SUMMARY
Varicella vaccine was licensed in Canada in 1998, and a publicly funded vaccination programme introduced in the province of Alberta in 2001. In theory the vaccination programme might increase the burden of disease from shingles, making it important to develop baseline data against which future comparisons can be made. The study's aim was to describe the epidemiology of non-fatal cases of shingles for which publicly funded health services were utilized for the period 1986–2002. Shingles cases were identified from the records of Alberta's universal, publicly funded health-care insurance system for 1986–2002. The earliest dated health service utilizations for ICD-9-CM codes of 053 or ICD-10-CA codes of B02 were classified as incident. Diagnostic codes at least 180 days after the first were classified as recurrent episodes. Denominators for rates were estimated using mid-year population estimates from the Alberta Health Care Insurance Plan Registry. Annual age- and sex-specific rates were estimated. We explored the pattern of rates for sex, age and year effects and their interactions. Shingles rates increased between 1986 and 2002. There was a sex effect and evidence of an age–sex interaction. Females had higher rates than males at every age; however, the difference between females and males was greatest for the 50–54 years age group and declined for older age groups. The increased rate of shingles in Alberta began before varicella vaccine was licensed or publicly funded in Alberta, and thus cannot be attributed to vaccination.
INTRODUCTION
Shingles (herpes zoster) occurs when latent varicella zoster virus (VZV) in the dorsal root ganglia reactivate after a primary episode of chickenpox. Increasing age and immunosuppression contribute to VZV reactivation. The introduction of mass vaccination programmes against varicella [1] raises concerns that shingles cases might increase in the long term, due to a loss of periodic exposure to varicella (contact with cases of varicella is significantly protective against shingles among persons who have already had chickenpox). By reducing varicella cases (and the accompanying risk of exposure to the virus), this protective effect could be lost, resulting in an increase in shingles cases.
Varicella vaccine was licensed in Canada in 1998. The province of Alberta initiated a publicly funded vaccination programme in April 2001 which initially targeted non-immune special groups (e.g. health-care workers, contacts of the immunosuppressed, women of child-bearing age) and fifth-grade school children who had no history of chickenpox, shingles or prior varicella vaccination. Non-immune adolescents aged 13–19 years became eligible in April 2003. These targeted programmes are scheduled for completion in 2010. A programme targeting preschoolers born on or after 1 January 1997 began in the spring of 2002 and is scheduled to end in April 2007. A universal programme for 1-year-old children began in July 2001 and will continue.
Surveillance of shingles is important in any jurisdiction in which a varicella vaccination programme is planned or implemented. Few data are available for Canada or Alberta. We used administrative data from Alberta's publicly funded and administered health-care system to describe the epidemiology of non-fatal cases of shingles in Alberta for the period 1986–2002.
METHODS
Data sources
Over 99% of Alberta residents are covered by the provincial health-care insurance plan. Information on each insured person is maintained in a provincial registration file that includes demographic information as well as a unique lifetime personal health number (PHN) that can be used to link the registration file to a variety of health data sources. Alberta collects a health-care insurance premium on a quarterly basis, therefore the accuracy of the registry is maintained during inter-censal periods [2, 3]. This source provides accurate population counts by geography and is used by Statistics Canada in the development of post-censal population projections. The system changed from a paper to an electronic system in the 1980s and extensive work was done to remove duplicates and the records of deceased persons. The data are thus considered to be reliable from 1986 onwards. An electronic fee-for-service data system (‘claims’) is maintained to pay practitioners through the provincial health insurance plan. Virtually all Alberta physicians (>99%) bill the provincial government on a fee-for-service basis. To support the payment of a claim, practitioners must supply information that includes the PHN, a unique practitioner identifier, up to three diagnostic (four-digit ICD-9-CM) codes, and a procedure code indicating the service performed [2]. The Hospital Morbidity Inpatient Database records hospital separations and includes up to 16 five-digit ICD-9-CM codes as well as the most responsible diagnosis (the diagnosis having the most influence on length of stay). ICD-10-CA codes were used starting in the fiscal year 2002/2003. From 1992/1993 onwards, this database also contains the unique PHN.
Definitions
The PHN was used for deterministic linkage across the databases. Shingles cases were identified as any individual visiting a physician where an ICD-9-CM code of 053 or an ICD-10-CA code of B02 was recorded in any of the three fields available in a physician record or in any of the 16 diagnostic fields available in a hospital record. Incident cases were defined on the basis of the earliest date for which a shingles diagnostic code in any diagnostic field was identified in any of the databases. Data for persons with more than one shingles code were included as incident only for the earliest date at which the code was identified. A person was considered to have experienced a shingles recurrence if a diagnostic code was found with a date more than 180 days after a prior code.
Data analysis
Incidence rates were estimated using mid-year population estimates from the provincial registration files. The proportions of cases identified from claims vs. the Hospital Morbidity database were examined.
Shingles rates were calculated for each year and examined using graphical displays. We used a sequence of logistic regression analyses to explore for sex, age, and year effects and their interactions. First, age group, sex, and year were each considered as categorical variables and a model was solved with main effects for age group, sex, and year, and an interaction for age group by sex. Then, after an examination of the year effect suggested that year could be treated as a polynomial, the models were re-solved. Finally, interactions between age group and the age factors were added to the model. SAS [4] and SPSS [5] software were used for data analysis.
Ethical approval
The Conjoint Health Research Ethics Board of the University of Calgary approved the study.
RESULTS
The proportion of cases that were identified only from the Hospital database was 4·9% in 1993 and declined steadily to 2·9% in 2002. Of the cases identified exclusively from the Hospital database, one-third or fewer had shingles as a primary diagnosis (data not shown).
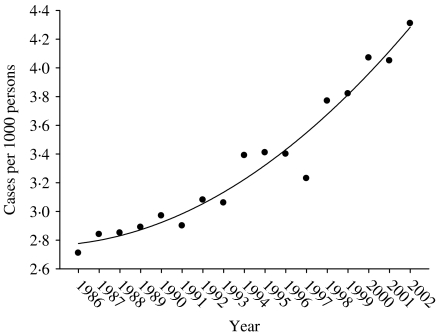
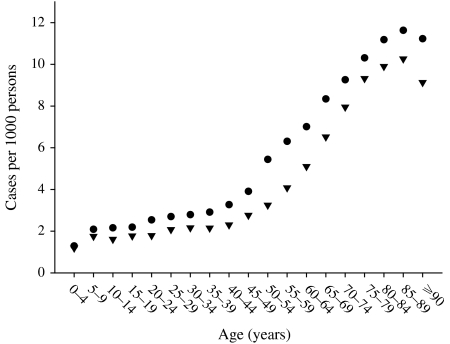
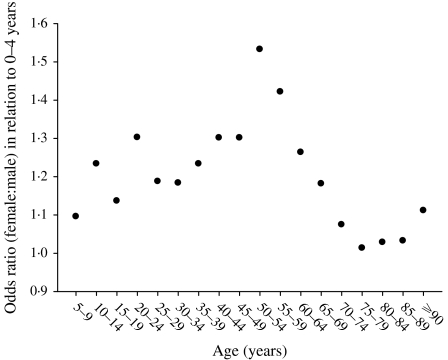
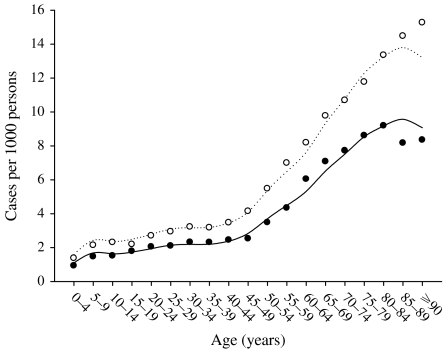
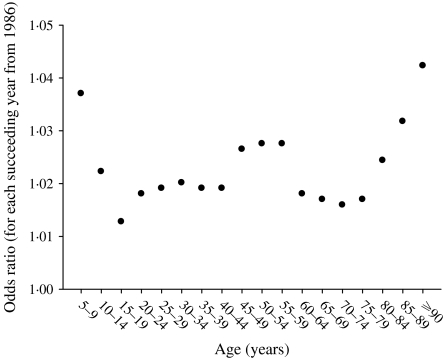
Figure 1 shows the main effect for year (with the quadratic fit superimposed). It shows that the zoster rate is increasing, and that this increase is accelerating. Figure 2 shows the age and sex effects. Females have higher rates at every age. The zoster rates increase markedly starting at ages in the mid 40s and reach an apparent maximum at ages in the late 80s. There is an interaction between age and sex that is presented separately in Figure 3, although it is also apparent in Figure 2. Specifically, the difference between female and male rates appears to increase gradually with age reaching a maximum at age range 50–54 years and then decreases at older ages (Fig. 3). Moreover, there has been a change in the age structure as the years have progressed. Figure 4 shows the sex-combined age structure for the first year (1986) and the last year (2002) respectively. The modelled fit is also shown. The interaction between age and year evident in Figure 4 is isolated in Figure 5. This figure shows a differential rate of increase among the middle ages (45–59) and the oldest (⩾85), and suggests that there may be an increase in the very young as well, although the small number of cases does not allow definitive conclusions.
Fig. 1.
Treated zoster rate by year, Alberta 1986–2002.
Fig. 2.
Treated zoster rate by age and sex, Alberta 1986–2002 combined. ●, Female; ▼, male.
Fig. 3.
Odds ratios, female to male by age, Alberta 1986–2002 combined.
Fig. 4.
Treated zoster rates by age. Alberta, 1986 (●) and 2002 (○). ––, 1986 fitted; ·······, 2002 fitted.
Fig. 5.
Odds ratio for age groups for each year after 1986.
Most persons (93·9%) had only one episode of shingles. A larger proportion of females than males had one or more recurrences at every age; however, there was no evidence of an age–sex interaction (data not shown).
DISCUSSION
We used administrative records for Alberta's universal, publicly funded health-care insurance plan; thus any temporal trend in incidence would not be due to a change over time in access to health care; a potential problem with studies that use data from health maintenance organizations or sentinel practices [6–8] where there may be financial or other barriers to accessing health care. Our study, like some others [9], has the advantage of being truly population based.
Throughout the period 1986–2002 we observed an increase in the rate of shingles in Alberta that accelerated over time. The increase began prior to 1998 when varicella vaccines were first licensed in Canada. Shingles rates were observed to be increasing elsewhere in Canada in the 1980s [9]. Publicly funded varicella vaccination programmes were introduced in Alberta in 2001, and mathematical models predict that any increase in shingles from the implementation of a vaccination programme would not be anticipated to be seen until at least 5 years after programme implementation [10].
We found that females have higher rates at every age and that there is an interaction between age and sex: the differences in rate increase with age to a maximum at age range 50–54 years and then decreases at older age. We further found that the rate of increase of shingles over time is higher among middle-aged and older persons than among younger persons. Investigators from the United Kingdom and the United States have similarly observed a higher rate of shingles among females compared to males and their data are also suggestive of an age–sex interaction [6, 7]; while another team, despite a display of data that is strongly suggestive of an age–sex interaction concluded simply that there was overall, no difference in rates between males and females [8].
In Massachusetts during 1998–2003 (post-vaccine era), an increase in the rate of shingles was observed while chickenpox rates declined according to data from the Behavioral Risk Factors Surveillance System [11]. Previous studies in the United Kingdom have found the mean annual incidence of shingles to be stable in each year for the period 1994–2001 [12], although the annual overall shingles consultation rate was observed to slowly increase during the 1990s in England and Wales [13].
We did not validate the diagnostic codes in our study. However, although the positive predictive values of a herpes zoster diagnosis vary by age, other investigators have found that the age-specific variation in rates of herpes zoster persist after adjustment for the predictive values of diagnoses [8]. The most likely bias resulting from failure to validate diagnostic codes would be an over-estimation of the numbers of cases. We may have underestimated the number of cases in our study as we included only those cases that sought medical care. Our findings for the secular trend over the period studied could be posited to be due to one or more of: a consistent secular trend of physicians making a diagnosis of shingles, changes in the validity of diagnostic codes for shingles, a true change in incidence, or an increase in consultation rates for the condition without a true increase in incidence (possibly due to changes in therapeutics such as availability of anti-viral drugs). A true change in incidence might be due to an increase in immunosuppressive conditions or therapies over time [14]. Racial and geographic factors have been associated with the epidemiology of both chickenpox and shingles [15]. Primary infection with VZV occurs at later ages in tropical regions than in temperate regions, which might lead such persons to have a later onset of shingles. In the United States racial differences have been noted in shingles: community-dwelling elderly black persons were less likely than whites to have had shingles. Despite high population growth, the proportion of Albertans who are foreign born declined during 1991–2001, while the proportion of visible minorities (primarily Chinese and South Asians) increased from 9·4% in 1991 to 11·2% in 2001 [16, 17]. We do not think the small increase in visible minorities in Alberta is sufficiently large to explain the observed temporal trends. We lacked the data to explore such explanations.
The observed higher rate of shingles in females compared to males may or may not reflect a true finding. If females had a higher rate of health service utilization at all ages than did males, there would be an increased opportunity for females to obtain a shingles diagnosis. Gender bias in diagnosis might also be explanatory, although we think it unlikely. There may also be age-sex specific differences in risks of immunosuppressive conditions and therapies [14]. Alternately, females may differ from males in their immune response to latent viral infections although such an inherent difference would not be expected to result in a secular trend of increase in disease incidence.
Females might have more contact with children (and thus exposure to chickenpox) than males. Thus, the observed higher rates of shingles among females are in the opposite direction of that which would be anticipated if repeated exposures to chickenpox reduced the risk of acquiring shingles. Persons who are at higher risk of shingles because of health or immunosuppressive treatment may be less likely to live and work with children; and people aged >50 years are less likely to live with children than others [18]. However, such a trend would have to be differential between males and females to explain our findings. If Albertans aged >50 years are less likely to live with children than younger persons, we would expect an increasing trend of shingles among older than younger persons (as observed); but such an explanation would also require variation by sex to explain our findings.
Ongoing surveillance of shingles is important given the introduction of varicella vaccination programmes, particularly since changes in disease patterns attributable to vaccination programmes may not be observed for many years after high rates of vaccination coverage and lower rates of circulating wild virus have been attained. However, surveillance of shingles alone will not sufficiently permit an understanding of the impact of chickenpox vaccination programmes on shingles incidence. Future epidemiological studies should consider co-existing health conditions; include validation sub-studies for diagnostic coding, information on the vaccination status of the population and test for age–sex interactions in rates.
ACKNOWLEDGEMENTS
This study was funded by an unconditional grant in aid from Merck Frosst Canada Ltd.
DECLARATION OF INTEREST
None.
REFERENCES
- 1.Brisson M, Edmunds WJ, Gay JN. Varicella vaccination: impact of vaccine efficacy on the epidemiology of VZV. Journal of Medical Virology. 2003;70:S31–S37. doi: 10.1002/jmv.10317. (Suppl. 1): [DOI] [PubMed] [Google Scholar]
- 2.Yiannakoulias N et al. Doctors, patients and influenza-like illness: clinicians or patients at risk? Public Health. 2004;118:527–531. doi: 10.1016/j.puhe.2004.02.005. [DOI] [PubMed] [Google Scholar]
- 3.Alberta Health and Wellness. Population Projections for Alberta and its Health Regions: models and methods. Edmonton: Alberta Health and Wellness; 1998. [Google Scholar]
- 4.Statistical Analysis System Institute Inc. SAS version 8.0. Cary, NC: SAS Inc.; 1999. [Google Scholar]
- 5.SPSS Inc. SPSS version 12. Chicago, IL, USA: [Google Scholar]
- 6.Fleming DM et al. Gender difference in the incidence of shingles. Epidemiology and Infection. 2004;132:1–5. doi: 10.1017/s0950268803001523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Insinga RP et al. The incidence of herpes zoster in a United States administrative database. Journal of General Internal Medicine. 2005;20:748–753. doi: 10.1111/j.1525-1497.2005.0150.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Donahue JG et al. The incidence of herpes zoster. Archives of Internal Medicine. 1995;155:1605–1609. [PubMed] [Google Scholar]
- 9.Brisson M et al. Epidemiology of varicella zoster virus infection in Canada and the United Kingdom. Epidemiology and Infection. 2001;127:305–314. doi: 10.1017/s0950268801005921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brisson M et al. Exposure to varicella boosts immunity to herpes-zoster: implications for mass vaccination against chickenpox. Vaccine. 2002;20:2500–2507. doi: 10.1016/s0264-410x(02)00180-9. [DOI] [PubMed] [Google Scholar]
- 11.Yih WK et al. The incidence of varicella and herpes zoster in Massachusetts as measured by the Behavioral Risk Factor Surveillance System (BRFSS) during a period of increasing varicella vaccine coverage, 1998–2003. BMC Public Health. 2005;5:68. doi: 10.1186/1471-2458-5-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chapman RS, Cross KW, Fleming DM. The incidence of shingles and its implications for vaccination policy. Vaccine. 2003;21:2541–2547. doi: 10.1016/s0264-410x(03)00034-3. [DOI] [PubMed] [Google Scholar]
- 13.Brisson M, Edmunds WJ. Epidemiology of Varicella-Zoster Virus in England and Wales. Journal of Medical Virology. 2003;70:S9–14. doi: 10.1002/jmv.10313. (Suppl. 1): [DOI] [PubMed] [Google Scholar]
- 14.Mullooly JP et al. Incidence of herpes zoster, 1997–2002. Epidemiology and Infection. 2005;133:245–253. doi: 10.1017/s095026880400281x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nagasako EM et al. Geographic and racial aspects of herpes zoster. Journal of Medical Virology. 2003;70:S20–23. doi: 10.1002/jmv.10315. [DOI] [PubMed] [Google Scholar]
- 16.Statistics Canada http://www12.statcan.ca/english/census01/products/analytic/companion/etoimm/provs.cfm. http://www12.statcan.ca/english/census01/products/analytic/companion/etoimm/provs.cfm . Proportion of foreign-born, Canada, provinces and territories, 1991, 1996 and 2001 ( ). Accessed 24 August 2006.
- 17.Statistics Canada http://www12.statcan.ca/english/census01/products/analytic/companion/etoimm/provs.cfm#diversity_varied. http://www12.statcan.ca/english/census01/products/analytic/companion/etoimm/provs.cfm#diversity_varied . 2001 census. Proportion of visible minorities, Canada, provinces and territories, 1991, 1996 and 2001. Canada's ethnocultural portrait: the changing mosaic ( ). Accessed 26 August 2006.
- 18.Jumaan AO et al. Incidence of herpes zoster, before and after varicella-vaccination-associated decreases in the incidence of varicella, 1992–2002. Journal of Infectious Diseases. 2005;191:2002–2007. doi: 10.1086/430325. [DOI] [PubMed] [Google Scholar]