Abstract
Background and Purpose
Telemedicine techniques can be employed to address the rural-metropolitan disparity in acute stroke care. The STRokE DOC trial reported more accurate decision making for telemedicine consultations compared with telephone-only, and that the California based research network facilitated a high rate of thrombolysis use, improved data collection, low risk of complications, low technical complications, and favorable assessment times.
The main objective of the STRokE DOC AZ TIME trial was to determine the feasibility of establishing, de novo, a single-hub, multi-rural spoke hospital telestroke research network across a large geographical area in Arizona by replicating the STRokE DOC protocol.
Methods
Prospective, single hub, two spoke, randomized, blinded, controlled trial of a 2-way, site independent, audiovisual telemedicine system designed for remote examination of adult patients with acute stroke versus telephone consultation to assess eligibility for treatment with intravenous thrombolysis. The primary outcome measure was whether the decision to give thrombolysis was correct. Secondary outcomes were rate of thrombolytic use, 90-day functional outcomes, incidence of intracerebral hemorrhages, and technical observations.
Results
From December 2007 to October 2008, 54 patients were assessed; 27 randomized to each arm. Mean National Institutes of Health stroke scale score at presentation was 7.3 (SD 6.2) points. No consultations were aborted, however technical problems (74%) were prevalent in the telemedicine arm. Overall, the correct treatment decision was established in 87% of the consultations. Both modalities, telephone (89% correct) and telemedicine (85% correct) performed well. Intravenous thrombolytic treatment was used in 30% of the telemedicine and telephone consultations. Good functional outcomes at 90 days were not significantly different. There were no statistically significant differences in mortality (4% in telemedicine and 11% in telephone) or rates of intracerebral hemorrhage (4% in telemedicine and 0% in telephone).
Conclusions
It is feasible to extend the original STRokE DOC trial protocol to a new state and establish an operational single hub, multi-spoke rural hospital telestroke research network in Arizona. The trial was not designed to have sufficient power to detect a difference between the two consultative modes, telemedicine and telephone-only. Whether by telemedicine or telephone consultative modalities, there were appropriate treatment decisions, high rates of thrombolysis use, improved data collection, low rates of intracerebral hemorrhage, and equally favorable time requirements. The learning curve was steep for the hub and spoke personnel of the new telestroke network as reflected by the frequent technical problems. Overall, the results support the effectiveness of highly organized and structured stroke telemedicine networks for extending expert stroke care into rural remote communities lacking sufficient neurological expertise.
Keywords: Stroke, telemedicine, rural hospitals, rural health, state government, randomized controlled trials
Introduction
Rural communities endure a shortage of stroke specialist care 1, 2. Telemedicine techniques can be implemented within stroke networks of care to address the rural-metropolitan disparity which exists 3, 4. The Stroke Team Remote Evaluation using a Digital Observation Camera (STRokE DOC) trial 5 reported both that stroke telemedicine (audio-video) consultations resulted in more accurate thrombolysis eligibility decision making compared with telephone-only consultations, and that the University of California San Diego (UCSD) based research network facilitated a high rate of thrombolysis use, improved data collection, low risk of complications, low technical complications, and favorable assessment times.
The main objective of the STRokE DOC Arizona - The Initial Mayo Clinic Experience (TIME) trial was to determine the feasibility of establishing, de novo, a single-hub, multi-rural spoke hospital telestroke research network in Arizona by replicating the UCSD STRokE DOC protocol. Additionally, the purpose was to determine the effectiveness of telemedicine and telephone-only consultation modes for decision making in acute stroke in a different state amongst different hospitals and providers.
Methods
Patients
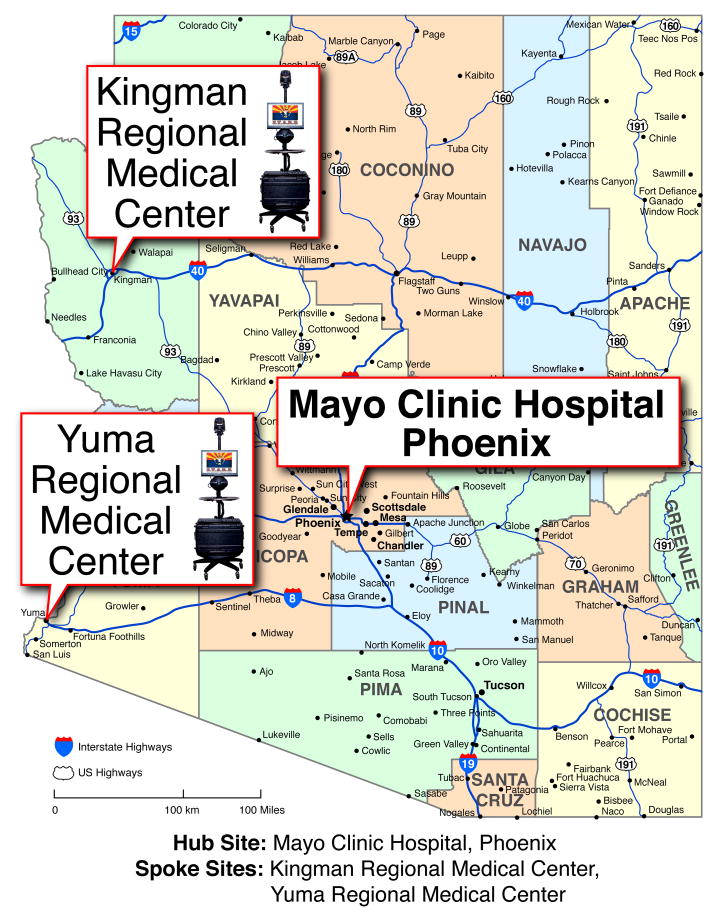
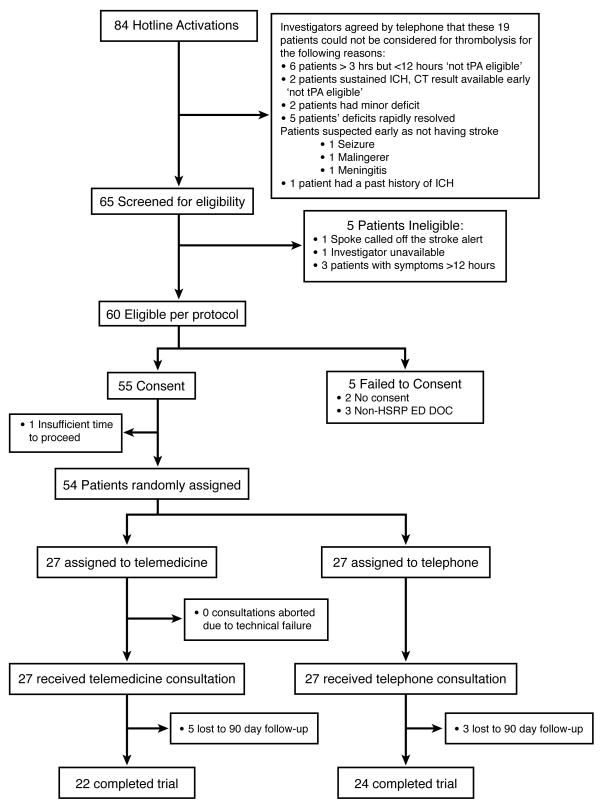
Between December 2007 and October 2008 the dedicated STRokE DOC AZ TIME Trial alpha-numeric group pager hotline was activated 84 times. Seventy-nine patients were determined to be potentially eligible, 55 patients were consented, and 54 were enrolled in the study and underwent random assignment to either telemedicine or telephone-only consultations when they presented with an acute stroke syndrome at one of two remote sites (spokes) that were located 186 to 205 miles (299 to 330 kilometers), respectively, from an academic metropolitan Joint Commission certified primary stroke center hub hospital. (Refer to figure 2) The trial flow figure (figure 1) describes the reasons patients were determined to be ineligible, the reasons why investigators elected not to consent, and the instances when either no surrogates or no certified spoke investigators were available for consent. The typical reasons that investigators elected not to proceed with consenting of otherwise eligible patients were ultra-early emergency medicine and vascular neurologist investigator collaborative perception that the patient was not eligible for intravenous thrombolysis, (for example time confirmed to exceed 3 hours, rapid resolution of focal neurological symptoms, stroke mimic, intracerebral hemorrhage reported on CT, minor persistent focal neurological deficits, and past history of intracerebral hemorrhage). This trial was undertaken prior to publication of the American Heart Association Scientific Statement concerning a 4½ hour expanded time window 6. We authorized a standard of clinical practice telephone-only run-in period of one month duration prior to trial start date while we acquired the STRokE DOC camera equipment and conducted the necessary hub and spoke training. This pre-trial period allowed time for spoke emergency medicine and hub vascular neurology investigators to familiarize themselves with the hotline activation protocol, the trial protocol, and each other. Trial inclusion criteria were age at least 18 years, ability for patient or surrogate to sign consent, symptoms and signs consistent with an acute stroke syndrome, and onset < 3 hours. Prisoners were excluded from enrollment. Written, informed consent was obtained at the spoke by emergency physician investigator and sent to the hub vascular neurology investigator by electronic fax (e-fax) for final verification prior to randomization.
Figure 2.
Figure 1.
Trial Flow
The trial was approved by each participating spoke hospital institutional review board and also by Mayo Clinic IRB, with authorization for central oversight. The trial was registered at ClinicalTrials.gov, number NCT00623350.
Procedures
A description of how the early Arizona hub and spoke telestroke network was established and descriptions of the technological factors, information technology, security, data encryption, the STRokE DOC camera system and technique, and prospective reliability have already been published 3, 5, 7–9.
The hub stroke team was contacted directly by the STRokE DOC AZ TIME trial alpha-numeric group pager system when a patient with acute stroke symptoms presented at the spoke emergency department. The designated on-call hub vascular neurologist telephoned the applicable spoke emergency department and spoke briefly with the spoke emergency physician in order to determine patient eligibility status. The published STRokE DOC trial protocol 5 was replicated. Eligible consented patients were randomized with permuted blocks that were stratified by study site to prevent group imbalances. The randomization to telemedicine or telephone-only consultation was performed in real-time using a web-based system, which eliminated investigator preference bias. For patients who were randomized to telemedicine, the consultation started with site-independent access to the telemedicine system. The hub consultant established audio and video contact with the spoke site and immediately acquired a medical history from patient and all accompanying relatives, supplemented by verbal and written reports from emergency medical systems (EMS), physicians, and nursing staff. Following the history acquisition, the hub consultant performed a National Institutes of Health stroke scale (NIHSS) examination with the aid of healthcare provider staff at the spoke site. Other relevant elements of the examination and were performed by, or reported to, the hub consultant as appropriate. Diagnostic test results were reported to the hub consultant by the spoke emergency physician, either verbally or by e-fax. Head CT images were viewed by hub neurologist with digital imaging and communications in medicine (DICOM) viewer. For patients who were randomized to telephone-only consultation, the hub consultant queried the spoke emergency physician about the history, physical examination including the NIHSS exam, the diagnostic test results, and the spoke radiologist’s report of the CT. The telephone communication with the emergency physician was supplemented, whenever feasible, by direct telephone communication between hub consultant and patient or surrogate. Neither the video nor the head CT images were viewed by the hub consultant with telephone only consultations.
In both consultative modalities, the hub consultant completed a pre-specified case report form. The consultant was free to repeat, or request a repeated, examination item and could speak with patient, relatives, witnesses, and emergency nurses. Clinical deficit and functional scale scores (including the NIHSS and prestroke and poststroke modified Rankin scale (mRS) score) were calculated by the hub consultant with the information provided by the bedside emergency physician or other healthcare providers. After review of the history, the examination findings, stroke scales, and head CT interpretation, the hub consultant presented a recommendation regarding patient eligibility for intravenous thrombolysis to the spoke emergency physician.
The main objective of the trial was to establish the feasibility of establishing a single hub, multi-rural spoke hospital telestroke research network in Arizona by replicating the STRokE DOC trial protocol in a new state, new hub, new spokes, amongst telemedicine naive practitioners.
An additional objective was to further assess the efficacy of telephone and telemedicine consultations for decision making. The outcome measure was whether the decision to give thrombolytic treatment was appropriate, as established with a rigorous multistage, masked adjudication process, the details of which have been published 9. Additional outcomes were rates of thrombolytic use, 90-day outcomes, rates of intracerebral hemorrhage, the completeness of the data, and technical observations.
Equipment included internet-enabled desktops and laptops used by a pool of four American Board of Psychiatry and Neurology (ABPN) certified, fellowship-trained vascular neurologists and the telemedicine systems at remote emergency departments. The software enabled site-independent access to two-way audio and high-resolution video, over standard internet connections (BF Technologies, San Diego, CA, USA).
The previously described STRokE DOC adjudication committee and levels 1, 2a, 2b, 3a, and 3b adjudication procedures were also utilized for the STRokE DOC AZ TIME trial 5,9.
Statistical Analysis
STRokE DOC AZ TIME trial was principally a feasibility trial. It was not designed to have sufficient power to detect a statistically significant difference between the two consultative modes, telemedicine and telephone-only. Determination of the trial sample size of 54 randomized subjects was made based on the number of participants the investigators estimated to be achievable in the course of the 1-year trial. The Cochran-Mantel-Haenszel (CMH) test, stratified by study site, was used to compare the correct decision rate (primary study outcome) between treatment groups. A Fisher’s exact test was used for all other correct decision outcomes, rate of thrombolytic use, rate of intracranial hemorrhage, mortality, and the 90-day modified Rankin scale score (0–1 versus 2–6). A Wilcoxan Rank Sum test was used for 90-day Barthel index and time point comparisons. All analyses were performed using the statistical software R (version 2.9.0).
Whereas in the STRokE DOC trial5, patients were enrolled for whom treatment disagreements were impossiblethus artificially enhancing agreement in both arms, which was not the case in STRokE DOC AZ TIME. (Refer to trial flow figure)
Role of the funding source
The sponsors of the trial had no role in the trial design, trial conduct, data acquisition, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all of the data in the study and final responsibility for the decision to submit for publication.
Results
The primary objective of this feasibility trial was satisfactorily achieved – an operational, single metropolitan hub primary stroke center, double rural hospital spoke research network was established in Arizona.
Fifty four patients presenting to a participating emergency department were randomized (figure 1). Overall 39/54 (72%) patients arrived by EMS. There were no demographic differences between groups. 90-day outcomes were available for 46/54 (85%) patients. Loss to 90-day follow-up was not significantly different across the two consultative methodology groups. The risk factor, myocardial infarction (p=0.0183), was present in a higher proportion of the telemedicine group subjects, but the proportions having other risk factors were not significantly different. (Table 1)
Table 1.
Subject Demographics and Risk Factors
| Patient Characteristics | Overall (n = 54) | Telemedicine (n = 27) | Telephone (n = 27) | P value |
|---|---|---|---|---|
| Age (years) | ||||
| Mean ± SD | 66.3 ± 13.5 | 66.4 ± 13.6 | 66.1 ± 13.6 | >0.99 |
| Female, n(%) | 27 (50) | 13(48) | 14 (52) | >0.99 |
| Race, n(%) | 52 (96) | 26 (96) | 26 (96) | >0.99 |
| White | 2 (4) | 1 (4) | 1 (4) | |
| Black | ||||
| Not Hispanic, n(%) | 47(87) | 23 (85) | 24 (89) | >0.99 |
| Weight (kg) | ||||
| Mean ± SD | 84.7 ± 19.7 | 88.2 ± 18.8 | 80.9 ± 20.3 | 0.49 |
| Risk Factors, n (%) & [% unknown] | ||||
| Coronary Disease | 16 (30) [6%] | 11 (41) [4%] | 5 (19) [7%] | 0.23 |
| MI | 10 (19) [9%] | 9(33) [7%] | 1(4) [11%] | 0.02 |
| Prior CVA | 14 (26) [2%] | 8 (30) [0%] | 6 (22) [4%] | 0.76 |
| Atrial Fibrillation | 10 (19) [6%] | 5 (19) [4%] | 5 (19) [7%] | >0.99 |
| Diabetes | 14 (26) [4%] | 6 (23) [0%] | 8 (30) [7%] | 0.32 |
| Hypertension | 40 (74) [2%] | 22 (82) [0%] | 18 (67) [4%] | 0.35 |
| Hyperlipidemia | 19 (35) [11%] | 10 (37) [7%] | 9 (33) [15%] | 0.85 |
| Fam Hx: Stroke/TIA | 6 (11) [60%] | 4 (15) [67%] | 2 (7) [52%] | 0.22 |
| Present Alcohol Use | 6 (11) [41%] | 2 (7) [33%] | 4(15) [48%] | 0.40 |
| Present Tobacco Use | 14 (26) [15%] | 8(30) [(7%] | 6 (22) [22%] | 0.44 |
Table 2 displays subjects’ pre-stroke functional status and stroke severity at baseline. The mean NIHSS score was 7.3 for the overall trial cohort and 12.2 for thrombolysis subset. Forty-six percent of the trial cohort presented with strokes resulting in moderate to severe disability at baseline. Baseline CT scans were interpreted to be normal in 30%, harbor a radiological contraindication to thrombolysis in 7%, and reveal ICH in 2%.
Table 2.
Subject Stroke Severity at Baseline
| Baseline Stroke Severity | Overall (n=54) | Telemedicine (n=27) | Telephone (n=27) | P value |
|---|---|---|---|---|
| Pre-Stroke mRS (Complete Scale) n (%) | ||||
| Dichotomized (0-1) | 46 (85) | 23 (85) | 23 (85) | >0.99 |
| 0=No symptoms | 42 (78) | 20 (74) | 22 (82) | |
| 1=No significant disability | 4 (7) | 3 (11) | 1 (4) | |
| 2=Slight disability | 5 (9) | 3 (11) | 2 (7) | |
| 3=Moderate disability | 3 (6) | 1 (4) | 2 (7) | |
| 4=Moderate to severe disability | 0 (0) | 0 (0) | 0 (0) | |
| 5=Severe disability | 0 (0) | 0 (0) | 0 (0) | |
| Baseline mRS (Complete Scale) n (%) | ||||
| Dichotomized (0-1) | 6 (11) | 3 (11) | 3 (11) | >0.99 |
| 0=No symptoms | 2 (4) | 1 (4) | 1 (5) | |
| 1=No significant disability | 4 (7) | 2 (7) | 2 (7) | |
| 2=Slight disability | 8 (15) | 3 (11) | 5 (19) | |
| 3=Moderate disability | 11 (20) | 6 (22) | 5 (19) | |
| 4=Moderate-severe disability | 25 (46) | 13 (48) | 12 (44) | |
| 5=Severe disability | 4 (7) | 2 (7) | 2 (7) | |
| NIHSS Mean ± SD (Median) | 7.3±6.2 (5.5) | 7.1±5.7 (5) | 7.6±6.7 (6) | 0.82 |
| mNIHSS Mean ± SD (Median) | 5.2±5.3 (3) | 5.3±5.2 (3) | 5.1±5.6 (3) | 0.89 |
| Baseline CT | ||||
| Scan Normal | 16 (30%) | 7 (26%) | 9 (33%) | 0.77 |
| Primary ICH * | 1 (2%) | 1 (4%) | 0 (0%) | >0.99 |
| CT Contraindication to rt-PA | 4 (7%) | 2 (7%) | 2 (7%) | >0.99 |
| rt-PA subset NIHSS (Mean ± SD) | 12.2±7.6 | 12.6±6.1 | 11.8±9.2 | 0.64 |
| rt-PA subset mNIHSS (Mean ± SD) | 9.1±6.7 | 10.1±5.6 | 8.0±7.8 | 0.49 |
A single subject was excluded from the evaluation time analysis because it was ultimately determined that the stroke onset had been several days prior to his hospital assessment. Table 3 reveals that the evaluation times for subjects in the two consultative modes featured no statistically significant differences. Overall, mean stroke symptom onset to emergency department door arrival time was 81.0 minutes. Mean door to emergency physician evaluation time 8.1 minutes, to telestroke hotline call 41.4 minutes, to consent 49.5 minutes, to neurological assessment 79.6 minutes, to CT interpretation 89.4 minutes, and to treatment decision 95.6 minutes. Mean telestroke hotline call to consent was 8.0 minutes, consent to neurological assessment 30.1 minutes, neurological assessment start to decision 16.1 minutes, and decision to thrombolysis (when applicable) was 19.8 minutes.
Table 3.
Subject Assessment, Decision, and Treatment Times
 |
Correct treatment decisions were made in similar proportions of the telemedicine and telephone-only consultation groups, as demonstrated by the primary outcome measure (level 2b adjudication) 87% overall (85% telemedicine and 89% telephone-only) (p > 0.999), and in identical proportions, 89%, when assessed at level 1. (Table 4A) Overall intravenous thrombolytic treatment was used in identical proportions, 30%, of the telemedicine and telephone consultations. There were no differences between the groups in the proportions of subjects who reached good 90-day functional outcomes as assessed by the Barthel Index (95–100), and the modified Rankin Scale scores (dichotomized 0–1), or mortality.
Table 4.
Table 4A. Overall Group Results and 4B: Thrombolysis Sub-Group Results
| Analyses | Telemedicine | Telephone | P value * |
|---|---|---|---|
| Overall | n=27 | n=27 | |
| Correct Decision | |||
| Level 2b (SDAC) (Primary) | 85% | 89% | >0.99 |
| Level 1 (SDAC) | 89% | 89% | >0.99 |
| Level 2a (MM) | 93% | 100% | 0.49 |
| Level 3a (MM) | 93% | 100% | 0.49 |
| Level 3b (SDAC) | 85% | 96% | 0.35 |
| Overall IV rt-PA treatment | 30% (n=8) | 30% (n=8) | >0.99 |
| Overall Post Consult ICH | 4% (n=1) | 0% (n=0) | >0.99 |
| 90d BI (95–100) | 59% (n=13/22) | 58% (n=14/24) | 0.77 |
| 90d mRS (Dichotomized 0-1) | 46% (n=10/22) | 38% (n=9/24) | 0.61 |
| Overall Mortality | 4% (n=1) | 11% (n=3) | |
| +rt-PA Subgroup | n=8 | n=8 | |
| Correct Decision | 0.57 | ||
| Level 2b (SDAC) | 63% | 88% | >0.99 |
| Level 1 (SDAC) | 89% | 89% | 0.47 |
| Level 2a (MM) | 75% | 100% | 0.47 |
| Level 3a (MM) | 75% | 100% | 0.57 |
| Level 3b (SDAC) | 63% | 88% | >0.99 |
| Post rt-PA ICH | 13% (n=1) | 0% (n=0) | 0.59 |
| 90d BI (95–100) | 57% (n=4/7) | 29% (n=2/7) | 0.56 |
| 90d mRS (Dichotomized 0-1) | 43% (n=3/7) | 14% (n=1/7) | >0.99 |
| Subgroup Mortality | 0% (n=0) | 13% (n=1) | |
SDAC=STRokE DOC Adjudicating Committee, MM=Medical Monitor,
P-values are from Fisher’s Exact test
There were no statistically significant differences in the proportion of correct treatment decisions between the two consultative modes in the thrombolysis subgroup (table 4B), and only 1 patient sustained a symptomatic intracranial hemorrhage, overall.
All level 2b thrombolysis eligibility protocol violations are displayed in table 5. The most common violation in the trial patients who were treated with thrombolysis was time of stroke symptom onset remaining unclear. The violations amongst trial patients that had not received thrombolytics were variable. Refer to Table 5.
Table 5.
Adjudication Disagreements
| Group | Thrombolysis | Reason for Disagreement | |
|---|---|---|---|
| 1 | Telemedicine | No | Committee would have attempted to lower blood pressure and treated. |
| 2 | Telemedicine | No | Committee would have treated a patient status post TURP 20 days ago without gross hematuria. |
| 3 | Telemedicine | Yes | Committee would not have treated as time of stroke onset remained unclear. |
| 4 | Telemedicine | Yes | Committee would not have treated as time of onset remained unclear. |
| 5 | Telephone | No | Committee would have treated the patient’s mild deficit. |
| 6 | Telephone | Yes | Committee would not have treated as time of onset was not documented and remained unclear. |
| 7 | Telephone | No | Committee would have treated a patient who fell, as there was no evidence of head trauma. |
Reasons for adjudication disagreements at the primary (level 2b) adjudication level, including number of disagreements at that level.
The demographics and NIHSS analysis combined showed no difference in non-completed data between telephone and telemedicine consultation groups.
Site independent evaluations were performed in 27 (100%) of the telemedicine consultations, using local area network (LAN) wired internet access or broadband wireless access.
The distribution of consultations across sites, emergency physicians, and neurology consultants were as follows: 34 (63%) consultations were conducted at one site, and 20 (37%) at the second site. Twenty different spoke emergency physicians requested consultations: 8 (40%) of them initiated a single consultation, 7 (35%) initiated between 2 to 4 consultations and 5 (25%) initiated 5 or more consultations. One remote consultant performed 31 (57%) of the consultations, the 2nd performed 13 (24%), the 3rd performed 9 (17%), and the 4th conducted a single (2%) consultation. At one site, emergency medicine residents, supervised by emergency physician investigators, participated in 16/34 (47%) of the consultations. There was no apparent relationship between provider or spoke hospital experience and the occurrence of adjudication disagreements.
Technical problems were noted in 20/27 (74%) of telemedicine consultations and in 0/27 (0%) telephone consultations. None of the technical problems prevented the determination of a treatment decision but some did impact the time to treatment decision. Of the 20 observations, 1 was due to video-only, 3 were due to internet connection-only, 5 were due to audio-only, 5 to DICOM-only, and 6 were due to multiple or other causes.
There were no differences in diagnoses. Only 3/54 (6%) subjects were discharged from the emergency department with a diagnosis other than stroke or TIA.
Patients were determined ineligible for thrombolysis for several reasons and, in order of descending proportions, they were: minor deficit (36%), time since stroke onset exceeded 3 hours (29%), rapid spontaneous resolution (14%), recent ischemic stroke (7%), active genitourinary bleeding (7%), anticoagulant use and international normalized ratio (INR) > 1.7 (7%).
Discussion
The results of this prospective, blinded, randomized trial confirm that the successful elements of the UCSD STRokE DOC hub and spoke telestroke network were transferable and reproducible in a completely new and separate geographic location amongst telemedicine naive healthcare institutions and personnel. The results demonstrate that both telemedicine and telephone, incorporated into a state network of emergency stroke care, are efficacious for making acute treatment decisions.
Telephone-only and telemedicine consultations resulted in 89% and 85% correct decisions, respectively, without any statistically significant difference. STRokE DOC AZ telephone-only consultations may have over-performed in comparison to STRokE DOC 5 (82% correct) for the following reasons: STRokE DOC AZ TIME trial authorized a 1-month telephone-only run-in experience prior to randomization, and the vascular neurology consultants devoted approximately the same time to telephone consultations as they did to telemedicine consultations (consent to decision time interval), 43.7 minutes versus 48.6 minutes, respectively. Consultants regularly and liberally re-acquired the history directly by telephone from patient, family members, and witnesses in addition to requesting repeat neurological assessments from emergency department nurses and physicians. A testament to the thoroughness of the telephone consultations was the comparable completeness of data collection between both telephone and telemedicine modalities. STRokE DOC AZ telemedicine consultations may have under performed in comparison to the original STRokE DOC trial (98% correct) due to the relative telemedicine inexperience of the hub and spoke healthcare personnel and due to the high proportion of technical observations witnessed by the telemedicine consultations. Ultimately, the trial was not designed to have sufficient power to detect a difference between the two consultation modalities, and no statistically significant differences were observed. Fundamentally, the results display that both telemedicine and telephone consultations rendered satisfactory levels of correct treatment decision making regarding thrombolysis eligibility amongst remote acute stroke patients in the context of a rigorous clinical trial protocol within an established stroke network of care.
Historically, rural Arizona hospitals, outside metropolitan Tucson and Phoenix, have thrombolysed an estimated 2 to 4% of all ischemic stroke patients requiring admission 4. The STRokE DOC AZ trial demonstrated a substantially higher proportion of patients at the two remote participating hospitals receiving thrombolysis (30%), regardless of consultative modality. Presumably the supportive network which was established and the real-time emergency vascular neurology consultative support, by telephone or telemedicine, for emergency physicians facilitated the accurate diagnoses, appropriate decision making, and thrombolysis administration to eligible stroke patients.
Regarding the measurement of long-term morbidity and mortality outcomes, Meyer et al 5 cautions against comparisons being made between different trials with different patient populations or different stroke care protocols, and instead recommends larger prospective trials with more patients given thrombolytic treatment after telemedicine consultations. To that end several research initiatives are underway or planned. For example, a new prospective stroke telemedicine trial in Arizona is already underway, Stroke Telemedicine for Arizona Rural Residents (STARR) Network Trial (Clinicaltrials.gov Identifier NCT00829361). The objective of the STARR Network Trial is to establish a system for the prospective collection, recording, and regular analysis of telestroke patient consultation and care data for the purpose of quality measure assessment and improvement and benchmarking against other national and international telestroke programs. A second example underway is the Stroke Team Remote Evaluation Using a Digital Observation Camera - Long Term Outcomes (STRokE DOC-LTO Clinicaltrials.gov Identifier NCT00936455). Finally a pooled analysis of the STRokE DOC and STRokE DOC AZ telemedicine stroke trials is planned.
The rate of post-thrombolytic intracerebral hemorrhage was low, 6% overall (13% for telemedicine and 0% for telephone, P = NS), as low as in the original STRokE DOC trial. It is reassuring that despite the potential risks of establishing a novel telestroke research network in a neighboring state amongst new consultant providers in new remote hospitals with technological complexities, major adverse outcomes to patient care were preventable.
Figure 3 demonstrates that there were no statistically significant differences in time increments between the telemedicine and telephone groups, even for the consent to decision (which includes the actual consultation time interval). This is remarkable and strongly suggests that the investigators invested sufficient time, regardless of the consultative modality, to determine the salient elements of the clinical assessment and to complete the requisite data fields in the case report forms. The actual consultation time interval is best described as the difference between consent to decision and consent to neurological examination, for example 46.1 minutes minus 30.1 minutes which equals 16 minutes.
Technical problems were noted in 74% of the STRokE DOC AZ telemedicine consultations (compared with only 19% in the original STRokE DOC trial). The reasons were plentiful. The explanation which had the greatest impact was that telemedicine use conflicted with Mayo Clinic corporate security. The network configuration and Mayo Clinic laptop security profiles adversely affected the use of telemedicine. The problems prevented the smooth and uninterrupted use of the Accessvideo client and DICOM. The technical issues witnessed ranged from trusted sites configuration to performance problems. Regular changes to the system arose from profile updates that reset the configuration of Accessclient and prevented the use of audio and video from spoke sites. Accessclient itself had built-in HIPPA compliant security which accounted for slowdown when coupled with Mayo Clinic corporate security profiles. What the trial experience eventually revealed was that the best way to run the telemedicine system was to have it installed on a standard laptop without being on the hub hospital domain. This allowed for full use of the product with limited issues on the remote side. Other explanations for high technical observations included the lack of telemedicine experience amongst the vascular neurology consultants, limited technical training and trouble shooting skills, lack of around the clock IT support, at hub and spokes, for the telestroke trial, and a definition for ‘technical observation’ that included any audio, video, internet, or DICOM issue, delay, or interruption, both major and minor.
Between the completion of STRokE DOC and STRokE DOC AZ TIME, the American Heart Association (AHA) has published a scientific statement reviewing the evidence for the use of telemedicine within stroke systems of care 10. The NIHSS-telestroke examination, when administered by a stroke specialist using HQ-VTC, is recommended when an NIHSS-bedside assessment by a stroke specialist is not immediately available for patients in the acute stroke setting, and this assessment is comparable to an NIHSS-bedside assessment (Class I, level of Evidence A). Additionally the AHA has published a policy statement concerning recommendations for the implementation of telemedicine within stroke systems of care 11. National telemedicine initiatives, like those in stroke, are essential to healthcare reform. Telemedicine is capable of addressing inequities in access to care, economic issues, and quality enhancement 12.
In summary, the authors of this trial report that it is feasible to extend the original UCSD STRokE DOC trial protocol to a new state and establish an operational single hub, multi-rural spoke hospital telestroke research network in Arizona. The feasibility trial was not designed to have sufficient power to detect a difference between the two consultative modes, telemedicine and telephone-only. Within the newly established telestroke network, whether by telemedicine or telephone consultative modalities, there were appropriate treatment decisions, high rates of thrombolysis use, improved data collection, low rates of intracerebral thrombolysis, and equally favorable time requirements. The learning curve was steep for the hub and spoke personnel of the new telestroke network as reflected by the frequent technical occurrences. Overall, the results support the effectiveness of highly organized and structured stroke telemedicine networks for extending expert stroke care into rural remote communities lacking sufficient neurological expertise.
Acknowledgments
Arizona Department of Health Services Research Grant and Mayo Clinic Research Grant
The work was supported by the Arizona Department of Health Services and the Mayo Clinic. The telemedicine application (AccessVideo TM) was purchased from BF Technologies. We acknowledge our participating facilities (Kingman and Yuma Regional Medical Centers) and the published list of STRokE DOC AZ TIME investigators 3. We acknowledge Laura C. Pappagallo, RN, Kelley J. Dunkowski, MBA, and Paola G. Scarberry for being the first Arizona telestroke project managers; Nicole L. Olmstead and Cristina T. Lipinski, RN, for serving as telestroke research monitors; Carol B. Garrison for assisting us with credentialing and privileging; Michael E. Brown, JD, and Page Y. Underwood, JD, for legal input; Kenneth G. Bobis for information technology; and Marvin H. Ruona for graphic designs.
Appendix 1
Contributors
BCM is credited with the idea and design for the original STRokE DOC trial protocol. BMD, BJB, and BCM had the idea for and designed this study, STRokE DOC AZ TIME; they participated in the acquisition, analysis, and interpretation of the data, drafting and revision of the manuscript, and provided administrative, technical, and material support, and supervised the study. BJB and BMB obtained the funding. MIA, TJI, DWD participated in the acquisition of the data and revision of the manuscript. KB and TCK participated in the research coordination, research administration, organization, data handling, and revision of the manuscript. TEJK and MLM participated in the overall network and trial management, logistics, organization, and revision of the manuscript. DAC developed and maintained the trial network information technology, and assisted with revision of the manuscript. CRHS served as trial monitor and assisted with revision of the manuscript. RR participated in the design of the study, the analysis and interpretation of data, revision of the manuscript, and statistical analysis. MPW and PCR participated in the organization of their respective spoke hospital site emergency departments’ participation, data acquisition, and revision of the manuscript. All authors have seen and approved the final version of the manuscript.
Footnotes
Trial Registration: ClinicalTrials.gov Number NCT00623350
Conflicts of interest: We have no conflicts of interest.
References
- 1.Joubert J, Prentice LF, Moulin T, Liaw ST, Joubert LB, Preux PM, Ware D, Medeiros de Bustos E, McLean A. Stroke in rural areas and small communities. Stroke. 2008;39:1920–1928. doi: 10.1161/STROKEAHA.107.501643. [DOI] [PubMed] [Google Scholar]
- 2.Leira EC, Hess DC, Torner JC, Adams HP., Jr Rural-urban differences in acute stroke management practices: A modifiable disparity. Archives of Neurology. 2008;65:887–891. doi: 10.1001/archneur.65.7.887. [DOI] [PubMed] [Google Scholar]
- 3.Demaerschalk BM, Miley ML, Kiernan TE, Bobrow BJ, Corday DA, Wellik KE, Aguilar MI, Ingall TJ, Dodick DW, Brazdys K, Koch TC, Ward MP, Richemont PC, Coinvestigators S. Stroke telemedicine.[see comment] Mayo Clinic Proceedings. 2009;84:53–64. doi: 10.4065/84.1.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Miley ML, Demaerschalk BM, Olmstead NL, Kiernan TE, Corday DA, Chikani V, Bobrow BJ. The state of emergency stroke resources and care in rural arizona: A platform for telemedicine. Telemedicine and e-Health. 2009;15:691–699. doi: 10.1089/tmj.2009.0018. [DOI] [PubMed] [Google Scholar]
- 5.Meyer BC, Raman R, Hemmen T, Obler R, Zivin JA, Rao R, Thomas RG, Lyden PD. Efficacy of site-independent telemedicine in the stroke doc trial: A randomised, blinded, prospective study.[see comment] Lancet Neurology. 2008;7:787–795. doi: 10.1016/S1474-4422(08)70171-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Del Zoppo GJ, Saver JL, Jauch EC, Adams HP, Jr American Heart Association Stroke C. Expansion of the time window for treatment of acute ischemic stroke with intravenous tissue plasminogen activator: A science advisory from the american heart association/american stroke association. Stroke. 2009;40:2945–2948. doi: 10.1161/STROKEAHA.109.192535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meyer BC, Lyden PD, Al-Khoury L, Cheng Y, Raman R, Fellman R, Beer J, Rao R, Zivin JA. Prospective reliability of the stroke doc wireless/site independent telemedicine system.[see comment] Neurology. 2005;64:1058–1060. doi: 10.1212/01.WNL.0000154601.26653.E7. [DOI] [PubMed] [Google Scholar]
- 8.Meyer BC, Raman R, Chacon MR, Jensen M, Werner JD. Reliability of site-independent telemedicine when assessed by telemedicine-naive stroke practitioners. Journal of Stroke & Cerebrovascular Diseases. 2008;17:181–186. doi: 10.1016/j.jstrokecerebrovasdis.2008.01.008. [DOI] [PubMed] [Google Scholar]
- 9.Meyer BC, Raman R, Rao R, Fellman RD, Beer J, Werner J, Zivin JA, Lyden PD. The stroke doc trial technique: ‘video clip, drip, and/or ship’. International Journal of Stroke. 2007;2:281–287. doi: 10.1111/j.1747-4949.2007.00153.x. [DOI] [PubMed] [Google Scholar]
- 10.Schwamm LH, Holloway RG, Amarenco P, Audebert HJ, Bakas T, Chumbler NR, Handschu R, Jauch EC, Knight WAt, Levine SR, Mayberg M, Meyer BC, Meyers PM, Skalabrin E, Wechsler LR American Heart Association Stroke C, Interdisciplinary Council on Peripheral Vascular D. A review of the evidence for the use of telemedicine within stroke systems of care: A scientific statement from the american heart association/american stroke association. Stroke. 2009;40:2616–2634. doi: 10.1161/STROKEAHA.109.192360. [DOI] [PubMed] [Google Scholar]
- 11.Schwamm LH, Audebert HJ, Amarenco P, Chumbler NR, Frankel MR, George MG, Gorelick PB, Horton KB, Kaste M, Lackland DT, Levine SR, Meyer BC, Meyers PM, Patterson V, Stranne SK, White CJ American Heart Association Stroke C. Council on Epidemiology and P, Interdisciplinary Council on Peripheral Vascular D, Council on Cardiovascular Radiology and I. Recommendations for the implementation of telemedicine within stroke systems of care: A policy statement from the american heart association. Stroke. 2009;40:2635–2660. doi: 10.1161/STROKEAHA.109.192361. [DOI] [PubMed] [Google Scholar]
- 12.Bashshur RL, Shannon GW, Krupinski EA, Grigsby J, Kvedar JC, Weinstein RS, Sanders JH, Rheuban KS, Nesbitt TS, Alverson DC, Merrell RC, Linkous JD, Ferguson AS, Waters RJ, Stachura ME, Ellis DG, Antoniotti NM, Johnston B, Doarn CR, Yellowlees P, Normandin S, Tracy J. National telemedicine initiatives: Essential to healthcare reform. Telemedicine and e-Health. 2009;15:600–610. doi: 10.1089/tmj.2009.9960. [DOI] [PubMed] [Google Scholar]