Abstract
OBJECTIVE
Compelling epidemiological and clinical evidence has identified a specific cardiomyopathy in diabetes, characterized by early diastolic dysfunction and adverse structural remodeling. Activation of the insulin-like growth factor 1 (IGF-1) receptor (IGF-1R) promotes physiological cardiac growth and enhances contractile function. The aim of the present study was to examine whether cardiac-specific overexpression of IGF-1R prevents diabetes-induced myocardial remodeling and dysfunction associated with a murine model of diabetes.
RESEARCH DESIGN AND METHODS
Type 1 diabetes was induced in 7-week-old male IGF-1R transgenic mice using streptozotocin and followed for 8 weeks. Diastolic and systolic function was assessed using Doppler and M-mode echocardiography, respectively, in addition to cardiac catheterization. Cardiac fibrosis and cardiomyocyte width, heart weight index, gene expression, Akt activity, and IGF-1R protein content were also assessed.
RESULTS
Nontransgenic (Ntg) diabetic mice had reduced initial (E)-to-second (A) blood flow velocity ratio (E:A ratio) and prolonged deceleration times on Doppler echocardiography compared with nondiabetic counterparts, indicative markers of diastolic dysfunction. Diabetes also increased cardiomyocyte width, collagen deposition, and prohypertrophic and profibrotic gene expression compared with Ntg nondiabetic littermates. Overexpression of the IGF-1R transgene markedly reduced collagen deposition, accompanied by a reduction in the incidence of diastolic dysfunction. Akt phosphorylation was elevated ∼15-fold in IGF-1R nondiabetic mice compared with Ntg, and this was maintained in a setting of diabetes.
CONCLUSIONS
The current study suggests that cardiac overexpression of IGF-1R prevented diabetes-induced cardiac fibrosis and diastolic dysfunction. Targeting IGF-1R–Akt signaling may represent a therapeutic target for the treatment of diabetic cardiac disease.
Diabetes represents a major threat to human health, with global incidence projected to reach 300 million by 2025 (1,2). Cardiovascular complications including coronary heart disease and peripheral vascular disease are regarded as primary causes of morbidity and mortality in both type 1 and type 2 diabetes (3,4). In addition, clinical and experimental evidence supports the existence of a distinct diabetic cardiomyopathy, associated with adverse changes to the structure and function of cardiomyocytes, which can occur independent of macrovascular complications (5,6). Left ventricular (LV) diastolic impairments (in LV filling, relaxation, and/or diastolic distensibility) are evident early in disease progression (7–9), often followed by later onset of systolic dysfunction (e.g., reduced LV ejection fraction and fractional shortening) (10). Functional alterations in the diabetic heart occur concomitantly with development of the structural abnormalities cardiomyocyte hypertrophy and cardiac fibrosis (11–13). In both type 1 and 2 diabetes, this cardiomyopathy is a prognostic indicator, particularly for mortality (14). New approaches to rescue LV remodeling and dysfunction specifically in diabetic myocardium are thus highly desirable.
Physiological heart growth or hypertrophy, which occurs during normal postnatal development and can be induced by exercise, is characterized by a normal cardiac structure and gene expression (15). In contrast, pathological hypertrophy is characterized by fibrosis, myofiber disarray, reduced cardiac output, and eventual heart failure (15). IGF-1, structurally and functionally related to insulin (16), plays a crucial role in stimulating physiological LV hypertrophy and conferring protection against cardiac dysfunction (17–19). The therapeutic potential of IGF-1 has thus been extensively examined in an array of cardiac pathologies, including heart failure and diabetes (18,19). In dilated cardiomyopathy, eccentric hypertrophy, and myocardial infarction (20–22), transgenic IGF-1 expression limited structural abnormalities such as myocyte necrosis and fibrosis (21). IGF-1 upregulation may also improve systolic function, via restoration of normal Ca2+ handling and increased cardiomyocyte contractility (17,20). Similar benefits have been proposed in diabetic myocardium (18,23), although in vivo cardiac functional studies have been limited. Furthermore, chronic IGF-1 may represent a flawed therapeutic approach for diabetes-induced LV dysfunction and remodeling, as both transgenic and pharmacological IGF-1 approaches significantly elevate systemic plasma IGF-1 concentrations and thus can have undesirable effects on nonmyocytes and other tissues. For instance, IGF-1 may induce fibroblast proliferation (24) and increase size of other organs including brain and kidney (17,25,26).
The current study seeks to circumvent the problem of potential noncardiac IGF-1 effects by using a cardiomyocyte-specific IGF-1R transgenic (Tg) mouse (27), which develops physiological cardiac hypertrophy and enhanced systolic function, without histopathology (27). Despite compelling evidence for IGF-1 preservation of cardiac function, the role of cardiac IGF-1R specifically in protecting LV function and structure in the context of diabetes-induced cardiomyopathy in vivo has not been examined. Thus, the present study tested the hypothesis that cardiomyocyte-specific IGF-1R protects against diabetes-induced LV diastolic dysfunction and remodeling, using a streptozotocin (STZ)–induced mouse model of diabetes in vivo.
RESEARCH DESIGN AND METHODS
Animal model.
This study was conducted in accordance to National Health and Medical Research Council of Australia guidelines, and approved by the Alfred Medical Research and Education Precinct Animal Ethics Committee. Heterozygote 7-week-old IGF-1R Tg mice and their age-matched nontransgenic (Ntg) male littermates (FVB/N background) were used. These mice were generated by cloning a cDNA insert for the human IGF-1R gene into a Sa1I-digested α-myosin heavy chain promoter construct (originated from the laboratory of Dr. Seigo Izumo, Beth Israel Deaconess Medical Center and Harvard Medical School). The bacterial sequence was removed from the construct and injected into the male pronucleus of fertilized single-cell FVB/N embryos on an FVB/N background. This transgenic line (no. 7) exhibited a 20-fold increase in IGF-1R expression specifically in the myocardium, without changes in IGF-1 plasma or tissue levels, as previously described (27). Diabetes was induced by five consecutive daily intraperitoneal STZ injections (55 mg/kg, in 0.1 mol/l citrate buffer, pH 4.5; Sigma Aldrich, St. Louis MO); sham mice were administered an equivalent volume of citrate buffer (28). Saphenous vein blood glucose levels were measured every 2 weeks using a hand-held glucometer (Accu-check Advantage; Roche, Basel, Switzerland), with blood glucose levels exceeding 28 mmol/l considered diabetic. Diabetes then progressed for 8 weeks (13), i.e., to 15 weeks of age, and submandibular blood was collected from conscious mice for final plasma glucose assessment (Austin Pathology Service, Melbourne, Victoria, Australia).
Analysis of LV function.
Animals were placed under light anesthesia with a cocktail of ketamine, xylazine, and atropine (100 mg/kg, 10 mg/kg, and 1.2 mg/kg, respectively, i.p.) for echocardiography. Baseline echocardiography (M-mode two-dimensional echocardiography) was performed at 6 weeks of age, using a Philips iE33 echocardiography system with a 15-MHz linear array transducer (29), prior to induction of diabetes (at 7 weeks). End point echocardiography (both two-dimensional M-mode and three-dimensional Doppler echocardiography) was performed at 15 weeks of age. Derived echocardiography parameters included interventricular septum, LV posterior wall thickness, LV diastolic dimension (LVDd), LV systolic dimension (LVDs), and fractional shortening (calculated as [(LVDd − LVDs)/LVDd] × 100%). LV filling was assessed on transmitral Doppler echocardiography, on the ratio of initial (E) and second (A) blood flow velocities (E:A ratio) and E velocity deceleration time (30). Cardiac catheterization was also performed after 8 weeks of diabetes in anesthetized mice (ketamine 100 mg/kg, xylazine 10 mg/kg, atropine 1.2 mg/kg i.p.), by placing a micromanometer-tipped catheter (1.4F; Millar Instrument Co, Houston, TX) in the LV or aorta (29). Parameters measured included aortic systolic and diastolic blood pressure, LV ± dP/dt (pressure change during contraction/relaxation), LV systolic pressure, and LV end-diastolic pressure (LVEDP).
Tissue collection and histology.
After cardiac catheterization, atria, ventricles, and lungs were rinsed in saline and lightly blotted and wet weight was determined. Whole blood was collected for analysis of glycated hemoglobin (GHb) (Austin Pathology Service), plasma insulin (n = 5–8/group, insulin ELISA kit; Millipore, Billerica, MA), and free fatty acids (n = 3/group, Nefa-C assay kit; Wako Chemicals, Richmond, VA, as per manufacturer's instructions). Tibias were collected and digested with 1 M NaOH (overnight, 37°C), and their lengths were measured using vernier calipers. Hearts were cut at the horizontal short-axis plane, and the middle portion of the ventricle was snap-frozen in liquid nitrogen and stored at −80°C for biochemical analysis. The basal portion of the ventricle was fixed in 10% neutral buffered formalin (Australian Biostain, Melbourne, Australia), and paraffin-embedded for histological analysis of 4-μm cross-sections. Hematoxylin-eosin (H-E)–stained sections were photographed under light microscopy at ×400 magnification (with resolution of 2,048 × 1,536 pixels and 300 × 300 dpi), and cardiomyocyte width was manually determined as the shortest dimension per cardiomyocyte (μm), using Olympus Image Pro-plus (Media Cybergenetics, Bethesda, MD), with a photographed scale of the ×400 objective lens for calibration. Mean values (± SEM) for each experimental group were calculated from the mean of 100 individual cardiomyocytes (from 6–8 fields) per mouse. Cardiac collagen deposition was also assessed, on 0.1% picrosirius red–stained sections. Ten fields were randomly chosen, taking care to avoid arterial collagen deposition, and photographed via light microscopy at ×200 magnification. Collagen stained an intense red color, which was measured and analyzed using Olympus Image Pro-plus as a ratio of area of collagen to total area of image.
RNA and protein extraction.
RNA and protein were extracted from frozen ventricle as previously described (31).
Analysis of gene expression.
Changes in cardiac β-myosin heavy chain (a marker of hypertrophy) and pro–collagen III (a marker of fibrosis) expression used real-time PCR. DNase-treated RNA was reverse-transcribed (Taqman Reverse Transcription reagents; Applied Biosystems) using the Gene-Amp PCR system 9700 (Applied Biosystems) to produce cDNA template concentrations of 20 ng/μl (13). β-myosin heavy chain expression was quantitated using SYBR Green chemistry; pro–collagen III used TaqMan universal mastermix (Applied Biosystems), using the ABI Prism 7700 Sequence Detection System, with ribosomal 18S as a control. All primers and probes were produced from murine-specific sequences published on GenBank at previously determined optimal concentrations (31). The comparative delta-delta cycle threshold method was used to analyze changes in gene expression as a relative fold change of Ntg sham mice, as previously described (31). Northern blot analysis was performed to analyze gene expression of the hypertrophic markers atrial (ANP) and B-type (BNP) natriuretic peptide, as previously described (27). Total RNA (7.5 μg) was denatured and loaded onto an agarose gel, and transferred to a Hybond–N membrane (Amersham Biosciences, Stockholm, Sweden).
Analysis of protein expression.
SDS-PAGE and Western blotting were performed to determine protein expression of phospho:total-Akt and IGF-1R:glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Phospho- and total Akt antibodies were obtained from Cell Signaling Technologies (catalog nos. 9271S and 9272; 1:1,000 dilution) and used according to manufacturer's instructions, with bands detected at 60 kDa. IGF-1R and GAPDH antibodies were obtained from Santa Cruz Biotechnology (catalog nos. sc713 and sc32233; 1:1,000 dilution) and used according to manufacturer's instructions, with bands detected at 97 and 37 kDa, respectively.
Statistical analysis.
Results are presented as mean ± SE. Two-way ANOVAs were run on all raw data using SigmaStat (Version 3.5; Erkath, Germany) and used to identify differences between genotype (Ntg or IGF-1R) and disease (diabetic or sham) groups, followed by Student Newman-Keuls post hoc test, unless otherwise stated. P ≤ 0.05 was considered significant. Unpaired t tests were also used to detect significance between Ntg sham and Ntg diabetic animals, where appropriate.
RESULTS
Induction of diabetes.
STZ-induced diabetic Ntg and IGF-1R Tg mice exhibited marked hyperglycemia, on both plasma glucose and GHb in comparison with age-matched nondiabetic shams (Table 1), without genotype-dependent differences. Plasma insulin decreased in both Ntg and IGF-1R Tg STZ mice (P < 0.001 on two-way ANOVA, n = 5–8, Table 1). Furthermore, plasma free fatty acids increased from basal (∼0.4 mmol/l in Ntg sham mice, n = 2) to 1.99 ± 0.14 and 1.57 ± 0.15 mmol/l in Ntg STZ and IGF-1R Tg STZ mice (P < 0.001 on two-way ANOVA, both n = 3), respectively. Body weight and tibia length were comparable across the four groups at the end of the study. IGF-1R Tg mice exhibited greater heart mass than Ntg mice, by ≥30% whether normalized to body weight or tibia length (Table 1), as previously observed in this strain (27). Diabetes however did not significantly change net heart weight–to–body weight or heart weight–to–tibia length ratio, in either Ntg or Tg mice.
TABLE 1.
Postmortem systemic analysis of Ntg and IGF-1R sham and diabetic mice (15 weeks of age)
| Ntg sham |
Ntg diabetic |
IGF-1R sham |
IGF-1R diabetic |
|
|---|---|---|---|---|
| n = 9 | n = 9 | n = 12 | n = 9 | |
| Blood glucose (mM) | 8.5 ± 0.8 | 35.5 ± 1.1* | 10.5 ± 1.0 | 35.4 ± 1.7* |
| GHb (%) | 2.4 ± 0.2 | 9.0 ± 0.6* | 2.3 ± 0.1 | 8.7 ± 0.5* |
| Plasma insulin (ng/ml) | 1.3 ± 0.2 | 0.4 ± 0.0* | 1.1 ± 0.2 | 0.6 ± 0.1* |
| BW (g) | 30.1 ± 1.4 | 28.7 ± 0.6 | 30.7 ± 0.9 | 29.4 ± 0.8 |
| HW (mg) | 122.4 ± 6.3 | 116.2 ± 3.2 | 165.5 ± 3.3†‡ | 154.8 ± 5.2†‡ |
| LW (mg) | 154.7 ± 4.2 | 154.8 ± 4.9 | 151.0 ± 3.5 | 148.8 ± 7.4 |
| TL (mm) | 16.3 ± 0.2 | 16.5 ± 0.1 | 16.5 ± 0.6 | 16.5 ± 0.2 |
| HW/BW (mg/g) | 4.06 ± 0.08 | 4.05 ± 0.05 | 5.46 ± 0.10†‡ | 5.27 ± 0.09†‡ |
| LW/BW (mg/g) | 5.31 ± 0.25 | 5.11 ± 0.24 | 5.27 ± 0.15 | 5.01 ± 0.32 |
| HW/TL (mg/mm) | 7.48 ± 0.32 | 7.04 ± 0.18 | 9.97 ± 0.13†‡ | 9.38 ± 0.24†‡ |
| LW/TL (mg/mm) | 9.41 ± 0.32 | 9.39 ± 0.32 | 9.15 ± 0.23 | 9.13 ± 0.47 |
BW, body weight; HW, heart weight; LW, lung weight; TL, tibial length.
*P < 0.05 vs. sham of the same genotype;
†P < 0.05 vs. Ntg sham;
‡P < 0.05 vs. Ntg diabetic.
IGF-1R prevents diabetes-induced cardiomyocyte hypertrophy.
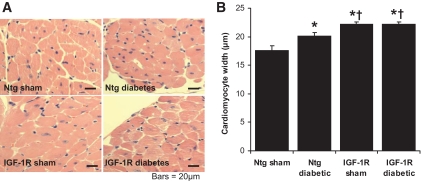
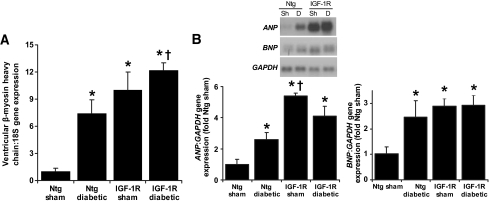
Cardiomyocyte width was significantly greater in Ntg diabetic mice versus Ntg sham (Fig. 1A and B), despite no net increase in heart weight–to–body weight or heart weight–to–tibia length ratios. IGF-1R Tg develop physiological cardiac hypertrophy with a 46% increase in myocyte volume (27). Consistent with the initial characterization of IGF-1R Tg mice, IGF-1R sham mice had enlarged cardiomyocytes versus Ntg sham. In contrast to the diabetes-induced increase in myocyte width in Ntg, diabetes failed to elicit any further increase in cardiomyocyte width in IGF-1R Tg mice (Fig. 1A and B). As with cardiomyocyte width, gene expression of β-myosin heavy chain, ANP, and BNP (Fig. 2A and B) were greater in Ntg diabetic mice versus Ntg sham. Expression of all three hypertrophic markers was also increased in IGF-1R sham versus Ntg sham, but again diabetes failed to induce further expression of these hypertrophic markers in the IGF-1R Tg animals.
FIG. 1.
Cardiomyocyte width is increased by diabetes and IGF-1R overexpression on histological analysis of ventricular cross-sections stained with H-E. A: Representative sections from the LV of sham and diabetic Ntg and IGF-1R mice (H-E stain; magnification ×400). Scale bars show 20 μm. B: Cardiomyocyte width pooled data. n = 8–12 in each group; *P < 0.001 vs. Ntg sham; †P < 0.001 vs. Ntg diabetes. (A high-quality digital representation of this figure is available in the online issue.)
FIG. 2.
A: Gene expression of β-myosin heavy chain (hypertrophic marker) was analyzed on real-time PCR and normalized against 18S expression. n = 8–11 for each group. B: Representative Northern blot showing total RNA. GAPDH was used to normalize for RNA loading (insert). Gene expression of ANP (n = 9–10 per group) and BNP (n = 4–5 per group) was assessed. Mean values for Ntg sham were normalized to 1. *P < 0.05 vs. Ntg sham; †P < 0.05 vs. Ntg diabetic. Sh, sham; D, diabetes.
IGF-1R limits diabetes-induced cardiac fibrosis.
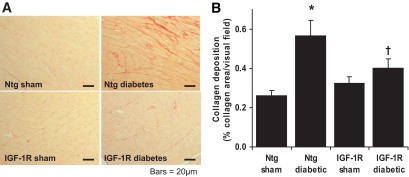
On sirius red–stained sections, diabetes significantly increased cardiac collagen deposition by 2.2 ± 0.3-fold in Ntg mice (P < 0.05; Fig. 3A and B). Cardiac collagen content in IGF-1R sham mice was similar to Ntg sham mice. Expression of the IGF-1R transgene significantly reduced the extent of cardiac fibrosis in diabetic mice. In contrast to Ntg animals, diabetes failed to increase collagen content in IGF-1R Tg hearts, where collagen deposition of IGF-1R diabetic mice was 1.2 ± 0.1-fold IGF-1R sham (P = not significant). Gene expression of the fibrotic marker pro–collagen III tended to increase in Ntg diabetic mice (2.4 ± 0.6-fold Ntg sham, P = 0.06; unpaired t test). Diabetes failed to upregulate pro–collagen III expression in IGF-1R Tg mice (pro–collagen III expression in IGF-1R diabetic mice was 0.87 ± 0.3-fold that of IGF-1R sham mice, P = not significant).
FIG. 3.
A: Representative sections from the LV of sham and diabetic Ntg and IGF-1R mice. Collagen deposition appears red (sirius red stain; magnification ×200). Scale bars show 40 μm. B: Quantitation of collagen area/total ventricular area pooled data. n = 8–12 in each group; *P < 0.05 vs. Ntg sham; †P < 0.05 vs. Ntg diabetic. A high-quality digital representation of this figure is available in the online issue.)
IGF-1R protects against diastolic dysfunction.
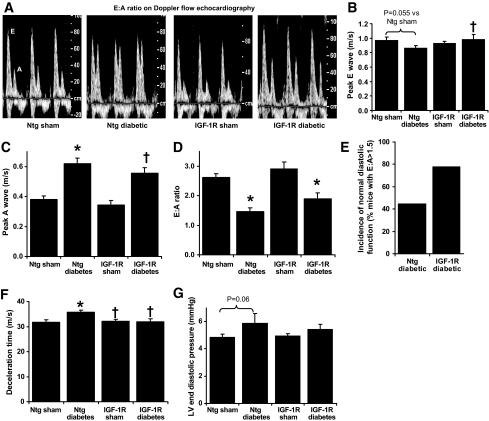
Consistent with IGF-1R mice developing physiological cardiac hypertrophy, IGF-1R Tg mice had increased LV wall thicknesses and enhanced systolic function (estimated by fractional shortening) versus Ntg at baseline by echocardiography (6–7 weeks of age, prior to induction of diabetes; Table 2). After 8 weeks of diabetes, systolic dysfunction was not evident; fractional shortening, LV posterior wall thickness, and LV systolic dimension were comparable in Ntg sham and Ntg diabetic mice (Table 2). The enhanced systolic function seen in IGF-1R Tg mice at baseline was maintained at 15 weeks of age, in both sham and diabetic mice. After 8 weeks of diabetes in Ntg mice, diastolic dysfunction was evident compared with Ntg sham mice (decreased E wave velocity and E:A ratio, increased A wave velocity and a trend for decreased E wave velocity, on Doppler flow echocardiography, Fig. 4A–E). Diastolic function was similar in IGF-1R sham and Ntg sham. A fall in the E:A ratio was also observed in IGF-1R diabetic mice compared with IGF-1R sham mice. Diabetes reduced the E:A ratio by 44 ± 5% in Ntg mice, and by 35 ± 7% in IGF-1R Tg mice. However, the number of mice with normal diastolic function (arbitrarily defined as E:A > 1.5; Fig. 4C) appeared greater in IGF-1R diabetic mice compared with Ntg diabetic mice. Diabetes-induced prolongation in deceleration time was significantly ameliorated by IGF-1R upregulation (Fig. 4F). LV catheterization performed after 8 weeks of diabetes further confirmed the presence of diastolic dysfunction in Ntg diabetic mice compared with sham mice (LV-dP/dt decreased by ∼20 ± 5%; P < 0.05, unpaired t test; Table 3). Diabetes also tended to increase LVEDP in Ntg diabetic mice (again by ∼20%, P = 0.06 on two-way ANOVA; Fig. 4G). In IGF-1R mice however, diabetes failed to significantly impact on either LV-dP/dt or LVEDP. Together, these data suggest that transgenic IGF-1R expression exhibited some protection against diabetes-induced diastolic dysfunction.
TABLE 2.
Echocardiographic analysis of heart dimensions and function
| Baseline (pre-diabetic, 7 weeks old) |
End point measurements (15 weeks old) |
|||||
|---|---|---|---|---|---|---|
| Ntg |
IGF-1R |
Ntg sham |
Ntg diabetic |
IGF-1R sham |
IGF-1R diabetic |
|
| n = 18 | n = 21 | n = 9 | n = 9 | n = 12 | n = 9 | |
| Heart rate (bpm) | 436 ± 14 | 423 ± 12 | 391 ± 20 | 414 ± 12 | 370 ± 13 | 402 ± 8 |
| LV posterior wall thickness (mm) | 0.75 ± 0.03 | 0.89 ± 0.03* | 0.71 ± 0.02 | 0.76 ± 0.03 | 0.96 ± 0.03† | 0.90 ± 0.03† |
| LV end diastolic dimension (mm) | 3.32 ± 0.06 | 3.27 ± 0.04 | 3.70 ± 0.07 | 3.52 ± 0.08 | 3.26 ± 0.10† | 3.58 ± 0.10‡ |
| LV end systolic dimension (mm) | 2.02 ± 0.08 | 1.65 ± 0.07* | 2.40 ± 0.07 | 2.19 ± 0.12 | 1.66 ± 0.14† | 1.95 ± 0.07 |
| Fractional shortening (%) | 40 ± 2 | 50 ± 2* | 35 ± 2 | 37 ± 2 | 50 ± 3† | 45 ± 1† |
*P < 0.05 vs. Ntg sham (baseline measurement);
†P < 0.05 vs. Ntg sham and Ntg diabetic;
‡P < 0.05 vs. IGF-1R sham.
FIG. 4.
Impact of cardiac-specific IGF-1R overexpression on diabetes-induced diastolic dysfunction. A: Representative mitral flow patterns from pulsed wave Doppler echocardiography. Diabetes (B) tends to decrease E velocity and (C) increases peak A velocity, with (D) net reduction in E:A wave ratio in both Ntg and IGF-1R diabetic mice. IGF-1R (E) reduces incidence of diastolic dysfunction, (F) restores normal deceleration time, and (G) tends to improve LV end diastolic pressure. n = 7–12 in each group; *P < 0.05 vs. sham of the same genotype; †P < 0.05 vs. Ntg counterpart.
TABLE 3.
Analysis of cardiac function via cardiac catheterization
| Ntg sham |
Ntg diabetic |
IGF-1R sham |
IGF-1R diabetic |
|
|---|---|---|---|---|
| n = 8 | n = 7 | n = 9 | n = 9 | |
| Heart rate (bpm) | 349 ± 10 | 364 ± 15 | 369 ± 17 | 372 ± 11 |
| Ao SBP (mmHg) | 107.8 ± 3.7 | 87.0 ± 3.3* | 99.4 ± 2.8 | 88.5 ± 2.8* |
| Ao DBP (mmHg) | 73.9 ± 3.6 | 59.2 ± 3.1* | 68.2 ± 2.5 | 60.5 ± 3.5 |
| LV+dP/dt (mmHg/s) | 7,237 ± 293 | 8,297 ± 586 | 8,628 ± 426† | 8,029 ± 557 |
| LV−dP/dt (mmHg/s) | −6,699 ± 361 | −5,543 ± 314† | −6,006 ± 284 | −5,500 ± 341 |
| LVSP (mmHg) | 102.8 ± 2.5 | 90.2 ± 2.2* | 96.9 ± 2.6 | 92.7 ± 3.1 |
Ao SBP, aortic systolic blood pressure; Ao DBP, aortic diastolic blood pressure; LVSP, left ventricular systolic pressure.
*P < 0.05 vs. sham of the same genotype;
†P < 0.05 vs. Ntg sham (on unpaired t test).
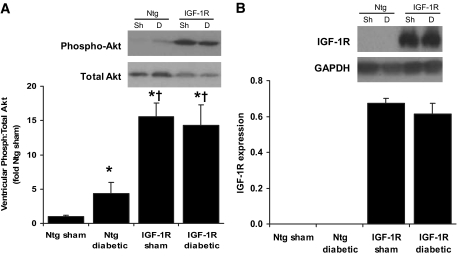
Preserved Akt activity in diabetic IGF-1R transgenic mice.
It has previously been shown that the physiological hypertrophy observed in IGF-1R is associated with activation of the phospho-inositide 3-kinase (PI3K) (p110α)–Akt pathway (27) and Akt is a critical mediator of physiological heart growth (32). In the current study, Akt phosphorylation was elevated in ventricles of IGF-1R sham mice by more than 15-fold versus Ntg sham, from 0.27 ± 0.04 to 4.26 ± 0.54 arbitrary units (AU) in Ntg and IGF-1R sham mice, respectively (Fig. 5A), which was maintained in diabetes. Akt phosphorylation was 3.92 ± 0.82 AU in IGF-1R Tg STZ mice (P(genotype) < 0.001 on two-way ANOVA). Interestingly, a modest increase in phospho-Akt (approximately fourfold) was also observed in Ntg diabetic mice compared with Ntg sham mice. No differences in extracellular signal–related kinase 1/2 phosphorylation however were detected in the groups of mice (results not shown). Lastly, as shown in Fig. 5B, the level of IGF-1R protein was comparable between IGF-1R sham and IGF-1R diabetic mice.
FIG. 5.
A: Representative Western blot showing phosphorylation of Akt and total Akt in sham (Sh) and diabetic (D) mice (insert), and quantitative analysis of Akt phosphorylation normalized to total Akt (left panel). Mean values for Ntg sham were normalized to 1. n = 4–6 in each group; *P < 0.05 vs. Ntg sham; †P < 0.05 vs. Ntg counterpart. B: Representative Western blot showing protein levels of IGF-1R and GAPDH protein (insert) and quantitative analysis of IGF-1R normalized to GAPDH (n = 3/group).
DISCUSSION
Diabetic cardiomyopathy is typically associated with diastolic dysfunction, myocardial fibrosis, and cardiomyocyte hypertrophy. The primary aim of the present study was to elucidate whether cardiac-specific upregulation of IGF-1R prevented cardiac dysfunction and adverse structural remodeling associated with pathophysiology of the diabetic heart. Key findings from the study provide evidence that IGF-1R specifically protects against diabetes-induced diastolic dysfunction and cardiac fibrosis. IGF-1R induced activation of the cell survival kinase Akt was maintained in a setting of diabetes, suggesting that beneficial actions of IGF-1R may be linked to promotion of physiological hypertrophy. The attenuation of diabetes-induced structural and functional cardiac damage mediated by IGF-1R signaling was independent of the degree of hyperglycemia and body weight.
IGF-1R promotes physiological hypertrophy and is protective in a setting of diabetes.
Physiological cardiac growth occurs during postnatal development, and in response to long-term exercise training (15). This form of hypertrophy is associated with normal or enhanced cardiac function and normal cardiac structure. In contrast, pathological cardiac hypertrophy may arise as a compensatory mechanism against increased myocyte stress in many disease states. Any initial benefit is however overridden by eventual derangement of myocardial architecture and deterioration of function (15). Diabetic cardiomyopathy is characterized by adverse structural changes to the heart, including increased cardiac fibrosis (pathological in nature) and cardiomyocyte hypertrophy. Our results show that diabetes increased several markers of cardiomyocyte hypertrophy, including cardiomyocyte width as well as gene expression of β-myosin heavy chain, ANP, and BNP as previously described (13). No changes in heart weight–to–body weight or heart weight–to–tibia length ratios were observed in Ntg diabetic mice, indicating no net increased heart size. Diabetes-induced cardiomyocyte hypertrophy in the absence of gross LV mass increase may result from loss of cardiomyocytes via hyperglycemia-induced apoptosis and necrosis (23,33). Compensatory hypertrophy of the remaining viable cardiomyocytes, in addition to fibrosis, may then occur, and therefore any increase in overall size of each cardiomyocyte is counterbalanced by a reduction in the total number of myocytes (33). The upregulation of hypertrophic gene expression observed in this study is consistent with this. The causes underlying the diabetes-induced hypertrophic response is multifaceted, and may involve metabolic derangements such as impaired glucose handling (34) and oxidative stress (23,31).
Adaptive physiological hypertrophy in both humans (35) and animal models (27,36) has been associated with increased IGF-1 formation and/or activation. This hypertrophy is characterized by normal heart structure and absence of myocardial fibrosis (17,27). In contrast, pathological hypertrophy (15) is accompanied by cardiac fibrosis and detrimental changes in LV function (such as that observed in diabetes in the present study). Consistent with previous findings (27), we demonstrated significant enlargement in cardiomyocyte size in IGF-1R Tg mice, accompanied by increased heart weight–to–body weight and heart weight–to–tibia length ratios, increased LV wall thicknesses, and enhanced systolic function compared with Ntg mice. Surprisingly, markers commonly associated with pathological hypertrophy (β-myosin heavy chain, ANP, and BNP) were also increased in IGF-1R Tg compared with Ntg sham mice. However, this is consistent with both the original characterization and the recent contention as to whether fetal gene expression alone is an indicator of whether hypertrophy is “pathological” or “physiological” in nature (27,29). Histological analysis of cardiac structure and analysis of LV function might thus be more appropriate markers of the type of hypertrophy present. Importantly, diabetes failed to elicit further increases in any parameter of hypertrophy studied in IGF-1R mice, suggesting that the IGF-1R–induced physiological hypertrophic response was able to blunt the pathological hypertrophic response induced by diabetes. Expression of the IGF-1R transgene was previously shown to blunt pressure-overload induced pathological growth (27). Together, this demonstrates that transgenic IGF-1R expression is associated with the preservation of normal cardiac structure and function in the setting of diabetes.
IGF-1R overexpression inhibits diabetes-induced cardiac fibrosis.
Increased cardiac fibrosis is a common hallmark of the diabetic heart (11,37). The resultant increased LV stiffness further compromises the diabetic heart's ability to contract and relax efficiently (14). In the present study, Ntg diabetic mice exhibited increased cardiac collagen deposition, consistent with previous findings in rats (38) and there was a tendency for elevated pro–collagen III gene expression compared with Ntg sham controls. IGF-1R overexpression alone was not associated with increased cardiac fibrosis on either marker (in line with its “physiological” phenotype), as collagen deposition in IGF-1R sham mice was similar to Ntg sham mice. Furthermore, in contrast to Ntg mice, no difference in collagen deposition was detectable between IGF-1R sham and IGF-1R diabetic mice. This suggests that IGF-1R overexpression confers protection against cardiac fibrosis induced by diabetes. Procollagen gene expression of the IGF-1R diabetic mice also tended to decrease compared with Ntg diabetic mice; however this change was not significant. The lack of cardiac fibrosis in diabetic IGF-1R Tg mice lends further support to the existing body of evidence that IGF-1R–induced hypertrophy is of a physiological nature, providing protection against adverse cardiac remodeling (27).
IGF-1R expression attenuates diastolic dysfunction.
In the present study, diastolic and systolic function was measured both noninvasively (echocardiography) and invasively (LV catheterization). LV diastolic dysfunction is considered one of the earliest hallmarks of diabetic cardiomyopathy, occurring prior to the onset of systolic dysfunction (8,39). Doppler echocardiography revealed that Ntg diabetic mice exhibited a pronounced decrease in the transmitral E:A ratio, ∼60% of the mice had an E:A ratio <1.5, and deceleration time was prolonged, all indicative of diastolic dysfunction. This was further supported by LV catheterization, on which diabetes reduced LV-dP/dt, with a trend for increased LVEDP. Taken together, these results support the presence of diabetes-induced diastolic dysfunction, as previously described (8,30,39). Our observation is consistent with increased collagen deposition observed in Ntg diabetic mice; the onset of LV diastolic dysfunction may be secondary to changes in the extracellular matrix scaffold of the heart, resulting in increased cardiac stiffness (37). Although transgenic IGF-1R expression itself did not enhance diastolic function in the absence of diabetes, it prevented the diabetes-induced prolongation of LV deceleration time and increased the number of diabetic mice with normal diastolic function. Diabetes also failed to elevate LVEDP in IGF-1R Tg mice. To our knowledge, we provide the first evidence that IGF-1R signaling specifically ameliorates diabetes-induced diastolic dysfunction and fibrosis in a mouse model in vivo.
We chose our 8-week study period of STZ-induced diabetes in mice to specifically assess the impact of IGF1-R on diastolic function in isolation. As such, no difference was detected in systolic function (assessed by both fractional shortening and LV+dP/dt) of Ntg diabetic mice compared with Ntg sham mice. The duration of diabetes is important, as systolic dysfunction in humans often manifests with a later onset than diastolic dysfunction (8,39). Our observations of diastolic dysfunction accompanied by a lack of systolic dysfunction in the diabetic heart are consistent with previous reports (40,41). However, species and strain clearly are important in dictating the presence of systolic dysfunction. Eight weeks of hyperglycemia is sufficient to induce systolic dysfunction in Wistar rats (9) and C57/BL6J mice (42). In contrast, 8 weeks of STZ-induced diabetes was not associated with impaired systolic function in our mice, which were generated on an FVB background. Importantly, FVB and C57 backgrounds show comparable cardiac (diastolic dysfunction, cardiomyocyte hypertrophy, and fibrosis) and blood glucose sensitivities to the STZ mouse model of diabetes (43,44). Consistent with previous reports (27), mice overexpressing IGF-1R exhibited enhanced systolic function (on echocardiography-derived fractional shortening) compared with Ntg mice, both prior to and after induction of diabetes (6 and 15 weeks of age, respectively). Because systolic function was preserved in our model of diabetes, IGF-1R Tg mice had increased systolic function compared with Ntg mice that was not impacted by the presence of diabetes.
Cardioprotective signaling in the diabetic heart.
PI3K (p110α) and Akt are important downstream effectors of IGF-1R signaling, critical for regulation of cardiac growth and survival (27,32,45,46). We observed a marked increase in ventricular phospho-Akt in IGF-1R Tg mice in the current study, in both the absence and presence of diabetes. Contrary to a previous report (47), Akt phosphorylation was increased in Ntg diabetic animals. This effect was relatively modest, and may be a compensatory response to the functional and structural derangements in the diabetic heart, thus warranting further investigation. The IGF-1R–PI3K(p110α)–Akt pathway plays a crucial role in inducing physiological (but not pathological) hypertrophy (19,27,46). Maintained elevated Akt activity in hearts of IGF-1R, together with improvements in both diastolic function and cardiac structure compared with Ntg diabetic mice, suggest the protective effects of IGF-1R signaling in diabetes may be attributable to IGF-1R induction of physiological hypertrophy. These findings may provide an additional mechanism by which regular exercise (which also activates this cardioprotective cascade [15]) improves morbidity and mortality in patients with diabetes (48–50).
A previous study reported that IGF-1 ligand transgenic expression in an STZ mouse model of diabetes protected the heart against diabetes-induced diastolic dysfunction (23). However, collagen deposition was not examined, and mice were studied only for up to 30 days after STZ treatment. The authors acknowledged that limitations of their study included higher circulating IGF-1 levels, which may have contributed to their results, in addition to the relatively short period of diabetes. Kajstura et al. attribute some of the IGF-1–mediated protection to inhibition of apoptosis, necrosis, and angiotensin II–mediated oxidative stress (23). Here, we demonstrate that IGF-1R overexpression is beneficial even in the absence of elevated circulating IGF-1 levels, and over a longer period of time (8 weeks vs. 30 days). Furthermore, we provide another potential mechanism to explain the protective properties of the IGF-1R, i.e., activation of Akt.
To our knowledge, this study is the first to assess the cardioprotective effect of IGF-1R overexpression on diabetes-induced cardiac dysfunction and remodeling. We have demonstrated that overexpression of IGF-1R specifically in the heart attenuated diabetes-induced cardiac fibrosis, and prevented diastolic dysfunction. This cardioprotective effect may be mediated via maintenance of enhanced IGF-1–PI3K(p110α)–Akt signaling, although further investigation is required. Given that cardiac complications are a major contributor to morbidity and mortality in both type 1 and type 2 diabetic patients, studies such as this may identify possible new therapeutic approaches toward reducing cardiac structural and functional damage induced by diabetes.
ACKNOWLEDGMENTS
This work was supported by the Diabetes Australia Research Trust and the National Health and Medical Research Council of Australia (NHMRC project ID526638). K.H. is supported by an Australian Postgraduate Award. J.R.M. was the recipient of a Career Development Award cofunded by the NHMRC and National Heart Foundation of Australia (317835/CR 04M1716) and is supported by an Australian Research Council Future Fellowship (FT0991657). X.-J.D. holds an NHMRC Senior Research Fellowship (ID317808). R.H.R. is the recipient of an NHMRC Senior Research Fellowship (ID472673).
No potential conflicts of interest relevant to this article were reported.
Parts of this study were presented in abstract form at the 20th Scientific Sessions of the American Heart Association, Orlando, Florida, November 14–18, 2009.
We thank Keith Buxton for assistance with animal studies and MaryAnn Arnstein for helpful advice on histological analysis.
Footnotes
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
REFERENCES
- 1.King H, Aubert RE, Herman WH: Global burden of diabetes, 1995–2025: prevalence, numerical estimates, and projections. Diabetes Care 1998; 21: 1414–1431 [DOI] [PubMed] [Google Scholar]
- 2.Zimmet P, Alberti KG, Shaw J: Global and societal implications of the diabetes epidemic. Nature 2001; 414: 782–787 [DOI] [PubMed] [Google Scholar]
- 3.Retnakaran R, Zinman B: Type 1 diabetes, hyperglycaemia, and the heart. Lancet 2008; 371: 1790–1799 [DOI] [PubMed] [Google Scholar]
- 4.Pierce GN, Maddaford TG, Russell JC: Cardiovascular dysfunction in insulin-dependent and non-insulin-dependent animal models of diabetes mellitus. In Symposium of the Canadian-Federation-of-Biological-Societies on Myocardial Ischemia and Reperfusion: Mechanisms of Injury and Protection London, Canada, National Research Council of Canada, 1996, p. 343–350 [PubMed] [Google Scholar]
- 5.Rubler S, Dlugash J, Yuceoglu YZ, Kumral T, Branwood AW, Grishman A: New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am J Cardiol 1972; 30: 595–602 [DOI] [PubMed] [Google Scholar]
- 6.Galderisi M, Anderson KM, Wilson PW, Levy D: Echocardiography evidence for the existence of a distinct diabetic cardiomyopathy (The Framingham-heart study). Am J Cardiol 1991; 68: 85–89 [DOI] [PubMed] [Google Scholar]
- 7.Zabalgoitia M, Ismaeil MF, Anderson L, Maklady FA: Prevalence of diastolic dysfunction in normotensive, asymptomatic patients with well-controlled type 2 diabetes mellitus. Am J Cardiol 2001; 87: 320–323 [DOI] [PubMed] [Google Scholar]
- 8.Schannwell CM, Schneppenheim M, Perings S, Plehn G, Strauer BE: Left ventricular diastolic dysfunction as an early manifestation of diabetic cardiomyopathy. Cardiology 2002; 98: 33–39 [DOI] [PubMed] [Google Scholar]
- 9.Hoit BD, Castro C, Bultron G, Knight S, Matlib MA: Noninvasive evaluation of cardiac dysfunction by echocardiography in streptozotocin-induced diabetic rats. J Card Fail 1999; 5: 324–333 [DOI] [PubMed] [Google Scholar]
- 10.Vinereanu D, Nicolaides E, Tweddel AC, Mädler CF, Holst B, Boden LE, Cinteza M, Rees AE, Fraser AG: Subclinical left ventricular dysfunction in asymptomatic patients with Type II diabetes mellitus, related to serum lipids and glycated haemoglobin. Clin Sci 2003; 105: 591–599 [DOI] [PubMed] [Google Scholar]
- 11.Shimizu M, Umeda K, Sugihara N, Yoshio H, Ino H, Takeda R, Okada Y, Nakanishi I: Collagen remodelling in myocardia of patients with diabetes. J Clin Path 1993; 46: 32–36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mizushige K, Yao L, Noma T, Kiyomoto H, Yu Y, Hosomi N, Ohmori K, Matsuo H: Alteration in left ventricular diastolic filling and accumulation of myocardial collagen at insulin-resistant prediabetic stage of a type II diabetic rat model. Circulation 2000; 101: 899–907 [DOI] [PubMed] [Google Scholar]
- 13.Rosenkranz AC, Hood SG, Woods RL, Dusting GJ, Ritchie RH: B-type natriuretic peptide prevents acute hypertrophic responses in the diabetic rat heart: importance of cyclic GMP. Diabetes 2003; 52: 2389–2395 [DOI] [PubMed] [Google Scholar]
- 14.Devereux RB, Roman MJ, Paranicas M, O'Grady MJ, Lee ET, Welty TK, Fabsitz RR, Robbins D, Rhoades ER, Howard BV: Impact of diabetes on cardiac structure and function: the strong heart study. Circulation 2000; 101: 2271–2276 [DOI] [PubMed] [Google Scholar]
- 15.McMullen JR, Jennings GL: Differences between pathological and physiological cardiac hypertrophy: novel therapeutic strategies to treat heart failure. Clin Exp Pharmacol Physiol 2007; 34: 255–262 [DOI] [PubMed] [Google Scholar]
- 16.Ranke MB: Insulin-like growth factor-I treatment of growth disorders, diabetes mellitus and insulin resistance. Trends Endocrinol Metab 2005; 16: 190–197 [DOI] [PubMed] [Google Scholar]
- 17.Duerr RL, Huang S, Miraliakbar HR, Clark R, Chien KR, Ross J, Jr: Insulin-like growth factor-1 enhances ventricular hypertrophy and function during the onset of experimental cardiac failure. J Clin Invest 1995; 95: 619–627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Norby FL, Aberle NS, II, Kajstura J, Anversa P, Ren J: Transgenic overexpression of insulin-like growth factor I prevents streptozotocin-induced cardiac contractile dysfunction and beta-adrenergic response in ventricular myocytes. J Endocrinol 2004; 180: 175–182 [DOI] [PubMed] [Google Scholar]
- 19.McMullen JR: Role of insulin-like growth factor 1 and phosphoinositide 3-kinase in a setting of heart disease. Clin Exp Pharmacol Physiol 2008; 35: 349–354 [DOI] [PubMed] [Google Scholar]
- 20.Welch S, Plank D, Witt S, Glascock B, Schaefer E, Chimenti S, Andreoli AM, Limana F, Leri A, Kajstura J, Anversa P, Sussman MA: Cardiac-specific IGF-1 expression attenuates dilated cardiomyopathy in tropomodulin-overexpressing transgenic mice. Circ Res 2002; 90: 641–648 [DOI] [PubMed] [Google Scholar]
- 21.Li B, Setoguchi M, Wang X, Andreoli AM, Leri A, Malhotra A, Kajstura J, Anversa P: Insulin-like growth factor-1 attenuates the detrimental impact of nonocclusive coronary artery constriction on the heart. Circ Res 1999; 84: 1007–1019 [DOI] [PubMed] [Google Scholar]
- 22.Li Q, Li B, Wang X, Leri A, Jana KP, Liu Y, Kajstura J, Baserga R, Anversa P: Overexpression of insulin-like growth factor-1 in mice protects from myocyte death after infarction, attenuating ventricular dilation, wall stress, and cardiac hypertrophy. J Clin Invest 1997; 100: 1991–1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kajstura J, Fiordaliso F, Andreoli AM, Li B, Chimenti S, Medow MS, Limana F, Nadal-Ginard B, Leri A, Anversa P: IGF-1 overexpression inhibits the development of diabetic cardiomyopathy and angiotensin II-mediated oxidative stress. Diabetes 2001; 50: 1414–1424 [DOI] [PubMed] [Google Scholar]
- 24.van Eickels M, Vetter H, Grohé C: Angiotensin-converting enzyme (ACE) inhibition attenuates insulin-like growth factor-I (IGF-1) induced cardiac fibroblast proliferation. Br J Pharmacol 2000; 131: 1592–1596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Reiss K, Cheng W, Ferber A, Kajstura J, Li P, Li B, Olivetti G, Homcy CJ, Baserga R, Anversa P: Overexpression of insulin-like growth factor-1 in the heart is coupled with myocyte proliferation in transgenic mice. Proc Natl Acad Sci U S A 1996; 93: 8630–8635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fiorotto ML, Schwartz RJ, Delaughter MC: Persistent IGF-1 overexpression in skeletal muscle transiently enhances DNA accretion and growth. FASEB J 2002; 16: 59–60 [DOI] [PubMed] [Google Scholar]
- 27.McMullen JR, Shioi T, Huang WY, Zhang L, Tarnavski O, Bisping E, Schinke M, Kong S, Sherwood MC, Brown J, Riggi L, Kang PM, Izumo S: The insulin-like growth factor 1 receptor induces physiological heart growth via the phosphoinositide 3-kinase(p110alpha) pathway. J Biol Chem 2004; 279: 4782–4793 [DOI] [PubMed] [Google Scholar]
- 28.Soro-Paavonen A, Watson AM, Li J, Paavonen K, Koitka A, Calkin AC, Barit D, Coughlan MT, Drew BG, Lancaster GI, Thomas M, Forbes JM, Nawroth PP, Bierhaus A, Cooper ME, Jandeleit-Dahm KA: Receptor for advanced glycation end products (RAGE) deficiency attenuates the development of atherosclerosis in diabetes. Diabetes 2008; 57: 2461–2469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kiriazis H, Wang K, Xu Q, Gao XM, Ming Z, Su Y, Moore XL, Lambert G, Gibbs ME, Dart AM, Du XJ: Knockout of beta(1)- and beta(2)-adrenoceptors attenuates pressure overload-induced cardiac hypertrophy and fibrosis. Br J Pharmacol 2008; 153: 684–692 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wichi R, Malfitano C, Rosa K, De Souza SB, Salemi V, Mostarda C, De Angelis K, Irigoyen MC: Noninvasive and invasive evaluation of cardiac dysfunction in experimental diabetes in rodents. Cardiovasc Diabet 2007; 6: 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ritchie RH, Quinn JM, Cao AH, Drummond GR, Kaye DM, Favaloro JM, Proietto J, Delbridge LM: The antioxidant tempol inhibits cardiac hypertrophy in the insulin-resistant GLUT4-deficient mouse in vivo. J Mol Cell Cardiol 2007; 42: 1119–1128 [DOI] [PubMed] [Google Scholar]
- 32.DeBosch B, Treskov I, Lupu TS, Weinheimer C, Kovacs A, Courtois M, Muslin AJ: Akt1 is required for physiological cardiac growth. Circulation 2006; 113: 2097–2104 [DOI] [PubMed] [Google Scholar]
- 33.Fiordaliso F, De Angelis N, Bai A, Cuccovillo I, Salio M, Serra DM, Bianchi R, Razzetti R, Latini R, Masson S: Effect of beta-adrenergic and renin-angiotensin system blockade on myocyte apoptosis and oxidative stress in diabetic hypertensive rats. Life Sci 2007; 81: 951–959 [DOI] [PubMed] [Google Scholar]
- 34.Yeshao W, Gu J, Peng X, Nairn AC, Nadler JL: Elevated glucose activates protein synthesis in cultured cardiac myocytes. Metab Clin Exp 2005; 54: 1453–1460 [DOI] [PubMed] [Google Scholar]
- 35.Neri Serneri GG, Boddi M, Modesti PA, Cecioni I, Coppo M, Padeletti L, Michelucci A, Colella A, Galanti G: Increased cardiac sympathetic activity and insulin-like growth factor-I formation are associated with physiological hypertrophy in athletes. Circ Res 2001; 89: 977–982 [DOI] [PubMed] [Google Scholar]
- 36.Fuller SJ, Mynett JR, Sugden PH: Stimulation of cardiac protein synthesis by insulin-like growth-factors. Biochem J 1992; 282: 85–90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Asbun J, Villarreal FJ: The pathogenesis of myocardial fibrosis in the setting of diabetic cardiomyopathy. J Am Coll Cardiol 2006; 47: 693–700 [DOI] [PubMed] [Google Scholar]
- 38.Candido R, Forbes JM, Thomas MC, Thallas V, Dean RG, Burns WC, Tikellis C, Ritchie RH, Twigg SM, Cooper ME, Burrell LM: A breaker of advanced glycation end products attenuates diabetes-induced myocardial structural changes. Circ Res 2003; 92: 785–792 [DOI] [PubMed] [Google Scholar]
- 39.Palmieri V, Capaldo B, Russo C, Iaccarino M, Pezzullo S, Quintavalle G, Di Minno G, Riccardi G, Celentano A: Uncomplicated type 1 diabetes and preclinical left ventricular myocardial dysfunction: insights from echocardiography and exercise cardiac performance evaluation. Diabetes Res Clin Pract 2008; 79: 262–268 [DOI] [PubMed] [Google Scholar]
- 40.Tsutsui H, Matsushima S, Kinugawa S, Ide T, Inoue N, Ohta Y, Yokota T, Hamaguchi S, Sunagawa K: Angiotensin II type 1 receptor blocker attenuates myocardial remodeling and preserves diastolic function in diabetic heart. Hypertens Res 2007; 30: 439–449 [DOI] [PubMed] [Google Scholar]
- 41.Matsushima S, Kinugawa S, Ide T, Matsusaka H, Inoue N, Ohta Y, Yokota T, Sunagawa K, Tsutsui H: Overexpression of glutathione peroxidase attenuates myocardial remodeling and preserves diastolic function in diabetic heart. Am J Physiol Heart Circ Physiol 2006; 291: H2237–H2245 [DOI] [PubMed] [Google Scholar]
- 42.Westermann D, Rutschow S, Van Linthout S, Linderer A, Bücker-Gärtner C, Sobirey M, Riad A, Pauschinger M, Schultheiss HP, Tschöpe C: Inhibition of p38 mitogen-activated protein kinase attenuates left ventricular dysfunction by mediating pro-inflammatory cardiac cytokine levels in a mouse model of diabetes mellitus. Diabetologia 2006; 49: 2507–2513 [DOI] [PubMed] [Google Scholar]
- 43.Wold LE, Ceylan-Isik AF, Fang CX, Yang X, Li SY, Sreejayan N, Privratsky JR, Ren J: Metallothionein alleviates cardiac dysfunction in streptozotocin-induced diabetes: role of Ca2+ cycling proteins, NADPH oxidase, poly(ADP-Ribose) polymerase and myosin heavy chain isozyme. Free Rad Biol Med 2006; 40: 1419–1429 [DOI] [PubMed] [Google Scholar]
- 44.Malhotra A, Begley R, Kang BP, Rana I, Liu J, Yang G, Mochly-Rosen D, Meggs LG: PKC-ϵ-dependent survival signals in diabetic hearts. Am J Physiol Heart Circ Physiol 2005; 289: H1343–H1350 [DOI] [PubMed] [Google Scholar]
- 45.Shioi T, Kang PM, Douglas PS, Hampe J, Yballe CM, Lawitts J, Cantley LC, Izumo S: The conserved phosphoinositide 3-kinase pathway determines heart size in mice. EMBO J 2000; 19: 2537–2548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.McMullen JR, Shioi T, Zhang L, Tarnavski O, Sherwood MC, Kang PM, Izumo S: Phosphoinositide 3-kinase(p110alpha) plays a critical role for the induction of physiological, but not pathological, cardiac hypertrophy. Proc Natl Acad Sci U S A 2003; 100: 12355–12360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bilim O, Takeishi Y, Kitahara T, Arimoto T, Niizeki T, Sasaki T, Goto K, Kubota I: Diacylglycerol kinase zeta inhibits myocardial atrophy and restores cardiac dysfunction in streptozotocin-induced diabetes mellitus. Cardiovasc Diabetol 2008; 7: 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sigal RJ, Kenny GP, Wasserman DH, Castaneda-Sceppa C, White RD: Physical activity/exercise and type 2 diabetes: a consensus statement from the American Diabetes Association. Diabetes Care 2006; 29: 1433–1438 [DOI] [PubMed] [Google Scholar]
- 49.Brassard P, Legault S, Garneau C, Bogaty P, Dumesnil JG, Poirier P: Normalization of diastolic dysfunction in type 2 diabetics after exercise training. Med Sci Sports Exerc 2007; 39: 1896–1901 [DOI] [PubMed] [Google Scholar]
- 50.Fossum E, Gleim GW, Kjeldsen SE, Kizer JR, Julius S, Devereux RB, Brady WE, Hille DA, Lyle PA, Dahlöf B: The effect of baseline physical activity on cardiovascular outcomes and new-onset diabetes in patients treated for hypertension and left ventricular hypertrophy: the LIFE study. J Intern Med 2007; 262: 439–448 [DOI] [PubMed] [Google Scholar]