Abstract
Clinical immunotherapy trials like dendritic cell-based vaccinations are hampered by the tumor's offensive repertoire that suppresses the incoming effector cells. Regulatory T cells are instrumental in suppressing the function of cytotoxic T cells. We studied the effect of low-dose cyclophosphamide on the suppressive function of regulatory T cells and investigated if the success rate of dendritic cell immunotherapy could be improved. For this, mesothelioma tumor-bearing mice were treated with dendritic cell-based immunotherapy alone or in combination with low-dose of cyclophosphamide. Proportions of regulatory T cells and the cytotoxic T cell functions at different stages of disease were analyzed. We found that low-dose cyclophosphamide induced beneficial immunomodulatory effects by preventing the induction of Tregs, and as a consequence, cytotoxic T cell function was no longer affected. Addition of cyclophosphamide improved immunotherapy leading to an increased median and overall survival. Future studies are needed to address the usefulness of this combination treatment for mesothelioma patients.
1. Introduction
Malignant mesothelioma (MM) is a cancer arising from mesothelial cells that lines the body's serous cavities (pleural, pericardial, and peritoneal) and the internal organs and is characterized by poor prognosis [1]. Chemotherapy or surgical treatments result in only limited improvements in response and survival. Novel therapeutic strategies are therefore needed. Immunotherapy is a promising but also challenging approach in the treatment of cancer.
Dendritic cells (DCs) are highly mobile antigen-presenting cells, capable of instructing and controlling the activation of NK cells, NKT cells, and B and T lymphocytes [2–4]. Previously we showed that DC-based immunotherapy in a murine MM model leads to protective immunity as well as regression of established tumors [5]. Currently we are investigating DC-based immunotherapy in MM patients. Although DC-vaccines are well tolerated by patients, further optimization is necessary to exploit the full potential of this therapeutic strategy [6].
It is becoming evident that immune suppression plays a crucial role in tumor progressing. Tumors secrete several mediators to recruit and/or activate suppressive cells. Regulatory T cells (Tregs) are prominent cells in this suppressive environment. These cells are instrumental in allowing a growing cancer to evade immunological attack by impairing T cell function [7]. Elevated levels of Tregs have been reported in many tumors and their presence predicts for poor survival. We have demonstrated previously the presence of Tregs within the tumors of MM patients [8]. It has also been described that Tregs are increased in the peripheral blood [9] and pleural effusions [10] of these patients.
Recent clinical studies have shown that low-dose cyclophosphamide (CTX) induces beneficial immunomodulatory effects in the context of active or adoptive immunotherapy [11–21]. CTX is widely used to treat various types of malignancies and some autoimmune disorders. It displays either immunosuppressive or immunopotentiating effects, depending on the dosage and the timing of drug administration [22]. Although the mechanisms underlying these modulations are not fully understood, low-dose CTX might prevent the development and functionality of the Tregs [23–27].
Van der Most et al. reported that CTX enhances the effectiveness of gemcitabine treatment in murine mesothelioma by reducing the amount of Tregs [28, 29]. Thereby they underline the immunogenic role of Tregs in the suppression of activated target cells. On the other hand, Jackaman et al. recently stated that Tregs are no potent regulators of antimesothelioma immunity in their murine model and that targeting of these cells will not improve results [30]. Taken together, the role of Tregs in mesothelioma is controversial and whether these cells will influence the outcome of immunotherapy is still unclear.
Here we investigated the effect of CTX on immunosuppression and the combination of CTX and DC-based immunotherapy was studied in a murine MM model. We found that CTX reduced the levels of Tregs and this induced beneficial immunomodulatory effects in the context of DC-based immunotherapy. These results anticipate that antitumor immune responses elicited by DC-based immunotherapy in humans might be improved by simultaneously depleting Tregs using low-dose CTX.
2. Methods
2.1. Animals and Cell Lines
Female 6–10 week old BALB/c (H-2d) mice (Harlan, Zeist, The Netherlands) were housed under pathogen-free conditions at the animal care facility of the Erasmus MC, Rotterdam. Experiments were approved by the local Ethical Committee for Animal Welfare and complied to the Guidelines for the Welfare of Animals in Experimental Neoplasia by the United Kingdom Coordinating Committee on Cancer Research (UKCCCR) and by the Code of Practice of the Dutch Veterinarian Inspection. The AB1 cell line, a mouse mesothelioma cell line, was kindly provided by Professor Bruce W.S. Robinson of the Queen Elizabeth II Medical Centre, Nedlands, Australia. The cell line was derived from tumors induced by Wittenoom Gorge crocidolite asbestos injected intraperitoneally into a BALB/c mouse [31]. Cells were cultured in RPMI 1640 medium containing 25 mM HEPES, Glutamax, 50 μg/ml gentamicin, and 5% (v/v) fetal bovine serum (FBS) (all obtained from GIBCO/Invitrogen, Breda, The Netherlands) in a humidified atmosphere at 37°C and 5% CO2, in air. AB1 cells were passaged once or twice a week to a new flask by treatment with 0.05% trypsin, 0.53 mM EDTA in phosphate buffered saline (PBS, all GIBCO/Invitrogen). The cell line was regularly tested and remained negative for mycoplasma contamination.
2.2. Reagents
Cyclophosphamide ([CTX], the generic name for Endoxan) was purchased from Baxter B.V., Utrecht, The Netherlands. A stock solution was prepared by dissolving 1 gram into 50 ml of PBS. It was further diluted in drinking water at a concentration of 0.13 mg/ml.
Every 4 days bottles were carefully examined (volume was determined) and replaced with fresh bottles containing 100 ml of drinking water or CTX-containing water. No changes in the drinking pattern of mice were observed between tumor-bearing mice with normal water or with CTX-containing drinking water (+/−3 ml per day per mouse). This equals approximately 20 mg CTX/kg body weight/day and is considered as a low-dose. Dehydration, being one of the points for signs of illness, was routinely checked. No signs of dehydration were observed during experiments except for a few cases with ill health or overt tumor growth.
2.3. Source of Tumor Antigen Derived from AB1 Tumor
AB1 cell line-derived tumor lysate was prepared from 50 × 106 cells suspended per ml PBS. The cell suspension was frozen in liquid nitrogen and disrupted by four cycles of freeze-thawing followed by sonication for 4 × 10 seconds with an amplitude of 10 microns, using a Soniprep 150 ultrasonic disintegrator equipped with a microtip (Sanyo Gallenkamp BV, Breda, The Netherlands) on ice. Cell lysate was aliquoted and stored at −80°C.
2.4. Culture Conditions of Bone Marrow-Derived DC Used for Vaccination
DCs were generated with only minor adaptations from a previously described protocol by Lutz [32]. After flushing femurs and tibias and red blood cell lysis, resulting bone marrow cells (2 × 106) were seeded in 100-mm Petri dishes (day 0) and cultured in 10 ml DC Culture Medium [DC-CM]: RPMI 1640 containing glutamax-I (GIBCO/Invitrogen) supplemented with 5% (v/v) FBS, 50 μM β-mercaptoethanol (Sigma-Aldrich), 50 μg/ml gentamicin (Invitrogen), and 20 ng/ml recombinant murine granulocyte macrophage-colony-stimulating factor [GM-CSF, kindly provided by Professor K. Thielemans, Free University Brussels, Belgium]. Cells were cultured at 37°C in a humidified atmosphere at 5% CO2, in air. At day 3, 10 ml of fresh DC-CM was added. On day 6, 10 ml of each plate was replaced with 10 ml of fresh DC-CM. After 8 days of culture, AB1 cell lysate was added to the DC cultures, to the equivalent of three AB1 cell-equivalents per DC. After 8 hours, 100 μg/ml LPS (E. coli 026:B6, Sigma-Aldrich) was added to the culture to allow complete maturation while incubated overnight. The next day, DCs were harvested by gentle pipetting and purified by Lympholyte-Mammal (Cedarlane, Hornby, ON, Canada) density gradient centrifugation, the interphase washed three times in PBS and resuspended at a concentration of 1 × 106 viable cells in 500 μl PBS. The quality of the DC preparation was determined by cell-counting, morphologic analysis and cell surface marker expression by flow cytometry, as previously described [5]. DCs (1 × 106) were delivered into the peritoneal cavity of BALB/c mice; control mice received 500 μl PBS.
2.5. Treatment with Tumor Lysate-Pulsed DCs and Cyclophosphamide on Outcome
Initial experiments were performed to determine the optimal concentration of CTX necessary to reduce Tregs in vivo. Continuous administration via the drinking water of low concentrations (“metronomic”) CTX had our preference because a single administration of a higher dose or multiple administrations i.p. engenders worse life expectancy as a consequence of peritoneal damage caused by the needle. Approximately 20 mg CTX/kg body weight/day in the drinking water of mice was the optimal concentration for further studies in mice, concentrations below 100 mg/kg are considered as a low dose. No effect on feeding, body weight, and physical well-being or other side effects were observed at this concentration.
On day 0, BALB/c mice (4 groups each consisting of 6 mice) were inoculated intraperitoneally with 0.5 × 106 AB1 tumor cells in 500 μl PBS. On day 12, 12 mice (2 groups) were vaccinated with 1 × 106 LPS-matured DCs pulsed with AB1 tumor cell line lysate in 500 μl PBS. From day 3 till day 10 and/or day 14 till day 21 mice were given drinking water supplemented with 0.13 mg/ml CTX. Mice were examined daily for evidence of ill health or overt tumor growth. Mice were killed if profoundly ill, according to UKCCCR regulations, and were scored as a death in survival analysis. All mice underwent extensive autopsy.
2.6. Immunohistology on Tumor Biopsies
Tumor biopsies were embedded in Tissue-Tek II optimum cutting temperature medium (Miles, Naperville, IL, USA), snap-frozen, and stored at −80°C. Tissue sections (6 μm) were cut on an HM-560 cryostat (Microm, Heidelberg, Germany) and immunostaining was carried out using a rat antimouse Foxp3 antibody (eBioscience [#14-5773], San Diego, CA, USA). Binding of antibody was detected using alkaline phosphatase- (AP-) conjugated goat antirat (Sigma-Aldrich Chemie B.V.). Naphtol-AS-MX-phosphate (0.30 mg·mL−1; Sigma-Aldrich Chemie B.V., Zwijndrecht, the Netherlands) and new fuchsine (160 mg·mL−1 in 2 M HCl; Chroma-Gesellschaft, Köngen, Germany) were used as substrate. The specificity was checked using a protein concentration-matched nonrelevant rat antibody and PBS. Double staining of Foxp3 and CD8 was performed using the AP-conjugated goat antirat followed by rabbit antiphycoerythrin (AbD Serotec, Düsseldorf, Germany) followed by incubation with horseradish peroxidase- (HRP-) conjugated swine antirabbit. Naphtol-AS-MX-phosphate and 1 mM Fast Blue substrate were used as substrate for AP, and NovaRed was used as substrate for HRP, according to the manufacturer's instructions (Vector, Burlingame, CA, USA). Foxp3 and phycoerythrin-labeled antimouse CD8 (BD Biosciences, San Jose, CA, USA) were both used at a dilution of 1 : 10. Signals were captured on a Leica microscope (Leica Microsystems B.V., Rijswijk, The Netherlands).
2.7. Preparation of Lymphocytes from Lymphoid Organs or Blood
Lymphocytes were collected from the spleens, lymph nodes, and blood of mice from each group. Briefly, spleens and lymph nodes were aseptically removed and mechanically dispersed in cold PBS. Cells suspensions were filtered through a 100 μm nylon mesh cell strainer (BD Biosciences, Bedford, MA, USA). Resulting suspensions and blood were depleted of erythrocytes by density gradient centrifugation, washed twice with PBS +2% BSA, and resuspended at a concentration of 1 × 106 cells/ml for flow cytometric analysis.
2.8. Flow Cytometry
The following antimouse antibodies were used: fluorescein isothiocyanate- (FITC-) conjugated anti-CD4 (1 : 800), anti-CD19 (1 : 200), phycoerythrin- (PE- )-conjugated anti-CD3 (1 : 25), anti-CD25 (1 : 200) (all from BD Biosciences), allophycocyanin- (APC)conjugated anti-Foxp3 (1 : 25) (eBioscience), and appropriate isotype-matched controls.
For cell surface marker staining, cells were washed with FACS-wash (0.05% NaN3, 2% BSA in PBS) and Fcγ II/III receptor blocking was performed using antimouse 2.4G2 antibody (1 : 100; kindly provided by L. Boon, Bioceros, Utrecht, The Netherlands) for 15 minutes on ice. After the blocking procedure, properly diluted antibodies for cell surface staining were added into each sample and placed on ice for 30 minutes protected from light. After two additional washes with FACS-wash, Fix/Perm buffer (eBiosience) was added and cells were incubated for 1 hour at 4°C in the dark. Cells were washed twice with Perm buffer. Intracellular staining was performed for Foxp3 according to the manufacturer's instructions (eBioscience). After blocking for 15 minutes with blocking antibody 2.4G2, properly diluted APC-conjugated Foxp3 antibody in Perm buffer was added and incubated for 30 minutes on ice protected from light. After washing the cells twice with Perm buffer, cells were washed with FACS-wash and measured. Data acquisition was performed by flow cytometry (LSR II; BD Biosciences) and data analysis was performed with FlowJo software (Tree Star, Inc.).
2.9. Coculture of Tregs and Activated Splenocytes
Splenocytes obtained from tumor-bearing mice were stained for CD3, CD4, CD8, and CD25. Four colour sample sorting was done on a FACS ARIA (BD Biosciences). Tregs were defined as CD3+CD4+CD25+bright cells and sorted with a purity >90% as determined by intracellular staining on Foxp3 expression. Splenocytes of DC-treated mice were cocultured with sorted Treg cells at different ratios. Splenocytes were restimulated for 4 hours using anti-CD3 and stained with mAbs directed against CD3, CD8, intracellular Granzyme B, and IFN-γ.
2.10. Statistical Analysis
Data are expressed as mean ± SD. Comparisons between groups were made using the Mann-Whitney U-test for independent samples. A two-tailed P-value <.05 was considered significant. Data presented as a percentage of tumor-free animals were analyzed with Kaplan-Meier survival curves, using the log-rank test to determine statistical significance.
3. Results
3.1. Regulatory T Cells in the Mouse Mesothelioma Model
The mesothelioma mouse model described in our previous study was used to examine the presence of CD4+CD25+Foxp3+ T cells in tumor progression [8]. In this protocol, BALB/c mice were intraperitoneally (i.p.) injected with a lethal dose of 0.5 × 106 AB1 tumor cells or 500 μl PBS as control. First signs of terminal illness (typically formation of ascites, ruffled hair, or marked loss of condition) appeared between 20 days and 30 days in tumor cell inoculated mice. Mice were sacrificed and subjected to extensive autopsy. All tumor inoculated mice showed solid tumor formation within the peritoneal cavity. The nature of these solid tumors varied from small nodules spreading throughout the mesentery and peritoneal lining to a single large mass. Tumor tissue, blood, spleen, and lymph nodes were collected from tumor-bearing mice for further analyses at day 10 or at first signs of terminal illness. No tissue abnormalities or formation of tumors could be detected in naive (PBS treated) mice.
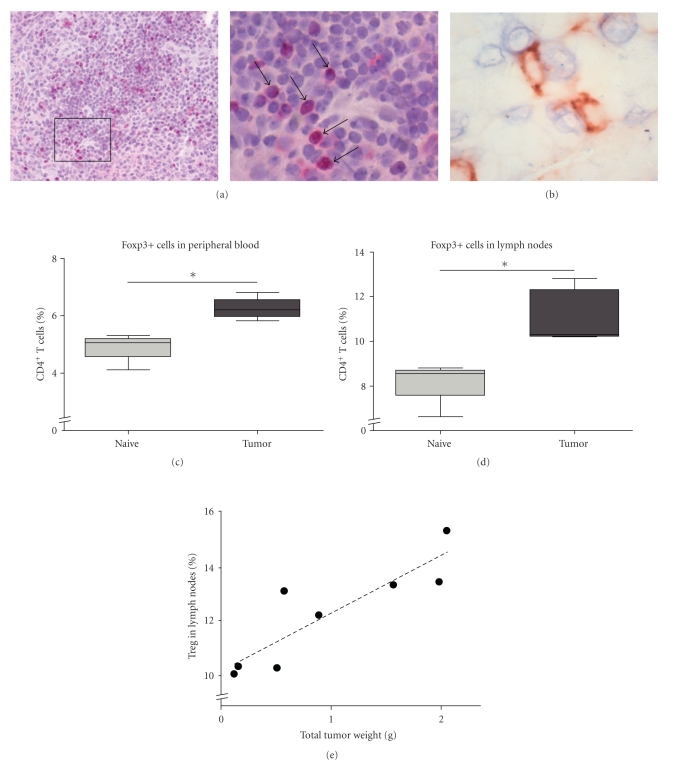
The presence of Tregs in mesothelioma tissue sections at day 10 was analyzed by immunohistochemistry for the phenotypic evidence of the transcription factor Foxp3, a hallmark of naturally arising CD4+CD25+ Treg cells [33–37]. Stainings confirmed that mouse tumor tissue obtained from AB1 inoculated mice contained significant amounts of Foxp3+ regulatory T cells, consisting with previous findings in human mesothelioma tissue [8]. Foxp3-expressing cells were located in the proximity of the tumor areas (Figure 1(a)). Double staining showed that the presence of these cells was occasionally in the vicinity of CD8+T cells, suggesting their possible direct suppressive role in antitumoral responses (Figure 1(b)).
Figure 1.
Increase in the proportion of regulatory T cells in a murine model for mesothelioma. (a) Left: The transcription factor Foxp3, a hallmark of naturally arising CD4+CD25+ Tregs, was expressed inside the tumor (red staining). Cells were counterstained with haematoxylin. [Magnification 200×] Right: A higher magnification (400×) shows the appearance of Foxp3+ cells as indicated by arrows. (b) Tregs (blue staining) were occasionally present in the near vicinity of CD8+ cells (red staining) [Magnification 1000×]. (c) and (d) Percentage of Tregs (defined as CD4+CD25+Foxp3+ cells) from total CD4+ T cells in the peripheral blood and draining lymph nodes is significantly higher at day 10 in tumor-bearing mice compared to healthy mice as observed by flow cytometry. (e) A positive correlation was found between the total tumor weight and the percentage of Tregs, measured in the lymph nodes of tumor-bearing mice. Five mice were euthanized at day 10 (tumor weight varied from 0.1 gram to 1 gram) and three mice at stage with ill health (tumor weight 1.5 to 2.1 gram). Correlation coefficient of the trend line (R-squared) is 0.780 (P value is .0039).
We then examined the blood and peripheral lymphoid organs for the presence of Tregs, determined by CD4, CD25, and Foxp3 positive expression using flow cytometry. In peripheral blood of control mice, the mean proportion of Treg cells was 4.9% of all CD4+ T cells. The percentage increased in the blood to 6.1% ten days after the inoculation of AB1 tumor cells (Figure 1(c)). Also the CD4+CD25+Foxp3+ Tregs were increased in the lymph nodes from 8.9% to 11.5% of the total CD4+ T cell population in tumor-bearing mice (Figure 1(d)). In the splenocytes from tumor-inoculated mice, the presence of Foxp3+ Tregs increased to 11% compared to 9% in the control group treated with PBS alone (data not shown).
To determine the impact of a growing tumor on Tregs, the percentage of Foxp3+ T cells in the draining lymph nodes was measured. Total weight of excised tumor tissue and the percentage of Tregs in the draining lymph nodes of corresponding mice are depicted in Figure 1(e). At ten days after AB1 inoculation (n = 5), the weight of the total tumor mass collected varied from 0.1 gram to 1 gram, or larger (1.5 to 2.1 gram) when mice showed evidence of ill health or overt tumor growth (n = 3).
In conclusion, Tregs were found within the tumor area and increased proportions of Tregs were found in the tumor-bearing mice. Moreover we show a significant increase in the proportion of Tregs that correlates with tumor burden (P = .0039).
3.2. Reduction of Tregs Improves Cytotoxic T Cell Function In Vivo
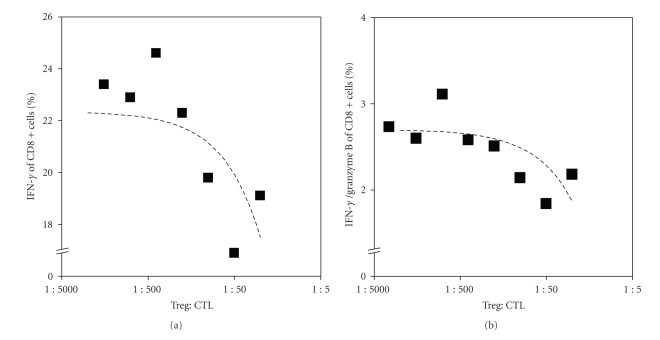
To demonstrate that cytotoxic T cell function is inhibited by Tregs, the intracellular expression of granzyme B and IFN-γ in activated CD8+T cells was measured. Tregs from tumor-bearing mice were isolated from spleen. These CD4+CD25+ T cells were then added in different ratios to the activated splenocytes isolated from DC-treated mice. Intracellular granzyme B expression by CD8+ T cells decreased in the fractions containing high amounts of CD4+CD25+ T cells. The same was observed for the IFN-γ expression (Figure 2). When CD4+CD25+ T cells are present in a ratio higher than 1 : 100, the function of CD8+ T cell is impaired.
Figure 2.
Tregs inhibit CTL function in vitro. (a) Activated splenocytes from DC-treated mice were cocultured with CD4+CD25+Foxp3+ cells (purified from tumor-bearing mice) in different ratios. The percentage of intracellular IFN-γ of CD8+ cells was measured using flow cytometry. And (b) the percentage of intracellular IFN-γ and granzyme B expression was measured using FACS. A reduction in the IFN-γ and granzyme B expression was found especially in those fractions where increasing doses of Treg were added to the CD8+ T cells.
Taken together, these data provide evidence that the addition of Tregs leads to impaired CD8 T cell activation.
3.3. Effects of Low-Dose Cyclophosphamide on the T and B Cell Populations
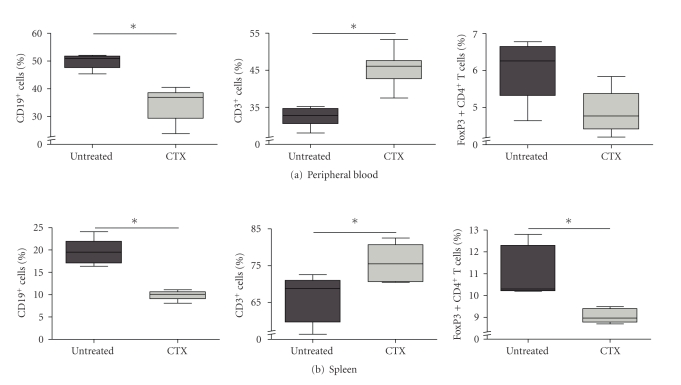
To examine the immunomodulating effects of CTX on Tregs, BALB/c mice were inoculated i.p. with 0.5 × 106 AB1 mesothelioma tumor cells and given drinking water with or without CTX. After 10 days peripheral blood and spleen were analyzed. Metronomic CTX treatment resulted in a significant decrease in the proportions of CD19+ B cells and an increase of CD3+ T cells (Figures 3(a) and 3(b)), in agreement with reports by others [13]. In contrast to the significant increase in the proportions of total CD3+ T cells, we observed a significant reduction in the fractions of Foxp3+ T cells (Figures 3(a) and 3(b)).
Figure 3.
CTX influences lymphocyte subsets in peripheral blood and draining lymph nodes of tumor-bearing mice. (a) The percentage of CD19+ cells was significantly decreased (P < .05) in CTX-treated tumor-bearing mice. CD3+ cells were significantly increased (P = .0325) in the peripheral blood of CTX-treated tumor-bearing mice compared to untreated tumor-bearing mice. (b) CD19+ cells were significantly decreased (P < .05) while CD3+ cells were significantly increased (P < .05), while the percentage of CD4+CD25+Foxp3+ cells was significantly decreased (P < .05) in the splenocytes of CTX-treated mice compared to untreated tumor-bearing mice.
In summary, these data show that metronomic administration of low-dose CTX has a strong immune-modulating effect in vivo, causing a shift in ratio between CD19+/CD3+ cells. Addition of CTX to drinking water of mice leads to a significant increase in the proportion of CD3+ T cells in the peripheral blood and the spleen, whereas the proportion of Tregs is reduced.
3.4. CTX Improves Suboptimal Dc-Based Immunotherapy
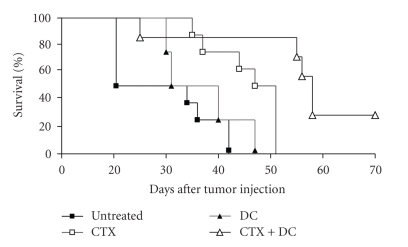
Next, we then investigated if combining CTX and DC vaccination enhanced the efficacy of the DC treatment as an antitumor treatment. In contrast to optimal DC-treatment protocols (as described by Hegmans et al. [5]), DCs are given at a relative late time point (day 12) in order to achieve suboptimal survival of DC-treated mice. Twelve days after i.p. tumor cell inoculation, mice were vaccinated with 1 × 106 LPS-matured DCs pulsed with AB1 tumor lysate. Mice that received CTX had an increased survival, as seen in our earlier experiments, compared to untreated mice.
When mice were given drinking water supplemented with 0.13 mg/ml CTX from day 3 till day 10 and day 14 till day 21, an increased survival was measured. However, the combination of DC-based immunotherapy and CTX administration significantly improved survival compared to DC-based immunotherapy (P < .0035) or CTX administration alone (P < .0056) (Figure 4).
Figure 4.
CTX combined with DC-based immunotherapy prolongs survival. Kaplan-Meier survival plot showing the effect of the combination of CTX and DC-based immunotherapy. Mice were given drinking water supplemented with 0.13 mg/ml CTX from day 3 till day 10 and day 14 till day 21. Tumor-lysate-pulsed DCs were given at day 12. Survival significantly improved when CTX and DC-based immunotherapy were combined compared to DC-based immunotherapy (P = .0035) or CTX administration alone (P = .0056). Each group contained 8 mice.
Therefore, we conclude that CTX is a powerful tool to optimize suboptimal DC-based immunotherapy. Although CTX alone also improves survival, the combination of both was significantly better.
3.5. Long-Term Administration of CTX Improves Survival Compared to Pre- or Postlmmunotherapy Treatment
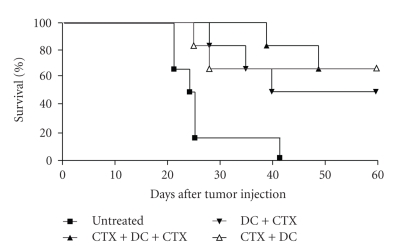
It has been reported that the timing of CTX administration is critical [38, 39], therefore we investigated the consequences of administration at different time points. To evaluate the effect of timing of CTX administration, mice were divided into three groups, receiving low-dose CTX before (day 3 till 10) or after (day 14 till 21) immunotherapy or long-term CTX (day 3 till 10 and day 14 till 21). Mice were treated with DC-immunotherapy on day 12. No significant differences were found between the groups (before [P = 037], after treatment [P = .84]). However long-term CTX administration may prolong the survival beneficial since no side effects of this low dose were observed (Figure 5).
Figure 5.
Long-period administration of CTX improves survival compared to pre- or post-immunotherapy treatment. Mice were divided into four groups (n = 6 per group) and inoculated with a lethal dose of AB1 tumor cells on day 0. Mice received low-dose CTX before (day 3 till 10) or after (day 14 till 21) immunotherapy or metronomic dosed CTX (day 3 till 10 and day 14 till 21). Groups 2, 3, and 4 were treated with DC-immunotherapy on day 12. Group 1 functioned as a tumor control group and did not receive any treatment. Administration of metronomic dosed CTX was not significantly better than CTX treatment before (P = .840) or after (P = .454) immunotherapy. However, the combination of CTX and immunotherapy was significantly better than no treatment (CTX before immunotherapy compared to untreated P = .0081, CTX after immunotherapy compared to untreated P = .0147, metronomic dosed CTX and immunotherapy compared to untreated P = .0018).
4. Discussion
Cancer immunotherapy attempts to harness the exquisite power and specificity of the immune system to recognize and destroy tumor cells or to prevent tumor recurrence. The finding that some patients with malignant pleural mesothelioma (MM) have tumors that regress spontaneously [40–43] or respond to immunotherapy [44–48] suggests that the immune system can generate antitumor reactivity under specific circumstances [41, 49].
DCs are extremely potent antigen-presenting cells specialized in inducing activation and proliferation of lymphocytes, which are essential for tumor killing [50]. Patient's own DCs can be used to present tumor-associated antigens and thereby generate tumor-specific immunity [3, 4]. Previously we used the mesothelioma mouse model to examine the impact of dendritic cell immunotherapy [5]. For malignant mesothelioma, as for most other cancer types, only a few tumor-associated antigens (TAAs) are known. These antigens are not expressed on the membranes of all tumors and therefore less suitable for antigen source for DC pulsing. Furthermore, none of these TAAs have been evaluated as a source of peptides to pulse DC or in a cancer vaccine trial. Tumor lysate priming strategies are advantageous in providing the full antigenic repertoire of the tumor and, particularly, unique individual tumor antigens, which will theoretically decrease the ability of tumors to evade the immune response by downregulation of a single antigen. Therefore, DCs were pulsed with autologous total tumor lysate fractions prepared by freeze thawing and sonication of AB1 tumor cells. Mice receiving tumor lysate-loaded DCs developed a protective antitumor immunity when animals were vaccinated before tumor inoculation. They showed no signs of tumor growth even after 3 months and after repeated injection of tumor cells (2nd tumor challenge). MM had a better outcome when DCs were injected early in tumor development indicating that tumor load played an important role in survival.
There are multiple levels to explain the limited benefit from DC vaccinations in mice with high tumor load. It has been suggested that immune suppression by tumor-derived factors is one of the main reasons for immunotherapy failure in general [7, 51]. Tumors escape immune recognition by attracting immune suppressive cells like Treg cells and myeloid-derived suppressor cells. Others and we have shown that mesothelioma cells are potent sources of a number of cytokines (e.g., IL-6, IL-8, VEGF, GRO, and RANTES) that might directly suppress immune activating cells, like DCs and/or recruit suppressive cells and thereby abolish an efficient immune response [8]. It has become evident that while protecting the host against cancer development, the immune cells also promote the emergence of tumors with reduced immunogenicity leading to a complex interplay of tumor growth and tumor regression mechanisms.
Like in human mesothelioma biopsies, a growing AB1 tumor in vivo contains significant amounts of CD4+CD25+Foxp3+ regulatory Tcells, which were previously shown to promote tumor progression in other cancer models. Exact mechanisms are not yet fully understood; however one of the mechanisms by which Tregs can abolish the effectiveness of immunotherapy may be by their capacity to produce granzyme B. This can lead to killing of cytotoxic T cells [52–54]. Depletion of Tregs by using a blocking anti-CD25 antibody (PC61), capable of specifically binding to the IL-2 receptor α-chain, led to an increased survival in mice; however, this depleting antibody is not suitable for human clinical use. Cyclophosphamide (CTX) is an alkylating cytotoxic cancer drug that, depending on its dose and timing of administration, has been used as a chemotherapeutic and disease-modifying agent or to enhance immune responses [14, 26, 27, 55–57]. Reports dating from the 1980s have shown that under some conditions low-dose CTX can potentiate antitumor immunity in mouse models [55]. Recently it has been suggested that this effect is caused by the depletion of Tregs [39]. Reductions in the amount of Tregs by using CTX were also found in mesothelioma [28]. In addition, Van der Most et al. showed that the combination of CTX and gemcitabine eradicates established murine mesothelioma whereas single treatment with gemcitabine did not lead to total tumor regression [29]. However the benefit of combining Treg depletion with DC-based immunotherapy for mesothelioma was not established till now.
CD4+CD25+Foxp3+ regulatory T cells comprise between 5 and 10% of the total CD4+ population in the blood of mice, and are important in controlling tolerance to self-antigens and thereby maintain immune system homeostasis [54, 58]. We found that the prevalence of Tregs increases in tumor-bearing mice in the tumor, peripheral blood, and lymph nodes (Figures 1 and 3) as compared with normal controls. Prolonged, lower-dose (“metronomic”) CTX inhibits the rise of Tregs (Figure 3). This effect augments the efficacy of dendritic cell-based immunotherapy (Figures 4 and 5). DC-based immunotherapy leads to an increase in survival that is further improved by depleting Treg cells.
5. Conclusion
We found that CTX is an applicable agent to reduce the proportion of Tregs in tumor-bearing mice. In addition, we showed that combining CTX and DC-based immunotherapy significantly prolongs the mean and overall survival in murine model for mesothelioma. We anticipate that CTX allows the host immune system to overcome the immunosuppressive mechanisms of Tregs, thereby allowing for a more complete and robust antitumor immune response initiated by DC-vaccination that is paramount to eliminate the tumor in vivo. Our findings suggest that CTX may also potentiate the immunogenicity of DC-immunotherapy in mesothelioma patients. In fact, these findings now form the basis of a new trial aiming the depletion of Tregs by CTX in combination with DC-based immunotherapy as a multimodality treatment in mesothelioma patients after chemotherapy.
Conflict of Interest
None of the listed authors on this paper had any contact with people or organizations that could inappropriately influence (bias) their work.
Acknowledgments
This study was funded by the “Stichting Asbestkanker Rotterdam” and Stichting Coolsingel.
Non-Standard Abbreviations
- CTL(s):
Cytotoxic T lymphocyte(s)
- CTX:
Cyclophosphamide
- MM:
Malignant mesothelioma
- DC(s):
Dendritic cell(s)
- Treg(s):
Regulatory T cell(s).
References
- 1.Robinson BWS, Musk AW, Lake RA. Malignant mesothelioma. The Lancet. 2005;366(9483):397–408. doi: 10.1016/S0140-6736(05)67025-0. [DOI] [PubMed] [Google Scholar]
- 2.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392(6673):245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 3.Banchereau J, Palucka AK. Dendritic cells as therapeutic vaccines against cancer. Nature Reviews Immunology. 2005;5(4):296–306. doi: 10.1038/nri1592. [DOI] [PubMed] [Google Scholar]
- 4.Steinman RM, Dhodapkar M. Active immunization against cancer with dendritic cells: the near future. International Journal of Cancer. 2001;94(4):459–473. doi: 10.1002/ijc.1503. [DOI] [PubMed] [Google Scholar]
- 5.Hegmans JPJJ, Hemmes A, Aerts JG, Hoogsteden HC, Lambrecht BN. Immunotherapy of murine malignant mesothelioma using tumor lysate-pulsed dendritic cells. American Journal of Respiratory and Critical Care Medicine. 2005;171(10):1168–1177. doi: 10.1164/rccm.200501-057OC. [DOI] [PubMed] [Google Scholar]
- 6.Figdor CG, de Vries IJM, Lesterhuis WJ, Melief CJM. Dendritic cell immunotherapy: mapping the way. Nature Medicine. 2004;10(5):475–480. doi: 10.1038/nm1039. [DOI] [PubMed] [Google Scholar]
- 7.Sakaguchi S, Sakaguchi N, Shimizu J, et al. Immunologic tolerance maintained by CD25+CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunological Reviews. 2001;182:18–32. doi: 10.1034/j.1600-065x.2001.1820102.x. [DOI] [PubMed] [Google Scholar]
- 8.Hegmans JPJJ, Hemmes A, Hammad H, Boon L, Hoogsteden HC, Lambrecht BN. Mesothelioma environment comprises cytokines and T-regulatory cells that suppress immune responses. European Respiratory Journal. 2006;27(6):1086–1095. doi: 10.1183/09031936.06.00135305. [DOI] [PubMed] [Google Scholar]
- 9.Meloni F, Morosini M, Solari N, et al. Foxp3 expressing CD4+CD25+ and CD8+CD8− T regulatory cells in the peripheral blood of patients with lung cancer and pleural mesothelioma. Human Immunology. 2006;67(1-2):1–12. doi: 10.1016/j.humimm.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 10.DeLong P, Carroll RG, Henry AC, et al. Regulatory T cells and cytokines in malignant pleural effusions secondary to mesothelioma and carcinoma. Cancer Biology and Therapy. 2005;4(3):342–346. doi: 10.4161/cbt.4.3.1644. [DOI] [PubMed] [Google Scholar]
- 11.Berd D, Mastrangelo MJ. Active immunotherapy of human melanoma exploiting the immunopotentiating effects of cyclophosphamide. Cancer Investigation. 1988;6(3):337–349. doi: 10.3109/07357908809080657. [DOI] [PubMed] [Google Scholar]
- 12.Mescher MF, Rogers JD. Immunotherapy of established murine tumors with large multivalent immunogen and cyclophosphamide. Journal of Immunotherapy. 1996;19(2):102–112. doi: 10.1097/00002371-199603000-00003. [DOI] [PubMed] [Google Scholar]
- 13.Liu J-Y, Wu Y, Zhang X-S, et al. Single administration of low dose cyclophosphamide augments the antitumor effect of dendritic cell vaccine. Cancer Immunology, Immunotherapy. 2007;56(10):1597–1604. doi: 10.1007/s00262-007-0305-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Taieb J, Chaput N, Schartz N, et al. Chemoimmunotherapy of tumors: cyclophosphamide synergizes with exosome based vaccines. Journal of Immunology. 2006;176(5):2722–2729. doi: 10.4049/jimmunol.176.5.2722. [DOI] [PubMed] [Google Scholar]
- 15.Goldfarb RH, Ohashi M, Brunson KW, et al. Augmentation of IL-2 activated natural killer cell adoptive immunotherapy with cyclophosphamide. Anticancer Research. 1998;18(3):1441–1446. [PubMed] [Google Scholar]
- 16.Proietti E, Greco G, Garrone B, et al. Importance of cyclophosphamide-induced bystander effect on T cells for a successful tumor eradication in response to adoptive immunotherapy in mice. Journal of Clinical Investigation. 1998;101(2):429–441. doi: 10.1172/JCI1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mihalyo MA, Doody ADH, McAleer JP, et al. In vivo cyclophosphamide and IL-2 treatment impedes self-antigen-induced effector CD4 cell tolerization: implications for adoptive immunotherapy. Journal of Immunology. 2004;172(9):5338–5345. doi: 10.4049/jimmunol.172.9.5338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li L, Okino T, Sugie T, et al. Cyclophosphamide given after active specific immunization augments antitumor immunity by modulation of Th1 commitment of CD4+ T cells. Journal of Surgical Oncology. 1998;67(4):221–227. doi: 10.1002/(sici)1096-9098(199804)67:4<221::aid-jso3>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- 19.Hermans IF, Chong TW, Palmowski MJ, Harris AL, Cerundolo V. Synergistic effect of metronomic dosing of cyclophosphamide combined with specific antitumor immunotherapy in a murine melanoma model. Cancer Research. 2003;63(23):8408–8413. [PubMed] [Google Scholar]
- 20.North RJ. Cyclophosphamide-facilitated adoptive immunotherapy of an established tumor depends on elimination of tumor-induced suppressor T cells. Journal of Experimental Medicine. 1982;155(4):1063–1074. doi: 10.1084/jem.155.4.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Salem ML, Kadima AN, El-Naggar SA, et al. Defining the ability of cyclophosphamide preconditioning to enhance the antigen-specific CD8+ T-cell response to peptide vaccination: creation of a beneficial host microenvironment involving type I IFNs and myeloid cells. Journal of Immunotherapy. 2007;30(1):40–53. doi: 10.1097/01.cji.0000211311.28739.e3. [DOI] [PubMed] [Google Scholar]
- 22.Nowak AK, Lake RA, Robinson BWS. Combined chemoimmunotherapy of solid tumours: improving vaccines? Advanced Drug Delivery Reviews. 2006;58(8):975–990. doi: 10.1016/j.addr.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 23.Ghiringhelli F, Menard C, Puig PE, et al. Metronomic cyclophosphamide regimen selectively depletes CD4+CD25+ regulatory T cells and restores T and NK effector functions in end stage cancer patients. Cancer Immunology, Immunotherapy. 2007;56(5):641–648. doi: 10.1007/s00262-006-0225-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Motoyoshi Y, Kaminoda K, Saitoh O, et al. Different mechanisms for anti-tumor effects of low- and high-dose cyclophosphamide. Oncology Reports. 2006;16(1):141–146. [PubMed] [Google Scholar]
- 25.Ikezawa Y, Nakazawa M, Tamura C, Takahashi K, Minami M, Ikezawa Z. Cyclophosphamide decreases the number, percentage and the function of CD25+CD4+ regulatory T cells, which suppress induction of contact hypersensitivity. Journal of Dermatological Science. 2005;39(2):105–112. doi: 10.1016/j.jdermsci.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 26.Lutsiak MEC, Semnani RT, De Pascalis R, Kashmiri SVS, Schlom J, Sabzevari H. Inhibition of CD4+25+ T regulatory cell function implicated in enhanced immune response by low-dose cyclophosphamide. Blood. 2005;105(7):2862–2868. doi: 10.1182/blood-2004-06-2410. [DOI] [PubMed] [Google Scholar]
- 27.Ghiringhelli F, Larmonier N, Schmitt E, et al. CD4+CD25+ regulatory T cells suppress tumor immunity but are sensitive to cyclophosphamide which allows immunotherapy of established tumors to be curative. European Journal of Immunology. 2004;34(2):336–344. doi: 10.1002/eji.200324181. [DOI] [PubMed] [Google Scholar]
- 28.van der Most RG, Currie AJ, Mahendran S, et al. Tumor eradication after cyclophosphamide depends on concurrent depletion of regulatory T cells: a role for cycling TNFR2-expressing effector-suppressor T cells in limiting effective chemotherapy. Cancer Immunology, Immunotherapy. 2009;58(8):1219–1228. doi: 10.1007/s00262-008-0628-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.van der Most RG, Currie AJ, Mahendran S, et al. Tumor eradication after cyclophosphamide depends on concurrent depletion of regulatory T cells: a role for cycling TNFR2-expressing effector-suppressor T cells in limiting effective chemotherapy. Cancer Immunology, Immunotherapy. 2009;58(8):1219–1228. doi: 10.1007/s00262-008-0628-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jackaman C, Cornwall S, Lew AM, Zhan Y, Robinson BWS, Nelson DJ. Local effector failure in mesothelioma is not mediated by CD4+CD25+ T-regulator cells. European Respiratory Journal. 2009;34(1):162–175. doi: 10.1183/09031936.00101008. [DOI] [PubMed] [Google Scholar]
- 31.Davis MR, Manning LS, Whitaker D, Garlepp MJ, Robinson BWS. Establishment of a murine model of malignant mesothelioma. International Journal of Cancer. 1992;52(6):881–886. doi: 10.1002/ijc.2910520609. [DOI] [PubMed] [Google Scholar]
- 32.Lutz MB, Kukutsch N, Ogilvie ALJ, et al. An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. Journal of Immunological Methods. 1999;223(1):77–92. doi: 10.1016/s0022-1759(98)00204-x. [DOI] [PubMed] [Google Scholar]
- 33.Miyara M, Sakaguchi S. Natural regulatory T cells: mechanisms of suppression. Trends in Molecular Medicine. 2007;13(3):108–116. doi: 10.1016/j.molmed.2007.01.003. [DOI] [PubMed] [Google Scholar]
- 34.Sakaguchi S. Regulatory T cells. Springer Seminars in Immunopathology. 2006;28(1):1–2. doi: 10.1007/s00281-006-0043-2. [DOI] [PubMed] [Google Scholar]
- 35.Yamaguchi T, Sakaguchi S. Regulatory T cells in immune surveillance and treatment of cancer. Seminars in Cancer Biology. 2006;16(2):115–123. doi: 10.1016/j.semcancer.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 36.Nishikawa H, Kato T, Tawara I, et al. Definition of target antigens for naturally occurring CD4+CD25+ regulatory T cells. Journal of Experimental Medicine. 2005;201(5):681–686. doi: 10.1084/jem.20041959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sakaguchi S. Immunologic tolerance maintained by regulatory T cells: implications for autoimmunity, tumor immunity and transplantation tolerance. Vox Sanguinis. 2002;83(supplement 1):151–153. doi: 10.1111/j.1423-0410.2002.tb05291.x. [DOI] [PubMed] [Google Scholar]
- 38.Machiels J-PH, Reilly RT, Emens LA, et al. Cyclophosphamide, doxorubicin, and paclitaxel enhance the antitumor immune response of granulocyte/macrophage-colony stimulating factor-secreting whole-cell vaccines in HER-2/neu tolerized mice. Cancer Research. 2001;61(9):3689–3697. [PubMed] [Google Scholar]
- 39.Wada S, Yoshimura K, Hipkiss EL, et al. Cyclophosphamide augments antitumor immunity: studies in an autochthonous prostate cancer model. Cancer Research. 2009;69(10):4309–4318. doi: 10.1158/0008-5472.CAN-08-4102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Maesen F, Willighagen R. Regression of a malignant tumour of the pleura. European Journal of Respiratory Diseases. 1987;71(2):135–138. [PubMed] [Google Scholar]
- 41.Robinson BWS, Robinson C, Lake RA. Localised spontaneous regression in mesothelioma—possible immunological mechanism. Lung Cancer. 2001;32(2):197–201. doi: 10.1016/s0169-5002(00)00217-8. [DOI] [PubMed] [Google Scholar]
- 42.Schwartz E, Maayan C, Mouallem M, Engelberg S, Friedman E. Malignant peritoneal mesothelioma: long-term spontaneous clinical remission. Medical and Pediatric Oncology. 1991;19(4):325–328. doi: 10.1002/mpo.2950190420. [DOI] [PubMed] [Google Scholar]
- 43.Pilling JE, Nicholson AG, Harmer C, Goldstraw P. Prolonged survival due to spontaneous regression and surgical excision of malignant mesothelioma. Annals of Thoracic Surgery. 2007;83(1):314–315. doi: 10.1016/j.athoracsur.2006.05.070. [DOI] [PubMed] [Google Scholar]
- 44.Yanagawa H, Sone S, Fukuta K, Nishioka Y, Ogura T. Local adoptive immunotherapy using lymphokine-activated killer cells and interleukin-2 against malignant pleural mesothelioma: report of two cases. Japanese Journal of Clinical Oncology. 1991;21(5):377–383. [PubMed] [Google Scholar]
- 45.Bielefeldt-Ohmann H, Marzo AL, Himbeck RP, Jarnicki AG, Robinson BWS, Fitzpatrick DR. Interleukin-6 involvement in mesothelioma pathobiology: inhibition by interferon α immunotherapy. Cancer Immunology Immunotherapy. 1995;40(4):241–250. doi: 10.1007/BF01519898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Astoul P, Picat-Joossen D, Viallat J-R, Boutin C. Intrapleural administration of interleukin-2 for the treatment of patients with malignant pleural mesothelioma: a phase II study. Cancer. 1998;83(10):2099–2104. doi: 10.1002/(sici)1097-0142(19981115)83:10<2099::aid-cncr8>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 47.Caminschi I, Venetsanakos E, Leong CC, Garlepp MJ, Robinson BWS, Scott B. Cytokine gene therapy of mesothelioma: immune and antitumor effects of transfected interleukin-12. American Journal of Respiratory Cell and Molecular Biology. 1999;21(3):347–356. doi: 10.1165/ajrcmb.21.3.3575. [DOI] [PubMed] [Google Scholar]
- 48.Kruklitis RJ, Singhal S, Delong P, et al. Immuno-gene therapy with interferon-β before surgical debulking delays recurrence and improves survival in a murine model of malignant mesothelioma. Journal of Thoracic and Cardiovascular Surgery. 2004;127(1):123–130. doi: 10.1016/j.jtcvs.2003.08.034. [DOI] [PubMed] [Google Scholar]
- 49.Robinson C, Callow M, Stevenson S, Scott B, Robinson BWS, Lake RA. Serologic responses in patients with malignant mesothelioma. Evidence for both public and private specificities. American Journal of Respiratory Cell and Molecular Biology. 2000;22(5):550–556. doi: 10.1165/ajrcmb.22.5.3930. [DOI] [PubMed] [Google Scholar]
- 50.Banchereau J, Briere F, Caux C, et al. Immunobiology of dendritic cells. Annual Review of Immunology. 2000;18:767–811. doi: 10.1146/annurev.immunol.18.1.767. [DOI] [PubMed] [Google Scholar]
- 51.Nagaraj S, Gabrilovich DI. Tumor escape mechanism governed by myeloid-derived suppressor cells. Cancer Research. 2008;68(8):2561–2563. doi: 10.1158/0008-5472.CAN-07-6229. [DOI] [PubMed] [Google Scholar]
- 52.Grossman WJ, Verbsky JW, Barchet W, Colonna M, Atkinson JP, Ley TJ. Human T regulatory cells can use the perforin pathway to cause autologous target cell death. Immunity. 2004;21(4):589–601. doi: 10.1016/j.immuni.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 53.Cao X, Cai SF, Fehniger TA, et al. Granzyme B and perforin are important for regulatory T cell-mediated suppression of tumor clearance. Immunity. 2007;27(4):635–646. doi: 10.1016/j.immuni.2007.08.014. [DOI] [PubMed] [Google Scholar]
- 54.Darrasse-Jèze G, Bergot A-S, Durgeau A, et al. Tumor emergence is sensed by self-specific CD44hi memory Tregs that create a dominant tolerogenic environment for tumors in mice. Journal of Clinical Investigation. 2009;119(9):2648–2662. doi: 10.1172/JCI36628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Awwad M, North RJ. Cyclophosphamide-induced immunologically mediated regression of a cyclophosphamide-resistant murine tumor: a consequence of eliminating precursor L3T4+ suppressor T-cells. Cancer Research. 1989;49(7):1649–1654. [PubMed] [Google Scholar]
- 56.Ercolini AM, Ladle BH, Manning EA, et al. Recruitment of latent pools of high-avidity CD8+ T cells to the antitumor immune response. Journal of Experimental Medicine. 2005;201(10):1591–1602. doi: 10.1084/jem.20042167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Xu L, Xu W, Jiang Z, Zhang F, Chu Y, Xiong S. Depletion of CD4+CD25high regulatory T cells from tumor infiltrating lymphocytes predominantly induces Th1 type immune response in vivo which inhibits tumor growth in adoptive immunotherapy. Cancer Biology and Therapy. 2009;8(1):66–72. doi: 10.4161/cbt.8.1.7131. [DOI] [PubMed] [Google Scholar]
- 58.Hori S, Sakaguchi S. Foxp3: a critical regulator of the development and function of regulatory T cells. Microbes and Infection. 2004;6(8):745–751. doi: 10.1016/j.micinf.2004.02.020. [DOI] [PubMed] [Google Scholar]