Abstract
The gene encoding COMM domain–containing 1 (COMMD1) is a prototypical member of the COMMD gene family that has been shown to inhibit both NF-κB– and HIF-mediated gene expression. NF-κB and HIF are transcription factors that have been shown to play a role in promoting tumor growth, survival, and invasion. In this study, we demonstrate that COMMD1 expression is frequently suppressed in human cancer and that decreased COMMD1 expression correlates with a more invasive tumor phenotype. We found that direct repression of COMMD1 in human cell lines led to increased tumor invasion in a chick xenograft model, while increased COMMD1 expression in mouse melanoma cells led to decreased lung metastasis in a mouse model. Decreased COMMD1 expression also correlated with increased expression of genes known to promote cancer cell invasiveness, including direct targets of HIF. Mechanistically, our studies show that COMMD1 inhibits HIF-mediated gene expression by binding directly to the amino terminus of HIF-1α, preventing its dimerization with HIF-1β and subsequent DNA binding and transcriptional activation. Altogether, our findings demonstrate a role for COMMD1 in tumor invasion and provide a detailed mechanism of how this factor regulates the HIF pathway in cancer cells.
Introduction
COMM domain–containing 1 (COMMD1), the first COMMD family member to be identified, is a pleiotropic factor that participates in multiple processes, including copper metabolism, sodium excretion, inflammatory responses, and adaptation to hypoxia. While COMMD gene family members are present in a variety of organisms, little is known about the functions of the other 9 genes that comprise this family (1). Studies emanating from the identification of COMMD1 as an XIAP-associated factor led to the discovery that COMMD1 is an inhibitor of the proinflammatory transcription factor NF-κB (2). Genetic studies in dogs implicate COMMD1 in copper excretion (3), while gene inactivation in mice indicates that COMMD1 also regulates HIF, a transcription factor that is a master regulator of oxygen homeostasis (4).
HIF controls energy metabolism, angiogenesis, erythropoiesis, and critical events during embryogenesis (5). This transcription factor is composed of a heterodimer between a constitutively expressed β-subunit (HIF-1β or ARNT) and an oxygen-regulated α-subunit (HIF-1α or HIF-2α) that belongs to the basic helix-loop-helix (bHLH), Per, ARNT, SIM (PAS) protein family (6). The bHLH and PAS domains at the amino terminus of HIF subunits are important for heterodimerization and DNA binding. Under normoxia, the levels of HIF-α subunits are low, due to rapid ubiquitination and degradation triggered by oxygen-dependent hydroxylation of proline residues in the oxygen-dependent degradation domain (ODDD) (7, 8). Prolyl hydroxylation of the α-subunits by the prolyl hydroxylase enzymes (PHD1, PHD2, and PHD3) promotes the binding of HIF-α subunits to a multimeric ubiquitin ligase complex, containing the von Hippel-Lindau (VHL) protein and Cul2 (9). Under hypoxic conditions, the activity of PHDs is attenuated, leading to HIF-α accumulation, dimerization with HIF-1β, and transcriptional activation of target genes containing hypoxia responsive elements (HREs).
In addition to its physiological roles, HIF participates in the pathophysiology of several disorders, including cancer, in which enhanced HIF activity is associated with tumor growth, neovascularization, local invasion, metastatic disease, and poor clinical outcomes (5). A variety of mechanisms, including local hypoxia within rapidly growing solid tumors, are thought to lead to HIF activation in cancer. Similarly, while under physiological conditions NF-κB plays critical roles in inflammatory responses and cellular survival to stress, activation of NF-κB is also a frequent occurrence in cancer. In particular, the ability of NF-κB to promote the expression of various antiapoptotic factors is thought to play a major role in the survival of cancer cells (10). In addition, both transcription factors promote cancer progression through the expression of genes involved in metastasis and tumor invasion (11). These gene targets include several genes involved in epithelial-to-mesenchymal transformation (EMT), a developmental program that can be abnormally activated in invasive epithelial malignancies and that is modulated by multiple transcriptional regulators, including both HIF and NF-κB (5, 12, 13).
Based on extensive evidence for the involvement of both HIF and NF-κB in tumor invasion, the dual role of COMMD1 in these pathways suggested the possibility that this factor may be inactivated or repressed in tumors. Here we report that COMMD1 expression is repressed in a variety of human malignancies. We show that decreased COMMD1 expression is accompanied by a more invasive phenotype and the upregulation of genes previously implicated in tumor invasion. Among these, several HIF targets were identified, and consistent with our prior reports, we found that COMMD1 suppression leads to derepressed HIF-mediated transcription. We demonstrate that COMMD1 competes with HIF-1β for binding to HIF-1α, resulting in decreased DNA binding and impaired transcriptional activation by HIF-1. Altogether, this study implicates COMMD1 in tumor invasion and provides additional mechanistic insights into the regulation of HIF.
Results
COMMD1 expression is decreased in cancer.
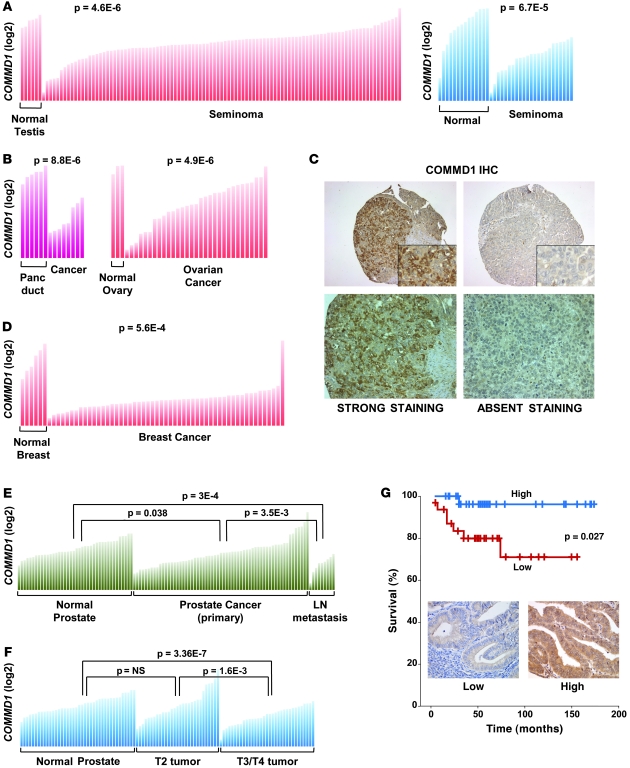
Given the role of COMMD1 as a suppressor of both the NF-κB and HIF pathways, we speculated that expression of COMMD1 might be reduced in the setting of cancer. To explore this notion, we queried the Oncomine database (14) for studies demonstrating differential COMMD1 expression in cancers compared with their counterpart normal tissues. The majority of the studies in the Oncomine database demonstrating differential expression in cancer found decreased COMMD1 expression in tumors, including 2 independent studies in seminoma (15, 16) as well as a study in pancreatic cancer (17) (Figure 1, A and B). COMMD1 expression was also reduced in ovarian cancer (18) (Figure 1B), and consistent with these data, COMMD1 was undetectable by immunohistochemistry staining in 18% of endometrioid and 12% of serous ovarian cancers in a tissue microarray (n = 22 and 26, respectively; see Figure 1C for representative examples). In addition, reduced COMMD1 gene expression involved not only neoplastic cells but also other cell types within the tumor microenvironment, as exemplified by a study using laser capture dissection to examine gene expression in stromal cells in breast cancer (19). Reduced COMMD1 gene expression was evident in stromal cells from primary breast tumors compared with those of normal breast tissue (Figure 1D). Moreover, this study found that COMMD1 was among the top 10% of underexpressed genes in patients that developed a recurrence 5 years later, suggesting that decreased COMMD1 expression might be associated with worse clinical outcomes.
Figure 1. Decreased COMMD1 expression is found in several cancers.
(A and B) COMMD1 expression in individual samples from normal tissues or the corresponding cancer is shown (as normalized expression in log2 scale). Comparisons between groups were performed using the Student’s t test, with P values indicated in each panel. Panc duct, pancreatic duct. (C) Lack of detectable COMMD1 protein expression is frequent in ovarian cancer. Representative examples of COMMD1 immunohistochemical (IHC) staining of ovarian cancer specimens are shown (original magnification, ×200 [top panels]; ×600 [insets]; ×400 [bottom panels]). (D) COMMD1 expression in stromal cells from normal breast tissue and breast cancer is shown in a similar format as in A. (E) COMMD1 expression in normal prostate, prostate cancer, and metastatic prostate cancer specimens from lymph nodes is shown. Comparisons between groups were performed using the Student’s t test, with P values indicated. (F) COMMD1 expression in the same sample set, this time subdivided according to the local tumor T stage. Similar to A, the Student’s t test was performed to compare the groups, and P values are shown. (G) COMMD1 expression by immunohistochemical staining correlates with patient survival in endometrial cancer (n = 63). The survival curves of patients with intense cytoplasmic staining (high, blue curve) and weak/absent nucleocytoplasmic staining (low, red curve) are compared. Representative images (original magnification, ×400) are included.
Decreased COMMD1 expression is associated with increased tumor invasion and worse survival.
We examined, in more detail, the possible relationship between reduced COMMD1 expression and the clinical behavior of a tumor in a prostate cancer study (20). The data indicated that greater decreases in COMMD1 expression were evident in lymph node metastatic tumors (Figure 1E). Furthermore, local tumor invasion (T3 or T4) was associated with greater reductions in COMMD1 expression compared with that of earlier stage tumors (T2) (Figure 1F). Next, we examined this question in a cohort of 63 patients with endometrial cancer that had long-term follow-up. Using a tissue microarray, COMMD1 expression was scored among the tumors. Upon separating the tumors into high expression (strong cytoplasmic staining) and low expression (low cytoplasmic and nuclear staining), it became evident that low COMMD1 expression was accompanied by a worse clinical outcome, primarily due to excess mortality in the first 5 years of follow-up (Figure 1G).
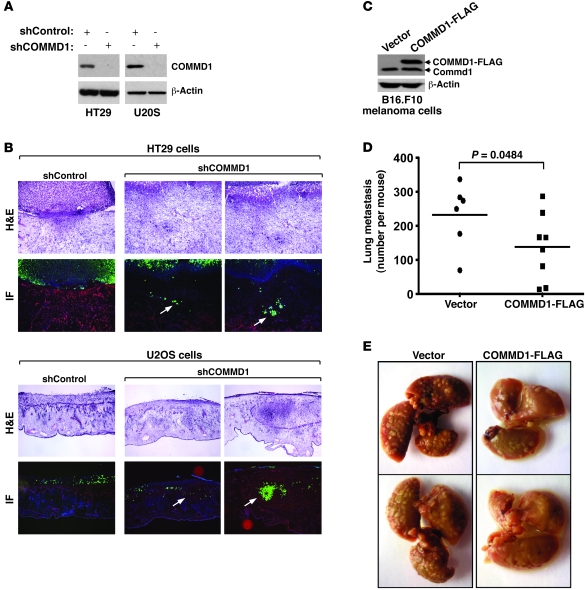
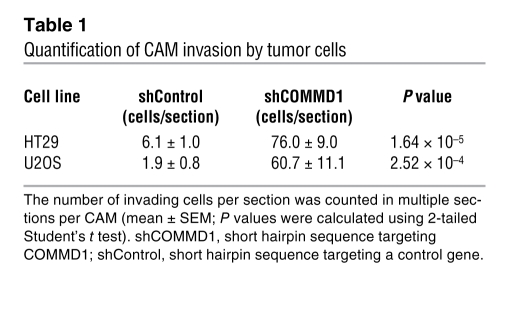
Altogether, these findings suggested the possibility that decreased COMMD1 expression might promote tumor invasion. To test this notion directly, we used lentiviral delivery of shRNA vectors to generate tumor cell lines defective in COMMD1 expression (Figure 2A) and examined their invasion potential in vivo. Control and COMMD1-deficient cells expressing GFP were placed over the chorioallantoic membrane (CAM) of chick embryos. Invasion into the CAM was assessed 3 days later by the presence of GFP-positive tumor cells within the laminin-stained stroma of the CAM (stained red in our experiments). In all instances, reduced COMMD1 expression resulted in increased invasiveness, as tumor cells were readily detectable within the CAM, while most of the control cells were restricted above the CAM (representative images and quantitation shown in Figure 2B and Table 1). In fact, this increased local invasion was also illustrated by the effacement of the boundary between the tumor mass above the CAM and the CAM tissue itself (seen most notably in the H&E-stained sections). Next, we used a different model to examine the notion that COMMD1 modulates tumor invasion and metastatic potential. In this case, we generated mouse melanoma cells stably overexpressing COMMD1 (Figure 2C) and injected these cells intravenously into C57BL6 mice. The number of subsequent lung metastases was significantly reduced when the cells expressed COMMD1 (Figure 2, D and E), indicating that this factor restrains the metastatic potential of this invasive cell line in vivo. Altogether, these findings suggested that suppressed COMMD1 gene expression is a frequent event in a number of cancers and might confer tumors with invasion advantages, perhaps acting collaboratively via changes in both neoplastic and stromal elements.
Figure 2. COMMD1 repression promotes tumor invasion.
(A–C) HT29 or U2OS cells were infected with lentiviruses expressing short hairpin sequences targeting COMMD1 (shCOMMD1) or a control gene (shControl). (A) Western blot analysis demonstrating the decrease in COMMD1 levels in the cell lines used is shown. These cells were tested for invasion of the chick CAM, as described in the Methods section. (B) Representative images of H&E and immunofluorescence staining (IF) are shown (laminin, red; DAPI, blue; cancer cells, green). Cell invasion into the CAM is indicated with white arrows (original magnification, ×200 [HT29]; ×100 [U2OS]). (C–E) B16.F10 mouse melanoma cells were stably transfected to express COMMD1-FLAG (as shown in the Western blot in C). These cells were subsequently injected into the tail vein of syngeneic C57BL6 mice (2 × 105 cells/mouse), and (D) after 15 days, the mice were sacrificed, and the number of lung metastasis per mouse was counted (the horizontal bars represent the mean in each group). The Student’s t test was used for statistical analysis. (E) Representative images of the lungs of these mice are shown.
Table 1 .
Quantification of CAM invasion by tumor cells
COMMD1 controls the expression of critical genes involved in invasion and metastasis.
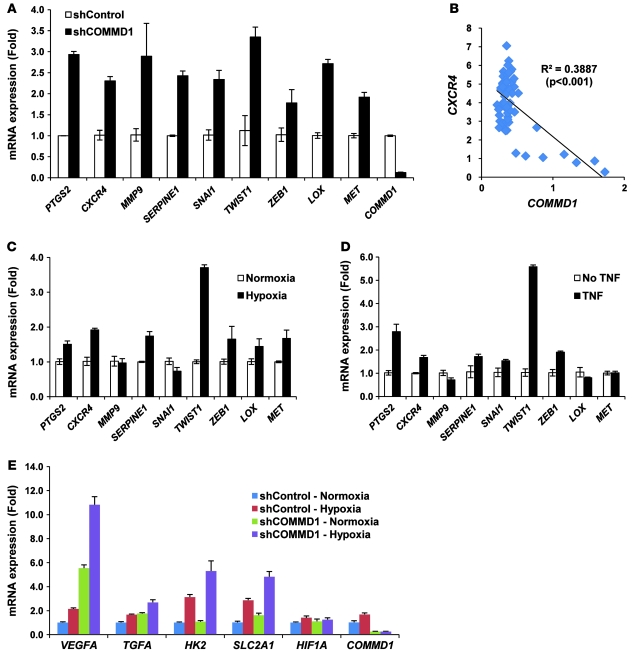
To try to ascertain in further detail the potential molecular targets responsible for the increased invasion observed after COMMD1 suppression, we examined, in a targeted manner, the expression of genes previously implicated in tumor invasion. COMMD1 suppression in HT29 cells was accompanied by increased expression of several genes that promote tumor invasion (Figure 3A), and similar findings were noted for LOX, MMP9, and CXCR4 in U2OS cells (data not shown). However, these findings were not universal, and some genes were not induced by COMMD1 deficiency (like MMP2) or were not expressed in HT29 cells (like ZEB2 and VIM). Additional evaluation of the Oncomine database also indicated a significant inverse correlation between COMMD1 and MMP9 expression (P < 0.05) in seminoma (data not shown) and between COMMD1 and CXCR4 expression in breast stromal cells (Figure 3B). Altogether, these data indicated that decreased COMMD1 levels lead to derepression of several genes that promote tumor invasion, a finding that is in agreement with the increased invasiveness of these cells.
Figure 3. COMMD1 inhibits the expression of HIF-regulated genes.
(A) Gene expression for a panel of invasion-promoting genes was determined in HT29 cells by quantitative RT-PCR and expressed as fold over the shControl sample (mean ± SEM of triplicate samples are shown). (B) Normalized COMMD1 and CXCR4 expression in breast stromal cells (shown in Figure 1E) was plotted and a Pearson’s correlation value was calculated. (C and D) The expression of the same genes was examined in HT29 cells exposed to (C) hypoxia or to (D) the NF-κB activator, TNF. The respective mRNA levels were determined by quantitative RT-PCR and expressed as fold over the untreated sample (mean ± SEM of triplicate samples are shown). (E) Expression of selected HIF target genes was determined by quantitative RT-PCR analysis and expressed as fold over the shControl sample (mean ± SEM of triplicate samples are shown).
COMMD1 inhibits HIF target genes in cancer cells.
Most of the invasion-promoting genes derepressed in COMMD1-deficient cells have been reported to be regulated by both NF-κB and HIF (12, 13, 21–28) (Supplemental Figure 1; supplemental material available online with this article; doi: 10.1172/JCI40583DS1). To further investigate whether these relationships hold true in the studied tumor cells, we examined whether hypoxia or TNF, a potent activator of NF-κB, led to increased expression of these genes (Figure 3, C and D). These studies confirmed that in these cells PTGS2 (encoding COX2), CXCR4, SERPINE1, TWIST1, and ZEB1 were responsive to both hypoxia and NF-κB activation, while MMP9 and SNAI1 were unresponsive to either stimulus. The data also revealed that LOX and MET expression was stimulated by hypoxia and not by NF-κB activation downstream of TNF, confirming prior reports that these genes are HIF responsive (29, 30). These findings indicated that in addition to the effects of COMMD1 on NF-κB, derepressed HIF-mediated transcription participates in the upregulation of invasion-promoting genes.
We turned our attention next to the effects of COMMD1 on HIF-mediated gene expression in cancer cells. We examined a panel of HIF-inducible gene targets and found that decreased COMMD1 expression resulted in the derepression of several HIF-responsive genes, such as VEGFA, TGFA (encoding TGF-α), HK2, and SLC2A1 (encoding GLUT1), under normoxic and hypoxic conditions (Figure 3E), and the same was true for several HIF targets that promote tumor invasion, such as LOX and CXCR4 (data not shown).
These findings were consistent with our prior report that Commd1–/– embryos demonstrate higher transcriptional activity of HIF-1 and abnormal placental development (4). Interestingly, unlike our observations in Commd1-deficient embryos, the HT29 and U2OS tumor lines did not demonstrate increased steady-state protein levels of HIF-1α or HIF-2α (Figure 4A). Detailed examination of HIF-1α accumulation during hypoxia did not reveal significantly greater levels of HIF-1α in the COMMD1-deficient cells either (Supplemental Figure 2). Nevertheless, this approach did demonstrate greater accumulation of Hif-1α in Commd1-deficient mouse embryo fibroblasts (MEFs) (Supplemental Figure 3), suggesting that the effects of COMMD1 on the stability of HIF-α subunits observed in normal and nonneoplastic cell lines is lost in some tumor cell lines, such as the ones investigated here.
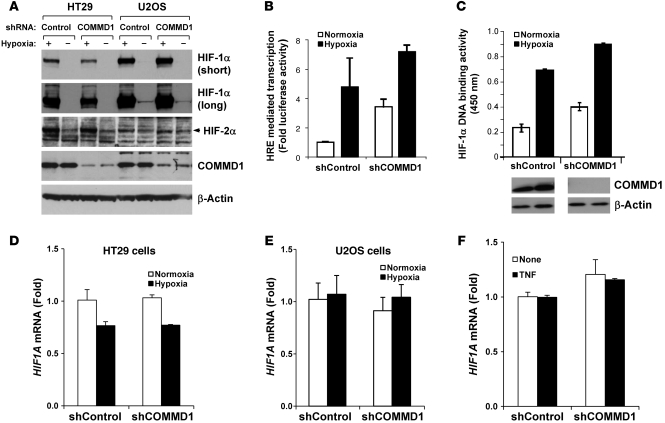
Figure 4. COMMD1 inhibits HIF-mediated transcription and DNA binding without affecting HIF-α expression.
(A) Protein expression of HIF-1α and HIF-2α was not affected by COMMD1 deficiency in HT29 and U2OS cells. Western blot analysis for HIF-1α, HIF-2α, COMMD1, and β-Actin from the indicated samples is shown. Long and short exposures of the HIF-1α Western blot are shown. (B) HEK 293T cells with a stable repression of COMMD1 or control cells were cotransfected with 5xHRE-firefly luciferase reporter and renilla luciferase control plasmid. Cells were incubated under normoxia or hypoxia for 16 hours. HRE reporter activity was determined by measuring firefly luciferase in the lysates and was corrected for transfection efficiency using renilla luciferase activities. Results are expressed as fold induction relative to normoxic conditions in shControl cells (mean ± SD of triplicate samples are shown). (C) The same cells examined in B were used for the HIF-1α HRE DNA binding assay. DNA binding is expressed as fold binding relative to normoxic conditions in shControl cells (mean ± SD of triplicate samples are shown). COMMD1 expression is shown by immunoblot analysis. (D–F) HIF1A mRNA expression (encoding for HIF-1α) was determined by qRT-PCR in (D) HT29 and (E) U2OS cells and normalized to shControl transfected cells under normoxia. (F) Similarly, the effect of TNF treatment on HIF1A expression in HT29 was also examined (mean ± SEM of triplicate samples are shown).
These data indicated that the increased expression of HIF target genes in tumor cells with decreased COMMD1 levels did not necessitate HIF-α accumulation. Furthermore, nuclear levels of HIF-1α were unaffected by COMMD1 expression (data not shown), suggesting that COMMD1 can modulate HIF transactivation directly. To examine this possibility, we investigated the effects of decreased COMMD1 expression on HIF transcriptional activity and DNA binding. Using a luciferase reporter containing multimerized HREs, we observed that decreased COMMD1 expression resulted in greater HIF activity, particularly during normoxia (Figure 4B). Interestingly, HIF-1α DNA binding was increased in COMMD1-deficient cells when compared with that of the corresponding control (Figure 4C), and similar results were seen using HeLa cells (data not shown). Finally, we examined the expression of the HIF1A gene encoding for HIF-1α, which has been reported to be NF-κB responsive in certain settings (31). We found no effect of COMMD1 deficiency, hypoxia, or TNF stimulation on HIF1A mRNA levels (Figure 4, D–F), further suggesting that the activation of HIF-dependent transcription in COMMD1-deficient cells is not an indirect effect mediated by NF-κB activation in these cells.
COMMD1 binds to HIF-1α in an oxygen-dependent manner.
To ascertain the mechanism of COMMD1-mediated inhibition of HIF, we examined in detail the interaction between COMMD1 and HIF-1α, the α-subunit predominantly expressed in these cells. Although HIF-1α expression was low under normoxic conditions, the protein was readily coimmunoprecipitated with endogenous COMMD1 (Figure 5A). Hypoxia resulted in the expected accumulation of HIF-1α, yet its coimmunoprecipitation with endogenous COMMD1 was not increased, suggesting that the binding between these molecules is regulated by oxygen. Further characterization of the interaction indicated that the COMM domain of COMMD1 is necessary and sufficient for HIF-1α binding (Figure 5, B and C).
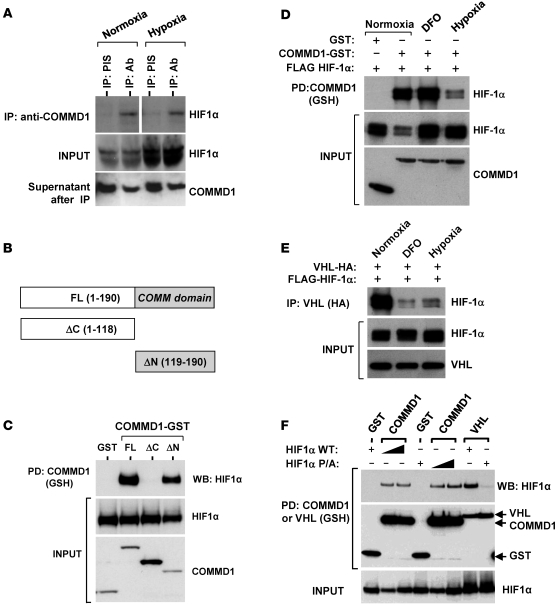
Figure 5. COMMD1 binds to HIF-1α preferentially under normoxic conditions.
(A) Endogenous COMMD1 immunoprecipitation using HEK 293T whole cell lysates incubated either under normoxia or hypoxia (for 1 hour). Whole cell lysates and immunoprecipitates were subjected to Western blot analysis. Supernatants after IP demonstrate immunodepletion of COMMD1. The lanes on the top panel were run on the same gel but were noncontiguous. (B) Truncation mutants of COMMD1. FL, full-length; ΔC, deletion of the carboxyl terminal COMM domain; ΔN, deletion of the amino terminus. (C) These proteins were coexpressed with HA-tagged HIF-1α in HEK293 cells and subsequently precipitated from cell lysates. Coprecipitation of HIF-1α with COMMD1 full-length and ΔN is shown. (D) HEK 293T cells expressing COMMD1-GST, GST, or FLAG–HIF-1α were cultured under normoxia (20% O2) or hypoxia (1% O2, 8 hours) or treated with DFO (0.1 mM, 8 hours), prior to glutahione-sepharose (GSH) precipitations and Western blot analysis as indicated. (E) HEK 293T cells expressing HA-VHL and FLAG–HIF-1α were cultured under normoxia, hypoxia, or treated with DFO as before. Immunoprecipitation of VHL (using anti-HA antibody) was followed by Western blot analysis. (F) HEK 293T cells were transfected with GST, COMMD1-GST, or GST-VHL, along with wild-type HIF-1α or HIF-1α P/A. Subsequently, GSH precipitations were followed by Western blot (WB) analysis as indicated. PD, pull down; PIS, preimmune serum.
Since oxygen-dependent proline hydroxylation is critical for the interaction between VHL and HIF-1α, we next investigated whether this modification is similarly involved in COMMD1 binding. In coprecipitation experiments, reduced binding between HIF-1α and COMMD1 was observed, despite the hypoxic stabilization of HIF-1α (Figure 5D, lanes 2 and 4). However, desferrioxamine (DFO), an inhibitor of prolyl hydroxylation, had no negative effect on this interaction (lane 3), suggesting that hydroxylation is not involved in the preferential interaction between COMMD1 and HIF-1α during normoxia. This is in contrast to the interaction between HIF-1α and VHL, in which both hypoxia and DFO treatment clearly reduced HIF-1α/VHL binding (Figure 5E). To directly address whether HIF-1α prolyl hydroxylation is involved in COMMD1 binding, we coexpressed COMMD1–glutathione-S-transferase (COMMD1-GST) with a proline-to-alanine mutant of HIF-1α (P402A/P564A, referred to herein as HIF-1α P/A). COMMD1 bound to wild-type HIF-1α and its P/A mutant to the same extent, while on the other hand, this mutation impaired the interaction with VHL as expected (Figure 5F). Together, these data demonstrated that COMMD1 binds to HIF-1α in an oxygen-dependent manner, albeit independent of prolyl hydroxylation.
COMMD1 prevents HIF-1α/β heterodimer formation.
The results thus far suggested that COMMD1 is part of the physiologic mechanisms that restrain HIF activity during normoxia. To elucidate further the mechanism of transcriptional inhibition, we next examined the domain of HIF-1α that is essential for COMMD1 binding (Figure 6A). As shown in Figure 6B, COMMD1 precipitation from cell lysates was capable of coprecipitating the amino-terminal region of HIF-1α (amino acids 1–300), which contains the bHLH and PAS domains involved in HIF-1β heterodimerization and DNA binding. In contrast, no binding was observed between COMMD1 and the transactivation domains or the ODDD of HIF-1α. To determine whether the interaction between COMMD1 and HIF-1α is direct, we examined the binding between recombinant COMMD1 and HIF-1α in a solid-phase ELISA binding assay. COMMD1 was able to bind to recombinant full-length HIF-1α and the 1–300 fragment (Figure 6C). These findings further suggested the possibility that COMMD1 may interact with other transcription factors that contain the bHLH/PAS domains, such as HIF-2α and HIF-1β. Indeed, we found that COMMD1 can coprecipitate HIF-2α (Supplemental Figure 4) and HIF-1β (Supplemental Figure 5). Since HIF-1β dimerizes with the HIF-α subunits, it is possible that the latter interaction could be indirect and mediated through the binding of COMMD1 with HIF-1α or HIF-2α. To examine these possibilities, we performed an in vitro binding experiment, in which recombinant COMMD1 protein produced in E. coli was mixed with cellular lysates prepared from normoxic or hypoxic cells. COMMD1 coprecipitated endogenous HIF-1β (Supplemental Figure 6), irrespective of the expression of HIF-1α in these lysates, further indicating a direct interaction between COMMD1 and the bHLH/PAS domains in these molecules. Indeed, like HIF-1α, the bHLH/PAS regions of HIF-1β and HIF-2α were able to bind to COMMD1 (Supplemental Figure 7).
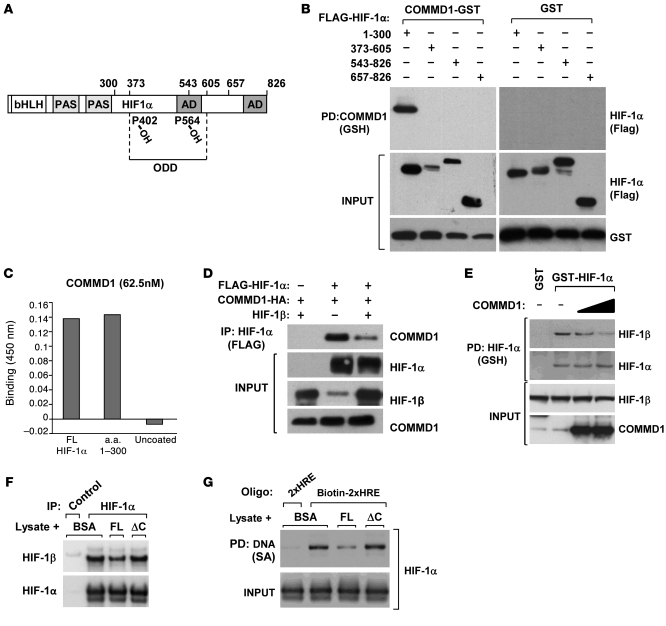
Figure 6. COMMD1 binds directly to the amino terminus of HIF-1α and inhibits HIF-1β dimerization.
(A) Schematic representation of the domains in HIF-1α. AD, activation domain. (B) HEK 293T cells expressing COMMD1-GST or GST and the indicated truncated fragments of FLAG–HIF-1α were used for GSH precipitations and Western blot analysis as shown. (C) Recombinant COMMD-1-MBP was incubated with immobilized recombinant HIF-1α full-length, HIF-1α fragment (1–300), or uncoated wells. Binding of the proteins was detected by absorbance at 450 nm, as described in the Methods section. Data represent the mean of duplicate experiments. (D) HEK 293T cells were transfected to express COMMD1-HA, FLAG–HIF-1α, or HIF-1β as indicated. HIF-1α was immunoprecipitated using an anti-FLAG antibody, and the resulting samples were subjected to Western blot analysis. (E) HEK 293T cell lysates expressing GST, GST-HIF-1α, or COMMD1 were subjected to GSH precipitations. The amount of coprecipitated endogenous HIF-1β was identified by Western blot analysis. (F and G) Lysates from HEK293 cells transfected with HA–HIF-1α were incubated with the indicated recombinant proteins. (F) Immunoprecipitation of HIF-1α (HA antibody, FLAG antibody as a control) was performed. The amount of HIF-1β recovered was assessed by immunoblotting. (G) The lysates were subsequently mixed with tandem HRE oligonucleotide probes, which were then precipitated and immunoblotted for HIF-1α. 2xHRE represents a control oligonucleotide that was not biotinylated, biotin-2xHRE represents the biotinylated version. FL, COMMD1 full-length; SA, streptavidin.
Given that COMMD1 and HIF-1β both bind to the amino terminus of HIF-1α, we investigated whether HIF-1β competes with COMMD1 for HIF-1α binding. As shown in Figure 6D, HIF-1β cotransfection diminished the interaction between COMMD1 and HIF-1α. Similarly, in a reciprocal experiment, overexpression of COMMD1 reduced the interaction between HIF-1α and HIF-1β (Figure 6E). We examined this notion further, using an in vitro binding system. Recombinant COMMD1 prepared in E. coli was incubated with cellular lysates and subsequently, HIF-α/β dimerization and HIF-1 DNA binding were evaluated. Incubation with full-length recombinant COMMD1 diminished the amount of HIF-1β that coprecipitated with HIF-1α (Figure 6F), but a truncated COMMD1 protein, with deletion of the carboxyl terminal COMM domain, that did not bind to HIF-1α (ΔC) had no effect in this assay. Using a similar approach, we assessed DNA binding by adding biotinylated oligonucleotides containing tandem HRE sites to precipitate DNA/protein complexes. Once more, full-length recombinant COMMD1 diminished HIF-1α/DNA binding, while the ΔC mutant that did not interact with HIF-1α had no effect (Figure 6G). Together, these experiments indicate that the ability of COMMD1 to bind directly to the bHLH/PAS domain of HIF-1α allowed it to compete with HIF-1β for binding. This provides a mechanism by which COMMD1 interferes directly with DNA binding and transcriptional activation, independent of any effects on HIF-1α stability.
Discussion
This study demonstrates that decreased COMMD1 gene expression is frequently observed in a variety of cancers and that this correlates with tumor invasion locally or to lymphatic nodes as well as with overall patient survival. Indeed, 2 separate in vivo tumor invasion models confirmed that cancer cell invasion was regulated by COMMD1, suggesting that decreased COMMD1 expression might represent a novel mechanism that confers cancer cells with invasion potential, perhaps by relieving the inhibitory influence of COMMD1 on HIF and NF-κB. In particular, the more invasive phenotype of COMMD1-deficient cells is consistent with their increased expression of various genes previously implicated in tumor invasion, such as the HIF-responsive gene LOX, encoding an enzyme responsible for collagen and elastin cross-linking that plays a critical role in tumor invasion (29). LOX is frequently overexpressed in metastatic cancers, promotes tumor invasion in xenograft models, and mediates critical changes in the extracellular matrix that result in a microenvironment conducive to metastasis (32). Interestingly, similar decreased expression in cancer has been reported for another COMMD family member, COMMD5, suggesting that some of these properties may be shared by other genes in this family (33).
This study also provides a detailed account of the mechanism by which COMMD1 inhibits HIF-mediated transcription. We have previously reported that COMMD1 can destabilize HIF-1α (4), an effect that has been linked to the ability of COMMD1 to compete for HSP90 binding (34). However, the data presented here indicate that COMMD1 can also inhibit the transcriptional activity of HIF independent of any effects on HIF-1α levels. These observations are not mutually exclusive, and the different findings made in mouse embryos, MEFs, and cancer cells may be the result of various mechanisms that affect HIF-α turnover in malignant cells (5). The data indicate that COMMD1 can impair HIF-1α/β dimerization and DNA binding. However, the regulation of HIF-mediated transcription by COMMD1 is likely more complex, as there is evidence for some gene target specificity. This suggests the possibility that, at the endogenous level, the competition between COMMD1 and HIF-1β may be a regulated event that might occur primarily at specific promoters, a possibility that will require future investigation.
Our findings indicate that COMMD1 can be added to a growing list of VHL-independent pathways of HIF regulation that are not always dependent on oxygen tension (5). For example, receptor for activated protein kinase C 1 (RACK1) also promotes HIF degradation through Cul2, in a VHL- and oxygen-independent manner, through binding to bHLH/PAS domain of HIF-1α, while HSP90 binds to the same domains and prevents RACK1-mediated degradation (35, 36). Interestingly COMMD1 can compete HSP90 but not RACK1 for binding to this domain, an effect that may underlie the ability of COMMD1 to destabilize HIF-1α in certain settings (34). We found that COMMD1/HIF-1α interactions are enhanced by oxygen, suggesting that the physiologic context for COMMD1-mediated inhibition is during normoxia, consistent with the observed effect of COMMD1 expression on HRE-responsive transcription. The mechanism for oxygen-dependent binding is unclear at this point but may involve HSP90 or hypoxia-mediated nuclear export of COMMD1 at a time when HIF-1α nuclear accumulation is taking place (37). It is tempting to speculate that decreased COMMD1 expression in certain tumors may account for upregulated HIF-mediated responses in the absence of overt tissue hypoxia, a phenomenon that is known to occur in cancer (5).
Finally, there is increasing recognition of the interrelationship between the NF-κB and HIF pathways. The activation of HIF is required for optimal phagocytic activation, a process also promoted by NF-κB, while on the other hand, HIF-1α has been shown to be a direct transcriptional target of NF-κB (31). In addition, it is notable that there is substantial overlap between these 2 transcription factors in the regulation of genes involved in EMT (25, 26, 38, 39). Moreover, several recent reports have demonstrated that the regulation of these pathways is further interconnected at the level of the prolyl hydroxylases, enzymes best known as regulators of HIF-α hydroxylation that have now been shown to modulate IKK activity and NF-κB–mediated transcription (40, 41). It is in this context that the dual role of COMMD1 in the regulation of both NF-κB and HIF seems to fit a larger physiologic paradigm, in which coordinated regulation of both of these pathways is probably required for certain biological processes.
Altogether, this study suggests that suppressed COMMD1 expression in a variety of malignancies results in derepressed NF-κB and HIF-mediated transcription, which is translated into a more aggressive tumor phenotype. The finding that decreased COMMD1 expression can be found also in stromal elements within the tumor suggests that these changes do not necessitate genetic alterations and may be mediated by factors present in the tumor microenvironment. Greater understanding of the mechanisms that control COMMD1 gene expression in normal and neoplastic cells is needed and might provide future opportunities for therapeutic intervention.
Methods
Plasmids and siRNA.
The plasmids pEBB, pEBG, pEBB-COMMD1-FLAG, pEBB-COMMD1 with deletion of the amino terminus–FLAG (ΔN-FLAG), pEBB-COMMD1 ΔC-FLAG, and pEBB-COMMD1-GST have been described previously (42, 43). pEBB–HIF-1α was generated by PCR amplification of the ORF, using pCEP4/HIF-1α (GenBank U22431) as template. All HIF-1α deletion mutants were generated by PCR amplification from the template FLAG–HIF-1α (4) and subcloned into p3xFLAG-CMV-10 expression vector (Sigma-Aldrich) for mammalian expression or into pETλHis for bacterial expression. pEBB-HIF-1α P/A was generated by PCR amplification of HIF-1α P/A from p(HA)-HIF-1α P/A, provided by Eric Huang (Department of Neurosurgery, University of Utah, Salt Lake City, Utah, USA) (44). pcDNA3-HA-VHL was provided by R.H. Giles (University Medical Center Utrecht).
Recombinant protein preparation.
Recombinant HIF-1α protein was prepared using immobilized metal ion affinity chromatography (IMAC), using a fused His6 tag as previously described (45). Recombinant COMMD1-GST full-length (used in Supplemental Figure 6) was prepared as previously described (46). The proteins used in Figure 6, F and G, were His6-tagged versions of COMMD1 full-length and ΔC, which were subcloned from the corresponding pEBB vectors into pET30a and purified from E. coli using IMAC.
Cell culture, transfection, and lentiviral production.
HEK 293, 293T, HeLa, HT29, U2OS, MEFs, and B16.F0 mouse melanoma cells were cultured in Dulbecco’s modified Eagle medium, supplemented with 10% fetal bovine serum, l-glutamine, and penicillin-streptomycin. Cells were cultured at 5% CO2 and 21% O2 for normoxia and 1%–3% O2 for hypoxia, as indicated. An Invivo2 Hypoxia Workstation 1000 (Biotrace International) and a regulated O2 tissue culture incubator (Napco) were used. Treatment with DFO consisted of supplementing the media with 0.1 mM DFO. A standard calcium phosphate transfection protocol was used to transfect plasmids into HEK 293 cells (42). Stable HeLa and 293T COMMD1 knockdown cells were generated, as previously described, by selection using medium supplemented with 1 μg/ml puromycin (4). Generation of U2OS shRNA cells lines (against COMMD1 or a control gene, chloramphenicol acetyl transferase [CAT]) has been previously described (47). An identical approach was taken to produce the corresponding HT29 cell lines.
Immunoblotting and immunoprecipitation.
Whole cell lysates were prepared by adding Triton lysis buffer (25 mM HEPES, 100 mM NaCl, 10 mM DTT, 1 mM EDTA, 10% glycerol, 1% Triton X-100) or RIPA buffer (PBS, 1% NP-40, 0.5% deoxycholate, 0.1% SDS, 10 mM DTT), supplemented with 1 mM sodium orthovanadate and protease inhibitors (Roche), as indicated in each experiment. Immunoprecipitations, glutathione (GSH) precipitations, and immunoblotting were performed as previously described (42, 43). The following antibodies were used in our studies: anti–β-actin (clone AC-74; Sigma-Aldrich), anti-COMMD1 (1:5,000) (43, 48), anti-GST (Santa Cruz Biotechnology Inc.), anti-FLAG (F1804; Sigma-Aldrich), anti-HA (clone HA-7; Sigma-Aldrich), anti–HIF-1α (clone 54; BD Biosciences), anti–HIF-2α (NB100-122; Novus Biologicals), anti–HIF-1β (clone 29; BD Biosciences — Transduction Laboratories), and anti–lamin A (clone 133A2; Monosan).
Studies using human tissue.
The use of ovarian cancer specimens was reviewed and approved by the Institutional Review Board at the University of Michigan Medical School. The informed consent requirement was waived, because tissues were retrospectively acquired from the Surgical Pathology archives, did not directly involve human subjects, and the study was considered no more than minimal risk. The use of leftover endometrial cancer specimens was reviewed and approved by the Scientific Advisory Council of the University Medical Center Utrecht Biobank. The samples were collected from the archives of the Department of Pathology of the University Medical Center Utrecht. According to Dutch law, no informed consent is needed when coded or anonymous leftover material is used for research, as long as patients have been informed of this possibility and offered the option to opt out, which was the case in the present study.
Immunohistochemistry.
Tissue microarrays for ovarian and endometrial cancers were stained for COMMD1. The ovarian tissue microarray block contained 3 representative cores from each of the 48 ovarian cancer specimens (26 serous and 22 endometrioid ovarian carcinomas). The endometrial cancer tissue microarray contained samples from 63 endometrioid endometrial carcinomas. Tissue microarray sections were deparaffinized in xylene and then rehydrated into distilled H2O through graded alcohols. Antigen retrieval was enhanced by microwaving the slides in citrate buffer (pH 6.0, Biogenex) for 10 minutes. Endogenous peroxidase activity was quenched by incubation with 3% hydrogen peroxide in methanol, and then the sections were blocked with 1.5% normal goat serum for 30 minutes. After blocking, the sections were incubated with a rabbit polyclonal anti-COMMD1 antibody (1:1,000 dilution) overnight at 4°C (43). Slides were washed in PBS and subsequently incubated with a biotinylated horse anti-rabbit secondary antibody for 30 minutes at room temperature. Antigen-antibody complexes were detected with the avidin-biotin peroxidase method, using Vector DAB as a chromogenic substrate (Vectastain ABC kit; Vector Laboratories). Immunostained sections were lightly counter-stained with hematoxylin and examined by light microscopy.
CAM invasion.
Fertilized eggs (Bilbie Aviaries) were incubated for 11 days at 37°C with 60% humidity. To grow tumor masses, 2.5 × 106 cells in 40 μl HBSS were applied on the CAM. After 3 days, the tumor masses and surrounding CAM were excised and fixed with 4% paraformaldehyde in PBS overnight at 4°C. Frozen sections of the CAM were made after immersion in 30% sucrose and were incubated with mouse anti-chicken laminin (3H11, diluted 1:10; Developmental Studies Hybridoma Bank) at room temperature for 2 hours, followed by incubation with Alexa Fluor 594–conjugated goat anti-mouse IgG (1:500; Invitrogen) for 60 minutes. Then, they were mounted with Vectashield mounting media with 4',6-diamidino-2-phenylindole (Vector Laboratories), visualized under a fluorescent microscope (Leica DMLB), and photographed with a SPOT cooled color digital camera (Diagnostic Instruments). Serial sections were also prepared and stained with H&E for light microscopic examination.
Lung metastasis mouse model.
The animal studies were reviewed and approved by the animal use committee at the University Medical Centre Groningen. Stable COMMD1-FLAG expression in B16.F10 cells was achieved through transfection of parental cells with pcDNA3.1-COMMD1-FLAG or empty vector and selection of stable transfectants through G418 resistance (1 mg/ml). Ten-week-old C57BL6 mice were injected in the tail vein with 2 × 105 cells resuspended in PBS. After 15 days, animals were sacrificed, and the number of lung tumors was counted and plotted as shown.
HIF-1α HRE plate binding assay.
HIF-1α binding assay was performed with DuoSet IC human/mouse active HIF-1α activity assay (R&D Systems), according to the manufacturer’s instructions. Briefly, HIF-1α antibody (400 ng/well) was coated overnight at room temperature in a Maxisorp 96-well ELISA plate (NUNC). Next, the plate was washed twice with PBS containing 0.5% Tween-20 (PBS-T) and blocked for 4 hours with 5% BSA in PBS-T at room temperature. Biotinylated oligonucleotide (4.5 μl) was added to 75 μg of nuclear extract and incubated for 30 minutes at room temperature and then added to the ELISA plate in triplicate. After a 2-hour incubation at room temperature, the plate was washed 5 times with PBS-T. Subsequently, the plate was incubated with streptavidin-HRP for 30 minutes, washed similarly in PBS-T, and then stained using the ImmunoPure TMB Substrate kit (Pierce), according to the manufacturer’s instructions. The signal intensities were measured using an ELISA reader (Bio-Rad) at 450 nm, and DNA binding was expressed as the fold binding relative to normoxic conditions in control cells.
HIF-1α/HRE coprecipitation assay.
Sense and antisense oligonucleotides encoding tandem HREs (sense strand, TGCATGCATACGTGGGCTCCAAGAGGCGCGTGCGTCGCGATGCA) were annealed after heating and slow cooling. In one case, the sense strand was biotinylated in the 5′ position (biotin, 2xHRE), while a nonbiotinylated oligonucleotide was used as control (2xHRE). Cell lysates prepared in Triton lysis buffer were pre-incubated with recombinant proteins overnight as indicated (final concentration of 1 μg/μl), and, thereafter, annealed oligonucleotides were added and mixed for 60 additional minutes at 4°C. The oligonucleotides were subsequently precipitated by streptavidin agarose beads and thoroughly rinsed 4 times before proceeding to Western blot analysis.
ELISA for direct binding of COMMD1.
ELISA plates (NUNC) were absorbed overnight at 4°C with 100 μl per well of His6-purified recombinant full-length and 1–300 HIF-1α proteins (at 1 mM). The wells were blocked with 2% BSA in PBS/0.1% Tween-20 (BSA-T) for 2 hours at 25°C. After 2 washes, the wells were incubated with either 100 μl of a 62.5 nM solution of COMMD1–maltose-binding protein (COMMD1-MBP) or MBP in BSA-T for 1 hour (48). The wells were washed 3 times with PBS-T (PBS, 0.1% Tween-20), incubated with a rabbit anti-COMMD1 (1:2,000) in BSA-T for 1 hour, and washed 3 times with PBS-T, followed by incubation with HRP-conjugated goat anti-rabbit IgG (H+L) antibodies (1:5,000; Bio-Rad) in BSA-T for 30 minutes. The plates were washed 5 times with PBS-T and 1 time with PBS, followed by staining using the ImmunoPure TMB Substrate kit (Pierce). Binding of the proteins was determined by measuring absorbance at 450 nm using an ELISA reader (Bio-Rad). MBP signals were subtracted from MBP-COMMD1 signals.
Quantitative RT-PCR.
Total RNA was extracted from cells using the RNeasy kit (Qiagen), according to the manufacturer’s instructions. A RT reaction with 500 ng total RNA in 25 μl was performed using random hexamers and TaqMan Reverse Transcription Reagents (Applied Biosystems). This was followed by quantitative PCR performed in the 7500 Real-Time PCR system (Applied Biosytems). In all reactions, TaqMan PCR Master Mix with β-Actin (Actb) mRNA quantitation was duplexed in the same well as an internal control. Quantitative RT-PCR of COMMD1 has been previously described (43). Oligonucleotides and internal probes were obtained from Applied Biosystems.
Cancer gene expression studies.
Gene expression profiles for COMMD1 and other genes were obtained from the Oncomine database ( https://www.oncomine.org/resource/login.html). All gene expression studies included here can be found at the Oncomine database or directly in the original reports (15–20). The normalized log2 expression data was downloaded for independent analysis, including graphing and calculation of P values using the Student’s t test and Pearson’s correlation index. In some cases, additional information about the samples, such as the tumor T stage in the study by Lapointe et al. (20), was obtained directly from the original publication and was included in the analysis.
Statistics.
The comparisons between groups in all gene expression studies presented in Figure 1 as well as in the CAM invasion presented in Table 1 were performed using the Student’s t test (2-tailed test, unequal variance analysis). The comparison between the groups in the lung metastasis study in Figure 2D was performed using the unpaired, 1-tailed Student’s t test. The survival curves presented in Figure 1G were compared using the log-rank test. The relationship between COMMD1 and CXCR4 expression was examined by the Pearson’s correlation coefficient using a 2-tailed test. For all comparisons, a P value of less than 0.05 was considered significant.
Supplementary Material
Acknowledgments
We are grateful to Eric Huang for providing a plasmid used in the course of our studies. We also thank Henk van der Molen and Petra van der Groep for their technical assistance. This work was supported by grant R01 DK073639 from the NIH and the University of Texas DOCS Award to E. Burstein, the Dutch Cancer Society (grant UU2003-2825 to M. Vooijs, P. van Diest, and E. van der Wall), the 2nd AEGON International Scholarship in Oncology to P. van Diest and E. van der Wall, and the Dutch Organization for Scientific Research (grant 40-00812-98-03106 to C. Wijmenga and L.W. Klomp and grant 817.02.022 to M. Vooijs, P. van Diest, B. van de Sluis, C. Wijmenga, and L.W. Klomp). Financial support was obtained for the hypoxic working station Ruskin Invivo1000 and a Turner systems luminometer from the Maurits Anna de Kock Foundation and Nijbakker Morra, respectively.
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Citation for this article: J Clin Invest. 2010;120(6):2119–2130. doi:10.1172/JCI40583.
References
- 1.Maine GN, Burstein E. COMMD proteins: COMMing to the scene. Cell Mol Life Sci. 2007;64(15):1997–2005. doi: 10.1007/s00018-007-7078-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ganesh L, et al. The gene product Murr1 restricts HIV-1 replication in resting CD4+ lymphocytes. . Nature. 2003;426(6968):853–857. doi: 10.1038/nature02171. [DOI] [PubMed] [Google Scholar]
- 3.van de Sluis B, Rothuizen J, Pearson PL, van Oost BA, Wijmenga C. Identification of a new copper metabolism gene by positional cloning in a purebred dog population. Hum Mol Genet. 2002;11(2):165–173. doi: 10.1093/hmg/11.2.165. [DOI] [PubMed] [Google Scholar]
- 4.van de Sluis B, et al. Increased activity of hypoxia-inducible factor 1 is associated with early embryonic lethality in Commd1 null mice. . Mol Cell Biol. 2007;27(11):4142–4156. doi: 10.1128/MCB.01932-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gordan JD, Simon MC. Hypoxia-inducible factors: central regulators of the tumor phenotype. Curr Opin Genet Dev. 2007;17(1):71–77. doi: 10.1016/j.gde.2006.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang GL, Jiang BH, Rue EA, Semenza GL. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. . Proc Natl Acad Sci U S A. 1995;92(12):5510–5514. doi: 10.1073/pnas.92.12.5510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bruick RK, McKnight SL. A conserved family of prolyl-4-hydroxylases that modify HIF. Science. 2001;294(5545):1337–1340. doi: 10.1126/science.1066373. [DOI] [PubMed] [Google Scholar]
- 8.Huang LE, Gu J, Schau M, Bunn HF. Regulation of hypoxia-inducible factor 1alpha is mediated by an O2-dependent degradation domain via the ubiquitin-proteasome pathway. . Proc Natl Acad Sci U S A. 1998;95(14):7987–7992. doi: 10.1073/pnas.95.14.7987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Maxwell PH, et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature. 1999;399(6733):271–275. doi: 10.1038/20459. [DOI] [PubMed] [Google Scholar]
- 10.Naugler WE, Karin M. NF-kappaB and cancer-identifying targets and mechanisms. Curr Opin Genet Dev. 2008;18(1):19–26. doi: 10.1016/j.gde.2008.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gort EH, Groot AJ, van der Wall E, van Diest PJ, Vooijs MA. Hypoxic regulation of metastasis via hypoxia-inducible factors. Curr Mol Med. 2008;8(1):60–67. doi: 10.2174/156652408783565568. [DOI] [PubMed] [Google Scholar]
- 12.Huber MA, et al. NF-kappaB is essential for epithelial-mesenchymal transition and metastasis in a model of breast cancer progression. J Clin Invest. 2004;114(4):569–581. doi: 10.1172/JCI21358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Min C, Eddy SF, Sherr DH, Sonenshein GE. NF-kappaB and epithelial to mesenchymal transition of cancer. J Cell Biochem. 2008;104(3):733–744. doi: 10.1002/jcb.21695. [DOI] [PubMed] [Google Scholar]
- 14.Rhodes DR, et al. Large-scale meta-analysis of cancer microarray data identifies common transcriptional profiles of neoplastic transformation and progression. Proc Natl Acad Sci U S A. 2004;101(25):9309–9314. doi: 10.1073/pnas.0401994101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Korkola JE, et al. Down-regulation of stem cell genes, including those in a 200-kb gene cluster at 12p13.31, is associated with in vivo differentiation of human male germ cell tumors. Cancer Res. 2006;66(2):820–827. doi: 10.1158/0008-5472.CAN-05-2445. [DOI] [PubMed] [Google Scholar]
- 16.Sperger JM, et al. Gene expression patterns in human embryonic stem cells and human pluripotent germ cell tumors. Proc Natl Acad Sci U S A. 2003;100(23):13350–13355. doi: 10.1073/pnas.2235735100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Buchholz M, et al. Transcriptome analysis of microdissected pancreatic intraepithelial neoplastic lesions. Oncogene. 2005;24(44):6626–6636. doi: 10.1038/sj.onc.1208804. [DOI] [PubMed] [Google Scholar]
- 18.Lancaster JM, et al. Gene expression patterns that characterize advanced stage serous ovarian cancers. J Soc Gynecol Investig. 2004;11(1):51–59. doi: 10.1016/j.jsgi.2003.07.004. [DOI] [PubMed] [Google Scholar]
- 19.Finak G, et al. Stromal gene expression predicts clinical outcome in breast cancer. Nat Med. 2008;14(5):518–527. doi: 10.1038/nm1764. [DOI] [PubMed] [Google Scholar]
- 20.Lapointe J, et al. Gene expression profiling identifies clinically relevant subtypes of prostate cancer. Proc Natl Acad Sci U S A. 2004;101(3):811–816. doi: 10.1073/pnas.0304146101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Staller P, Sulitkova J, Lisztwan J, Moch H, Oakeley EJ, Krek W. Chemokine receptor CXCR4 downregulated by von Hippel-Lindau tumour suppressor pVHL. Nature. 2003;425(6955):307–311. doi: 10.1038/nature01874. [DOI] [PubMed] [Google Scholar]
- 22.Helbig G, et al. NF-kappaB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J Biol Chem. 2003;278(24):21631–21638. doi: 10.1074/jbc.M300609200. [DOI] [PubMed] [Google Scholar]
- 23.Leufgen H, et al. Collagenase expression and activity is modulated by the interaction of collagen types, hypoxia, and nutrition in human lung cells. J Cell Physiol. 2005;204(1):146–154. doi: 10.1002/jcp.20289. [DOI] [PubMed] [Google Scholar]
- 24.Li X, et al. Synergistic effect of hypoxia and TNF-alpha on production of PAI-1 in human proximal renal tubular cells. Kidney Int. 2005;68(2):569–583. doi: 10.1111/j.1523-1755.2005.00435.x. [DOI] [PubMed] [Google Scholar]
- 25.Sosic D, Richardson JA, Yu K, Ornitz DM, Olson EN. Twist regulates cytokine gene expression through a negative feedback loop that represses NF-kappaB activity. Cell. 2003;112(2):169–180. doi: 10.1016/S0092-8674(03)00002-3. [DOI] [PubMed] [Google Scholar]
- 26.Yang MH, et al. Direct regulation of TWIST by HIF-1alpha promotes metastasis. Nat Cell Biol. 2008;10(3):295–305. doi: 10.1038/ncb1691. [DOI] [PubMed] [Google Scholar]
- 27.Higgins DF, et al. Hypoxia promotes fibrogenesis in vivo via HIF-1 stimulation of epithelial-to-mesenchymal transition. J Clin Invest. 2007;117(12):3810–3820. doi: 10.1172/JCI30487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chua HL, Bhat-Nakshatri P, Clare SE, Morimiya A, Badve S, Nakshatri H. NF-kappaB represses E-cadherin expression and enhances epithelial to mesenchymal transition of mammary epithelial cells: potential involvement of ZEB-1 and ZEB-2. Oncogene. 2007;26(5):711–724. doi: 10.1038/sj.onc.1209808. [DOI] [PubMed] [Google Scholar]
- 29.Erler JT, et al. Lysyl oxidase is essential for hypoxia-induced metastasis. Nature. 2006;440(7088):1222–1226. doi: 10.1038/nature04695. [DOI] [PubMed] [Google Scholar]
- 30.Pennacchietti S, Michieli P, Galluzzo M, Mazzone M, Giordano S, Comoglio PM. Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell. 2003;3(4):347–361. doi: 10.1016/s1535-6108(03)00085-0. [DOI] [PubMed] [Google Scholar]
- 31.Rius J, et al. NF-kappaB links innate immunity to the hypoxic response through transcriptional regulation of HIF-1alpha. Nature. 2008;453(7196):807–811. doi: 10.1038/nature06905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Erler JT, et al. Hypoxia-induced lysyl oxidase is a critical mediator of bone marrow cell recruitment to form the premetastatic niche. Cancer Cell. 2009;15(1):35–44. doi: 10.1016/j.ccr.2008.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Solban N, et al. HCaRG, a novel calcium-regulated gene coding for a nuclear protein, is potentially involved in the regulation of cell proliferation. . J Biol Chem. 2000;275(41):32234–32243. doi: 10.1074/jbc.M001352200. [DOI] [PubMed] [Google Scholar]
- 34.van de Sluis B, et al. COMMD1 promotes pVHL and O2-independent proteolysis of HIF-1alpha via HSP90/70. . PLoS ONE. 2009;4(10):e7332. doi: 10.1371/journal.pone.0007332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Isaacs JS, Jung YJ, Mimnaugh EG, Martinez A, Cuttitta F, Neckers LM. Hsp90 regulates a von Hippel Lindau-independent hypoxia-inducible factor-1 alpha-degradative pathway. J Biol Chem. 2002;277(33):29936–29944. doi: 10.1074/jbc.M204733200. [DOI] [PubMed] [Google Scholar]
- 36.Liu YV, Baek JH, Zhang H, Diez R, Cole RN, Semenza GL. RACK1 competes with HSP90 for binding to HIF-1alpha and is required for O(2)-independent and HSP90 inhibitor-induced degradation of HIF-1alpha. Mol Cell. 2007;25(2):207–217. doi: 10.1016/j.molcel.2007.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Muller PA, et al. Nuclear-cytosolic transport of COMMD1 regulates NF-kappaB and HIF-1 activity. Traffic. 2009;10(5):514–527. doi: 10.1111/j.1600-0854.2009.00892.x. [DOI] [PubMed] [Google Scholar]
- 38.Gort EH, et al. The TWIST1 oncogene is a direct target of hypoxia-inducible factor-2alpha. Oncogene. 2008;27(11):1501–1510. doi: 10.1038/sj.onc.1210795. [DOI] [PubMed] [Google Scholar]
- 39.Huber MA, Kraut N, Beug H. Molecular requirements for epithelial-mesenchymal transition during tumor progression. Curr Opin Cell Biol. 2005;17(5):548–558. doi: 10.1016/j.ceb.2005.08.001. [DOI] [PubMed] [Google Scholar]
- 40.Cummins EP, et al. Prolyl hydroxylase-1 negatively regulates IkappaB kinase-beta, giving insight into hypoxia-induced NFkappaB activity. Proc Natl Acad Sci U S A. 2006;103(48):18154–18159. doi: 10.1073/pnas.0602235103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chan DA, Kawahara TL, Sutphin PD, Chang HY, Chi JT, Giaccia AJ. Tumor vasculature is regulated by PHD2-mediated angiogenesis and bone marrow-derived cell recruitment. Cancer Cell. 2009;15(6):527–538. doi: 10.1016/j.ccr.2009.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Burstein E, et al. A novel role for XIAP in copper homeostasis through regulation of MURR1. EMBO J. 2004;23(1):244–254. doi: 10.1038/sj.emboj.7600031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Burstein E, et al. COMMD proteins: A novel family of structural and functional homologs of MURR1. J Biol Chem. 2005;280(23):22222–22232. doi: 10.1074/jbc.M501928200. [DOI] [PubMed] [Google Scholar]
- 44.Kageyama Y, Koshiji M, To KK, Tian YM, Ratcliffe PJ, Huang LE. Leu-574 of human HIF-1alpha is a molecular determinant of prolyl hydroxylation. FASEB J. 2004;18(9):1028–1030. doi: 10.1096/fj.03-1233fje. [DOI] [PubMed] [Google Scholar]
- 45.Groot AJ, et al. Identification by phage display of single-domain antibody fragments specific for the ODD domain in hypoxia-inducible factor 1alpha. Lab Invest. 2006;86(4):345–356. doi: 10.1038/labinvest.3700395. [DOI] [PubMed] [Google Scholar]
- 46.Maine GN, Mao X, Muller PA, Komarck CM, Klomp LW, Burstein E. COMMD1 expression is controlled by critical residues that determine XIAP binding. Biochem J. 2009;417(2):601–609. doi: 10.1042/BJ20080854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Maine GN, Mao X, Komarck CM, Burstein E. COMMD1 promotes the ubiquitination of NF-kappaB subunits through a Cullin-containing ubiquitin ligase. EMBO J. 2007;26(2):436–447. doi: 10.1038/sj.emboj.7601489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Klomp AE, van de Sluis B, Klomp LW, Wijmenga C. The ubiquitously expressed MURR1 protein is absent in canine copper toxicosis. J Hepatol. 2003;39(5):703–709. doi: 10.1016/S0168-8278(03)00380-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.