Abstract
We used Duffin's isoxic hyperoxic ( mmHg) and hypoxic (
mmHg) and hypoxic ( mmHg) rebreathing tests to compare the control of breathing in eight (7 male) Andean highlanders and six (4 male) acclimatizing Caucasian lowlanders after 10 days at 3850 m. Compared to lowlanders, highlanders had an increased non-chemoreflex drive to breathe, characterized by higher basal ventilation at both hyperoxia (10.5 ± 0.7 vs. 4.9 ± 0.5 l min−1, P= 0.002) and hypoxia (13.8 ± 1.4 vs. 5.7 ± 0.9 l min−1, P < 0.001). Highlanders had a single ventilatory sensitivity to CO2 that was lower than that of the lowlanders (P < 0.001), whose response was characterized by two ventilatory sensitivities (VeS1 and VeS2) separated by a patterning threshold. There was no difference in ventilatory recruitment thresholds (VRTs) between populations (P= 0.209). Hypoxia decreased VRT within both populations (highlanders: 36.4 ± 1.3 to 31.7 ± 0.7 mmHg, P < 0.001; lowlanders: 35.3 ± 1.3 to 28.8 ± 0.9 mmHg, P < 0.001), but it had no effect on basal ventilation (P= 0.12) or on ventilatory sensitivities in either population (P= 0.684). Within lowlanders, VeS2 was substantially greater than VeS1 at both isoxic tensions (hyperoxic: 9.9 ± 1.7 vs. 2.8 ± 0.2, P= 0.005; hypoxic: 13.2 ± 1.9 vs. 2.8 ± 0.5, P < 0.001), although hypoxia had no effect on either of the sensitivities (P= 0.192). We conclude that the control of breathing in Andean highlanders is different from that in acclimatizing lowlanders, although there are some similarities. Specifically, acclimatizing lowlanders have relatively lower non-chemoreflex drives to breathe, increased ventilatory sensitivities to CO2, and an altered pattern of ventilatory response to CO2 with two ventilatory sensitivities separated by a patterning threshold. Similar to highlanders and unlike lowlanders at sea-level, acclimatizing lowlanders respond to hypobaric hypoxia by decreasing their VRT instead of changing their ventilatory sensitivity to CO2.
mmHg) rebreathing tests to compare the control of breathing in eight (7 male) Andean highlanders and six (4 male) acclimatizing Caucasian lowlanders after 10 days at 3850 m. Compared to lowlanders, highlanders had an increased non-chemoreflex drive to breathe, characterized by higher basal ventilation at both hyperoxia (10.5 ± 0.7 vs. 4.9 ± 0.5 l min−1, P= 0.002) and hypoxia (13.8 ± 1.4 vs. 5.7 ± 0.9 l min−1, P < 0.001). Highlanders had a single ventilatory sensitivity to CO2 that was lower than that of the lowlanders (P < 0.001), whose response was characterized by two ventilatory sensitivities (VeS1 and VeS2) separated by a patterning threshold. There was no difference in ventilatory recruitment thresholds (VRTs) between populations (P= 0.209). Hypoxia decreased VRT within both populations (highlanders: 36.4 ± 1.3 to 31.7 ± 0.7 mmHg, P < 0.001; lowlanders: 35.3 ± 1.3 to 28.8 ± 0.9 mmHg, P < 0.001), but it had no effect on basal ventilation (P= 0.12) or on ventilatory sensitivities in either population (P= 0.684). Within lowlanders, VeS2 was substantially greater than VeS1 at both isoxic tensions (hyperoxic: 9.9 ± 1.7 vs. 2.8 ± 0.2, P= 0.005; hypoxic: 13.2 ± 1.9 vs. 2.8 ± 0.5, P < 0.001), although hypoxia had no effect on either of the sensitivities (P= 0.192). We conclude that the control of breathing in Andean highlanders is different from that in acclimatizing lowlanders, although there are some similarities. Specifically, acclimatizing lowlanders have relatively lower non-chemoreflex drives to breathe, increased ventilatory sensitivities to CO2, and an altered pattern of ventilatory response to CO2 with two ventilatory sensitivities separated by a patterning threshold. Similar to highlanders and unlike lowlanders at sea-level, acclimatizing lowlanders respond to hypobaric hypoxia by decreasing their VRT instead of changing their ventilatory sensitivity to CO2.
Introduction
Ventilatory acclimatization to hypoxia in lowlanders has a complicated time course that results in a progressive increase in resting ventilation occurring over hours to days (Powell et al. 1998). This acclimatization is accompanied by an increased hypoxic ventilatory response (HVR) (Sato et al. 1992; Howard & Robbins, 1995). In contrast, Andean highlanders are known to have lower alveolar ventilation compared to acclimatized lowlanders (Chiodi, 1957; Severinghaus et al. 1966; Lahiri, 1968; Cudkowicz et al. 1972; Beall et al. 1997; Moore, 2000), and blunted HVR (Severinghaus et al. 1966; Sorensen & Severinghaus, 1968; Velasquez et al. 1968; Lahiri et al. 1969; Cudkowicz et al. 1972; Beall et al. 1997; Gamboa et al. 2003; Brutsaert et al. 2005) despite their chronic exposure to hypoxia. The mechanisms responsible for these differences are unclear, but they probably involve alterations in the control of breathing (Moore, 2000; Brutsaert, 2007).
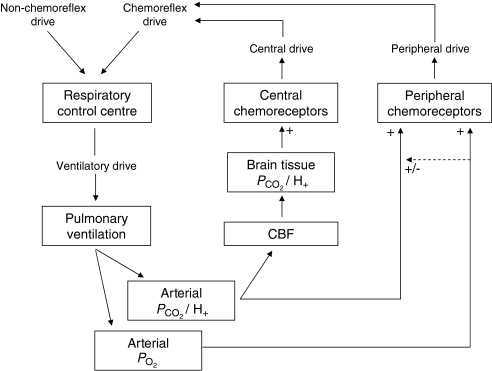
The control of breathing can be divided into chemoreflex and non-chemoreflex drives to breathe (Fig. 1) (Lloyd & Cunningham, 1963). Non-chemoreflex drives (e.g. wakefulness drive, voluntary cortical drive, hormonal factors) have never been measured in highlanders, while a single study of acclimatization in lowlanders showed that these drives remain unchanged from sea-level values after 5 days of high altitude exposure (Somogyi et al. 2005). Chemoreflex drives to breathe can be further divided into central and peripheral drives. Both central and peripheral chemoreceptors respond to changes in hydrogen ion concentration ([H+]) in their immediate environments (Torrance, 1996; Nattie & Li, 2009), while peripheral chemoreceptors are also responsive to changes in  via hypoxia-mediated changes in their [H+] ion sensitivity (Cunningham, 1987; Torrance, 1996; Kumar & Bin-Jaliah, 2007). As a result, most studies in acclimatizing lowlanders and Andean highlanders have induced acute changes in
via hypoxia-mediated changes in their [H+] ion sensitivity (Cunningham, 1987; Torrance, 1996; Kumar & Bin-Jaliah, 2007). As a result, most studies in acclimatizing lowlanders and Andean highlanders have induced acute changes in  and
and  and measured the ensuing changes in ventilation to assess the chemoreflex control of breathing (Duffin, 2007).
and measured the ensuing changes in ventilation to assess the chemoreflex control of breathing (Duffin, 2007).
Figure 1. The control of breathing model (Lloyd & Cunningham, 1963).
The total ventilatory drive is the sum of chemoreflex and non-chemoreflex drives to breathe, which are integrated in the respiratory centre. The ventilatory drive exerts its action on the respiratory muscles that affect pulmonary ventilation and result in changes in arterial  and
and  . Arterial [H+] is ‘sampled’ by the peripheral chemoreceptors located in the carotid bodies, where it determines the peripheral chemoreflex drive. Hypoxia exerts its effect on ventilation via peripheral chemoreceptors, where it acts indirectly via increasing the ventilatory sensitivity to [H+] in most individuals but may also act directly by increasing the overall activity of the receptor. Central chemoreceptors respond to changes in the local [H+] environment, which is affected by brain tissue
. Arterial [H+] is ‘sampled’ by the peripheral chemoreceptors located in the carotid bodies, where it determines the peripheral chemoreflex drive. Hypoxia exerts its effect on ventilation via peripheral chemoreceptors, where it acts indirectly via increasing the ventilatory sensitivity to [H+] in most individuals but may also act directly by increasing the overall activity of the receptor. Central chemoreceptors respond to changes in the local [H+] environment, which is affected by brain tissue  . Brain tissue
. Brain tissue  is a function of both arterial
is a function of both arterial  and cerebral blood flow, which acts to decrease the brain tissue
and cerebral blood flow, which acts to decrease the brain tissue  at a higher flow (Berkenbosch et al. 1989; Mohan et al. 1999). The central and peripheral chemoreflex drives add together to form a total chemoreflex drive to breathe.
at a higher flow (Berkenbosch et al. 1989; Mohan et al. 1999). The central and peripheral chemoreflex drives add together to form a total chemoreflex drive to breathe.
These studies suggest that acclimatization to high altitude in lowlanders affects both central and peripheral chemosensitivity. However, the findings are contradictory, such that central chemosensitivity is either increased (Schoene et al. 1990; Fatemian & Robbins, 1998; Ainslie et al. 2003) or remains unchanged (Sato et al. 1992; Somogyi et al. 2005; Ainslie & Burgess, 2008) and peripheral chemosensitivity is either increased (Ainslie & Burgess, 2008) or remains unchanged (Somogyi et al. 2005). On the other hand, Andean highlanders have been shown to have higher total and peripheral chemosensitivity to CO2 during euoxia (100 mmHg) compared to sea-level lowlanders, with this difference abolished during hypoxia (50 mmHg) (Fatemian et al. 2003). The comparison of these findings in acclimatizing lowlanders and Andean highlanders is complicated by the different methodologies used (Moore, 2000; Duffin, 2007). To date, there have been no studies that specifically compared both chemoreflex and non-chemoreflex drives to breathe between highlanders and acclimatizing lowlanders.
To resolve these conflicting findings we used Duffin's rebreathing technique (Duffin et al. 2000) to compare chemoreflex and non-chemoreflex drives to breathe between acclimatizing Caucasian lowlanders following 10 days at 3850 m and Andean highlanders.
Methods
Subjects
The study was approved by the Ethics Review Board of the University Health Network, Toronto, Canada and Comisión de Ética de la Investigación (CEI) del Comité Nacional de Bioética, La Paz, Bolivia. All studies were performed in accordance with the Declaration of Helsinki of the World Medical Association (2004). For acclimatized lowlander studies, six healthy Caucasian subjects (4 male) were recruited from the members of the 2008 Bolivia expedition and were all members of a respiratory research laboratory at the University of Toronto in Canada. All six lowlander subjects were also authors on the present manuscript. Their mean ±s.d. age, height and body mass were 32.7 ± 13.8 years, 174.2 ± 13.6 cm and 72.0 ± 17.0 kg, respectively (Table 1). For highlander studies, 8 healthy (7 male) long term residents of La Paz, Bolivia (altitude 3850 m) were recruited. The highlander subjects identified themselves as belonging to the native Aymara population. However, since no studies were done to determined the extent of their Aymara heritage, we are not able to comment on the degree of European admixture among our highlander subjects. The duration of residence at high altitude of our highlander subjects is unknown, but in general it is thought that the Andean highlanders have resided at altitude for at least 11,000 years (Aldenderfer, 2003). Their mean ±s.d. age, height and body mass were 38.1 ± 8.6 years, 158.8 ± 8.5 cm and 63.8 ± 4.9 kg, respectively (Table 1). Resting arterial blood gas data were obtained from all subjects (except highlander 7, in whom we were unable to obtain an arterial blood sample). Anthropomorphic and resting arterial blood gas data are summarized in Table 1. Each subject gave an informed written consent before commencing the study.
Table 1.
Resting anthropomorphic and arterial blood gas data from all subjects
| Subject | Age (yrs) | Height (cm) | Weight (kg) | Hb (g dl−1) | Hct (%) | pH |
 (mmHg) (mmHg) |
 (mmHg) (mmHg) |
BE (mequiv l−1) | HCO3− (mmol l−1) | TCO2 (mmol l−1) |
 (%) (%) |
Lac (mmol l−1) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Highlanders (H) | H1 | 46 | 160 | 70 | 16.7 | 49 | 7.455 | 26 | 63 | −6 | 18.2 | 19 | 93 | 0.36 |
| H2 | 41 | 169 | 62 | 16 | 47 | 7.436 | 27 | 57 | −6 | 18.2 | 19 | 91 | 0.9 | |
| H3 | 33 | 160 | 70 | 17 | 50 | 7.446 | 25.8 | 59 | −6 | 17.8 | 19 | 92 | 1.17 | |
| H4 | 30 | 145 | 60 | 17.3 | 51 | 7.441 | 28 | 61 | −5 | 19.1 | 20 | 93 | 0.72 | |
| H6 | 50 | 155 | 68.5 | 15 | 44 | 7.428 | 35.5 | 50 | −1 | 23.5 | 25 | 86 | 0.71 | |
| H7 | 45 | 162 | 62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
| H8 | 26 | 169 | 60 | 16 | 47 | 7.439 | 32.7 | 53 | −2 | 22.2 | 23 | 89 | 0.59 | |
| H9 | 34 | 150 | 58 | 14.6 | 43 | 7.45 | 27.2 | 60 | −5 | 18.9 | 20 | 92 | 0.66 | |
| n | 8 | 8 | 8 | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 | |
| Mean | 38.1 | 158.8 | 63.8 | 16.1 | 47.3 | 7.4 | 28.9 | 57.6 | −4.4 | 19.7 | 20.7 | 90.9 | 0.7 | |
| (s.d.) | (8.6) | (8.5) | (4.9) | (1) | (3) | (0) | (3.7) | (4.6) | (2.1) | (2.2) | (2.4) | (2.5) | (0.3) | |
| Lowlanders (L) | L1 | 26 | 173 | 61 | 14.6 | 42 | 7.433 | 27.9 | 58 | −6 | 18.6 | 19 | n/a | n/a |
| L2 | 32 | 170 | 73 | 15.3 | 45 | 7.436 | 33.9 | 56 | −1 | 22.8 | 24 | n/a | n/a | |
| L3 | 22 | 150 | 44 | 13.6 | 40 | 7.473 | 26.7 | 57 | −4 | 19.6 | 20 | n/a | n/a | |
| L4 | 30 | 188 | 84 | 13.6 | 40 | 7.445 | 26 | 64 | −6 | 17.9 | 19 | 94 | 1.21 | |
| L5 | 60 | 180 | 80 | 16.3 | 48 | 7.416 | 30.3 | 53 | −5 | 19.5 | 20 | 88 | 1.04 | |
| L6 | 26 | 184 | 90 | 16.3 | 48 | 7.438 | 30 | 62 | −4 | 20.3 | n/a | n/a | n/a | |
| n | 6 | 6 | 6 | 6 | 6 | 6 | 6 | 6 | 6 | 6 | 5 | 2 | 2 | |
| Mean | 32.7 | 174.2 | 72 | 15 | 43.8 | 7.4 | 29.1 | 58.3 | −4.3 | 19.8 | 20.4 | 91 | 1.1 | |
| (s.d.) | (13.8) | (13.6) | (17) | (1.2) | (3.7) | (0) | (2.9) | (4) | (1.9) | (1.7) | (2.1) | (4.2) | (0.1) |
Note that in one subject (H7) no arterial blood gas data were obtained due to inability to obtain a blood sample on multiple attempts. Also note that arterial oxygen saturation and lactate values are missing (n/a) in some lowlander subjects due to malfunction of blood analyser cartridges. Hb, haemoglobin concentration; Hct, haematocrit;  , arterial
, arterial  ;
;  , arterial
, arterial  ; BE, base excess; HCO3−, bicarbonate ion concentration; TCO2, total CO2;
; BE, base excess; HCO3−, bicarbonate ion concentration; TCO2, total CO2;  , arterial oxygen saturation.
, arterial oxygen saturation.
General protocol
Prior to experiments, all lowlander and highlander subjects were familiarized with the rebreathing apparatus and were allowed to complete familiarization tests. For highlanders, the familiarization protocol entailed first watching one of the research scientists perform both hypoxic and hyperoxic rebreathing tests on themselves, and then breathing on the rebreathing apparatus and performing several trial rebreathing runs. At the same time, the nature of the experiments and specific instructions were provided in the highlanders' native language through an interpreter, and all their questions were answered. All of the lowlander subjects were members of the respiratory research laboratory and were familiar with the rebreathing experiments. All tests were conducted in the laboratory at the High Altitude Pathology Institute, Clinica IPPA, La Paz, Bolivia (altitude 3850 m, ambient barometric pressure 484 mmHg, ambient temperature 18–25°C; temperature and pressure were measured with a standard thermometer and barometer, respectively). Each subject performed a hypoxic and a hyperoxic test in random order, with at least 20 min of rest time between the tests. Lowlanders were tested 10 days after arrival in La Paz (Altitude 3850 m) from sea-level (Toronto, Canada, altitude 105 m).
Chemoreflex assessment
Testing method
Duffin's rebreathing tests (Mohan & Duffin, 1997) were used to assess subjects’ chemoreflexes. Their interpretation is given in (Duffin et al. 2000), and they have been extensively discussed (Duffin et al. 2000; Mahamed & Duffin, 2001; Mateika et al. 2004). In this modification of Read's original method (Read, 1967), the rebreathing stage is preceded by 5 min of hyperventilation and isoxia is maintained throughout the test. The hyperventilation ensures that the rebreathing stage starts at a CO2 level below the ventilatory recruitment threshold (VRT), which enables both the VRT and the sub-threshold (basal) ventilation to be measured (Mohan et al. 1999). The latter measures the contribution of the non-chemoreflex drives to breathe (Shea, 1996), such as the wakefulness drive (Fink, 1961). Rebreathing is repeated at two different isoxic  tensions: once during hyperoxia (
tensions: once during hyperoxia ( mmHg), to minimize the contribution of the peripheral chemoreceptors and preferential measurement of the central chemoreceptor response (Lloyd & Cunningham, 1963), and once during hypoxia (
mmHg), to minimize the contribution of the peripheral chemoreceptors and preferential measurement of the central chemoreceptor response (Lloyd & Cunningham, 1963), and once during hypoxia ( mmHg), to allow measurement of the combined central and peripheral chemoreceptor responses. The difference between the hypoxic and hyperoxic responses represents the contribution of the peripheral chemoreflex and can be used to calculate HVR at any given isocapnic end-tidal
mmHg), to allow measurement of the combined central and peripheral chemoreceptor responses. The difference between the hypoxic and hyperoxic responses represents the contribution of the peripheral chemoreflex and can be used to calculate HVR at any given isocapnic end-tidal  (
( ) (Duffin, 2007).
) (Duffin, 2007).
Testing protocol
All subjects were seated comfortably in an upright position during the rebreathing tests and wore a finger pulse-oximetry probe. The testing environment was quiet, with minimal distractions. Subjects wore nose clips while breathing via a bacterial–viral filter (Allegiance, Healthcare Corp., McGaw Park, IL, USA) connected to one side of a Series 2870 three-way sliding valve (Hans Rudolph Inc., Kansas City, MO, USA) that allowed the operator to alternate the subject's breathing between ambient air and the rebreathing bag. The rebreathing bag had a volume of 5 litres and an oxygen inlet. It was primed with a CO2–O2 mixture to ensure appropriate equilibration at the beginning of the rebreathing test (Mohan & Duffin, 1997). Inspired and expired partial pressures of CO2 and O2 were sampled at the mouth and monitored throughout the test using infra-red CO2 and polarographic O2 sensors (RespirAct, Thornhill Research Inc, Toronto, Canada). The linearity of both gas analysers in the measurement range used in the current study is ±1% full scale for the O2 analyser and ±0.1% full scale for the CO2 analyser. Ventilation was measured using a mass flow sensor (AWM720P1 Airflow, Honeywell, Freeport, IL, USA). Analog data were digitized (DAQCard-6024E, National Instruments, Austin, TX, USA) and entered continuously into a computer.
A custom-written program (LabVIEW, National Instruments; source code available on request) analysed the data to provide a file of breath-by-breath end-tidal  (
( ), end-tidal
), end-tidal  (
( ), and ventilation. In addition, the program operated a solenoid valve controlling the flow of oxygen to the rebreathing bag to maintain desired isoxic tensions during rebreathing: a
), and ventilation. In addition, the program operated a solenoid valve controlling the flow of oxygen to the rebreathing bag to maintain desired isoxic tensions during rebreathing: a  of 150 mmHg (hyperoxic) or 50 mmHg (hypoxic). The
of 150 mmHg (hyperoxic) or 50 mmHg (hypoxic). The  and
and  analysers were calibrated using gas from cylinders of analysed medical grade compressed gases, and ventilation was calibrated using a 3 litre calibration syringe (Model R5530B, Vacumed, Ventura, CA, USA). A portable pulse oximeter (Autocorr Plus Vital Signs Monitor, BCI international, USA) was used to measure arterial oxygen saturation (
analysers were calibrated using gas from cylinders of analysed medical grade compressed gases, and ventilation was calibrated using a 3 litre calibration syringe (Model R5530B, Vacumed, Ventura, CA, USA). A portable pulse oximeter (Autocorr Plus Vital Signs Monitor, BCI international, USA) was used to measure arterial oxygen saturation ( ) and heart rate.
) and heart rate.
Each rebreathing test began with a 5 min hyperventilation of ambient air, with subjects coached to maintain their  between 10 and 15 mmHg. Subjects then exhaled completely and were switched to the rebreathing bag, from which they took three deep breaths to facilitate rapid equilibration of
between 10 and 15 mmHg. Subjects then exhaled completely and were switched to the rebreathing bag, from which they took three deep breaths to facilitate rapid equilibration of  in the bag, lungs and arterial blood with that of mixed venous blood. This equilibration was verified by observing a plateau in the end-tidal
in the bag, lungs and arterial blood with that of mixed venous blood. This equilibration was verified by observing a plateau in the end-tidal  , and was a prerequisite for continuation of the test. The rebreathing test ended when either ventilation exceeded 100 l min−1,
, and was a prerequisite for continuation of the test. The rebreathing test ended when either ventilation exceeded 100 l min−1,  exceeded 60 mmHg, ventilatory discomfort was no longer tolerable, or sufficient data were obtained to show the ventilatory response. The majority of the rebreathing tests were terminated when sufficient data were obtained or when discomfort was indicated by the subject.
exceeded 60 mmHg, ventilatory discomfort was no longer tolerable, or sufficient data were obtained to show the ventilatory response. The majority of the rebreathing tests were terminated when sufficient data were obtained or when discomfort was indicated by the subject.
Rebreathing test analysis
Rebreathing test data were analysed using a custom-written program (LabVIEW, National Instruments; source code available on request). After eliminating the initial three equilibration breaths, as well as sighs, swallows and breaths incorrectly detected by the acquisition software, breath-by-breath  values were plotted against time and fitted with a least squares regression line. This line provided a predicted value of
values were plotted against time and fitted with a least squares regression line. This line provided a predicted value of  as a function of time, thereby minimizing inter-breath variability. Tidal volume (ml BTPS), respiratory rate (breaths min−1) and ventilation (l min−1 BTPS) were then plotted against the predicted
as a function of time, thereby minimizing inter-breath variability. Tidal volume (ml BTPS), respiratory rate (breaths min−1) and ventilation (l min−1 BTPS) were then plotted against the predicted  (mmHg).
(mmHg).
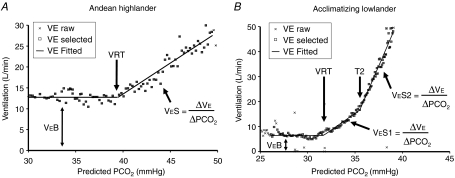
In highlanders (Fig. 2A), these plots were fitted by dividing the data into two segments separated by a breakpoint corresponding to the VRT, defined as the  where ventilation started to increase in response to rising
where ventilation started to increase in response to rising  (Duffin et al. 2000). In lowlanders (Fig. 2B), the plots were fitted by dividing the data into three segments separated by two thresholds: the VRT as in highlanders and a second threshold (T2) defined as the
(Duffin et al. 2000). In lowlanders (Fig. 2B), the plots were fitted by dividing the data into three segments separated by two thresholds: the VRT as in highlanders and a second threshold (T2) defined as the  at which the slope of the ventilatory response to
at which the slope of the ventilatory response to  changed. In both populations, the first segment was fitted with either an exponential decline to a final value, or a mean, and measured sub-VRT ventilation (VeB) that represented non-chemoreflex ventilation drive. In the present study, all first segments were fitted using means. Note that all responses were fitted with a physiological model in mind rather than an empirical curve-fitting technique.
changed. In both populations, the first segment was fitted with either an exponential decline to a final value, or a mean, and measured sub-VRT ventilation (VeB) that represented non-chemoreflex ventilation drive. In the present study, all first segments were fitted using means. Note that all responses were fitted with a physiological model in mind rather than an empirical curve-fitting technique.
Figure 2. Breath-by-breath ventilation vs. end-tidal during a representative Duffin's rebreathing test for one highlander (A) and one lowlander (B) illustrating fitting of the plots.
during a representative Duffin's rebreathing test for one highlander (A) and one lowlander (B) illustrating fitting of the plots.
In highlanders (A), the plots were fitted by dividing them into 2 segments, separated by a breakpoint corresponding to the ventilatory recruitment threshold (VRT). In lowlander subjects (B), the plots were fitted by dividing them into 3 segments, separated by two breakpoints, VRT and T2. T2 was defined as the  at which there was a change in the slope of the ventilatory response to CO2 above VRT. In both populations, the first segment was fitted with either an exponential decline to a final value, or a mean, and measured sub-VRT ventilation (VeB) that represents non-chemoreflex ventilation drive. In highlanders, the second segment was fitted with a straight line where the slope measured the sensitivity (VeS). In lowlanders, the second and third segments were also fitted with straight lines where the slopes measured the first (VeS1) and second (VeS2) ventilatory sensitivities to CO2.
at which there was a change in the slope of the ventilatory response to CO2 above VRT. In both populations, the first segment was fitted with either an exponential decline to a final value, or a mean, and measured sub-VRT ventilation (VeB) that represents non-chemoreflex ventilation drive. In highlanders, the second segment was fitted with a straight line where the slope measured the sensitivity (VeS). In lowlanders, the second and third segments were also fitted with straight lines where the slopes measured the first (VeS1) and second (VeS2) ventilatory sensitivities to CO2.
In highlanders, the second segment was fitted with a straight line, whose slope represented the ventilatory sensitivity (VeS). In lowlanders, the second and third segments were also fitted with straight lines, with slopes representing the first (VeS1) and second (VeS2) ventilatory sensitivities to CO2, respectively. Model fitting was based on minimizing the sum of least squares for non-linear regression using the LabVIEW software (Levenberg–Marquardt algorithm). The furthest outlying points were automatically discarded until an r2 value of >0.95 was achieved.
Statistical analyses
All results are reported as mean ±s.e.m. (except where noted), with significance set at P < 0.05. A two-way repeated measures ANOVA, with factors ‘population’ (highlanders vs. lowlanders) and ‘isoxia’ (hypoxic vs. hyperoxic), and post hoc analysis using Tukey's test were used to detect differences in VeB, VRT and VeS. The highlander VeS was tested against both the VeS1 and VeS2 of lowlanders. In lowlanders, a two-way repeated measures ANOVA, with factors ‘sensitivity’ (VeS1 vs. VeS2) and ‘isoxia’ (hypoxic vs. hyperoxic), and post hoc analysis using Tukey's test were used to detect differences between VeS1 and VeS2.
Results
All subjects completed both rebreathing protocols. Table 2 shows values of VeB, VRT and VeS for all highlander and VeB, VRT, VeS1, T2 and VeS2 for all lowlander subjects. Figure 3 summarizes these findings for both populations.
Table 2.
Individual subject values for non-chemoreflex drives to breathe (VeB), ventilatory recruitment thresholds (VRT) and sensitivities to CO2 (VeS in highlanders; VeS1 and VeS2 in lowlanders) for all rebreathing tests
| Lowlanders after 10 days at 3850 m | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
Isoxic hyperoxic tests ( mmHg) mmHg) |
Isoxic hypoxic tests ( mmHg) mmHg) |
|||||||||
| Subject | VeB (l min−1) | VRT (mmHg) | VeS1 (l min−1 mmHg−1) | T2 (mmHg) | VeS2 (l min−1 mmHg−1) | VeB (l min−1) | VRT (mmHg) | VeS1 (l min−1 mmHg−1) | T2 (mmHg) | VeS2 (l min−1 mmHg−1) |
| L1 | 6.6 | 34.0 | 2.8 | 38.0 | 5.3 | 6.8 | 29.0 | 1.9 | 32.0 | 10.4 |
| L2 | 2.9 | 40.0 | 1.8 | 48.0 | 4.5 | 1.8 | 33.0 | 2.7 | 43.0 | 12.8 |
| L3 | 6.5 | 31.8 | 2.7 | 35.4 | 8.8 | 4.7 | 26.0 | 1.0 | 29.0 | 7.8 |
| L4 | 5.3 | 39.0 | 3.5 | 44.0 | 11.1 | 9.5 | 29.0 | 5.0 | 32.5 | 13.2 |
| L5 | 4.1 | 34.0 | 3.0 | 40.0 | 15.9 | 5.7 | 28.0 | 3.4 | 32.0 | 12.3 |
| L6 | 4.3 | 33.0 | 3.2 | 41.0 | 14.1 | 5.9 | 28.0 | 2.7 | 33.0 | 23.0 |
| Mean (s.e.m.) | 4.9 (0.5)† | 35.3 (1.3)* | 2.8 (0.2)† | 41.1 (1.7) | 9.9 (1.7)† | 5.7 (0.9)† | 28.8 (0.9)* | 2.8 (0.5)† | 33.6 (1.8) | 13.2 (1.9)† |
| Andean highlanders |
||||||||||
Isoxic hyperoxic tests ( mmHg) mmHg) |
Isoxic hypoxic tests ( mmHg) mmHg) |
|||||||||
| Subject | VeB (l min−1) | VRT (mmHg) | VeS (l min−1 mmHg−1) | VeB (l min−1) | VRT (mmHg) | VeS (l min−1 mmHg−1) | ||||
| H1 | 12.9 | 39.0 | 1.5 | 16.0 | 36.0 | 1.9 | ||||
| H2 | 8.1 | 36.3 | 2.7 | 23.2 | 30.3 | 3.0 | ||||
| H3 | 9.6 | 32.0 | 1.0 | 11.7 | 32.0 | 0.8 | ||||
| H4 | 11.9 | 35.5 | 2.2 | 9.8 | 30.0 | 2.2 | ||||
| H6 | 13.8 | 39.0 | 1.8 | 10.2 | 31.0 | 2.1 | ||||
| H7 | 9.6 | 31.0 | 1.6 | 14.1 | 30.0 | 2.3 | ||||
| H8 | 10.0 | 43.5 | 2.2 | 11.9 | 34.0 | 2.2 | ||||
| H9 | 8.2 | 35.0 | 1.4 | 13.8 | 30.5 | 1.6 | ||||
| Mean (s.e.m.) | 10.5 (0.7)† | 36.4 (1.3)* | 1.8 (0.2)† | 13.8 (1.4)† | 31.7 (0.7)* | 2 (0.2)† | ||||
Mean ±s.e.m. values for each condition and subject populations are compared with (*) values indicating differences between hypoxic and hyperoxic tests within populations and (†) values indicating differences between populations determined by repeated measures ANOVA (P < 0.05). VeS in highlanders was compared against both VeS1 and VeS2 of lowlanders.
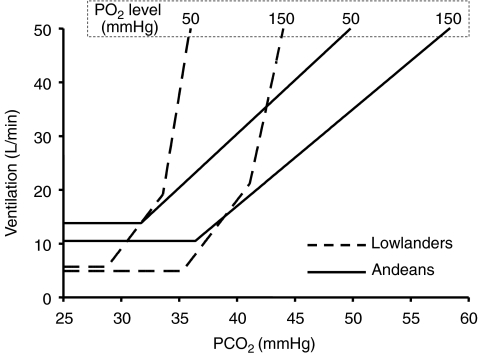
Figure 3. Mean breath-by-breath ventilation vs.  during isoxic hyperoxic and hypoxic Duffin's rebreathing tests for all Andean highlander (continuous lines) and lowlander (dashed lines) subjects.
during isoxic hyperoxic and hypoxic Duffin's rebreathing tests for all Andean highlander (continuous lines) and lowlander (dashed lines) subjects.
Note the presence of two ventilatory sensitivity slopes, VeS1 and VeS2, in acclimatized lowlanders, while highlander responses have only one slope.
Highlanders had greater basal ventilation (hyperoxic: 10.5 ± 0.7 vs. 4.9 ± 0.5 l min−1, P= 0.002; hypoxic: 13.8 ± 1.4 vs. 5.7 ± 0.9 l min−1, P < 0.001), similar ventilatory recruitment thresholds (P= 0.209), and lower ventilatory sensitivities to CO2 than lowlanders, even when compared to first (VeS1, P= 0.038) and second (VeS2, P < 0.001) lowlander sensitivities.
Hypoxia decreased VRT within both populations (highlanders: 36.4 ± 1.3 to 31.7 ± 0.7 mmHg, P < 0.001; lowlanders: 35.3 ± 1.3 to 28.8 ± 0.9 mmHg, P < 0.001), but it had no statistically detectable effect on basal ventilation (P= 0.12) or ventilatory sensitivities in either population (P= 0.684).
Within lowlanders, VeS2 was substantially greater than VeS1 at both isoxic tensions (hyperoxic: 9.9 ± 1.7 vs. 2.8 ± 0.2 l min−1 mmHg−1, P= 0.005; hypoxic: 13.2 ± 1.9 vs. 2.8 ± 0.5 l min−1 mmHg−1, P < 0.001), although hypoxia had no statistically detectable effect on either of the sensitivities (P= 0.192).
Discussion
This study is the first direct comparison of all three components of the ‘Oxford’ control of breathing model (Fig. 1) (Lloyd & Cunningham, 1963), specifically looking at the central and peripheral chemoreflex drives to breathe and the non-chemoreflex drive to breathe in acclimatized lowlanders and Andean highlanders. As Fig. 3 illustrates, the control of breathing in acclimatized lowlanders differed from that of Andean highlanders in several aspects that are discussed below.
Non-chemoreflex drives to breathe
Basal ventilation in acclimatized lowlanders was lower than that in highlanders at both isoxic tensions. Since basal sub-VRT ventilation represents non-chemoreflex drives to breathe (Mohan et al. 1999; Duffin et al. 2000), our findings suggest that the non-chemoreflex drive to breathe is either increased in Andean highlanders compared to lowlanders, or that the acclimatization process results in an early suppression of the non-chemoreflex drives to breathe in lowlanders. The latter hypothesis is unlikely, as five days of hypoxic exposure have been shown to have no effect on basal ventilation in acclimatizing lowlanders (Somogyi et al. 2005); and basal ventilations measured by Somogyi et al. (hyperoxic: 2.9 ± 0.7 l min−1, hypoxic: 4.5 ± 1.1 l min−1) are comparable to those measured in the present study (hyperoxic: 4.9 ± 0.5 l min−1, hypoxic: 5.7 ± 0.9 l min−1). Furthermore, studies using 3 h of isobaric, isocapnic hypoxia (Mahamed et al. 2003) and 20 min of intermittent isobaric, isocapnic hypoxia for 2 weeks (Mahamed & Duffin, 2001) also found no changes in basal ventilation in lowlanders residing at sea-level. Unfortunately, the lack of sea-level control data in our lowlanders does not allow for direct comparison between their resting sea-level basal ventilations and those following 10 days of altitude exposure. The purpose of this study was to compare control of breathing in Andean highlanders to that of acclimatized lowlanders, so sea-level baseline data for acclimatized lowlanders was not collected during the present study. An investigation of acclimatization and the control of breathing parameters in lowlanders has been previously reported (Somogyi et al. 2005) and found no change in basal ventilation. We therefore cannot conclusively rule out the possibility of acclimatization-induced depression of the non-chemoreflex drive to breathe in lowlanders as a cause of their lower basal ventilations relative to highlanders.
Alternatively, the relatively high basal ventilation in highlanders can be explained by their higher non-chemoreflex drives to breathe compared to acclimatizing lowlanders. One source of this drive to breathe could be the carotid body, which may increase its basal level of activity in response to chronic hypoxia. Evidence for this notion is the carotid body hypertrophy observed in highlanders (Arias-Stella & Valcarcel, 1976; Kay & Laidler, 1977; Heath et al. 1985; Khan et al. 1988; Lahiri et al. 2000). The change in carotid body function can be the result of chronic exposure to hypobaric hypoxia and, if found to be true in a larger case series and in other high altitude populations, could represent a form of adaptation to chronic hypoxia.
Another source of the increased non-chemoreflex drive to breathe in Andean highlanders could be related to their experimental anxiety due to unfamiliarity with respiratory experiments, especially since all lowlander subjects were members of the respiratory research laboratory who were familiar with respiratory experiments. Interestingly, the basal ventilations observed in Andean highlanders in the present study are comparable to those measured in Himalayan highlanders and in sea-level lowlanders (university students) who are naive to respiratory experiments (Jensen et al. 2005, 2010; Slessarev et al. 2010). Furthermore, all Andean highlanders were familiarized with the rebreathing experiments on their first visit to the laboratory (see Methods), which should have reduced their experimental anxiety.
Since acclimatized lowlanders in the present study were experienced respiratory physiologists, it is possible that repeated exposure to rebreathing experiments has resulted in the attenuation of their experimental anxiety and basal ventilations to levels below those achieved during the brief familiarization protocol used with highlanders in the present study. However, recent repeatability studies failed to show such attenuation in experimentally naive sea-level lowlanders (Jensen et al. 2010). Lastly, other methods of studying the control of breathing, including classic HVR tests, are affected by experimental anxiety to the same extent as the current experiments. As a result, the reported basal ventilations are likely to be the best representation of the non-chemoreflex drives to breathe in the studied populations during the described testing conditions. Further studies with larger sample sizes may offer an alternative explanation for the observed phenomena.
Pattern of ventilatory response to CO2
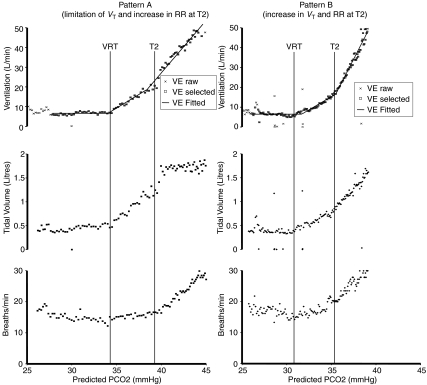
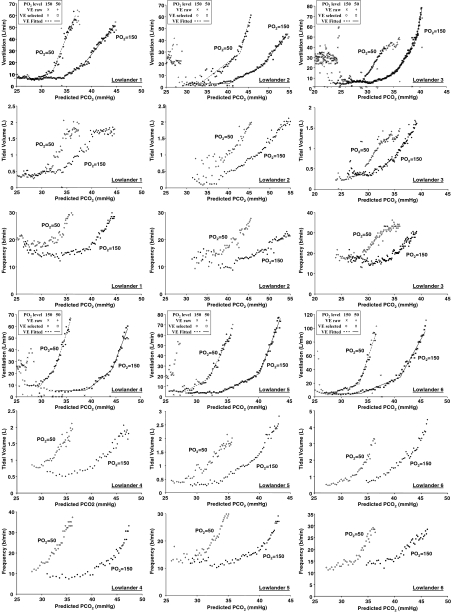
There was a distinct difference in the pattern of ventilatory response to CO2 between the two populations; in lowlanders it was characterized by two distinct sensitivities (VeS1 and VeS2) separated by a threshold (T2), as compared to a single sensitivity (VeS) in highlanders (Fig. 2). Interestingly, VeS2 was greater than VeS1 in lowlanders, and both of these were greater than the VeS of highlanders (Table 2). The occasional presence of two ventilatory sensitivities separated by a threshold has been previously reported in lowlanders residing at sea-level (Duffin et al. 2000; Duffin & Mahamed, 2003), where it has been attributed to a change in breathing pattern, with tidal volume reaching a plateau and further increase in ventilation with increasing CO2 driven by an increase in respiratory rate. In such a case the second threshold, T2, has therefore been termed a patterning threshold (Duffin et al. 2000).
The presence of T2 and two sensitivities in acclimatizing lowlanders has only been observed once previously (Burgess et al. 2008), with another study identifying only the VRT and one ventilatory sensitivity (Somogyi et al. 2005). Further analysis of our data shows that T2 corresponds to a change in pattern of breathing in all of our subjects. However, only one subject (L1) showed the classic pattern of tidal volume plateau and an augmented increase in respiratory rate (Fig. 4 pattern A). The rest of the subjects displayed an augmentation in the rate of rise in both tidal volume and respiratory rate at T2 (Fig. 5), such that sensitivity post T2 (VeS2) is greater than prior to T2 (VeS1) (Fig. 4 pattern B). The reason for this newly observed pattern of ventilatory change at T2 is unclear and may be simply a characteristic of our particular small group of subjects. With T2 marking a simultaneous increase in the rate of rise of both tidal volume and respiratory rate in response to increasing CO2, it appears that there is an overall increase in the ventilatory drive that occurs at T2.
Figure 4. Two types of patterning threshold (T2) were identified in acclimatizing lowlanders.
Pattern A has been previously seen in lowlanders at sea-level (Duffin & Mahamed, 2003) and is characterized by a decrease in the rate of rise of tidal volume (VT) and an increase in the rate of rise in respiratory rate (RR), leading to overall increase in ventilatory sensitivity to CO2 above T2. Pattern B is characterized by an increased rate of rise of both VT and RR above T2, leading to an overall increase in ventilatory sensitivity to CO2 above T2. Most of the acclimatizing lowlander subjects (5 out of 6) displayed pattern B.
Figure 5. Patterns of ventilatory response to hypoxic and hyperoxic rebreathing from all lowlander subjects illustrating the patterning thresholds.
Note that only Lowlander 1 displayed the classic patterning threshold with tidal volume reaching a maximum so that a further increase in ventilation was driven by an increase in respiratory rate alone. The rest of lowlanders increased both their tidal volume and respiratory rate at T2.
One possible explanation for this may be that the acclimatizing lowlanders in the group that we tested have different central and peripheral chemoreflex thresholds (Mohan & Duffin, 1997), with the central threshold corresponding to VRT and the peripheral threshold corresponding to T2. The lack of T2 in our highlander population may suggest either that their central and peripheral thresholds are similar, as is the case in most lowlanders studied at sea-level, or that the peripheral threshold is absent altogether due to lack of peripheral chemosensitivity to CO2. The latter hypothesis is supported by the absence of a hypoxia-induced increase in ventilatory CO2 sensitivity in highlanders (Fig. 3), which is a characteristic pattern observed in lowlanders at sea-level (Mohan & Duffin, 1997). This absence may be attributed to a change in function of the carotid body and related to the observed hypertrophy and histological changes of this organ in highlanders (Arias-Stella & Valcarcel, 1976; Kay & Laidler, 1977; Heath et al. 1985; Khan et al. 1988; Lahiri et al. 2000).
Ventilatory sensitivity to CO2
A third difference in the control of breathing between acclimatized lowlanders and Andean highlanders was the relatively greater ventilatory sensitivity to CO2 in acclimatized lowlanders both below (VeS1) and above (VeS2) T2, although the difference between VeS1 in lowlanders and VeS in highlanders was significant only during hyperoxic (P= 0.029), but not hypoxic (P= 0.100) tests (Table 2). However, this non-significance may have been the result of a type-2 error due to the small number of subjects in the study (see ‘Methodological considerations’ below). Since hyperoxia effectively silences the peripheral chemoreceptors (Lloyd & Cunningham, 1963; Mohan & Duffin, 1997), the ventilatory sensitivities during hyperoxic rebreathing tests primarily represent central chemoreceptor sensitivity (Duffin, 2007). Our results suggest that this sensitivity is greater in acclimatized lowlanders than in highlanders, although the mechanism responsible for this difference is unclear.
Previous studies of acclimatizing lowlanders showed either no change (Somogyi et al. 2005) or an increase (Ainslie & Burgess, 2008) in the sensitivity of the ventilatory response to CO2 following acclimatization to high altitude. However, these two studies differed in the duration and altitude of hypoxic exposure (5 days at 3480 m in Somogyi et al., and 9 days at >5000 m in Ainslie & Burgess), which could explain the observed difference in the results. Our study was done at an altitude (3850 m) similar to that in Somogyi et al. but the duration of hypoxic exposure in our study (10 days) was closer to that in Ainslie & Burgess, and the ventilatory CO2 sensitivities measured in our study (hyperoxic: 2.8 ± 0.2 l min−1 mmHg−1; hypoxic: 2.0 ± 0.2 l min−1 mmHg−1) are closer to those measured by Somogyi et al. (hyperoxic: 2.3 ± 0.5 l min−1 mmHg−1; hypoxic: 3.7 ± 0.8 l min−1 mmHg−1) than those measured by Ainslie & Burgess (hyperoxic: 4.8 ± 3.5 l min−1 mmHg−1; hypoxic: 5.4 ± 2.2 l min−1 mmHg−1). This comparison suggests that the change in ventilatory CO2 sensitivity in acclimatizing lowlanders is affected more by the absolute altitude of exposure (with higher altitude increasing ventilatory sensitivity to CO2) than by its duration.
Ventilatory recruitment thresholds
There was no difference in the VRT between the acclimatizing lowlanders and the Andean highlanders. A previous study (Somogyi et al. 2005) showed that acclimatization to high altitude in lowlanders is accompanied by a decrease in the VRT from sea-level values. This finding is also supported by the previously observed leftward shift in the x-axis intercept of the ventilation– relationship with acclimatization (Ainslie & Burgess, 2008). The leftward shift of the VRT can be attributed to an altered [H+]–
relationship with acclimatization (Ainslie & Burgess, 2008). The leftward shift of the VRT can be attributed to an altered [H+]– relationship that results from respiratory alkalosis caused by hypoxia-induced hyperventilation. The change in the [H+]–
relationship that results from respiratory alkalosis caused by hypoxia-induced hyperventilation. The change in the [H+]– relationship is such that the same [H+] corresponds to different
relationship is such that the same [H+] corresponds to different  values at sea-level and at altitude, with the altitude value being lower (Duffin, 2005).
values at sea-level and at altitude, with the altitude value being lower (Duffin, 2005).
The VRTs measured in our study were similar between acclimatized lowlanders and Andean highlanders at both isoxic tensions (hyperoxic: 35.3 ± 1.3 vs. 36.4 ± 1.3 mmHg; hypoxic: 28.8 ± 0.9 vs. 31.7 ± 0.7 mmHg, P= 0.209), while both were lower than those measured by Somogyi et al. (2005) (hyperoxic: 40.1 ± 1.8 mmHg; hypoxic: 33.2 ± 1.6 mmHg). Since lowlanders in our study were exposed to a similar altitude, but for a longer duration of hypoxia (10 vs. 5 days), compared to the lowlanders in the study by Somogyi et al. the observed results suggest that the increased duration of hypoxic exposure results in progressive lowering of VRTs in acclimatizing lowlanders so that they are similar to those of adapted highlanders after 10 days. It is therefore likely that an ongoing correction of respiratory-induced changes in acid–base status is responsible for the observed changes in VRT; however, this hypothesis requires further investigation.
Ventilatory response to hypoxia
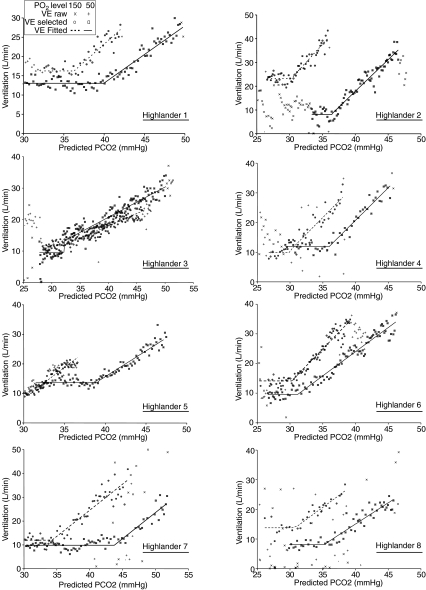
The ventilatory response to hypoxia in both populations was marked by a decrease in VRT (highlanders: 36.4 ± 1.3 to 31.7 ± 0.7 mmHg, P < 0.001; lowlanders: 35.3 ± 1.3 to 28.8 ± 0.9 mmHg, P < 0.001), with no change in basal ventilation (P= 0.12) or ventilatory sensitivities in either population (P= 0.684). This is distinctly different from the response seen in lowlanders at sea-level, who were previously shown to respond to hypoxia by increasing their ventilatory sensitivity to CO2 (Mohan & Duffin, 1997). Interestingly, Andean highlanders, like Himalayan highlanders from our companion study (Slessarev et al. 2010), displayed marked heterogeneity in their ventilatory response to hypoxia (Fig. 6). While most highlanders seemed to decrease their VRT in response to hypoxia (the only change that was statistically significant), some also increased their basal ventilation without change in sensitivity or VRT, while others increased their sensitivity in addition to a change in VRT. Our limited sample size prevents us from commenting on whether these observations represent certain subsets within the highlander population, but it demonstrates that the ventilatory response to hypoxia in highlanders may be heterogeneous, which could further confound the interpretation of ventilatory control studies. More studies with larger sample sizes are required to further elucidate the nature of this heterogeneity.
Figure 6. Hypoxic and hyperoxic rebreathing responses from all highlander subjects.
Note the different patterns of responses between hypoxic and hyperoxic rebreathing (see text for more detailed discussion).
A decrease in VRT with hypoxia without an increase in sensitivity has been reported previously (Somogyi et al. 2005), suggesting that this pattern of hypoxic response is ubiquitous in acclimatizing lowlanders. In our rebreathing tests the difference in ventilatory CO2 sensitivities between hyperoxic and hypoxic tests represents the contribution of the peripheral chemoreflex. The absence of a hypoxia-induced change in ventilatory CO2 sensitivity in acclimatized lowlanders and Andean highlanders therefore suggests that the hypoxic enhancement of peripheral CO2 chemosensitivity is blunted.
Summary
Our findings suggest that the control of breathing in acclimatizing lowlanders is distinctly different from that in Andean highlanders. Specifically, acclimatizing lowlanders have lower basal ventilations and relatively lower non-chemoreflex drives to breathe, higher ventilatory CO2 sensitivities, and show a patterning threshold. The nature of higher basal ventilations in highlanders is not clear and further dedicated studies are required to identify its source. However, there are also some similarities between Andean highlanders and acclimatizing lowlanders. For example, both populations lack the hypoxia-induced increase in ventilatory CO2 sensitivity seen in sea-level lowlanders, which suggests that peripheral CO2–O2 interaction is blunted in these populations. Instead, hypoxia induces a decrease in their VRT.
Comparison of our data with previous studies suggests that a longer duration of hypoxic exposure in lowlanders results in a progressive decrease in VRTs, such that those of acclimatizing lowlanders are similar to those of Andean highlanders following 10 days at high altitude, while exposure of only 5 days results in VRTs that are higher than those of Andean highlanders, but lower than those of sea-level lowlanders (Somogyi et al. 2005). It is not clear which of the observed differences or similarities between the two populations are adaptive to life at high altitude. However, further studies and comparison of this data with other high altitude populations may help to identify specific changes in the control of breathing that may represent adaptive strategies.
Methodological considerations
The absence of statistically significant changes in basal ventilations and ventilatory sensitivities with hypoxia should be interpreted with caution, since the power of the performed within-population tests was relatively low due to small sample size in the present study. For instance, in highlanders a change in basal ventilation of 30% and in ventilatory sensitivity of 45% with hypoxia would have been required to produce tests with a power greater than 80%. Similarly, in lowlanders a change in basal ventilation of 52%, VeS1 of 36% and VeS2 of 84% with hypoxia would have been required to produce tests with a power greater than 80%. We therefore suggest that the general patterns of change should be taken into account when interpreting results.
We used Duffin's rebreathing technique (Mohan et al. 1999; Duffin et al. 2000) to measure the chemoreflex and non-chemoreflex drives to breathe. In contrast, previous studies of ventilatory control in acclimatizing lowlanders and highlanders have employed either steady-state methods or Read's rebreathing; the latter only measures the hyperoxic ventilatory response to CO2. Although these methods provide useful information about ventilatory sensitivities to O2 and CO2, the separation of the contributions of individual components of the control of breathing model is difficult (Fig. 1), thereby complicating comparison between studies and between populations (Duffin, 2007). Furthermore, increasing cerebral blood flow secondary to hypoxia or hypercapnia results in washout of CO2 and lower [H+] at the central chemoreceptors with steady-state methods, thereby attenuating the measured ventilatory response (Berkenbosch et al. 1989; Mohan et al. 1999). Since control of cerebral blood flow in highlanders may be altered (Norcliffe et al. 2005; Appenzeller et al. 2006; Claydon et al. 2008), the effects of this attenuation are hard to predict, which may further complicate the comparison of results between populations (Ainslie & Duffin, 2009).
We only studied Andean highlanders residing at one altitude (3850 m). Previous studies have shown that the level of altitude residence may affect control of breathing in highlanders (Curran et al. 1995), and this factor requires further evaluation, possibly with the rebreathing technique employed in the current study. Moreover, the ancestry of highlanders, which may have an effect on the control of breathing (Curran et al. 1997; Brutsaert et al. 2005; Beall, 2007), was not established in the present study. Future studies using Duffin's rebreathing method in larger samples of highlanders with a known pedigree of highlander ancestry may help identify specific aspects of the control of breathing model that are adaptive to life at high altitude.
Lastly, recent studies suggest that there are several patterns of adaptation to life at high altitude (Moore, 2000; Beall, 2007), with probable differences in the control of breathing between different high altitude populations. It would be interesting to compare the results of the present study with rebreathing studies from other high altitude populations in an attempt to identify specific differences in the control of breathing between populations.
Conclusion
The control of breathing in acclimatizing lowlanders is distinctly different from that in Andean highlanders, although some similarities exist. Compared to Andean highlanders, acclimatized lowlanders have relatively lower non-chemoreflex drives to breathe and a greater ventilatory response to CO2. Both highlanders and acclimatized lowlanders display blunted peripheral CO2 chemosensitivity and respond to hypoxia with a decrease in ventilatory recruitment threshold.
Acknowledgments
We thank Thornhill Research Inc., Toronto, Canada and Canadian Institute for Health Research for helping to fund the expedition to LaPaz, Bolivia. We thank Professors Gustavo Zubieta-Castillo and Gustavo Zubieta-Calleja from the High Altitude Pathology Institute, Clinica IPPA, Zubieta University in La Paz, Bolivia for providing us with a laboratory space and assisting us with experiments and highlander subject recruitment. We also thank volunteer subjects from LaPaz, Bolivia and from Isocapnia Laboratory High Altitude Team for volunteering their time in the name of science. We thank Mrs Cathie Kessler and Mr Tim Clark for their invaluable help in preparing for the expedition. Disclosures. Three of the study authors (M.S., D.P. and J.A.F.) contributed to the development of the RespirAct, a device used for data acquisition during the Bolivia expedition. These authors stand to gain financially if the device is successfully commercialized by Thornhill Research Inc., a University of Toronto/University Health Network-related company.
Glossary
Abbreviations
- HVR
hypoxic ventilatory response

end-tidal


end-tidal

- T2
second ventilatory threshold
- VeB
sub-ventilatory recruitment threshold basal ventilation
- VeS
ventilatory sensitivity to CO2 in highlanders
- VeS1
first ventilatory sensitivity to CO2 in lowlanders
- VeS2
second ventilatory sensitivity to CO2 in lowlanders
- VRT
ventilatory recruitment threshold
Author contributions
All the listed authors were involved in the conception and design, or analysis and interpretation of data, and drafting the article or revising it critically for important intellectual content. All authors approved the final version of the manuscript.
References
- Ainslie PN, Burgess KR. Cardiorespiratory and cerebrovascular responses to hyperoxic and hypoxic rebreathing: effects of acclimatization to high altitude. Respir Physiol Neurobiol. 2008;161:201–209. doi: 10.1016/j.resp.2008.02.003. [DOI] [PubMed] [Google Scholar]
- Ainslie PN, Duffin J. Integration of cerebrovascular CO2 reactivity and chemoreflex control of breathing: mechanisms of regulation, measurement, and interpretation. Am J Physiol Regul Integr Comp Physiol. 2009;296:R1473–R1495. doi: 10.1152/ajpregu.91008.2008. [DOI] [PubMed] [Google Scholar]
- Ainslie PN, Kolb JC, Ide K, Poulin MJ. Effects of five nights of normobaric hypoxia on the ventilatory responses to acute hypoxia and hypercapnia. Respir Physiol Neurobiol. 2003;138:193–204. doi: 10.1016/s1569-9048(03)00190-3. [DOI] [PubMed] [Google Scholar]
- Aldenderfer MS. Moving up in the world. Am Sci. 2003;91:542–549. [Google Scholar]
- Appenzeller O, Claydon VE, Gulli G, Qualls C, Slessarev M, Zenebe G, Gebremedhin A, Hainsworth R. Cerebral vasodilatation to exogenous NO is a measure of fitness for life at altitude. Stroke. 2006;37:1754–1758. doi: 10.1161/01.STR.0000226973.97858.0b. [DOI] [PubMed] [Google Scholar]
- Arias-Stella J, Valcarcel J. Chief cell hyperplasia in the human carotid body at high altitudes; physiologic and pathologic significance. Hum Pathol. 1976;7:361–373. doi: 10.1016/s0046-8177(76)80052-4. [DOI] [PubMed] [Google Scholar]
- Beall CM. Two routes to functional adaptation: Tibetan and Andean high-altitude natives. Proc Natl Acad Sci U S A. 2007;104(Suppl 1):8655–8660. doi: 10.1073/pnas.0701985104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beall CM, Strohl KP, Blangero J, Williams-Blangero S, Almasy LA, Decker MJ, Worthman CM, Goldstein MC, Vargas E, Villena M, Soria R, Alarcon AM, Gonzales C. Ventilation and hypoxic ventilatory response of Tibetan and Aymara high altitude natives. Am J Phys Anthropol. 1997;104:427–447. doi: 10.1002/(SICI)1096-8644(199712)104:4<427::AID-AJPA1>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- Berkenbosch A, Bovill JG, Dahan A, DeGoede J, Olievier IC. The ventilatory CO2 sensitivities from Read's rebreathing method and the steady-state method are not equal in man. J Physiol. 1989;411:367–377. doi: 10.1113/jphysiol.1989.sp017578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brutsaert TD. Population genetic aspects and phenotypic plasticity of ventilatory responses in high altitude natives. Respir Physiol Neurobiol. 2007;158:151–160. doi: 10.1016/j.resp.2007.03.004. [DOI] [PubMed] [Google Scholar]
- Brutsaert TD, Parra EJ, Shriver MD, Gamboa A, Rivera-Ch M, Leon-Velarde F. Ancestry explains the blunted ventilatory response to sustained hypoxia and lower exercise ventilation of Quechua altitude natives. Am J Physiol Regul Integr Comp Physiol. 2005;289:R225–R234. doi: 10.1152/ajpregu.00105.2005. [DOI] [PubMed] [Google Scholar]
- Burgess K, Burgess K, Subedi P, Ainslie P, Topor Z, Whitelaw W. Prediction of periodic breathing at altitude. In: Poulin MJ, Wilson RJA, editors. Integration in Respiratory Control: From Genes to Systems. Springer; 2008. [Google Scholar]
- Chiodi H. Respiratory adaptations to chronic high altitude hypoxia. J Appl Physiol. 1957;10:81–87. doi: 10.1152/jappl.1957.10.1.81. [DOI] [PubMed] [Google Scholar]
- Claydon VE, Gulli G, Slessarev M, Appenzeller O, Zenebe G, Gebremedhin A, Hainsworth R. Cerebrovascular responses to hypoxia and hypocapnia in Ethiopian high altitude dwellers. Stroke. 2008;39:336–342. doi: 10.1161/STROKEAHA.107.491498. [DOI] [PubMed] [Google Scholar]
- Cudkowicz L, Spielvogel H, Zubieta G. Respiratory studies in women at high altitude (3,600 m or 12,200 ft and 5,200 m or 17,200 ft) Respiration. 1972;29:393–426. doi: 10.1159/000192911. [DOI] [PubMed] [Google Scholar]
- Cunningham DJ. Studies on arterial chemoreceptors in man. J Physiol. 1987;384:1–26. doi: 10.1113/jphysiol.1987.sp016440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curran LS, Zhuang J, Droma T, Land L, Moore LG. Hypoxic ventilatory responses in Tibetan residents of 4400 m compared with 3658 m. Respir Physiol. 1995;100:223–230. doi: 10.1016/0034-5687(94)00110-l. [DOI] [PubMed] [Google Scholar]
- Curran LS, Zhuang J, Sun SF, Moore LG. Ventilation and hypoxic ventilatory responsiveness in Chinese-Tibetan residents at 3658 m. J Appl Physiol. 1997;83:2098–2104. doi: 10.1152/jappl.1997.83.6.2098. [DOI] [PubMed] [Google Scholar]
- Duffin J. Role of acid-base balance in the chemoreflex control of breathing. J Appl Physiol. 2005;99:2255–2265. doi: 10.1152/japplphysiol.00640.2005. [DOI] [PubMed] [Google Scholar]
- Duffin J. Measuring the ventilatory response to hypoxia. J Physiol. 2007;584:285–293. doi: 10.1113/jphysiol.2007.138883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duffin J, Mahamed S. Adaptation in the respiratory control system. Can J Physiol Pharmacol. 2003;81:765–773. doi: 10.1139/y03-049. [DOI] [PubMed] [Google Scholar]
- Duffin J, Mohan RM, Vasiliou P, Stephenson R, Mahamed S. A model of the chemoreflex control of breathing in humans: model parameters measurement. Respir Physiol. 2000;120:13–26. doi: 10.1016/s0034-5687(00)00095-5. [DOI] [PubMed] [Google Scholar]
- Fatemian M, Gamboa A, Leon-Velarde F, Rivera-Ch M, Palacios JA, Robbins PA. Selected contribution: Ventilatory response to CO2 in high-altitude natives and patients with chronic mountain sickness. J Appl Physiol. 2003;94:1279–1287. doi: 10.1152/japplphysiol.00859.2002. [DOI] [PubMed] [Google Scholar]
- Fatemian M, Robbins PA. Human ventilatory response to CO2 after 8 h of isocapnic or poikilocapnic hypoxia. J Appl Physiol. 1998;85:1922–1928. doi: 10.1152/jappl.1998.85.5.1922. [DOI] [PubMed] [Google Scholar]
- Fink BR. Influence of cerebral activity in wakefulness on regulation of breathing. J Appl Physiol. 1961;16:15–20. doi: 10.1152/jappl.1961.16.1.15. [DOI] [PubMed] [Google Scholar]
- Gamboa A, Leon-Velarde F, Rivera-Ch M, Palacios JA, Pragnell TR, O’Connor DF, Robbins PA. Selected contribution: Acute and sustained ventilatory responses to hypoxia in high-altitude natives living at sea level. J Appl Physiol. 2003;94:1255–1262. doi: 10.1152/japplphysiol.00856.2002. [DOI] [PubMed] [Google Scholar]
- Heath D, Smith P, Fitch R, Harris P. Comparative pathology of the enlarged carotid body. J Comp Pathol. 1985;95:259–271. doi: 10.1016/0021-9975(85)90012-x. [DOI] [PubMed] [Google Scholar]
- Howard LS, Robbins PA. Alterations in respiratory control during 8 h of isocapnic and poikilocapnic hypoxia in humans. J Appl Physiol. 1995;78:1098–1107. doi: 10.1152/jappl.1995.78.3.1098. [DOI] [PubMed] [Google Scholar]
- Jensen D, Mask G, Tschakovsky ME. Variability of the ventilatory response to Duffin's modified hyperoxic and hypoxic rebreathing procedure in healthy awake humans. Respir Physiol Neurobiol. 2010;170:185–97. doi: 10.1016/j.resp.2009.12.007. [DOI] [PubMed] [Google Scholar]
- Jensen D, Wolfe LA, O’Donnell DE, Davies GA. Chemoreflex control of breathing during wakefulness in healthy men and women. J Appl Physiol. 2005;98:822–828. doi: 10.1152/japplphysiol.01208.2003. [DOI] [PubMed] [Google Scholar]
- Kay JM, Laidler P. Hypoxia and the carotid body. J Clin Pathol Suppl (R Coll Pathol) 1977;11:30–44. doi: 10.1136/jcp.s3-11.1.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan Q, Heath D, Smith P, Norboo T. The histology of the carotid bodies in highlanders from Ladakh. Int J Biometeorol. 1988;32:254–259. doi: 10.1007/BF01080024. [DOI] [PubMed] [Google Scholar]
- Kumar P, Bin-Jaliah I. Adequate stimuli of the carotid body: more than an oxygen sensor? Respir Physiol Neurobiol. 2007;157:12–21. doi: 10.1016/j.resp.2007.01.007. [DOI] [PubMed] [Google Scholar]
- Lahiri S. Alveolar gas pressures in man with life-time hypoxia. Respir Physiol. 1968;4:373–386. doi: 10.1016/0034-5687(68)90042-x. [DOI] [PubMed] [Google Scholar]
- Lahiri S, Kao FF, Velasquez T, Mart-Inez C, Pezzia W. Irreversible blunted respiratory sensitivity to hypoxia in high altitude natives. Respir Physiol. 1969;6:360–374. doi: 10.1016/0034-5687(69)90034-6. [DOI] [PubMed] [Google Scholar]
- Lahiri S, Rozanov C, Cherniack NS. Altered structure and function of the carotid body at high altitude and associated chemoreflexes. High Alt Med Biol. 2000;1:63–74. doi: 10.1089/152702900320694. [DOI] [PubMed] [Google Scholar]
- Lloyd BB, Cunningham DJC. A quantitative approach to the regulation of human respiration. In: Cunningham DJC, Lloyd BB, editors. The Regulation of Human Respiration. Oxford: Blackwell; 1963. pp. 331–349. [Google Scholar]
- Mahamed S, Cunningham DA, Duffin J. Changes in respiratory control after three hours of isocapnic hypoxia in humans. J Physiol. 2003;547:271–281. doi: 10.1113/jphysiol.2002.030965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahamed S, Duffin J. Repeated hypoxic exposures change respiratory chemoreflex control in humans. J Physiol. 2001;534:595–603. doi: 10.1111/j.1469-7793.2001.00595.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mateika JH, Mendello C, Obeid D, Badr MS. Peripheral chemoreflex responsiveness is increased at elevated levels of carbon dioxide after episodic hypoxia in awake humans. J Appl Physiol. 2004;96:1197–1205. doi: 10.1152/japplphysiol.00573.2003. [DOI] [PubMed] [Google Scholar]
- Mohan R, Duffin J. The effect of hypoxia on the ventilatory response to carbon dioxide in man. Respir Physiol. 1997;108:101–115. doi: 10.1016/s0034-5687(97)00024-8. [DOI] [PubMed] [Google Scholar]
- Mohan RM, Amara CE, Cunningham DA, Duffin J. Measuring central-chemoreflex sensitivity in man: rebreathing and steady-state methods compared. Respir Physiol. 1999;115:23–33. doi: 10.1016/s0034-5687(99)00003-1. [DOI] [PubMed] [Google Scholar]
- Moore LG. Comparative human ventilatory adaptation to high altitude. Respir Physiol. 2000;121:257–276. doi: 10.1016/s0034-5687(00)00133-x. [DOI] [PubMed] [Google Scholar]
- Nattie E, Li A. Central chemoreception is a complex system function that involves multiple brain stem sites. J Appl Physiol. 2009;106:1464–1466. doi: 10.1152/japplphysiol.00112.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norcliffe LJ, Rivera-Ch M, Claydon VE, Moore JP, Leon-Velarde F, Appenzeller O, Hainsworth R. Cerebrovascular responses to hypoxia and hypocapnia in high-altitude dwellers. J Physiol. 2005;566:287–294. doi: 10.1113/jphysiol.2005.086629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell FL, Milsom WK, Mitchell GS. Time domains of the hypoxic ventilatory response. Respir Physiol. 1998;112:123–134. doi: 10.1016/s0034-5687(98)00026-7. [DOI] [PubMed] [Google Scholar]
- Read DJ. A clinical method for assessing the ventilatory response to carbon dioxide. Aust Ann Med. 1967;16:20–32. doi: 10.1111/imj.1967.16.1.20. [DOI] [PubMed] [Google Scholar]
- Sato M, Severinghaus JW, Powell FL, Xu FD, Spellman MJ., Jr Augmented hypoxic ventilatory response in men at altitude. J Appl Physiol. 1992;73:101–107. doi: 10.1152/jappl.1992.73.1.101. [DOI] [PubMed] [Google Scholar]
- Schoene RB, Roach RC, Hackett PH, Sutton JR, Cymerman A, Houston CS. Operation Everest II: ventilatory adaptation during gradual decompression to extreme altitude. Med Sci Sports Exerc. 1990;22:804–810. [PubMed] [Google Scholar]
- Severinghaus JW, Bainton CR, Carcelen A. Respiratory insensitivity to hypoxia in chronically hypoxic man. Respir Physiol. 1966;1:308–334. doi: 10.1016/0034-5687(66)90049-1. [DOI] [PubMed] [Google Scholar]
- Shea SA. Behavioural and arousal-related influences on breathing in humans. Exp Physiol. 1996;81:1–26. doi: 10.1113/expphysiol.1996.sp003911. [DOI] [PubMed] [Google Scholar]
- Slessarev M, Prisman E, Ito S, Watson R, Jensen D, Preiss D, Greene R, Norboo T, Stobdan T, Diskit D, Norboo A, Kunzang M, Appenzeller O, Duffin J, Fisher JA. Differences in the control of breathing between Himalayan and sea-level residents. J Physiol. 2010 doi: 10.1113/jphysiol.2009.185504. doi 10.1113/jphysiol.2009.185504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Somogyi RB, Preiss D, Vesely A, Fisher JA, Duffin J. Changes in respiratory control after 5 days at altitude. Respir Physiol Neurobiol. 2005;145:41–52. doi: 10.1016/j.resp.2004.11.004. [DOI] [PubMed] [Google Scholar]
- Sorensen SC, Severinghaus JW. Respiratory sensitivity to acute hypoxia in man born at sea level living at high altitude. J Appl Physiol. 1968;25:211–216. doi: 10.1152/jappl.1968.25.3.211. [DOI] [PubMed] [Google Scholar]
- Torrance RW. Prolegomena. Chemoreception upstream of transmitters. Adv Exp Med Biol. 1996;410:13–38. [PubMed] [Google Scholar]
- Velasquez T, Martinez C, Pezzia W, Gallardo N. Ventilatory effects of oxygen in high altitude natives. Respir Physiol. 1968;5:211–220. doi: 10.1016/0034-5687(68)90088-1. [DOI] [PubMed] [Google Scholar]