Abstract
Purpose
The target antigens of graft-versus-leukemia that are tumor-associated are incompletely characterized.
Experimental Design
We examined responses developing against CML66, an immunogenic antigen preferentially expressed in myeloid progenitor cells identified from a patient with chronic myelogenous leukemia who attained long-lived remission following CD4+ donor lymphocyte infusion (DLI).
Results
From this patient, CML66-reactive CD8+ T cell clones were detected against an endogenously presented HLA-B*4403-restricted epitope (HDVDALLW). Neither CML66-specific antibody nor T cell responses were detectable in peripheral blood before DLI. However, by one month after DLI, CD8+ T cells were present in peripheral blood, and at 10-fold higher frequency in marrow. Subsequently, plasma antibody to CML66 developed in association with disease remission. Donor-derived CML66-reactive T cells were detected at low levels in vivo in marrow prior to DLI by ELISpot and by a nested polymerase chain reaction-based assay to detect clonotypic T cell receptor sequences, but not in blood of the patient pre-DLI, nor of the graft donor.
Conclusions
CD4+ DLI results in rapid expansion of pre-existing marrow-resident leukemia-specific donor CD8+ T cells, followed by a cascade of antigen-specific immune responses detectable in blood. Our single-antigen analysis thus demonstrates that durable post-transplant tumor immunity is directed in part against nonpolymorphic overexpressed leukemia antigens, that elicit coordinated cellular and humoral immunity.
Keywords: Donor lymphocyte infusion, graft-versus-leukemia, leukemia antigen, chronic myeloid leukemia, T cell
INTRODUCTION
While much progress has been recently made in the development of potent immunotherapy reagents, the generation of consistent tumor immunity through defined-antigen vaccination remains elusive (1–3). In this context, dissecting the mechanisms of response in instances of clinically evident anti-tumor immunity can be highly instructive. Allogeneic hematopoietic stem cell transplant (HSCT) for leukemia remains the most established example of human immunity, whose curative effect depends on mounting donor-derived immune responses, called the graft versus leukemia (GvL) effect (4). The results of donor lymphocyte infusion (DLI) for treating relapsed chronic myelogenous leukemia (CML) further illustrate the potency of GvL (5, 6). In this procedure, lymphocytes derived from the donor are infused—typically without further radiation or chemotherapy—and consistently result in durable remissions in 70–80% CML patients.
One approach to dissecting immunity following DLI and HSCT is through identification of antigens targeted by these therapies. Studies in patients and mouse models have firmly established allo-antigens as an important antigen class targeted by GvL, but mounting evidence suggests that tumor-associated antigens elicit at least part of this response (7–12). We therefore focused our analysis on immune responses directed against CML66. This CML-associated antigen was previously identified to elicit high-titer antibodies in a CD4+-DLI treated individual, who developed prompt GvL without clinically evident graft-versus-host disease (GvHD). CML66 is not an allo-antigen, as no sequence differences within CML66 between donor and recipient were discovered (13). On the other hand, CML66 is highly expressed in leukemia cells, and particularly in CD34+ myeloid progenitor cells (14).
In the current study, we analyzed immunity against CML66 as a means to characterize the kinetics of developing GvL responses. Since CD4+ T cell responses can aid generation of both antigen-specific antibody and CD8+ T cell responses, we queried whether development of potent B cell responses indicated presence of antigen-specific cytolytic T cell immunity. From peripheral blood, we isolated CD8+ cytotoxic T cell clones directed against a CML66-derived HLA-B4403 restricted 8mer peptide. We found expansion of functional CML66-specific T cells in peripheral blood and marrow as early as 1 month after DLI, in close temporal association with antibody responses. Intriguingly, donor-derived CML66-specific T cells were detected in the marrow even prior to DLI, that were not present in T cells of the original donor. Together, our analysis of the responses to this single leukemia-associated antigen suggests that DLI-induced GvL is a multi-component process. This process involves both antigen-priming to generate a pool of pre-existing leukemia-specific T cells residing in the marrow reservoir before DLI, functional anti-leukemia cytotoxic T cell responses in marrow and peripheral blood, together with leukemia-directed antibody responses and recruitment of new antigen-specific T cells developing after DLI. Our study provides evidence that effective GvL responses comprise long-lived and coordinated immunity against overexpressed leukemia antigens.
MATERIALS AND METHODS
Patient samples
Heparinized blood, skin biopsies or marrow aspirates were obtained from patients and normal donors enrolled on clinical research protocols at the Dana-Farber Cancer Institute (DFCI). All clinical protocols were approved by the DFCI Human Subjects Protection Committee. Peripheral blood (PBMC) or bone marrow mononuclear cells (BMMC) from normal donors and patients were isolated by Ficoll/Hypaque density-gradient centrifugation, cryopreserved with 10% DMSO, and stored in vapor-phase liquid nitrogen until the time of analysis. Plasma was isolated after centrifugation of whole blood and cryopreserved at −80°C until use. Primary skin fibroblast cultures were generated from 5 mm diameter skin punch biopsies that were minced into <0.5 mm fragments, affixed to culture dishes with fibrin clots, and fed with MCDB106/M199 medium (1:1 v/v) (GIBCO/Life Technologies, Carlsbad, CA) +10 ng/ml EGF (Millipore, Billerica, MA) + 0.4µg/ml hydrocortisone (EMD Biosciences, San Diego, CA). Fibroblasts that migrated out and divided from these explants were suspended 7 days later with trypsin/EDTA, subcultured in the same medium and expanded.
Sources of antigen
We synthesized ninety-one overlapping 15- to 18-mers (unpurified) encompassing the coding region of CML661 and a series of 8-mer, 9-mer and 10-mers (>85% purity) to define the peptide epitope of CML66-reactive T cells (New England Peptide, Gardner, MA). Synthetic peptides encoding the cytomegalovirus (CMV) pp65 (NLVPMVATV) and the Mart126–35 epitopes (M26, EAAGIGILTV) were used as controls. Peptides were reconstituted in DMSO (10 mg/ml) and stored at −20°C. CML66 was cloned into a standard E1- and E3-deleted adenoviral vector, and expressed under the control of a bi-directional CMV immediate early gene promoter that expressed GFP simultaneously. The Ad/CML66 plasmid was constructed and produced following standard adenoviral vector production protocols (Jeng-Shin Lee and Richard Mulligan; Harvard Gene Therapy Initiative). CML66 was also PCR-cloned into pcDNA3.1 (5’-primer: AGCTTTAAGCTTCACCATGGAGGTGGCGGCTAATTGCCCCTAC; 3’primer: ATAGAATTCTGCAGTCAATGGTGATGGTGATGATGATTCTCTGTATTTACTTTTATTAA).
Generation of antigen presenting cells (APCs)
Patient EBV-transformed B cell lines were generated by incubating PBMC with supernatant from B95-8 Marmoset cells, and maintained in RPMI 1640, supplemented with 10% FCS, and antibiotics. For the generation of dendritic cells (DCs), CD14+ cells were immunomagnetically isolated (Miltenyi Biotec, Auburn CA) per manufacturer´s guidelines, and cultured in IMDM (Invitrogen, Carlsbad, CA) supplemented with 10% human AB serum (Cellgro, Herndon, VA), 2 mM glutamine, 50 µg/ml human transferrin (Roche, Basel, Switzerland), 5 µg/ml human insulin (Sigma-Aldrich, St. Louis, MO), and 15 µg/ml gentamycin (Invitrogen) in the presence of 50 ng/ml GM-CSF (Genzyme, Cambridge, MA) and 20 ng/ml IL-4 (R&D Systems, Minneapolis, MN). On days three and six, 50% fresh medium with cytokines was added. On day seven, cells were matured for 48 hours with 1 µg/ml prostaglandin E2 (Sigma-Aldrich), 10 ng/ml IL-1β (BD Bioscience, San Jose, CA), 1000 U/ml IL-6 (BD Bioscience) and 10 ng/ml TNFα (Genzyme). CD40-B cells were generated from PBMC by activation on CD40L-expressing irradiated feeder cells in the presence of IL-4 (R&D Systems) and cyclosporin A (Novartis, Basel, Switzerland) as described (15). The MHC class I-deficient immortalized B lymphoblastoid cell line 721.221 was retrovirally infected with individual plasmids encoding the full-length B*4403, B*4402 A*203, A*3301 alleles (16), and maintained in DMEM supplemented with 10% FCS, 2mM L-glutamine, antibiotics and 1mM sodium pyruvate. MUTZ-3 cells (DSMZ, Braunschweig, Germany) were cultured in alpha-MEM (Invitrogen), supplemented with 20% FBS and GM-CSF (10 ng/ml; R&D Systems).
Expression of CML66 in APCs
CML66 was introduced into DCs by adenoviral transduction. Ad/CML66 viral supernatant was spin-infected (MOI=100; 1250 g at 37C for 2 hours) onto 0.4 × 106 immature DCs, that were seeded in 24-well plates in IMDM supplemented with 10% human sera. Immediately following spin-infection, DC maturation cytokines were added. Infected DCs were used 24–48 hours later. By flow cytometry, infection routinely generated GFP expression in 70–90% of APCs. For some experiments, CML66 was introduced into DCs or CD40L-stimulated B cells using RNA nucleofection. For production of CML66 transcript, endotoxin-free CML66/pcDNA3.1 plasmid (Endofree Maxiprep Kit, Invitrogen, Carlsbad CA) was linearized with XbaI (NE Biolabs, Beverly, MA) and in vitro transcribed and polyadenylated (mMessage mMachine; Ambion, Foster City, CA). Mart1 transcripts were similarly generated following linearization of pOBT7-Mart1 cDNA (ATCC) with Ssp1 (NE Biolabs, Beverly, MA). Polyadenylated RNA (2–10 µg) was nucleofected into 2 million CD40-B cells in 100 µl of PBS/10% HEPES buffer (Program Q-004, Amaxa Nucleofector Device; Lonza Inc., Walkersville, MD).
Cloning of CML66-specific T cells
Autologous mature DCs (1 × 105 cells/well) were adenovirally transduced to express CML66, and cultured with thawed post-DLI CD3+ T cells (2 × 106 cells/2ml) with IL-7 (10 ng/ml; Endogen Inc., Woburn, MA) on day 0 and IL-2 (100 IU/ml; Amgen, Thousand Oaks, CA) starting on day 1. Medium was replenished twice weekly with fresh IL-2. Ten days following this single stimulation, CD8+ T cells were immunomagnetically-selected (Miltenyi Biotec, Auburn CA), and tested by ELISpot against autologous matured DCs pulsed with various pools of CML66-derived overlapping peptides (10 µg/ml/peptide). Reactive T cells were expanded using irradiated peptide 66–72-pulsed autologous EBV cells in the presence of 1% PHA and 100 IU/ml of recombinant IL-2. This population was further enriched by selection of cells secreting IFNγ in response to peptide 72-pulsed autologous EBV cells (IFN-γ Catch Reagent, Miltenyi Biotec, Auburn CA), and cloned by limiting dilution on feeder cells (irradiated allogeneic PBMC and EBV cells, with 100 U/ml recombinant human IL2 and PHA). Clones displaying specific anti-CML66 reactivity (see assays below) were further expanded using similar culture conditions.
Detection of antigen-specific T cells
Cytolytic assays were performed with Europium-labeled target cells (PerkinElmer, Wellesley MA) per manufacturer’s directions. Target cells (5,000 cells/well) were labelled for 30 minutes with Europium, washed extensively, co-incubated at different effector-to-target cell ratios in triplicate for 2 hours, and specific Europium release was measured. ELISpot was performed using peptide-pulsed target cells (50,000 cells/well) coincubated with 200–1000 T cell clones/well in duplicate in ELISpot plates (Millipore, Billerica, MA) for 24 hours. Interferon-γ secretion (IFNγ) was detected using capture and detection antibodies as directed (Mabtech AB, Mariemont, OH) and imaged (ImmunoSpot Series Analyzer; Cellular Technology, Cleveland, OH). To test dependence on class I of T cell reactivity, ELISpot plates were first coated with APCs in the presence of class I blocking antibody (W6/32) for 2 hours at room temperature, prior to introduction of T cells into the wells. Antigen-specific T cell reactivity was also detected by IFNγ secretion assay, per manufacturer’s recommendations (Miltenyi Biotec, Auburn CA), and labelled cells were analyzed by flow cytometry (Beckman-Coulter FC500).
Chimerism analysis
Quantitative sequencing of single nucleotide polymorphisms (SNPs) informative between Patient A and her donor was performed, as previously described (17). We identified informative polymorphisms using a panel of SNPs with high minor allele frequency, that were tested against recipient-derived genomic DNA (gDNA) (extracted from patient fibroblasts) and donor-derived gDNA (from donor PBMC). Once identified, these loci were tested against gDNA extracted from immunomagnetically-selected (Miltenyi Biotec, Auburn CA) marrow-derived CD8+ T cells (Pyrosequencing AB, Uppsala, Sweden).
Detection and quantitation of TCR Vβ clonotypes
We modified our previous method for T cell receptor (TCR) spectratyping (18) into a two-step process. First, we identified the dominant Vβ subfamily among the 24 known Vβ subfamilies. We generated 5 pools of Vβ forward primers (pool 1: Vβ 1–5.1; pool 2: Vβ 5.2–9; pool 3: Vβ 10–13.2; pool 4: Vβ 14–19; and pool 5: Vβ 20–24; see Supplementary Table 3). RNA extracted from the T cell clones (QIAamp RNA Blood Mini-kit; Qiagen, Valencia, CA), was reverse transcribed into cDNA (Superscript, GIBCO BRL, Gaithersburg, MD) using random hexamers, and PCR-amplified in five separate 20 µl volume reactions, each consisting of: 10 µl of GeneAmp Fast Master Mix (Applied Biosystems, Foster City, CA), pooled Vβ subfamily forward primers (0.5 µM per primer, per pool), 0.5 µM of reverse primer specific for the TCR constant region (Cβ external primer), and 100–200 ng cDNA. Second, T cell clone-derived cDNA was reamplified with each of the 5 individual primers contained within a positive pool together with a FAM-conjugated Cβ reverse (internal) primer. For both steps, the amplification conditions were: 95C for 20 seconds; followed by 35 cycles of 95C for 3 seconds, 55C for 7 seconds, 60C for 30 seconds; and then 60 C for 2 minutes. The amplified fragment was cloned into pCR2.1-TOPO (Invitrogen, Carlsbad, CA), and sequenced. Based on this sequence, a quantitative Taqman PCR assay was designed such that a sequence-specific probe was located within the region of junctional diversity (Applied Biosystems; Foster City, CA). To determine the size distribution of the amplified Vβ subfamily, the fluorescent PCR product was elecrophoresed on a 3730 DNA sequencer (Applied Biosystems, Foster City, CA) and analyzed (Genemapper software, v.4.0; Applied Biosystems, Foster City, CA).
To detect clonotypic Vβ transcripts directly from patient RNA, nested PCR was performed first using the clone-specific Vβ forward primer (0.5 µM) and Cβ reverse primer (0.5 µM), together with cDNA (4 µl) and GeneAmp Fast PCR Master Mix (10 µl) in a 20 µl reaction volume, using identical amplification conditions as described above. Subsequently, 4 µl of this PCR product was amplified with 1 µl of the clone CDR3 region-specific primer and probe, and 10 µl of Taqman Fast Universal PCR Master Mix (Applied Biosystems, Foster City, CA) in a total volume of 20 µl. The PCR amplification conditions were: 95C for 20 minutes × 1 cycle, and 40 cycles of 95C for 3 seconds followed by 60C for 30 seconds (7500 Fast Real-time PCR cycler; Applied Biosystems, Foster City, CA). Test transcripts were quantified relative to GAPDH transcripts by calculating 2^(GAPDH CT-target CT).
Peptide ELISA
Individual or pools of peptides (5 µg/mL) were applied to Nunc C96 Maxisorp plates (Fisher, Pittsburgh, PA) in carbonate buffer (pH 9.6) overnight at 4C, then blocked in PBS-0.05% Tween with 2% milk. Plasma samples were added to duplicate wells in blocking buffer (1:200 dilution) and incubated 3 hrs at 25C. Peptide binding of patient immunoglobulin was detected using an alkaline phosphatase-conjugated anti-human IgG secondary antibody (1:1000 dilution, Jackson ImmunoResearch; West Grove, PA) and visualized with p-nitrophenyl phosphate substrate (Sigma-Aldrich, St. Louis, MO). Absorbance was read at 405 nm. Positive peptide-reactivity was defined as signal greater than two standard deviations above the mean of 10 normal volunteers.
RESULTS
Detection of CD8+ T cells reactive against CML66 in a DLI responder
We previously discovered potent antibody responses (at titers > 1:64,000) against the CML-associated protein CML66 that developed in close temporal correlation with disease regression following donor lymphocyte infusion (DLI) in Patient A (13). CML66 has unknown function, although residues 268–399 are homologous to NUDC, which is involved in nuclear distribution during mitosis (19). Patient A relapsed with CML within one year after T cell-depleted myeloablative matched-related donor HSCT, and was infused with 3 × 108 donor CD4+ T cells/kg 4.5 years after transplant, to which she developed prompt cytogenetic remission followed by durable molecular remission (>10 years) (Suppl. Table 1). We speculated that such high-titer antibody responses would coordinately develop with an antigen-specific cellular memory response. We therefore stimulated T cells isolated from the Patient A, 5 years after DLI, against autologous dendritic cells (DCs) that were adenovirally transduced to express whole CML66 antigen, since this led to efficient expression of this protein within DCs (Suppl. Figure 1A).
Ten days after this single stimulation against CML66-expressing DCs, CD8+ T cells from Patient A were tested by ELISpot for reactivity against a panel of overlapping peptides, encompassing the entire coding region of CML66. A total of ninety-one 15- to 18mers, which overlapped by 11 residues, were divided into 18 pools (Supplementary Table 2). Patient A was known to express HLA-A2+, and was CMV-seropositive. As expected, Patient A CD8+ T cells stimulated with DCs expressing either the empty adenoviral vector or CML66 both elicited equivalent frequencies of CMV-specific reactivity against the known HLA-A2+ restricted PP65 peptide epitope (Suppl. Figure 1B). Of the CML66-derived peptide pools, pool 15 elicited the greatest T cell reactivity (80 spots per 1 × 105 cells) by ELISpot, with 2.6-fold greater reactivity from CML66-stimulated cells compared with T cells stimulated against control DCs. Further testing of the 6 peptides contained within pool 15 revealed that the CD8+ T cell reactivity was directed against the 18mer 66–72 (CML66 residues 449–466: PKEMPCFCLRHDVDALLW) (Suppl. Fig. 2).
Peptide 66–72 contains a naturally processed, class I-restricted epitope of CML66
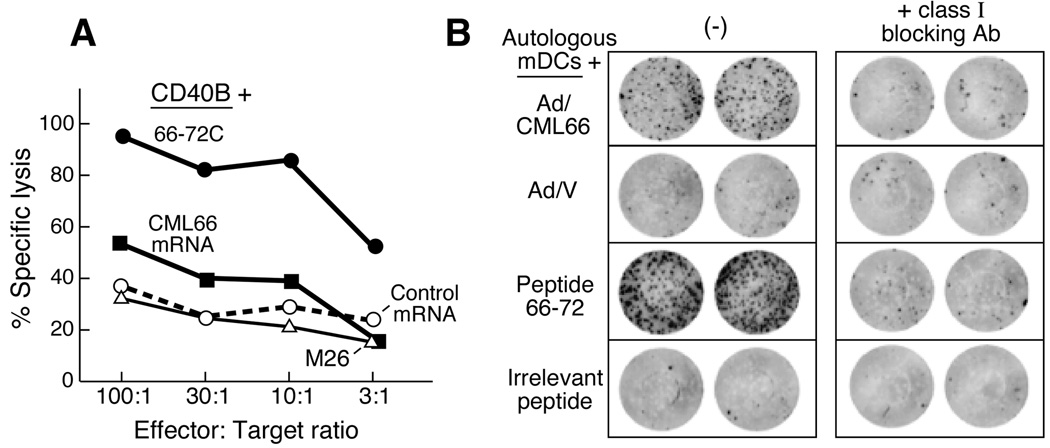
We sought to determine whether peptide 66–72 reactive CD8+ T cells could recognize an epitope derived from processed whole antigen. As antigen-presenting cells (APCs), we used CD40L-expanded autologous B cells, that could efficiently express CML66 or irrelevant/control proteins following nucleofection with gene-specific mRNA (Suppl. Figure 3). As shown in Figure 1A, peptide 66–72-reactive T cells were cytolytic to autologous CD40L-expanded B cells expressing CML66 and APCs pulsed with peptide 66–72, but not against autologous CD40L-B cells expressing irrelevant antigen or pulsed with irrelevant peptide (i.e. the melanoma antigen MART1 or its peptide M26). These results demonstrate that with forced expression, CML66 is processed to express an epitope containing peptide 66–72.
Figure 1. Identification of a CML66-derived peptide epitope eliciting a T cell response in DLI responder Patient A.
A. Peptide 66–72 contains a naturally processed epitope of CML66. By cytolysis assay, peptide 66–72-reactive T cells are cytolytic to cells expressing CML66 whole antigen (using autologous CD40L activated B cells, nucleofected with CML66 RNA as targets, black squares), or pulsed with peptide 66–72C (black circles), compared to CD40L-B cells nucleofected to expressing irrelevant antigen (MART1, open circles) or pulsed with irrelevant peptide (M26, open triangles). This figure demonstrates representative results from 3 independent experiments. B. Recognition of the peptide 66–72 epitope is HLA class I restricted. Peptide 66–72-specific T cell clones (500 cells/well) were tested by ELISpot assay against autologous mature DCs either transduced to express whole CML66 antigen (Ad/CML66) or vector (Ad/V); or pulsed with peptide 66–72 or irrelevant peptide; in the presence or absence of class I blocking antibody (W6/32 antibody).
Peptide 66–72-reactive T cells were cloned by limiting dilution. Clones 3A1, 9A8 and 11A5 all specifically recognized autologous DCs pulsed with peptide 66–72 or expressing whole CML66 protein, and were further expanded. Figure 1B demonstrates that these interactions were class I-restricted, since IFN-γ secretion was blocked in the presence of the class I blocking antibody, W6/32.
Peptide 66–72C is the HLA*B4403-restricted epitope of CML66
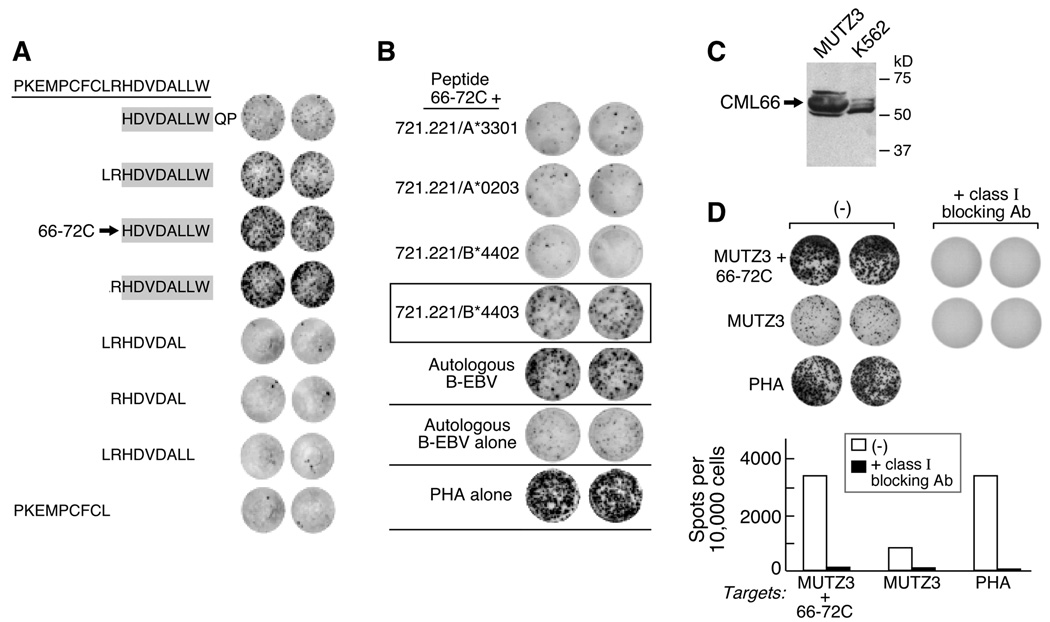
To identify the minimal class I-binding epitope of CML66, 8-, 9- and 10-mer peptides derived from the parental 18mer, peptide 66–72, were pulsed onto autologous DCs and tested against clones 3A1, 9A8 and 11A5 by ELISpot. Figure 2 shows that maximal IFNγ secretion was observed by all three clones when tested against either the 8mer HDVDALLW (designated as peptide 66–72C), or the 9mer RHDVDALLW. The C-terminal residue W appeared to be critical for the TCR-MHC complex interaction, since its absence resulted in loss of IFN-γ secretion.
Figure 2. CML66 is processed and presented by CD34+ CML66+ myeloid leukemia cells in the context of HLA B4403.
A. The minimal epitope of the 18mer parental peptide 66–72 (PKEMPCFCLRHDVDALLW) is a 8-mer peptide (indicated by the arrow, designed peptide 66–72C). By ELISpot, 5000 antigen–specific T cells were tested per well, against autologous matured DCs pulsed with various 8-, 9- and 10mer peptides derived from peptide 66–72. Reactivity was detected among the peptides containing the 8mer HDVDALLW (shaded). B. Recognition of peptide 66–72C is restricted to HLA-B*4403 (boxed). Peptide 66–72C pulsed-721.221 cells (50,000 cells per well) expressing the individual HLA A and B alleles of Patient A were tested against 1000 cells of clone 9A8. Similar results were observed with clones 3A1 and 11A5. C. Protein lysates generated from MUTZ3 and K562 cells (20 µg/lane) were immunoblotted against the CML66 monoclonal antibody 22F. D. The 66–72 specific T cell clone recognizes MUTZ3 cells on ELISpot assay. This interaction can be blocked in the presence of class I MHC blocking antibody. This results were observed for clones 3A1, 9A8 and 11A5.
Patient A was known to express HLA A*0203, A*3301, B*1301, B*4403, Cw*0304, and Cw*0702. Comparison with the binding motifs from the SYFPEITHI database (20) showed that B*4403 is the only HLA allele from Patient A’s haplotype that could be predicted to accommodate W at the peptide C-terminus (motif x[EMILDV]xxxxxx[YFW]). The peptide binding scores were calculated using the NetMHCpan prediction system.(21) The predicted score for the 8-mer peptide HDVDALLW and B*4403 was 469.49 (weak binding) while all other combinations of peptides were predicted as non-binding interactions. Therefore, the 8-mer HDVDALLW was considered the best candidate epitope and B*4403 as restriction element.
We verified this prediction, by testing the peptide 66–72-reactive T cell clones against a series of EBV-immortalized B cell lines derived from individuals who shared at least one HLA allele with Patient A, that were pulsed with peptide 66–72. Of the 7 lines tested, 4 of 4 expressing HLA-B44 elicited reactivity from the CML66-reactive clones (Suppl Figure 4). These results were further confirmed in experiments using 721.22 cells, that in parental form are MHC class I-deficient, but which were engineered to express only a single MHC class I allele (16). As shown in Figure 2B, the CML66-reactive T cell clones were all reactive against HLA-B*4403 expressing cells, but not cells expressing HLA-A*0203, HLA-A*3301 or HLA-B*4402.
To confirm that peptide 66–72C is an endogenously processed, we tested the CML66-specific T cell clones against the HLA-B4403-expressing human CD34+ acute myeloid leukemia cell line, MUTZ-3 (22). MUTZ-3 cells express high levels of CML66 (Figure 2C). By ELISpot, CML66-reactive T cell clones recognized MUTZ3, and this recognition was abrogated in the presence of class I blocking antibody (Figure 2D).
CML66-specific CD8+ T cell responses in peripheral blood and marrow are coordinated with B cell responses following DLI
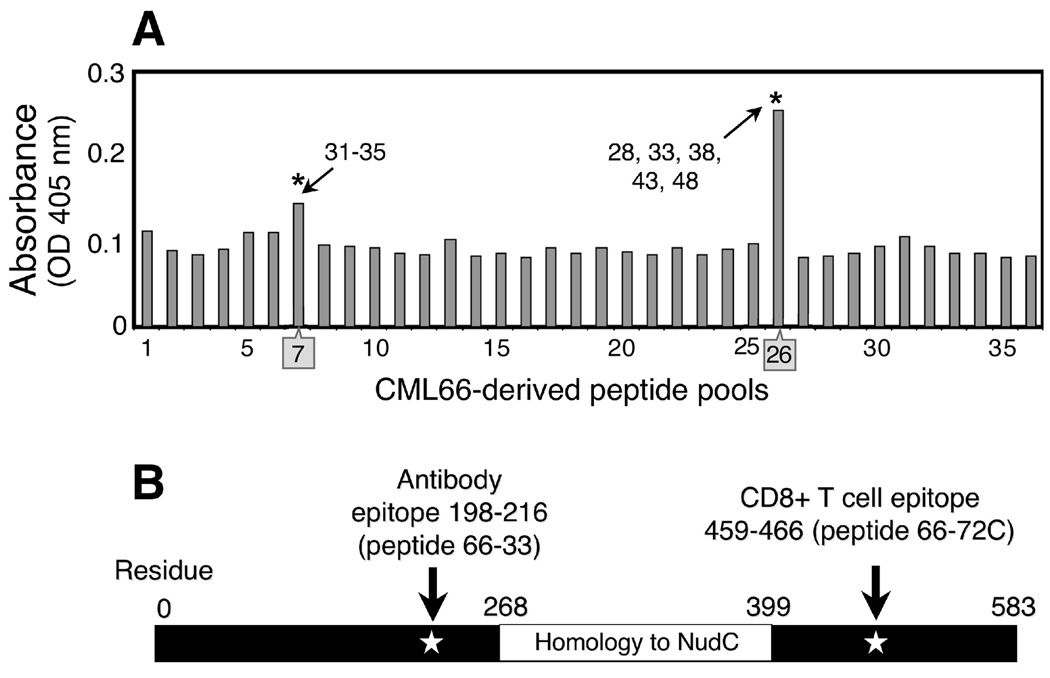
We previously demonstrated that CML66 elicits high-titer antibody reactivity in Patient A. To define the CML66-associated antibody epitope, we tested patient plasma against an array of 36 pools of overlapping CML66-derived 18mers (see Supplementary Table 2). Each peptide was represented in two separate pools. Post-DLI plasma, diluted 1:200, was reactive on ELISA assay against two pools—one containing peptides 66-31 through 66-35, and the other containing peptides 66-28, 66-33, 66-38, 66-43 and 66-48 (Figure 3A). Hence, we concluded that the CML66 B cell epitope was contained within peptide 66-33 (GFYVSLEWVTISKKNQDNK, residues 198–216). Plasma reactivity to peptide 66-33 was also detected at dilutions 1:500 and 1:1000. Although we cannot exclude that the patient plasma also contained antibodies reactive to nonlinear epitopes from CML66, we found the pattern of reactivity against 66-33 consistent with our previous results using recombinant whole protein as coating ELISA antigen (13). We detected clear reactivity against peptide 66-33 developing 2–3 months, that increased by 12 months following DLI, when the patient developed molecular remission (Figure 4), not present prior to DLI. The B and T cell epitopes of the CML66 antigen are thus positioned at different regions of CML66 (Figure 3B).
Figure 3. The CML66 B and T cell epitopes are positioned in different regions of the antigen.
A. Post-DLI plasma of Patient A is reactive against peptide pools 7 and 26, which contain peptide 66-33 (*). Plasma (1:200 dilution) was tested on ELISA in duplicate, against an array of peptide pools, each containing 5–6 peptides/pool (pools described in Supplementary Table 2). B. Schematic representation of the positions of the B and T cell epitopes of CML66.
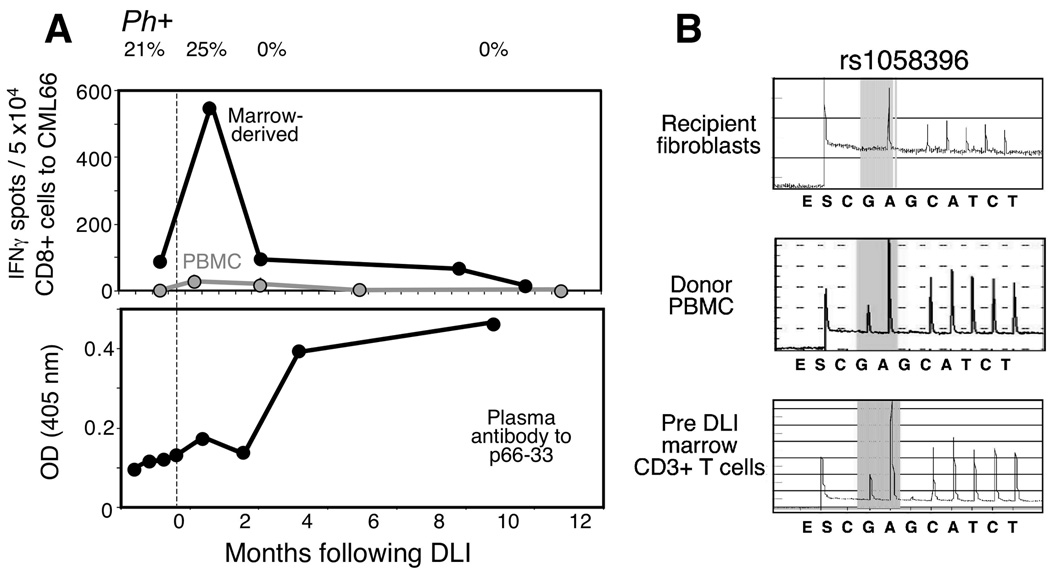
Figure 4. Expansion of the CML66-specific T cell population following DLI, that is coordinated with antigen-specific B cell response.
A. Expansion of CML66-reactive T cells from peripheral blood and marrow 1–3 months following DLI, detected by IFNγ ELISpot following presensitization with autologous DCs transduced to express CML66, and then restimulation with peptide 66–72C pulsed DCs. T cell reactivity was measured by calculating the number of spots in duplicate wells against CML66-expressing targets over control target cells. Plasma reactivity (diluted 1:200) before and after DLI against peptide 66-33 was measured by ELISA assay. Peptide specific OD was calculated as the difference in duplicate wells between OD (peptide) and OD (reagent control). Development of immune responses is depicted in relationship to % BCR-ABL (or, Philadelphia chromosome positive [Ph+]) cells in the marrow, B. Marrow-infiltrating T cells prior to DLI are donor-derived. Genomic DNA was extracted from recipient fibroblasts, donor PBMC and pre-DLI marrow CD3+ cells, and amplified for the region of polymorphism around SNP rs 1058396. Pyrosequencing revealed the amplicon sequence of marrow infiltrating T cells to be identical to the donor.
We next tested serial peripheral blood mononuclear cell (PBMC) and marrow samples for evidence of in vivo T cell reactivity against peptide 66–72C. PBMC and marrow before and in the months following DLI were thawed and presensitized to CML66 once using autologous DCs expressing CML66. Ten days later, CD8+ T cells were isolated and tested by ELISpot against peptide 66–72C-pulsed autologous DCs. In peripheral blood, antigen-specific CD8+ T cell reactivity was absent prior to DLI, but then readily detectable between 1–3 months following DLI (~45 IFNγ-secreting CD8+ T cells/50,000 cells), and thereafter waned (Figure 4A). More dramatically, marrow CML66-specific T cell reactivity peaked at approximately 1 month following DLI, coinciding with maximal peripheral blood response, but was of more than 10-fold greater frequency. We observed that marrow and blood-derived CML66-specific T cell responses peaked in the few weeks preceding cytogenetic elimination of BCR-ABL expressing cells within the marrow.
Donor-derived CML66-specific T cell clones are long-lived and present in marrow prior to DLI
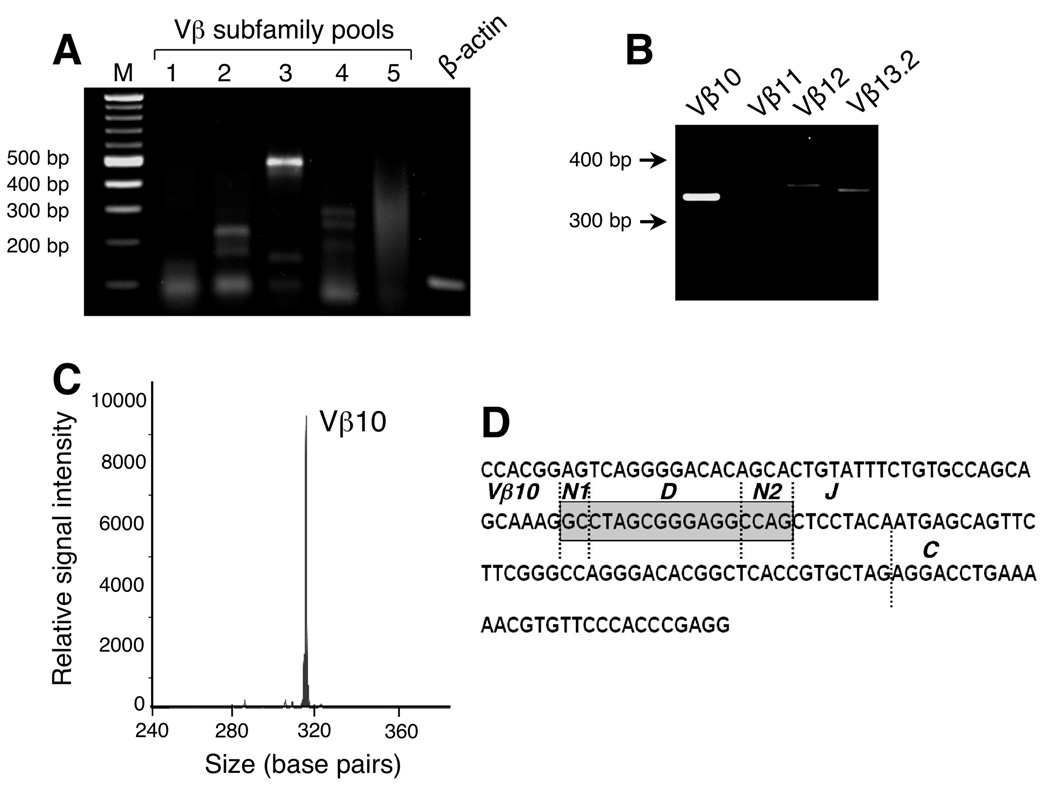
Intriguingly, ELISpot analysis revealed that CML66-reactive T cells were present in marrow prior to DLI, although at 5-fold less levels than the height of the DLI-associated response (100 spots per 5 × 104 cells). Chimerism analysis of pre-DLI marrow-derived T cells revealed that they were fully donor-derived (Figure 4B). To detect the CML66 epitope-specific T cell clones in vivo, we developed a sensitive nested PCR assay to amplify CDR3-specific sequences unique to the TCRs of the clones. As summarized in Figure 5, we first identified the specific CDR3 sequences of clones 3A1, 9A8 and 11A5 by RT-PCR using primers specific for each of the 24 Vβ subfamilies, that were pooled into 5 mixtures. We had insufficient material to complete the analysis for clone 3A1. However, as shown in Figure 5A, clone 11A5 expressed a Vβ subfamily included in pool 3, that was identified as Vβ10 (Figure 5B), while clone 9A8 expressed Vβ6 (data not shown). By spectratyping, the amplified Vβ6 and Vβ10 bands were confirmed to be derived from clonal cells, since only single peaks – representing CDR3 sequences derived from a single rearrangement -- were observed (Figure 5C). Next, CDR3-region specific quantitative PCR primers were designed, such that the probe sequences were positioned within the region of junctional diversity (Figure 5D).
Figure 5. Design of T cell clone-specific primers.
A. RNA from the CML66 T cell clone 11A5 expressed a Vβ subfamily included in primer pool 3. All Vβ subfamilies generated amplicons between 300–400 basepairs in length when using the Cβ reverse internal primer. B. Of the primers contained in primer pool 3, clone 11A5 was found to express Vβ10. C. The Vβ10 amplicon generated a monoclonal peak by spectratyping. D. Clone-specific primers for 11A5 were designed based on the sequence of the clone-specific CDR3 rearrangement, such that the quantitative PCR probe was positioned in the region of junctional diversity (shaded region).
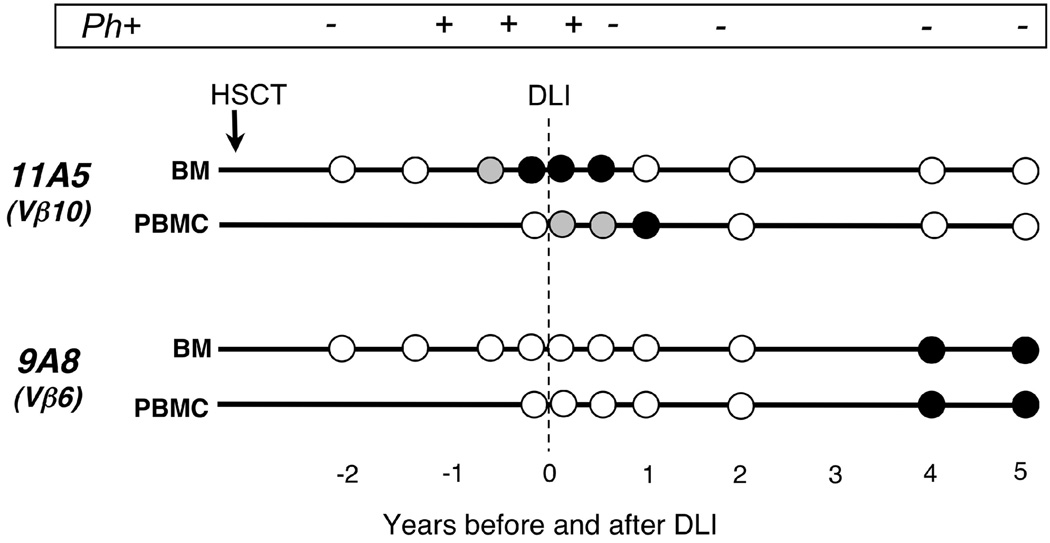
The results of our analysis are depicted in Figure 6. As expected, our assays efficiently amplified RNA derived from the 9A8 and 11A5 clones, but not from irrelevant T cell clones, normal marrow, nor normal PBMC, including T cells from the transplant donor (Suppl Table 4). Clone 11A5, however, was detectable by nested PCR in RNA directly extracted from Patient A’s peripheral blood only following DLI, while this same clone was detectable in marrow prior and again following DLI, with detectability tapering by 12 months post-DLI. As shown in Figure 6, clone 11A5 was detectable in marrow for several months before the patient received DLI treatment, when disease relapse was apparent by marrow cytogenetic evaluation. Five years after DLI, this clone was still present at very low frequency as it could only be detected following antigen-specific stimulation (data not shown). In contrast, clone 9A8 demonstrated a very different kinetic: while it was not detectable in blood nor marrow prior to DLI, nor within the first 24 months after DLI, it could be detected by nested PCR directly from PBMC and marrow 4–5 years after DLI. Together, these results demonstrate that CML66 elicits long-lived cellular immunity.
Figure 6. Pacing of accumulation of CML66-specific T cells before and after DLI in peripheral blood and marrow of Patient A, detected by clone-specific CDR3 nested PCR.
Each circle represents a tested sample. White circle = Negative: no amplification; Grey circle=Positive: amplification detected; black circle=double positive: amplification detected and amplification level is more than median level of all positive samples. Also shown is monitoring of tumor burden based on detection of Ph+ (BCR-ABL) chromosomes by marrow cytogenetic evaluation.
DISCUSSION
The role of adaptive B cell immunity in GvL responses following allogeneic HSCT has not been well characterized. We previously uncovered high-titer antibodies, developing in the setting of DLI-induced GvL responses, that targeted a broad array of intracellular, overexpressed and nonpolymoprhic leukemia-associated antigens. Although our studies are admittedly restricted to the analysis of responses against a single antigen in a single patient, our data provide intriguing evidence that leukemic antigen-specific antibodies can mark functional cytolytic CD8+ responses after HSCT. We performed detailed studies to dissect the kinetics of B and T cell immunity developing against a response-associated antigen, CML66, in an individual with long-lasting remission in the absence of graft-versus-host disease. We delineate a temporal sequence of activity by immune effectors in this patient, in which CML66-specific CD8+ T cell responses are first detectable in the marrow prior to DLI. Following infusion of donor CD4+ cells, we then observed antigen-specific CD8+ T cell reactivity expanding in both blood and marrow together with appearance of high-titer B cell responses directed against the same antigen coincident with attaining clinical remission. These data provide the first description of coordinated adaptive immunity developing against a bona fide leukemia-associated antigen in marrow and blood following transplantation.
We previously demonstrated that patients with robust GvL responses to DLI therapy develop potent humoral immunity, while nonresponders do not. Other investigators have demonstrated the potential of post-transplant B cell responses to provide functional anti-tumor immune effects (23). We discovered that T cell responses immediately preceded cytogenetic and increased B cell responses, and these results suggest T cell-mediated lysis of leukemia cells increases the accessibility of overexpressed antigens to B cells for antibody production. Given the long-lasting immunity associated with DLI, a focus of ongoing studies is determining whether antibodies also serve to enhance and amplify GvL to increase cross-presentation of antigen to APCs through antibody-mediated uptake of intracellular antigens (24–26).
GvL effects have been commonly attributed to cellular targeting of allo-antigens with lympho-hematopoietic restricted expression (27), while the extent and identity of GvL targets that are leukemia-associated, such as CML66, are unknown. Antigens with leukemia-restricted expression include products of chromosomal translocations, and mutated or over-, or aberrantly expressed self-proteins (28). For CML, these include the gene product of BCR-ABL, as well as WT-1 and proteinase 3, although the ability of these to elicit functional T cell responses following allotransplant has been unclear (29, 30). Our current studies uniquely provide evidence that GvL responses are comprised at least partly of donor immunity directed against nonpolymorphic leukemia-associated antigens. Furthermore, serologic responses may represent an efficient approach to identify response-associated leukemia-associated antigens. In addition to CML66, we have previously identified numerous B cell-defined antigens targeted by GvL in patients who have undergone allotransplant or DLI (10, 23). These were found to be highly expressed in leukemic cells and were without sequence polymorphisms between donor and recipient, and hence are not alloantigens. While we have not systematically examined concurrent T cell responses to these antigens, our current results suggest that a subset will likely elicit T-cell mediated tumor-specific recognition. Consistent with these findings, Nishida et al. recently identified several T cell clones reactive against tumor and not to recipient hematopoietic cells, developing in patients with clinical response following nonmyeloablative transplant for chronic lymphocytic leukemia.(9) Together, a major implication of our data is that leukemia-associated antigens discovered in the context of therapeutic response to allotransplant may be more numerous than previously appreciated. Their discovery may enable the development of strategies to more rationally enhance GvL effects without inciting GvHD, or even anti-tumor immunity in the absence of allo-grafting.
Our analysis of individual CML66-specific CD8+ T cell clones reveal several important insights regarding GvL. Firstly, as an antigen, CML66 appears capable of eliciting polyclonal T cell reactivity, as we discovered different Vβ specificities among the analyzed clones. While we predominantly focused on two clones here, our original limiting dilution analysis yielded far more antigen-specific clones, and probably several other Vβ specificities could have been uncovered. Moreover, CML66 likely contains other immunogenic regions (i.e within pools 13 and 17, Suppl Figure 1B), and epitopes within the context of other MHC class I restricting alleles. Suemori et al., for example, recently described a HLA-A24 defined epitope (residues 76–84) which elicits recognition of whole CML66-expressing leukemia cells (31, 32).
Secondly, CML66-specific clones are remarkably enduring in vivo. Even 5 years after the patient’s clinical response, we could still identify the 11A5 TCR that was present prior to DLI. Moreover, new CML66-specific T cells were recruited over time, as the 9A8 clone appeared only years after DLI. DLI results in T cell neogenesis and reconstitution of T cell repertoire diversity (33–35). Intermittent molecular detection of BCR-ABL transplant has been reported in successfully transplanted CML patients, and continued immune surveillance is likely critical in maintaining durable remission (36). Thus, our data suggest that one facet of DLI-induced GvL responses is the generation of enduring leukemia antigen-specific cellular immunity.
Thirdly, our studies provide supportive evidence that leukemia cells actively suppress the function of donor CD8+ cytolytic T cells. We could identify CML66-specific T cells residing in marrow prior to DLI, but we only observed functional T cell responses resulting in high IFN-γ secretion after DLI. Marrow has been long-appreciated as a reservoir for high-avidity memory B and T cells that are critical for sustaining long-term humoral and cellular immunity (37–39). Marrow is also a major priming site for T cell responses to blood-borne antigens (40). Recently, model systems have underscored the essential role of central memory effector T cells for sustaining long-lasting immunity (41), and the marrow compartment has been shown to harbor antigen-specific central memory T cells to tumor associated antigens (8, 42–44). At the same time, marrow is a known site of residual disease in hematologic malignancies. Studies in viral immunity have described T cell “exhaustion” in the setting of chronic antigen exposure (45). Leukemia cells that are residing in the marrow are a source of persistent antigen, and likely exert a myriad of inhibitory effects of immunity (29).
In closing, our studies suggest a new paradigm for understanding GvL, that involves immunity against leukemia-associated antigens together with allo-antigens, and that elicit complex immunity. Evidence of tumor-specific immunity following HSCT has several implications for developing approaches to stimulate immunity against malignancy in even the autologous setting. For example, generating coordinated B and T cell immunity may be an important characteristic to mimic. Secondly, our studies underscore the importance of efficient antigen priming to expand the pool of T cells with desired specificity whose function can be secondarily modulated. Many more questions are raised by our studies, including defining the relationship between antigen-specific CD4+ T cells against CML66 with developing CD8+ and antibody responses, and detailing how B cell immunity functions to modulate T cell responses. Future studies with this GvL antigen will likely provide further mechanistic insights that can be applied to developing effective vaccination strategies.
STATEMENT OF TRANSLATIONAL RELEVANCE.
A robust example of long-lasting human tumor immunity is the excellent results of donor lymphocyte infusion (DLI) for the treatment of relapsed chronic myeloid leukemia, but the underlying mechanisms of this complex immune response are poorly characterized. We identify and track CD8+ T cell immunity against a graft-versus-leukemia (GvL) antigen CML66 within the context of DLI-induced remission. Our results indicate that CD4+ DLI results in the rapid expansion of pre-existing marrow-resident leukemia-specific CD8+ T cells, followed by a cascade of peripheral B and T antigen-specific immune responses. Effective GvL thus comprises at least partly of immunity against nonpolymorphic B and T cell epitopes. The discovery of leukemia antigens within the context of therapeutic response, such as CML66, provide important tools for dissecting effective tumor immunity; insights gained from these studies provide a rational path for devising more specific and less toxic immunotherapy.
Supplementary Material
ACKNOWLEDGEMENTS
We would like to acknowledge the generous support from the DFCI Pasquarello Tissue Bank, and from the clinical transplant teams at the DFCI, Boston. We would like especially to thank excellent technical help from Linyun Chen, Sean McDonough and Patricia Barron and invaluable advice and assistance from Zach Herbert, Alicja Piechoka-Trocha, and Dr. Christian Brander.
J.G.R. and the Cell Culture Core of the Harvard Skin Disease Research Center were supported by P30 AR42689 from NIAMS. C.J.W. acknowledges support from the Department of Defense (W81XWH-07-1-0080), the Miles and Eleanor Shore Award, NCI (5R21CA115043-2), the Early Career Physician-Scientist Award of the Howard Hughes Medical Institute, and is a Damon-Runyon Clinical Investigator supported in part by the Damon-Runyon Cancer Research Foundation (CI-38-07)
Footnotes
None of the authors have conflicts of interest to disclose.
AUTHORSHIP CONTRIBUTIONS
Contribution: C.J.W. designed the study, performed research, analyzed data, and wrote the manuscript. W.Z., WYZ, S.A.R,, J.C. and T.S. performed critical experiments and analyzed data. V.B. analyzed data. C.M.C., E.P.A., and R.J.S. provided vital clinical samples. J.G.R. initiated skin fibroblast lines from transplant recipients. All authors edited the paper.
REFERENCES
- 1.Cheever MA. Twelve immunotherapy drugs that could cure cancers. Immunol Rev. 2008;222:357–368. doi: 10.1111/j.1600-065X.2008.00604.x. [DOI] [PubMed] [Google Scholar]
- 2.Cheever MA, Allison JP, Ferris AS, et al. The prioritization of cancer antigens: a national cancer institute pilot project for the acceleration of translational research. Clin Cancer Res. 2009;15(17):5323–5337. doi: 10.1158/1078-0432.CCR-09-0737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rosenberg SA, Yang JC, Restifo NP. Cancer immunotherapy: moving beyond current vaccines. Nat Med. 2004;10(9):909–915. doi: 10.1038/nm1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wu CJ, Ritz J. Induction of tumor immunity following allogeneic stem cell transplantation. Adv Immunol. 2006;90:133–173. doi: 10.1016/S0065-2776(06)90004-2. [DOI] [PubMed] [Google Scholar]
- 5.Collins RH, Jr, Shpilberg O, Drobyski WR, et al. Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol. 1997;15(2):433–444. doi: 10.1200/JCO.1997.15.2.433. [DOI] [PubMed] [Google Scholar]
- 6.Kolb HJ, Schattenberg A, Goldman JM, et al. Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. Blood. 1995;86(5):2041–2050. [PubMed] [Google Scholar]
- 7.Bellucci R, Wu CJ, Chiaretti S, et al. Complete response to donor lymphocyte infusion in multiple myeloma is associated with antibody responses to highly expressed antigens. Blood. 2004;103(2):656–663. doi: 10.1182/blood-2003-07-2559. [DOI] [PubMed] [Google Scholar]
- 8.Melenhorst JJ, Scheinberg P, Chattopadhyay PK, et al. High avidity myeloid leukemia-associated antigen-specific CD8+ T cells preferentially reside in the bone marrow. Blood. 2009;113(10):2238–2244. doi: 10.1182/blood-2008-04-151969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nishida T, Hudecek M, Kostic A, et al. Development of tumor-reactive T cells after nonmyeloablative allogeneic hematopoietic stem cell transplant for chronic lymphocytic leukemia. Clin Can Res. 2009 doi: 10.1158/1078-0432.CCR-09-0199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu CJ, Yang XF, McLaughlin S, et al. Detection of a potent humoral response associated with immune-induced remission of chronic myelogenous leukemia. J Clin Invest. 2000;106(5):705–714. doi: 10.1172/JCI10196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bellucci R, Alyea EP, Chiaretti S, et al. Graft-versus-tumor response in patients with multiple myeloma is associated with antibody response to BCMA, a plasma-cell membrane receptor. Blood. 2005;105(10):3945–3950. doi: 10.1182/blood-2004-11-4463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bellucci R, Oertelt S, Gallagher M, et al. Differential epitope mapping of antibodies to PDC-E2 in patients with hematologic malignancies after allogeneic hematopoietic stem cell transplantation and primary biliary cirrhosis. Blood. 2007;109(5):2001–2007. doi: 10.1182/blood-2006-06-030304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yang XF, Wu CJ, McLaughlin S, et al. CML66, a broadly immunogenic tumor antigen, elicits a humoral immune response associated with remission of chronic myelogenous leukemia. Proc Natl Acad Sci U S A. 2001;98(13):7492–7497. doi: 10.1073/pnas.131590998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wu CJ, Biernacki M, Kutok JL, et al. Graft-versus-leukemia target antigens in chronic myelogenous leukemia are expressed on myeloid progenitor cells. Clin Cancer Res. 2005;11(12):4504–4511. doi: 10.1158/1078-0432.CCR-05-0036. [DOI] [PubMed] [Google Scholar]
- 15.Schultze JL, Cardoso AA, Freeman GJ, et al. Follicular lymphomas can be induced to present alloantigen efficiently: a conceptual model to improve their tumor immunogenicity. Proc Natl Acad Sci U S A. 1995;92(18):8200–8204. doi: 10.1073/pnas.92.18.8200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Reche PA, Keskin DB, Hussey RE, Ancuta P, Gabuzda D, Reinherz EL. Elicitation from virus-naive individuals of cytotoxic T lymphocytes directed against conserved HIV-1 epitopes. Med Immunol. 2006;5:1. doi: 10.1186/1476-9433-5-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Armistead PM, Mohseni M, Gerwin R, et al. Erythroid-lineage-specific engraftment in patients with severe hemoglobinopathy following allogeneic hematopoietic stem cell transplantation. Exp Hematol. 2008;36(9):1205–1215. doi: 10.1016/j.exphem.2008.04.004. [DOI] [PubMed] [Google Scholar]
- 18.Wu CJ, Chillemi A, Alyea EP, et al. Reconstitution of T-cell receptor repertoire diversity following T-cell depleted allogeneic bone marrow transplantation is related to hematopoietic chimerism. Blood. 2000;95(1):352–359. [PubMed] [Google Scholar]
- 19.Nishino M, Kurasawa Y, Evans R, Lin SH, Brinkley BR, Yu-Lee LY. NudC is required for Plk1 targeting to the kinetochore and chromosome congression. Curr Biol. 2006;16(14):1414–1421. doi: 10.1016/j.cub.2006.05.052. [DOI] [PubMed] [Google Scholar]
- 20.Rammensee H, Bachmann J, Emmerich NP, Bachor OA, Stevanovic S. SYFPEITHI: database for MHC ligands and peptide motifs. Immunogenetics. 1999;50(3–4):213–219. doi: 10.1007/s002510050595. [DOI] [PubMed] [Google Scholar]
- 21.Nielsen M, Lundegaard C, Blicher T, et al. NetMHCpan, a method for quantitative predictions of peptide binding to any HLA-A and -B locus protein of known sequence. PLoS One. 2007;2(8):e796. doi: 10.1371/journal.pone.0000796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Masterson AJ, Sombroek CC, De Gruijl TD, et al. MUTZ-3, a human cell line model for the cytokine-induced differentiation of dendritic cells from CD34+ precursors. Blood. 2002;100(2):701–703. doi: 10.1182/blood.v100.2.701. [DOI] [PubMed] [Google Scholar]
- 23.Biernacki MA, Marina O, Liu FL, et al. Antigen targets of remission-inducting immune therapy are expressed on CML progenitor cells. 2009 (in revision) [Google Scholar]
- 24.Amigorena S. Fc gamma receptors and cross-presentation in dendritic cells. J Exp Med. 2002;195(1):F1–F3. doi: 10.1084/jem.20011925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Odunsi K, Qian F, Matsuzaki J, et al. Vaccination with an NY-ESO-1 peptide of HLA class I/II specificities induces integrated humoral and T cell responses in ovarian cancer. Proc Natl Acad Sci U S A. 2007;104(31):12837–12842. doi: 10.1073/pnas.0703342104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Valmori D, Souleimanian NE, Tosello V, et al. Vaccination with NY-ESO-1 protein and CpG in Montanide induces integrated antibody/Th1 responses and CD8 T cells through cross-priming. Proc Natl Acad Sci U S A. 2007;104(21):8947–8952. doi: 10.1073/pnas.0703395104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mullally A, Ritz J. Beyond HLA: the significance of genomic variation for allogeneic hematopoietic stem cell transplantation. Blood. 2007;109(4):1355–1362. doi: 10.1182/blood-2006-06-030858. [DOI] [PubMed] [Google Scholar]
- 28.Wu CJ, Ritz J. Revealing tumor immunity after hematopoietic stem cell transplantation. Clin Cancer Res. 2009;15(14):4515–4517. doi: 10.1158/1078-0432.CCR-09-0873. [DOI] [PubMed] [Google Scholar]
- 29.Beatty GL, Smith JS, Reshef R, et al. Functional unresponsiveness and replicative senescence of myeloid leukemia antigen-specific CD8+ T cells after allogeneic stem cell transplantation. Clin Cancer Res. 2009;15(15):4944–4953. doi: 10.1158/1078-0432.CCR-08-3332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gannage M, Abel M, Michallet AS, et al. Ex vivo characterization of multiepitopic tumor-specific CD8 T cells in patients with chronic myeloid leukemia: implications for vaccine development and adoptive cellular immunotherapy. J Immunol. 2005;174(12):8210–8218. doi: 10.4049/jimmunol.174.12.8210. [DOI] [PubMed] [Google Scholar]
- 31.Suemori K, Fujiwara H, Ochi T, et al. Identification of an epitope derived from CML66, a novel tumor-associated antigen expressed broadly in human leukemia, recognized by human leukocyte antigen-A*2402-restricted cytotoxic T lymphocytes. Cancer Sci. 2008;99(7):1414–1419. doi: 10.1111/j.1349-7006.2008.00823.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Suemori K, Fujiwara H, Ochi T, et al. Identification of a novel epitope derived from CML66 that is recognized by anti-leukaemia cytotoxic T lymphocytes. Br J Haematol. 2009;146(1):115–118. doi: 10.1111/j.1365-2141.2009.07695.x. [DOI] [PubMed] [Google Scholar]
- 33.Bellucci R, Alyea EP, Weller E, et al. Immunologic effects of prophylactic donor lymphocyte infusion after allogeneic marrow transplantation for multiple myeloma. Blood. 2002;99(12):4610–4617. doi: 10.1182/blood.v99.12.4610. [DOI] [PubMed] [Google Scholar]
- 34.Claret EJ, Alyea EP, Orsini E, et al. Characterization of T cell repertoire in patients with graft-versus-leukemia after donor lymphocyte infusion. J Clin Invest. 1997;100(4):855–866. doi: 10.1172/JCI119601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hochberg EP, Chillemi AC, Wu CJ, et al. Quantitation of T-cell neogenesis in vivo after allogeneic bone marrow transplantation in adults. Blood. 2001;98(4):1116–1121. doi: 10.1182/blood.v98.4.1116. [DOI] [PubMed] [Google Scholar]
- 36.Miyamura K, Tahara T, Tanimoto M, et al. Long persistent bcr-abl positive transcript detected by polymerase chain reaction after marrow transplant for chronic myelogenous leukemia without clinical relapse: a study of 64 patients. Blood. 1993;81(4):1089–1093. [PubMed] [Google Scholar]
- 37.Becker TC, Coley SM, Wherry EJ, Ahmed R. Bone marrow is a preferred site for homeostatic proliferation of memory CD8 T cells. J Immunol. 2005;174(3):1269–1273. doi: 10.4049/jimmunol.174.3.1269. [DOI] [PubMed] [Google Scholar]
- 38.Slifka MK, Antia R, Whitmire JK, Ahmed R. Humoral immunity due to long-lived plasma cells. Immunity. 1998;8(3):363–372. doi: 10.1016/s1074-7613(00)80541-5. [DOI] [PubMed] [Google Scholar]
- 39.Slifka MK, Whitmire JK, Ahmed R. Bone marrow contains virus-specific cytotoxic T lymphocytes. Blood. 1997;90(5):2103–2108. [PubMed] [Google Scholar]
- 40.Feuerer M, Beckhove P, Garbi N, et al. Bone marrow as a priming site for T-cell responses to blood-borne antigen. Nat Med. 2003;9(9):1151–1157. doi: 10.1038/nm914. [DOI] [PubMed] [Google Scholar]
- 41.Berger C, Jensen MC, Lansdorp PM, Gough M, Elliott C, Riddell SR. Adoptive transfer of effector CD8+ T cells derived from central memory cells establishes persistent T cell memory in primates. J Clin Invest. 2008;118(1):294–305. doi: 10.1172/JCI32103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Choi C, Witzens M, Bucur M, et al. Enrichment of functional CD8 memory T cells specific for MUC1 in bone marrow of patients with multiple myeloma. Blood. 2005;105(5):2132–2134. doi: 10.1182/blood-2004-01-0366. [DOI] [PubMed] [Google Scholar]
- 43.Letsch A, Keilholz U, Assfalg G, Mailander V, Thiel E, Scheibenbogen C. Bone marrow contains melanoma-reactive CD8+ effector T cells and, compared with peripheral blood, enriched numbers of melanoma-reactive CD8+ memory T cells. Cancer Res. 2003;63(17):5582–5586. [PubMed] [Google Scholar]
- 44.Letsch A, Knoedler M, Na IK, et al. CMV-specific central memory T cells reside in bone marrow. Eur J Immunol. 2007;37(11):3063–3068. doi: 10.1002/eji.200636930. [DOI] [PubMed] [Google Scholar]
- 45.Wherry EJ, Blattman JN, Murali-Krishna K, van der Most R, Ahmed R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J Virol. 2003;77(8):4911–4927. doi: 10.1128/JVI.77.8.4911-4927.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.