set to 100% for each parameter analyzed. Basal androgen release was 40.0 ± 4.9 pg 11-KT/mg tissue. Gene expression levels were normalized to β-actin1 mRNA levels. *, Values are significantly different (P < 0.05) from the respective basal condition in the absence of recombinant gonadotropin; #, significant difference (P < 0.05) between treatment with rzfFSH and rzfLH.
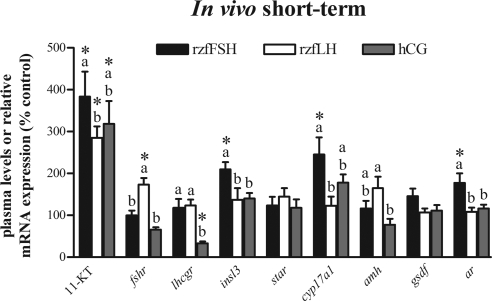
Both recombinant gonadotropins exhibited bioactivity also in vivo, as reflected in significantly elevated plasma 11-KT levels and the change in the mRNA levels of several genes expressed in zebrafish testis (Fig. 6). At the dose (100 ng/g body weight) and time after administration (2 h) tested, rzfFSH showed significantly higher steroidogenic potency than rzfLH, although the difference between the two hormones was less pronounced than that observed in vitro. Considering changes in gene expression, the rzfFSH treatment up-regulated insl3, cyp17a1, and ar mRNA levels (1.8- to 2.5-fold). In vivo administration of rzfLH, conversely, had only minor effects on testicular gene expression. An interesting exception was the significant 1.7-fold up-regulation of fshr mRNA levels. Injection of hCG also elicited a significant increase in plasma 11-KT levels that was intermediate between those observed after the treatments with rzfFSH and rzfLH (Fig. 6). Similar to rzfLH, hCG injection did not change the mRNA levels of the steroidogenesis-related genes (e.g. star and cyp17a1) or the Leydig cell-specific gene insl3, the only exception being the previously unreported down-regulation of lhcgr transcription.
Figure 6.
In vivo short-term (2 h) modulation of plasma androgen levels and testicular gene expression by rzf gonadotropins and hCG. Circulating 11-KT concentrations and relative mRNA expression levels of several testicular genes (fshr, lhcgr, insl3, star, cyp17a1, amh, gsdf, and ar) 2 h after injection with 100 ng/g body weight rzfFSH (black bars), rzfLH (white bars) or 10 IU/g body weight hCG (gray bars). Data (mean ± sem) come from an experiment with eight fish per condition and are expressed as percentage of control levels, which were set to 100% for each parameter analyzed. Control androgen levels were 3.6 ± 0.9 ng 11-KT/ml plasma. Gene expression levels were normalized to β-actin1 mRNA levels. *, Values are significantly different (P < 0.05) from the basal control condition. Different letters denote significant differences among groups (P < 0.05).
Discussion
The pituitary gonadotropins LH and FSH, acting via their receptors, Lhcgr and Fshr, are the main factors controlling testis functions across vertebrates, including teleost fish (1,2,3,40). Knowledge about the identity and characteristics of testicular cell types responding to gonadotropic signals, i.e. expressing the gonadotropin receptors, is crucial for understanding how gonadotropins regulate testis functions. This information is, however, lacking in most fish species. The available data are restricted to coho salmon, Japanese eel, and African catfish (7,17,18), although a complete data set has been reported only for the latter; African catfish Leydig cells express both lhcgr and fshr mRNA, whereas Sertoli cells solely express fshr mRNA (7). In view of the importance of zebrafish as an experimental model and the essential roles that pituitary gonadotropins play in the regulation of testis physiology, we considered it imperative to identify gonadotropin receptor-expressing cells in the zebrafish testis.
In situ hybridization and qPCR analysis of laser microdissected testicular interstitial tissue showed that, also in zebrafish, the steroid-producing Leydig cells express both fshr and lhcgr mRNAs. These results support the view that fshr/Fshr expression by Leydig cells is a common feature among teleost fish, which is further supported by the strong steroidogenic activity displayed by several piscine FSH proteins (7,8,9,10,11,12,13,14,15,16). Therefore, we propose to explain the steroidogenic activity of FSH-like piscine gonadotropins by a trophic, direct effect on Leydig cells steroid release (7).
qPCR-based analyses of samples obtained by laser microdissection and FACS demonstrated fshr expression in zebrafish Sertoli cells, as typically reported in other vertebrates (1,2,4) including fish (7,17,18). Surprisingly, we also found, for the first time in any vertebrate, lhcgr mRNA expression in zebrafish Sertoli cells. This striking testicular expression pattern of lhcgr in zebrafish may be related to its undifferentiated gonochoristic mode of gonadal sex differentiation, in which the indifferent gonad initially develops as an ovary-like structure in all individuals, but in future males, developing oocytes soon degenerate and the gonads transform into testes (41,42,43). Accordingly, in undifferentiated gonochoristic fish, the default differentiation pathway of the germ-cell-supporting somatic cell precursor (common for both granulosa and Sertoli cells) (44,45) may be toward granulosa-like cells, which may retain the potential to transdifferentiate to Sertoli cells (46,47,48,49) and/or may regress, whereas another population of undifferentiated somatic cells differentiates into Sertoli cells (50). Because mammalian as well as salmon granulosa cells (51,52,53) express both Fshr and Lhcgr protein and the respective mRNAs, it seems possible that the coexpression of both fshr and lhcgr mRNA in zebrafish Sertoli cells is related to the transitory female developmental stage observed in this species.
In the present report, we show that rzfFSH was at least 20-fold more potent in stimulating androgen production than rzfLH, whereas both hormones reached similar maximal stimulation levels. This difference in the steroidogenic potency may be related to the higher mRNA expression levels shown by fshr in comparison with lhcgr, which may lead to a lower abundance of Lhcgr protein on the Leydig cell membrane. This may also represent an adaptive mechanism to balance the constitutive activity of the zebrafish Lhcgr (54) in favor of a tight regulation of Leydig cell steroid release. Functional characterization of zebrafish gonadotropin receptors has shown that the Lhcgr was exclusively activated by LH, whereas both FSH and LH were able to activate the Fshr, which showed a slightly higher preference for FSH (24). Therefore, we conclude that the stimulation of steroid release elicited by rzfFSH was exclusively mediated via Fshr-dependent mechanisms.
Incubation of zebrafish testicular explants with increasing concentrations of the adenylate cyclase activator forskolin resulted in a concentration-dependent stimulation of androgen release, as previously shown in other fish (8,55,56,57). In the current study, forskolin and both recombinant gonadotropins reached similar maximal induction levels, whereas the PKA inhibitor H-89 strongly inhibited gonadotropin-stimulated androgen release. Together, these data suggest a prominent role for the cAMP/PKA pathway in both Fshr- and Lhcgr-mediated regulation of testicular steroid production (7,8,55,56,57), although other signaling pathways may have (a quantitatively minor) role in the process, as proposed for other fish (8,55,58,59) and mammalian species (5,60,61).
Although both FSH and LH make use of the cAMP/PKA pathway in zebrafish Leydig cells, other downstream mechanisms that the gonadotropins use seem to differ, as demonstrated by analyzing expression levels of steroidogenesis-relevant transcripts; only rzfFSH up-regulated the testicular mRNA levels of star and cyp17a1. Our results are similar to those recently obtained for Japanese eel where the two gonadotropins showed similar potencies in stimulating testicular androgen release, but only recombinant FSH induced major changes in steroidogenic gene expression (16). Because up-regulation of gene expression is essential for the long-term maintenance of steroid production (60,62), piscine FSH seems to be the main hormone sustaining Leydig cell steroidogenesis.
The finding that zebrafish (this study) and Japanese eel (16) recombinant LH proteins have limited capacities to support expression of steroidogenesis-related genes may indicate that their bioactivity regarding steroid production relies predominantly on nongenomic mechanisms. Although the present results do not allow identifying such mechanisms, possible explanations could include direct stimulation of the catalytic activity of steroidogenic enzymes, recruitment of mRNAs encoding for steroidogenic enzymes and/or steroidogenic acute regulatory protein from previously produced and stabilized pools, and/or activation (e.g. by phosphorylation) of already synthesized but inactive protein pools (60,62). However, all these assumptions would postulate the use by FSH of yet unidentified downstream pathways, which may or may not involve the participation of cAMP and/or PKA, to induce the observed up-regulation of steroidogenesis genes expression. As mentioned previously, the latter is essential for the sustained production of steroids, suggesting that the effects exerted by piscine LH on steroidogenesis have a limited duration. This may constitute an additional mechanism in zebrafish Leydig cells to counterbalance the constitutive activity of the Lhcgr (54). Hence, we propose that the LH bioactivity on Leydig cells may be restricted to a transient response of the testicular steroidogenic system, such as described in spawning goldfish, a close relative of the zebrafish, with processes of short duration requiring a quick response in males once they perceive the stimuli from ovulatory females (27,63). Such events are characterized by the presence of high plasma sex steroid levels, and the two gonadotropins may cooperate to produce the high steroid output required for semen hydration, for stimulating courtship/spawning behavior, and/or for the release of pheromones into the water (7,27,63,64).
Although rzfLH did not change the mRNA levels of steroidogenesis-related genes analyzed, it did increase fshr mRNA levels in our short-term experiments (2 h). Interestingly, rzfFSH induced a partial down-regulation of fshr mRNA levels after 2 d culture, which, together with the short-term LH-induced up-regulation, may represent a regulatory loop to achieve a balanced testicular gonadotropin receptor expression.
The ability of hCG to stimulate steroidogenesis in fish is well known (65) and is explained by its capacity to activate the Lhcgr but not the Fshr in different species, including the zebrafish (23,54). In our in vivo study, hCG elicited downstream effects on zebrafish testis similar to rzfLH (i.e. stimulation of androgen release without up-regulating gene expression), although the effects at thereceptor transcription level were different: lhcgr mRNA down-regulation occurred after hCG but not after rzfLH treatment. This could be associated with the long half-life of hCG (65) or the comparatively high concentration used (10 IU to ∼715 ng); also, the high specificity of hCG for the Lhcgr of fish is remarkable because piscine (purified or recombinant) LH proteins, although preferring the Lhcgr, also activate the Fshr (7,22,24,52).
The experimental setup involving the 3β-Hsd inhibitor trilostane revealed inhibitory effects of androgens on rzfFSH-induced changes in both fshr and star mRNA levels. Both genes are situated far upstream in the steroidogenic process (60), and thus, androgen-mediated inhibition may be part of a negative feedback loop to prevent overstimulation of steroidogenesis. This is supported by ongoing studies in our laboratory showing that testicular star mRNA levels are down-regulated by exposure to 11-KT (unpublished) as well as by the information available for mammalian models (66,67).
Further confirmation of the differential roles of FSH and LH on zebrafish Leydig cell functions was obtained in the current study by measuring mRNA levels of the Leydig cell-specific gene insl3. Its expression remained unchanged after exposure to rzfLH or hCG but was strongly up-regulated by rzfFSH in a steroid-independent manner. The latter observation agrees with own in vitro assays in zebrafish testis showing no effect of increasing concentrations of 11-KT on insl3 mRNA transcription (unpublished), although it contrasts with previous studies reporting stimulatory effects of androgens on testicular insl3 expression in amphibians (68). In mammals, although one study has reported that Insl3 expression was regulated by androgens both in primary cultures of rat Leydig cells and in the mouse MA-10 tumor Leydig cell line (69), another study using the same cell line concluded that Insl3 was expressed in a constitutive manner (70). To our knowledge, the present report is the first demonstrating in any teleost species a stimulatory effect of FSH on insl3 expression, a Leydig cell-specific factor with antiapoptotic effects on meiotic germ cells in mammals (71) but with yet unknown functions in fish.
Although there is substantial evidence supporting the critical nursing functions of Sertoli cells during germ cell proliferation and differentiation in fish spermatogenesis (40), very little is known about the mechanisms by which Sertoli cells relay the gonadotropic signals toward the developing germ cells. Thus, in an attempt to characterize gonadotropin effects on zebrafish Sertoli cells, we measured the mRNA levels of a number of Sertoli cell-specific genes in several bioassays. However, among the genes examined, we found little response to both gonadotropins, the exceptions being the transient increases in fshr or ar mRNA levels after 2 h in vivo rzfLH and rzfFSH treatment, respectively. In general, our candidate gene approach regarding gonadotropin-sensitive Sertoli cell transcripts was of limited success; future studies should therefore include unbiased approaches.
Whether the effects elicited by recombinant hormones (in this and other studies, e.g. Refs. 11,13,16, and 72,73,74,75,76) reflect the biological activities of the pituitary hormones could be answered only by comparing highly purified gonadotropins with homologous recombinant proteins, a setting not feasible in zebrafish due to its small size. Nevertheless, different recombinant gonadotropins, including single-chain and/or N-terminal His-tagged molecules (like the hormones used in this study), have been shown to elicit biological effects comparable to heterodimeric purified native hormones, which has been explained by the receptors’ capacity to specifically recognize their ligands even if those are presented in different conformations (77,78,79,80,81,82). Moreover, based on structural analysis of human FSH in complex with its receptor (80), the His tag used for purification is pointing away from the major receptor interaction sites, and therefore, it is unlikely to interfere with receptor activation. Whether this applies to fish gonadotropins and their receptors as well remains to be explored with species large enough to allow purification of pituitary hormones for comparative studies. However, our His-tagged, single-chain gonadotropins were able to fully exploit the steroidogenic potency of zebrafish testis tissue (as measured by forskolin incubations), demonstrating their suitability as bona fide ligands to entirely activate their receptors.
In summary, the zebrafish testis shows a unique, previously unreported among vertebrates, cellular pattern of lhcgr and fshr expression, because both Leydig and Sertoli cells express the mRNAs for both receptors. Particularly remarkable is the lhcgr expression by Sertoli cells, which may be related to the undifferentiated gonochoristic mode of sex differentiation in zebrafish. In a series of functional bioassays, rzfFSH showed a higher potency in stimulating the testicular steroidogenic system and a higher capacity to alter testicular gene expression profiles than rzfLH. These differences highlight the distinct functional domains of FSH and LH in regulating testis physiology in adult zebrafish, with a role for FSH as the constitutive driving force for both steroidogenesis and spermatogenesis, whereas the function of LH might be seen in context with an acute, additional steroid demand. Moreover, although pharmacological data show that the Fshr can be cross-activated by LH, this does not appear to happen in vivo or under culture conditions, because only FSH induced clear changes in the expression of selected testicular genes.
Supplementary Material
Acknowledgments
We thank DSM Food Specialties (Delft, The Netherlands) and SenterNovem (The Netherlands) for using their PALM MicroBeam Instrument at the Department of Cell Architecture and Dynamics (Utrecht University), G. Arkesteijn for technical assistance with FACS, Roland Romijn (U-Protein Express B.V.) for assistance in expression and purification of recombinant zebrafish gonadotropins, and H. C. Schriek and J. van Rootselaar (Utrecht University aquarium facility) for taking care of zebrafish stocks.
Footnotes
This work was supported by the Norwegian Research Council (Grant 159645/S40 to G.L.T., R.W.S., and J.B.), the National Institutes of Health (Grant DK69711 to J.B.), the Ramón Areces Foundation (Spain; to Á.G.-L), and the National Council for Scientific and Technological Development (Brazil; to R.H.N.).
Current address for Á.G.-L.: Institute of Marine Sciences of Andalucía (High Spanish Council for Scientific Research), Avenida República Saharaui 2, 11510 Puerto Real, Cádiz, Spain.
Disclosure Summary: The authors have nothing to disclose.
First Published Online March 22, 2010
Abbreviations: Cq, Threshold cycle; FACS, fluorescence-activated cell sorting; Fshr, FSH receptor; hCG, human chorionic gonadotropin; 3β-Hsd, 3β-hydroxysteroid dehydrogenase; 11-KT, 11-ketotestosterone; Lhcgr, LH/choriogonadotropin receptor; OHA, 11β-hydroxyandrostenedione; PKA, protein kinase A; qPCR, quantitative PCR; rzf, recombinant zebrafish.
References
- Pierce JG, Parsons TF 1981 Glycoprotein hormones: structure and function. Annu Rev Biochem 50:465–495 [DOI] [PubMed] [Google Scholar]
- McLachlan RI, Wreford NG, O'Donnell L, de Kretser DM, Robertson DM 1996 The endocrine regulation of spermatogenesis: independent roles for testosterone and FSH. J Endocrinol 148:1–9 [DOI] [PubMed] [Google Scholar]
- Kumar TR 2005 What have we learned about gonadotropin function from gonadotropin subunit and receptor knockout mice? Reproduction 130:293–302 [DOI] [PubMed] [Google Scholar]
- Petersen C, Soder O 2006 The Sertoli cell: a hormonal target and ‘super’ nurse for germ cells that determines testicular size. Horm Res 66:153–161 [DOI] [PubMed] [Google Scholar]
- Saez JM 1994 Leydig cells: endocrine, paracrine, and autocrine regulation. Endocr Rev 15:574–626 [DOI] [PubMed] [Google Scholar]
- Meng X, Lindahl M, Hyvönen ME, Parvinen M, de Rooij DG, Hess MW, Raatikainen-Ahokas A, Sainio K, Rauvala H, Lakso M, Pichel JG, Westphal H, Saarma M, Sariola H 2000 Regulation of cell fate decision of undifferentiated spermatogonia by GDNF. Science 287:1489–1493 [DOI] [PubMed] [Google Scholar]
- García-López A, Bogerd J, Granneman JC, van Dijk W, Trant JM, Taranger GL, Schulz RW 2009 Leydig cells express follicle-stimulating hormone receptors in African catfish. Endocrinology 150:357–365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Planas JV, Swanson P, Dickhoff WW 1993 Regulation of testicular steroid production in vitro by gonadotropins (GTH I and GTH II) and cyclic AMP in cocho salmon (Oncorhynchus kisutch). Gen Comp Endocrinol 91:8–24 [DOI] [PubMed] [Google Scholar]
- Planas JV, Swanson P 1995 Maturation-associated changes in the response of the salmon testis to the steroidogenic actions of gonadotropins (GTH I and GTH II) in vitro. Biol Reprod 52:697–704 [DOI] [PubMed] [Google Scholar]
- Kagawa H, Tanaka H, Okuzawa K, Kobayashi M 1998 GTH II but not GTH I induces final maturation and development of maturational competence of oocytes of red seabream in vitro. Gen Comp Endocrinol 112:80–88 [DOI] [PubMed] [Google Scholar]
- Kamei H, Ohira T, Yoshiura Y, Uchida N, Nagasawa H, Aida K 2003 Expression of a biologically active recombinant follicle stimulating hormone of Japanese eel Anguilla japonica using methylotropic yeast, Pichia pastoris. Gen Comp Endocrinol 134:244–254 [DOI] [PubMed] [Google Scholar]
- Weltzien FA, Norberg B, Swanson P 2003 Isolation and characterization of FSH and LH from pituitary glands of Atlantic halibut (Hippoglossus hippoglossus L.). Gen Comp Endocrinol 131:97–105 [DOI] [PubMed] [Google Scholar]
- Aizen J, Kasuto H, Golan M, Zakay H, Levavi-Sivan B 2007 Tilapia follicle-stimulating hormone (FSH): immunochemistry, stimulation by gonadotropin-releasing hormone, and effect of biologically active recombinant FSH on steroid secretion. Biol Reprod 76:692–700 [DOI] [PubMed] [Google Scholar]
- Zmora N, Kazeto Y, Kumar RS, Schulz RW, Trant JM 2007 Production of recombinant channel catfish (Ictalurus punctatus) FSH and LH in S2 Drosophila cell line and an indication of their different actions. J Endocrinol 194:407–416 [DOI] [PubMed] [Google Scholar]
- Molés G, Gómez A, Rocha A, Carrillo M, Zanuy S 2008 Purification and characterization of follicle-stimulating hormone from pituitary glands of sea bass (Dicentrarchus labrax). Gen Comp Endocrinol 158:68–76 [DOI] [PubMed] [Google Scholar]
- Kazeto Y, Kohara M, Miura T, Miura C, Yamaguchi S, Trant JM, Adachi S, Yamauchi K 2008 Japanese eel follicle-stimulating hormone (Fsh) and luteinizing hormone (Lh): production of biologically active recombinant Fsh and Lh by Drosophila S2 cells and their differential actions on the reproductive biology. Biol Reprod 79:938–946 [DOI] [PubMed] [Google Scholar]
- Miwa S, Yan L, Swanson P 1994 Localization of two gonadotropin receptors in the salmon gonad by in vitro ligand autoradiography. Biol Reprod 59:629–642 [DOI] [PubMed] [Google Scholar]
- Ohta T, Miyake H, Miura C, Kamei H, Aida K, Miura T 2007 Follicle-stimulating hormone induces spermatogenesis mediated by androgen production in Japanese eel, Anguilla japonica. Biol Reprod 77:970–977 [DOI] [PubMed] [Google Scholar]
- Prat F, Sumpter JP, Tyler CR 1996 Validation of radioimmunoassays for two salmon gonadotropins (GTH I and GTH II) and their plasma concentrations throughout the reproductive cycle in male and female rainbow trout (Oncorhynchus mykiss). Biol Reprod 54:1375–1382 [DOI] [PubMed] [Google Scholar]
- Gomez JM, Weil C, Ollitrault M, Le Bail PY, Breton B, Le Gac F 1999 Growth hormone (GH) and gonadotropin subunit gene expression and pituitary and plasma changes during spermatogenesis and oogenesis in rainbow trout (Oncorhynchus mykiss). Gen Comp Endocrinol 113:413–428 [DOI] [PubMed] [Google Scholar]
- Sohn YC, Yoshiura Y, Kobayashi M, Aida K 1999 Seasonal changes in mRNA levels of gonadotropin and thyrotropin subunits in the goldfish, Carassius auratus. Gen Comp Endocrinol 113:436–444 [DOI] [PubMed] [Google Scholar]
- Vischer HF, Granneman JC, Linskens MH, Schulz RW, Bogerd J 2003 Both recombinant African catfish LH and FSH are able to activate the African catfish FSH receptor. J Mol Endocrinol 31:133–140 [DOI] [PubMed] [Google Scholar]
- Bogerd J, Granneman JC, Schulz RW, Vischer HF 2005 Fish FSH receptors bind LH: How to make the human FSH receptor to be more fishy? Gen Comp Endocrinol 142:34–43 [DOI] [PubMed] [Google Scholar]
- So WK, Kwok HF, Ge W 2005 Zebrafish gonadotropins and their receptors. II. Cloning and characterization of zebrafish follicle-stimulating hormone and luteinizing hormone subunits: their spatial-temporal expression patterns and receptor specificity. Biol Reprod 72:1382–1396 [DOI] [PubMed] [Google Scholar]
- Sambroni E, Le Gac F, Breton B, Lareyre JJ 2007 Functional specificity of the rainbow trout (Oncorhynchus mykiss) gonadotropin receptors as assayed in a mammalian cell line. J Endocrinol 195:213–228 [DOI] [PubMed] [Google Scholar]
- Bogerd J, Blomenröhr M, Andersson E, van der Putten HH, Tensen CP, Vischer HF, Granneman JC, Janssen-Dommerholt C, Goos HJ, Schulz RW 2001 Discrepancy between molecular structure and ligand selectivity of a testicular follicle-stimulating hormone receptor of the African catfish (Clarias gariepinus). Biol Reprod 64:1633–1643 [DOI] [PubMed] [Google Scholar]
- Kobayashi M, Aida K, Hanyu I 1986 Gonadotropin surge during spawning in male goldfish. Gen Comp Endocrinol 62:70–79 [DOI] [PubMed] [Google Scholar]
- Krøvel AV, Olsen LC 2002 Expression of a vas::EGFP transgene in primordial germ cells of the zebrafish. Mech Dev 116:141–150 [DOI] [PubMed] [Google Scholar]
- Westerfield M 1995 The zebrafish book. A guide for the laboratory use of zebrafish (Danio rerio). Eugene, OR: University of Oregon Press; 1–26 [Google Scholar]
- Good-Avila SV, Yegorov S, Harron S, Bogerd J, Glen P, Ozon J, Wilson BC 2009 Relaxin gene family in teleosts: phylogeny, syntenic mapping, selective constraint, and expression analysis. BMC Evol Biol 9:293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leal MC, Cardoso ER, Nóbrega RH, Batlouni SR, Bogerd J, França LR, Schulz RW 2009 Histological and stereological evaluation of zebrafish (Danio rerio) spermatogenesis with an emphasis on spermatogonial generations. Biol Reprod 81:177–187 [DOI] [PubMed] [Google Scholar]
- Kurita K, Sakai N 2004 Functionally distinctive testicular cell lines of zebrafish to support male germ cell development. Mol Reprod Dev 67:430–438 [DOI] [PubMed] [Google Scholar]
- Houwing S, Kamminga LM, Berezikov E, Cronembold D, Girard A, van den Elst H, Filippov DV, Blaser H, Raz E, Moens CB, Plasterk RH, Hannon GJ, Draper BW, Ketting RF 2007 A role for piwi and piRNAs in germ cell maintenance and transposon silencing in zebrafish. Cell 129:69–82 [DOI] [PubMed] [Google Scholar]
- Iwai T, Yoshii A, Yokota T, Sakai C, Hori H, Kanamori A, Yamashita M 2006 Structural components of the synaptonemal complex, SYCP1 and SYCP3, in the medaka fish Oryzias latipes. Exp Cell Res 312:2528–2537 [DOI] [PubMed] [Google Scholar]
- Yano A, Suzuki K, Yoshizaki G 2008 Flow-cytometric isolation of testicular germ cells from rainbow trout (Oncorhynchus mykiss) carrying the green fluorescent protein gene driven by trout vasa regulatory regions. Biol Reprod 78:151–158 [DOI] [PubMed] [Google Scholar]
- Sawatari E, Shikina S, Takeuchi T, Yoshizaki G 2007 A novel transforming growth factor-β superfamily expressed in gonadal somatic cells enhances primordial germ cell and spermatogonial proliferation in rainbow trout (Oncorhynchus mykiss). Dev Biol 301:266–275 [DOI] [PubMed] [Google Scholar]
- de Waal PP, Wang DS, Nijenhuis WA, Schulz RW, Bogerd J 2008 Functional characterization and expression analysis of the androgen receptor in zebrafish (Danio rerio) testis. Reproduction 136:225–234 [DOI] [PubMed] [Google Scholar]
- Leal MC, de Waal PP, García-López A, Chen SX, Bogerd J, Schulz RW 2009 Zebrafish primary testis tissue culture: an approach to study testis function ex vivo. Gen Comp Endocrinol 162:134–138 [DOI] [PubMed] [Google Scholar]
- Schulz RW, van der Corput L, Janssen-Dommerholt C, Goos HJTh 1994 Sexual steroids during puberty in male African catfish (Clarias gariepinus): serum levels and gonadotropin-stimulated testicular secretion in vitro. J Comp Physiol B 164:195–205 [Google Scholar]
- Schulz RW, de França LR, Lareyre JJ, LeGac F, Chiarini-Garcia H, Nobrega RH, Miura T 2010 Spermatogenesis in fish. Gen Comp Endocrinol 165:390–411 [DOI] [PubMed] [Google Scholar]
- Takahashi H 1977 Juvenile hermaphroditism in the zebrafish, Brachydanio rerio. Bull Fac Fish Hokkaido Univ 28:57–65 [Google Scholar]
- Orban L, Sreenivasan R, Olsson PE 2009 Long and winding roads: testis differentiation in zebrafish. Mol Cell Endocrinol 312:35–41 [DOI] [PubMed] [Google Scholar]
- Maack G, Segner H 2003 Morphological development of the gonads in zebrafish. J Fish Biol 62:895–906 [Google Scholar]
- Albrecht KH, Eicher EM 2001 Evidence that Sry is expressed in pre-Sertoli cells and Sertoli and granulosa cells have a common precursor. Dev Biol 240:92–107 [DOI] [PubMed] [Google Scholar]
- Nakamura S, Aoki Y, Saito D, Kuroki Y, Fujiyama A, Naruse K, Tanaka M 2008 Sox9b/sox9a2-EGFP transgenic medaka reveals the morphological reorganization of the gonads and a common precursor of the female and male supporting cells. Mol Reprod Dev 75:472–476 [DOI] [PubMed] [Google Scholar]
- Wang XG, Orban L 2007 Anti-Müllerian hormone and 11β-hydroxylase show reciprocal expression to that of aromatase in the transforming gonad of zebrafish males. Dev Dyn 236:1329–1338 [DOI] [PubMed] [Google Scholar]
- Guigon CJ, Coudouel N, Mazaud-Guittot S, Forest MG, Magre S 2005 Follicular cells acquire Sertoli cell characteristics after oocyte loss. Endocrinology 146:2992–3004 [DOI] [PubMed] [Google Scholar]
- Couse JF, Hewitt SC, Bunch DO, Sar M, Walker VR, Davis BJ, Korach KS 1999 Postnatal sex reversal of the ovaries in mice lacking estrogen receptors α and β. Science 286:2328–2331 [DOI] [PubMed] [Google Scholar]
- Uhlenhaut NH, Jakob S, Anlag K, Eisenberger T, Sekido R, Kress J, Treier AC, Klugmann C, Klasen C, Holter NI, Riethmacher D, Schütz G, Cooney AJ, Lovell-Badge R, Treier M 2009 Somatic sex reprogramming of adult ovaries to testes by FOXL2 ablation. Cell 139:1130–1142 [DOI] [PubMed] [Google Scholar]
- Uchida D, Yamashita M, Kitano T, Iguchi T 2002 Oocyte apoptosis during the transition from ovary-like tissue to testes during sex differentiation of juvenile zebrafish. J Exp Biol 205:711–718 [DOI] [PubMed] [Google Scholar]
- Themmen APN, Huhtaniemi IT 2000 Mutations of gonadotropins and gonadotropin receptors: elucidating the physiology and pathophysiology of pituitary-gonad function. Endocr Rev 21:551–583 [DOI] [PubMed] [Google Scholar]
- Yan L, Swanson P, Dickhoff WW 1992 A two-receptor model for salmon gonadotropins (GTH I and GTH II). Biol Reprod 47:418–427 [DOI] [PubMed] [Google Scholar]
- Andersson E, Nijenhuis W, Male R, Swanson P, Bogerd J, Taranger GL, Schulz RW 2009 Pharmacological characterization, localisation and quantification of expression of gonadotropin receptors in Atlantic salmon (Salmo salar L.) ovaries. Gen Comp Endocrinol 163:329–339 [DOI] [PubMed] [Google Scholar]
- Kwok HF, So WK, Wang Y, Ge W 2005 Zebrafish gonadotropins and their receptors. I. Cloning and characterization of zebrafish follicle-stimulating hormone and luteinizing hormone receptors: evidence for their distinct functions in follicle development. Biol Reprod 72:1370–1381 [DOI] [PubMed] [Google Scholar]
- Wade MG, Van der Kraak G 1991 The control of testicular androgen production in the goldfish: effects of activators of different intracellular signaling pathways. Gen Comp Endocrinol 83:337–344 [DOI] [PubMed] [Google Scholar]
- Amiri RM, Maebayashi M, Adachi S, Moberg GP, Doroshov SI, Yamauchi K 1999 In vitro steroidogenesis by testicular fragments and ovarian follicles in a hybrid sturgeon, Bester. Fish Physiol Biochem 21:1–14 [Google Scholar]
- Schulz RW, Liemburg M, García-López A, Dijk W, Bogerd J 2008 Androgens modulate testicular androgen production in African catfish (Clarias gariepinus) depending on the stage of maturity and type of androgen. Gen Comp Endocrinol 156:154–163 [DOI] [PubMed] [Google Scholar]
- Lister A, Van Der Kraak G 2002 Modulation of goldfish testicular testosterone production in vitro by tumor necrosis factor α, interleukin-1β, and macrophage conditioned media. J Exp Zool 292:477–486 [DOI] [PubMed] [Google Scholar]
- Martins RS, Fuentes J, Almeida O, Power DM, Canario AV 2009 Ca2+-calmodulin regulation of testicular androgen production in Mozambique tilapia (Oreochromis mossambicus). Gen Comp Endocrinol 162:153–159 [DOI] [PubMed] [Google Scholar]
- Stocco DM, Wang X, Jo Y, Manna PR 2005 Multiple signaling pathways regulating steroidogenesis and steroidogenic acute regulatory protein expression: more complicated than we thought. Mol Endocrinol 19:2647–2659 [DOI] [PubMed] [Google Scholar]
- Manna PR, Chandrala SP, Jo Y, Stocco DM 2006 cAMP-independent signaling regulates steroidogenesis in mouse Leydig cells in the absence of StAR phosphorylation. J Mol Endocrinol 37:81–95 [DOI] [PubMed] [Google Scholar]
- Stocco DM, Clark BJ 1996 Regulation of the acute production of steroids in steroidogenic cells. Endocr Rev 17:221–244 [DOI] [PubMed] [Google Scholar]
- Kyle AL, Stacey NE, Peter RE, Billard R 1985 Elevation in gonadotrophin concentrations and milt volumes as a result of spawning behavior in the goldfish. Gen Comp Endocrinol 57:10–22 [DOI] [PubMed] [Google Scholar]
- Chen SX, Bogerd J, García-López A, de Jonge H, de Waal PP, Hong WS, Schulz RW 2010 Molecular cloning and functional characterization of a zebrafish nuclear progesterone receptor. Biol Reprod 82:171–181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mylonas CC, Fostier A, Zanuy S 2010 Broodstock management and hormonal manipulations of fish reproduction. Gen Comp Endocrinol 165:516–534 [DOI] [PubMed] [Google Scholar]
- Houk CP, Pearson EJ, Martinelle N, Donahoe PK, Teixeira J 2004 Feedback inhibition of steroidogenic acute regulatory protein expression in vitro and in vivo by androgens. Endocrinology 145:1269–1275 [DOI] [PubMed] [Google Scholar]
- Eacker SM, Agrawal N, Qian K, Dichek HL, Gong EY, Lee K, Braun RE 2008 Hormonal regulation of testicular steroid and cholesterol homeostasis. Mol Endocrinol 22:623–635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Rienzo G, Aniello F, Branno M, Izzo G, Minucci S 2006 The expression level of frog relaxin mRNA (fRLX), in the testis of Rana esculenta, is influenced by testosterone. J Exp Biol 209:3806–3811 [DOI] [PubMed] [Google Scholar]
- Laguë E, Tremblay JJ 2008 Antagonistic effects of testosterone and the endocrine disruptor mono-(2-ethylhexyl) phthalate on INSL3 transcription in Leydig cells. Endocrinology 149:4688–4694 [DOI] [PubMed] [Google Scholar]
- Sadeghian H, Anand-Ivell R, Balvers M, Relan V, Ivell R 2005 Constitutive regulation of the Insl3 gene in rat Leydig cells. Mol Cell Endocrinol 241:10–20 [DOI] [PubMed] [Google Scholar]
- Kawamura K, Kumagai J, Sudo S, Chun SY, Pisarska M, Morita H, Toppari J, Fu P, Wade JD, Bathgate RA, Hsueh AJ 2004 Paracrine regulation of mammalian oocyte maturation and male germ cell survival. Proc Natl Acad Sci USA 101:7323–7328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X, Lin SW, Kobayashi M, Ge W 11 July 2008 Expression of recombinant zebrafish follicle-stimulating hormone (FSH) in methylotropic yeast Pichia pastoris. Fish Physiol Biochem 10.1007/s10695-008-9244-z [DOI] [PubMed] [Google Scholar]
- Ko H, Park W, Kim DJ, Kobayashi M, Sohn YC 2007 Biological activities of recombinant Manchurian trout FSH and LH: their receptor specificity, steroidogenic and vitellogenic potencies. J Mol Endocrinol 38:99–111 [DOI] [PubMed] [Google Scholar]
- Kasuto H, Levavi-Sivan B 2005 Production of biologically active tethered tilapia LHβα by the methylotrophic yeast Pichia pastoris. Gen Comp Endocrinol 140:222–232 [DOI] [PubMed] [Google Scholar]
- Hayakawa Y, Morita T, Kitamura W, Kanda S, Banba A, Nagaya H, Hotta K, Sohn YC, Yoshizaki G, Kobayashi M 2008 Biological activities of single-chain goldfish follicle-stimulating hormone and luteinizing hormone. Aquaculture 274:408–415 [Google Scholar]
- Kobayashi M, Morita T, Ikeguchi K, Yoshizaki G, Suzuki T, Watabe S 2006 In vivo biological activity of recombinant goldfish gonadotropins produced by baculovirus in silkworm larvae. Aquaculture 256:433–442 [Google Scholar]
- Ben-Menahem D 2004 Single chain variants of the glycoprotein hormones and their receptors as tools to study receptor activation and analogue design. J Neuroendocrinol 16:171–177 [DOI] [PubMed] [Google Scholar]
- Fidler AE, Lin JS, Lun S, Ng Chie W, Western A, Stent V, McNatty KP 2003 Production of biologically active tethered ovine FSHβα by the methylotrophic yeast Pichia pastoris. J Mol Endocrinol 30:213–225 [DOI] [PubMed] [Google Scholar]
- Legardinier S, Poirier JC, Klett D, Combarnous Y, Cahoreau C 2008 Stability and biological activities of heterodimeric and single-chain equine LH/chorionic gonadotropin variants. J Mol Endocrinol 40:185–198 [DOI] [PubMed] [Google Scholar]
- Fan QR, Hendrickson WA 2005 Structure of human follicle-stimulating hormone in complex with its receptor. Nature 433:269–277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugahara T, Pixley MR, Minami S, Perlas E, Ben-Menahem D, Hsueh AJ, Boime I 1995 Biosynthesis of a biologically active single peptide chain containing the human common α and chorionic gonadotropin β subunits in tandem. Proc Natl Acad Sci USA 92:2041–2045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fralish GB, Narayan P, Puett D 2003 Consequences of single-chain translation on the structures of two chorionic gonadotropin yoked analogs in α-β and β-α configurations. Mol Endocrinol 17:757–767 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.