Abstract
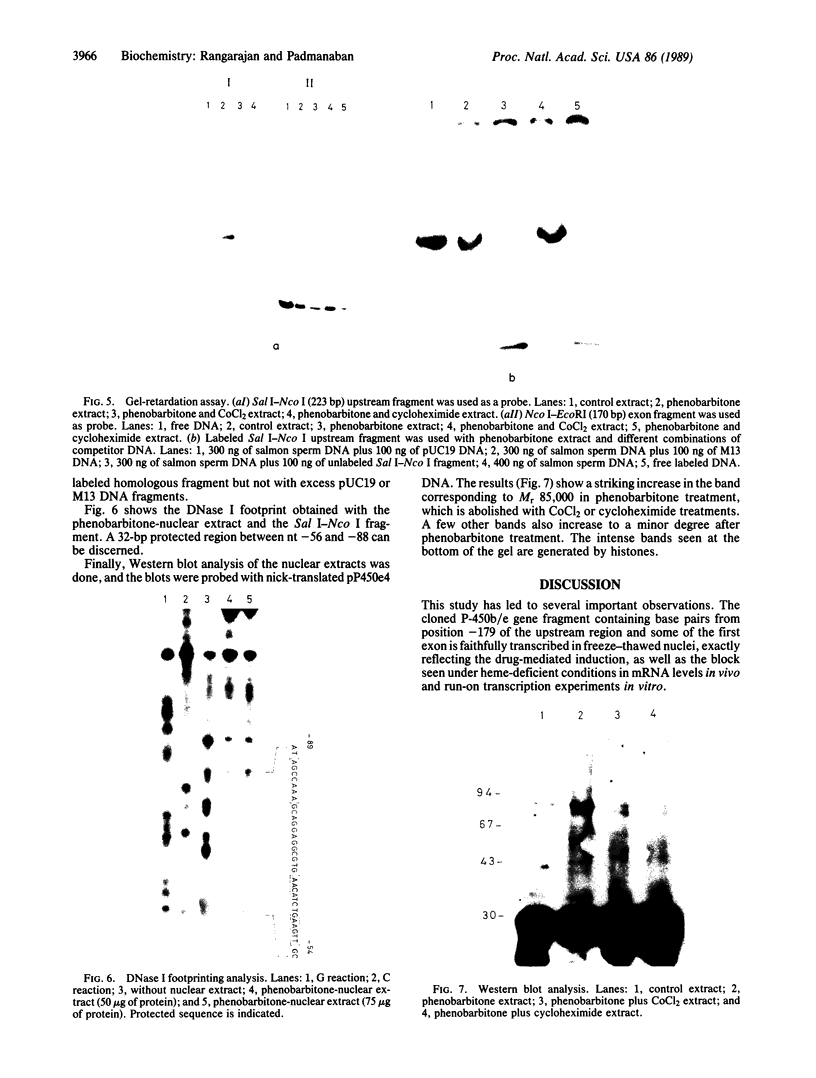
The cloned DNA fragment of the cytochrome P-450b/e gene containing the upstream region from position -179 through part of the first exon is faithfully transcribed in freeze-thawed rat liver nuclei. Phenobarbitone treatment of the animal strikingly increases this transcription, and the increase is blocked by cycloheximide (protein synthesis inhibitor) or CoCl2 (heme biosynthetic inhibitor) treatment of animals. This picture correlates very well with the reported cytochrome P-450b/e mRNA levels in vivo and run-on transcription rates in vitro under these conditions. The upstream region (from position -179) was assessed for protein binding with nuclear extracts by nitrocellulose filter binding, gel retardation, DNase I treatment ("footprinting"), and Western blot analysis. Phenobarbitone treatment dramatically increases protein binding to the upstream region, an increase once again blocked by cycloheximide or CoCl2 treatments. Addition of heme in vitro to heme-deficient nuclei and nuclear extracts restores the induced levels of transcription and protein binding to the upstream fragment, respectively. Thus, drug-mediated synthesis and heme-modulated binding of a transcription factor(s) appear involved in the transcriptional activation of the cytochrome P-450b/e genes, and an 85-kDa protein may be a major factor in this regard.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bhat G. J., Rangarajan P. N., Padmanaban G. Differential effects of cycloheximide on rat liver cytochrome P-450 gene transcription in the whole animal and hepatoma cell culture. Biochem Biophys Res Commun. 1987 Nov 13;148(3):1118–1123. doi: 10.1016/s0006-291x(87)80248-6. [DOI] [PubMed] [Google Scholar]
- Chodosh L. A., Olesen J., Hahn S., Baldwin A. S., Guarente L., Sharp P. A. A yeast and a human CCAAT-binding protein have heterologous subunits that are functionally interchangeable. Cell. 1988 Apr 8;53(1):25–35. doi: 10.1016/0092-8674(88)90484-9. [DOI] [PubMed] [Google Scholar]
- Diffley J. F., Stillman B. Purification of a cellular, double-stranded DNA-binding protein required for initiation of adenovirus DNA replication by using a rapid filter-binding assay. Mol Cell Biol. 1986 May;6(5):1363–1373. doi: 10.1128/mcb.6.5.1363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dwarki V. J., Francis V. N., Bhat G. J., Padmanaban G. Regulation of cytochrome P-450 messenger RNA and apoprotein levels by heme. J Biol Chem. 1987 Dec 15;262(35):16958–16962. [PubMed] [Google Scholar]
- Favaloro J., Treisman R., Kamen R. Transcription maps of polyoma virus-specific RNA: analysis by two-dimensional nuclease S1 gel mapping. Methods Enzymol. 1980;65(1):718–749. doi: 10.1016/s0076-6879(80)65070-8. [DOI] [PubMed] [Google Scholar]
- Fujisawa-Sehara A., Sogawa K., Yamane M., Fujii-Kuriyama Y. Characterization of xenobiotic responsive elements upstream from the drug-metabolizing cytochrome P-450c gene: a similarity to glucocorticoid regulatory elements. Nucleic Acids Res. 1987 May 26;15(10):4179–4191. doi: 10.1093/nar/15.10.4179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujisawa-Sehara A., Yamane M., Fujii-Kuriyama Y. A DNA-binding factor specific for xenobiotic responsive elements of P-450c gene exists as a cryptic form in cytoplasm: its possible translocation to nucleus. Proc Natl Acad Sci U S A. 1988 Aug;85(16):5859–5863. doi: 10.1073/pnas.85.16.5859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galas D. J., Schmitz A. DNAse footprinting: a simple method for the detection of protein-DNA binding specificity. Nucleic Acids Res. 1978 Sep;5(9):3157–3170. doi: 10.1093/nar/5.9.3157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guarente L., Lalonde B., Gifford P., Alani E. Distinctly regulated tandem upstream activation sites mediate catabolite repression of the CYC1 gene of S. cerevisiae. Cell. 1984 Feb;36(2):503–511. doi: 10.1016/0092-8674(84)90243-5. [DOI] [PubMed] [Google Scholar]
- Guertin M., Baril P., Bartkowiak J., Anderson A., Bélanger L. Rapid suppression of alpha 1-fetoprotein gene transcription by dexamethasone in developing rat liver. Biochemistry. 1983 Aug 30;22(18):4296–4302. doi: 10.1021/bi00287a021. [DOI] [PubMed] [Google Scholar]
- Jones P. B., Durrin L. K., Fisher J. M., Whitlock J. P., Jr Control of gene expression by 2,3,7,8-tetrachlorodibenzo-p-dioxin. Multiple dioxin-responsive domains 5'-ward of the cytochrome P1-450 gene. J Biol Chem. 1986 May 25;261(15):6647–6650. [PubMed] [Google Scholar]
- Maniatis T., Goodbourn S., Fischer J. A. Regulation of inducible and tissue-specific gene expression. Science. 1987 Jun 5;236(4806):1237–1245. doi: 10.1126/science.3296191. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- Miskimins W. K., Roberts M. P., McClelland A., Ruddle F. H. Use of a protein-blotting procedure and a specific DNA probe to identify nuclear proteins that recognize the promoter region of the transferrin receptor gene. Proc Natl Acad Sci U S A. 1985 Oct;82(20):6741–6744. doi: 10.1073/pnas.82.20.6741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nebert D. W., Gonzalez F. J. P450 genes: structure, evolution, and regulation. Annu Rev Biochem. 1987;56:945–993. doi: 10.1146/annurev.bi.56.070187.004501. [DOI] [PubMed] [Google Scholar]
- Poland A., Mak I., Glover E. Species differences in responsiveness to 1,4-bis[2-(3,5-dichloropyridyloxy)]-benzene, a potent phenobarbital-like inducer of microsomal monooxygenase activity. Mol Pharmacol. 1981 Sep;20(2):442–450. [PubMed] [Google Scholar]
- Rangarajan P. N., Ravishankar H., Padmanaban G. Isolation of a cytochrome P-450e gene variant and characterization of its 5' flanking sequences. Biochem Biophys Res Commun. 1987 Apr 14;144(1):258–263. doi: 10.1016/s0006-291x(87)80504-1. [DOI] [PubMed] [Google Scholar]
- Ravishankar H., Padmanaban G. Effect of cobalt chloride and 3-amino-1,2,4-triazole on the induction of cytochrome P-450 synthesis by phenobarbitone in rat liver. Arch Biochem Biophys. 1983 Aug;225(1):16–24. doi: 10.1016/0003-9861(83)90002-4. [DOI] [PubMed] [Google Scholar]
- Ravishankar H., Padmanaban G. Regulation of cytochrome P-450 gene expression. Studies with a cloned probe. J Biol Chem. 1985 Feb 10;260(3):1588–1592. [PubMed] [Google Scholar]
- Santoro C., Mermod N., Andrews P. C., Tjian R. A family of human CCAAT-box-binding proteins active in transcription and DNA replication: cloning and expression of multiple cDNAs. Nature. 1988 Jul 21;334(6179):218–224. doi: 10.1038/334218a0. [DOI] [PubMed] [Google Scholar]
- Satyabhama S., Seelan R. S., Padmanaban G. Expression of cytochrome P-450 and albumin genes in rat liver: effect of xenobiotics. Biochemistry. 1986 Aug 12;25(16):4508–4512. doi: 10.1021/bi00364a009. [DOI] [PubMed] [Google Scholar]
- Strauss F., Varshavsky A. A protein binds to a satellite DNA repeat at three specific sites that would be brought into mutual proximity by DNA folding in the nucleosome. Cell. 1984 Jul;37(3):889–901. doi: 10.1016/0092-8674(84)90424-0. [DOI] [PubMed] [Google Scholar]
- Taylor C. W., Yeoman L. C., Daskal I., Busch H. Two-dimensional electrophoresis of proteins of citric acid nuclei prepared with aid of a Tissumizer. Exp Cell Res. 1973 Nov;82(1):215–226. doi: 10.1016/0014-4827(73)90264-4. [DOI] [PubMed] [Google Scholar]
- Wasylyk B. Transcription elements and factors of RNA polymerase B promoters of higher eukaryotes. CRC Crit Rev Biochem. 1988;23(2):77–120. doi: 10.3109/10409238809088317. [DOI] [PubMed] [Google Scholar]
- Ye Z. S., Samuels H. H. Cell- and sequence-specific binding of nuclear proteins to 5'-flanking DNA of the rat growth hormone gene. J Biol Chem. 1987 May 5;262(13):6313–6317. [PubMed] [Google Scholar]