Abstract
LIGHT, a ligand for the lymphotoxin-beta receptor, establishes lymphoid-like tissues inside tumor sites and recruits naïve T-cells into the tumor. However, whether these infiltrating T-cells are specific for tumor antigens is not known. We hypothesized that therapy with LIGHT can expand functional tumor-specific CD8+ T-cells that can be boosted using HPV16E6E7-Venezuelan Equine Encephalitis Virus Replicon Particles (HPV16-VRP) and that this combined therapy can eradicate HPV16-induced tumors. Our data show that forced expression of LIGHT in tumors results in an increase in expression of interferon gamma (IFNg) and chemottractant cytokines such as IL-1a, MIG and MIP-2 within the tumor and that this tumor microenvironment correlates with an increase in frequency of tumor-infiltrating CD8+ T-cells. Forced expression of LIGHT also results in the expansion of functional T-cells that recognize multiple tumor-antigens, including HPV16 E7, and these T-cells prevent the outgrowth of tumors upon secondary challenge. Subsequent boosting of E7-specific T-cells by vaccination with HPV16-VRP significantly increases their frequency in both the periphery and the tumor, and leads to the eradication of large well-established tumors, for which either treatment alone is not successful. These data establish the safety of Ad-LIGHT as a therapeutic intervention in pre-clinical studies and suggest that patients with HPV16+ tumors may benefit from combined immunotherapy with LIGHT and antigen-specific vaccination.
Keywords: Cervical Cancer, Immunotherapy, Human papillomavirus, HPV16, LIGHT, TNFSF14, Adenovirus, VEE Virus Replicon Particles, VRP, T-cells
Introduction
The importance of the cellular immune system in controlling the pathogenesis of HPV and associated cervical lesions in humans is very well-established (1-7). In the case of HPV16-induced cancers, the most frequently targeted antigens are the E6 and E7 proteins, because they are oncogenic and sustained expression is required for the maintenance of the cancerous phenotype. This blatant expression of foreign antigens in a tumor makes HPV a very attractive target for “proof of concept” studies in the development of therapeutic vaccines. Several therapeutic vaccines that target the E6 and/or E7 proteins have been developed over the last fifteen years (reviewed in (8)). These include regimens based on peptides, proteins, plasmids and viral vectors, each having its strengths and limitations. Of the viral vector based approaches, Venezuelan Equine Encephalitis (VEE) alphavirus replicon particles (VRP) (9) are very promising. VRP encoding several viral genes have been shown to be immunogenic and protective (10-14). Previous studies have tested the anti-tumor efficacy of VRP containing the HPV16 E7 gene and shown protective responses as well as some therapeutic efficacy for early stage tumors (15). However, whether HPV16-VRP vaccination can induce regression of large tumors has not been evaluated.
Increased frequency of tumor-specific lymphocytes after vaccination argues that vaccine interventions are doing their job, which is to induce tumor-specific T-cells. However, the induced T-cells are frequently ineffective in controlling tumor growth, likely due to lack of an inflammatory environment in the tumor. Altering the tumor to create a pro-inflammatory environment in situ that can sustain T-cell activation may lead to regression of large, well-established tumors.
Lymphotoxin-β receptor (LTβR) signaling plays an important role in the formation of lymphoid structures (16-19) and regulates the expression of chemokines and adhesion molecules, which control the migration and positioning of DC and lymphocytes in the spleen (18). TNFSF14/LIGHT, a ligand for LTβR and herpes virus entry mediator (HVEM), is predominantly expressed on activated immune cells, especially on the surface of activated DCs and T-cells (16, 20-23). Expression of LIGHT restores lymphoid structures in lymphotoxin β knockout mice, establishes lymphoid-like tissues inside tumor sites via its interactions with LTβR on stromal cells, induces intra-tumoral production of chemokines such as CCL21 and recruits naïve T-cells into the tumor (16, 22-24). It coordinately induces activation and expansion of incoming T-cells through HVEM, thereby functioning as a co-stimulatory molecule and generating stronger anti-tumor immunity (20, 21, 23, 24). While enhanced T-cell infiltration is observed in LIGHT-expressing tumors, their specificity is unknown.
The C3.43 cell line is an aggressive derivative of the C3 line, which was transformed using a pRSVneo-derived plasmid containing the HPV16 genome (25). The cell line expresses an H-2Db-bound peptide - E7(49-57) (RAHYNIVTF) and CTL that recognize E7(49-57) are capable of eradicating HPV16-transformed cells in vitro and in vivo (25, 26). The ampitope (SSPVNSLRNVV)–peptide is also a highly immunogenic H-2Db restricted epitope that was encoded by a cryptic ORF in the antisense strand of the ampicillin resistance gene of the plasmid backbone and vaccination with the ampitope peptide protects against challenge with C3-derived cell lines (27). Therefore, the ampitope peptide also functions as a tumor-specific antigen in this model. In the present study, we examined whether therapy with either HPV16-VRP or forced expression of LIGHT in large well-established tumors HPV16+ tumors could induce regression and whether combined therapy had superior efficacy in inducing anti-tumor immunity that could eradicate tumors.
Materials and Methods
Mice and Cell Lines
Specific pathogen-free female C57Bl/6 mice 6-8 weeks of age were purchased from Taconic Farms (Germantown, NY). Tumor challenge studies were performed using the C3.43 cell line, an in vivo-passaged derivative of the C3 HPV16+ transformed murine tumor cell line (25). C3.43 cells were clonally expanded in vitro from a progressively growing C3 tumor in vivo and low passage number working vials were frozen as seed stocks. C3.43 cells retained expression of the HVP16 E6 and E7 (by RT-PCR and Western blot), expressed similar levels of MHC class I molecules on the surface compared to the parental C3 line (by flow cytometry), and responded to prophylactic vaccination with various HPV16 E7-containing vaccines in vivo. C3.43 cells tested negative for mycoplasma contamination (MycoAlert Mycoplasma Detection Kit, Cambrex Inc). Cells used for tumor challenge were cultured no longer than two weeks from original seed stocks prior to in vivo injection. C3.43 cells were cultured in IMDM media supplemented with 10%FBS. All procedures were performed in accordance with institutional guidelines and approved by the USC Institutional Animal Care and Use Committee (IACUC).
Peptides
The HPV16 E7(49-57) RAHYNIVTF peptide (25), the SSPVNSLRNVV Ampitope peptide (aa 49-59) (27) and the AQMNNRDCL peptide from prostate stem cell antigen (PSCA(23-31)) (28) were synthesized at the University of Chicago (Chicago, IL) and purified by reverse phase HPLC. Purity was assessed by analytical HPLC and was determined to be >90% pure.
Tumor Challenge and Treatment
Groups of fifteen female 8-week old C57BL/6 mice were challenged subcutaneously in the right flank with 5×105 C3.43 tumor cells in PBS. Throughout the duration of the experiments, tumor growth was monitored 2-3 times per week with a caliper in three dimensions. Mice were euthanized per USC IACUC guidelines when tumor volume exceeded 1500 mm3 or when ulcerated. In experiments examining efficacy of therapeutic vaccination with HPV16-VRP (29), mice were vaccinated with 107 infectious units of VRP subcutaneously in 50μl of PBS on days 14 and 21 after tumor challenge, as boosting after 1 week is optimal for generating immune responses. In studies examining the effect of LIGHT, recombinant adenovirus carrying DNA encoding for the LIGHT gene (Ad-LIGHT) were injected intra-tumorally at 1010 virus particles per mouse on day 14 and the 2nd injection was done after an interval of 72h, which has been previously (30) optimized to prevent adenovirus-specific immune responses from neutralizing the delivery vector. Particles carrying either LacZ or green fluorescent protein (GFP) were used as controls (Ad-CTRL or VRP-CTRL). For combined therapy, mice were treated with adenovirus particles as described and vaccinated with 107 infectious units of either HPV16-VRP or GFP-VRP on days 24 and 31. No treatment-related toxicities were noted.
Tumor Resection and Re-challenge
Survival surgeries were performed under the guidance of an experienced veterinarian according to protocols approved by USC IACUC. Briefly, mice were anesthetized and tumors were surgically excised under sterile conditions. Skin flaps were closed using Nexabond tissue adhesive followed by suturing. Mice were re-challenged with 5×105 C3.43 tumor cells in the left flank and monitored until revival. Tumor growth and survival was monitored 2 times per week. Mice were euthanized per USC IACUC guidelines.
Isolating tumor-infiltrating lymphocytes (TILS)
TILS were isolated as described previously (28, 31). Tumor tissues were cut into small pieces and digested in media supplemented with 0.25% w/v dispase for 2 hours with continuous stirring at 37°C. The tumor digest was passed through a nylon mesh to obtain a single cell suspension, washed with PBS and analyzed by flow cytometry to determine the frequency of E7-specific T-cells in the tumors.
Flow Cytometry and Tetramer Staining
H-2Db tetramers containing the HPV16 E7(49–57) peptide were obtained from the National Institute of Allergy and Infectious Diseases Tetramer Facility (Atlanta, GA). Single cell suspension of tumors, which contain tumor-infiltrating lymphocytes (TILs), was obtained as described. CD8+ T-cells were isolated from individual spleens and pooled lymph nodes using MACS cell sorting (Miltenyi). One million unsorted tumor cells or CD8-enriched splenocytes or CD8-enriched lymphocytes were incubated for 1 h at 4°C with 0.5μg/ml tetramer, 1:100 diluted anti-CD3 antibody and 1:100 diluted anti-CD8 antibody (BD PharMingen). Cells were washed twice and fixed in 2% paraformaldehyde. At least 100,000 cells were acquired on the Beckman Coulter FC 500 flow cytometer and analyzed using the CXP software. Gating was done on CD8+ cells and percentage of CD3+/CD8+/tetramer+ cells was determined.
ELISPOT Assay
Functional tumor antigen-specific T-cells specific for HPV16 E7(49–57) and the Ampitope peptides were detected. The Db-binding PSCA(23-31) peptide was used to set background. Multiscreen HA plates (Millipore) were coated with 10μg/ml anti-IFNg antibody (BD PharMingen). Plates were washed and blocked with culture medium. Splenocytes were added in 2-fold serial dilutions ranging from 5×105 to 6.25×104 cells per well in medium containing 5 international units interleukin (IL)-2 and 10μg/ml peptide. After 40-h, plates were washed and incubated with 1μg/ml biotinylated anti-IFNg antibody. Washed plates were incubated with 100μl of 1/4000 diluted streptavidin-horseradish peroxidase (HRP, Sigma Chemical Co.) per well. Spots were developed using 3-amino-9-ethyl-carbazole (Promega) for 5 min and the reaction was stopped with water. Spots were counted using the Zeiss KS ELISPOT system. Assays were performed in triplicate and results were calculated as spot-forming cells per 106 splenocytes, after subtracting background.
Measuring Intra-tumoral Cytokines
Tumors were collected, weighed, and homogenized in PBS containing 2× Halt Protease Inhibitor Cocktail (Pierce). Supernatants were collected by centrifugation at 4°C. Cytokine and chemokine levels were quantified using the Milliplex Mouse Cytokine/Chemokine - Premixed 32 Plex (Millipore) and a Bio-Plex Suspension Array system following manufacturer's instructions. TGFb levels were quantified separately using a Milliplex TGFb kit according to the manufacturer's instructions.
Statistical Analysis
Each figure is representative of at least two independent experiments. Tumor growth, multiplex ELISA, ELISPOT and flow cytometry data were analyzed by either a two-tailed Student's t test or a one-way analysis of variance (ANOVA) when more than 2 groups were compared. Survival was analyzed by the log-rank test. All statistical analyses were performed using the GraphPad Prism™ version 4.0 software.
Results
Therapy with HPV16-VRP does not induce regression of well-established C3.43 tumors
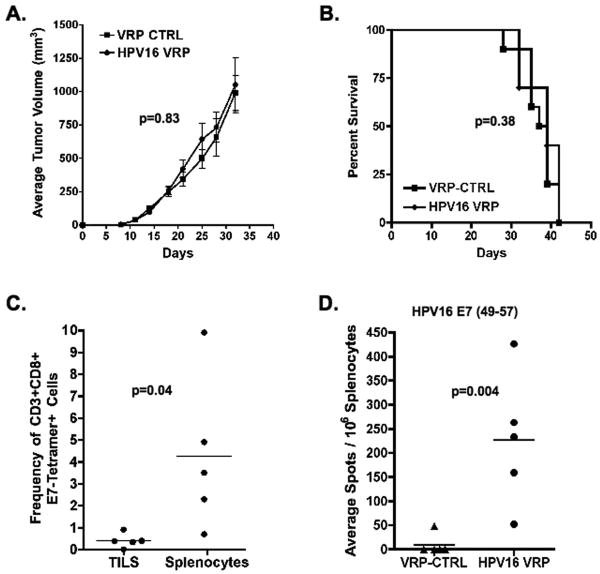
To determine whether therapy of well-established HPV16+ tumors with HPV16-VRP could induce tumor regression, two groups of C57BL/6 mice were challenged and tumors were allowed to grow for 14 days, until average tumor volume was greater than 100mm3, and vaccinated with HPV16-VRP. When compared to control vaccinated mice, mice that were vaccinated with HPV16-VRP did not show statistically significant differences in either average tumor volume (p=0.83, two-tailed T test, Fig. 1A) or in survival (p=0.38, Fig. 1B). To ensure that lack of efficacy of HPV16-VRP vaccination on survival was not due to a failure in inducing functional E7-specific T-cells, the frequency and function of E7-specific CD8+ T-cells were analyzed one week after final vaccination. Figure 1C shows that HPV16-VRP vaccination resulted in the expansion of E7-specific T-cells in the spleens of tumor-bearing mice, but not in the tumors of the same mice (p=0.04, two-tailed T test). Figure 1D shows that the E7-specific T-cells expanded in the spleen upon vaccination with HPV16-VRP were able to secrete interferon gamma upon stimulation with the E7(49-57) peptide (p=0.004, two-tailed T test). Additionally, the cells were able to secrete both IL-2 and TNFa and proliferate in response to peptide stimulation (data not shown). Together, these data show that HPV16-VRP vaccination does not induce regression of large HPV16+ tumors because tumor-specific T cells are absent in the tumor despite the induction of strong functional immunity in the peripheral lymphoid tissue.
Figure 1. Therapy of well-established C3.43 tumors with HPV16-VRP alone results in an expansion of HPV16 E7-specific T-cells but does not induce significant tumor regression.
Tumor-bearing mice were treated with either VRP-CTRL (GFP-VRP) or HPV16-VRP. (A & B) Average tumor volumes ± SEM and survival are plotted against days after tumor challenge (n=10 per group). (C) Lymphocytes isolated from individual spleens (n=5 per group) or tumors of HPV16-VRP vaccinated mice were analyzed by FACS and the average frequency of CD8+/CD3+/E7-tetramer+ cells is shown. (D) IFN-gamma ELISPOT assay was used to detect functional tumor-specific T-cells after stimulation with the HPV16 E7(49-57) peptide (n=5 per group).
Ad-LIGHT treatment leads to the expansion of E7-specific T-cells and increases T-cell infiltration into tumors
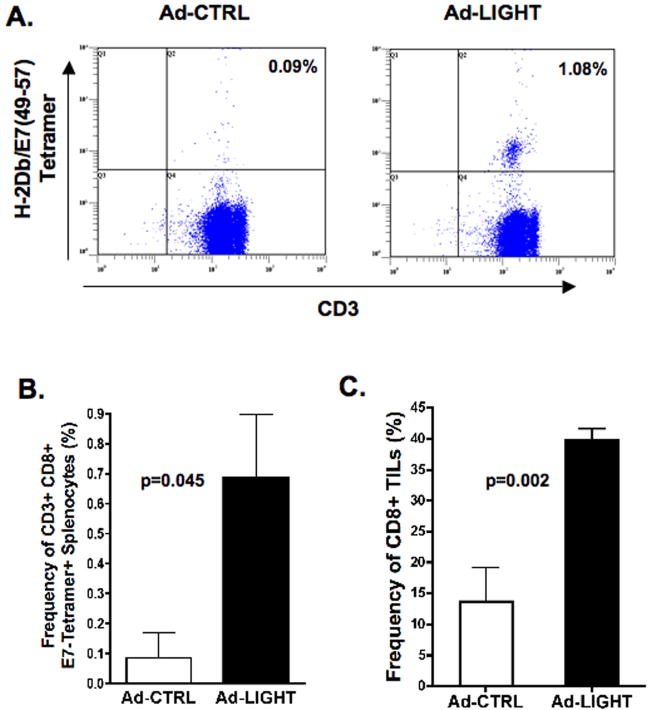
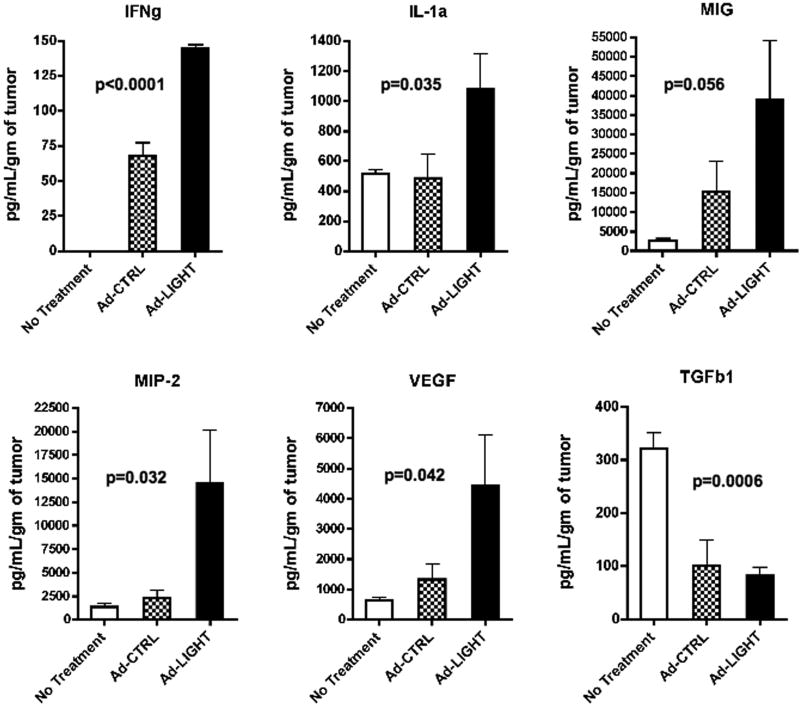
We hypothesized that altering the tumor microenvironment by over-expressing LIGHT may induce functional tumor-specific T-cells that control tumor growth and increase the survival of tumor-bearing mice. To test this hypothesis, mice were challenged with tumor and treated with intra-tumoral injections of either Ad-CTRL or Ad-LIGHT. To determine whether expression of LIGHT induced the expansion of HPV16+ tumor specific T-cells, individual spleens and pooled lymph nodes from mice were assessed for frequency of E7-specific CD8+ T-cells one week after final treatment. Figure 2A shows that the average frequency of CD3+CD8+tetramer+ T-cells in the draining inguinal lymph nodes from Ad-CTRL treated mice was 0.09%, which is similar to naïve mice, and that treatment with Ad-LIGHT increased the average frequency of CD3+CD8+tetramer+ T-cells to 1.08%. Data comparing the frequency of CD3+CD8+tetramer+ T-cells in individual spleens from groups of five mice treated with either Ad-CTRL or Ad-LIGHT shows that intra-tumoral injection of Ad-LIGHT significantly increased the frequency of E7-specific T-cells ranging from 0.31% to 1.49% (p=0.045, two-tailed T test, Fig. 2B). To determine whether intra-tumoral injection of Ad-LIGHT altered the tumor microenvironment to increase infiltration of CD8+ T-cells into tumors, individual tumors were analyzed for CD8+ T-cells by flow cytometry and for cytokine/chemokine expression by multiplex ELISA. Our data show that frequency of CD8+ TILs increases significantly in Ad-LIGHT treated tumors as compared with control treated tumors (p=0.002, two-tailed T test, Fig. 2C). As shown in figure 3, multiplex ELISA analysis showed a statistically significant increase in the intra-tumoral levels of IFNg, IL-1a, macrophage inflammatory protein-2 (MIP-2) and vascular endothelial growth factor (VEGF) as well a trend towards an increase in monokine induced by gamma interferon (MIG/CXCL9). Similar trends were seen for IP-10 and GM-CSF. Additionally, there was an increase in RANTES, TNFa, G-CSF, IL-1b, IL-2, IL-6, IL-9, IL-10, IL-12p70, IL-13, IL-15, KC and LIX in the treated groups as compared with no treatment (data not shown). However, there were no statistically significant differences between control-treated and Ad-LIGHT treated mice. No differences in the intra-tumoral levels of IL-3, IL-4, IL-5, IL-7, IL-12p40, IL-17, LIF, MCP-1 and M-CSF were observed between any of the groups (data not shown). Finally, treatment with Ad-CTRL resulted in a decrease in intra-tumoral concentration of the immunosuppressive cytokine, TGFb1, which was further decreased upon expression of LIGHT. Together, these results show that intra-tumoral injection of Ad-LIGHT in tumors increases the concentration of IFNg and chemo-attractant cytokines while reducing the concentration of immunosuppressive TGFb1, which may lead to the observed increase in T-cell infiltration into tumors and the expansion of E7-specific T-cells.
Figure 2. Forced expression of LIGHT induces the expansion of HPV16 E7-specific T-cells and significantly increases T-cell infiltration into C3.43 tumors.
Individual spleens and draining lymph nodes were harvested 7 days after last treatment from tumor-bearing mice treated with either Ad-LIGHT or Ad-CTRL (n=5 per group). CD8+ T-cells were isolated by MACS sorting and incubated with HPV16 E7(49–57)/H-2Db tetramer, anti-CD3 and anti-CD8 antibodies. Cells were analyzed by FACS and gating was done on CD8+ cells. (A) Upper right-hand quadrant shows frequency of CD8+/CD3+/E7-tetramer+ cells in pooled lymphocytes. (B) Average frequency of CD8+/CD3+/E7-tetramer+ cells in splenocytes. (C) TILS were isolated from tumors, stained with anti-CD3 and anti-CD8 antibodies. Average frequency of CD3+CD8+ cells is shown.
Figure 3. Forced expression of LIGHT in C3.43 tumors induces increased expression of IFNg and chemokines.
Three groups of 5 tumor-bearing mice were left either untreated or treated with Ad-CTRL or treated with Ad-LIGHT. On day 24, tumors were resected and homogenized. Intra-tumoral cytokines and chemokines were measured. Cytokine/chemokine concentration per gram of tumor ± SD is shown.
LIGHT expression in HPV16+ tumors leads to increased survival
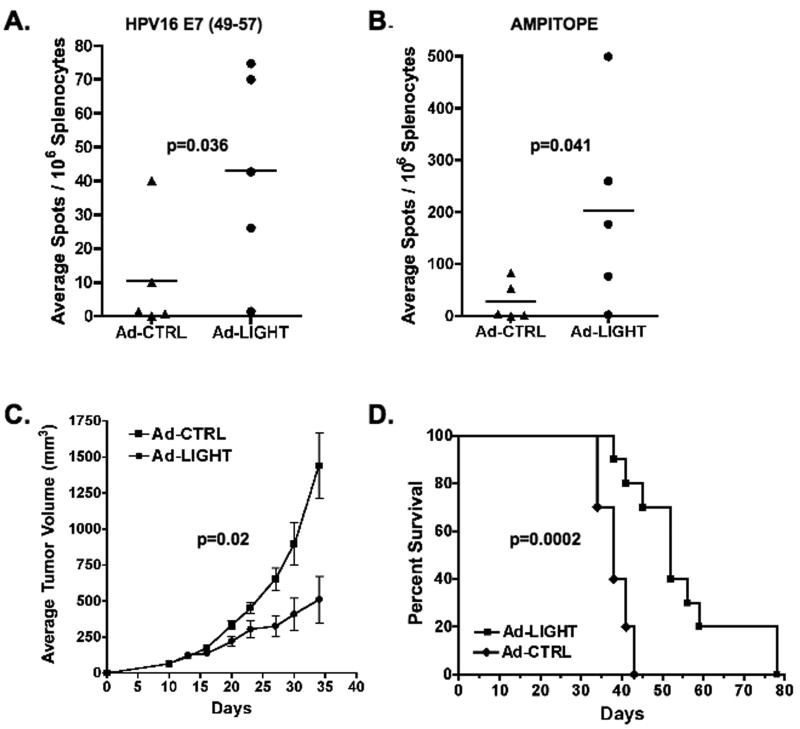
Since treatment with Ad-LIGHT induced the expansion of E7-specific T-cells, we sought to determine whether these T-cells were functional. The number of splenic T-cells secreting IFNg in response to peptide stimulation was measured by ELISPOT one week after final treatment. Our data show that T-cells from Ad-LIGHT treated mice were able to secrete IFNg upon stimulation with the E7(49-57) peptide whereas those treated with the control did not (p=0.036, two-tailed T test, Fig. 4A). To assess whether therapy with Ad-LIGHT could induce functional T-cells to multiple tumor antigens, we also assessed the T-cell response to the ampitope (SSPVNSLRNVV) peptide (27). Our data show that T-cells from mice treated with Ad-LIGHT were also able to specifically secrete IFNg when stimulated with the ampitope peptide, whereas control-treated mice did not (p=0.041, two-tailed T test, Fig. 4B). We next sought to determine whether the functional tumor-specific T-cells expanded by intra-tumoral expression of LIGHT could induce tumor regression. Our data show that Ad-LIGHT therapy partially controlled tumor growth (p=0.02, two-tailed T test, Fig. 4C) and resulted in a statistically significant increase in the duration of survival of tumor-bearing mice as compared to mice treated with control particles (p=0.0002, log-rank test, Fig. 4D). Together, these data show that functional tumor-specific T-cells induced by treatment with Ad-LIGHT partially controlled growth of HPV16+ C3.43 tumor but did not induce complete tumor regression.
Figure 4. Forced expression of LIGHT in C3.43 tumors induces functional T-cells that are specific for multiple tumor antigens and increases survival of tumor-bearing mice.
Groups of 5 tumor-bearing mice treated with either Ad-LIGHT or Ad-CTRL. IFN-gamma ELISPOT assay was used to detect functional peptide-specific T-cells in individual spleens. (A) HPV16 E7(49-57) (B) Ampitope (SSPVNSLRNVV) peptide (C & D) Tumor growth was monitored in groups of 10 tumor-bearing mice treated with either Ad-LIGHT or Ad-CTRL. Average tumor volumes ± SEM and survival are plotted against days after tumor challenge.
LIGHT delivered by adenovirus to primary tumors prevents outgrowth of distal tumors upon re-challenge
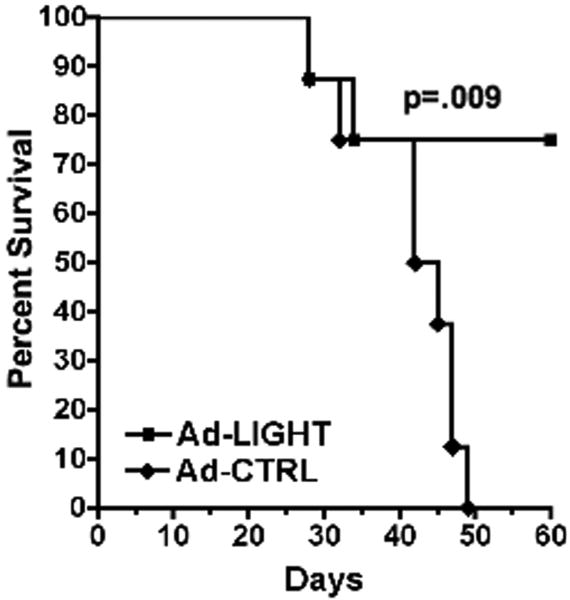
Intra-tumoral expression of LIGHT may not lead to tumor regression due to tumor growth outpacing the immune response or due to the bulk of the tumor at the time of intervention. To determine whether tumor-specific T-cells induced by LIGHT could prevent the outgrowth of smaller tumors, mice were first challenged with tumor and treated with either Ad-LIGHT or Ad-CTRL. One week after final treatment, primary tumors were surgically resected from the right flank and mice were re-challenged on the left flank. Primary tumors were resected because the fast growth rate of well-established C3.43 tumors in the Ad-CTRL treated group would require that mice be sacrificed prior to completion of the evaluation of whether secondary tumors were rejected. Only the 80% of mice that survived surgery and did not have recurring tumor at the primary site were included in the analysis. We observed that by day 50 after tumor re-challenge, 0% of mice in the control-treated group (n=8, analyzed) survived as compared with 75% of mice in the Ad-LIGHT treated group (n=8, analyzed) (p=0.009, log-rank test, Fig. 5). Additionally, we found that 5 out of 8 mice in the Ad-LIGHT treated group were tumor-free, showing that the functional tumor-specific T-cells expanded by intra-tumoral expression of LIGHT could prevent outgrowth of tumors.
Figure 5. Forced expression of LIGHT in primary tumors prevents outgrowth of tumors upon secondary challenge.
Groups of 10 tumor-bearing mice were treated with either Ad-LIGHT or Ad-CTRL. On day 24 after challenge, primary tumors were resected from the right flank and mice were given a secondary tumor challenge on the left flank. Percent survival is plotted against days after secondary tumor challenge (n=8 analyzed per group).
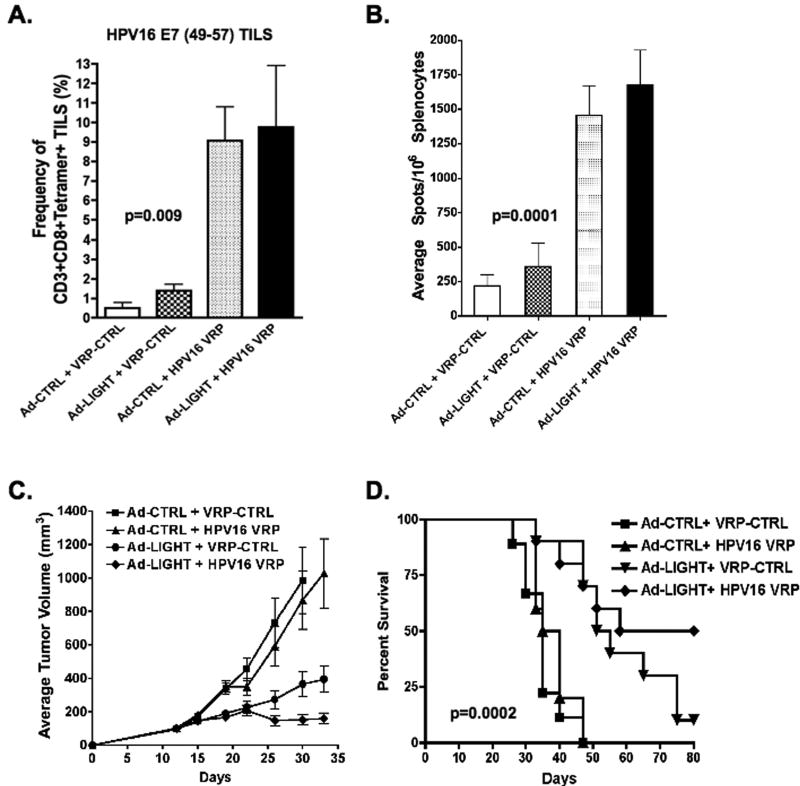
Combined treatment with Ad-LIGHT followed by vaccination with HPV16-VRP induces regression of large HPV16+ tumors
Since treatment of 14-day old tumor with either HPV16-VRP alone or with Ad-LIGHT alone did not result in tumor regression despite induction of tumor-specific immunity, we hypothesized that inducing E7-specific immunity via intra-tumoral injection of Ad-LIGHT and boosting with HPV16-VRP would increase the frequency of E7-specific T-cells and result in regression. To test this hypothesis, tumor-bearing mice were treated with Ad-LIGHT and subsequently vaccinated with HPV16-VRP. FACS analysis of tumors and spleens with E7(49–57) tetramers showed that mice treated with either Ad-LIGHT alone or HPV16-VRP alone had increased frequencies of tumor-infiltrating E7-specific T-cells as compared to control-treated mice and that combined treatment with both Ad-LIGHT and HPV16-VRP further increased the frequency of E7-specific T-cells in the tumors (p=0.009, one-way ANOVA, Fig. 6A). IFNg ELISPOT analysis revealed that mice treated with both Ad-LIGHT and HPV16-VRP had stronger functional tumor-specific T-cell responses as compared to mice treated with Ad-LIGHT (p=0.0001, one-way ANOVA, Fig. 6B), showing that Ad-LIGHT treatment followed by vaccination with HPV16-VRP increases the frequency of functional tumor-specific T cells. Figure 6C shows that treatment with HPV16-VRP alone did not impact tumor growth whereas treatment with Ad-LIGHT resulted in partial control of tumor growth. Most importantly, the data show that combined treatment with Ad-LIGHT and HPV16-VRP reduced the average tumor volume as compared to treatment with Ad-LIGHT alone (p=0.09, one-way ANOVA, Fig. 6C). Furthermore, the data show that control-treated mice or mice treated with HPV16-VRP alone died by day 47 whereas 70% of mice treated with Ad-LIGHT and 70% of mice treated with both Ad-LIGHT and HPV16-VRP survived. By day 80, only 10% of mice treated with Ad-LIGHT survived whereas 50% of mice treated with both Ad-LIGHT and HPV16-VRP were tumor-free (p=0.0002, log-rank test, Fig. 6D). Together, these data support our hypothesis that intra-tumoral expression of Ad-LIGHT alters the tumor microenvironment by increasing the expression of proinflammatory and chemotactic cytokines/chemokines and that boosting LIGHT-induced E7-specific T cells with HPV16-VRP would increase the frequency of tumor-specific T-cells, which leads to the regression of large, well-established HPV16+ tumors.
Figure 6. Vaccination with HPV16-VRP boosts the frequency of tumor-specific T-cells induced by intra-tumoral expression of LIGHT and leads to significant survival.
Tumor-bearing mice treated with either Ad-CTRL (Ad-LacZ) or Ad-LIGHT followed by treatment with either VRP-CTRL (GFP-VRP) or HPV16-VRP. (A) TILS were analyzed by FACS and the average frequency of CD8+/CD3+/E7-tetramer+ TILS is shown (n=5 per group). (B) IFN-gamma ELISPOT assay was used to detect functional tumor-specific T-cells from individual spleens after stimulation with both the Ampitope and E7(49-57) peptides (n=5 per group). (C & D) Average tumor volumes ± SEM and survival are plotted against days after tumor challenge (n=10 per group).
Discussion
VRP are excellent immunogens with numerous advantages over other vaccination platforms. Previously, our lab has shown that HPV16-VRP vaccination is protective and partially therapeutic when administered to treat small palpable tumors (15). In this study, we tested whether HPV16-VRP vaccination could eradicate large tumors that mimic more advanced stage of disease and show that treatment with HPV16-VRP alone does not induce regression. Our data show that HPV16-VRP vaccination can induce high frequencies of functional E7-specific T-cells in the peripheral lymphoid organs of tumor-bearing hosts, however, the CD8+ T cells are absent within the tumors themselves. In essence, these data recapitulate observations from the multitude of clinical trials in patients with various stages of HPV16+ lesions that show an induction of HPV-specific immunity, but have not been successful in terms of inducing objective clinical responses. Thus, inducing inflammation within the tumor may be critical for successful immunotherapy of HPV16+ lesions such as cervical cancer.
LIGHT/TNFSF14 is a molecule that promotes a variety of pro-inflammatory processes through its interaction with LTβR on stromal cells and HVEM on activated immune cells. In this study, we tested whether expression of LIGHT, delivered using adenoviruses, could lead to the expansion of tumor antigen-specific T-cells and overcome tumor-mediated immune suppression by altering the tumor microenvironment. To do so, we used the C3.43 tumor challenge model in which tumor cells express HPV16 E7, a clinically relevant antigen, under its natural promoter. We first showed that using adenoviruses to deliver LIGHT to HPV16+ tumors resulted in an increased frequency of E7(49-57)-specific T-cells in both the periphery and within the tumor. Treatment with Ad-LIGHT also resulted increased pro-inflammatory Th1 cytokines/chemokines that recruit T-cells, NK cells, neutrophils and macrophages. Of these, we found a statistically significant increase in the concentrations of IFNg, IL-1a, MIP-2 and MIG that was dependent upon the expression of LIGHT. For other Th1 cytokines, expression seemed to increase upon treatment with either Ad-CTRL or Ad-LIGHT, which suggests an adjuvant-like effect of adenovirus-mediated delivery. For molecules such as IL-15, IP-10 and MIP-1a, there was a large increase and for IL-10, there was a large decrease in concentration upon treatment with Ad-LIGHT as compared with Ad-CTRL. However, the difference was not statistically significant. Additionally, there was a decrease in TGFb1, an immunosuppressive cytokine that inhibits secretion and activity of proinflammatory cytokines such as IFNg and TNFa. Previous studies have shown that LIGHT-LTβR signaling induces CCL21 production that co-localizes T-cells and dendritic cells (32). CCL21 may induce IFNg expression and IFNg, which is also induced by LIGHT-mediated signaling, is necessary for anti-tumor immunity. MIG and IP-10 are induced by IFNg, have anti-tumor properties and are known to be necessary for IFNg-mediated anti-tumor responses (33, 34). Our novel finding that the intra-tumoral expression of LIGHT results in increased expression of MIG and IP-10 fits with the role of IFNg and IFNg-induced chemokines in anti-tumor immunity. Remarkably, we found no increase in the expression of Th2 cytokines. Another interesting observation was the statistically significant increase in VEGF expression. A previous study has shown that VEGF can increase expression of LIGHT on macrophages (35), however, whether LIGHT can increase the expression of VEGF through the ligation of LTβR on endothelial cells has not been established thus far. The mechanism of LIGHT-mediated VEGF expression and its biological relevance remain to be understood.
In addition to the E7(49-57) peptide, C3.43 cells express the ampitope peptide, which functions as the dominant tumor antigen (27). Our data show that intra-tumoral expression of LIGHT leads to the induction of functional T-cell responses to both antigens. More importantly, immune responses strongly correlated with control of tumor growth, though it did not result in tumor clearance. Thus, our results show that adenovirus mediated delivery of LIGHT can induce the expansion of functional T-cells specific to both dominant and sub-dominant tumor antigens, which provides a significant clinical advantage over antigen-specific therapies because T-cells specific for the dominant antigens may become tolerized by the tumor (36). Our data showing that LIGHT-induced immunity can significantly prevent the development of tumors upon re-challenge at a distal site is in agreement with the study by Yu et al, data from which supports the notion that expression of LIGHT delivered using adenoviruses results in the prevention or clearance of metastasis (30). However, our study also suggests that the bulk of tumor and pace of tumor growth are important factors in the efficacy of therapeutic intervention with Ad-LIGHT. To evaluate whether improving the kinetics of the immune response could enhance the efficacy of treatment with LIGHT, we boosted the frequency of tumor-antigen specific T-cells by HPV16-VRP vaccination and found a significant increase in functional E7-specific T-cells after combined therapy as compared to treatment with LIGHT alone. More importantly, combined treatment resulted in a significant increase in tumor-free survival. We also observed comparable frequency of E7-specific T-cells infiltrating into the tumors upon treatment with Ad-CTRL/HPV16-VRP, which could be a result of the increase in Th1 type cytokines in the tumors, but this did not have any impact on survival. This could be the result of treatment with HPV16-VRP beginning on day 24 in the Ad-CTRL/HPV16-VRP group, by which time tumor volume is significantly smaller in the LIGHT-treated groups. However, data in figures 2 and 4 shows that treatment with Ad-CTRL alone does not yield functional immunity in either the periphery or within the tumor and does not impact tumor growth. This suggests that while kinetics of the immune response is important, LIGHT-mediated alteration of the tumor microenvironment plays a significant role in activating tumor-specific T-cell immunity that is critical for achieving tumor-free survival.
In summary, our study provides the first evidence of LIGHT mediated induction of T-cells specific for a clinically relevant oncogenic protein both peripherally and within the tumor, the frequency of which can be further increased by vaccination. Moreover, immunity is induced against both dominant and subdominant antigens and results in the rejection of tumors upon secondary challenge. Combined treatment of intra-tumoral injection of Ad-LIGHT and vaccination with HPV16 VRP has significantly greater efficacy in inducing tumor-specific immunity that leads to the eradiation of large well-established HPV16+ tumors than either treatment alone. Therefore, this type of combination therapy may be of significant advantage to enhance tumor-specific immunity.
Acknowledgments
Footnotes: These studies were supported by the NIH P01 CA97296 and R01 CA74397 grants to W.M.K. S.K. is the recipient of a fellowship from the Achievement Rewards for College Scientists (ARCS) Foundation, L.B. is a Fulbright Research Scholar supported by a grant from the National Research Foundation – Flanders (FWO) and W.M.K. holds the Walter A. Richter Cancer Research Chair.
References
- 1.Nasiell K, Roger V, Nasiell M. Behavior of mild cervical dysplasia during long-term follow-up. Obstet Gynecol. 1986;67:665–9. doi: 10.1097/00006250-198605000-00012. [DOI] [PubMed] [Google Scholar]
- 2.Benton C, Shyahidullah H, Hunter JAM. Human papillomavirus in the immunocompromised. Papillomavirus Rep. 1999;3:23–6. [Google Scholar]
- 3.Laga M, Icenogle JP, Marsella R, Manoka AT, Nzila N, Ryder RW, et al. Genital papillomavirus infection and cervical dysplasia--opportunistic complications of HIV infection. IntJCancer. 1992;50:45–8. doi: 10.1002/ijc.2910500110. [DOI] [PubMed] [Google Scholar]
- 4.Maiman M, Fruchter RG, Serur E, Remy JC, Feuer G, Boyce J. Human immunodeficiency virus infection and cervical neoplasia. GynecolOncol. 1990;38:377–82. doi: 10.1016/0090-8258(90)90077-x. [DOI] [PubMed] [Google Scholar]
- 5.Rudlinger R, Smith IW, Bunney MH, Hunter JA. Human papillomavirus infections in a group of renal transplant recipients. BrJDermatol. 1986;115:681–92. doi: 10.1111/j.1365-2133.1986.tb06649.x. [DOI] [PubMed] [Google Scholar]
- 6.Coleman N, Birley HD, Renton AM, Hanna NF, Ryait BK, Byrne M, et al. Immunological events in regressing genital warts. AmJClinPathol. 1994;102:768–74. doi: 10.1093/ajcp/102.6.768. [DOI] [PubMed] [Google Scholar]
- 7.Rogozinski TT, Jablonska S, Jarzabek-Chorzelska M. Role of cell-mediated immunity in spontaneous regression of plane warts. IntJDermatol. 1988;27:322–6. doi: 10.1111/j.1365-4362.1988.tb02362.x. [DOI] [PubMed] [Google Scholar]
- 8.Kanodia S, Da Silva DM, Kast WM. Recent advances in strategies for immunotherapy of human papillomavirus-induced lesions. Int J Cancer. 2008;122:247–59. doi: 10.1002/ijc.23252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schlesinger S, Dubensky TW. Alphavirus vectors for gene expression and vaccines. Curr Opin Biotechnol. 1999;10:434–9. doi: 10.1016/s0958-1669(99)00006-3. [DOI] [PubMed] [Google Scholar]
- 10.Pushko P, Parker M, Ludwig GV, Davis NL, Johnston RE, Smith JF. Replicon-helper systems from attenuated Venezuelan equine encephalitis virus: expression of heterologous genes in vitro and immunization against heterologous pathogens in vivo. Virology. 1997;239:389–401. doi: 10.1006/viro.1997.8878. [DOI] [PubMed] [Google Scholar]
- 11.Hevey M, Negley D, Pushko P, Smith J, Schmaljohn A. Marburg virus vaccines based upon alphavirus replicons protect guinea pigs and nonhuman primates. Virology. 1998;251:28–37. doi: 10.1006/viro.1998.9367. [DOI] [PubMed] [Google Scholar]
- 12.Caley IJ, Betts MR, Davis NL, Swanstrom R, Frelinger JA, Johnston RE. Venezuelan equine encephalitis virus vectors expressing HIV-1 proteins: vector design strategies for improved vaccine efficacy. Vaccine. 1999;17:3124–35. doi: 10.1016/s0264-410x(99)00142-5. [DOI] [PubMed] [Google Scholar]
- 13.Davis NL, Caley IJ, Brown KW, Betts MR, Irlbeck DM, McGrath KM, et al. Vaccination of macaques against pathogenic simian immunodeficiency virus with Venezuelan equine encephalitis virus replicon particles. J Virol. 2000;74:371–8. doi: 10.1128/jvi.74.1.371-378.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pushko P, Bray M, Ludwig GV, Parker M, Schmaljohn A, Sanchez A, et al. Recombinant RNA replicons derived from attenuated Venezuelan equine encephalitis virus protect guinea pigs and mice from Ebola hemorrhagic fever virus. Vaccine. 2000;19:142–53. doi: 10.1016/s0264-410x(00)00113-4. [DOI] [PubMed] [Google Scholar]
- 15.Velders MP, McElhiney S, Cassetti MC, Eiben GL, Higgins T, Kovacs GR, et al. Eradication of established tumors by vaccination with Venezuelan equine encephalitis virus replicon particles delivering human papillomavirus 16 E7 RNA. Cancer Res. 2001;61:7861–7. [PubMed] [Google Scholar]
- 16.Ware CF. Network communications: lymphotoxins, LIGHT, and TNF. Annu Rev Immunol. 2005;23:787–819. doi: 10.1146/annurev.immunol.23.021704.115719. [DOI] [PubMed] [Google Scholar]
- 17.Fu YX, Chaplin DD. Development and maturation of secondary lymphoid tissues. Annu Rev Immunol. 1999;17:399–433. doi: 10.1146/annurev.immunol.17.1.399. [DOI] [PubMed] [Google Scholar]
- 18.Cyster JG. Chemokines and cell migration in secondary lymphoid organs. Science. 1999;286:2098–102. doi: 10.1126/science.286.5447.2098. [DOI] [PubMed] [Google Scholar]
- 19.Ruddle NH. Lymphoid neo-organogenesis: lymphotoxin's role in inflammation and development. Immunol Res. 1999;19:119–25. doi: 10.1007/BF02786481. In Process Citation. [DOI] [PubMed] [Google Scholar]
- 20.Mauri DN, Ebner R, Montgomery RI, Kochel KD, Cheung TC, Yu GL, et al. LIGHT, a new member of the TNF superfamily, and lymphotoxin alpha are ligands for herpesvirus entry mediator. Immunity. 1998;8:21–30. doi: 10.1016/s1074-7613(00)80455-0. [DOI] [PubMed] [Google Scholar]
- 21.Rooney IA, Butrovich KD, Glass AA, Borboroglu S, Benedict CA, Whitbeck JC, et al. The lymphotoxin-beta receptor is necessary and sufficient for LIGHT-mediated apoptosis of tumor cells. J Biol Chem. 2000;275:14307–15. doi: 10.1074/jbc.275.19.14307. [DOI] [PubMed] [Google Scholar]
- 22.Gommerman JL, Browning JL. Lymphotoxin/light, lymphoid microenvironments and autoimmune disease. Nat Rev Immunol. 2003;3:642–55. doi: 10.1038/nri1151. [DOI] [PubMed] [Google Scholar]
- 23.Tamada K, Shimozaki K, Chapoval AI, Zhu G, Sica G, Flies D, et al. Modulation of T-cell-mediated immunity in tumor and graft-versus-host disease models through the LIGHT co-stimulatory pathway. Nat Med. 2000;6:283–9. doi: 10.1038/73136. [DOI] [PubMed] [Google Scholar]
- 24.Wang J, Lo J, Foster A, Yu P, Chen H, W Y, et al. The regulation of T cell homeostasis and autoimmunity by T cell derived LIGHT. J Clinic Invest. 2001:1771–1780. doi: 10.1172/JCI13827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Feltkamp MC, Smits HL, Vierboom MP, Minnaar RP, de Jongh BM, Drijfhout JW, et al. Vaccination with cytotoxic T lymphocyte epitope-containing peptide protects against a tumor induced by human papillomavirus type 16-transformed cells. Eur J Immunol. 1993;23:2242–9. doi: 10.1002/eji.1830230929. [DOI] [PubMed] [Google Scholar]
- 26.Feltkamp MC, Vreugdenhil GR, Vierboom MP, Ras E, van der Burg SH, ter Schegget J, et al. Cytotoxic T lymphocytes raised against a subdominant epitope offered as a synthetic peptide eradicate human papillomavirus type 16-induced tumors. Eur J Immunol. 1995;25:2638–42. doi: 10.1002/eji.1830250935. [DOI] [PubMed] [Google Scholar]
- 27.van Hall T, van de Rhee NE, Schoenberger SP, Vierboom MP, Verreck FA, Melief CJ, et al. Cryptic open reading frames in plasmid vector backbone sequences can provide highly immunogenic cytotoxic T-lymphocyte epitopes. Cancer Res. 1998;58:3087–93. [PubMed] [Google Scholar]
- 28.Garcia-Hernandez Mde L, Gray A, Hubby B, Klinger OJ, Kast WM. Prostate stem cell antigen vaccination induces a long-term protective immune response against prostate cancer in the absence of autoimmunity. Cancer Res. 2008;68:861–9. doi: 10.1158/0008-5472.CAN-07-0445. [DOI] [PubMed] [Google Scholar]
- 29.Cassetti MC, McElhiney SP, Shahabi V, Pullen JK, Le Poole IC, Eiben GL, et al. Antitumor efficacy of Venezuelan equine encephalitis virus replicon particles encoding mutated HPV16 E6 and E7 genes. Vaccine. 2004;22:520–7. doi: 10.1016/j.vaccine.2003.07.003. [DOI] [PubMed] [Google Scholar]
- 30.Yu P, Lee Y, Wang Y, Liu X, Auh S, Gajewski TF, et al. Targeting the primary tumor to generate CTL for the effective eradication of spontaneous metastases. J Immunol. 2007;179:1960–8. doi: 10.4049/jimmunol.179.3.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Garcia-Hernandez Mde L, Gray A, Hubby B, Kast WM. In vivo effects of vaccination with six-transmembrane epithelial antigen of the prostate: a candidate antigen for treating prostate cancer. Cancer Res. 2007;67:1344–51. doi: 10.1158/0008-5472.CAN-06-2996. [DOI] [PubMed] [Google Scholar]
- 32.Wang J, Foster A, Chin R, Yu P, Sun Y, Wang Y, et al. The complementation of lymphotoxin deficiency with LIGHT, a newly discovered TNF family member, for the restoration of secondary lymphoid structure and function. Eur J Immunol. 2002;32:1969–79. doi: 10.1002/1521-4141(200207)32:7<1969::AID-IMMU1969>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- 33.Sharma S, Stolina M, Luo J, Strieter RM, Burdick M, Zhu LX, et al. Secondary lymphoid tissue chemokine mediates T cell-dependent antitumor responses in vivo. J Immunol. 2000;164:4558–63. doi: 10.4049/jimmunol.164.9.4558. [DOI] [PubMed] [Google Scholar]
- 34.Sharma S, Yang SC, Hillinger S, Zhu LX, Huang M, Batra RK, et al. SLC/CCL21-mediated anti-tumor responses require IFNgamma, MIG/CXCL9 and IP-10/CXCL10. Mol Cancer. 2003;2:22. doi: 10.1186/1476-4598-2-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Petreaca ML, Yao M, Ware C, Martins-Green MM. Vascular endothelial growth factor promotes macrophage apoptosis through stimulation of tumor necrosis factor superfamily member 14 (TNFSF14/LIGHT) Wound Repair Regen. 2008;16:602–14. doi: 10.1111/j.1524-475X.2008.00411.x. [DOI] [PubMed] [Google Scholar]
- 36.Schreiber H, Wu TH, Nachman J, Kast WM. Immunodominance and tumor escape. Semin Cancer Biol. 2002;12:25–31. doi: 10.1006/scbi.2001.0401. [DOI] [PubMed] [Google Scholar]